Topographic Relationship between the Supratrochlear Nerve and Corrugator Supercilii Muscle—Can This Anatomical Knowledge Improve the Response to Botulinum Toxin Injections in Chronic Migraine?
Abstract
:1. Introduction
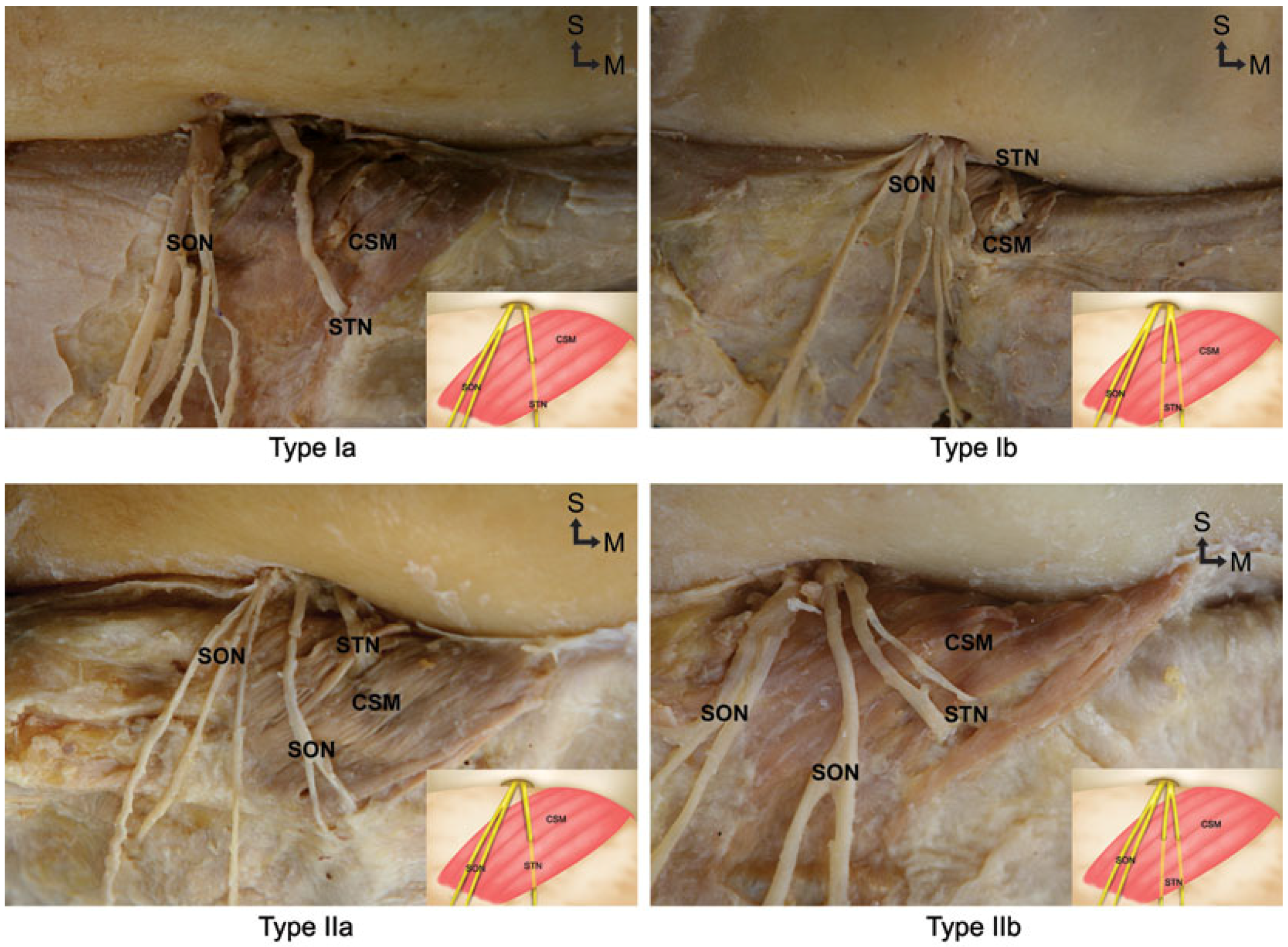
2. Results
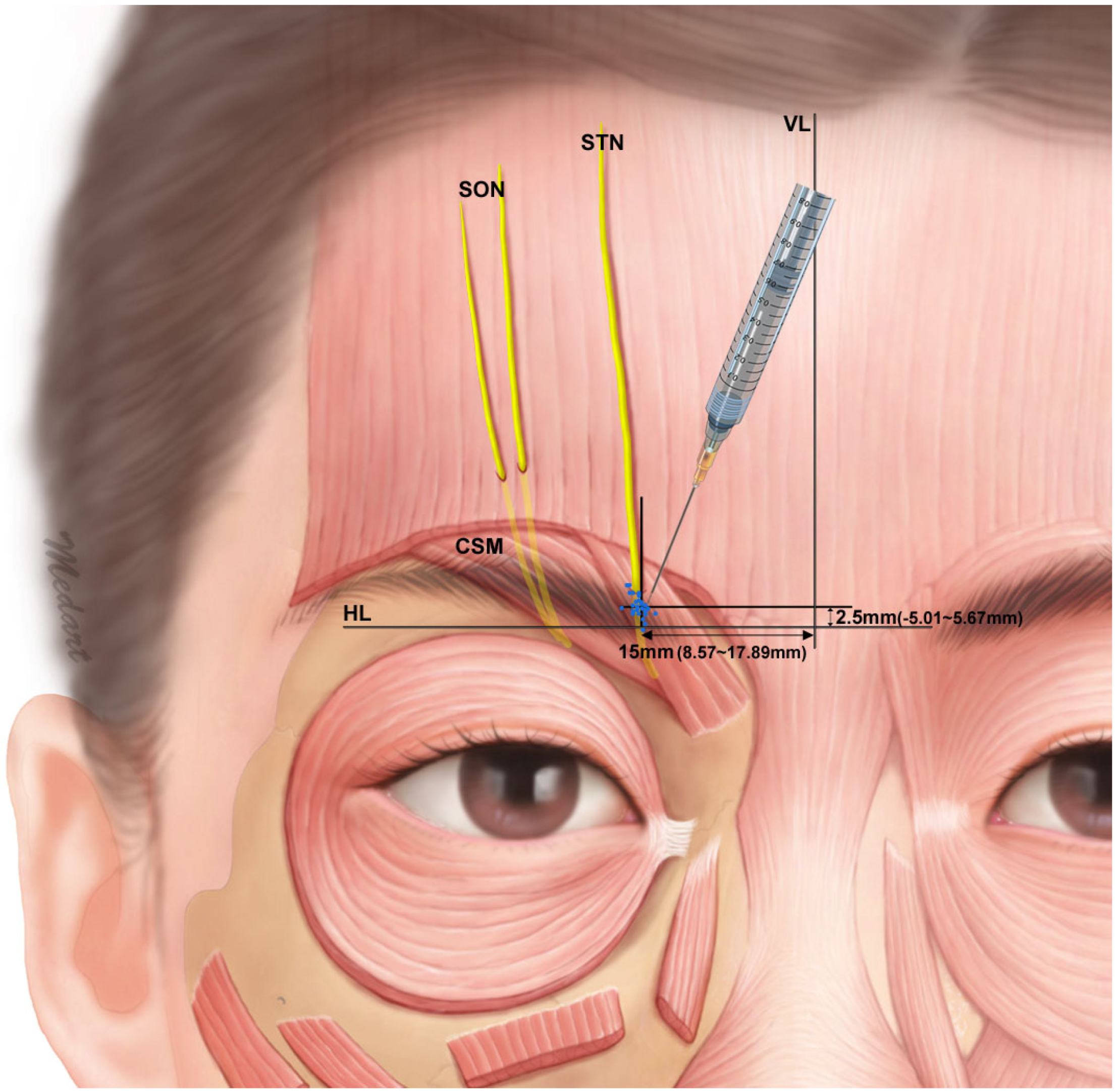
3. Discussion
4. Materials and Methods
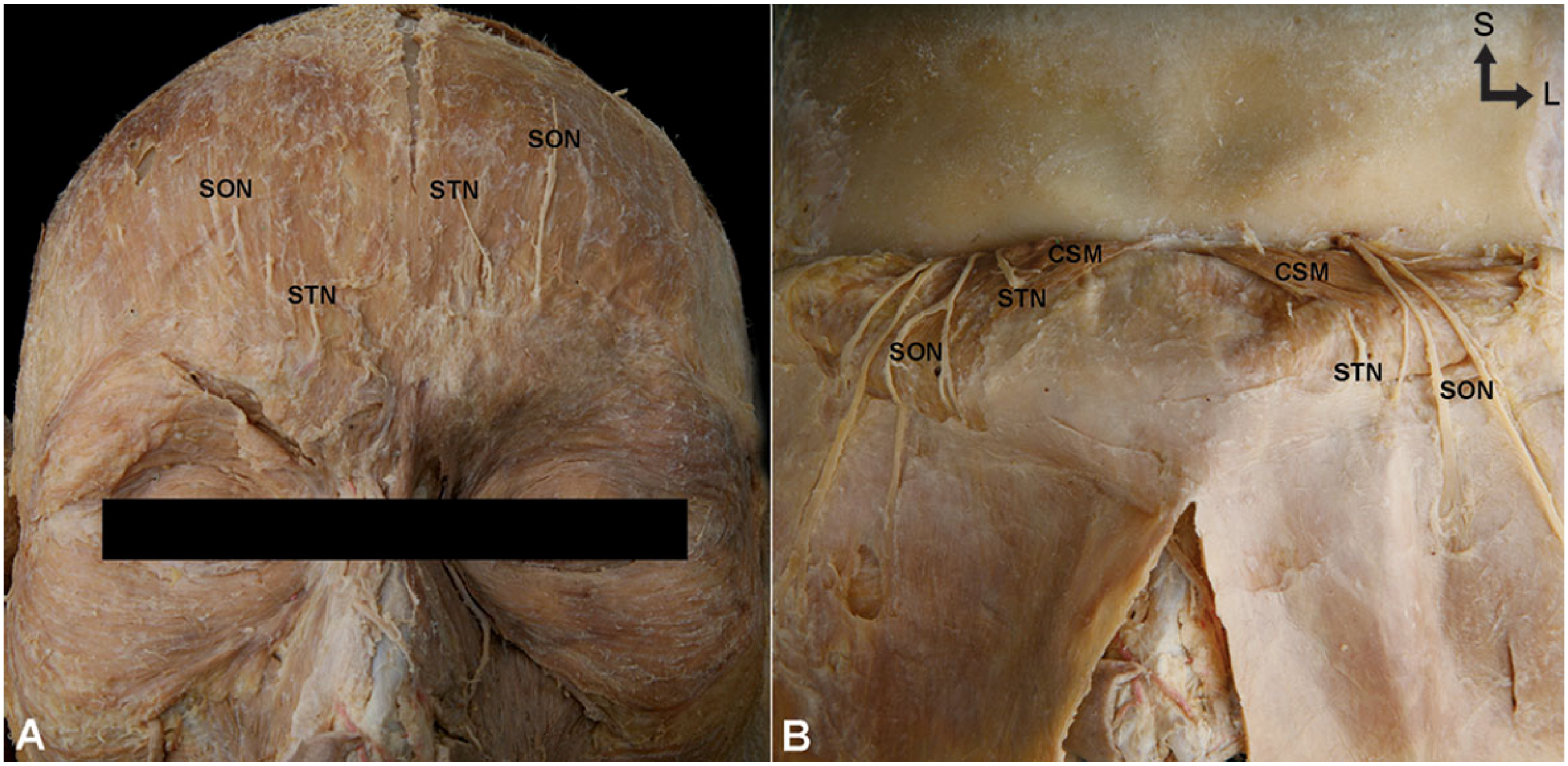
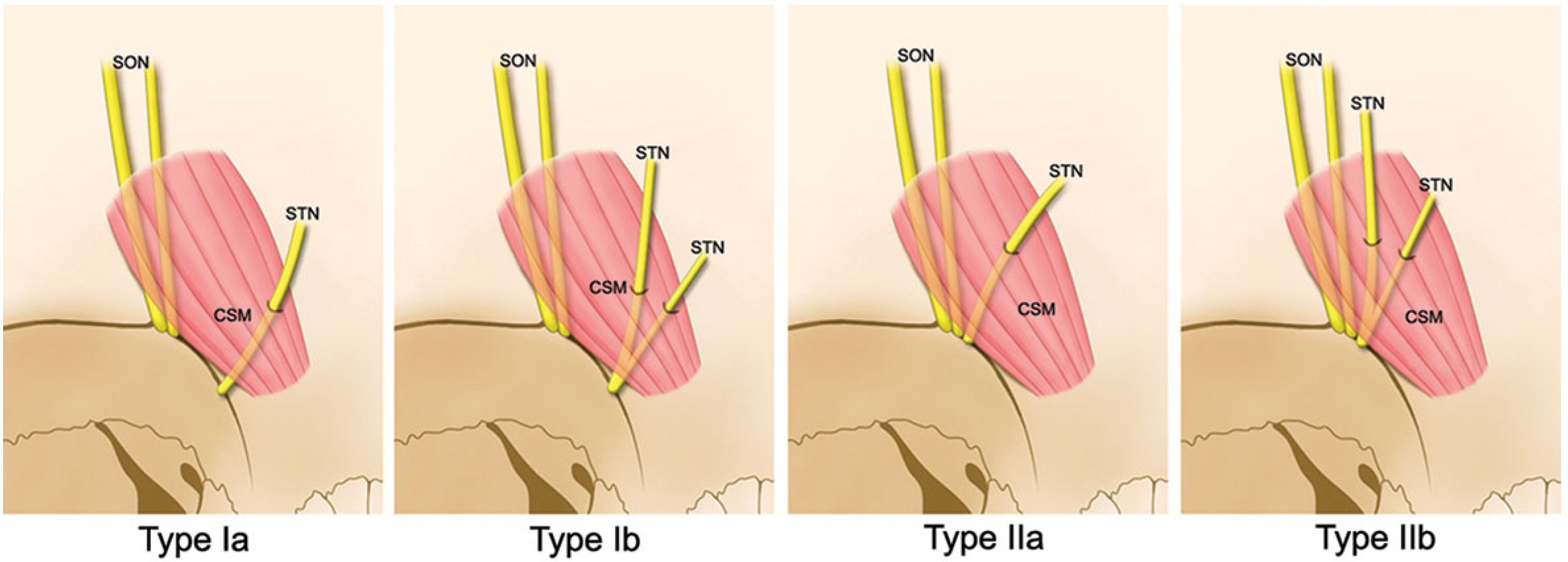
- Type Ia:
- The STN emerges independently from the supraorbital notch region, and passes through the CSM as a single nerve branch.
- Type Ib:
- The STN emerges independently from the supraorbital notch region, and bifurcates into two branches prior to entering the CSM.
- Type IIa:
- The STN emerges from the supraorbital notch region with the SON and passes through the CSM as a single nerve branch.
- Type IIb:
- The STN emerges from the supraorbital notch region with the SON and then bifurcates into two branches prior to entering the CSM.
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Moore, K.L.; Dalley, A.F. Clinically Oriented Anatomy, 5th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006. [Google Scholar]
- Standring, S.; Gray, H. Gray’s Anatomy the Anatomical Basis of Clinical Practice, 40th ed.; Churchill Livingstone: Edinburgh, UK, 2008. [Google Scholar]
- Afifi, A.M.; Alghoul, M.; Zor, F.; Kusuma, S.; Zins, J.E. Comparison of the transpalpebral and endoscopic approaches in resection of the corrugator supercilii muscle. Aesthet. Surg. J. 2012, 32, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Bidros, R.S.; Salazar-Reyes, H.; Friedman, J.D. Subcutaneous temporal