A Severe Accident Caused by an Ocellate River Stingray (Potamotrygon motoro) in Central Brazil: How Well Do We Really Understand Stingray Venom Chemistry, Envenomation, and Therapeutics?
Abstract
:1. Introduction
2. Case Report
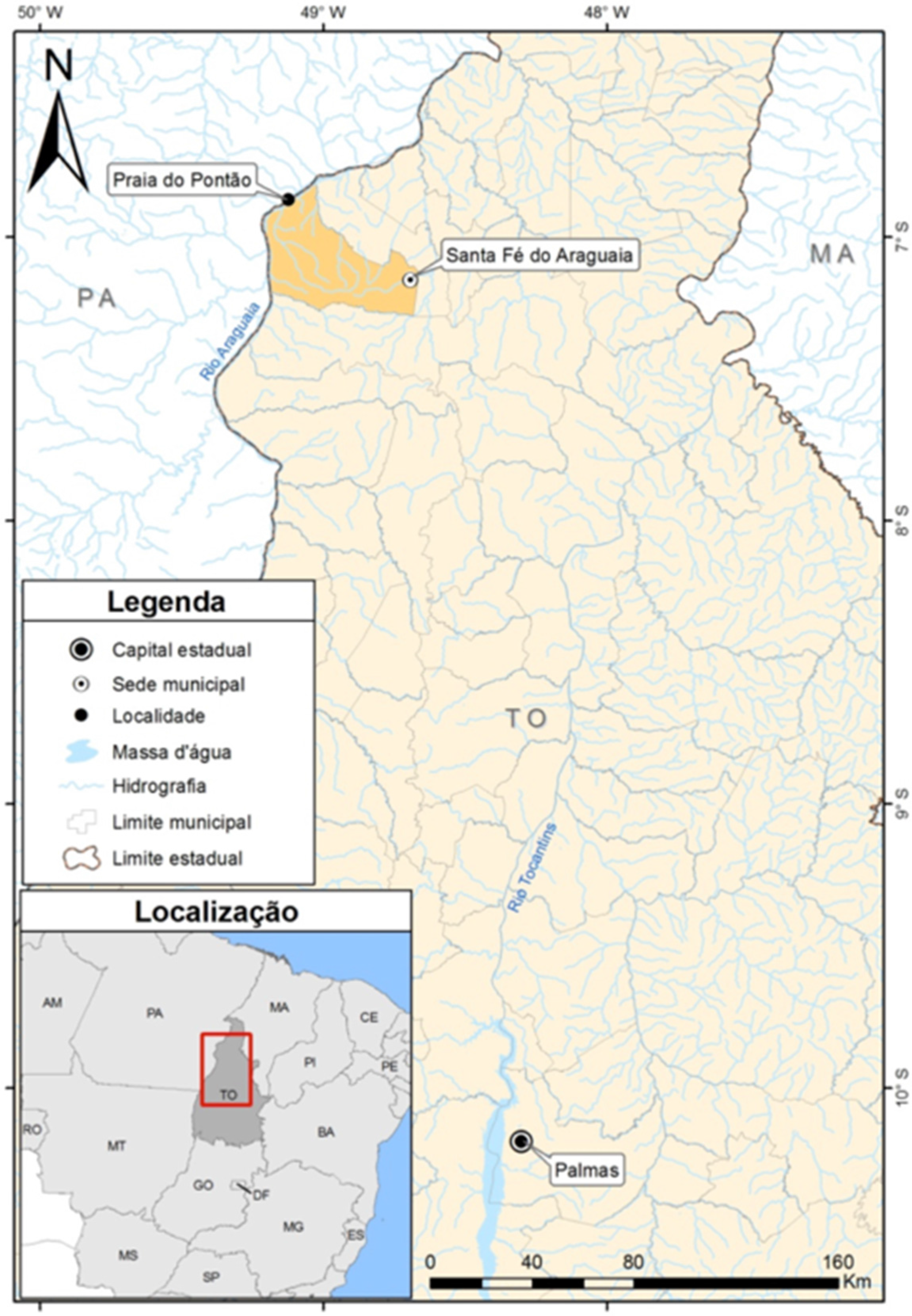
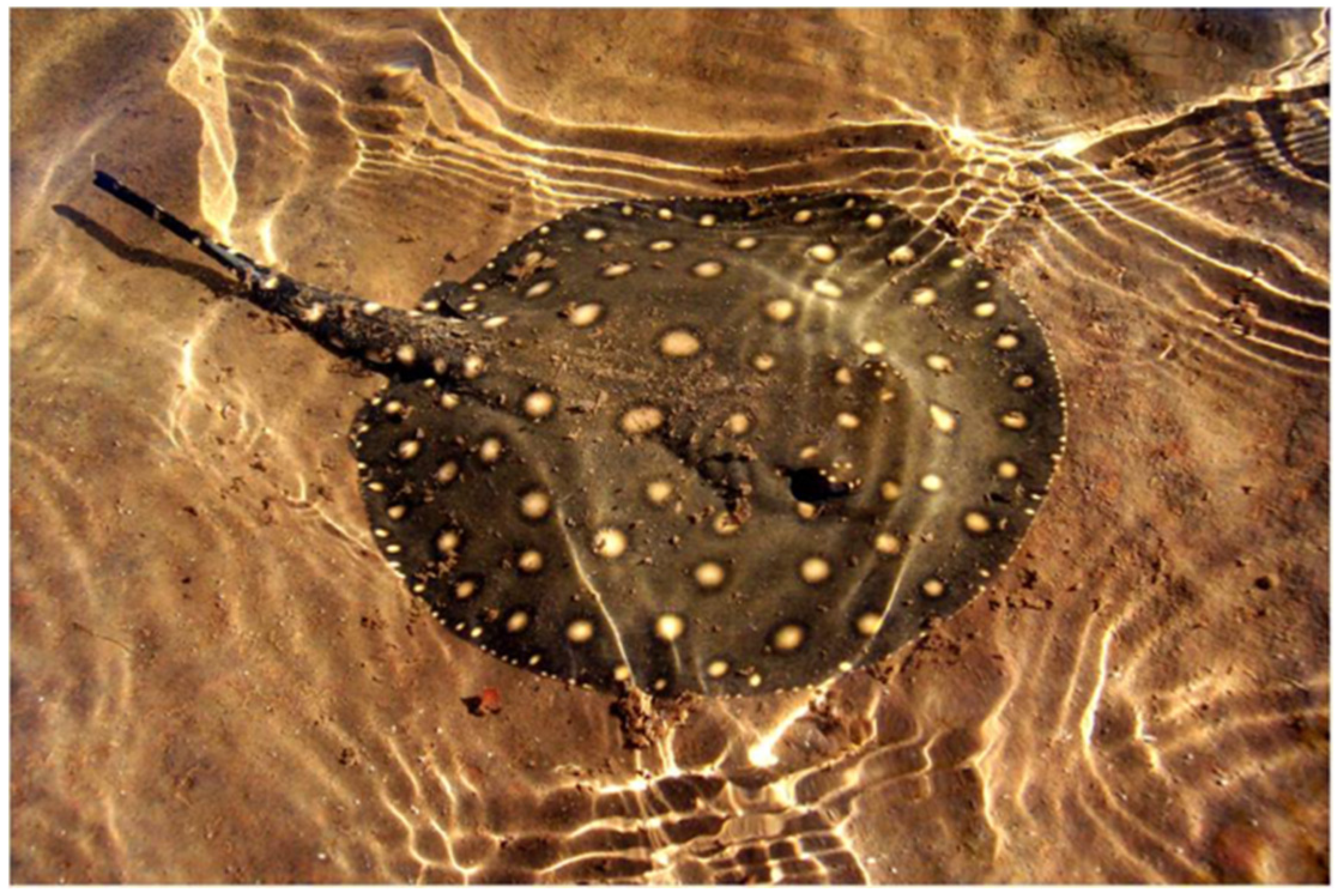
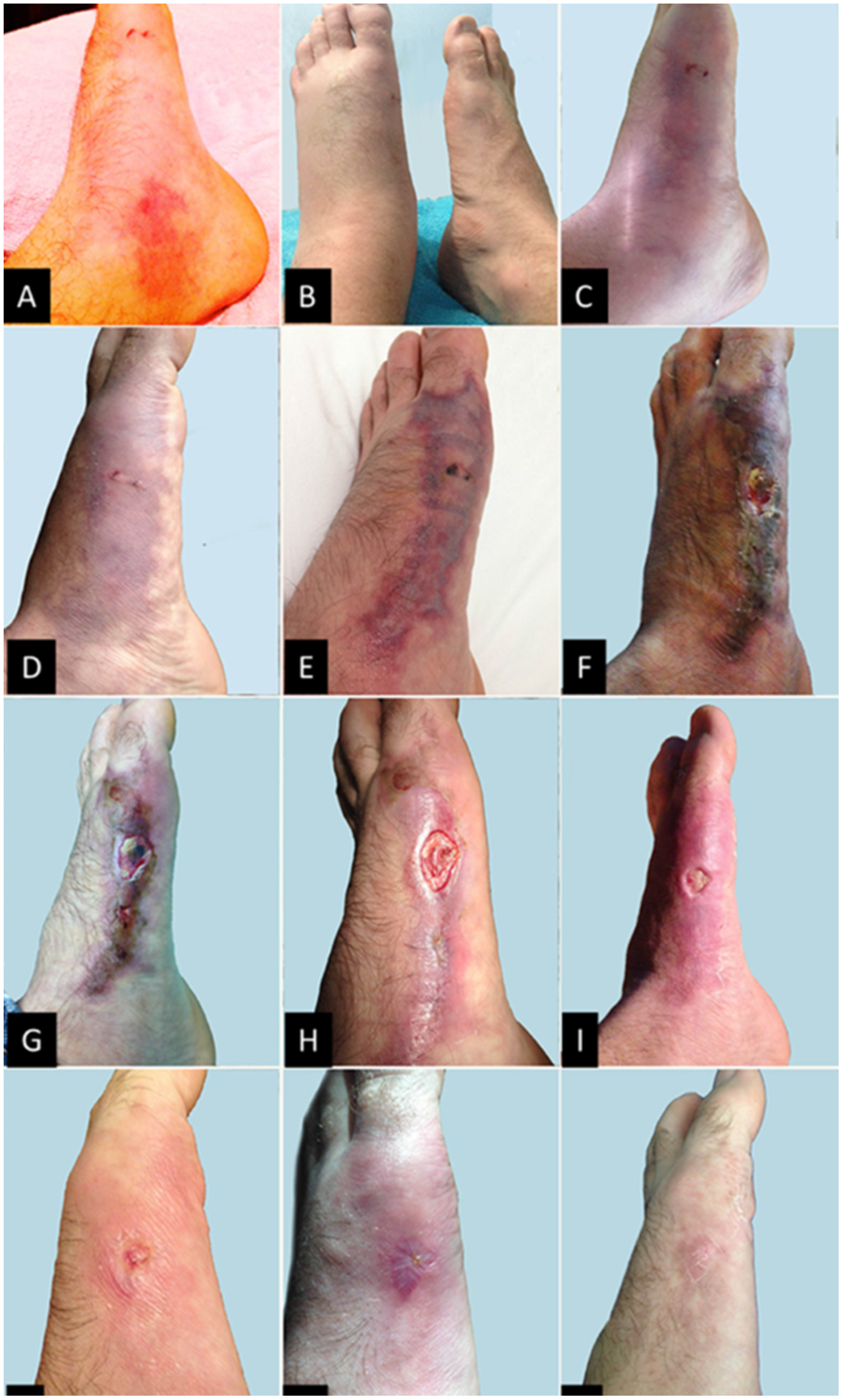
3. Discussion
3.1. Characterizations of Potamotrygon Venoms
3.2. Components Responsible for the Pharmacology of Stingray Venom and Mucus
- What biochemical components are produced by stingrays specifically to serve as toxins?
- To what extent do cytoplasmic components of stingray venom-producing cells contribute, if at all, to early symptoms of stingray envenomation?
- Does stingray mucus itself possess pharmacological activity relevant to envenomation?
- Why is the pain caused by freshwater stingray envenomations so intractable and persistent?
- What non-mucoid components of mucus, if any, are produced by bacteria in the mucus, rather than by the stingrays themselves?
- To what degree do bacteria in the mucus and in the water contribute to the sequelae of stingray envenomation?
3.3. Clinical Treatment of the Patient
4. Conclusions and Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lovejoy, N.R. Stingrays, Parasites, and Neotropical Biogeography: A Closer Look at Brooks et al/s Hypotheses Concerning the Origins of Neotropical Freshwater Rays (Potamotrygonidae). Syst. Biol. 1997, 46, 218–230. [Google Scholar] [CrossRef]
