Reduction of Aflatoxins in Apricot Kernels by Electronic and Manual Color Sorting
Abstract
:1. Introduction
2. Results and Discussion
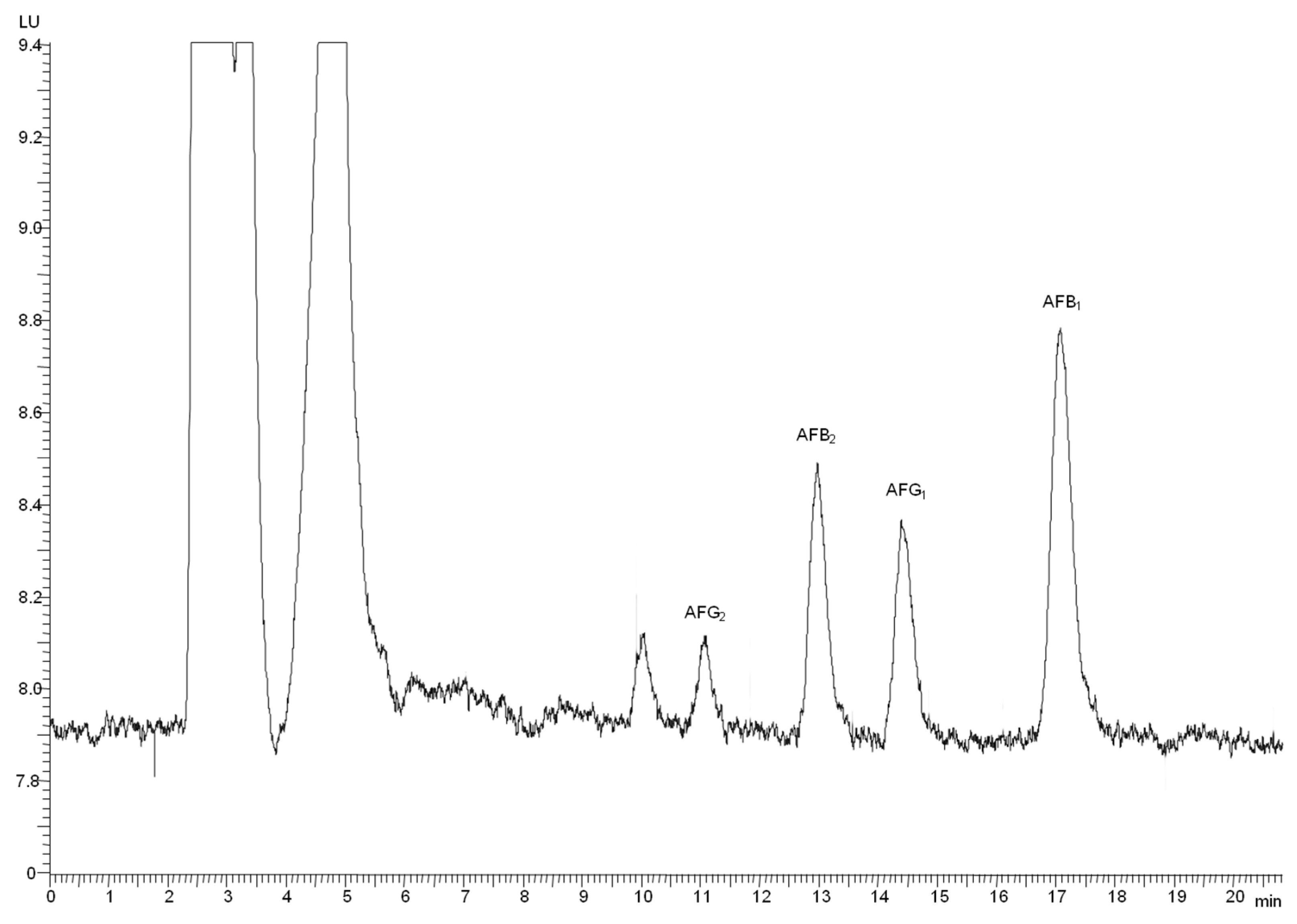
2.1. Method Performance
| Matrices | LOD μg/kg | LOQ μg/kg | ||||||
|---|---|---|---|---|---|---|---|---|
| AFB1 | AFB2 | AFG1 | AFG2 | AFB1 | AFB2 | AFG1 | AFG2 | |
| Healthy apricot kernels a | 0.4 | 0.08 | 0.56 | 0.15 | 0.8 | 0.16 | 1.12 | 0.30 |
| Discoloured apricot kernels b | 24 | 17.6 | - | - | 48 | 35.2 | - | - |
| Apricot kernel skins c | 1.40 | 0.48 | 3.10 | 1.26 | 2.80 | 0.96 | 5.40 | 2.52 |
| Commercial products d | 0.03 | 0.01 | 0.05 | 0.02 | 0.06 | 0.02 | 0.1 | 0.04 |
| Matrices | AFB1 | Total AFS | ||||
|---|---|---|---|---|---|---|
| Spiking level (µg/kg) | Recovery (%) | RSDr (%) | Spiking level (µg/kg) | Recovery (%) | RSDr (%) | |
| Almonds | 0.2 | 107 | 6.9 | 0.5 | 100 | 9.8 |
| 1.0 | 96 | 3.4 | 2.5 | 94 | 2.9 | |
| Peeled almonds | 0.2 | 102 | 4.1 | 0.5 | 94 | 10.6 |
| 1.0 | 94 | 4.6 | 2.5 | 91 | 6.6 | |