browlift under local anesthesia: A useful technique for periorbital rejuvenation. Aesthet Surg. J. 2010, 30, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Behmand, R.A.; Tucker, T.; Guyuron, B. Single-Site botulinum toxin type a injection for elimination of migraine trigger points. Headache 2003, 43, 1085–1089. [Google Scholar] [CrossRef] [PubMed]
- Ducic, I.; Felder, J.M., 3rd; Fantus, S.A. A systematic review of peripheral nerve interventional treatments for chronic headaches. Ann. Plast. Surg. 2014, 72, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Robbins, M.S.; Kuruvilla, D.; Blumenfeld, A.; Charleston, L., 4th; Sorrell, M.; Robertson, C.E.; Grosberg, B.M.; Bender, S.D.; Napchan, U.; Ashkenazi, A. Trigger point injections for headache disorders: Expert consensus methodology and narrative review. Headache 2014, 54, 1441–1459. [Google Scholar] [CrossRef] [PubMed]
- Liberini, P.; Pari, E.; Gazzina, S.; Caratozzolo, S.; Rao, R.; Padovani, A. Technique of injection of onabotulinumtoxin A for chronic migraine: The PREEMPT injection paradigm. Neurol. Sci. 2014, 35 (Suppl. 1), 41–43. [Google Scholar] [CrossRef] [PubMed]
- De Ru, J.A.; Buwalda, J. Botulinum toxin A injection into corrugator muscle for frontally localised chronic daily headache or chronic tension-type headache. J. Laryngol. Otol. 2009, 123, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Dodick, D.; Blumenfeld, A.; Silberstein, S.D. Botulinum neurotoxin for the treatment of migraine and other primary headache disorders. Clin. Dermatol. 2004, 22, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Guyuron, B.; Varghai, A.; Michelow, B.J.; Thomas, T.; Davis, J. Corrugator supercilii muscle resection and migraine headaches. Plast Reconstr. Surg. 2000, 106, 429–434; discussion 435–437. [Google Scholar] [CrossRef] [PubMed]
