Aflatoxin M1 in Cow’s Milk: Method Validation for Milk Sampled in Northern Italy
Abstract
:1. Introduction
2. Results
2.1. Validation of ELISA
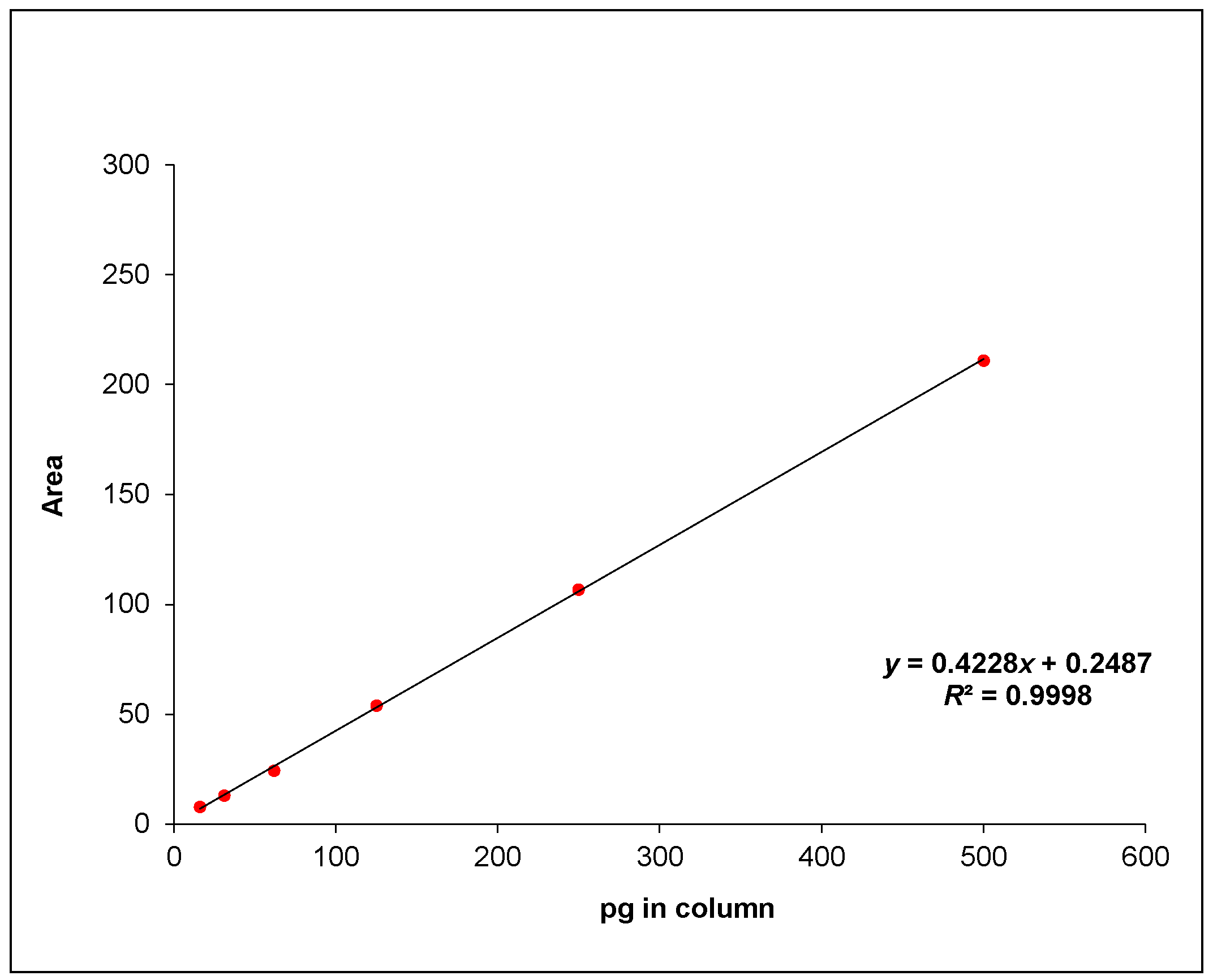
2.2. Validation of HPLC
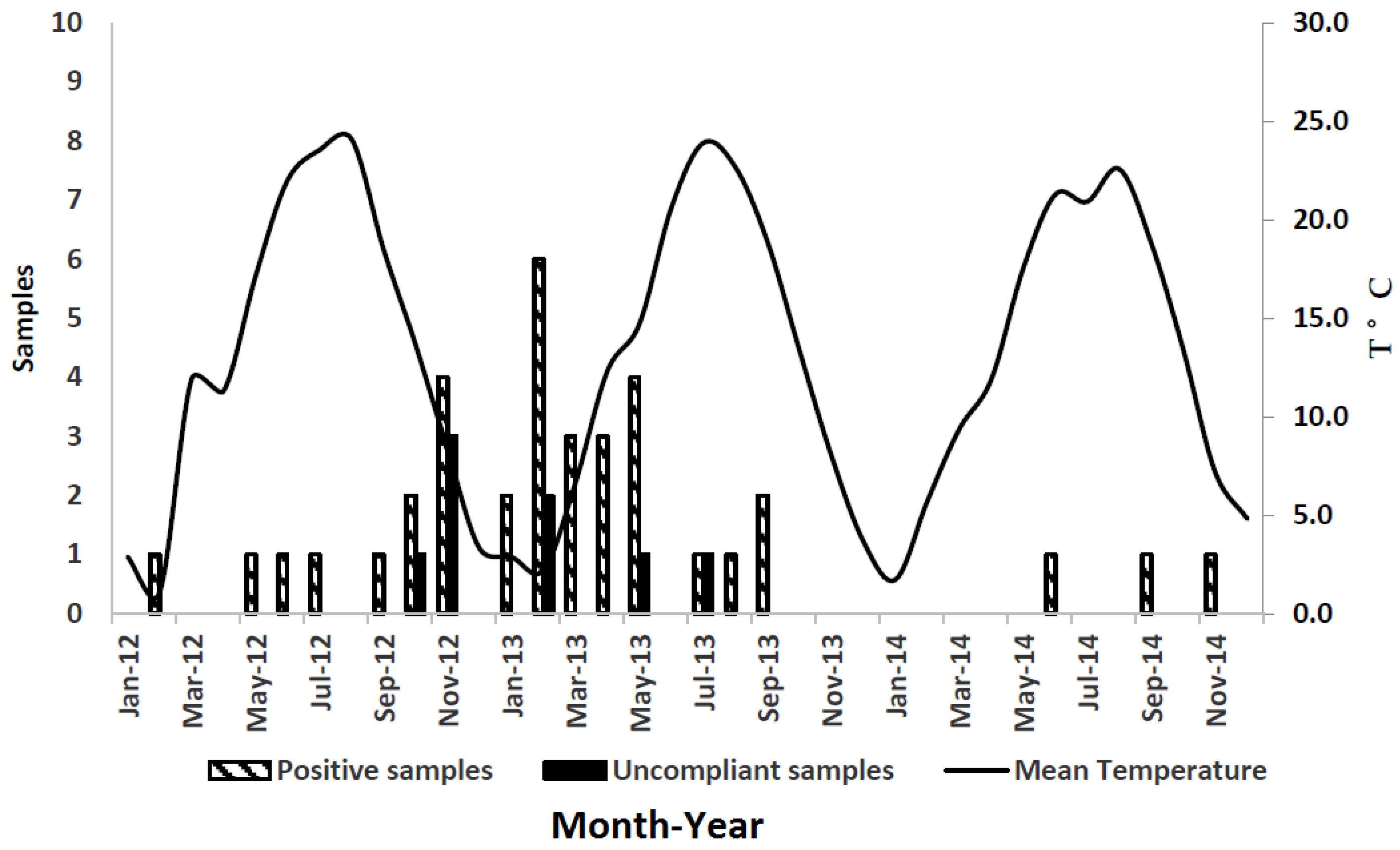
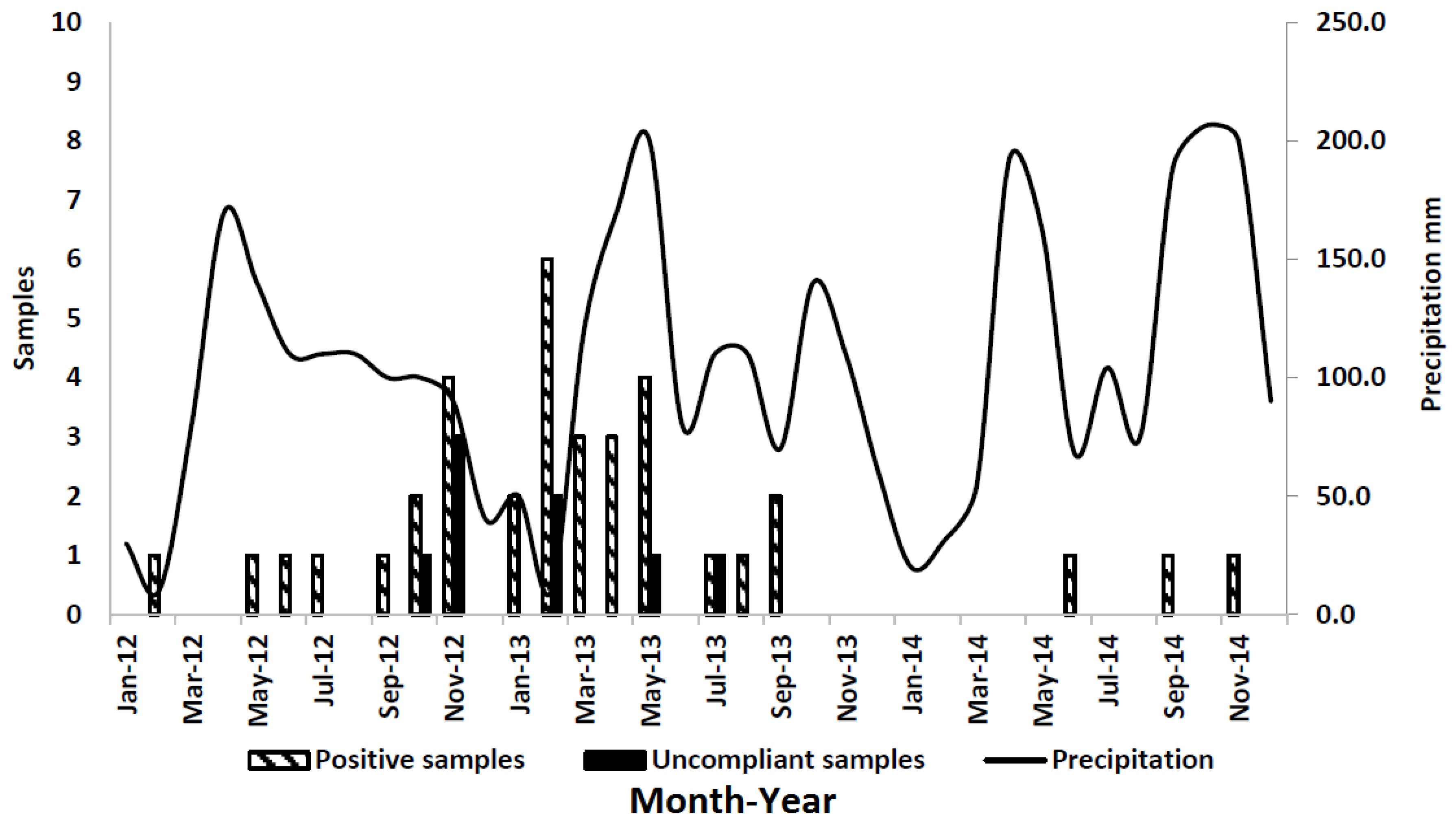
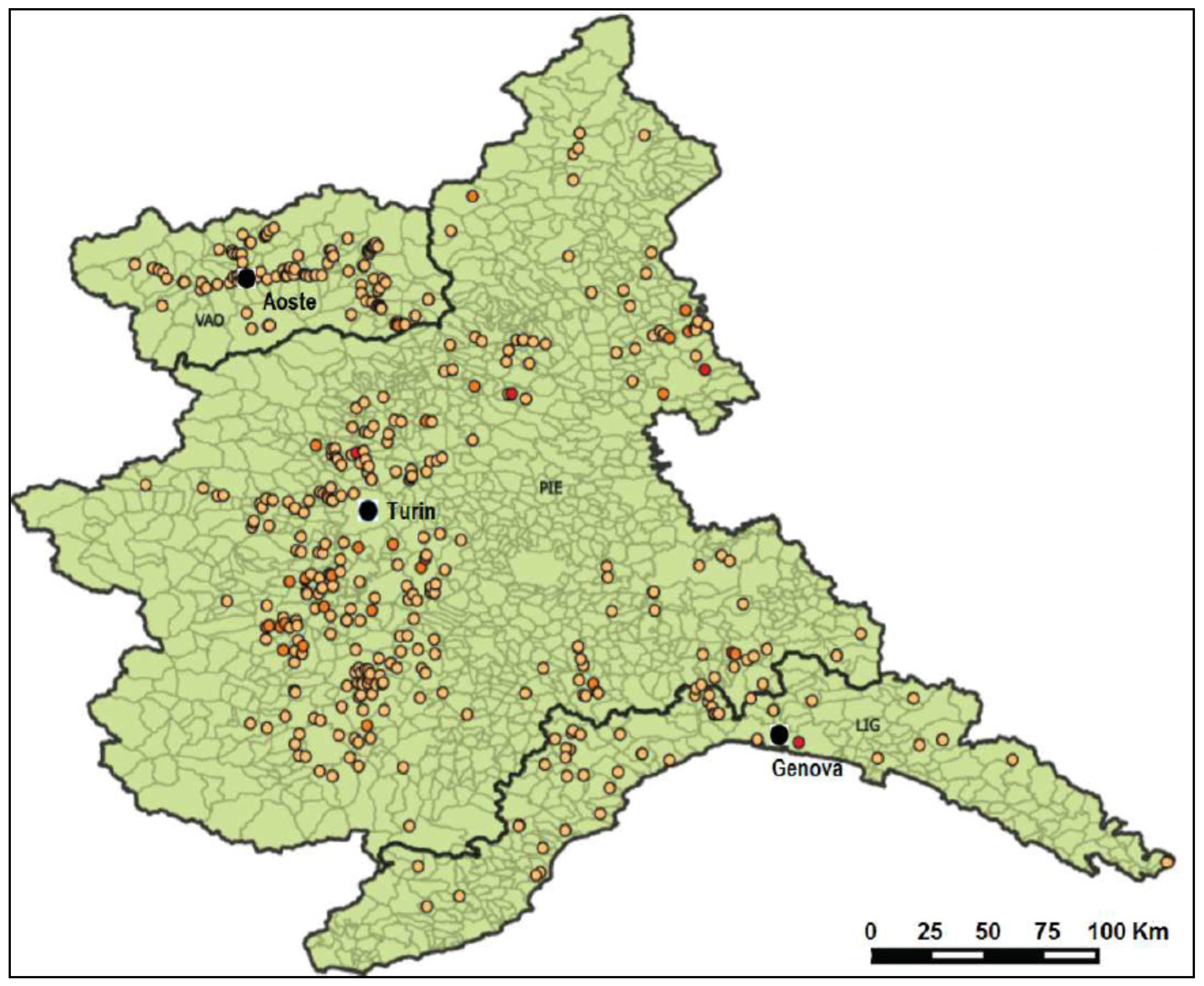
2.3. Results of Sample Analysis
3. Discussion
4. Experimental Section
4.1. Sample Collection
4.2. ELISA Screening
4.3. HPLC Confirmatory Method
4.4. Validation of ELISA
4.5. Validation of HPLC
4.6. Statistical Analyses
5. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| CI | Confidence Interval |
| EC | European Commission |
| ELISA | Enzyme-Linked ImmunoSorbent Assay |
| FLD | fluorometric detection |
| HPLC | High-Performance Liquid Chromatography |
| ppm | parts per million corresponding to mg/kg |
| ppb | parts per billion corresponding to μg/kg |
| ppt | parts per trillion corresponding to ng/kg |
| LOD | Limit of Detection |
| LOQ | Limit of Quantification |
| TLC | Thin-layer chromatography |
| UV | Ultraviolet |
References
- Bezerra da Rocha, M.E.; da Chagas Oliveira Freire, F.; Erlan Feitosa Maia, F.; Florindo Guedes, M.I.; Rondina, R. Mycotoxins and their effects on human and animal health. Food Contr. 2014, 36, 159–165. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Trucksess, M.W.; Diaz-Amigo, C. Mycotoxins in foods. Encycl. Environ. Health 2012, 888–897. [Google Scholar]
- Iqbal, S.Z.; Bhatti, I.A.; Asi, M.R.; Bhatti, H.N.; Sheikh, M.A. Aflatoxin contamination in chillies from Punjab Pakistan with reference to climate change. Int. J. Agric. Biol. 2011, 13, 261–265. [Google Scholar]
