Botulinum Neurotoxin Serotypes Detected by Electrochemical Impedance Spectroscopy
Abstract
:1. Introduction
| Serotype | Target Protein | Sequence Cleaved from Protein | No. Amino Acids Cleaved |
|---|---|---|---|
| BoNT/A | SNAP-25 | RATKMLGSG | 9 |
| BoNT/B | VAMP | FETSAAKLKRKYW | 13 |
| BoNT/C | SNAP-25 | ATKMLGSG | 8 |
| BoNT/D | VAMP | LSELDDRADALQAGASQFETSAAKLKRKYW | 30 |
| BoNT/E | SNAP-25 | IMEKADSNKTRIDEANQRATKMLGSG | 26 |
2. Results and Discussion
2.1. Formation of SNAP-25 and VAMP Monolayers
2.2. Electrochemical Impedance Spectroscopy for the Detection of Botulinum Neurotoxin
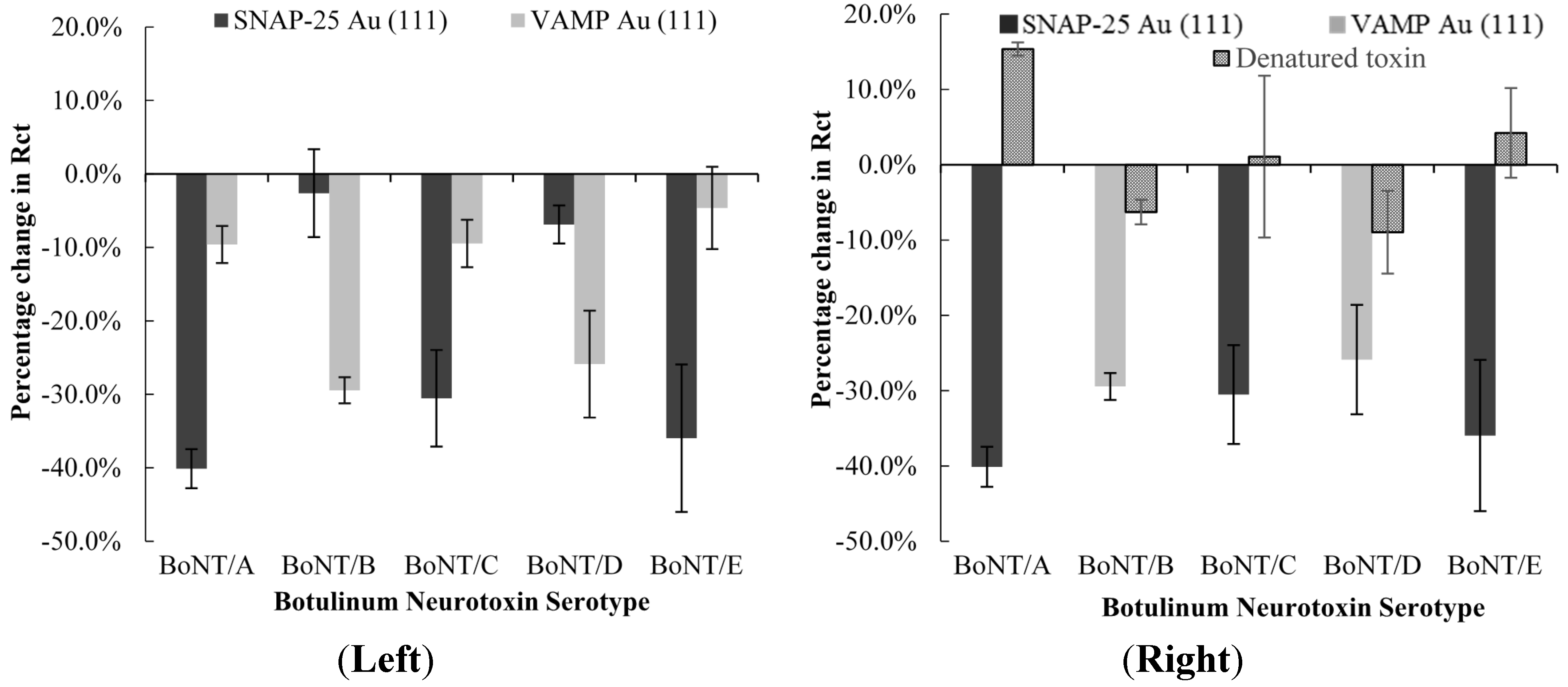
2.2.1. Detection of Botulinum Neurotoxin Serotypes A, C, and E
2.2.2. Detection of Botulinum Neurotoxin Serotypes B and D
2.3. Cross Reactivity of Different Serotypes
3. Experimental Section
3.1. Self-Assembled Monolayers of SNAP-25 and VAMP
3.2. Cyclic Voltammetry
3.3. Electrochemical Impedance Spectroscopy
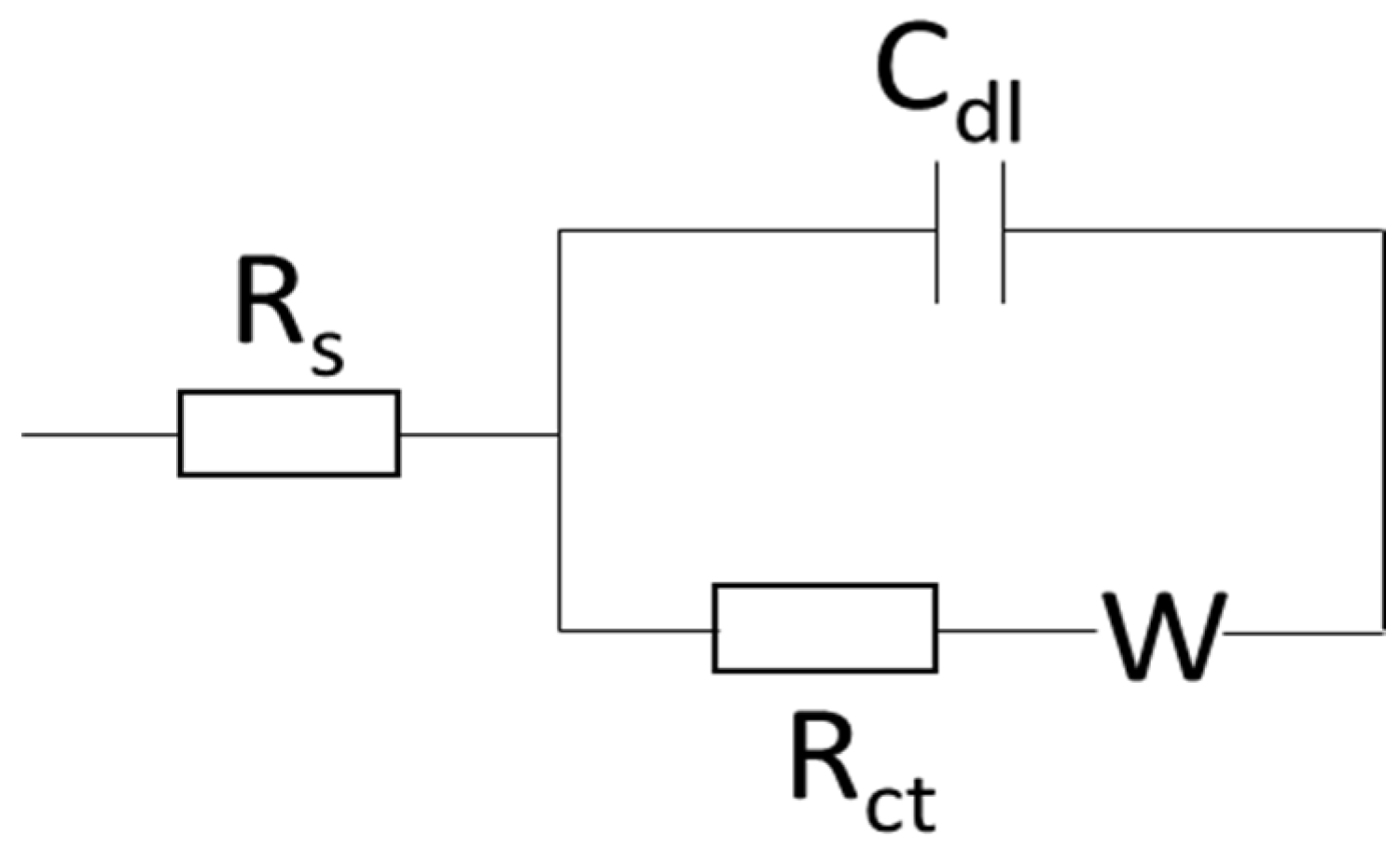
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Balali-Mood, M.; Moshiri, M.; Etemad, L. Medical aspects of bio-terrorism. Toxicon 2013, 69, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Thanongsaksrikul, J.; Chaicumpa, W. Botulinum neurotoxins and botulism: A novel therapeutic approach. Toxins 2011, 3, 469–488. [Google Scholar] [CrossRef] [PubMed]
- Arnon, S.S.; Schechter, R.; Inglesby, T.V.; Henderson, D.A.; Bartlett, J.G.; Ascher, M.S.; Eitzen, E.; Fine, A.D.; Hauer, J.; Layton, M.; et al. Botulinum toxin as a biological weapon. JAMA J. Am. Med. Assoc. 2001, 285, 1059–1070. [Google Scholar] [CrossRef]
- Čapek, P.; Dickerson, T.J. Sensing the deadliest toxin: Technologies for botulinum neurotoxin detection. Toxins 2010, 2, 24–53. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Stanker, L.H.; Sharma, S.K. Botulinum neurotoxin: Where are we with detection technologies? Crit. Rev. Microbiol. 2013. [Google Scholar] [CrossRef]
- Fernández-Salas, E.; Wang, J.; Molina, Y.; Nelson, J.B.; Jacky, B.P.S.; Aoki, K.R. Botulinum neurotoxin serotype a specific cell-based potency assay to replace the mouse bioassay. PLoS One 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Kiris, E.; Kota, K.P.; Burnett, J.C.; Soloveva, V.; Kane, C.D.; Bavaria, S. Recent developments in cell-based assays and stem cell technologies for botulinum neurotoxin research and drug discovery. Expert Rev. Mol. Diagn. 