- Janis, J.E.; Hatef, D.A.; Hagan, R.; Schaub, T.; Liu, J.H.; Thakar, H.; Bolden, K.M.; Heller, J.B.; Kurkjian, T.J. Anatomy of the supratrochlear nerve: Implications for the surgical treatment of migraine headaches. Plast Reconstr. Surg. 2013, 131, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.C.; Bogart, M.M.; Wee, S.A.; Burstein, R.; Arndt, K.A.; Dover, J.S. Predicting migraine responsiveness to botulinum toxin type A injections. Arch. Dermatol. 2010, 146, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Levin, M. Nerve blocks in the treatment of headache. Neurotherapeutics 2010, 7, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Grazzi, L. Onabotulinum toxin A for treatment of chronic migraine with medication overuse. Neurol. Sci. 2013, 34 (Suppl. 1), S27–S28. [Google Scholar] [CrossRef] [PubMed]
- Gerwin, R. Treatment of chronic migraine headache with onabotulinumtoxinA. Curr. Pain Headache Rep. 2011, 15, 336–338. [Google Scholar] [CrossRef] [PubMed]
- Benecke, R.; Heinze, A.; Reichel, G.; Hefter, H.; Gobel, H.; Dysport myofascial pain study group. Botulinum type A toxin complex for the relief of upper back myofascial pain syndrome: How do fixed-location injections compare with trigger point-focused injections? Pain Med. 2011, 12, 1607–1614. [Google Scholar] [CrossRef] [PubMed]
- Guyuron, B.; Kriegler, J.S.; Davis, J.; Amini, S.B. Comprehensive surgical treatment of migraine headaches. Plast Reconstr. Surg. 2005, 115, 1–9. [Google Scholar] [PubMed]
