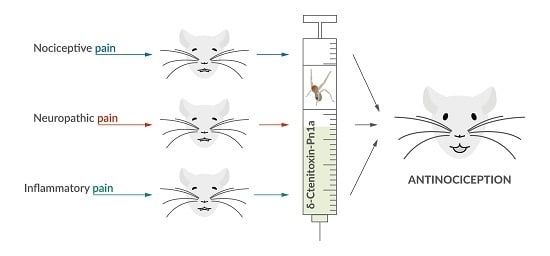
δ-Ctenitoxin-Pn1a, a Peptide from Phoneutria nigriventer Spider Venom, Shows Antinociceptive Effect Involving Opioid and Cannabinoid Systems, in Rats
Abstract
:1. Introduction
2. Results
2.1. Effect of δ-CNTX-Pn1a on Carrageenan-Induced Inflammatory Hyperalgesia
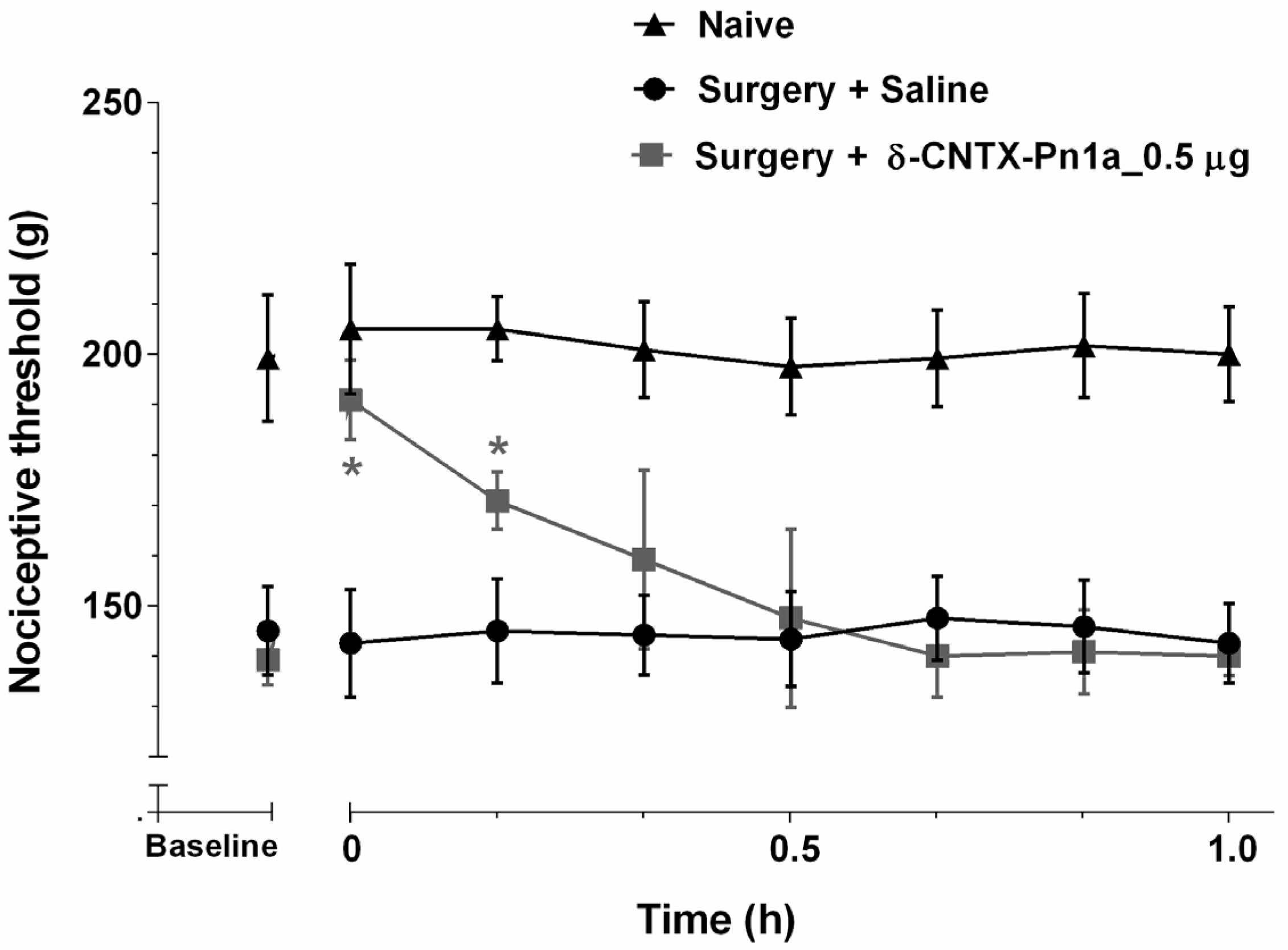
2.2. Effect of δ-CNTX-Pn1a on Neuropathic Hyperalgesia