| Amaretti | 0.2 | 115 | 4.1 | 0.5 | 103 | 5.6 |
| 1.0 | 98 | 1.3 | 2.5 | 94 | 1.8 | |
| Cantucci | 0.2 | 91 | 5.3 | 0.5 | 86 | 4.0 |
| 1.0 | 92 | 2.9 | 2.5 | 89 | 3.5 | |
| Almond nougat | 0.2 | 98 | 5.3 | 0.5 | 85 | 7.1 |
| 1.0 | 96 | 3.2 | 2.5 | 91 | 2.5 | |
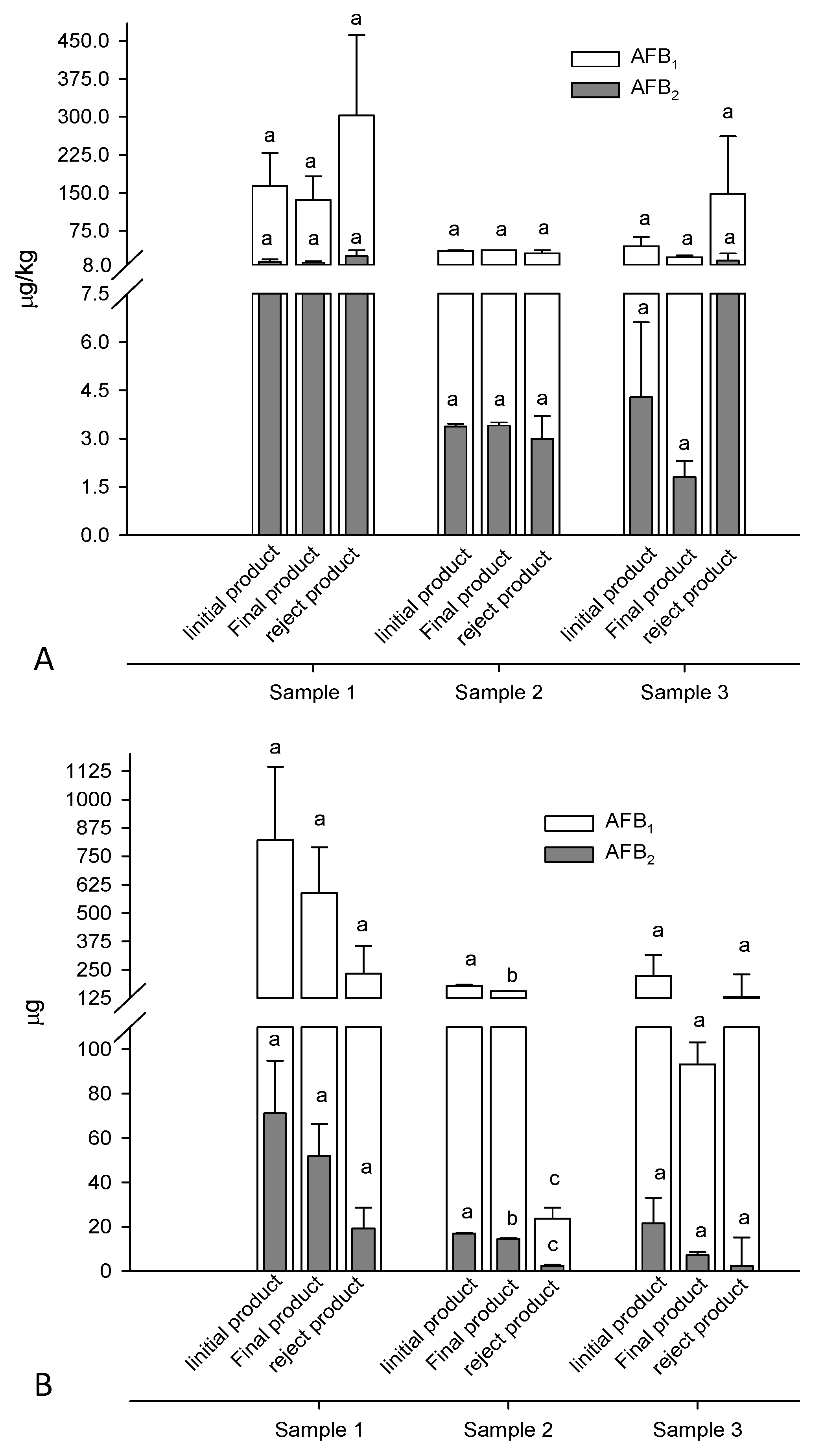
2.2. Electronic Color Sorting (e-Sorting) of Apricot Kernels
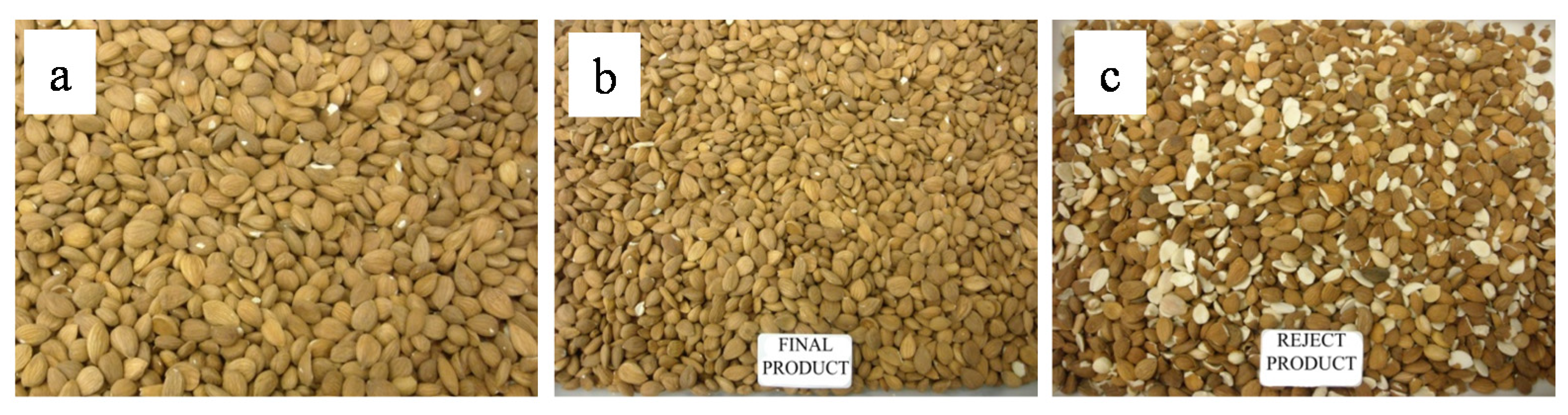
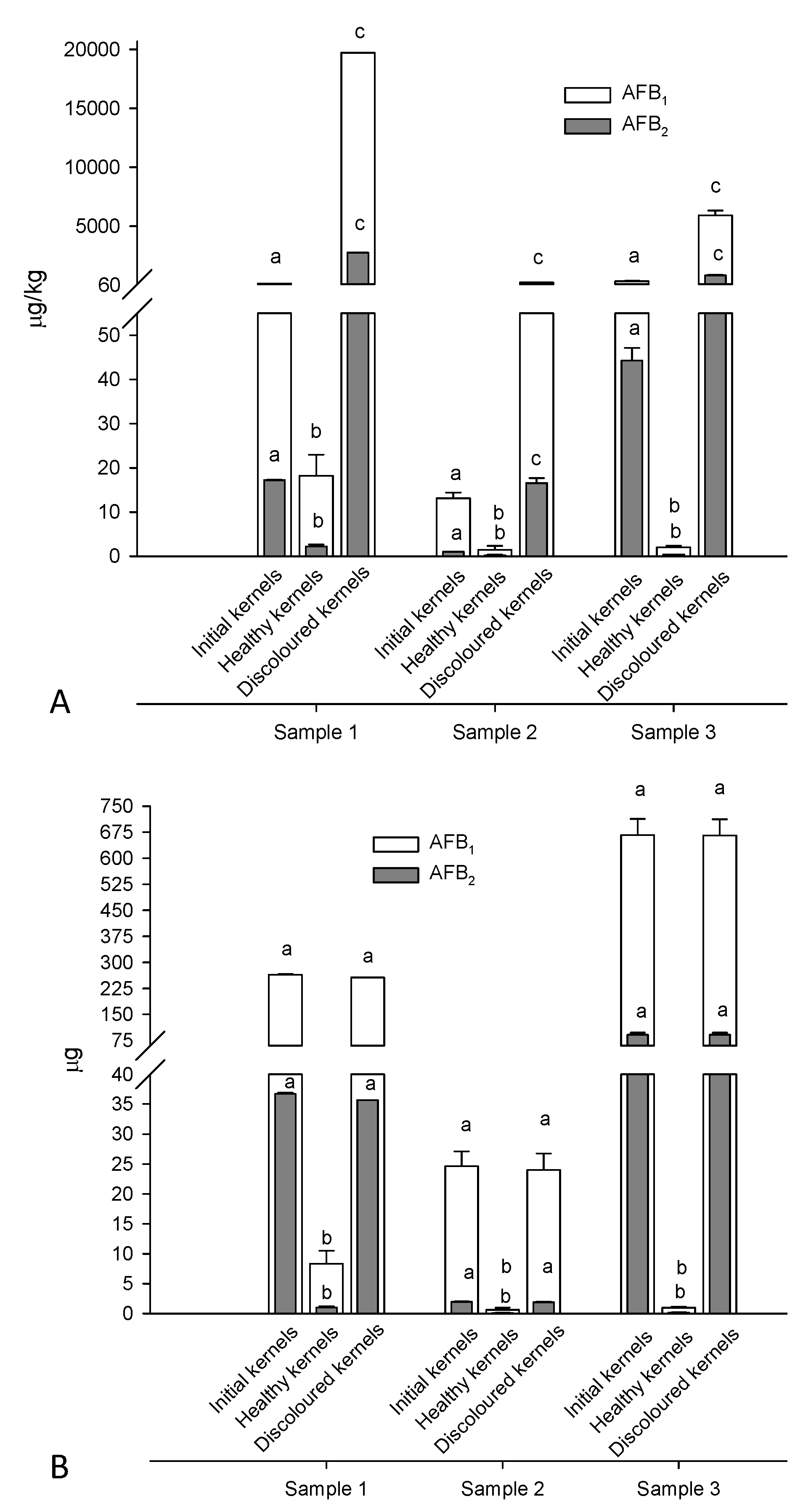
2.3. Blanching, Peeling, and Manual Sorting of Apricot kernels

2.4. Occurrence of Aflatoxins in Commercial Products Containing Apricot Kernels and/or Almonds
| Commercial Product | % of Almonds or Apricot Kernels | Origin | AFB1 (µg/kg) | Total AFs (µg/kg) |
|---|---|---|---|---|
| Almonds | 100% | Italy | 0.06 | 0.06 |
| Almonds | 100% | California, US | n.d. | n.d. |
| Almonds | 100% | California, US | n.d. | n.d. |
| Almonds | 100% | Italy b | n.d. | n.d. |
| Almonds | 100% | Italy | n.d. | n.d. |
| Almonds | 100% | Italy b | n.d. | n.d. |
| Almonds | 100% | Italy | n.d. | n.d. |