2014, 14, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Barash, J.R.; Arnon, S.S. A novel strain of Clostridium botulinum that produces type B and type H botulinum toxins. J. Infect. Dis. 2014, 209, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Dover, N.; Barash, J.R.; Hill, K.K.; Xie, G.; Arnon, S.S. Molecular characterization of a novel botulinum neurotoxin type H gene. J. Infect. Dis. 2014, 209, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Dolly, J.O.; Aoki, K.R. The structure and mode of action of different botulinum toxins. Eur. J. Neurol. 2006, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.; Smith, H.S. Botulinum toxins: Mechanisms of action, antinociception and clinical applications. Toxicology 2013, 306, 124–146. [Google Scholar] [CrossRef] [PubMed]
- Boyer, A.E.; Moura, H.; Woolfitt, A.R.; Kalb, S.R.; McWilliams, L.G.; Pavlopoulos, A.; Schmidt, J.G.; Ashley, D.L.; Barr, J.R. From the mouse to the mass spectrometer: Detection and differentiation of the endoproteinase activities of botulinum neurotoxins A–G by mass spectrometry. Anal. Chem. 2005, 77, 3916–3924. [Google Scholar] [CrossRef] [PubMed]
- Kalb, S.R.; Moura, H.; Boyer, A.E.; McWilliams, L.G.; Pirkle, J.L.; Barr, J.R. The use of Endopep-MS for the detection of botulinum toxins A, B, E, and F in serum and stool samples. Anal. Biochem. 2006, 351, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Ferreira, J.L.; Eblen, B.S.; Whiting, R.C. Detection of type A, B, E, and F Clostridium botulinum neurotoxins in foods by using an amplified enzyme-linked immunosorbent assay with digoxigenin-labeled antibodies. Appl. Environ. Microbiol. 2006, 72, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lou, J.; Jenko, K.L.; Marks, J.D.; Varnum, S.M. Simultaneous and sensitive detection of six serotypes of botulinum neurotoxin using enzyme-linked immunosorbent assay-based protein antibody microarrays. Anal. Biochem. 2012, 430, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Lindström, M.; Keto, R.; Markkula, A.; Nevas, M.; Hielm, S.; Korkeala, H. Multiplex PCR assay for detection and identification of Clostridium botulinum types A, B, E, and F in food and fecal material. Appl. Environ. Microbiol. 2001, 67, 5694–5699. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Xu, M.; Freeman, C.; James, T.; Davis, J.J. Ultrasensitive label free electrical detection of insulin in neat blood serum. Anal. Chem. 2013, 85, 4129–4134. