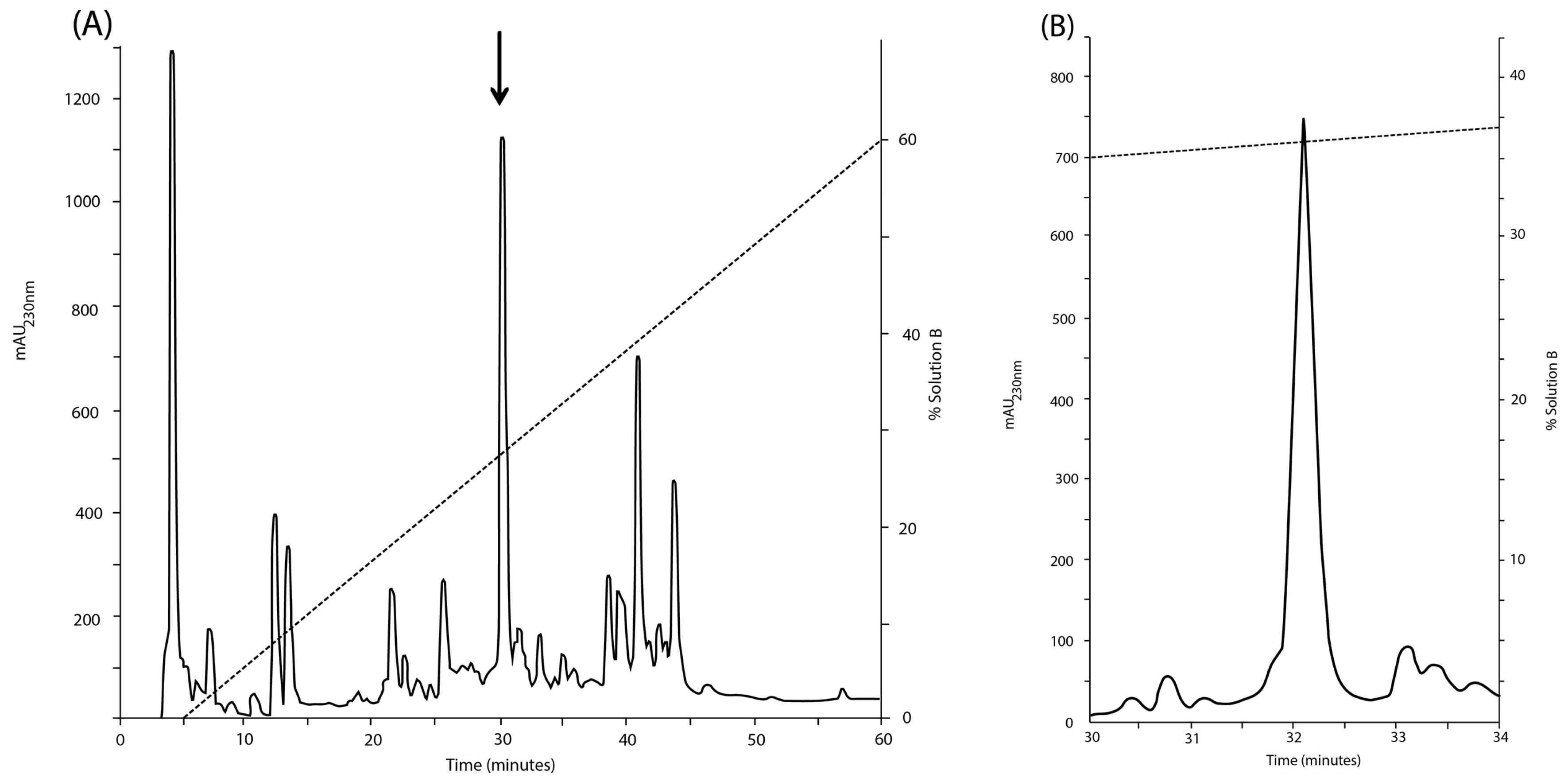
2.3. Amino Acid Sequence of the Purified Peptide
ESI-MS analysis of the reduced and S-alkylated peptide yielded a mass spectrum with two intense signals at
m/z 861.79 (4+) and 1148.65 (3+), respectively (data not shown). Both multiply-charged ions were fragmented by collision induced dissociation (CID) and the tandem mass spectrometry (MS/MS) spectra are shown in
Figure 3A,B. Manual interpretation of the MS/MS spectra rendered a 25-mer peptide,
1CDAOTHYCTNYWECCSGYCEHSHCW
25 (where O, denotes hydroxyproline).
