Firing the Sting: Chemically Induced Discharge of Cnidae Reveals Novel Proteins and Peptides from Box Jellyfish (Chironex fleckeri) Venom
Abstract
:1. Introduction
2. Results and Discussion
2.1. Microscopy Examination of Undischarged and Chemically Discharged Nematocysts

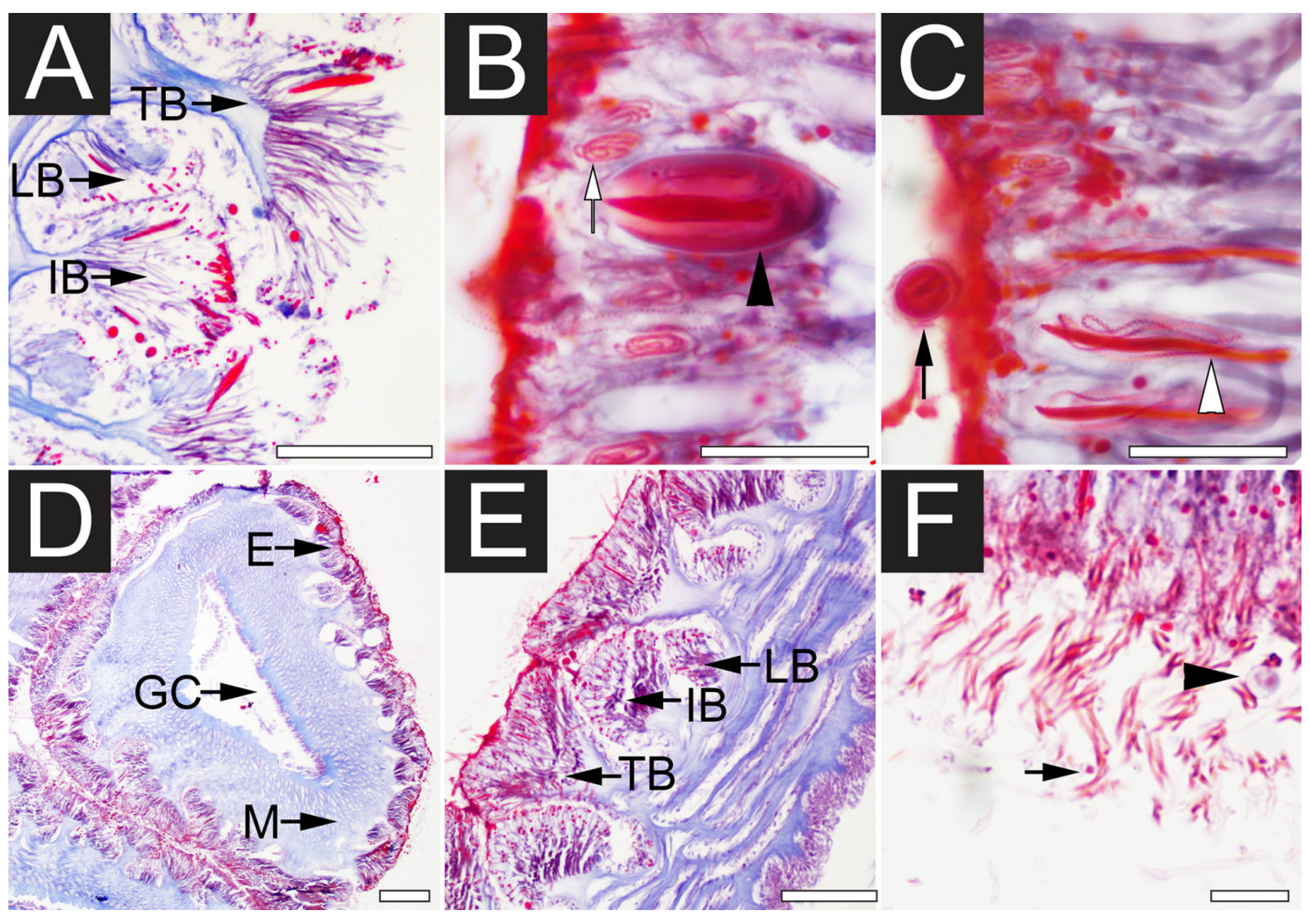
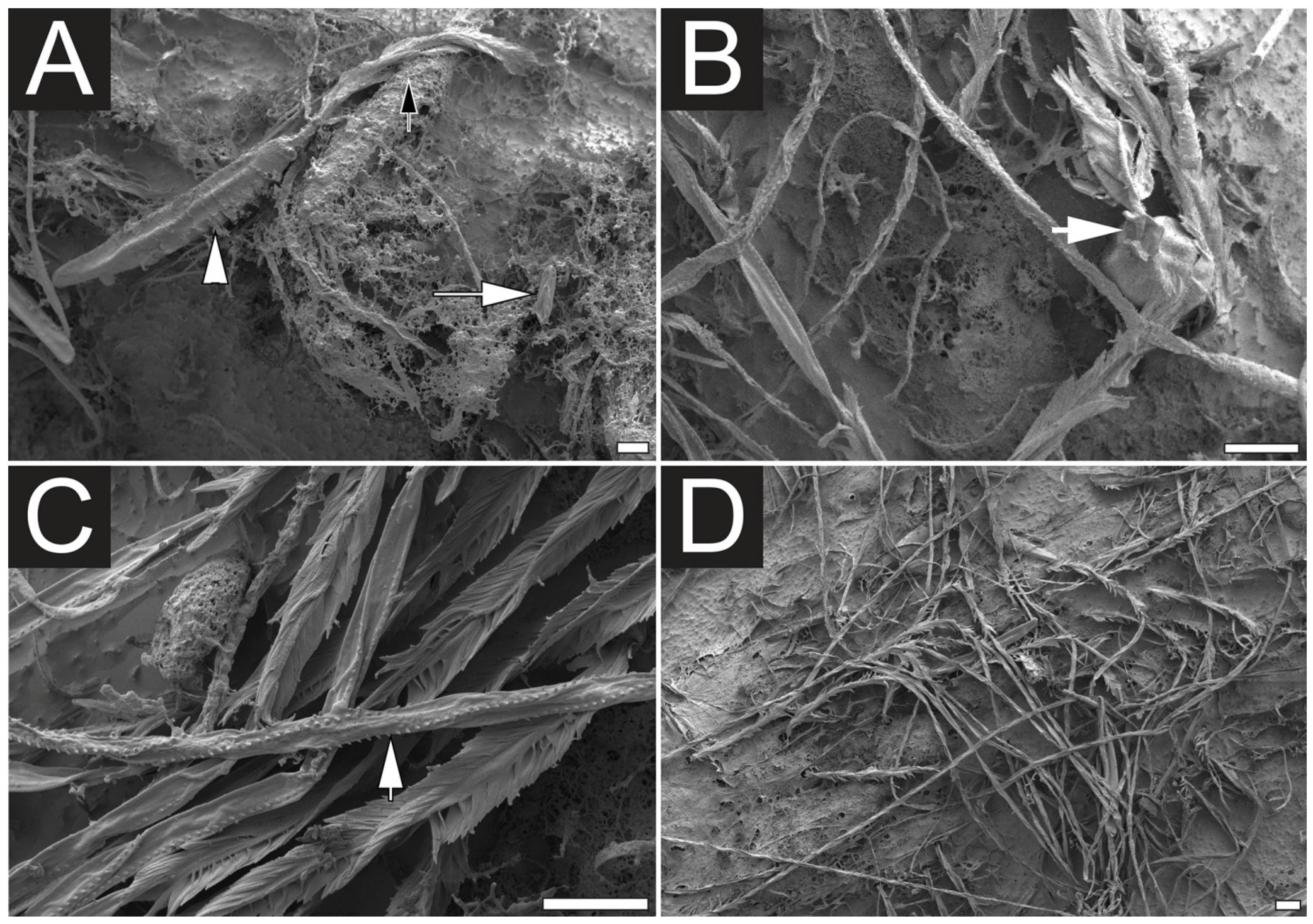
2.2. Transcriptome Assembly and Functional Annotation
2.3. Comparative Proteomic Analyses and Identified Proteins
| Protein ID Transcriptome Match | Uniprot Accession #(s)/Best Cnidaria BLAST Hit | Conserved Domain | Comparative Protein Level a | %Cov (95%) b | Matched Peptides # (%95) c | |||
|---|---|---|---|---|---|---|---|---|
| PDN | CDN | PDN | CDN | PDN | CDN | |||
| Known Cubozoa Toxins | ||||||||
| T0179 | T1PQV6/Toxin B precursor | – | 44 | 6 | 65.11 | 60.15 | 8 | 63 |
| T0362 | T1PRE3/Toxin A precursor | – | 25 | 7 | 29.7 | 55.3 | 12 | 49 |
| T2746 | A7L035/Toxin CfTX-1 precursor | – | 93 | 40 | 45.9 | 77.9 | 3 | 14 |
| T2621 | A7L036/Toxin CfTX-2 precursor | – | Not found | 68 | Not found | 42.4 | Not found | 10 |
| Metalloproteases | ||||||||
| T0344 | A7S336; Predicted protein; Nematostella vectensis | ShKT domain (IPR003582) | 47 | 27 | 16.92 | 23.69 | 6 | 27 |
| T0690 | A7SNJ4; Metalloendopeptidase; Nematostella vectensis | Peptidase M12A, astacin domain (IPR001506); ShKT domain (IPR003582) | 72 | 61 | 17.49 | 45.73 | 4 | 16 |
| T2821 | A7S5S4; Metalloendopeptidase; Nematostella vectensis | Peptidase M12A, astacin domain (IPR001506) | 177 | 77 | 25 | 47.5 | 2 | 12 |
| T1091 | – | Peptidase M12A, astacin domain (IPR001506) | 207 | 189 | 12.7 | 7.7 | 1 | 2 |
| T2460 | A7T0S0; Metalloendopeptidase; Nematostella vectensis | Peptidase M12A, astacin domain (IPR001506) | 138 | 200 | 25.6 | 41 | 3 | 7 |
| Serine Protease inhibitors | ||||||||