- Fallucco, M.; Janis, J.E.; Hagan, R.R. The anatomical morphology of the supraorbital notch: Clinical relevance to the surgical treatment of migraine headaches. Plast Reconstr. Surg. 2012, 130, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Afridi, S.K.; Shields, K.G.; Bhola, R.; Goadsby, P.J. Greater occipital nerve injection in primary headache syndromes--prolonged effects from a single injection. Pain 2006, 122, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Blumenfeld, A.; Ashkenazi, A.; Grosberg, B.; Napchan, U.; Narouze, S.; Nett, B.; DePalma, T.; Rosenthal, B.; Tepper, S.; Lipton, R.B. Patterns of use of peripheral nerve blocks and trigger point injections among headache practitioners in the USA: Results of the American Headache Society Interventional Procedure Survey (AHS-IPS). Headache 2010, 50, 937–942. [Google Scholar] [CrossRef] [PubMed]
- Andersen, N.B.; Bovim, G.; Sjaastad, O. The frontotemporal peripheral nerves. Topographic variations of the supraorbital, supratrochlear and auriculotemporal nerves and their possible clinical significance. Surg. Radiol. Anat. 2001, 23, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Konofaos, P.; Soto-Miranda, M.A.; Ver Halen, J.; Fleming, J.C. Supratrochlear and supraorbital nerves: An anatomical study and applications in the head and neck area. Ophthal Plast. Reconstr. Surg. 2013, 29, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.A.; Rudkin, G.; Honig, M.; Elahi, M.; Adams, J. Lateral subcutaneous brow lift and interbrow muscle resection: Clinical experience and anatomic studies. Plast Reconstr. Surg. 2000, 105, 1120–1127; discussion 1128. [Google Scholar] [CrossRef] [PubMed]