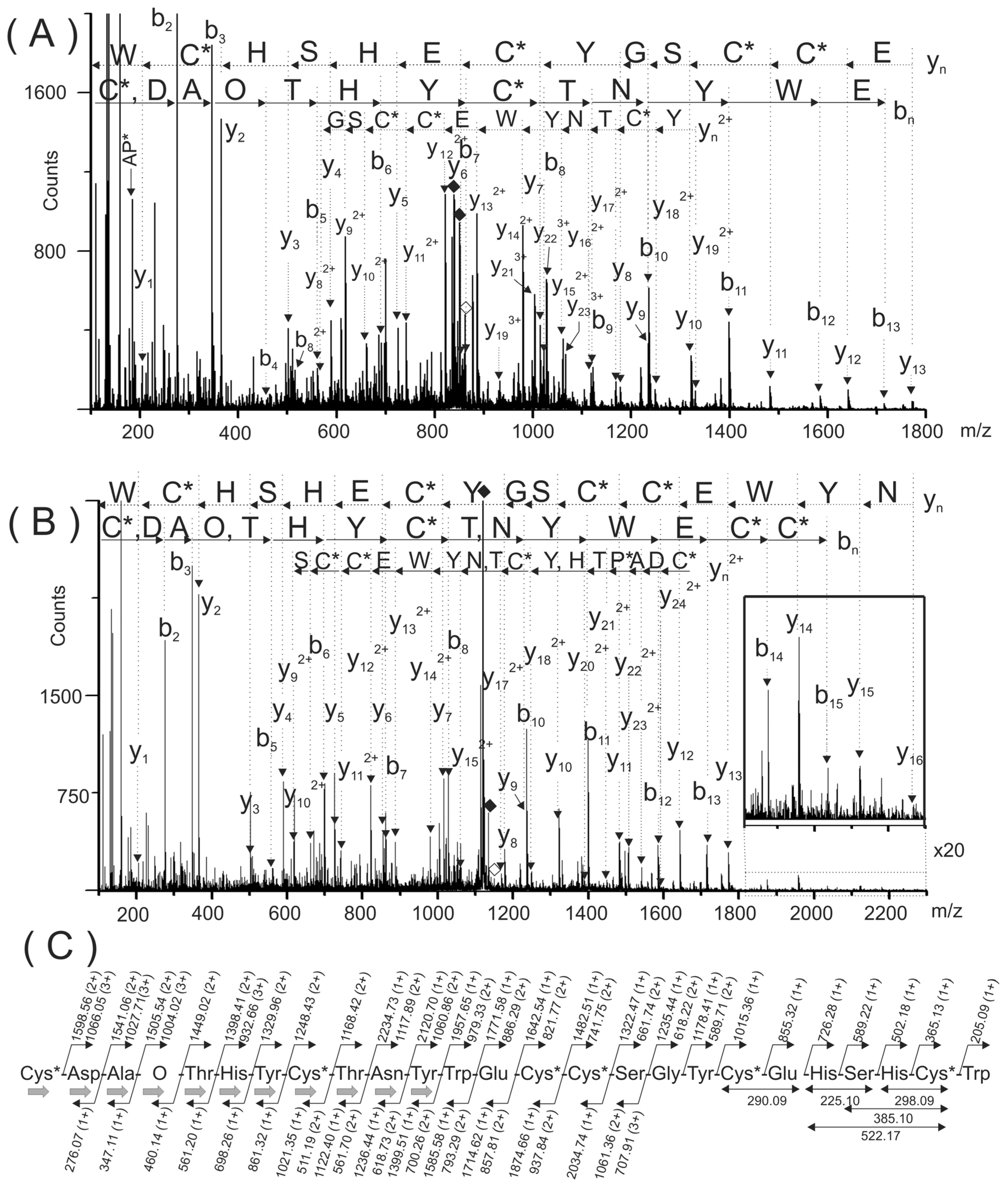
Figure 3.
Electrospray ionization tandem mass spectrometry (ESI-MS/MS) spectra of the multiply-charged species (M + 4H)4+ and (M + 3H)3+ of the reduced and carbamidomethylated PiVIIA peptide detected at m/z 861.79 (A) and 1148.65 (B), respectively. The sequences deduced form the bn and yn ions are indicated with continuous and broken arrows, respectively. (C) Summary of the sequencing of peptide PiVIIA. The gray arrows in (C) indicated the eleven amino acids verified by stepwise Edman degradation. The solid and empty rhombuses indicate the precursor ion and neutral losses of CO2 molecules, respectively. O represents a hydroxyproline residue. The six cysteine residues are carbamidomethylated and are represented as C*.
Figure 3.
Electrospray ionization tandem mass spectrometry (ESI-MS/MS) spectra of the multiply-charged species (M + 4H)4+ and (M + 3H)3+ of the reduced and carbamidomethylated PiVIIA peptide detected at m/z 861.79 (A) and 1148.65 (B), respectively. The sequences deduced form the bn and yn ions are indicated with continuous and broken arrows, respectively. (C) Summary of the sequencing of peptide PiVIIA. The gray arrows in (C) indicated the eleven amino acids verified by stepwise Edman degradation. The solid and empty rhombuses indicate the precursor ion and neutral losses of CO2 molecules, respectively. O represents a hydroxyproline residue. The six cysteine residues are carbamidomethylated and are represented as C*.
The sequences deduced form the C- (y
n+, y
n2+ and y
n3+) and N-terminal ions (b
n) were highly complementary and yielded a very trustworthy sequencing. Immonium and internal ions are in a good agreement with the proposed sequence.
Figure 3C summarizes the molecular masses of the backbone and some of the internal fragment ions used to deduce the amino acid sequence of the purified peptide. Due to the arrangement of Cys residues in the purified peptide (C-C-CC-C-C, where “-” denotes one or more non-Cys residues; framework VI/VII), it was named as PiVIIA, according to the current nomenclature for conotoxins [
9].
Taking into account the mass accuracy of this measurement (0.01–0.02 Da mass error), the 113.04 Da mass differences between backbone ions y222+ (m/z 1505.54) and y212+ (m/z 1449.02) was assigned to hydroxyproline residue (O, 113.0477 Da) and not to Leu/Ile (113.0841 Da). The mass difference (m/z 113.03) between b4 (m/z 460.14) and b3 (m/z 347.11) ions confirmed this assignment. Other internal fragment ions were also in a good agreement with the assignment of this modified amino acid.
