Snake Genome Sequencing: Results and Future Prospects
Abstract
:1. Introduction
1.1. Why Snakes Are Interesting
1.2. What Genomes Can Tell Us
1.3. Aims and Objectives of This Review
2. Status of Snake Genome Sequencing Projects
3. Genome Data in the Reconstruction of Toxin Evolution
3.1. Overview of Possible Mechanisms of Toxin Evolution
3.2. Moonlighting: The Strange Case of Nerve Growth Factor
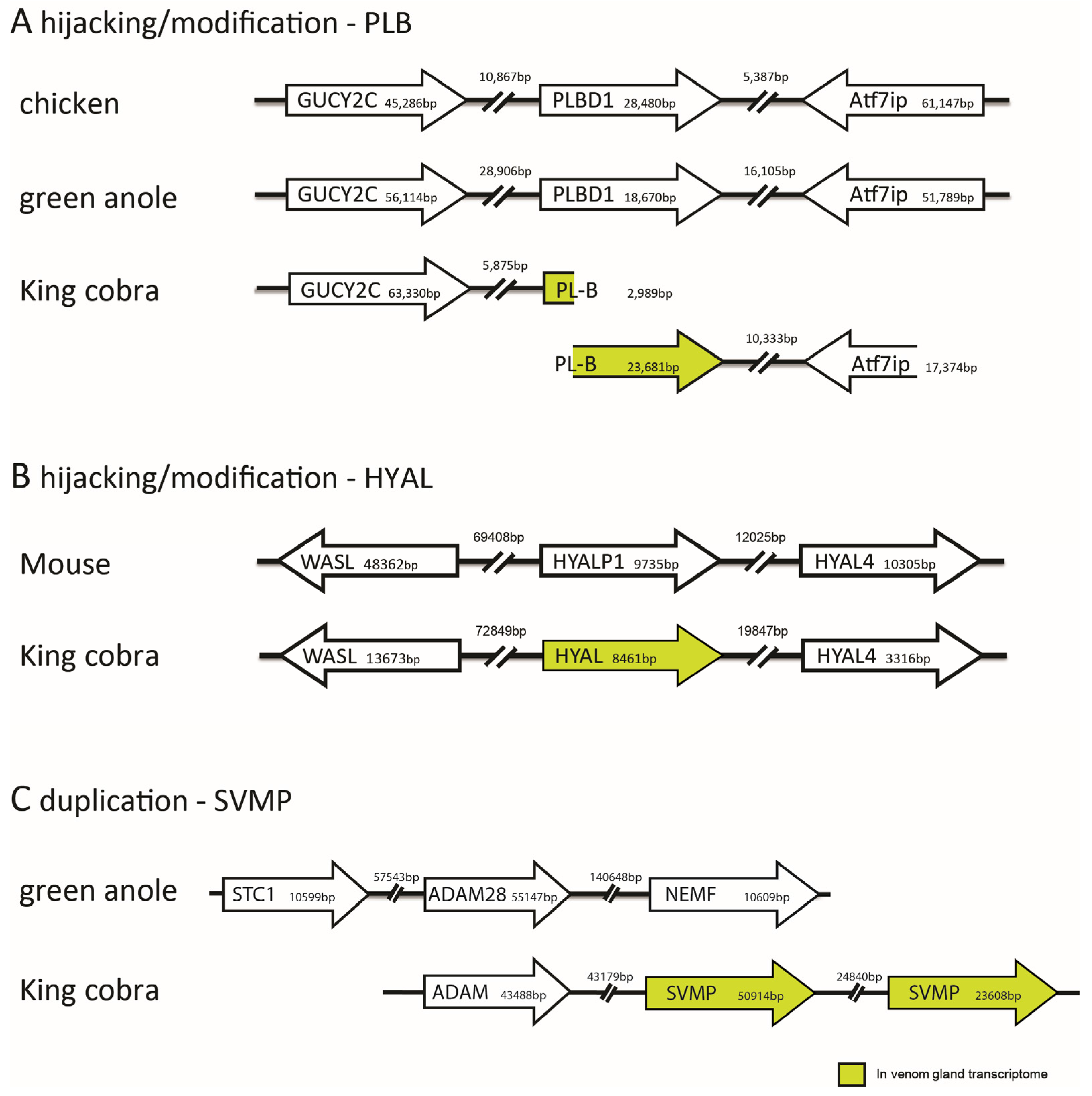
3.3. Gene Duplication
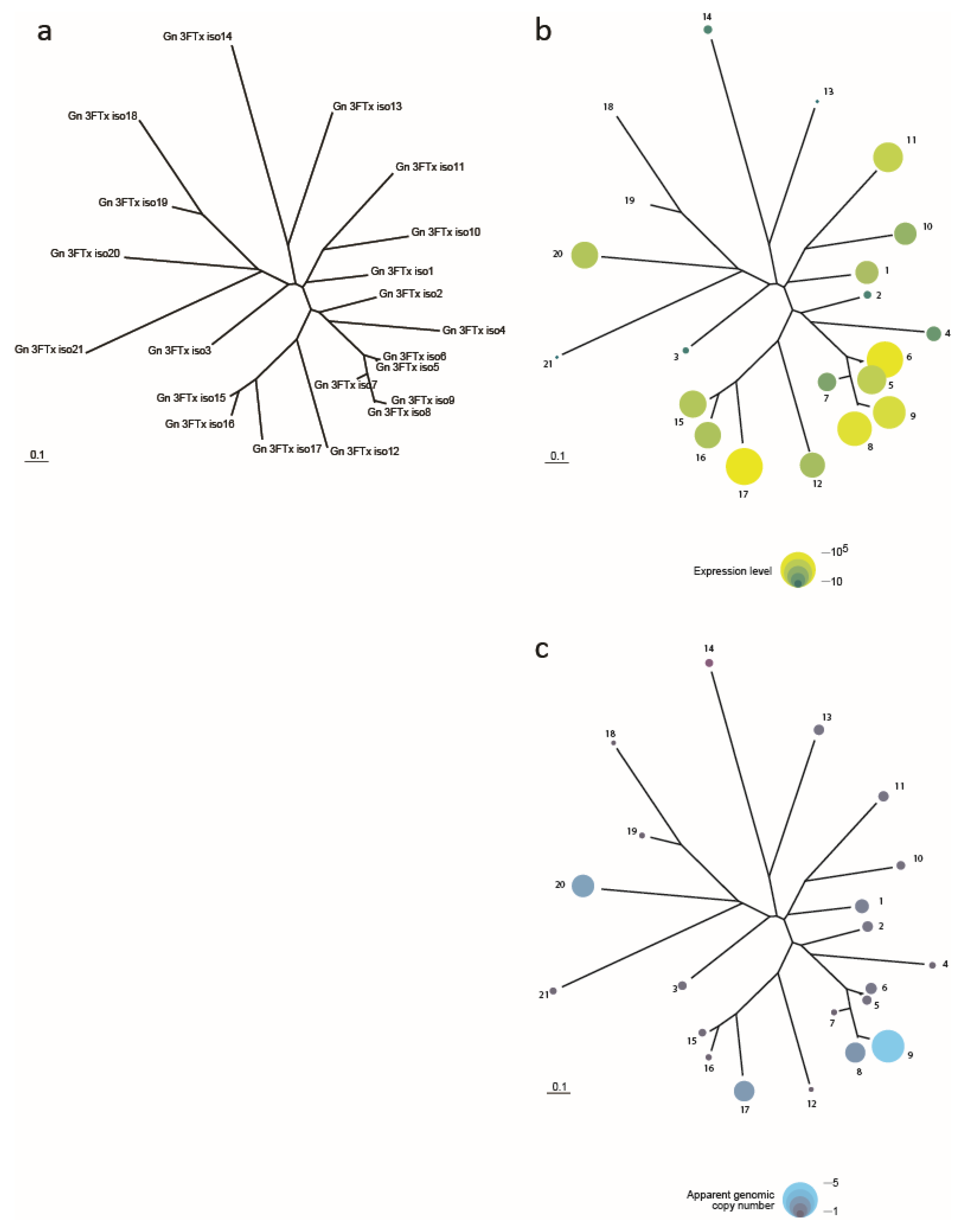
3.4. Possible Selective Advantage of Possessing Multigene Toxin Families
3.5. The Selective Expression of Toxin Genes, or Their Ancestral Orthologues, in the Venom Gland
3.5.1. Recruitment and Neo-Functionalisation Hypothesis
3.5.2. Restriction and Sub-Functionalisation Hypothesis
3.5.3. Testing the Recruitment and Restriction Hypotheses
3.6. Mechanisms of Transcriptional Regulation That Might Have Led to Selective Expression of Toxin Genes in the Venom-Gland
3.6.1. Non-Coding RNA Genes
3.6.2. Transposable Elements
3.6.3. VERSE
3.6.4. AG-Rich Motifs
3.7. Evolution of Toxin Resistance in Snakes as Studied with Genomic Data
4. Transposable Elements and Other Repetitive Sequences in Snake Genomes
5. Future Prospects in Snake Genomics
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Goldman, A.D.; Landweber, L.F. What is a genome? PLoS Genet. 2016, 12. [Google Scholar] [CrossRef] [PubMed]