- Filazi, A.; Sireli, U.T. Occurrence of Aflatoxins in Food. In Aflatoxins—Recent Advances and Future Prospects; Mehdi, R.-A., Ed.; InTech: Rijeka, Croatia, 2013; pp. 406–408. [Google Scholar]
- Creppy, E.E. Update of survey, regulation and toxic effect of mycotoxins in Europe. Toxicol. Lett. 2002, 127, 19–28. [Google Scholar] [CrossRef]
- Dohnal, V.; Wu, Q.; Kuca, K. Metabolism of aflatoxins: Key enzymes and interindividual as well as interspecies differences. Arch. Toxicol. 2014, 88, 1635–1644. [Google Scholar] [CrossRef] [PubMed]
- Lopez, C.E.; Ramos, L.L.; Ramadàn, S.S.; Bulacio, L.C. Presence of aflatoxin M1 in milk for human consumption in Argentina. Food Control 2003, 114, 31–34. [Google Scholar] [CrossRef]
- Pitet, A. Natural occurrence of mycotoxins in foods and feeds an updated review. Rev. Med. Vet. 1998, 6, 479–492. [Google Scholar]
- Gürbay, A.; Aydin, S.; Girgin, G.; Engin, A.B.; Sahin, G. Assessment of aflatoxin M1 levels in milk in Ankara, Turkey. Food Contr. 2006, 17, 1–4. [Google Scholar] [CrossRef]
- The Commission of the European Communities. Commission Regulation (EC) No 1881/2006 of 19 December 2006. Setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L 364, 1–24. [Google Scholar]
- Turner, N.W.; Bramhmbhatt, H.; Szabo-Vezse, M.; Poma, A.; Coker, R.; Piletsky, S.A. Analytical methods for determination of mycotoxins: An update (2009–2014). Anal. Chim. Acta 2015, 901, 12–33. [Google Scholar] [CrossRef] [PubMed]