| Peeled almonds | 100% | Italy | 0.06 | 0.07 |
| Peeled almonds | 100% | California, US | 0.06 | 0.06 |
| Peeled almonds | 100% | Italy b | 0.06 | 0.06 |
| Peeled almonds | 100% | Italy | 0.06 | 0.06 |
| Peeled almonds | 100% | California, US | n.d. | n.d. |
| Peeled almonds | 100% | California, US | n.d. | n.d. |
| Almond flour | 100% | Italy | 0.06 | 0.06 |
| Chopped almonds | 100% | Italy b | 0.06 | 0.06 |
| Bakery product (4% almonds) a | 100% | Italy b | 0.06 | 0.16 |
| Bakery product (4% almonds) a | 100% | Italy b | n.d. | n.d. |
| Bakery product (3% chopped peeled almonds) a | 100% | Italy b | n.d. | n.d. |
| Bakery product (3% almonds) a | 100% | Italy b | n.d. | n.d. |
| Bakery product (3% peeled almonds) a | 100% | Italy b | n.d. | n.d. |
| Bakery product (4% peeled almonds) a | 100% | Italy b | n.d. | n.d. |
| Bakery product (4% peeled almonds) a | 100% | Italy b | n.d. | n.d. |
| Bakery pastries | 7% Almonds | Italy b | n.d. | n.d. |