- Thorson, T.B.; Brooks, D.R.; Mayes, M.A. The evolution of freshwater adaptation in stingrays. Natl. Geogr. Soc. Res. Rep. 1983, 15, 663–694. [Google Scholar]
- Rosa, R.S. A Systematic Revision of the South American Freshwater Stingrays (Chondrichthyes, Potamotrygonidae); The College of William and Mary: Williamsburg, VA, USA, 1985. [Google Scholar]
- Lovejoy, N.R. Systematics of myliobatoid elasmobranchs with emphasis on the phylogeny and historical biogeography of Neotropical freshwater stingrays (Potamotrygonidae: Rajiformes). Zool. J. Linn. Soc. 1996, 117, 207–257. [Google Scholar] [CrossRef]
- Lovejoy, N.R.; Albert, J.S.; Crampton, W.G.R. Miocene marine incursions and marine/freshwater transitions: Evidence from Neotropical fishes. J. S. Am. Earth Sci. 2006, 21, 5–13. [Google Scholar] [CrossRef]
- Carvalho, M.R.; Lovejoy, N.R.; Rosa, R.S. Family Potamotrygonidae. In Checklist of the Freshwater Fishes of South and Central America; Reis, R.E., Ferraris, C.J., Jr., Kullander, S.O., Eds.; EDIPUCRS: Porto Alegre, Rio Grande do Sul, Brazil, 2003; pp. 22–29. [Google Scholar]
- Carvalho, M.R.; Maisey, J.G.; Grande, L. Freshwater stingray of the Green River Formation of Wyoming (Early Eocene), with description of a new genus and species and an analysis of its phylogenetic relationships (Chondrichthyes: Myliobatiformes). Bull. Am. Mus. Nat. Hist. 2004, 284, 1–136. [Google Scholar] [CrossRef]