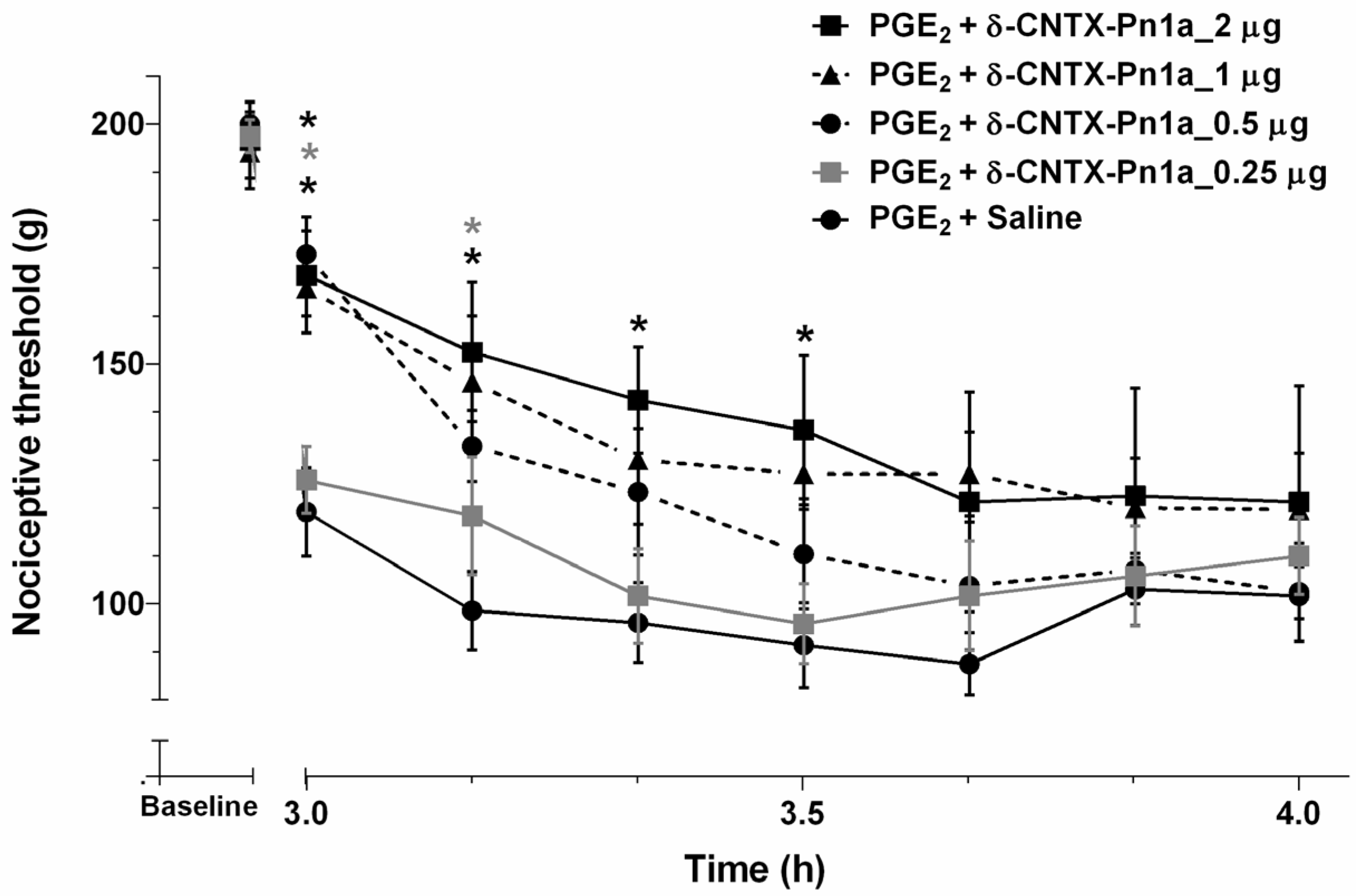
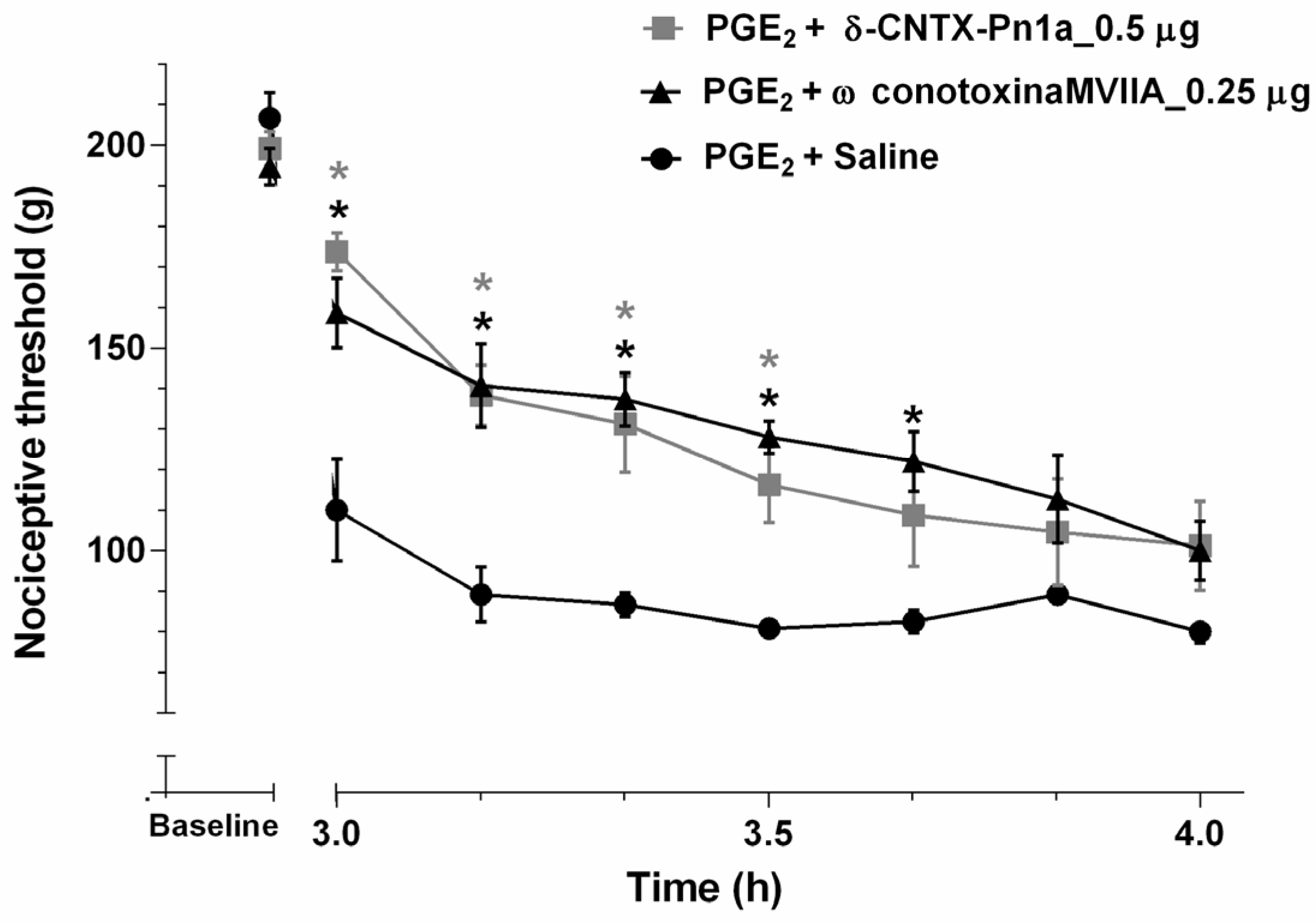
2.3. Effect of δ-CNTX-Pn1a on Prostaglandin E2-Induced Hyperalgesia and Comparison between δ-CNTX-Pn1a and ω-Conotoxina MVIIA Antinociceptive Activity
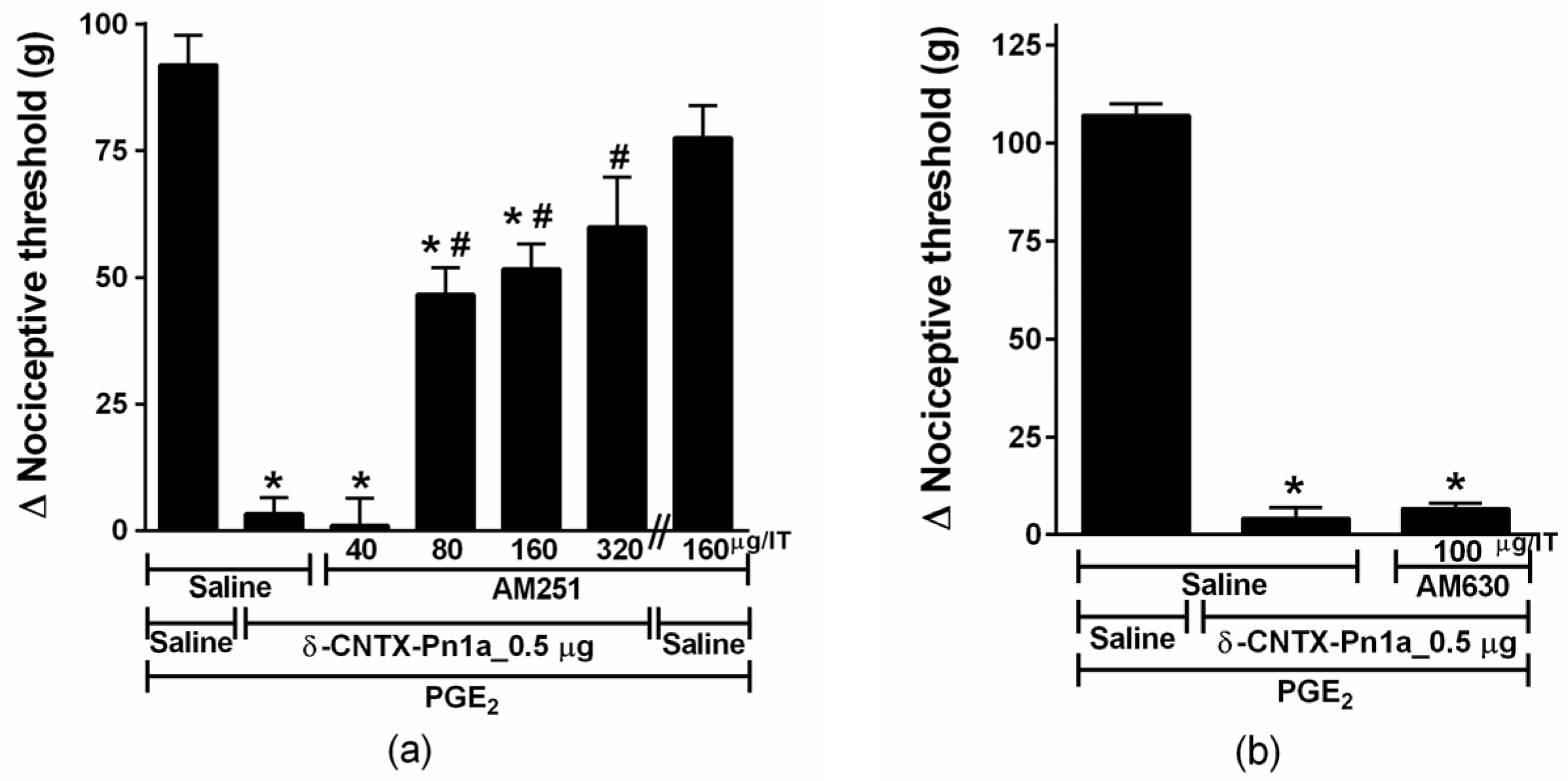
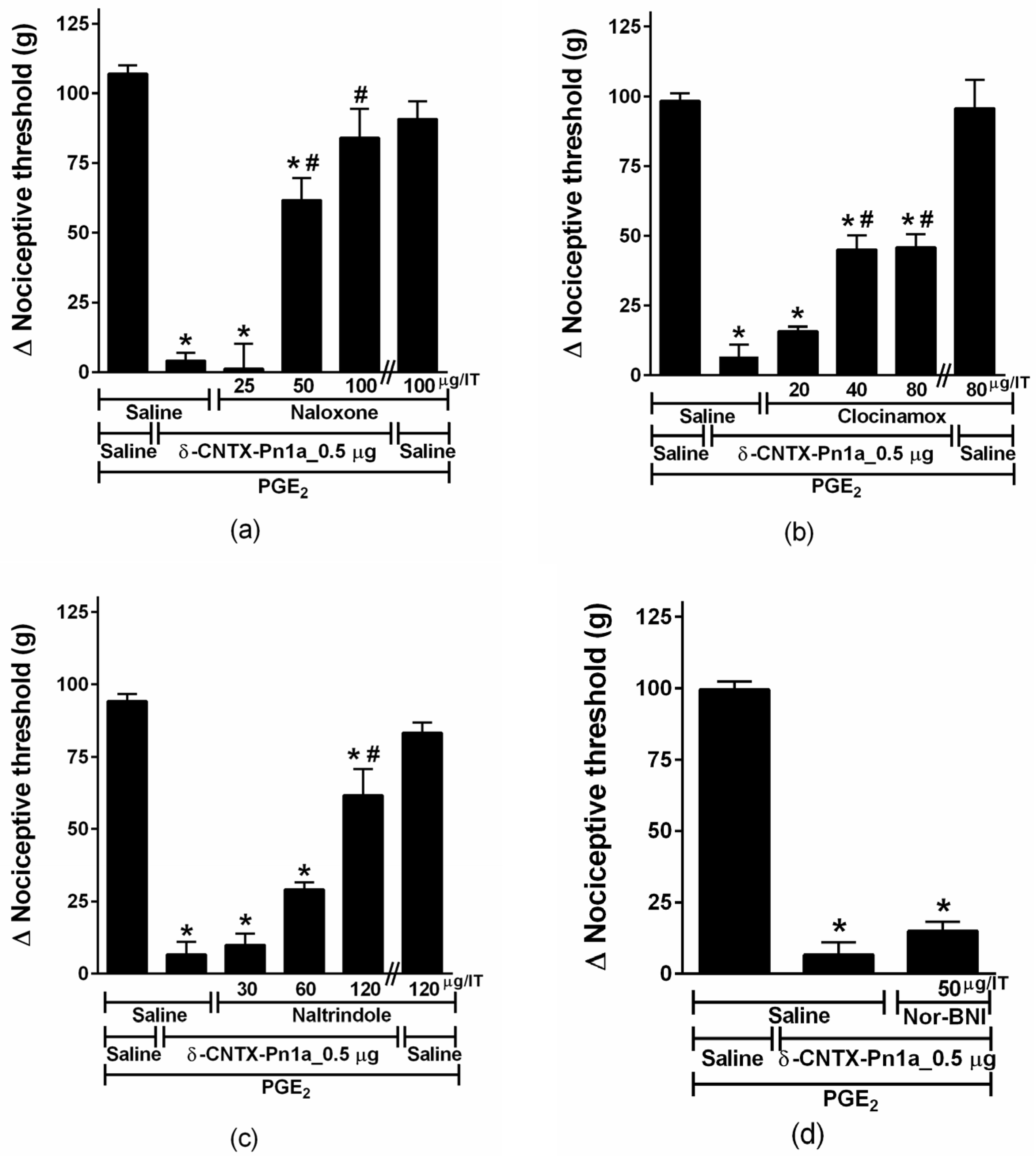
2.4. Involvement of Cannabinoid and Opioid Systems in δ-CNTX-Pn1a Antinociceptive Effect
3. Discussion
4. Conclusions
5. Material and Methods
5.1. Animals
5.2. Drugs
5.3. Drug Treatments
5.4. Carrageenan-Induced Inflammatory Hyperalgesia
5.5. Prostaglandin E2-Induced Nociceptive Hyperalgesia
5.6. Nociceptive Test
5.7. Induction of Neuropathy
5.8. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| δ-CNTX-Pn1a | δ-Ctenitoxin-Pn1a |
| NMDA | N-methyl-d-aspartate |
| Cg | Carrageenan |
| PGE2 | Prostaglandin E2 |
| AM251 | N-(piperidin-1-yl)-5-(4-iodophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide |
| AM630 | 6-iodo-2-methyl-1-[2-(4-morpholinyl)ethyl]-1H-indol-3-yl(4-ethoxyphenyl)methanone |
| Nor-BNI | Nor-Binaltorphimine dihydrochloride |
References
- Harper, D. Online Etymology Dictionary, “Penal-Etymology”. Available online: http://www.etymonline.com/index.php?term=penal&allowed_in_frame=0 (Accessed on 17 January 2016).
