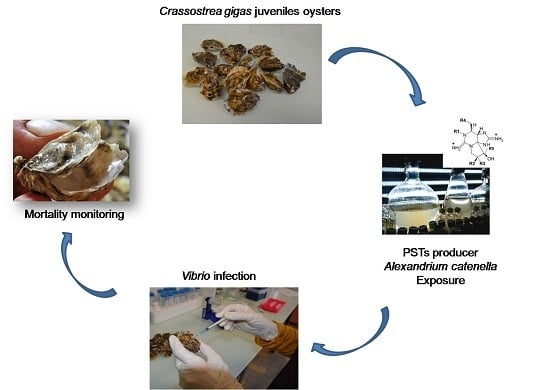
Exposure to the Paralytic Shellfish Toxin Producer Alexandrium catenella Increases the Susceptibility of the Oyster Crassostrea gigas to Pathogenic Vibrios
Abstract
:1. Introduction
2. Results
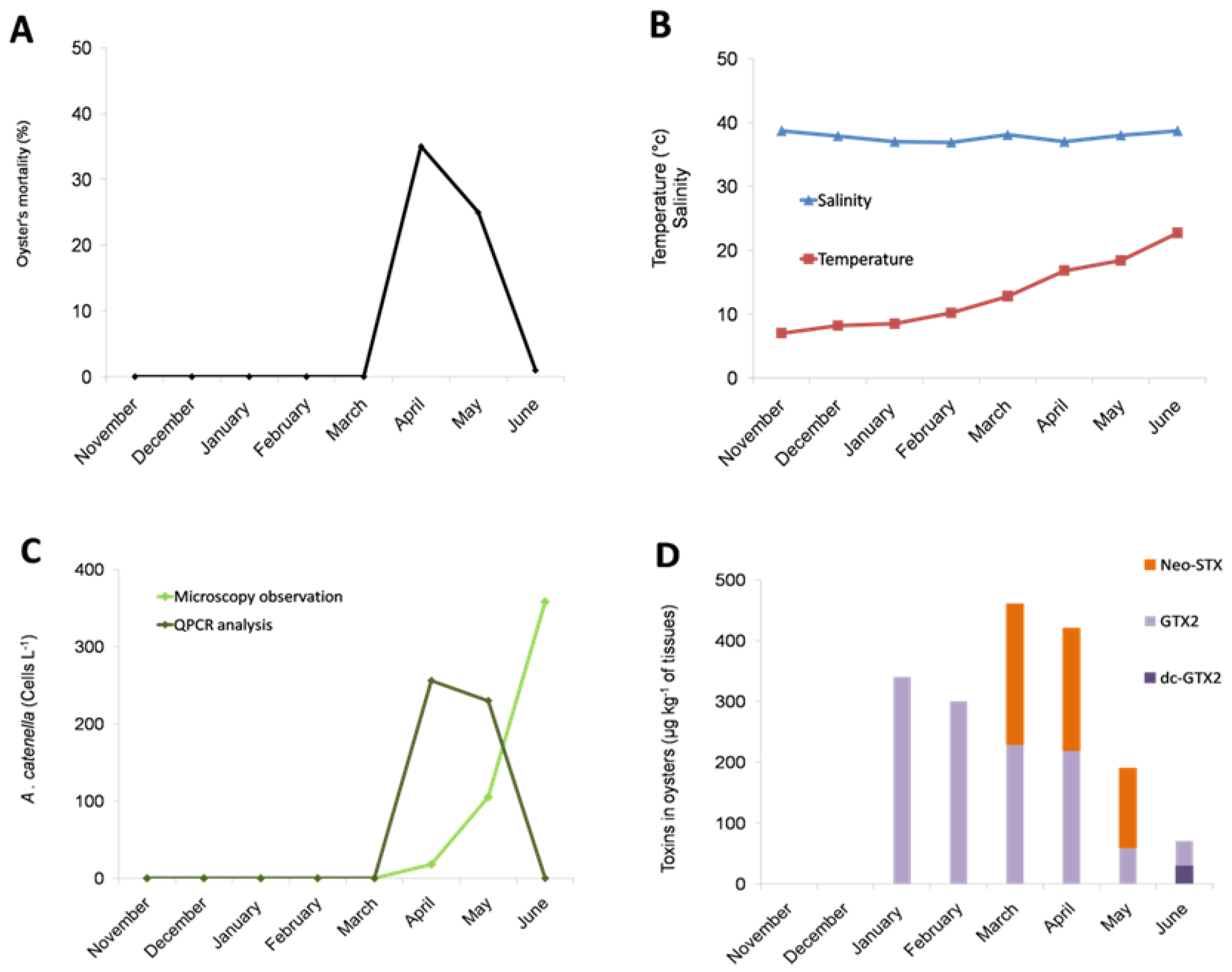
2.1. Occurrence of an Oyster Mortality Event during a Toxic Alexandrium catenella in the Thau Lagoon
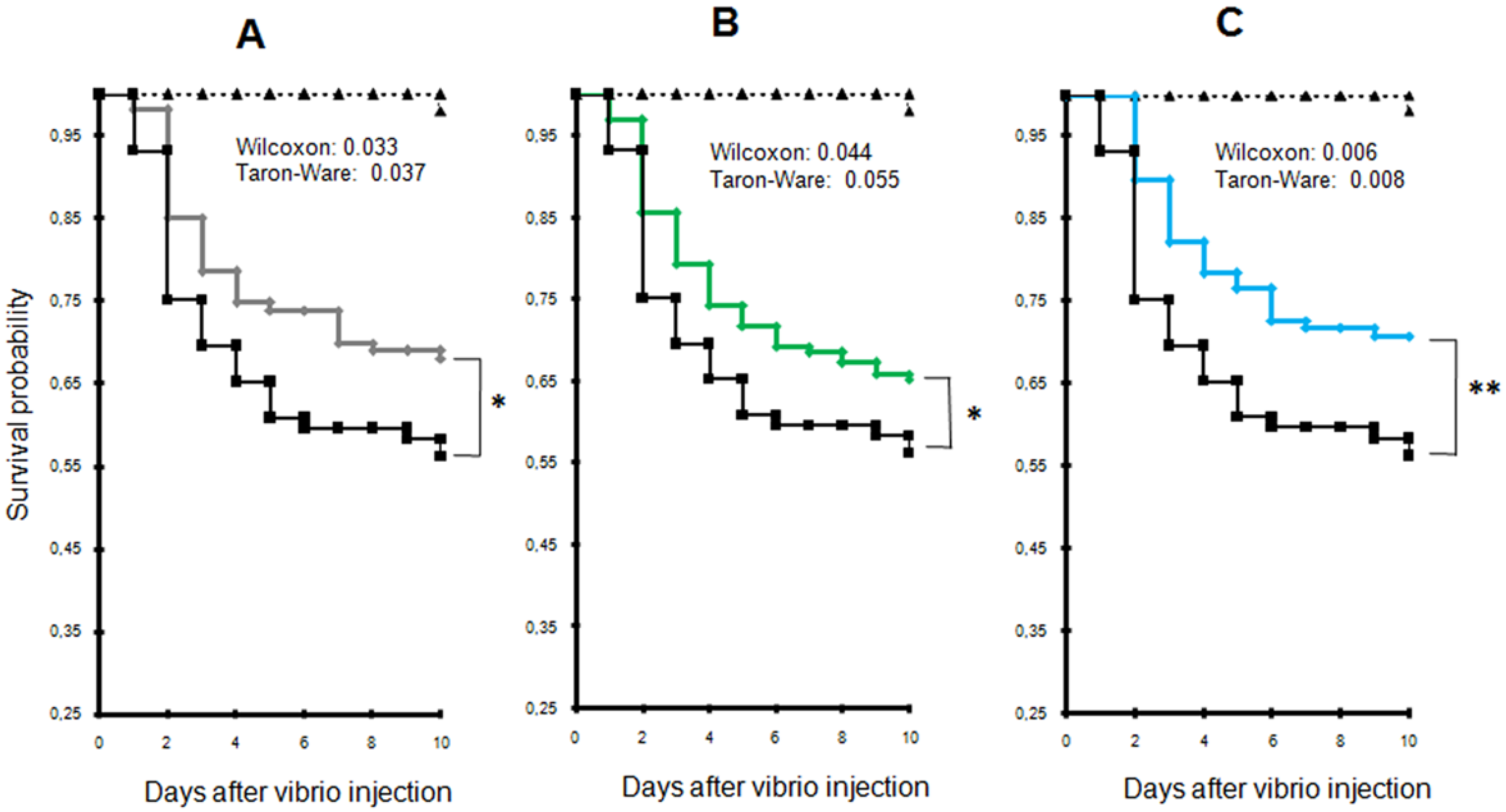
2.2. Exposure to Alexandrium catenella Increases Oyster Mortality
2.3. PSTs Accumulate in Infected Oysters
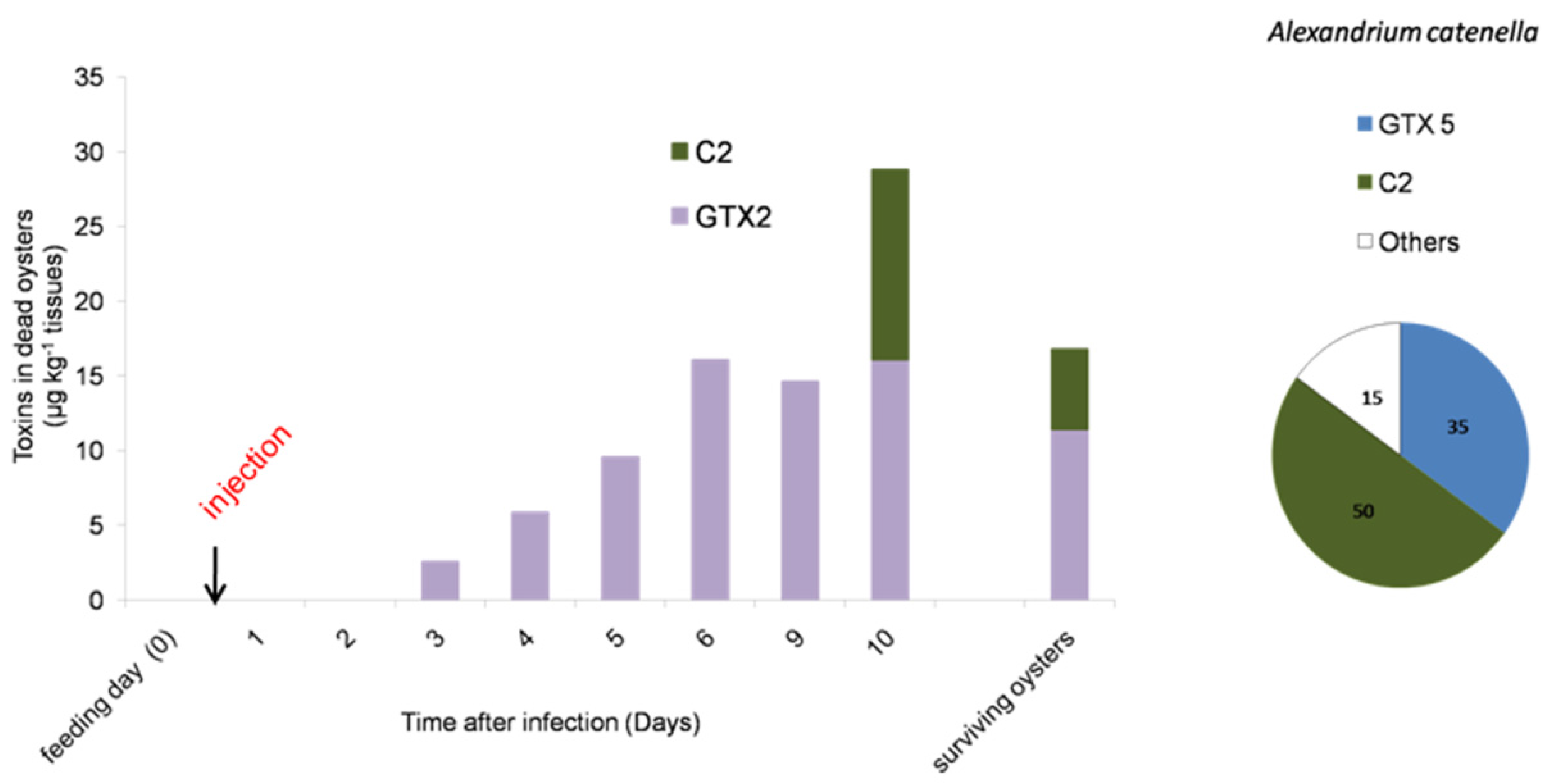
3. Discussion
4. Experimental Section
4.1. Field Survey in Thau Lagoon
4.1.1. Collection of Environmental Samples and Processing
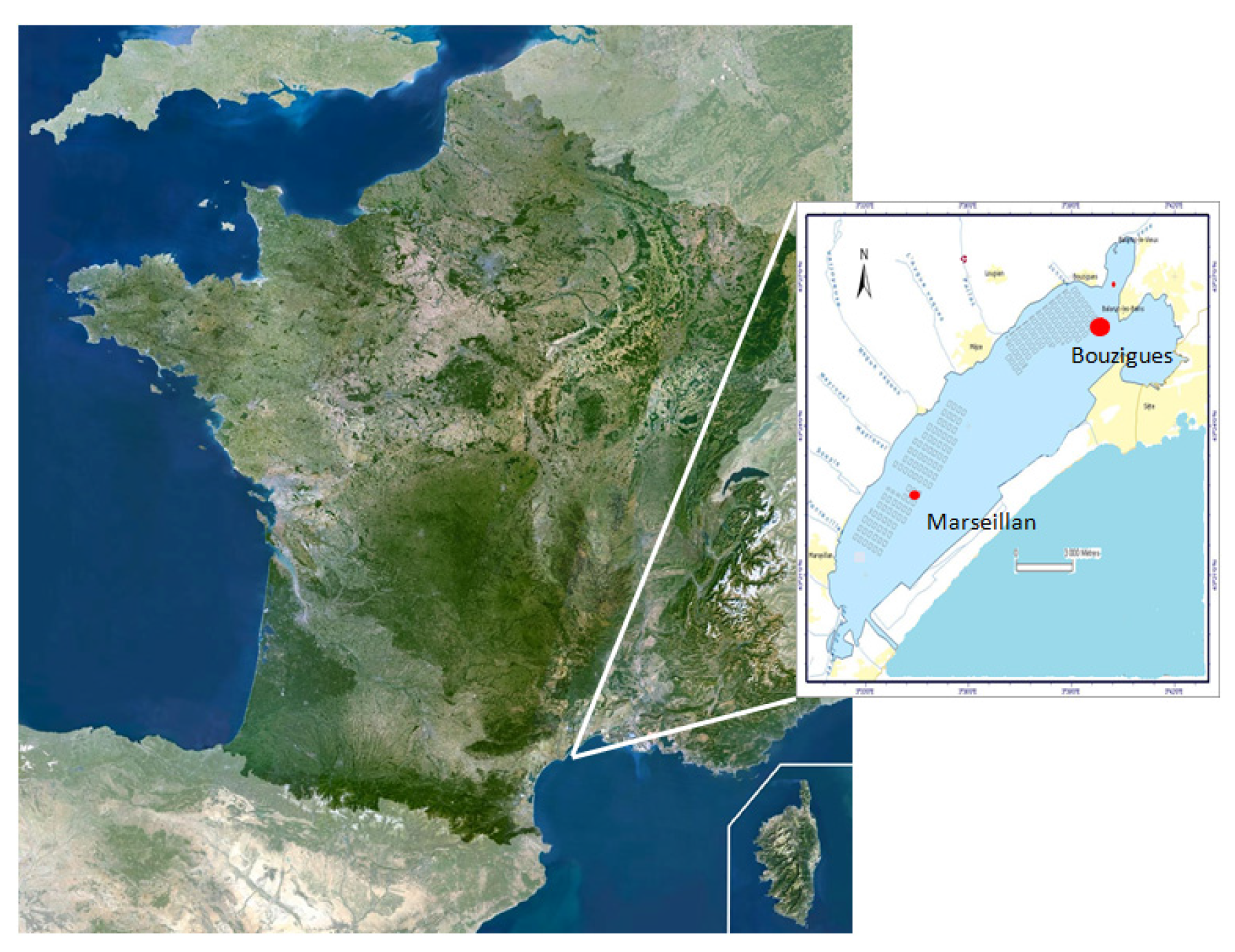
4.1.2. Dynamics of Alexandrium catenella
4.1.3. Oyster Mortality
4.1.4. Oyster Biotoxin Contamination
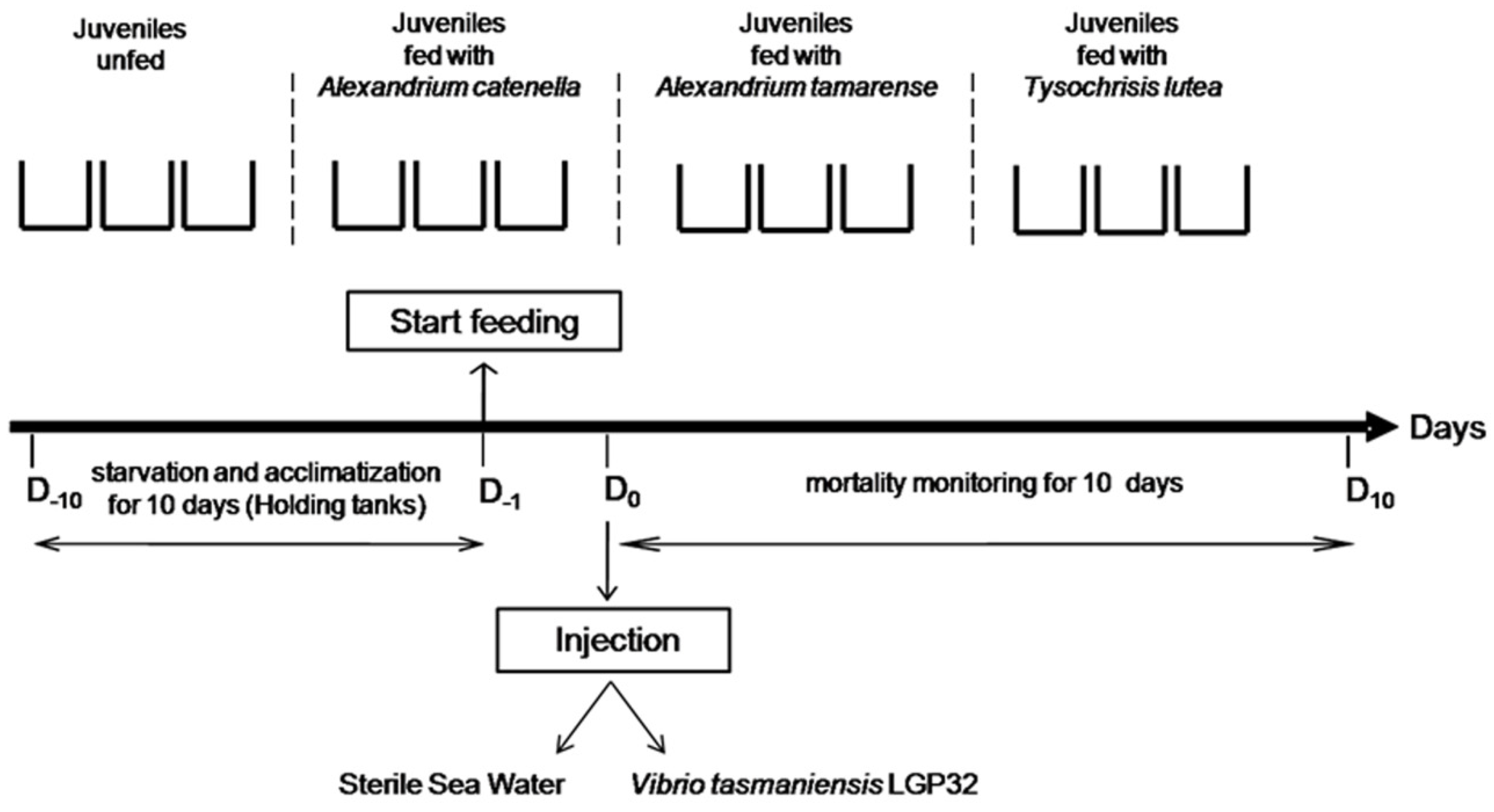
4.2. Experimental Infections
4.2.1. Oysters
4.2.2. Algae Production
4.2.3. Pathogenic Bacterial Strain
4.2.4. Experimental Design
4.3. Neurotoxins Analysis
4.4. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sabry, R.C.; da Silva, P.M.; Gesteira, T.C.V.; Pontinha, V.D.; Magalhaes, A.R.M. Pathological study of oysters Crassostrea gigas from culture and C. rhizophorae from natural stock of Santa Catarina Island, SC, Brazil. Aquaculture 2011, 320, 43–50. [Google Scholar] [CrossRef]