- Hoorn, C.; Wesselingh, F.P.; ter Steege, H.; Bermudez, M.A.; Mora, A.; Sevink, J.; Sanmartin, I.; Sanchez-Meseguer, A.; Anderson, C.L.; Figueiredo, J.P.; et al. Amazonia through time: Andean uplift, climate change, landscape evolution, and biodiversity. Science 2010, 330, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, M.R.; Lovejoy, N.R. Morphology and phylogenetic relationships of a remarkable new genus and two new species of Neotropical freshwater stingrays from the Amazon basin (Chondrichthyes: Potamotrygonidae). Zootaxa 2011, 48, 13–48. [Google Scholar]
- Rosa, R.S.; Charvet-Almeida, P.; Quijada, C.C.D. Biology of the South American Potamotrygonid stingray. In Sharks and Their Relatives II: Biodiversity, Adaptative Physiology and Conservation; Carrier, J.C., Musick, J.A., Heithaus, M.R., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 241–286. [Google Scholar]
- Carvalho, M.R.; Perez, M.H.S.; Lovejoy, N.R. Potamotrygon tigrina, a new species of freshwater stingray from the upper Amazon basin, closely related to Potamotrygon schroederi Fernandez-Yépez, 1958 (Chondrichthyes: Potamotrygonidae). Zootaxa 2011, 2827, 1–30. [Google Scholar]
- Silva, J.P.C.B.; Carvalho, M.R. A new species of Neotropical freshwater stingray of the genus Potamotrygon Garman, 1877 from the Río Madre de Díos, Peru (Chondrichthyes: Potamotrygonidae). Pap. Avulsos Zool. 2011, 51, 139–154. [Google Scholar] [CrossRef]
- Fontenelle, J.P.; da Silva, J.P.; de Carvalho, M.R. Potamotrygon limai, sp. nov., a new species of freshwater stingray from the upper Madeira River system, Amazon basin (Chondrichthyes: Potamotrygonidae). Zootaxa 2014, 3765, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Rosa, R.S.; Carvalho, M.R. Família Potamotrygonidae. Catálogo das Espécies de Peixes de Água Doce do Brasil; Museu Nacional: Rio de Janeiro, Brazil, 2007. [Google Scholar]