- Castoe, T.A.; de Koning, A.P.; Hall, K.T.; Card, D.C.; Schield, D.R.; Fujita, M.K.; Ruggiero, R.P.; Degner, J.F.; Daza, J.M.; Gu, W.; et al. The burmese python genome reveals the molecular basis for extreme adaptation in snakes. Proc. Natl. Acad. Sci. USA 2013, 110, 20645–20650. [Google Scholar] [CrossRef] [PubMed]
- Vonk, F.J.; Casewell, N.R.; Henkel, C.V.; Heimberg, A.M.; Jansen, H.J.; McCleary, R.J.; Kerkkamp, H.M.; Vos, R.A.; Guerreiro, I.; Calvete, J.J.; et al. The king cobra genome reveals dynamic gene evolution and adaptation in the snake venom system. Proc. Natl. Acad. Sci. USA 2013, 110, 20651–20656. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.R.; Good, J.M. Targeted capture in evolutionary and ecological genomics. Mol. Ecol. 2016, 25, 185–202. [Google Scholar] [CrossRef] [PubMed]
- Dowell, N.L.; Giorgianni, M.W.; Kassner, V.A.; Selegue, J.E.; Sanchez, E.E.; Carroll, S.B. The deep origin and recent loss of venom toxin genes in rattlesnakes. Curr Biol 2016, 26, 2434–2445. [Google Scholar] [CrossRef] [PubMed]
- Ledford, H. Astrazeneca launches project to sequence 2 million genomes. Nature 2016, 532. [Google Scholar] [CrossRef] [PubMed]
- Koepfli, K.P.; Paten, B.; O’Brien, S.J. The genome 10K project: A way forward. Annu. Rev. Anim. Biosci. 2015, 3, 57–111. [Google Scholar] [CrossRef] [PubMed]
- Schield, D.R.; Card, D.C.; Reyes-Velasco, J.; Andrew, A.L.; Modahl, C.A.; Mackessy, S.M.; Pollock, D.D.; Castoe, T.A. A role for genomics in rattlesnake research—Current knowledge and future potential. In Rattlesnakes of Arizona; Schuett, G.W., Porras, L.W., Reiserer, R.S., Eds.; Eco Books: Rodeo, NM, USA, in press.