- Burge, C.A.; Judah, L.R.; Conquest, L.L.; Griffin, F.J.; Cheney, D.P.; Suhrbier, A.; Vadopalas, B.; Olin, P.G.; Renault, T.; Friedman, C.S. Summer seed mortality of the Pacific oyster, Crassostrea gigas Thunberg grown in Tomales Bay, California, USA: The influence of oyster stock, planting time, pathogens, and environmental stressors. J. Shellfish Res. 2007, 26, 163–172. [Google Scholar] [CrossRef]
- Nicolas, J.L.; Ansquer, D.; Cochard, J.C. Isolation and characterization of a pathogenic bacterium specific to Manila clam Tapes-philippinarum larvae. Dis. Aquat. Org. 1992, 14, 153–159. [Google Scholar] [CrossRef]
- Jenkins, C.; Hick, P.; Gabor, M.; Spiers, Z.; Fell, S.A.; Gu, X.N.; Read, A.; Go, J.; Dove, M.; O’Connor, W.; et al. Identification and characterisation of an ostreid herpes virus-1 microvariant (OsHV-1 mu-var) in Crassostrea gigas (Pacific oysters) in Australia. Dis. Aquat. Org. 2013, 105, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Keeling, S.E.; Brosnahan, C.L.; Williams, R.; Gias, E.; Hannah, M.; Bueno, R.; McDonald, W.L.; Johnston, C. New Zealand juvenile oyster mortality associated with ostreid herpesvirus 1—An opportunistic longitudinal study. Dis. Aquat. Org. 2014, 109, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Domeneghetti, S.; Varotto, L.; Civettini, M.; Rosani, U.; Stauder, M.; Pretto, T.; Pezzati, E.; Arcangeli, G.; Turolla, E.; Pallavicini, A.; et al. Mortality occurrence and pathogen detection in Crassostrea gigas and Mytilus galloprovincialis close-growing in shallow waters (Goro lagoon, Italy). Fish Shellfish Immun. 