- Janis, J.E.; Ghavami, A.; Lemmon, J.A.; Leedy, J.E.; Guyuron, B. The anatomy of the corrugator supercilii muscle: Part II. Supraorbital nerve branching patterns. Plast Reconstr. Surg. 2008, 121, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Aurora, S.K.; Dodick, D.W.; Diener, H.C.; DeGryse, R.E.; Turkel, C.C.; Lipton, R.B.; Silberstein, S.D. OnabotulinumtoxinA for chronic migraine: Efficacy, safety, and tolerability in patients who received all five treatment cycles in the PREEMPT clinical program. Acta Neurol. Scand. 2014, 129, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Aurora, S.K.; Dodick, D.W.; Turkel, C.C.; DeGryse, R.E.; Silberstein, S.D.; Lipton, R.B.; Diener, H.C.; Brin, M.F.; PREEMPT 1 Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: Results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 1 trial. Cephalalgia 2010, 30, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Aurora, S.K.; Winner, P.; Freeman, M.C.; Spierings, E.L.; Heiring, J.O.; DeGryse, R.E.; VanDenburgh, A.M.; Nolan, M.E.; Turkel, C.C. OnabotulinumtoxinA for treatment of chronic migraine: Pooled analyses of the 56-week PREEMPT clinical program. Headache 2011, 51, 1358–1373. [Google Scholar] [CrossRef] [PubMed]
- Dodick, D.W.; Turkel, C.C.; DeGryse, R.E.; Aurora, S.K.; Silberstein, S.D.; Lipton, R.B.; Diener, H.C.; Brin, M.F.; PREEMPT Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: Pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache 2010, 50, 921–936. [Google Scholar] [CrossRef] [PubMed]