In addition, the automated Edman sequencing of the native peptide allowed the verification of the eleven N-terminal amino acids including the presence of hydroxyproline residue at the fourth position (see the black solid arrows in
Figure 3C). During this analysis, no PTH-derivatives were detected at positions 1 and 8, and they were tentatively assumed to be Cys residues, which agrees with the sequence deduced from the results of
de novo sequencing of the reduced and carbamidomethylated peptide.
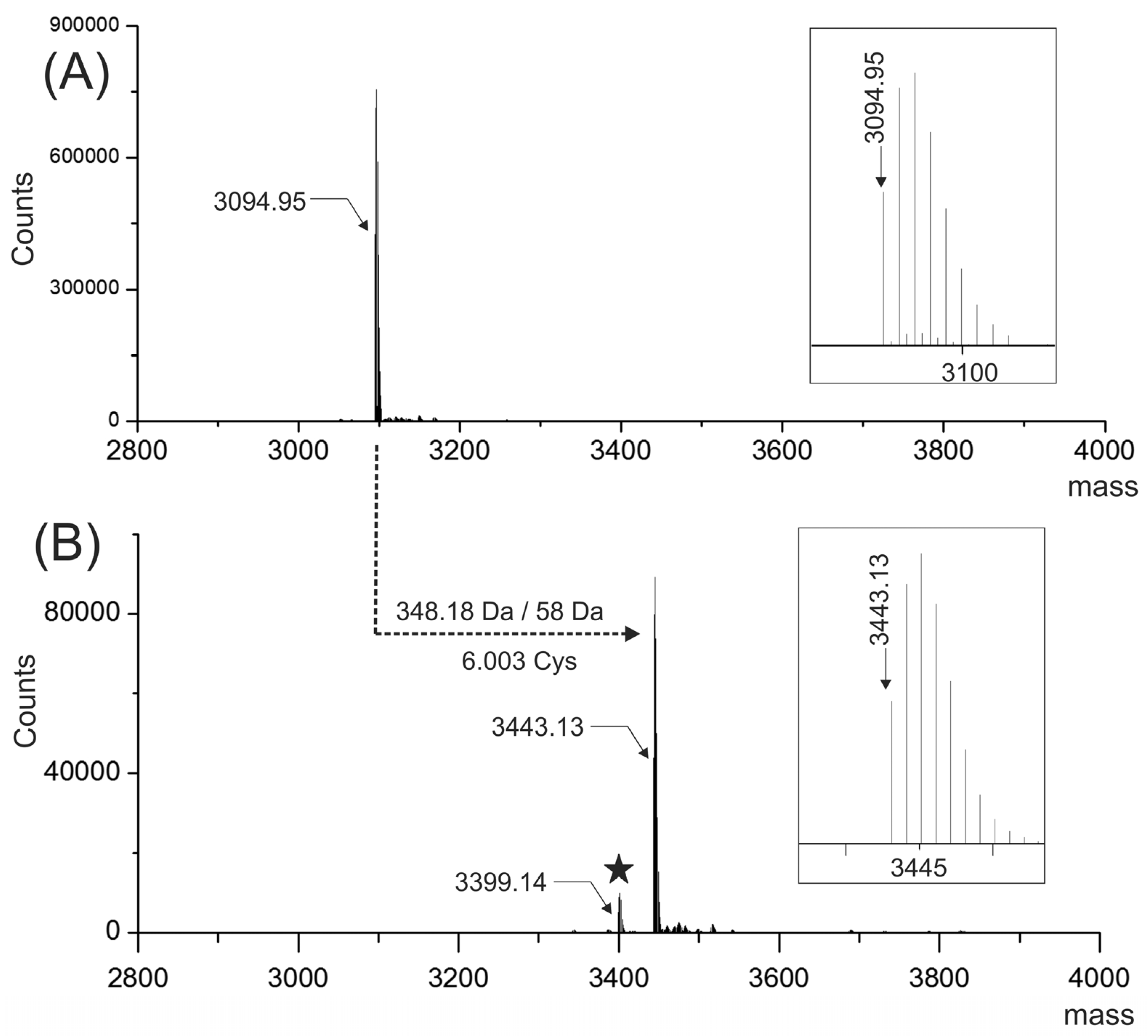
However, the molecular mass (3006.96 Da, monoisotopic mass) of the sequence deduced from the ESI-MS/MS spectra (
Figure 3A,B), considering three disulfide bonds, a hydroxyl proline residue and a free C-terminus is 87.99 Da lower than the experimental molecular mass (3094.95 Da) (
Figure 2A).
This mass difference cannot be assigned to any of the 20 amino acids commonly present in proteins. In the region close to the [M+4H]
4+ (
m/z 861.79) and [M+3H]
3+ (
m/z 1148.65) precursor ions, two signals separated by 44 Da (see solid rhombuses in
Figure 3A (
m/z 850.79 and 839.79) and
Figure 3B (
m/z 1134.05 and 1119.39) were detected. These signals were tentatively assigned to the losses of two molecules of carbon dioxide, which is in agreement with the putative decarboxylation observed in
Figure 2B (signal at 3399.14 Da). This evidence suggested that two labile posttranslational modifications (PTM) were lost upon CID conditions. As a consequence, two gamma-carboxy glutamic acids at positions 13 and 20 were assigned. In the MS/MS spectra (
Figure 3A,B), the glutamic acids were detected in an unmodified form due to the poor stability of this PTM inside the collision cell. If the presence of two gamma-carboxy glutamic acids are considered beside the hydroxyproline residue, the calculated (3443.12 Da) and experimental monoisotopic mass (3443.13 Da) of the carbamidomethylated peptide are in a good agreement with a mass error of 0.01 Da (2.9 ppm).