- Rajendra, W.; Armugam, A.; Jeyaseelan, K. Toxins in anti-nociception and anti-inflammation. Toxicon 2004, 44, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Pineda, S.S.; Undheim, E.A.; Rupasinghe, D.B.; Ikonomopoulou, M.P.; King, G.F. Spider venomics: Implications for drug discovery. Future Med. Chem. 2014, 6, 1699–1714. [Google Scholar] [CrossRef] [PubMed]
- King, G. The future of venoms-based drug discovery: An interview with Glenn King. Future Med. Chem. 2014, 6, 1613–1615. [Google Scholar] [CrossRef] [PubMed]
- Castro, Jr.; Dalmolin, G.D.; da Silva, J.F.; Gomez, M.V. Uso de Toxinas Animais para o Tratamento da Dor: Fundamentos e Aplicações. In Biotecnologia Aplicada à Saúde: Fundamentos e Aplicações; Resende, R.R., Soccol, C.R., Eds.; Publisher: Blucher, Brazil, 2015; Volume 1, pp. 575–609. (In Portuguese) [Google Scholar]
- Sylvie, D. Pain Modulating Peptides in Spider Venoms: Good and Evil. In Spider Venom; Gopalakrishnakone, P., Corzo, G.A., Diego-Garcia, E., de Lima, M.E., Eds.; Springer: Amsterdam, The Netherlands, 2015; Volume 1, pp. 121–154. [Google Scholar]
- De Lima, M.E.; Torres, F.S.; Magalhães, B.L.E.; Freitas, A.C.N. Perspectivas inovadoras para o uso terapêutico de toxinas da aranha “armadeira” Phoneutria nigriventer (Keyserling, 1891) na dor e na disfunção erétil. In Biotecnologia Aplicada à Saúde: Fundamentos e Aplicações; Resende, R.R., Soccol, C.R., Eds.; Blucher: São Paulo, Brazil, 2015; Volume 1, pp. 539–572. (In Portuguese) [Google Scholar]
- Chen, B.; Ji, Y. Antihyperalgesia effect of BmK AS, a scorpion toxin, in rat by intraplantar injection. Brain Res. 2002, 952, 322–326. [Google Scholar] [CrossRef]
- De lima, M.E.; Stankiewicz, M.; Hamon, A.; Figueiredo, S.G.; Cordeiro, M.N.; Diniz, C.R.; Martin-Eauclaire, M.-F.; Pelhate, M. The toxin Tx4(6-1) from the spider Phoneutria nigriventer slows down Na+ current inactivation in insect CNS via binding to receptor site 3. J. Insect Physiol. 2002, 48, 53–61. [Google Scholar] [CrossRef]
- Rosa, F.; Trevisan, G.; Rigo, F.K.; Tonello, R.; Andrade, E.L.; Cordeiro, M.N.; Calixto, J.B.; Gomez, M.V.; Ferreira, J. Phα1β, a peptide from the venom of the spider Phoneutria nigriventer shows antinociceptive effects after continuous infusion in a neuropathic pain model in rats. Anesth Anal. 2014, 119, 196–202. [Google Scholar] [CrossRef] [PubMed]
- De Souza, A.H.; da Costa Lopes, A.M.; Castro, C.J., Jr.; Pereira, E.M.; Klein, C.P.; da Silva, C.A., Jr.; da Silva, J.F.; Ferreira, J.; Gomez, M.V. The effects of Phα1β, a spider toxin, calcium channel blocker, in a mouse fibromyalgia model. Toxicon 2014, 81, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, J.F.; Castro-Junior, C.J.; Oliveira, S.M.; Dalmolin, G.D.; Silva, C.R.; Vieira, L.B.; Diniz, D.M.; Cordeiro, M.N.; Ferreira, J.; Souza, A.H.; et al. Characterization of the antinociceptive effect of PhTx3–4, a toxin from Phoneutria nigriventer, in models of thermal, chemical and incisional pain in mice. Toxicon 2015, 108, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Olivera, B.; Bulaj, G.; Garrett, J.; Terlau, H.; Imperial, J. Peptide Toxins from the venoms of cone snails and other toxoglossan gastropods. In Animal Toxins: State of the Art, Perspectives in Health and Biotechnology; de Lima, M.E., Pimenta, A.M.C., Martin-Eauclaire, M.F., Zingali, R.B., Hochat, H., Eds.; Editora UFMG: Belo Horizonte, Brazil, 2009; Volume 1, pp. 25–48. [Google Scholar]
- De Marco Almeida, F.; de Castro Pimenta, A.M.; Oliveira, M.C.; de Lima, M.E. Venoms, toxins and derivatives from the Brazilian fauna: Valuable sources for drug discovery. Sheng Li Xue Bao 2015, 67, 261–270. [Google Scholar] [PubMed]
- Wallace, M.S. Ziconotide: A new nonopioid intrathecal analgesic for the treatment of chronic pain. Expert. Rev. Neurother. 2006, 6, 1423–1428. [Google Scholar] [CrossRef] [PubMed]