- World Health Organization. Rabies and Envenomings: A Neglected Public Health Issue: Report of a Consultative Meeting, World Health Organization, Geneva, 10 January 2007; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Vonk, F.J.; Jackson, K.; Doley, R.; Madaras, F.; Mirtschin, P.J.; Vidal, N. Snake venom: From fieldwork to the clinic: Recent insights into snake biology, together with new technology allowing high-throughput screening of venom, bring new hope for drug discovery. Bioessays 2011, 33, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Coates, M.; Ruta, M. Nice snake, shame about the legs. Trends Ecol. Evol. 2000, 15, 503–507. [Google Scholar] [CrossRef]
- Greene, H.W. Snakes: The Evolution of Mystery in Nature; University of California Press: Berkeley, CA, USA, 1997; p. 351. [Google Scholar]
- Vicoso, B.; Emerson, J.J.; Zektser, Y.; Mahajan, S.; Bachtrog, D. Comparative sex chromosome genomics in snakes: Differentiation, evolutionary strata, and lack of global dosage compensation. PLoS Biol. 2013, 11. [Google Scholar] [CrossRef] [PubMed]
- Bradnam, K.R.; Fass, J.N.; Alexandrov, A.; Baranay, P.; Bechner, M.; Birol, I.; Boisvert, S.; Chapman, J.A.; Chapuis, G.; Chikhi, R.; et al. Assemblathon 2: Evaluating de novo methods of genome assembly in three vertebrate species. Gigascience 2013, 2. [Google Scholar] [CrossRef] [PubMed]
- Ullate-Agote, A.; Milinkovitch, M.C.; Tzika, A.C. The genome sequence of the corn snake (Pantherophis guttatus), a valuable resource for evodevo studies in squamates. Int. J. Dev. Biol. 2014, 58, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Di-Poi, N.; Montoya-Burgos, J.I.; Miller, H.; Pourquie, O.; Milinkovitch, M.C.; Duboule, D. Changes in hox genes’ structure and function during the evolution of the squamate body plan. Nature 2010, 464, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Wang, Z.; Li, Q.; Lian, J.; Zhou, Y.; Lu, B.; Jin, L.; Qiu, P.; Zhang, P.; Zhu, W.; et al. Evolution trajectories of snake genes and genomes revealed by comparative analyses of five-pacer viper. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, C.; Meik, J.M.; Dashevsky, D.; Card, D.C.; Castoe, T.A.; Schaack, S. Endogenous hepadnaviruses, bornaviruses and circoviruses in snakes. Proc. R. Soc. 2014, 281. [Google Scholar] [CrossRef] [PubMed]
- Pubmed taxonomy database. Available online: https://www.Ncbi.Nlm.Nih.Gov/taxonomy (accessed on 30 November 2016).
- Vidal, N.; Delmas, A.S.; David, P.; Cruaud, C.; Couloux, A.; Hedges, S.B. The phylogeny and classification of caenophidian snakes inferred from seven nuclear protein-coding genes. C. R. Biol. 2007, 330, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.R.; Martins, A.R.; Curcio, F.; Ramos, L.O. Osteology and cartilaginous elements of trilepida salgueiroi (amaral, 1954) (scolecophidia: Leptotyphlopidae). Anat. Rec. (Hoboken) 2015, 298, 1722–1747. [Google Scholar] [CrossRef] [PubMed]
- Boulenger, G.A. Catalogue of the Snakes in the British Museum (Natural History); British Museum (Natural History): London, UK, 1893; Volume 1, p. 448. [Google Scholar]
- Cohn, M.J.; Tickle, C. Developmental basis of limblessness and axial patterning in snakes. Nature 1999, 399, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Head, J.J.; Polly, P.D. Evolution of the snake body form reveals homoplasy in amniote hox gene function. Nature 2015, 520, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Van Soldt, B.J.; Metscher, B.D.; Poelmann, R.E.; Vervust, B.; Vonk, F.J.; Muller, G.B.; Richardson, M.K. Heterochrony and early left-right asymmetry in the development of the cardiorespiratory system of snakes. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Vonk, F.J.; Admiraal, J.F.; Jackson, K.; Reshef, R.; de Bakker, M.A.; Vanderschoot, K.; van den Berge, I.; van Atten, M.; Burgerhout, E.; Beck, A.; et al. Evolutionary origin and development of snake fangs. Nature 2008, 454, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Jackson, K. Evolution of the venom conducting fang in snakes. Integr. Comp. Biol. 2002, 42, 1249. [Google Scholar]