2014, 41, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Soletchnik, P.; le Moine, O.; Faury, N.; Razet, D.; Geairon, P.; Goulletquer, P. Summer mortality of the oyster in the Bay Marennes-Oleron: Spatial variability of environment and biology using a geographical information system (GIS). Aquat. Living Resour. 1999, 12, 131–143. [Google Scholar] [CrossRef]
- Berthelin, C.; Kellner, K.; Mathieu, M. Storage metabolism in the Pacific oyster (Crassostrea gigas) in relation to summer mortalities and reproductive cycle (west coast of France). Comp. Biochem. Physiol. 2000, 125, 359–369. [Google Scholar] [CrossRef]
- Garnier, M.; Labreuche, Y.; Garcia, C.; Robert, M.; Nicolas, J.L. Evidence for the involvement of pathogenic bacteria in summer mortalities of the Pacific oyster Crassostrea gigas. Microb. Ecol. 2007, 53, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Gay, M.; Renault, T.; Pons, A.M.; le Roux, F. Two vibrio splendidus related strains collaborate to kill Crassostrea gigas: Taxonomy and host alterations. Dis. Aquat. Org. 2004, 62, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Lacoste, A.; Jalabert, F.; Malham, S.; Cueff, A.; Gelebart, F.; Cordevant, C.; Lange, M.; Poulet, S.A. A Vibrio splendidus strain is associated with summer mortality of juvenile oysters Crassostrea gigas in the Bay of Morlaix (North Brittany, France). Dis. Aquat. Org. 2001, 46, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Le Roux, F.; Binesse, J.; Saulnier, D.; Mazel, D. Construction of a Vibrio splendidus mutant lacking the metalloprotease gene vsm by use of a novel counterselectable suicide vector. Appl. Environ. Microbiol. 