- Soares, E.C. Peixes do Mearim; Editora Geia: São Luís, Maranhão, Brazil, 2005. [Google Scholar]
- Garman, S. The Plagiostoma (sharks, skates and rays). Mem. Mus. Comp. Zool. 1913, 36, 1–515. [Google Scholar]
- Compagno, L.J.V.; Cook, S.F. The exploitation and conservation of freshwater elasmobranchs: Status of taxa and prospects for the future. J. Aquaricult. Aqua. Sci. 1995, 7, 62–90. [Google Scholar]
- Loboda, T.S.; Carvalho, M.R. Systematic revision of the Potamotrygon motoro (Müller & Henle, 1841) species complex in the Paraná-Paraguay basin, with description of two new ocellated species (Chondrichthyes: Myliobatiformes: Potamotrygonidae). Neotrop. Ichthyol. 2013, 11, 693–737. [Google Scholar]
- Almeida, M.P.; Lins, P.M.; Charvet-Almeida, P.; Barthem, R.B. Diet of the freshwater stingray Potamotrygon motoro (Chondrichthyes: Potamotrygonidae) on Marajo Island (Para, Brazil). Braz. J. Biol. 2010, 70, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.P.; Barthem, R.B.; da Silva Viana, A.; Charvet-Almeida, P. Factors affecting the distribution and abundance of freshwater stingrays (Chondrichthyes: Potamotrygonidae) at Marajó Island, mouth of the Amazon River. Pan Am. J. Aquat. Sci. 2009, 4, 1–11. [Google Scholar]
- Halstead, B.W. Poisonous and Venomous Marine Animals of the World; United States Government Printing Office: Washington, DC, USA, 1970; Volume 3.