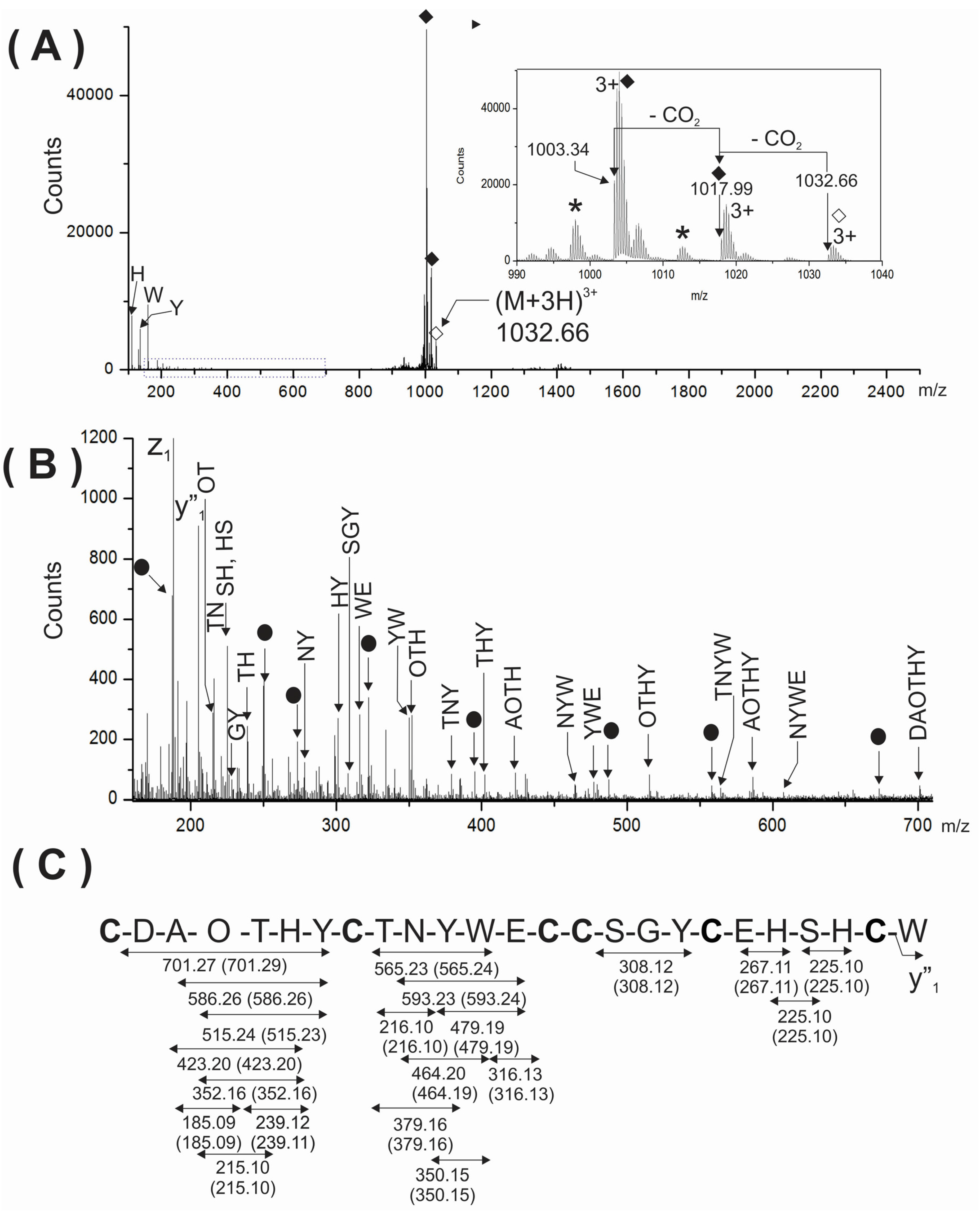
To further support the presence of two gamma-carboxy glutamic acids in the analyzed peptide, the triply-charged ion (
m/z 1032.66) of the intact peptide was analyzed by ESI-MS/MS (
Figure 4A). As expected, very few backbone fragment ions were detected due to the considerable structural constrains imposed by three disulfide linkages. This MS/MS spectrum showed the immonium ions corresponding to the aromatic amino acids (H, Y and W) present in the peptide and two very intense signals at
m/z 1003.34 and 1017.99 (see solid rhombuses, inset
Figure 4A) close to precursor ions. These signals were also assigned to two subsequent losses of two CO
2 from the precursor ions. This behavior observed in CID experiments for the intact peptide, containing disulfide linkages and two gamma-carboxy glutamic acids are in agreement with the previous reports for conotoxins sharing similar characteristics [
7,
8]. Unexpectedly, the MS/MS spectrum of the intact peptide (
Figure 4A) was also very useful and informative when the region from
m/z 150–705 was magnified (
Figure 4B). Segments of sequences comprised between cysteine residues were well represented in the MS/MS spectrum by several internal fragment ions devoid of cysteine. Their expected and experimental molecular masses are summarized in
Figure 4C.