- Mwanza, M.; Abdel-Hadi, A.; Ali, A.M.; Egbuta, M. Evaluation of analytical assays efficiency to detect aflatoxin M1 in milk from selected areas in Egypt and South Africa. J. Dairy Sci. 2015, 98, 6660–6667. [Google Scholar] [CrossRef] [PubMed]
- The Commission of the European Communities. Commission Regulation (EC) No 401/2006 of 23 February 2006. Laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. Eur. Commun. 2006, L 70, 12–34. [Google Scholar]
- Bianchi, D.M.; Barbaro, A.; Gallina, S.; Vitale, N.; Chiavacci, L.; Caramelli, M.; Decastelli, L. Monitoring of foodborne pathogenic bacteria in vending machine raw milk in Piedmont, Italy. Food Control 2013, 32, 435–439. [Google Scholar] [CrossRef]
- Giacometti, F.; Bonilauri, P.; Albonetti, S.; Amatiste, S.; Arrigoni, N.; Bianchi, D.M.; Bertasi, B.; Bilei, S.; Bolzoni, G.; Cascone, G.; et al. Quantitative risk assessment of human salmonellosis and listeriosis related to the consumption of raw milk in Italy. J. Food Protect. 2015, 78, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Cavaliere, C.; Foglia, P.; Guarino, C.; Motto, M.; Nazzaria, M.; Samperi, R.; Laganà, A.; Berardo, N. Mycotoxins produced by Fusarium genus in maize: Determination by screening and confirmatory methods based on liquid chromatography tandem mass spectrometry. Food Chem. 2007, 105, 700–710. [Google Scholar] [CrossRef]
- Grasso, L.; Scarano, G.; Salzillo, A.; Serpe, L. Studio sulla presenza di aflatossina M1 nel latte e nei suoi derivati provenienti dalle regioni Campania e Calabria. La Rivista di scienza dell’alimentazione 2001, 30, 29–34. (In Italian) [Google Scholar]