2007, 73, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Lemire, A.; Goudenege, D.; Versigny, T.; Petton, B.; Calteau, A.; Labreuche, Y.; le Roux, F. Populations, not clones, are the unit of vibrio pathogenesis in naturally infected oysters. ISME J. 2015, 9, 1523–1531. [Google Scholar] [CrossRef] [PubMed]
- Duperthuy, M.; Binesse, J.; le Roux, F.; Romestand, B.; Caro, A.; Got, P.; Givaudan, A.; Mazel, D.; Bachere, E.; Destoumieux-Garzon, D. The major outer membrane protein OmpU of Vibrio splendidus contributes to host antimicrobial peptide resistance and is required for virulence in the oyster Crassostrea gigas. Environ. Microbiol. 2010, 12, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Duperthuy, M.; Schmitt, P.; Garzon, E.; Caro, A.; Rosa, R.D.; le Roux, F.; Lautredou-Audouy, N.; Got, P.; Romestand, B.; de Lorgeril, J.; et al. Use of OmpU porins for attachment and invasion of Crassostrea gigas immune cells by the oyster pathogen Vibrio splendidus. Proc. Natl. Acad. Sci. USA 2011, 108, 2993–2998. [Google Scholar] [CrossRef] [PubMed]
- Vanhove, A.S.; Duperthuy, M.; Charriere, G.M.; le Roux, F.; Goudenege, D.; Gourbal, B.; Kieffer-Jaquinod, S.; Coute, Y.; Wai, S.N.; Destoumieux-Garzon, D. Outer membrane vesicles are vehicles for the delivery of Vibrio tasmaniensis virulence factors to oyster immune cells. Environ. Microbiol. 2015, 17, 1152–1165. [Google Scholar] [CrossRef] [PubMed]