| Mixed nuts snack (almonds, peanuts, sultana grapes, cranberry) | 25% Almonds | California, US | n.d. | n.d. |
| Salt roasted almonds with EVO oil | 100% | Italy | 0.11 | 0.33 |
| Salt roasted almonds | 100% | Italy b | n.d. | n.d. |
| Smoked almonds | 100% | Italy b | n.d. | n.d. |
| Sugared almonds | 100% | Italy | n.d. | n.d. |
| Almonds soft nougat | 50% Almonds | Italy b | n.d. | n.d. |
| Almonds nougat | 45% Almonds | Italy b | n.d. | n.d. |
| Almonds nougat | 45% Almonds | Italy b | n.d. | n.d. |
| Almonds soft nougat | 40% Almonds | Italy b | n.d. | n.d. |
| Almonds nougat | 36% Almonds | Italy b | n.d. | n.d. |
| Almonds soft nougat | 30% Almonds | Italy b | n.d. | n.d. |
| Cantucci | 22% Almonds | Italy b | 0.06 | 0.07 |
| Cantucci | 22% Almonds | Italy b | 0.06 | 0.06 |
| Cantucci | 22% Almonds | Italy b | n.d. | n.d. |
| Cantucci | 20% Almonds | Italy b | n.d. | n.d. |
| Cantucci | 19% Almonds | Italy b | n.d. | n.d. |
| Mean of positive samples | - | - | 0.07 | 0.11 |
| Amaretti | 38% Apricot kernels, 4% Almonds, 4% Pistachios | Italy b | 1.30 | 1.79 |