- Hofstadler Deiques, C. The development of the pit organ of bothrops jararaca and crotalus durissus terrificus (serpentes, viperidae): Support for the monophyly of the subfamily crotalinae. Acta Zool. 2002, 83, 175–182. [Google Scholar] [CrossRef]
- Gracheva, E.O.; Ingolia, N.T.; Kelly, Y.M.; Cordero-Morales, J.F.; Hollopeter, G.; Chesler, A.T.; Sanchez, E.E.; Perez, J.C.; Weissman, J.S.; Julius, D. Molecular basis of infrared detection by snakes. Nature 2010, 464, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, S.A. Snake venoms: A brief treatise on etymology, origins of terminology, and definitions. Toxicon 2015, 103, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Roelants, K.; Champagne, D.E.; Scheib, H.; Tyndall, J.D.; King, G.F.; Nevalainen, T.J.; Norman, J.A.; Lewis, R.J.; Norton, R.S.; et al. The toxicogenomic multiverse: Convergent recruitment of proteins into animal venoms. Annu. Rev. Genom. Hum. Genet. 2009, 10, 483–511. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wuster, W.; Vonk, F.J.; Harrison, R.A.; Fry, B.G. Complex cocktails: The evolutionary novelty of venoms. Trends Ecol. Evol. 2013, 28, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Velasco, J.; Card, D.C.; Andrew, A.L.; Shaney, K.J.; Adams, R.H.; Schield, D.R.; Casewell, N.R.; Mackessy, S.P.; Castoe, T.A. Expression of venom gene homologs in diverse python tissues suggests a new model for the evolution of snake venom. Mol. Biol. Evol. 2015, 32, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Majoros, W.H.; Pertea, M.; Salzberg, S.L. Tigrscan and glimmerhmm: Two open source ab initio eukaryotic gene-finders. Bioinformatics 2004, 20, 2878–2879. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.E.; White, S.; Searle, S.M.; Stemple, D.L. Incorporating RNA-seq data into the zebrafish ensembl genebuild. Genome Res. 2012, 22, 2067–2078. [Google Scholar] [CrossRef] [PubMed]
- Spielman, S.J.; Wan, S.; Wilke, C.O. A comparison of one-rate and two-rate inference frameworks for site-specific dn/ds estimation. Genetics 2016, 24, 2499–2511. [Google Scholar] [CrossRef] [PubMed]
- Tekaia, F. Inferring orthologs: Open questions and perspectives. Genom. Insights 2016, 9, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Simoes, B.F.; Sampaio, F.L.; Jared, C.; Antoniazzi, M.M.; Loew, E.R.; Bowmaker, J.K.; Rodriguez, A.; Hart, N.S.; Hunt, D.M.; Partridge, J.C.; et al. Visual system evolution and the nature of the ancestral snake. J. Evol. Biol. 2015, 28, 1309–1320. [Google Scholar] [CrossRef] [PubMed]
- Irimia, M.; Maeso, I.; Roy, S.W.; Fraser, H.B. Ancient cis-regulatory constraints and the evolution of genome architecture. Trends Genet. 2013, 29, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Tattini, L.; D’Aurizio, R.; Magi, A. Detection of genomic structural variants from next-generation sequencing data. Front. Bioeng. Biotechnol. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Castoe, T.A.; Hall, K.T.; Mboulas, M.L.G.; Gu, W.; de Koning, A.P.; Fox, S.E.; Poole, A.W.; Vemulapalli, V.; Daza, J.M.; Mockler, T.; et al. Discovery of highly divergent repeat landscapes in snake genomes using high-throughput sequencing. Genome Biol. Evol. 2011, 3, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Telford, M.J.; Copley, R.R. Improving animal phylogenies with genomic data. Trends Genet. 2011, 27, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.S.; Van de Peer, Y.; Braasch, I.; Meyer, A. Comparative genomics provides evidence for an ancient genome duplication event in fish. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2001, 356, 1661–1679. [Google Scholar] [CrossRef] [PubMed]
- Hillier, L.W.; Miller, W.; Birney, E.; Warren, W.; Hardison, R.C.; Ponting, C.P.; Bork, P.; Burt, D.W.; Groenen, M.A.; Delany, M.E.; et al. Sequence and comparative analysis of the chicken genome provide unique perspectives on vertebrate evolution. Nature 2004, 432, 695–716. [Google Scholar] [CrossRef] [PubMed]