- Segarra, A.; Pepin, J.F.; Arzul, I.; Morga, B.; Faury, N.; Renault, T. Detection and description of a particular Ostreid herpesvirus 1 genotype associated with massive mortality outbreaks of Pacific oysters, Crassostrea gigas, in France in 2008. Virus Res. 2010, 153, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Martenot, C.; Oden, E.; Travaille, E.; Malas, J.P.; Houssin, M. Detection of different variants of Ostreid Herpesvirus 1 in the Pacific oyster, Crassostrea gigas between 2008 and 2010. Virus Res. 2011, 160, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Thebault, A.; Degremont, L.; Arzul, I.; Miossec, L.; Robert, M.; Chollet, B.; Francois, C.; Joly, J.P.; Ferrand, S.; et al. Ostreid herpes virus1 detection and relationship with Crassostrea gigas spat mortality in France between 1998 and 2006. Vet. Res. 2011, 42, 416–417. [Google Scholar] [CrossRef] [PubMed]
- Samain, J.F. Review and perspectives of physiological mechanisms underlying genetically-based resistance of the Pacific oyster Crassostrea gigas to summer mortality. Aquat. Living Resour. 2011, 24, 227–236. [Google Scholar] [CrossRef]
- Jolivel, A.; Elodie, F. Statistical Analysisof Oystermortalitydata Acquired by the National Observatory shellfish. Available online: http://archimer.ifremer.fr/doc/00130/24095/ (accessed on 3 December 2015).