- Ashkenazi, A.; Silberstein, S. Botulinum toxin type A for the treatment of headache: Why we say yes. Arch. Neurol. 2008, 65, 146–149. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.-J.; Choi, K.-S.; Won, S.-Y.; Apinuntrum, P.; Hu, K.-S.; Kim, S.-T.; Tansatit, T.; Kim, H.-J. Topographic Relationship between the Supratrochlear Nerve and Corrugator Supercilii Muscle—Can This Anatomical Knowledge Improve the Response to Botulinum Toxin Injections in Chronic Migraine? Toxins 2015, 7, 2629-2638. https://doi.org/10.3390/toxins7072629
Lee H-J, Choi K-S, Won S-Y, Apinuntrum P, Hu K-S, Kim S-T, Tansatit T, Kim H-J. Topographic Relationship between the Supratrochlear Nerve and Corrugator Supercilii Muscle—Can This Anatomical Knowledge Improve the Response to Botulinum Toxin Injections in Chronic Migraine? Toxins. 2015; 7(7):2629-2638. https://doi.org/10.3390/toxins7072629
Chicago/Turabian StyleLee, Hyung-Jin, Kwang-Seok Choi, Sung-Yoon Won, Prawit Apinuntrum, Kyung-Seok Hu, Seong-Taek Kim, Tanvaa Tansatit, and Hee-Jin Kim. 2015. "Topographic Relationship between the Supratrochlear Nerve and Corrugator Supercilii Muscle—Can This Anatomical Knowledge Improve the Response to Botulinum Toxin Injections in Chronic Migraine?" Toxins 7, no. 7: 2629-2638. https://doi.org/10.3390/toxins7072629
APA StyleLee, H.-J., Choi, K.-S., Won, S.-Y., Apinuntrum, P., Hu, K.-S., Kim, S.-T., Tansatit, T., & Kim, H.-J. (2015). Topographic Relationship between the Supratrochlear Nerve and Corrugator Supercilii Muscle—Can This Anatomical Knowledge Improve the Response to Botulinum Toxin Injections in Chronic Migraine? Toxins, 7(7), 2629-2638. https://doi.org/10.3390/toxins7072629