| Amaretti | 40% Apricot kernels, 5% Almonds | Italy b | 1.50 | 1.56 |
| Amaretti | 20% Apricot kernels | Italy b | 0.55 | 0.99 |
| Amaretti | 20% Apricot kernels | Italy b | 0.53 | 0.56 |
| Amaretti | 19% Apricot kernels | Italy b | 0.40 | 0.41 |
| Amaretti | 16% Apricot kernels, Sweet and bitter almonds | Italy b | 0.08 | 0.30 |
| Amaretti | 20% Apricot kernels | Italy b | 0.06 | 0.06 |
| Amaretti | 20% Apricot kernels | Italy b | n.d. | n.d. |
| Mean of positive samples | - | - | 0.63 | 0.81 |
3. Experimental Section
3.1. Samples
3.2. Electronic Color Sorting of Apricot Kernels
3.3. Blanching, Peeling, and Manual Sorting of Apricot Kernels
3.4. Determination of AFB1, AFB2, AFG1 and AFG2
3.5. Chemicals and Reagents
3.6. HPLC-FLD Apparatus and Conditions
3.7. Calibration Curves
3.8. Recovery Experiments
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations Statistics Division. Available online: http://faostat3.fao.org/browse/Q/QC/ (accessed on 28 December 2015).