- McGivern, J.G. Ziconotide: A review of its pharmacology and use in the treatment of pain. Neuropsychiatr. Dis. Treat. 2007, 3, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Parolaro, D.; Rubino, T.; Viganò, D.; Massi, P.; Guidali, C.; Realini, N. Cellular mechanisms underlying the interaction between cannabinoide and opioid system. Curr. Drug Targets 2010, 11, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Desroches, J.; Beaulieu, P. Opioids and cannabinoids interactions: Involvement in pain management. Curr. Drug Targets 2010, 11, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Konno, K.; Picolo, G.; Gutierrez, V.P.; Brigatte, P.; Zambelli, V.O.; Camargo, A.C.; Cury, Y. Crotalphine, a novel potent analgesic peptide from the venom of the South American rattlesnake Crotalus durissus terrificus. Peptides 2008, 29, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Machado, F.C.; Zambelli, V.O.; Fernandes, A.C.; Heimann, A.S.; Cury, Y.; Picolo, G. Peripheral interactions between cannabinoid and opioid systems contribute to the antinociceptive effect of crotalphine. Br. J. Pharmacol. 2014, 171, 961–972. [Google Scholar] [CrossRef] [PubMed]
- Martin-Eauclaire, M.F.; Abbas, N.; Sauze, N.; Mercier, L.; Berge-Lefranc, J.L.; Condo, J.; Bougis, P.E.; Guieu, R. Involvement of endogenous opioid system in scorpion toxin-induced antinociception in mice. Neurosci. Lett. 2010, 482, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Freitas, A.C.; Pacheco, D.F.; Machado, M.F.; Carmona, A.K.; Duarte, I.D.; de Lima, M.E. PnPP-19, a spider toxin analogue, induces peripheral antinociception through opioid and cannabinoid receptors and inhibition of Neutral endopeptidase. Br. J. Pharmacol. 2016, 7. [Google Scholar] [CrossRef]
- Borges, M.H.; de Lima, M.E.; Stankiewiscz, M.; Pelhate, M.; Cordeiro, M.N.; Beirão, P.S.L. Structural and functional diversity in the venom of spiders of the genus Phoneutria. In Animal Toxins: State of the Art. Perspectives on Health and Biotechnology; de Lima, M.E., Pimenta, A.M.C., Martin-Eauclaire, M.F., Zingali, R.B., Hochat, H., Eds.; Editora UFMG: Belo Horizonte, Brazil, 2009; Volume 1, pp. 291–312. [Google Scholar]
- De Lima, M.E.; Figueiredo, S.F.; Matavel, A.; Nunes, K.P.; da Silva, C.N.; de Marco Almeida, F.; Diniz, M.R.V.; do Cordeiro, M.N.; Stankiewicz, M.; Beirão, P.S.L. Phoneutria nigriventer Venom and Toxins: A Review. In Spider Venom; Gopalakrishnakone, P., Corzo, G.A., Diego-Garcia, E., de Lima, M.E., Eds.; Springer: Amsterdam, The Netherlands, 2015; Volume 1, pp. 71–100. [Google Scholar]
- Martin-Moutot, N.; Mansuelle, P.; Alcaraz, G.; dos Santos, R.G.; Cordeiro, M.N.; de Lima, M.E.; Seagar, M.; van Renterghem, C. Phoneutria nigriventer toxin 1: A novel, state-dependent inhibitor of neuronal sodium channels that interacts with micro conotoxin binding sites. Mol. Pharmacol. 2006, 69, 1931–1937. [Google Scholar] [CrossRef] [PubMed]
- Matavel, A.; Fleury, C.; Oliveira, L.C.; Molina, F.; de Lima, M.E.; Cruz, J.S.; Cordeiro, M.N.; Richardson, M.; Ramos, C.H.; Beirão, P.S. Structure and activity analysis of two spider toxins that alter sodium channel inactivation kinectics. Biochemistry 2009, 48, 3078–3088. [Google Scholar] [CrossRef] [PubMed]
- Leão, R.M.; Cruz, J.S.; Diniz, C.R.; Cordeiro, M.N.; Beirão, P.S. Inhibition of neuronal high-voltage activated calcium channels by the omega-Phoneutria nigriventer Tx3-3 peptide toxin. Neuropharmacology 2000, 39, 1756–1767. [Google Scholar] [CrossRef]
- Dos Santos, R.G.; van Renterghem, C.; Martin-Moutot, N.; Mansuelle, P.; Cordeiro, M.N.; Diniz, C.R.; Mori, Y.; de Lima, M.E.; Seagar, M. Phoneutria nigriventer omega-phonetoxin IIA blocks the Cav2 family of calcium channels and interacts with omega-conotoxin-binding sites. J. Biol. Chem. 2002, 277, 13856–13862. [Google Scholar] [CrossRef] [PubMed]