- Charvet-Almeida, P.; Araújo, M.L.G.; Rosa, R.S.; Rincón, G. Neotropical freshwater stingray: Diversity and conservation status. Shark News 2002, 14, 47–51. [Google Scholar]
- Haddad, V., Jr.; Neto, D.G.; de Paula Neto, J.B.; de Luna Marques, F.P.; Barbaro, K.C. Freshwater stingrays: Study of epidemiologic, clinic and therapeutic aspects based on 84 envenomings in humans and some enzymatic activities of the venom. Toxicon 2004, 43, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Barbaro, K.C.; Lira, M.S.; Malta, M.B.; Soares, S.L.; Garrone Neto, D.; Cardoso, J.L.; Santoro, M.L.; Haddad Junior, V. Comparative study on extracts from the tissue covering the stingers of freshwater (Potamotrygon falkneri) and marine (Dasyatis guttata) stingrays. Toxicon 2007, 50, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Pedroso, C.M.; Jared, C.; Charvet-Almeida, P.; Almeida, M.P.; Garrone Neto, D.; Lira, M.S.; Haddad, V., Jr.; Barbaro, K.C.; Antoniazzi, M.M. Morphological characterization of the venom secretory epidermal cells in the stinger of marine and freshwater stingrays. Toxicon 2007, 50, 688–697. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, M.R.; da Silva, N.J., Jr.; Ulhoa, C.J. A hyaluronidase from Potamotrygon motoro (freshwater stingrays) venom: Isolation and characterization. Toxicon 2008, 51, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Garrone Neto, D.; Haddad, V., Jr. Arraias em rios da região Sudeste do Brasil: Locais de ocorrência e impactos sobre a população. Rev. Soc. Bras. Med. Trop. 2010, 43, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Antoniazzi, M.M.; Benvenuti, L.A.; Lira, M.S.; Jared, S.G.; Neto, D.G.; Jared, C.; Barbaro, K.C. Histopathological changes induced by extracts from the tissue covering the stingers of Potamotrygon falkneri freshwater stingrays. Toxicon 2011, 57, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Haddad, V., Jr. Atlas de Animais Aquáticos Perigosos do Brasil: Guia Médico de Identificação e Tratamento; Editora Roca: São Paulo, Brazil, 2000. [Google Scholar]
- Junghanss, T.; Bodio, M. Medically important venomous animals: Biology, prevention, first aid, and clinical management. Clin. Infect. Dis. 2006, 43, 1309–1317. [Google Scholar] [PubMed]
- Garrone Neto, D.; Haddad, V., Jr. Acidentes por raias. In Animais Peçonhentos no Brasil: Biologia, Clínica e Terapêutica dos Acidentes; Cardoso, J.L.C., França, F.O.D.S., Wen, F.H., Málaque, C.M.S.A., Haddad, V., Jr., Eds.; Sarvier: São Paulo, Brazil, 2009; pp. 295–305. [Google Scholar]
- Pardal, P.P.O. Ictismo por arraia. In Animais Peçonhentos no Brasil: Biologia, Clínica e Terapêutica dos Acidentes; Sarvier: São Paulo, Brazil, 2009; pp. 523–529. [Google Scholar]
- Lameiras, J.L.V.; Costa, O.T.F.; Santos, M.C.; Duncan, W.L.P. Arraias de água doce (Chondrichthyes—Potamotrygonidae): Biologia, veneno e acidentes. Sci. Amazon. 2013, 2, 11–27. [Google Scholar]
- Domingos, M.O.; Franzolin, M.R.; dos Anjos, M.T.; Franzolin, T.M.; Barbosa Albes, R.C.; de Andrade, G.R.; Lopes, R.J.; Barbaro, K.C. The influence of environmental bacteria in freshwater stingray wound-healing. Toxicon 2011, 58, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Barber, G.R.; Swygert, J.S. Necrotizing fasciitis due to Photobacterium damsela in a man lashed by a stingray. N. Engl. J. Med. 2000, 342, 824. [Google Scholar] [CrossRef] [PubMed]
- Torrez, P.P.; Quiroga, M.M.; Said, R.; Abati, P.A.; Franca, F.O. Tetanus after envenomations caused by freshwater stingrays. Toxicon 2015, 97, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.F.; Girard, R.H.; Rao, D.; Ly, B.T.; Davis, D.P. Stingray envenomation: A retrospective review of clinical presentation and treatment in 119 cases. J. Emerg. Med. 2007, 33, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Haddad, V., Jr.; Cardoso, J.L.C.; Garrone Neto, D. Injuries by marine and freshwater stingray: History, clinical aspects of the envenomations and current status of a neglected problem in Brazil. J. Venom. Anim. Toxins Incl. Trop. Dis. 2013, 19, 1–10. [Google Scholar]