- Viridis, S.; Scarano, C.; Spanu, V.; Murittu, G.; Ibba, I.; de Santis, E. A survey on Aflatoxin M1 content in sheep and goat milk produced in Sardinia Region, Italy (2005–2013). Ital. J. Food Saf. 2014, 3, 206–209. [Google Scholar] [CrossRef]
- Driehuis, F.; Spanjer, M.C.; Scholten, J.M.; te Giffel, M.C. Occurrence of mycotoxins in feedstuffs of dairy cows and estimation of total dietary intakes. J. Dairy Sci. 2008, 91, 4261–4271. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.L.; Wang, Y.M.; Mac, M.R.; Liu, J.X. Seasonal variation of aflatoxin M1 in raw milk from the Yangtze River Delta region of China. Food Control 2013, 34, 703–706. [Google Scholar] [CrossRef]
- Bilandzic, N.; Varenina, I.; Solomun, B. Aflatoxin M1 in raw milk in Croatia. Food Control 2010, 21, 1279–1281. [Google Scholar] [CrossRef]
- Battilani, P.; Barbano, C.; Piva, G. Aflatoxin B1 contamination in maize related to the aridity index in North Italy. World Mycotoxin J. 2008, 1, 449–456. [Google Scholar] [CrossRef]
- Dashti, B.; Al-Hamli, S.; Alomirah, H.; Al-Zenki, S.; Abbas, A.B.; Sawaya, W. Levels of aflatoxin M1 in milk, cheese consumed in Kuwait and occurrence of total aflatoxin in local and imported animal feed. Food Control 2009, 20, 686–690. [Google Scholar] [CrossRef]
- Bianchi, D.M.; Marello, G.; Mastrodomenico, M.; Dalla Mutta, M.; Decastelli, L.; Gallina, S.; Gili, M.; Gramaglia, M. Aflatossina nel latte bovino: Tecniche di screening e di conferma per il monitoraggio in Piemonte. LAR 2013, 19, 59–63. (In Italian) [Google Scholar]