When this MS/MS spectrum (
Figure 4B) was compared with the corresponding for the reduced and carbamidomethylated peptide (
Figure 2A,B), the assignment of, backbone (b
n and y
n) and cysteine-containing internal fragment ions, was a more reliable and easier task. This comparison was a key element for a reliable
de novo sequencing, of a region in the MS/MS spectrum difficult to interpret due to the coexistence of several fragment ions of different origins: N-, C-terminal ions, internal fragment ions as well and their associated ions originated by neutral losses (-NH
3, -H
2O, -CO
2). Furthermore, several internal fragment ions comprised between
1Cys and
8Cys, were detected, and most of them contain the hydroxyproline residue at the fourth position. The high resolution and mass accuracy achieved in the MS/MS spectrum also supported the presence of hydroxyproline (113.047 Da) and avoided its misassignment as Leu/Ile (113.084 Da). See the good agreement obtained between the expected and experimental mass for internal fragment ions containing hydroxyproline (
Figure 4C).
Figure 4.
(A) MS/MS spectra of the (M + 3H)3+ ion at m/z 1033.32 of the native PiVIIA peptide. The inset at the right represents the region close to the precursor ion. Two consecutive losses of 44 Da were assigned to CO2 molecules from the precursor due to the presence of two γ-carboxyglutamic acids residues and are indicated with solid rhombuses. The asterisks represent losses of water. The precursor ion is indicated with an empty rhombus; (B) The expanded region (m/z 150–705) of the MS/MS spectrum shown in (A) is enriched in cysteine-free internal fragment ions. The filled circles correspond to YnAm ions. The mass errors between the expected and experimental masses for all fragments ions were between 0.01 and 0.02 Da; (C) Summary of the internal fragment ions detected in (B). Calculated masses of internal ions are indicated between parentheses. O represents a hydroxyproline residue.
Figure 4.
(A) MS/MS spectra of the (M + 3H)3+ ion at m/z 1033.32 of the native PiVIIA peptide. The inset at the right represents the region close to the precursor ion. Two consecutive losses of 44 Da were assigned to CO2 molecules from the precursor due to the presence of two γ-carboxyglutamic acids residues and are indicated with solid rhombuses. The asterisks represent losses of water. The precursor ion is indicated with an empty rhombus; (B) The expanded region (m/z 150–705) of the MS/MS spectrum shown in (A) is enriched in cysteine-free internal fragment ions. The filled circles correspond to YnAm ions. The mass errors between the expected and experimental masses for all fragments ions were between 0.01 and 0.02 Da; (C) Summary of the internal fragment ions detected in (B). Calculated masses of internal ions are indicated between parentheses. O represents a hydroxyproline residue.
From the methodological point of view, the fragmentation of the intact peptide was very useful for the assignment of two gamma carboxyglutamic acids as well as for a more reliable interpretation of the MS/MS spectra of the reduced and S-alkylated peptide. To what extent the fragmentation of an intact conotoxin with several disulfide bonds could be a general approach that may contribute to a more reliable sequencing by mass spectrometry of the reduced and S-alkylated derivative will depend, of course, on the arrangement of cysteine residues.
At least in the present case, where the sequencing has been based mainly on mass spectrometry data, this approach permitted an unambiguous assignment of the internal fragment ions, and a more reliable sequencing because it avoids their wrong assignment as either N- or C-terminal ions.
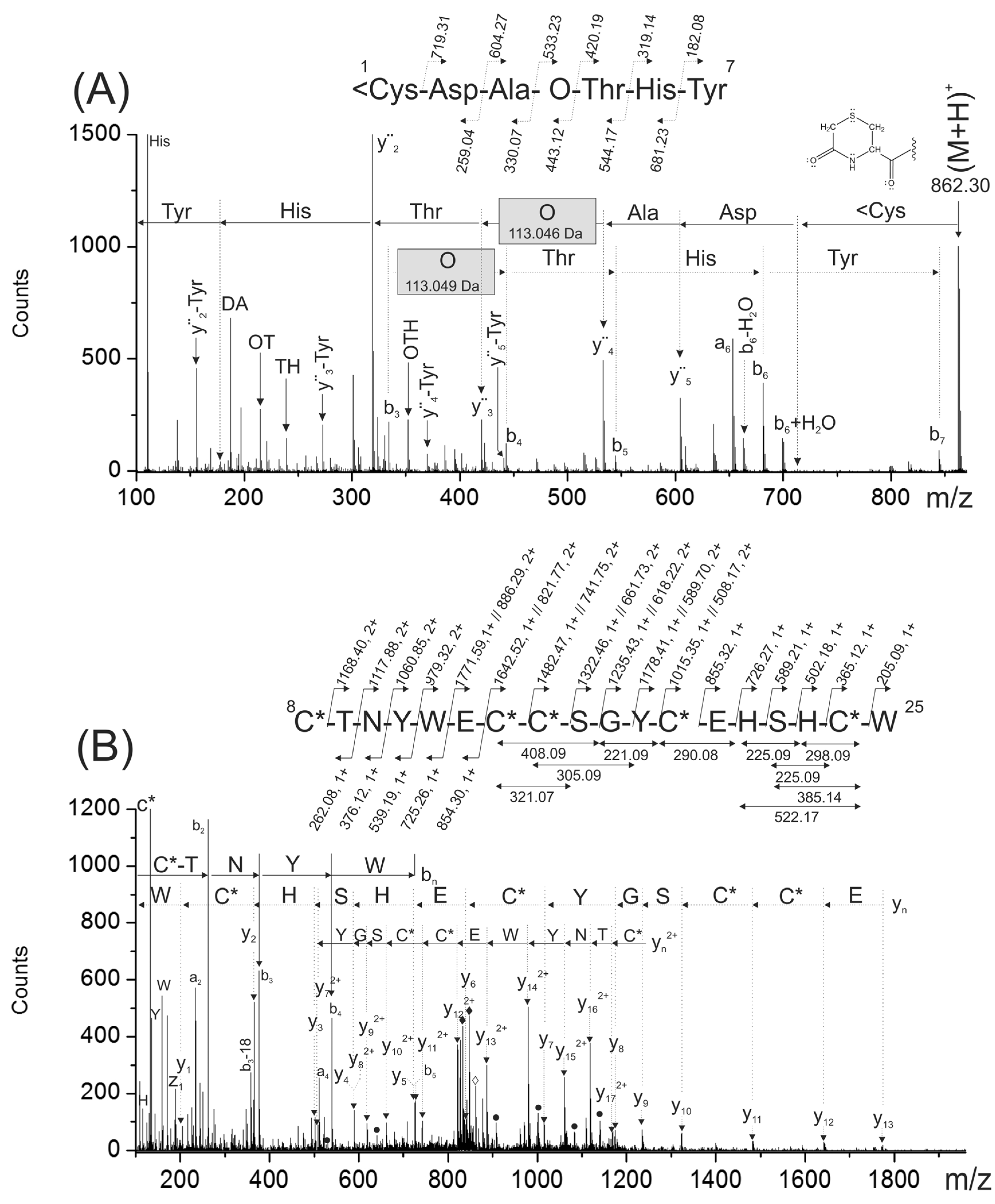
To confirm the peptide sequencing depicted in
Figure 2A,B, the reduced and S-alkylated peptide was digested with chymotrypsin. This digestion was useful because it separated into two chymotryptic peptides, the modified amino acids present in the original peptide (hydroxyproline and gamma-carboxy glutamic acids). A singly- and a triply-charged ions detected at
m/z 862.30 (
1<CDAOTHY
7) and
m/z 861.95 (