| T0134 | A7SCV8; Predicted protein; Nematostella vectensis | Kazal domain (IPR002350) | Not found | 149 | Not found | 5.34 | Not found | 3 |
3. Experimental Section
3.1. Specimen Collection
3.2. Nematocyst Morphology
3.2.1. Cryo-Scanning Electron Microscopy
3.2.2. Histology
3.3. Transcriptome Library Construction
3.4. Venom Extraction
3.4.1. Chemically Induced Discharge of Nematocysts by Ethanol
3.4.2. Pressure Disrupted, Pre-Purified Nematocysts
3.5. Proteomics Methods
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Abbreviation
| PDN | Pressure disrupted nematocysts |
| CDN | chemically discharged nematocysts |
| LC | liquid chromatography |
| MS/MS | tandem MS |
| SPI | serine protease inhibitor |
| NBF | 10% neutral-buffered formalin |
| contig | contiguous |
| PBS | phosphate buffered saline |
Conflicts of Interest
References
- Mariottini, G.L.; Pane, L. Cytotoxic and cytolytic cnidarian venoms. A review on health implications and possible therapeutic applications. Toxins 2014, 6, 108–151. [Google Scholar]
- Suput, D. In vivo effects of cnidarian toxins and venoms. Toxicon 2009, 54, 1190–1200. [Google Scholar] [CrossRef] [PubMed]
- Purcell, J.E. The functions of nematocysts in prey capture by epipelagic siphonophores (Coelenterata, Hydrozoa). Biol. Bull. 1984, 166, 310–327. [Google Scholar] [CrossRef]
- Beckmann, A.; Ozbek, S. The nematocyst: a molecular map of the cnidarian stinging organelle. Int. J. Dev. Biol. 2012, 56, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Tardent, P. The cnidarian cnidocyte, a hightech cellular weaponory. BioEssays 1995, 17, 351–362. [Google Scholar] [CrossRef]
- Burnett, J.W. Treatment of Atlantic cnidarian envenomations. Toxicon 2009, 54, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
- Yanagihara, A.A.; Shohet, R.V. Cubozoan venom-induced cardiovascular collapse is caused by hyperkalemia and prevented by zinc gluconate in mice. PLoS One 2012, 7, e51368. [Google Scholar] [CrossRef] [PubMed]