- Tuncel, G.; Nout, M.J.R.; Brimec, L. Degradation of cyanogenic glycosides of bitter apricot seeds (Prunus armeniaca) by endogenous and added enzymes as affected by heat treatments and particle size. Food Chem. 1998, 63, 65–69. [Google Scholar] [CrossRef]
- Haciseferogullari, H.; Gezer, I.; Ozcan, M.M.; Murat Asma, B. Post harvest and physical-mechanical properties of some apricot varieties cultivated in Turkey. J. Food Eng. 2007, 79, 364–373. [Google Scholar] [CrossRef]
- Gezer, I.; Haciseferogullari, H.; Demir, F. Some physical properties of Hacıhaliloglu apricot pit and its kernel. J. Food Eng. 2002, 56, 49–57. [Google Scholar] [CrossRef]
- Femenia, A.; Rossellò, C.; Mulet, A.; Cañellas, J. Chemical composition of bitter and sweet apricot kernels. J. Agric. Food Chem. 1995, 43, 356–361. [Google Scholar] [CrossRef]
- Hallabo, S.A.S.; El-Wakeil, F.A.; Morsi, M.; Khairy, S. Chemical and physical properties of apricot kernel oil and almond kernel oil. Egypt. J. Food Sci. 1975, 3, 1–5. [Google Scholar]
- Durmaz, G.; Alpaslan, M. Antioxidant properties of roasted apricot (Prunus armeniaca L.) kernels. Food Chem. 2007, 63, 1177–1181. [Google Scholar] [CrossRef]
- Haase, I.; Brüning, P.; Matissek, R.; Fisher, M. Real-Time PCR Assays for the Quantification of rDNA from Apricot and other plant species in Marzipan. J. Agric. Food Chem. 2013, 61, 3414–3418. [Google Scholar] [CrossRef] [PubMed]
- RASFF (Rapid Alert System for Food and Feed). RASFF Portal. Available online: https://webgate.ec.europa.eu/rasff-window/portal/index.cfm?event=searchForm&cleanSearch=1 (accessed on 28 December 2015).
- EFSA (European Food Safety Authority). Opinion of The Scientific Panel on Contaminants in the Food Chain on a Request from the Commission Related to the Potential Increase of Consumer Health Risk by a Possible Increase of the Existing Maximum Levels for Aflatoxins in Almonds, Hazelnuts and Pistachios Derived Products; EFSA: Parma, Italy, 2007; pp. 1–127. [Google Scholar]
- European Commission. Commission Regulation (EC) No. 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L364, 5–24. [Google Scholar]
- European Commission. Commission Regulation (EC) No. 165/2010 of 26 February 2010 amending Regulation (EC) No. 1881/2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2010, L50, 8–11. [Google Scholar]
- Codex Standard 193–1995. Codex general Standard for Contaminants and Toxins in Food and Feed. Available online: http://www.codexalimentarius.net/download/standards/17/CXS_193e.pdf (accessed on 28 December 2015).
- Whitaker, T.B. Efficiency of the blanching and electronic colour sorting process for reducing aflatoxin in raw shelled peanuts. Peanut Sci. 1997, 24, 62–66. [Google Scholar] [CrossRef]
- Schatzki, T.; Pan, J. Distribution of aflatoxins in pistachios. 3. Distribution in pistachios process streams. J. Agric. Food Chem. 1996, 44, 1076–1084. [Google Scholar] [CrossRef]
- Stainer, W.E.; Brunschweiler, K.; Leimbacher, E.; Schneider, R. Aflatoxins and fluorescence in Brazil nuts and pistachio Nut. J. Agric. Food Chem. 1992, 40, 2453–2457. [Google Scholar] [CrossRef]
- Shade, J.E.; McGreevy, K.; King, A.D., Jr.; Mackey, B.; Fuller, G. Incidence of aflatoxin in California Almonds. Appl. Microbiol. 1975, 29, 48–53. [Google Scholar]
- Dorner, J.W. Management and prevention of mycotoxins in peanuts. Food Addit. Contam. 2008, 25, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Gloria, E.M. Aflatoxin Contamination Distribution Among Grains and Nuts, Aflatoxins—Detection, Measurement and Control; Torres-Pacheco, I., Ed.; University of Sao Paulo: Sao Paulo, Brazil, 2011; pp. 75–90. [Google Scholar]