- González Pereyra, M.L.; Alonso, V.A.; Sager, R.; Morlaco, M.B.; Magnoli, C.E.; Astoreca, A.L.; Rosa, C.A.R.; Chiacchiera, S.M.; Dalcero, A.M.; Cavaglieri, L.R. Fungi and selected mycotoxins from pre- and post-fermented corn silage. J. Appl. Microbiol. 2008, 104, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- Prandini, A.; Tansini, G.; Sigolo, S.; Filippi, L.; Laporta, M.; Piva, G. On the occurrence of aflatoxin M1 in milk and dairy products. Food Chem. Toxicol. 2009, 47, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, E.; Bonyadian, M.; Rafei, M.; Kazemeini, H.R. Occurrence of aflatoxin M1 in raw milk of 5 dairy species in Ahvaz, Iran. Food Chem. Toxicol. 2010, 48, 129–131. [Google Scholar] [CrossRef] [PubMed]
- Suriyasathaporna, W.; Watinee Nakpraserta, W. Seasonal patterns of aflatoxin M1 contamination in commercial pasteurised milk from different areas in Thailand milk from different areas in Thailand. Food Addit. Contam. 2012, 5, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Visenuo, A.; Alka, M. Occurrence, detection and detoxification of mycotoxins. BioScience 2015, 40, 943–954. [Google Scholar]
- Boudra, H.; Barnouin, J.; Dragacci, S.; Morgavi, D.P. Aflatoxin M1 and ochratoxin A in raw bulk milk from French dairy herds. J. Dairy Sci. 2007, 90, 3197–3201. [Google Scholar] [CrossRef] [PubMed]
- Pirestani, A.; Tabatabaei, S.N.; Fazeli, M.H.; Antikchi, M.; Baabaei, M. Comparison of HPLC and Elisa for Determination of Aflatoxin concentration in the milk and feeds of dairy cattle. J. Res. Agric. Sci. 2011, 7, 71–78. [Google Scholar]