- Vieira, L.B.; Kushmerick, C.; Hildebrand, M.E.; Garcia, E.; Stea, A.; Cordeiro, M.N.; Richardson, M.; Gomez, M.V.; Snutch, T.P. Inhibition of high voltage-activated calcium channels by spider toxin PnTx3-6. J. Pharmacol. Exp. Ther. 2005, 314, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- Kushmerick, C.; Kalapothakis, E.; Beirão, P.S.; Penaforte, C.L.; Prado, V.F.; Cruz, J.S.; Diniz, C.R.; Cordeiro, M.N.; Gomez, M.V.; Romano-Silva, M.A.; et al. Phoneutria nigriventer toxin Tx3-1 blocks A-type K+ currents controlling Ca2+ oscillation frequency in GH3 cells. J. Neurochem. 1999, 72, 1472–1481. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, S.G.; Garcia, M.E.L.-P.; Valentim, A.C.; Cordeiro, M.N.; Diniz, C.R.; Richardson, M. Purification and amino acid sequence of the insecticidal neurotoxin Tx4(6-1) from the venom of the “armed” spider Phoneutria nigriventer (Keys). Toxicon 1995, 33, 83–93. [Google Scholar] [CrossRef]
- Figueiredo, S.G.; de Lima, M.E.; Cordeiro, M.N.; Diniz, C.R.; Pattend, D.; Halliwelld, R.F.; Gilroyd, J.; Richardson, M. Purification and amino acid sequence of a highly insecticidal toxin from the venom of the Brazilian spider Phoneutria nigriventer which inhibits NMDA-evoked currents in rat hippocampal neurons. Toxicon 2001, 39, 309–317. [Google Scholar] [CrossRef]
- Oliveira, L.C.; de Lima, M.E.; Pimenta, A.M.C.; Mansuelle, P.; Rochat, H.; Cordeiro, M.N.; Richardson, M.; Figueiredo, S.G. PnTx4-3, a new insect toxin from Phoneutria nigriventer venom elicits the glutamate uptake inhibition exhibited by PhTx4 toxic fraction. Toxicon 2003, 42, 793–800. [Google Scholar] [CrossRef] [PubMed]
- De Lima, M.E.; Figueiredo, S.G.; Pimenta, A.M.; Santos, D.M.; Borges, M.H.; Cordeiro, M.N.; Richardson, M.; Oliveira, L.C.; Stankiewicz, M.; Pelhate, M. Peptides of arachnid venoms with insecticidal activity targeting sodium channels. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2007, 146, 264–279. [Google Scholar] [CrossRef] [PubMed]
- Paiva, A.L.; Matavel, A.; Peigneur, S.; Cordeiro, M.N.; Tytgat, J.; Diniz, M.R.V.; de Lima, M.E. Differential effects of the recombinant toxin PnTx4(5-5) from the spider Phoneutria nigriventer on mammalian and insect sodium channels. Biochimie 2016, 121, 326–335. [Google Scholar] [CrossRef] [PubMed]
- King, G.F.; Gentz, M.C.; Escoubas, P.; Nicholson, G.M. A rational nomenclature for naming peptide toxins from spiders and other venomous animals. Toxicon 2008, 52, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Penaforte, C.L.; Prado, V.F.; Prado, M.A.; Romano-Silva, M.A.; Guimarães, P.E.; de Marco, L.; Gomez, M.V.; Kalapothakis, E. Molecular cloning of cDNAs encoding insecticidal neurotoxic peptides from the spider Phoneutria nigriventer. Toxicon 2000, 38, 1443–1449. [Google Scholar] [CrossRef]
- Oliveira, C.F.B. Estudo da ação antihiperalgésica da toxina PnTx4(5-5) do veneno da aranha armadeira Phoneutria nigriventer (Keyserling, 1981). Master’s Thesis, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brasil, 2010. [Google Scholar]
- Silva, F.R.; Batista, E.M.L.; Gomez, M.V.; Kushmerick, C.; da Silva, J.F.; Cordeiro, M.N.; Vieira, L.B.; Ribeiro, F.M. The Phoneutria nigriventer spider toxin, PnTx4-5-5, promotes neuronal survival by blocking NMDA receptors. Toxicon 2016, 112, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Bleakman, D.; Alt, A.; Nisenbaum, E.S. Glutamate receptors and pain. Semin. Cell Dev. Biol. 2006, 17, 592–604. [Google Scholar] [CrossRef] [PubMed]
- Chizh, B.A. Low dose ketamine: A therapeutic and research tool to explore N-methyl-d-aspartate (NMDA) receptor-mediated plasticity in pain pathways. J. Psychopharmacol. 2007, 21, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, D.C.; Willis, A.L. Proceedings: Prostaglandin E2, inflammation and pain threshold in rat paws. Br. J. Pharmacol. 1973, 49, 183–184. [Google Scholar]
- Picolo, G.; Giorgi, R.; Cury, Y. Delta-opioid receptors and nitric oxide mediate the analgesic effect of Crotalus durissus terrificus snake venom. Eur. J. Pharmacol. 2000, 391, 55–62. [Google Scholar] [CrossRef]
- Picolo, G.; Cury, Y. Peripheral neuronal nitric oxide synthase activity mediates the antinociceptive effect of Crotalus durissus terrificus snake venom, a delta- and kappa-opioid receptor agonist. Life Sci. 2004, 75, 559–573. [Google Scholar] [CrossRef] [PubMed]