8C*TNYWγC*C*SGYC*γHSHC*W
25), respectively, were generated during this proteolytic digestion.
The N-terminal chymotryptic peptide (
1<CDAOTHY
7) has a molecular mass 17 Da lower than expected. At the N-terminal end, it has a cyclic Cys residue forming a six membered-ring (<C), probably originated during proteolytic digestion due to the combined effect of a basic pH and temperature [
10]. The analysis of the MS/MS spectra corresponding for this peptide (
Figure 5A) confirmed the N-terminal sequence (
Figure 2A,B). In this MS/MS spectrum, a C-terminal rearrangement ion (b
6 + H
2O) typical of peptides containing internal basic amino acids [
11] was observed with appreciable intensity. Probably, the presence of a basic amino acid (His) located at
n − 1 position make this rearrangement more favorable [
12]. In addition, when this rearrangement ion (b
6 + H
2O) was further fragmented upon CID, several fragment ions devoid of the C-terminal Tyr were observed. These ions were assigned as y
n-Tyr, (
Figure 5A) and they were also useful to confirm the peptide sequencing.
The mass differences between consecutive backbone ions y
4–y
3 (113.049 Da) and b
4–b
3 (113.046 Da) confirmed the presence of a hydroxyproline residue (see gray rectangles in
Figure 5A). The good agreement between the experimental masses of internal ions and backbone ions that contain hydroxyproline within their structures also confirmed the presence of this modified amino acid (data not shown). In this MS/MS spectra, signals previously assigned to CO
2 losses from the precursor, were not observed since this fragment of PiVIIA (1–7) does not contain any gamma-carboxy glutamic acid.
On the other hand, the analysis of the MS/MS spectrum of the triply-charged chymotryptic peptide at
m/z 861.95 (
Figure 5B) confirmed the C-terminal sequence of the peptide. This MS/MS showed signals assigned to two CO
2 losses from the precursor confirming by this way, the presence of two gamma-carboxy glutamic acids. In addition, y
n2+ ion series with masses increased by 44 Da were observed (see filled circles in
Figure 5B). Internal fragment ions were useful to confirm the C-terminal part of the sequence that was not verified simultaneously by the b
n and y
n ions.
The experimental molecular mass (m/z 861.95) and the calculated mass for the triple-charged ion of this peptide (m/z 861.95), considering the presence of two gamma-carboxy glutamic acids, agrees very well.
Although the Glutamic acids were detected in all ESI-MS/MS spectra as a free Glu residue (129.04 Da), the presence of two neutral losses of 44 Da from the precursor confirm that they are modified as γ residues.
Figure 5.
The ESI-MS/MS spectra shown in (A) and (B) correspond to singly- and triply-charged peptides 1<Cys-Tyr7 (m/z 862.30) and 8Cys-Trp25 (m/z 861.95) obtained from the chymotryptic digestion of the reduced and carbamidomethylated PiVIIA peptide. The gray rectangles indicate the mass difference assigned to the hydroxyproline residue. <C represents a cyclized cysteine residue located at the N-terminal end forming a cyclic of six-membered ring. In (B), the black rhombuses indicate triply-charged signals detected at m/z 847.28 and 832.62 that were assigned to two subsequent losses of CO2 molecules from the precursor. The filled circles correspond to yn2+ ions with masses increased by 44 Da due to the presence of a γ residue within their sequence.
Figure 5.
The ESI-MS/MS spectra shown in (A) and (B) correspond to singly- and triply-charged peptides 1<Cys-Tyr7 (m/z 862.30) and 8Cys-Trp25 (m/z 861.95) obtained from the chymotryptic digestion of the reduced and carbamidomethylated PiVIIA peptide. The gray rectangles indicate the mass difference assigned to the hydroxyproline residue. <C represents a cyclized cysteine residue located at the N-terminal end forming a cyclic of six-membered ring. In (B), the black rhombuses indicate triply-charged signals detected at m/z 847.28 and 832.62 that were assigned to two subsequent losses of CO2 molecules from the precursor. The filled circles correspond to yn2+ ions with masses increased by 44 Da due to the presence of a γ residue within their sequence.