- Lupo, A.; Roebuck, C.; Dutcher, M.; Kennedy, J.; Abouzied, M. Validation study of a rapid ELISA for detection of aflatoxin in corn. J. AOAC Int. 2010, 93, 587–599. [Google Scholar] [PubMed]
- Iqbal, J.; Asghar, M.A.; Ahmed, A.; Khan, M.A.; Jamil, K. Aflatoxins contamination in Pakistani brown rice: A comparison of TLC, HPLC, LC–MS/MS and ELISA techniques. Toxicol. Mech. Methods 2014, 24, 544–551. [Google Scholar] [CrossRef] [PubMed]
- AOAC Official Method 2000.08-2004. In Aflatoxin M1 in Liquid Milk. Immunoaffinity Column by Liquid Chromatography; AOAC International: Rockville, MD, USA, 2004; pp. 1–3.
- The Commission of the European Communities. Decision 2002/657/CE Commission Decision of 12 August 2002. Implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Off. J. Eur. Commun. 2002, L 221, 8–36. [Google Scholar]
- Youden, W.J. Index for rating diagnostic tests. Cancer 1950, 3, 32–35. [Google Scholar] [PubMed]
- Sergeant, E.S.G. EpiTools epidemiological calculators. Available online: http://epitools.ausvet.com.au (accessed on 28 December 2015).
- Horwitz, W.; Albert, R. The Horwitz ratio (HorRat): A useful index of method performance with respect to precision. J. AOAC Int. 2006, 89, 1095–1099. [Google Scholar] [PubMed]
- Eurachem/CITAC. Guide: Quantifying Uncertainty in Analytical Measurement, 3rd ed.; Ellison, S.L.R., Williams, A., Eds.; Eurachem/CITAC: Leoben, Austria, 2012. [Google Scholar]

| Parameters | AFM1 Spike Doses | ||
|---|---|---|---|