- Whitaker, T.B.; Slate, A.; Birmingham, T.; Adams, J.; Jacobs, M.; Grary, G. Correlation between aflatoxin contamination and various USDA grade categories of shelled almonds. Food Addit. Contam. 2010, 93, 943–947. [Google Scholar]
- Whitaker, T.B.; Dorner, J.V.; Lamb, M.; Slate, A.B. The effect of sorting farmers’ stock peanuts by size and colour on partitioning aflatoxin into various shelled peanut grade sizes. Peanut Sci. 2005, 32, 103–118. [Google Scholar] [CrossRef]
- Campbell, B.C.; Molyneux, R.J.; Schatzki, T.F. Current research on reducing pre- and post-harvest aflatoxin contamination of U.S. almond, pistachio, and walnut. J. Toxicol. Toxin Rev. 2003, 22, 225–266. [Google Scholar] [CrossRef]
- De Mello, F.R.; Scussel, V.M. Characteristic of in-shell Brazil nuts and their relationship to aflatoxin contamination: Criteria for sorting. J. Agric. Food Chem. 2007, 55, 9305–9310. [Google Scholar] [CrossRef] [PubMed]
- Calderari, T.O.; Iamanaka, B.T.; Frisvad, J.C.; Pitt, J.I.; Sartori, D.; Pereira, J.L.; Fungaro, M.H.P.; Taniwaki, M.H. The biodiversity of Aspergillus section Flavi in brazil nuts: From rainforest to consumers. Int. J. Food Microbiol. 2013, 160, 267–272. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EC) No. 401/2006 of 23 February 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. Eur. Union 2010, L70, 12–34. [Google Scholar]
- Shakerardekani, A.; Karim, R.; Mirdamadiha, F. The effect of sorting on aflatoxin reduction on pistachio nuts. J. Food Agric. Environ. 2012, 11, 459–461. [Google Scholar] [CrossRef]
- Cole, R.J.; Dorner, J.W.; Holbrook, C.C. Advances in mycotoxin elimination and resistance. In Advantage in Peanut Science; Pattee, H.E., Stalker, H.T., Eds.; American Peanut research Educational Society: Stillwater, OK, USA, 1995; pp. 456–474. [Google Scholar]
- Zivoli, R.; Gambacorta, L.; Perrone, G.; Solfrizzo, M. Effect of almond processing on levels and distribution of aflatoxins in finished products and by-products. J. Agric. Food Chem. 2014, 62, 5707–5715. [Google Scholar] [CrossRef] [PubMed]
- Kabak, B.; Dobson, A.D.V.; Var, I. Strategies to prevent Mycotoxin contamination of food and animal feed: A review. Crit. Rev. Food Sci. 2006, 46, 593–619. [Google Scholar] [CrossRef] [PubMed]
- Imperato, R.; Campone, L.; Piccinelli, A.L.; Veneziano, A.; Rastrelli, L. Survey of aflatoxins and ochatoxin A contamination food products imported in Italy. Food Control 2011, 22, 1905–1910. [Google Scholar] [CrossRef]
- Luttfullah, G.; Hussain, A. Studies on contamination level of aflatoxins in some dried fruits and nuts of Pakistan. Food Control 2011, 22, 426–429. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zivoli, R.; Gambacorta, L.; Piemontese, L.; Solfrizzo, M. Reduction of Aflatoxins in Apricot Kernels by Electronic and Manual Color Sorting. Toxins 2016, 8, 26. https://doi.org/10.3390/toxins8010026
Zivoli R, Gambacorta L, Piemontese L, Solfrizzo M. Reduction of Aflatoxins in Apricot Kernels by Electronic and Manual Color Sorting. Toxins. 2016; 8(1):26. https://doi.org/10.3390/toxins8010026
Chicago/Turabian StyleZivoli, Rosanna, Lucia Gambacorta, Luca Piemontese, and Michele Solfrizzo. 2016. "Reduction of Aflatoxins in Apricot Kernels by Electronic and Manual Color Sorting" Toxins 8, no. 1: 26. https://doi.org/10.3390/toxins8010026
APA StyleZivoli, R., Gambacorta, L., Piemontese, L., & Solfrizzo, M. (2016). Reduction of Aflatoxins in Apricot Kernels by Electronic and Manual Color Sorting. Toxins, 8(1), 26. https://doi.org/10.3390/toxins8010026