- Diochot, S.; Baron, A.; Salinas, M.; Douguet, D.; Scarzello, S.; Dabert-Gay, A.S.; Debayle, D.; Friend, V.; Alloui, A.; Lazdunski, M.; et al. Black mamba venom peptides target acid-sensing ion channels to abolish pain. Nature 2012, 490, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Dalmolin, G.D.; Silva, C.R.; Rigo, F.K.; Gomes, G.M.; Cordeiro, M.N.; Richardson, M.; Silva, M.A.; Prado, M.A.; Gomez, M.V.; Ferreira, J. Antinociceptive effect of Brazilian armed spider venom toxin Tx3–3 in animal models of neuropathic pain. Pain 2011, 152, 2224–2232. [Google Scholar] [CrossRef] [PubMed]
- Willis, A.L. Parallel assay of prostaglandin-like activity in rat inflammatory exudate by means of cascade superfusion. J. Pharm. Pharmacol. 1969, 21, 126–128. [Google Scholar] [CrossRef] [PubMed]
- Di Rosa, M.; Giroud, J.P.; Willoughby, D.A. Studies on the mediators of the acute inflammatory response induced in rats in different sites by carrageenan and turpentine. J. Pathol. 1971, 104, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Bridges, D.; Thompson, S.W.; Rice, A.S. Mechanisms of neuropathic pain. Br. J. Anaesth. 2001, 87, 12–26. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.M.; Silva, C.R.; Trevisan, G.; Villarinho, J.G.; Cordeiro, M.N.; Richardson, M.; Borges, M.H.; Castro, C.J., Jr.; Gomez, M.V.; Ferreira, J. Antinociceptive effect of a novel armed spider peptide Tx3-5 in pathological pain models in mice. Pflugers Arch. Eur. J. Physiol. 2016, 22, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983, 16, 109–110. [Google Scholar] [CrossRef]
- Richardson, M.; Pimenta, A.M.; Bemquerer, M.P.; Santoro, M.M.; Beirão, P.S.; Lima, M.E.; Figueiredo, S.G.; Bloch, C.J.; Vasconcelos, E.A.; Campos, F.A.; et al. Comparison of the partial proteomes of the venoms of Brazilian spiders of the genus Phoneutria. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2006, 142, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Mestre, C.; Pélissier, T.; Fialip, J.; Wilcox, G.; Eschalier, A. A method to perform direct transcutaneous intrathecal injection in rats. J. Pharmacol. Toxicol. Methods 1994, 32, 197–200. [Google Scholar] [CrossRef]
- Randall, L.O.; Selitto, J.J. A method for measurement of analgesic activity on inflamed tissues. Arch. Int. Pharmacody 1957, 111, 409–419. [Google Scholar]
- Bennett, G.J.; Xie, Y.K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988, 33, 87–107. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Emerich, B.L.; Ferreira, R.C.M.; Cordeiro, M.N.; Borges, M.H.; Pimenta, A.M.C.; Figueiredo, S.G.; Duarte, I.D.G.; De Lima, M.E. δ-Ctenitoxin-Pn1a, a Peptide from Phoneutria nigriventer Spider Venom, Shows Antinociceptive Effect Involving Opioid and Cannabinoid Systems, in Rats. Toxins 2016, 8, 106. https://doi.org/10.3390/toxins8040106
Emerich BL, Ferreira RCM, Cordeiro MN, Borges MH, Pimenta AMC, Figueiredo SG, Duarte IDG, De Lima ME. δ-Ctenitoxin-Pn1a, a Peptide from Phoneutria nigriventer Spider Venom, Shows Antinociceptive Effect Involving Opioid and Cannabinoid Systems, in Rats. Toxins. 2016; 8(4):106. https://doi.org/10.3390/toxins8040106
Chicago/Turabian StyleEmerich, Bruna Luiza, Renata C. M. Ferreira, Marta N. Cordeiro, Márcia Helena Borges, Adriano M. C. Pimenta, Suely G. Figueiredo, Igor Dimitri G. Duarte, and Maria Elena De Lima. 2016. "δ-Ctenitoxin-Pn1a, a Peptide from Phoneutria nigriventer Spider Venom, Shows Antinociceptive Effect Involving Opioid and Cannabinoid Systems, in Rats" Toxins 8, no. 4: 106. https://doi.org/10.3390/toxins8040106
APA StyleEmerich, B. L., Ferreira, R. C. M., Cordeiro, M. N., Borges, M. H., Pimenta, A. M. C., Figueiredo, S. G., Duarte, I. D. G., & De Lima, M. E. (2016). δ-Ctenitoxin-Pn1a, a Peptide from Phoneutria nigriventer Spider Venom, Shows Antinociceptive Effect Involving Opioid and Cannabinoid Systems, in Rats. Toxins, 8(4), 106. https://doi.org/10.3390/toxins8040106