- Pernet, F.; Barret, J.; le Gall, P.; Corporeau, C.; Degremont, L.; Lagarde, F.; Pepin, J.F.; Keck, N. Mass mortalities of Pacific oysters Crassostrea gigas reflect infectious diseases and vary with farming practices in the Mediterranean Thau lagoon, France. Aquac. Environ. Interact. 2012, 2, 215–237. [Google Scholar] [CrossRef]
- Pernet, F.; Lagarde, F.; Jeannee, N.; Daigle, G.; Barret, J.; le Gall, P.; Quere, C.; D’orbcastel, E.R. Spatial and Temporal Dynamics of Mass Mortalities in Oysters Is Influenced by Energetic Reserves and Food Quality. PLoS ONE 2014. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M.; Alpermann, T.J.; Cembella, A.D.; Collos, Y.; Masseret, E.; Montresor, M. The globally distributed genus Alexandrium: Multifaceted roles in marine ecosystems and impacts on human health. Harmful Algae 2012, 14, 10–35. [Google Scholar] [CrossRef] [PubMed]
- Scholin, C.A.; Herzog, M.; Sogin, M.; Anderson, D.M. Identification of group- and strain-specific genetic markers for globally distributed Alexandrium (Diniphyceae). Sequence analysis of LSU rRNA gene. J. Phycol. 1994, 30, 744–754. [Google Scholar] [CrossRef]
- Cόrdova, J.L.; Müller, I. Use of PCR and partial sequencing of the large-subunit rRNA gene to indentify Alexandrium catenella (Dinophycea) from the South of Chile. Harmful Algae 2002, 1, 343–350. [Google Scholar] [CrossRef]
- MacKenzie, L.; de Salas, M.; Adamson, J.; Beuzenberg, V. The dinoflagellate genus Alexandrium (Halim) in New Zealand coastal waters: Comparative morphology, toxicity and molecular genetics. Harmful Algae 2004, 3, 71–92. [Google Scholar] [CrossRef]
- Adachi, M.; Sako, Y.; Ishida, Y. Analyses of Alexandrium (Dinophyceae) species using sequences of the 5.8S ribosomal DNA and internal transcribed spacer regions. J. Phycol. 1996, 32, 424–432. [Google Scholar] [CrossRef]
- Yeung, P.K.K.; Wong, F.T.W.; Wong, J.T.Y. Large subunit rDNA sequences from Alexandrium catenevilalla strains isolated during algal blooms in Hong Kong. J. Appl. Phycol. 2002, 14, 147–150. [Google Scholar] [CrossRef]
- Vila, M.; Garces, E.; Maso, M.; Camp, J. Is the distribution of the toxic dinoflagellates Alexandrium catenella expanding along the NW Mediterranean coast? Mar. Ecol. Prog. Ser. 2001, 23, 497–514. [Google Scholar] [CrossRef]
- Penna, A.; Garcés, E.; Vila, M.; Giacobbe, M.G.; Fraga, S.; Luglie, A.; Bravo, I.; Bertozzini, E.; Vernesi, C. Alexandrium catenella (Dinophycea), a toxic ribotype expanding in the NW Mediterranean sea. Mar. Biol. 2005, 148, 13–23. [Google Scholar] [CrossRef]
- Garces, E.; Maso, M.; Camp, J.A. Recurrent and localized dinoflagellate bloom in a Mediterranean beach. J. Plankton Res. 1999, 21, 2373–2391. [Google Scholar] [CrossRef]
- Bravo, I.; Vila, M.; Maso, M.; Figueroa, R.I.; Ramilo, I. Alexandrium catenella and Alexandrium minutum blooms in the Mediterranean Sea: Toward the identification of ecological niches. Harmful Algae 2008, 7, 515–522. [Google Scholar] [CrossRef]
- Abadie, E.; Amzil, Z.; Belin, C.; Comps, M.A.; Elzière-Papayanni, P.; Lassus, P.; le Bec, C.; Baut, C.M.-L.; Nezan, E.; Poggi, R. Contamination de l’étang de Thau par Alexandrium tamarense. Available online: http://www.ifremer.fr/docelec/doc/1999/rapport-884.pdf (accessed on 3 December 2015).
- Lilly, E.L.; Kulis, D.M.; Gentien, P.; Anderson, D.M. Paralytic shellfish poisoning toxins in France linked to a human-introduced strain of Alexandrium catenella from the western Pacific: Evidence from DNA and toxin analysis. J. Plankton Res. 2002, 24, 443–452. [Google Scholar] [CrossRef]
- Genovesi, B.; Shin-Grzebyk, M.S.; Grzebyk, D.; Laabir, M.; Gagnaire, P.A.; Vaquer, A.; Pastoureaud, A.; Lasserre, B.; Collos, Y.; Berrebi, P.; et al. Assessment of cryptic species diversity within blooms and cyst bank of the Alexandrium tamarense complex (Dinophyceae) in a Mediterranean lagoon facilitated by semi-multiplex PCR. J. Plankton Res. 2011, 33, 405–414. [Google Scholar] [CrossRef]
- Laabir, M.; Amzil, Z.; Lassus, P.; Masseret, E.; Tapilatu, Y.; de Vargas, R.; Grzebyk, D. Viability, growth and toxicity of Alexandrium catenella and Alexandrium minutum (Dinophyceae) following ingestion and gut passage in the oyster Crassostrea gigas. Aquat. Living Resour. 2007, 20, 51–57. [Google Scholar] [CrossRef]
- Laabir, M.; Jauzein, C.; Genovesi, B.; Masseret, E.; Grzebyk, D.; Cecchi, P.; Vaquer, A.; Perrin, Y.; Colos, Y. Influence of temperature, salinity and irradiance on the growth and cell yield of the harmful red tide dinoflagellate Alexandrium catenella colonizing Mediterranean waters. J. Plankton Res. 2011, 33, 1550–1563. [Google Scholar] [CrossRef]
- Masselin, P.; Amzil, Z.; Abadie, E.; Nézan, E.; le Bec, C.; Chiantella, C.; Truquet, P. Paralytic shellfish poisoning on the French Mediterranean coast in the autumn 1998: Alexandrium tamarense complex (Dinophyceae) as causative agent. In Harmful Algae Blooms; Hallegraeff, G.M., Blackburn, S.I., Bolch, C.J., Lewis, R.J., Eds.; JOC-UNESCO: Paris, France, 2000; pp. 407–441. [Google Scholar]