- Wicker, T.; Robertson, J.S.; Schulze, S.R.; Feltus, F.A.; Magrini, V.; Morrison, J.A.; Mardis, E.R.; Wilson, R.K.; Peterson, D.G.; Paterson, A.H.; et al. The repetitive landscape of the chicken genome. Genome Res. 2005, 15, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Ezkurdia, I.; Juan, D.; Rodriguez, J.M.; Frankish, A.; Diekhans, M.; Harrow, J.; Vazquez, J.; Valencia, A.; Tress, M.L. Multiple evidence strands suggest that there may be as few as 19,000 human protein-coding genes. Hum. Mol. Genet. 2014, 23, 5866–5878. [Google Scholar] [CrossRef] [PubMed]
- De Koning, A.P.; Gu, W.; Castoe, T.A.; Batzer, M.A.; Pollock, D.D. Repetitive elements may comprise over two-thirds of the human genome. PLoS Genet. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Alfoldi, J.; Di Palma, F.; Grabherr, M.; Williams, C.; Kong, L.; Mauceli, E.; Russell, P.; Lowe, C.B.; Glor, R.E.; Jaffe, J.D.; et al. The genome of the green anole lizard and a comparative analysis with birds and mammals. Nature 2011, 477, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G. From genome to “venome”: Molecular origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences and related body proteins. Genome Res. 2005, 15, 403–420. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, A.D.; Swain, M.T.; Hegarty, M.J.; Logan, D.W.; Mulley, J.F. Restriction and recruitment-gene duplication and the origin and evolution of snake venom toxins. Genome Biol. Evol. 2014, 6, 2088–2095. [Google Scholar] [CrossRef] [PubMed]
- Cousin, X.; Bon, S.; Massoulie, J.; Bon, C. Identification of a novel type of alternatively spliced exon from the acetylcholinesterase gene of bungarus fasciatus. Molecular forms of acetylcholinesterase in the snake liver and muscle. J. Biol. Chem. 1998, 273, 9812–9820. [Google Scholar] [CrossRef] [PubMed]
- Sunagar, K.; Fry, B.G.; Jackson, T.N.; Casewell, N.R.; Undheim, E.A.; Vidal, N.; Ali, S.A.; King, G.F.; Vasudevan, K.; Vasconcelos, V.; et al. Molecular evolution of vertebrate neurotrophins: Co-option of the highly conserved nerve growth factor gene into the advanced snake venom arsenalf. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Kostiza, T.; Meier, J. Nerve growth factors from snake venoms: Chemical properties, mode of action and biological significance. Toxicon 1996, 34, 787–806. [Google Scholar] [CrossRef]
- Wijeyewickrema, L.C.; Gardiner, E.E.; Gladigau, E.L.; Berndt, M.C.; Andrews, R.K. Nerve growth factor inhibits metalloproteinase-disintegrins and blocks ectodomain shedding of platelet glycoprotein vi. J. Biol. Chem. 2010, 285, 11793–11799. [Google Scholar] [CrossRef] [PubMed]
- Junqueira-de-Azevedo, I.L.; Bastos, C.M.; Ho, P.L.; Luna, M.S.; Yamanouye, N.; Casewell, N.R. Venom-related transcripts from bothrops jararaca tissues provide novel molecular insights into the production and evolution of snake venom. Mol. Biol. Evol. 2015, 32, 754–766. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, C.J. Protein species and moonlighting proteins: Very small changes in a protein’s covalent structure can change its biochemical function. J. Proteom. 2016, 134, 19–24. [Google Scholar] [CrossRef] [PubMed]
- True, J.R.; Carroll, S.B. Gene co-option in physiological and morphological evolution. Annu. Rev. Cell Dev. Biol. 2002, 18, 53–80. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.S.; Raes, J. Duplication and divergence: The evolution of new genes and old ideas. Annu. Rev. Genet. 2004, 38, 615–643. [Google Scholar] [CrossRef] [PubMed]
- Loewe, L.; Hill, W.G. The population genetics of mutations: Good, bad and indifferent. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 1153–1167. [Google Scholar] [CrossRef] [PubMed]
- Force, A.; Lynch, M.; Pickett, F.B.; Amores, A.; Yan, Y.L.; Postlethwait, J. Preservation of duplicate genes by complementary, degenerative mutations. Genetics 1999, 151, 1531–1545. [Google Scholar] [PubMed]
- Fry, B.G.; Wuster, W.; Kini, R.M.; Brusic, V.; Khan, A.; Venkataraman, D.; Rooney, A.P. Molecular evolution and phylogeny of elapid snake venom three-finger toxins. J. Mol. Evol. 2003, 57, 110–129. [Google Scholar] [CrossRef] [PubMed]
- Nei, M.; Rooney, A.P. Concerted and birth-and-death evolution of multigene families. Annu. Rev. Genet. 2005, 39, 121–152. [Google Scholar] [CrossRef] [PubMed]