- Diaz, J.H. The evaluation, management, and prevention of stingray injuries in travelers. J. Travel Med. 2008, 15, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, R.J. Pharmacology of South American freshwater stingray venom (Potamotrygon motoro). Trans. N. Y. Acad. Sci. 1972, 34, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Baumann, K.; Casewell, N.R.; Ali, S.A.; Jackson, T.N.; Vetter, I.; Dobson, J.S.; Cutmore, S.C.; Nouwens, A.; Lavergne, V.; Fry, B.G. A ray of venom: Combined proteomic and transcriptomic investigation of fish venom composition using barb tissue from the blue-spotted stingray (Neotrygon kuhlii). J. Proteomics 2014, 109, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Sivan, G. Fish venom: Pharmacological features and biological significance. Fish Fish. 2009, 10, 159–172. [Google Scholar] [CrossRef]
- Conceicao, K.; Konno, K.; Melo, R.L.; Marques, E.E.; Hiruma-Lima, C.A.; Lima, C.; Richardson, M.; Pimenta, D.C.; Lopes-Ferreira, M. Orpotrin: A novel vasoconstrictor peptide from the venom of the Brazilian stingray Potamotrygon gr. orbignyi. Peptides 2006, 27, 3039–3046. [Google Scholar] [CrossRef] [PubMed]
- Conceicao, K.; Santos, J.M.; Bruni, F.M.; Klitzke, C.F.; Marques, E.E.; Borges, M.H.; Melo, R.L.; Fernandez, J.H.; Lopes-Ferreira, M. Characterization of a new bioactive peptide from Potamotrygon gr. orbignyi freshwater stingray venom. Peptides 2009, 30, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, K.W.; Lima, C.; Piran-Soares, A.A.; Marques, E.E.; Hiruma-Lima, C.A.; Lopes-Ferreira, M. Biological and biochemical properties of the Brazilian Potamotrygon stingrays: Potamotrygon cf. scobina and Potamotrygon gr. orbignyi. Toxicon 2006, 47, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, M.R. Estudos Bioquímicos do Veneno de Raias Potamotrygon Motoro (Chondrichthyes: Dasyatidae, Potamotrygoninae)—Purificação e Caracterização de uma Hialuronidase. Dissertação de Mestrado, Instituto de Ciências Biológicas, Universidade Federal de Goiás, Goiânia, Goiás, Brasil, 2001. [Google Scholar]
- Conceicao, K.; Monteiro-dos-Santos, J.; Seibert, C.S.; Silva, P.I., Jr.; Marques, E.E.; Richardson, M.; Lopes-Ferreira, M. Potamotrygon cf. henlei stingray mucus: Biochemical features of a novel antimicrobial protein. Toxicon 2012, 60, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Kirchhoff, K.N.; Klingelhofer, I.; Dahse, H.M.; Morlock, G.; Wilke, T. Maturity-related changes in venom toxicity of the freshwater stingray Potamotrygon leopoldi. Toxicon 2014, 92, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-dos-Santos, J.; Conceicao, K.; Seibert, C.S.; Marques, E.E.; Silva, P.I., Jr.; Soares, A.B.; Lima, C.; Lopes-Ferreira, M. Studies on pharmacological properties of mucus and sting venom of Potamotrygon cf. henlei. Int. Immunopharmacol. 2011, 11, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Hiemenz, J.W.; Kennedy, B.; Kwon-Chung, K.J. Invasive fusariosis associated with an injury by a stingray barb. J. Med. Vet. Mycol. 1990, 28, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Ho, P.L.; Tang, W.M.; Lo, K.S.; Yuen, K.Y. Necrotizing fasciitis due to Vibrio alginolyticus following an injury inflicted by a stingray. Scand. J. Infect. Dis. 1998, 30, 192–193. [Google Scholar] [PubMed]
- Minnaganti, V.R.; Patel, P.J.; Iancu, D.; Schoch, P.E.; Cunha, B.A. Necrotizing fasciitis caused by Aeromonas hydrophila. Heart Lung J. Crit. Care 2000, 29, 306–308. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, S. Extracellular proteolytic enzymes produced by human pathogenic vibrio species. Front. Microbiol. 2013, 4, 339. [Google Scholar] [CrossRef] [PubMed]
- Mullins, J.F.; Wilson, C.J.; Best, W.C. Cryotherapy in the treatment of stingray wounds. South. Med. J. 1957, 50, 533–535. [Google Scholar] [CrossRef] [PubMed]
- Brisset, I.B.; Schaper, A.; Pommier, P.; de Haro, L. Envenomation by Amazonian freshwater stingray Potamotrygon motoro: 2 cases reported in Europe. Toxicon 2006, 47, 32–34. [Google Scholar] [CrossRef] [PubMed]
- Russell, F.E.; Lewis, R. Evaluation of the current status of therapy for stingray injuries. In Venoms; American Association for the Advancement of Science: Washington, DC, USA, 1956; pp. 43–53. [Google Scholar]