- Twarog, B.M.; Hidaka, T.; Yamaguchi, H. Resistance to totrodoxin and saxitoxin in nerves of bivalves mollusks: A possible correlation with paralytic shellfish poisoning. Toxicon 1972, 10, 273–278. [Google Scholar] [CrossRef]
- Shumway, S.E.; Cucci, T.L. The effects of the toxic dinoflagellate Protogonyaulax tamarensis on the feeding and behavior of bivalve molluscs. Aquat. Toxicol. 1987, 10, 9–27. [Google Scholar] [CrossRef]
- Shumway, S.E. A review of the effects of algal blooms on shellfish and aquaculture. J. World. Aquac. Soc. 1990, 21, 65–104. [Google Scholar] [CrossRef]
- Bricelj, V.M.; Connell, L.; Konoki, K.; MacQuarrie, S.P.; Scheuer, T.; Catterall, W.A.; Trainer, V.L. Sodium channel mutation leading to saxitoxin resistance in clams increases risk of PSP. Nature 2005, 434, 763–767. [Google Scholar] [CrossRef] [PubMed]
- Bardouil, M.; Bohec, M.; Cormerais, M.; Bougrier, S.; Lassus, P. Experimental study of the effects of a toxic microalgal diet on feeding of the oyster Crassostrea gigas Thunberg. J. Shellfish Res. 1993, 1, 417–422. [Google Scholar]
- Wildish, D.; Lassus, P.; Martin, J.; Saulnier, A.; Bardouil, M. Effect of the PSP-causing dinoflagellate, Alexandrium sp. on the initial feeding response of Crassostrea gigas. Aquat. Living Resour. 1998, 11, 35–43. [Google Scholar] [CrossRef]
- Lassus, P.; Baron, R.; Garen, P.; Truquet, P.; Masselin, P.; Bardouil, M.; Leguay, D.; Amzil, Z. Paralytic shellfish poison outbreaks in the Penze estuary: Environmental factors affecting toxin uptake in the oyster, Crassostrea gigas. Aquat. Living Resour. 2004, 17, 207–214. [Google Scholar] [CrossRef]
- Rolland, J.L.; Pelletier, K.; Masseret, E.; Rieuvilleneuve, F.; Savar, V.; Santini, A.; Amzil, Z.; Laabir, M. Paralytic toxins accumulation and tissue Expression of α-amylase and lipase genes in the pacific oyster Crassostrea gigas fed with the neurotoxic dinoflagellate Alexandrium catenella. Mar. Drugs 2012, 10, 2519–2534. [Google Scholar] [CrossRef] [PubMed]
- Gueguen, M.; Bardouil, M.; Baron, R.; Lassus, P.; Truquet, P.; Massardier, J.; Amzil, Z. Detoxification of Pacific oyster Crassostrea gigas fed on diets of Skeletonema costatum with and without silt, following PSP contamination by Alexandrium minutum. Aquat. Living Resour. 2008, 21, 13–20. [Google Scholar] [CrossRef]
- Medhioub, W.; Lassus, P.; Truquet, P.; Bardouil, M.; Amzil, Z.; Séchet, V.; Sibat, M.; Soudant, P. Physiological responses of Crassostrea gigas when exposed to the toxic dinoflagellates Alexandrium ostenfeldii: Toxin uptake and detoxification. Aquaculture 2012, 358, 108–115. [Google Scholar] [CrossRef]
- Hegaret, H.; Wikfors, G.H.; Soudant, P.; Lambert, C.; Shumway, S.E.; Berard, J.B.; Lassus, P. Toxic dinoflagellates (Alexandrium fundyense and A. catenella) have minimal apparent effect on oyster hemocytes. Mar. Biol. 2007, 152, 441–447. [Google Scholar] [CrossRef]
- Hegaret, H.; da Silva, P.M.; Wikfors, G.H.; Haberkorn, H.; Shumway, S.E.; Soudant, P. In vitro interactions between several species of harmful algae and haemocytes of bivalve mollusks. Cell Biol. Toxicol. 2011, 27, 249–266. [Google Scholar] [CrossRef] [PubMed]
- Mello, D.F.; da Silva, P.M.; Barracco, M.A.; Soudant, P.; Hegaret, H. Effects of the dinoflagellates Alexandrium minutum and its toxin (saxitoxin) on the functional activity and gene expression of Crassostrea gigas hemocytes. Harmful Algae 2013, 26, 45–51. [Google Scholar] [CrossRef]
- Medhioub, W.; Ramondenc, S.; Vanhove, A.; Vergnes, A.; Masseret, E.; Savar, V.; Amzil, Z.; Laabir, M.; Rolland, J.L. Exposure to the Neurotoxic Dinoflagellate Alexandrium catenella induces apoptosis of the oyster Crassostrea gigas hemocytes. Mar. Drugs 2013, 11, 4799–4814. [Google Scholar] [CrossRef] [PubMed]
- Lassudrie, M.; Soudant, P.; Nicolas, J.L.; Fabioux, C.; Lambert, C.; Miner, P.; le Grand, J.; Petton, B.; Hegaret, H. Interaction between toxic dinoflagellate Alexandrium catenella exposure and disease associated with herpesvirus OsHV-1 mu Var in Pacific oyster spat Crassostrea gigas. Harmful Algae 2015, 45, 53–61. [Google Scholar] [CrossRef]
- Bachere, E.; Rosa, R.D.; Schmitt, P.; Poirier, A.C.; Merou, N.; Charriere, G.M.; Destoumieux-Garzon, D. The new insights into the oyster antimicrobial defense: Cellular, molecular and genetic view. Fish Shellfish Immunol. 2015, 46, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, L.; Guo, X.; Litman, G.W.; Dishaw, L.J.; Zhang, G. Massive expansion and functional divergence of innate immune genes in a protostome. Sci. Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Fistarol, G.O.; Legrand, C.; Selander, E.; Hummert, C.; Stolte, W.; Graneli, E. Allelopathy in Alexandrium spp.: Effect on a natural plankton community and on algal monocultures. Aquat. Microb. Ecol. 2004, 35, 45–56. [Google Scholar] [CrossRef]
- Oshima, Y. Chemical and enzymatic transformation of paralytic shellfish toxins in marine organisms. In Harmful Marine Algal Blooms; Lassus, P., Arzul, G., Erard, E., Gentien, P., Marcaillou, C., Eds.; Lavoisier/Intercept: Paris, France, 1995; pp. 475–480. [Google Scholar]