2.4. Electrophysiological Characterization
A total of 30 DRG neurons (mean capacity = 44 ± 22 pF, Standard Deviation) were successfully voltage-clamped for a sufficient time to allow the study of PiVIIA actions. The mean capacitance of these neurons corresponds to a cell diameter of about 36 ± 9 μm. All voltage clamp experiments were performed using the whole-cell mode. In order to minimize the effects of time-dependent shifts in our results, recordings were not initiated until about 5–10 min after the whole-cell configuration was achieved, that is, when the signal had stabilized.
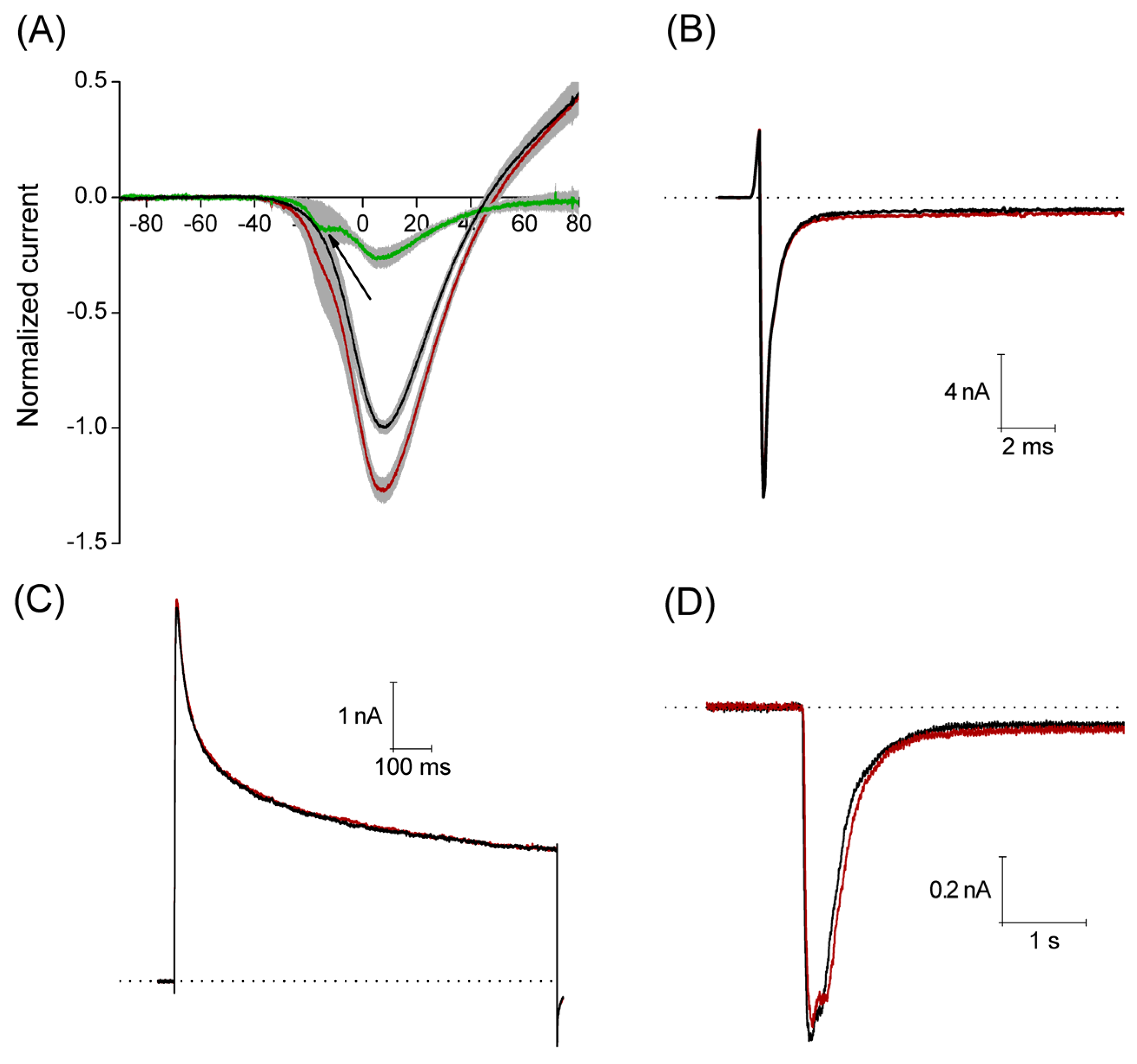
The Ca2+ currents were recorded applying a voltage ramp from −100 mV to +100 mV, with a duration of 500 ms, interval between sweeps was 10 s. Under control conditions, this stimulation protocol produced a current that activated around −35 mV reaching its maximum amplitude (1.7 ± 0.2 nA) around 8 mV, having a reversal potential around 45 mV. An estimated concentration of free Ca2+ is about 1.5 × 10−9 M in the pipette solution for the recording of Ca2+ current (0.1 mM Ca2+ and 10 mM EGTA). A calculated equilibrium potential for Ca2+ in this condition is about +180 mV, then the outward currents observed at voltages higher than +45 mV are most probably carried by Cs+ flow through voltage-gated Ca2+ channels. With perfusion of 3 μM PiVIIA in eight of 11 neurons, the peak amplitude increased by 29% ± 5% (p < 0.05), compared to the control. In the additional three neurons, a non-significant (p > 0.05) decrease in the peak amplitude of 6.8% ± 1.7% with respect to the control (data not shown) was observed.