- Russell, F.E.; Panos, T.C.; Kang, L.W.; Warner, A.M.; Colket, T.C., 3rd. Studies on the mechanism of death from stingray venom; a report of two fatal cases. Am. J. Med. Sci. 1958, 235, 566–584. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.J.; Davies, R.S. Stingray injury. J. Accid. Emerg. Med. 1996, 13, 224–225. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.K. Stingray injuries. Wilderness Environ. Med. 1997, 8, 24–28. [Google Scholar] [CrossRef]
- Giltner, C.L.; Bobenchik, A.M.; Uslan, D.Z.; Deville, J.G.; Humphries, R.M. Ciprofloxacin-resistant Aeromonas hydrophila cellulitis following leech therapy. J. Clin. Microbiol. 2013, 51, 1324–1326. [Google Scholar] [CrossRef] [PubMed]
- Figueira, V.; Vaz-Moreira, I.; Silva, M.; Manaia, C.M. Diversity and antibiotic resistance of Aeromonas spp. in drinking and waste water treatment plants. Water Res. 2011, 45, 5599–5611. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.T. Recent progress in novel macrolides, quinolones, and 2-pyridones to overcome bacterial resistance. Med. Res. Rev. 1999, 19, 497–520. [Google Scholar] [CrossRef]
- Chang, V.S.; Dhaliwal, D.K.; Raju, L.; Kowalski, R.P. Antibiotic Resistance in the Treatment of Staphylococcus aureus Keratitis: A 20-Year Review. Cornea 2015, 34, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ogutu, J.O.; Gu, J.; Ding, F.; You, Y.; Huo, Y.; Zhao, H.; Li, W.; Zhang, Z.; Zhang, W.; et al. Comparative analysis of quinolone resistance in clinical isolates of Klebsiella pneumoniae and Escherichia coli from Chinese children and adults. BioMed Res. Int. 2015, 2015, 168292. [Google Scholar] [CrossRef] [PubMed]
- Maleki, M.H.; Jalilian, F.A.; Khayat, H.; Mohammadi, M.; Pourahmad, F.; Asadollahi, K.; Pakzad, I.; Sadeghifard, N.; Soroush, S.; Emaneini, M.; et al. Detection of highly ciprofloxacin resistance acinetobacter baumannii isolated from patients with burn wound infections in presence and absence of efflux pump inhibitor. Maedica (Buchar) 2014, 9, 162–167. [Google Scholar] [PubMed]
- Park, M.; Sutherland, J.B.; Kim, J.N.; Rafii, F. Effect of fluoroquinolone resistance selection on the fitness of three strains of Clostridium perfringens. Microb. Drug Resist. 2013, 19, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.S. Peripheral neuropathy associated with fluoroquinolones. Ann. Pharmacother. 2001, 35, 1540–1547. [Google Scholar] [CrossRef] [PubMed]
- Jumma, O.K.; Dick, J.; Marshall, A.; Mellor, K. Ciprofloxacin induced acute small fibre neuropathy. Case report. Can. J. Neurol. Sci. 2013, 40, 127–128. [Google Scholar] [PubMed]
- Samarakoon, N.; Harrisberg, B.; Ell, J. Ciprofloxacin-induced toxic optic neuropathy. Clin. Exp. Ophthalmol. 2007, 35, 102–104. [Google Scholar] [CrossRef] [PubMed]
- Ghatole, M.; Manthalkar, P.; Kandle, S.; Yemul, V.; Jahagirdar, V. Correlation of extended spectrum beta lactamases production with cephalosporin resistance in gram negative bacilli. Indian J. Pathol. Microbiol. 2004, 47, 82–84. [Google Scholar] [PubMed]
- Chuang, Y.M.; Tseng, S.P.; Teng, L.J.; Ho, Y.C.; Hsueh, P.R. Emergence of cefotaxime resistance in Citrobacter freundii causing necrotizing fasciitis and osteomyelitis. J. Infect. 2006, 53, e161–e163. [Google Scholar] [CrossRef] [PubMed]
- Brusselaers, N.; Vogelaers, D.; Blot, S. The rising problem of antimicrobial resistance in the intensive care unit. Ann. Intensive Care 2011, 1, 47. [Google Scholar] [CrossRef] [PubMed]
- Ko, W.C.; Wu, H.M.; Chang, T.C.; Yan, J.J.; Wu, J.J. Inducible beta-lactam resistance in Aeromonas hydrophila: Therapeutic challenge for antimicrobial therapy. J. Clin. Microbiol. 1998, 36, 3188–3192. [Google Scholar] [PubMed]
- Harada, K.; Arima, S.; Niina, A.; Kataoka, Y.; Takahashi, T. Characterization of Pseudomonas aeruginosa isolates from dogs and cats in Japan: Current status of antimicrobial resistance and prevailing resistance mechanisms. Microbiol. Immunol. 2012, 56, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Schechter, N.M. Structure of the dermal-epidermal junction and potential mechanisms for its degradation: The possible role of inflammatory cells. Immunol. Ser. 1989, 46, 477–507. [Google Scholar] [PubMed]