| 0.025 ppb | 0.050 ppb | 0.075 ppb | |
| Repeatability (Coefficient of variation CV%) | 9.7 | 7.6 | 7.0 |
| Recovery (% ±standard deviation SD) | 95 ± 7.8 | 93 ± 5.8 | 96 ± 5.8 |
| Purpose | 2012 (N = 548) | 2013 (N = 625) | 2014 (N = 495) | Total (N = 1668) | % |
|---|---|---|---|---|---|
| Raw Milk Vending Machine Survey | 381 | 332 | 255 | 968 | 58.0 |
| Study Purposes | 6 | 35 | 142 | 183 | 11.0 |
| Extraordinary AFM1 Local Survey | 31 | 116 | 33 | 180 | 10.8 |
| National Residues Plan | 57 | 85 | 37 | 179 | 10.7 |
| Regional Food Safety Survey | 52 | 14 | 14 | 80 | 4.8 |
| Tank Milk Survey (Regulation EC 853/2004) | 3 | 31 | 3 | 37 | 2.2 |
| Import-Export Survey | 7 | 9 | 5 | 21 | 1.3 |
| Other | 11 | 3 | 6 | 20 | 1.2 |
| Survey Year | Number of Samples (N = 1668) | Positive Samples (ELISA/HPLC) (N = 36) | Positive Samples (%) (% = 2.2) | Noncompliant with EU Limit (N = 8) | Noncompliant with EU Limit (%) (% = 0.5) | AFM1 Level in Noncompliant Samples (ppt) 1 |
|---|---|---|---|---|---|---|
| 2012 | 548 | 11 | 2.0 | 4 | 0.7 | 74 ± 10 |
| 83 ± 11 | ||||||
| 89 ± 12 | ||||||
| 208 ± 27 | ||||||
| 2013 | 625 | 22 | 3.5 | 4 | 0.6 | 86 ± 11 |
| 98 ± 13 | ||||||
| 137 ± 18 | ||||||
| 195 ± 25 | ||||||
| 2014 | 495 | 3 | 0.6 | 0 | 0.0 | - |
| Month | Positive by ELISA (N) | Non-Compliant (N) | Month | Positive by ELISA (N) | Non-Compliant (N) |
|---|---|---|---|---|---|
| September 2012 | 1 | 0 | September 2013 | 2 | 0 |
| October 2012 | 2 | 1 | October 2013 | 0 | 0 |
| November 2012 | 4 | 3 | November 2013 | 0 | 0 |
| December 2012 | 0 | 0 | December 2013 | 0 | 0 |