The maximum effect was reached within the first 2.5 min of the toxin perfusion, and it did not reverse after 2–5 min washout (
Figure 6A). In the additional three neurons, a non-significant (
p > 0.05) decrease in the peak amplitude of 6.8% ± 1.7% with respect to the control (data not shown) was observed. It is worth noting that the use of PiVIIA toxin reveals a low voltage activated component of the Ca
2+ current that was not evident in the control condition (see arrow in
Figure 6A), and that most probably corresponds to the T-type Ca
2+ current (Ca
v 3.1–3.3).
With the aim of studying the selectivity of the toxin, in a set of experiments, we analyzed the actions of PiVIIA on sodium, potassium and acid sensing ionic channel (ASIC) currents. Sodium currents were evoked by a 40 ms step depolarization to a membrane potential of −10 mV from a holding potential of −100 mV, with an interval between sweeps of 8 s. The actions of PiVIIA were evaluated on:
- (a)
the maximum amplitude of the current (INamax),
- (b)
the time constant of the current inactivation (τh), as derived from an exponential fit, and
- (c)
the relation between the current amplitude at the end of the voltage pulse and the peak current amplitude (INaend/INamax) which gives an estimate of the probability for the channels not to be inactivated at the end of the voltage pulse.
Perfusion of 10 μM PiVIIA (
n = 5) for 2 min had no significant effect (
p > 0.05) on any of the parameters studied: INa
max = 6.4 ± 2.8 nA
versus 6.5 ± 2.7 nA; τ
h = 5.6 ± 1.9 ms
versus 5.5 ± 1.8 ms; INa
end/INa
max = 0.12 ± 0.09
versus 0.13 ± 0.1, under control conditions and in the presence of PiVIIA, respectively (
Figure 6B).
Potassium currents were elicited by a single-step voltage protocol, where 1 s depolarizing pulses to 30 mV were applied from a holding potential of −90 mV every 8 s. The parameters measured were:
- (a)
the maximum amplitude IKmax,
- (b)
the decay time constant τ, and
- (c)
the steady-state level (IKss) of the current.
None of these parameters were significantly affected by the perfusion of 10 μM PiVIIA for about 2 min (
p > 0.05,
n = 5): IK
max = 4.1 ± 1.2 nA
versus 4.0 ± 1.1 nA; τ = 308.3 ± 50.3 ms
versus 329.4 ± 63.0 ms; IK
ss = 2.2 ± 0.7 nA
versus 2.2 ± 0.7 nA, under control conditions and in the presence of PiVIIA, respectively (
Figure 6C).
The ASIC currents were elicited by a fast (about 40 ms) pH change from 7.4 to 6.1 for 5 s, while keeping the cell at a holding potential of −60 mV. The interval between the pH change steps was 1 min to guarantee that the ASIC current was completely recovered from desensitization.
The parameters measured to characterize the action of PiVIIA on the ASIC currents were:
- (a)
Maximum peak amplitude (Imax).
- (b)
Current desensitization time constant (τdes), determined by fitting the decay phase of the current with a single exponential function).
- (c)
The amplitude at the end of the 5 s acid pulse (Iss), which is a measure of the steady state current and it was calculated as the mean of the current in the last 200 ms of the acid pulse).
The compound was applied 20 s before the pH change to 6.1 and during the whole 5 s pH pulse. 3 μM PiVIIA did not produce any significant effect on the parameters studied (
p > 0.05,
n = 7): I
max = 1.1 ± 0.2 nA
versus 1.0 ± 0.2 nA; τ
des = 343 ± 76 ms
versus 358 ± 84 ms; I
ss = 0.04 ± 0.02 nA
versus 0.05 ± 0.02 nA, under control conditions and in the presence of PiVIIA, respectively (
Figure 6D).
Figure 6.
Peptide PiVIIA increases the amplitude of the calcium current without significantly modifying the Na+, K+ and ASIC currents. (A) Current-voltage relationship generated from a ramp pulse protocol. Solid lines represent the mean of eight experiments under control conditions (black line) and in the presence of 3 μM PiVIIA (red line). Shadows in light gray represent the standard error of the mean. Green trace depicts the PiVIIA-sensitive component of the Ca2+ current obtained by subtraction of the Ca2+ current in the presence of the toxin from that in the absence of the compound. Perfusion with PiVIIA revealed a low voltage activated component that probably corresponds to the T-type Ca2+ current (arrow); (B–D) Representative traces of sodium (B), potassium (C) and ASIC (D) currents in the absence (black lines) and presence (red lines) of PiVIIA. The dotted lines represent the zero current.
Figure 6.
Peptide PiVIIA increases the amplitude of the calcium current without significantly modifying the Na+, K+ and ASIC currents. (A) Current-voltage relationship generated from a ramp pulse protocol. Solid lines represent the mean of eight experiments under control conditions (black line) and in the presence of 3 μM PiVIIA (red line). Shadows in light gray represent the standard error of the mean. Green trace depicts the PiVIIA-sensitive component of the Ca2+ current obtained by subtraction of the Ca2+ current in the presence of the toxin from that in the absence of the compound. Perfusion with PiVIIA revealed a low voltage activated component that probably corresponds to the T-type Ca2+ current (arrow); (B–D) Representative traces of sodium (B), potassium (C) and ASIC (D) currents in the absence (black lines) and presence (red lines) of PiVIIA. The dotted lines represent the zero current.