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.F.D.; Norena, N.; Kaushik, A.; Bhansali, S. A low-cost miniaturized potentiostat for point-of-care diagnosis. Biosens. Bioelectron. 2014, 62, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Bryan, T.; Luo, X.; Forsgren, L.; Morozova-Roche, L.A.; Davis, J.J. The robust electrochemical detection of a Parkinson’s disease marker in whole blood sera. Chem. Sci. 2012, 3, 3468–3473. [Google Scholar] [CrossRef]
- Barreiros dos Santos, M.; Agusil, J.P.; Prieto-Simón, B.; Sporer, C.; Teixeira, V.; Samitier, J. Highly sensitive detection of pathogen Escherichia coli O157: H7 by electrochemical impedance spectroscopy. Biosens. Bioelectron. 2013, 45, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.; Guo, J.; Chen, S.; Yang, M. Nanoporous membrane based impedance sensors to detect the enzymatic activity of botulinum neurotoxin A. J. Mater. Chem. B 2013, 1, 6544–6550. [Google Scholar] [CrossRef]
- Bogomolova, A.; Komarova, E.; Reber, K.; Gerasimov, T.; Yavuz, O.; Bhatt, S.; Aldissi, M. Challenges of electrochemical impedance spectroscopy in protein biosensing. Anal. Chem. 2009, 81, 3944–3949. [Google Scholar] [CrossRef] [PubMed]
- Daniels, J.S.; Pourmand, N. Label-free impedance biosensors: Opportunities and challenges. Electroanalysis 2007, 19, 1239–1257. [Google Scholar] [CrossRef] [PubMed]
- Gwenin, C.D.; Halliwell, J. Detection of Botulinum Toxin. UK patent application No. 1310090.4, 2013. [Google Scholar]
- Halliwell, J.; Savage, A.C.; Buckley, N.; Gwenin, C.D. Electrochemical impedance spectroscopy biosensor for detection of active botulinum neurotoxin. Sens. Bio-Sens. Res. 2014, 2, 12–15. [Google Scholar] [CrossRef]
- Schiavo, G.; Santucci, A.; Dasgupta, B.R.; Mehta, P.P.; Jontes, J.; Benfenati, F.; Wilson, M.C.; Montecucco, C. Botulinum neurotoxins serotypes A and E cleave SNAP-25 at distinct COOH-terminal peptide bonds. FEBS Lett. 1993, 335, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, G.; Rossetto, O.; Catsicas, S.; de Laureto, P.P.; DasGupta, B.R.; Benfenati, F.; Montecucco, C. Identification of the nerve terminal targets of botulinum neurotoxin serotypes A, D, and E. J. Biol. Chem. 1993, 268, 23784–23787. [Google Scholar] [PubMed]
- Strong, A.E. Self-assembling chiral monolayers of helical peptides bound to gold via side-chain thioethers. Chem. Commun. 1998, 4, 473–474. [Google Scholar] [CrossRef]