| January 2013 | 2 | 0 | January 2014 | 0 | 0 |
| February 2013 | 6 | 2 | February 2014 | 0 | 0 |
| March 2013 | 3 | 0 | March 2014 | 0 | 0 |
| April 2013 | 3 | 0 | April 2014 | 0 | 0 |
| May 2013 | 4 | 1 | May 2014 | 0 | 0 |
| June 2013 | 0 | 0 | June 2014 | 1 | 0 |
| July 2013 | 1 | 1 | July 2014 | 0 | 0 |
| August 2013 | 1 | 0 | August 2014 | 0 | 0 |
| Total (N) | 27 | 8 | Total (N) | 3 | 0 |
| Mean | 2.25 | 0.7 | Mean | 0.25 | 0 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bellio, A.; Bianchi, D.M.; Gramaglia, M.; Loria, A.; Nucera, D.; Gallina, S.; Gili, M.; Decastelli, L. Aflatoxin M1 in Cow’s Milk: Method Validation for Milk Sampled in Northern Italy. Toxins 2016, 8, 57. https://doi.org/10.3390/toxins8030057
Bellio A, Bianchi DM, Gramaglia M, Loria A, Nucera D, Gallina S, Gili M, Decastelli L. Aflatoxin M1 in Cow’s Milk: Method Validation for Milk Sampled in Northern Italy. Toxins. 2016; 8(3):57. https://doi.org/10.3390/toxins8030057
Chicago/Turabian StyleBellio, Alberto, Daniela Manila Bianchi, Monica Gramaglia, Andrea Loria, Daniele Nucera, Silvia Gallina, Marilena Gili, and Lucia Decastelli. 2016. "Aflatoxin M1 in Cow’s Milk: Method Validation for Milk Sampled in Northern Italy" Toxins 8, no. 3: 57. https://doi.org/10.3390/toxins8030057
APA StyleBellio, A., Bianchi, D. M., Gramaglia, M., Loria, A., Nucera, D., Gallina, S., Gili, M., & Decastelli, L. (2016). Aflatoxin M1 in Cow’s Milk: Method Validation for Milk Sampled in Northern Italy. Toxins, 8(3), 57. https://doi.org/10.3390/toxins8030057