- Meisler, M.H.; Antonucci, T.K.; Treisman, L.O.; Gumucio, D.L.; Samuelson, L.C. Interstrain variation in amylase gene copy number and mRNA abundance in three mouse tissues. Genetics 1986, 113, 713–722. [Google Scholar] [PubMed]
- Olivera, B.M.; Seger, J.; Horvath, M.P.; Fedosov, A.E. Prey-capture strategies of fish-hunting cone snails: Behavior, neurobiology and evolution. Brain Behav. Evol. 2015, 86, 58–74. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Vidal, N.; Norman, J.A.; Vonk, F.J.; Scheib, H.; Ramjan, S.F.; Kuruppu, S.; Fung, K.; Hedges, S.B.; Richardson, M.K.; et al. Early evolution of the venom system in lizards and snakes. Nature 2006, 439, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Sanz-Soler, R.; Sanz, L.; Calvete, J.J. Distribution of rptln genes across reptilia: Hypothesized role for rptln in the evolution of svmps. Integr. Comp. Biol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wagstaff, S.C.; Wuster, W.; Cook, D.A.; Bolton, F.M.; King, S.I.; Pla, D.; Sanz, L.; Calvete, J.J.; Harrison, R.A. Medically important differences in snake venom composition are dictated by distinct postgenomic mechanisms. Proc. Natl. Acad. Sci. USA 2014, 111, 9205–9210. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Huttley, G.A.; Wuster, W. Dynamic evolution of venom proteins in squamate reptiles. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, A.D.; Swain, M.T.; Logan, D.W.; Mulley, J.F. Testing the toxicofera: Comparative transcriptomics casts doubt on the single, early evolution of the reptile venom system. Toxicon 2014, 92, 140–156. [Google Scholar] [CrossRef] [PubMed]
- Sanz, L.; Calvete, J.J. Insights into the evolution of a snake venom multi-gene family from the genomic organization of echis ocellatus svmp genes. Toxins (Basel) 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Han, S.X.; Kwong, S.; Ge, R.; Kolatkar, P.R.; Woods, A.E.; Blanchet, G.; Kini, R.M. Regulation of expression of venom toxins: Silencing of prothrombin activator trocarin d by ag-rich motifs. FASEB J. 2016, 30, 2411–2425. [Google Scholar] [CrossRef] [PubMed]
- Feldman, C.R.; Durso, A.M.; Hanifin, C.T.; Pfrender, M.E.; Ducey, P.K.; Stokes, A.N.; Barnett, K.E.; Brodie, E.D., 3rd; Brodie, E.D., Jr. Is there more than one way to skin a newt? Convergent toxin resistance in snakes is not due to a common genetic mechanism. Heredity (Edinb) 2016, 116, 84–91. [Google Scholar] [CrossRef] [PubMed]
- McGlothlin, J.W.; Kobiela, M.E.; Feldman, C.R.; Castoe, T.A.; Geffeney, S.L.; Hanifin, C.T.; Toledo, G.; Vonk, F.J.; Richardson, M.K.; Brodie, E.D.; et al. Historical contingency in a multigene family facilitates adaptive evolution of toxin resistance. Curr. Biol. 2016, 26, 1616–1621. [Google Scholar] [CrossRef] [PubMed]
- Soong, T.W.; Venkatesh, B. Adaptive evolution of tetrodotoxin resistance in animals. Trends Genet. 2006, 22, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Padeken, J.; Zeller, P.; Gasser, S.M. Repeat DNA in genome organization and stability. Curr. Opin. Genet. Dev. 2015, 31, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Marcinkiewicz, C. Functional characteristic of snake venom disintegrins: Potential therapeutic implication. Curr. Pharm. Des. 2005, 11, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Laing, G.D.; Moura-da-Silva, A.M. Jararhagin and its multiple effects on hemostasis. Toxicon 2005, 45, 987–996. [Google Scholar] [CrossRef] [PubMed]
- McCleary, R.J.; Kini, R.M. Non-enzymatic proteins from snake venoms: A gold mine of pharmacological tools and drug leads. Toxicon 2013, 62, 56–74. [Google Scholar] [CrossRef] [PubMed]
- Kini, R.M.; Doley, R. Structure, function and evolution of three-finger toxins: Mini proteins with multiple targets. Toxicon 2010, 56, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Wagstaff, S.C.; Laing, G.D.; Theakston, R.D.; Papaspyridis, C.; Harrison, R.A. Bioinformatics and multiepitope DNA immunization to design rational snake antivenom. PLoS Med. 2006, 3. [Google Scholar] [CrossRef] [PubMed]