- Yu, J.; Becker, M.L.; Carri, G.A. The influence of amino acid sequence and functionality on the binding process of peptides onto gold surfaces. Langmuir 2012, 28, 1408–1417. [Google Scholar] [CrossRef] [PubMed]
- Hager, G.; Brolo, A.G. Adsorption/desorption behaviour of cysteine and cystine in neutral and basic media: Electrochemical evidence for differing thiol and disulfide adsorption to a Au(1 1 1) single crystal electrode. J. Electroanal. Chem. 2003, 550–551, 291–301. [Google Scholar] [CrossRef]
- Widrig, C.A.; Chung, C.; Porter, M.D. The electrochemical desorption of n-alkanethiol monolayers from polycrystalline Au and Ag electrodes. J. Electroanal. Chem. Interfacial Electrochem. 1991, 310, 335–359. [Google Scholar] [CrossRef]
- Foran, P.; Lawrence, G.W.; Shone, C.C.; Foster, K.A.; Dolly, J.O. Botulinum neurotoxin C1 cleaves both syntaxin and SNAP-25 in intact and permeabilized chromaffin cells: Correlation with its blockade of catecholamine release. Biochemistry 1996, 35, 2630–2636. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, J.; Gwenin, C. A label free colorimetric assay for the detection of active botulinum neurotoxin type A by SNAP-25 conjugated colloidal gold. Toxins 2013, 5, 1381–1391. [Google Scholar] [CrossRef] [PubMed]
- Haiss, W.; Lackey, D.; Sass, J.K.; Besocke, K.H. Atomic resolution scanning tunneling microscopy images of Au(111) surfaces in air and polar organic solvents. J. Chem. Phys. 1991, 95, 2193. [Google Scholar] [CrossRef]
- Park, S.-M.; Yoo, J.-S. Electrochemical impedance spectroscopy for better electrochemical measurements. Anal. Chem. 2003, 75, 455A–461A. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savage, A.C.; Buckley, N.; Halliwell, J.; Gwenin, C. Botulinum Neurotoxin Serotypes Detected by Electrochemical Impedance Spectroscopy. Toxins 2015, 7, 1544-1555. https://doi.org/10.3390/toxins7051544
Savage AC, Buckley N, Halliwell J, Gwenin C. Botulinum Neurotoxin Serotypes Detected by Electrochemical Impedance Spectroscopy. Toxins. 2015; 7(5):1544-1555. https://doi.org/10.3390/toxins7051544
Chicago/Turabian StyleSavage, Alison C., Nicholas Buckley, Jennifer Halliwell, and Christopher Gwenin. 2015. "Botulinum Neurotoxin Serotypes Detected by Electrochemical Impedance Spectroscopy" Toxins 7, no. 5: 1544-1555. https://doi.org/10.3390/toxins7051544
APA StyleSavage, A. C., Buckley, N., Halliwell, J., & Gwenin, C. (2015). Botulinum Neurotoxin Serotypes Detected by Electrochemical Impedance Spectroscopy. Toxins, 7(5), 1544-1555. https://doi.org/10.3390/toxins7051544