- Paquet, P.; Nusgens, B.V.; Pierard, G.E.; Lapiere, C.M. Gelatinases in drug-induced toxic epidermal necrolysis. Eur. J. Clin. Investig. 1998, 28, 528–532. [Google Scholar] [CrossRef]
- Burns, E.H., Jr.; Marciel, A.M.; Musser, J.M. Structure-function and pathogenesis studies of Streptococcus pyogenes extracellular cysteine protease. Adv. Exp. Med. Biol. 1997, 418, 589–592. [Google Scholar] [PubMed]
- Jackson, R.J.; Lim, D.V.; Dao, M.L. Identification and analysis of a collagenolytic activity in Streptococcus mutans. Curr. Microbiol. 1997, 34, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Seddon, S.V.; Hemingway, I.; Borriello, S.P. Hydrolytic enzyme production by Clostridium difficile and its relationship to toxin production and virulence in the hamster model. J. Med. Microbiol. 1990, 31, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Tytell, A.A.; Hewson, K. Production, purification, and some properties of Clostridium histolyticum collagenase. Proc. Soc. Exp. Biol. Med. 1950, 74, 555–558. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.D. Virulence factors of Clostridium perfringens. Rev. Infect. Dis. 1979, 1, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Rood, J.I. Virulence genes of Clostridium perfringens. Annu. Rev. Microbiol. 1998, 52, 333–360. [Google Scholar] [CrossRef] [PubMed]
- Diener, B.; Carrick, L., Jr.; Berk, R.S. In vivo studies with collagenase from Pseudomonas aeruginosa. Infect. Immun. 1973, 7, 212–217. [Google Scholar] [PubMed]
- Wellisch, G.; Cohen, E.; Cahane, Z.; Horowitz, J. Simple method for collagenase determination in 38 Pseudomonas aeruginosa strains. J. Clin. Microbiol. 1984, 20, 1020–1021. [Google Scholar] [PubMed]
- Reid, G.C.; Woods, D.R.; Robb, F.T. Peptone induction and rifampin-insensitive collagenase production by Vibrio alginolyticus. J. Bacteriol. 1980, 142, 447–454. [Google Scholar] [PubMed]
- Hare, P.; Scott-Burden, T.; Woods, D.R. Characterization of extracellular alkaline proteases and collagenase induction in Vibrio alginolyticus. J. Gen. Microbiol. 1983, 129, 1141–1147. [Google Scholar] [CrossRef] [PubMed]
- Keil, B. Vibrio alginolyticus (“Achromobacter”) collagenase: Biosynthesis, function and application. Matrix Suppl. 1992, 1, 127–133. [Google Scholar] [PubMed]
- Takeuchi, H.; Shibano, Y.; Morihara, K.; Fukushima, J.; Inami, S.; Keil, B.; Gilles, A.M.; Kawamoto, S.; Okuda, K. Structural gene and complete amino acid sequence of Vibrio alginolyticus collagenase. Biochem. J. 1992, 281, 703–708. [Google Scholar] [PubMed]
- Xiong, P.; Peng, X.; Wei, S.; Chen, Y.; Zhao, H.; Tang, S.; Wu, X. [Virulence-related genes of Vibrio alginolyticus and its virulence in mice]. Wei Sheng Wu Xue Bao 2014, 54, 80–88. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Silva, N.J., Jr.; Ferreira, K.R.C.; Pinto, R.N.L.; Aird, S.D. A Severe Accident Caused by an Ocellate River Stingray (Potamotrygon motoro) in Central Brazil: How Well Do We Really Understand Stingray Venom Chemistry, Envenomation, and Therapeutics? Toxins 2015, 7, 2272-2288. https://doi.org/10.3390/toxins7062272
Da Silva NJ Jr., Ferreira KRC, Pinto RNL, Aird SD. A Severe Accident Caused by an Ocellate River Stingray (Potamotrygon motoro) in Central Brazil: How Well Do We Really Understand Stingray Venom Chemistry, Envenomation, and Therapeutics? Toxins. 2015; 7(6):2272-2288. https://doi.org/10.3390/toxins7062272
Chicago/Turabian StyleDa Silva, Nelson Jorge, Jr., Kalley Ricardo Clementino Ferreira, Raimundo Nonato Leite Pinto, and Steven Douglas Aird. 2015. "A Severe Accident Caused by an Ocellate River Stingray (Potamotrygon motoro) in Central Brazil: How Well Do We Really Understand Stingray Venom Chemistry, Envenomation, and Therapeutics?" Toxins 7, no. 6: 2272-2288. https://doi.org/10.3390/toxins7062272
APA StyleDa Silva, N. J., Jr., Ferreira, K. R. C., Pinto, R. N. L., & Aird, S. D. (2015). A Severe Accident Caused by an Ocellate River Stingray (Potamotrygon motoro) in Central Brazil: How Well Do We Really Understand Stingray Venom Chemistry, Envenomation, and Therapeutics? Toxins, 7(6), 2272-2288. https://doi.org/10.3390/toxins7062272