- Suzuki, T.; Ichimi, K.; Oshima, Y.; Kamiyama, T. Paralytic shellfish poisoning (PSP) toxin profiles and short-term detoxification kinetics in mussels Mytilus galloprovincialis fed with the toxic dinoflagellate Alexandrium tamarense. Harmful Algae 2003, 2, 201–206. [Google Scholar] [CrossRef]
- Bougrier, S.; Lassus, P.; Bardouil, M.; Masselin, P.; Truquet, P. Paralytic shellfish poison accumulation yields and feeding time activity in the Pacific oyster (Crassostrea gigas) and king scallop (Pecten maximus). Aquat. Living Resour. 2003, 16, 347–352. [Google Scholar] [CrossRef]
- Cembella, A.D.; Shumway, S.E.; Larocque, R. Sequestering and putative biotransformation of paralytic shellfish toxins by the sea scallop Placopecten magellanicus: Seasonal and special scales in natural populations. J. Exp. Mar. Biol. Ecol. 2005, 180, 1–22. [Google Scholar] [CrossRef]
- Rolland, J.L.; Medhioub, W.; Vergnes, A.; Abi-khalil, C.; Savar, V.; Abadie, E.; Masseret, E.; Amzil, Z.; Laabir, M. A feedback mechanism to control apoptosis occurs in the digestive gland of the oyster Crassostrea gigas exposed to the paralytic shellfish toxins producer Alexandrium catenella. Mar. Drugs 2014, 12, 5035–5054. [Google Scholar] [CrossRef] [PubMed]
- Petton, B.; Bruto, M.; James, A.; Labreuche, Y.; Alunno-Bruscia, M.; le Roux, F. Crassostrea gigas mortality in France: The usual suspect, a herpes virus, may not be the killer in this polymicrobial opportunistic disease. Front. Microbiol. 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ausubel, F.M.; Brent, R.; Kingston, R.E.; Moore, D.D.; Seidman, J.G.; Smith, J.A.; Struhl, K. Preparation of genomic DNA from mammalian tissue. In Current Protocol in Molecular Biology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2003; Volume 1, pp. 2.2.1–2.2.3. [Google Scholar]
- John, U.; Fensome, R.A.; Medlin, L.K. The application of a molecular clock based on molecular sequences and the fossil record to explain biogeographic distributions within the Alexandrium tamarense “species complex” (Dinophyceae). Mol. Biolog. Evol. 2003, 20, 1015–1027. [Google Scholar] [CrossRef] [PubMed]
- John, U.; Litaker, W.; Montresor, M.; Murray, S.; Brosnahan, M.L.; Anderson, D.M. Proposal to reject the name Gonyaulux catenella (Alexandrium catenella) (Dinophyceae). Taxon 2014, 63, 932–933. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhuang, Y.; Zhang, H.; Lin, X.; Lin, S. DNA barcoding species in Alexandrium tamarense complex using ITS and proposing designation of five species. Harmful Algae 2014, 31, 100–113. [Google Scholar] [CrossRef]
- Lilly, E.L.; Halanych, K.M.; Anderson, D.M. Species boundaries and global biogeography of the Alexandrium tamarense complex (Dinophyceae). J. Phycol. 2007, 43, 1329–1338. [Google Scholar] [CrossRef]
- Shellfish Observatory Network (RESCO). Ifremer, 155 rue Jean-Jacques Rousseau, 92138, Issy-les-Moulineaux, France. Available online: http://wwz.ifremer.fr/observatoire_conchylicole (accessed on 3 December 2015).
- Harrison, P.J.; Waters, R.E.; Taylor, F.J.R. A broad-spectrum artificial seawater medium for coastal and open ocean phytoplankton. J. Phycol. 1980, 16, 28–35. [Google Scholar] [CrossRef]
- Van de Riet, J.; Gibbs, R.S.; Muggah, P.M.; Rourke, W.A.; MacNeil, J.D.; Quilliam, M.A. Liquid Chromatography Post-Column Oxidation (PCOX) Method for the Determination of Paralytic Shellfish Toxins in Mussels, Clams, Oysters, and Scallops: Collaborative Study. J. AOAC Int. 2011, 94, 1154–1176. [Google Scholar] [PubMed]
- Kaplan, E.L.; Meier, P. Non parametric estimation for incomplete observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abi-Khalil, C.; Lopez-Joven, C.; Abadie, E.; Savar, V.; Amzil, Z.; Laabir, M.; Rolland, J.-L. Exposure to the Paralytic Shellfish Toxin Producer Alexandrium catenella Increases the Susceptibility of the Oyster Crassostrea gigas to Pathogenic Vibrios. Toxins 2016, 8, 24. https://doi.org/10.3390/toxins8010024
Abi-Khalil C, Lopez-Joven C, Abadie E, Savar V, Amzil Z, Laabir M, Rolland J-L. Exposure to the Paralytic Shellfish Toxin Producer Alexandrium catenella Increases the Susceptibility of the Oyster Crassostrea gigas to Pathogenic Vibrios. Toxins. 2016; 8(1):24. https://doi.org/10.3390/toxins8010024
Chicago/Turabian StyleAbi-Khalil, Celina, Carmen Lopez-Joven, Eric Abadie, Veronique Savar, Zouher Amzil, Mohamed Laabir, and Jean-Luc Rolland. 2016. "Exposure to the Paralytic Shellfish Toxin Producer Alexandrium catenella Increases the Susceptibility of the Oyster Crassostrea gigas to Pathogenic Vibrios" Toxins 8, no. 1: 24. https://doi.org/10.3390/toxins8010024
APA StyleAbi-Khalil, C., Lopez-Joven, C., Abadie, E., Savar, V., Amzil, Z., Laabir, M., & Rolland, J.-L. (2016). Exposure to the Paralytic Shellfish Toxin Producer Alexandrium catenella Increases the Susceptibility of the Oyster Crassostrea gigas to Pathogenic Vibrios. Toxins, 8(1), 24. https://doi.org/10.3390/toxins8010024