- Bird, A. Perceptions of epigenetics. Nature 2007, 447, 396–398. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, E.; Larkeryd, A.; Sjodin, A.; Forsman, M.; Stenberg, P. Scaffolding of a bacterial genome using minion nanopore sequencing. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Giordano, F.; Ning, Z. Oxford nanopore minion sequencing and genome assembly. Genom. Proteom. Bioinform. 2016, 31, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.; Wu, L.; Yavas, G.; Simonyan, V.; Ning, B.; Hong, H. Challenges, solutions, and quality metrics of personal genome assembly in advancing precision medicine. Pharmaceutics 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Levy-Sakin, M.; Ebenstein, Y. Beyond sequencing: Optical mapping of DNA in the age of nanotechnology and nanoscopy. Curr. Opin. Biotechnol. 2013, 24, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Saliba, A.E.; Westermann, A.J.; Gorski, S.A.; Vogel, J. Single-cell RNA-seq: Advances and future challenges. Nucleic Acids Res. 2014, 42, 8845–8860. [Google Scholar] [CrossRef] [PubMed]
| Trivial Name | Scientific Name | Family | Notes |
|---|---|---|---|
| Prong-snouted blind snake | Anilios bituberculatus | Typhlopidae | F.J. Vonk et al., in progress |
| Texas blind snake | Rena dulcis | Leptotyphlopidae | T.A. Castoe et al., in progress |
| Boa constrictor | Boa constrictor | Boidae | Ref. [13]; GenB: PRJNA210004 |
| Boa constrictor | Boa constrictor | Boidae | Ref. [14] |
| Burmese python | Python bivittatus | Pythonidae | Published [2]; GenB: AEQU00000000 |
| Garter snake | Thamnophis sirtalis | Colubridae | GenB: LFLD00000000 |
| Thamnophis elegans | Colubridae | Ref. [13]; GenB: PRJNA210004 | |
| Corn snake | Pantherophis guttatus | Colubridae | Ref. [15]; GenB: JTLQ01000000 |
| Corn snake | Pantherophis guttatus | Colubridae | Targeted sequencing: 5′ hox genes [16] |
| King cobra | Ophiophagus hannah | Elapidae | Published [3]; GenB: AZIM00000000 |
| Malayan pit viper | Calloselasma rhodostoma | Viperidae | F.J. Vonk et al., in progress |
| Five-pacer viper | Deinagkistrodon acutus | Viperidae | Ref. [17] |
| European adder | Vipera berus berus | Viperidae | Baylor College of Medicine, Human Genome Sequencing Center; GenB: JTGP00000000 |
| Habu | Protobothrops flavoviridis | Viperidae | H. Shibata et al., in progress |
| Brown spotted pit viper | Protobothrops mucrosquamatus | Viperidae | A.S. Mikheyev et al., in progress; GenB: PRJDB4386 |
| Prairie rattlesnake | Crotalus viridis viridis | Viperidae | T.A. Castoe et al., in progress |
| Western diamond-backed rattlesnake | Crotalus atrox | Viperidae | Ref. [5] |
| Timber rattlesnake | Crotalus horridus | Viperidae | GenB: LVCR00000000.1 |
| Speckled rattlesnake | Crotalus mitchellii pyrrhus | Viperidae | Ref. [18]; GenB: JPMF01000000 |
| Western Diamondback rattlesnake, Mojave rattlesnake and Eastern Diamondback rattlesnake | Crotalus atrox, C. scutulatus, and C. adamanteus | Viperidae | Targeted sequencing of bacterial artificial chromosome (BAC) clones containing phospholipase A2 genes. |
| Pygmy rattlesnake | Sistrurus miliarius | Viperidae | Ref. [13]; GenB: PRJNA210004 |
| Temple pit viper | Tropidolaemus wagleri | Viperidae | R.M. Kini et al., in progress |
| Species | Coding Genes (k) | Genome Size (Gb) | Repeats (%) |
|---|---|---|---|
| Burmese python | 25 [2] | 1.44 [2] | 31.8–59.4 [2] |
| King cobra | 21.19 [3] | 1.36–1.59 [3] | 35.2–60.4 [2] |
| Chicken | 20–23 * [44] | 1.05 [44] | 4.3–8.0 [45]; 9.4 [44] |
| Human | 20.4 ¶; 19 [46] | 3.54 ¶ | >66–69 [47] |
| Anolis | 18.5 † | 1.70 † | 30% ‡ [48] |
| Venom Toxin or Toxin Family | Number of Paralogues |
|---|---|
| 3FTx (three-finger toxin) * | 21 |
| PLA2 (phospholipase A2) * | 12 |
| Lectin * | 11 |
| Kunitz * | 10 |
| Waprins * | 6 |
| Cystatin | 5 |
| CRISP (cysteine-rich secretory protein) | 3 |
| CVF (cobra venom factor) | 3 |
| Kallikrein | 3 |
| SVMP (snake venom metalloproteinase) | 3 |
| LAAO (L-amino acid oxidase) | 2 |
| NGF (nerve growth factor) | 2 |
| NP (natriuretic peptide) * | 2 |
| Acetylcholinesterase | 1 |
| Hyaluronidase | 1 |
| PLB (phospholipase-B) | 1 |
| VEGF (vascular endothelial growth factors) | 1 |
| Vespryn | 1 |
| 3FTX Isoform | Nucleotide Sequence | Accession Code Genbank |
|---|---|---|
| Iso1 | GATACACCTTGACATGTCTAACACATGAATCATTATTTTTTGAAACCACTGAGACTTGTTCAGATGGGCAGAACCTATGCTATGCAAAATGGTTTGCAGTTTTTCCAGGTG | AZIM01011044.1 |
| Iso2 | GATACACCAGGATATGCCACAAATCTTCTTTTATCTCTGAGACTTGTCCAGATGGGCAGAACCTATGCTATTTAAAATCGTGGTGTGACATTTTTT | AZIM01016929.1 |
| Iso3 | GATACACCTTGACATGCATCACATCTGCTCGTAACTTTGAGACTTGTCCACCTGGGCAGAACCTATGCTTTTTAAAATCATGGTATGAAGCTTCAT | AZIM01214498.1 |
| Iso4 | TACAAAACCGGTGAACGTATTATTTCTGAGACTTGTCCCCCTGGGCAGGACCTATGCTATATGAAGACTTGGTGTGACGTTTTTT | AZIM01146344.1 |
| Iso5 | GATACACCATGACATGTTACACACAGTACTCATTGTCTCCTCCAACCACTAAGACTTGTCCAGATGGGCAGAACCTATGCTATAAAAGGTGATTTGCGTTTATTCCACATG | AZIM01015434.1 |
| Iso6 | GATACACCACGAAATGCTACGTAACACCTGATGCTACCTCTCAGACTTGTCCAGATGGGGAGAACATATGCTATACAAAGTCTTGGTGTGACGGTTTTT | AZIM01133918.1 |
| Iso7 | GATACACCACGAAATGCTATGTAACACCTGATGCTACCTCTCAGACTTGTCCAGATGGGGAGAACATATGCTATACAAAGTCTTGGTGTGACGTTTTTT | AZIM01229389.1 |
| Iso8 | GATACACCACGAAATGCTACATAACACCTGATGTGAAGTCTCAGACTTGTCCAGATGGGGAGAACATATGCTATACAAAGACTTGGTGTGATGTTTGGT | AZIM01229389.1 |
| Iso9 | GATACACCACGAAATGCTACGTAACACCTGATGTTAAGTCTGAGACTTGTCCAGATGGGCAGGACATATGCTATACAGAGACTTGGTGTGACGTTTGGT | AZIM01028336.1 |
| Iso10 | GATACACCACGAAATGCTACGTAACACCTGATGTTAAGTCTGAGACTTGTCCAGCTGGGCAGGACATATGCTATACAGAGACTTGGTGTGATGCTTGGT | AZIM01097792.1 |
| Iso11 | GACACACCAGGATATGTCTCACAGACTACTCAAAAGTTAGTGAAACCATTGAGATTTGTCCAGATGGGCAGAACTTCTGCTTTAAAAAGTTTCCTAAGGGTATTCCATTTT | AZIM01006046.1 |
| Iso12 | GATACACCATGAAATGTCTCACAAAGTACTCCCGGGTTAGTGAAACCTCTCAGACTTGTCACGTTTGGCAGAACCTATGTTTTAAAAAGTGGCAGAAGG | AZIM01011575.1 |
| Iso13 | GACACACCTTGATATGTGTCAAACAGTACACAATTTTTGGTGTAACCCCTGAGATTTGCGCAGATGGGCAGAACCTATGCTATAAAACATGGCATATGGTGTATCCAGGTG | AZIM01011969.1 |
| Iso14 | GATACACCACGAAATGTTACAACCACCAGTCAACGACTCCTGAAACCACTGAAATTTGTCCAGATTCAGGGTACTTTTGCTATAAAAGCTCTTGGATTGATGGACGTG | AZIM01034614.1 |
| Iso15 | GATACACCCTGATATGTCACCGAGTGCATGGACTTCAGACTTGTGAACCAGATGAGAAGTTTTGCTTTAGAAAGACGACAATGTTTTTTCCAAATC | AZIM01009352.1 |
| Iso16 | GATACACCAGGAAATGTCTCAACACACCGCTTCCTTTGATCTATANTTAAAATGACTATTAAGAAGTTGCCATCTA | AZIM01009586.1 |
| Iso17 | NATACACCAGGATATGTTTAAAGCAAGAGCCATTTCAACCTGAAACCAGTACAACTTGTCCAGATGGGGAAGATGCTTGCTATAGTACATTTTGGAGTGATAACC | AZIM01019523.1 |
| Iso18 | NATACACCAGGATATGTTTAAAGCAAGAGCCGTTTCAACCTGAAACCACTACAACTTGTCCAGAAGGGGAGGATGCTTGCTATAATTTGTTTTGGAGTGATCACA | AZIM01052732.1 |
| Iso19 | GATACAGCTTGATATGTTTTAACCAAGAGACGTATCGACCTGAAACCACTACAACTTGTCCAGATGGGGAGGACACTTGCTATAGTACATTTTGGAATGATCACCATG | AZIM01009977.1 |
| Iso20 | CACAAACCAAGACATGTTACTCATGCACTGGAGCATTTTGTTCTAATCGTCAAAAATGTTCGGGTGGGCAGGTCATATGCTTTAAAAGTTGGAAAAATACTCTTCTGATAT | AZIM01013260.1 |
| Iso21 | CACACACCCTGACATGTTACTCATGCAATGGATTATTATGTTCTGACCGTGAACAATGTCCAGATGGGTAGGACATATGCTTTAAGAGATGGAATGATACTGATTGGTCAG | AZIM01013561.1 |
| Iso22 | GATACAGCTTGACATGTCTCAATTGCCCAGAACAGTATTGTAAAAGAATTCACACTTGTCGAGATGGGGAGAACGTATGCTTTAAAAGGTTTTACGAGGGTAAACTATTAT | AZIM01071124.1 |
| Iso23 | GATACACTCTGTTGTGTTGCAAATGCAATCAAACGGTTTGTGATCTCAATTCGTATTGTTCAGCAGGCAAGAACCAATGCTATATATTGCAGAATAATA | AZIM01008565.1 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kerkkamp, H.M.I.; Kini, R.M.; Pospelov, A.S.; Vonk, F.J.; Henkel, C.V.; Richardson, M.K. Snake Genome Sequencing: Results and Future Prospects. Toxins 2016, 8, 360. https://doi.org/10.3390/toxins8120360
Kerkkamp HMI, Kini RM, Pospelov AS, Vonk FJ, Henkel CV, Richardson MK. Snake Genome Sequencing: Results and Future Prospects. Toxins. 2016; 8(12):360. https://doi.org/10.3390/toxins8120360
Chicago/Turabian StyleKerkkamp, Harald M. I., R. Manjunatha Kini, Alexey S. Pospelov, Freek J. Vonk, Christiaan V. Henkel, and Michael K. Richardson. 2016. "Snake Genome Sequencing: Results and Future Prospects" Toxins 8, no. 12: 360. https://doi.org/10.3390/toxins8120360
APA StyleKerkkamp, H. M. I., Kini, R. M., Pospelov, A. S., Vonk, F. J., Henkel, C. V., & Richardson, M. K. (2016). Snake Genome Sequencing: Results and Future Prospects. Toxins, 8(12), 360. https://doi.org/10.3390/toxins8120360