- Tibballs, J.; Yanagihara, A.A.; Turner, H.C.; Winkel, K. Immunological and toxinological responses to jellyfish stings. Inflamm. Allergy Drug Targets 2011, 10, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Marino, A.; Crupi, R.; Rizzo, G.; Morabito, R.; Musci, G.; La Spada, G. The unusual toxicity and stability properties of crude venom from isolated nematocysts of Pelagia noctiluca (Cnidaria, Scyphozoa). Mol. Cell. Biol. 2007, 53, OL994–OL1002. [Google Scholar]
- Rocha, J.; Peixe, L.; Gomes, N.C.; Calado, R. Cnidarians as a source of new marine bioactive compounds-an overview of the last decade and future steps for bioprospecting. Mar. Drugs 2011, 9, 1860–1886. [Google Scholar] [CrossRef] [PubMed]
- Haefner, B. Drugs from the deep: marine natural products as drug candidates. Drug Discov. Today 2003, 8, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Schwartsmann, G.; Da Rocha, A.B.; Mattei, J.; Lopes, R. Marine-derived anticancer agents in clinical trials. Expert Opin. Investig. Drugs 2003, 12, 1367–1383. [Google Scholar] [CrossRef] [PubMed]
- Mariottini, G.L.; Pane, L. The role of Cnidaria in drug discovery. A review on CNS implications and new perspectives. Recent Pat. CNS Drug Discov. 2013, 8, 110–122. [Google Scholar] [CrossRef]
- Morabito, R.; La Spada, G.; Crupi, R.; Esposito, E.; Marino, A. Crude venom from nematocysts of the jellyfish Pelagia noctiluca as a tool to study cell physiology. Cent. Nerv. Syst. Agents Med. Chem. 2015, in press. [Google Scholar]
- Li, R.; Yu, H.; Xing, R.; Liu, S.; Qing, Y.; Li, K.; Li, B.; Meng, X.; Cui, J.; Li, P. Application of nanoLC-MS/MS to the shotgun proteomic analysis of the nematocyst proteins from jellyfish Stomolophus meleagris. J. Chromatogr. B 2012, 899, 86–95. [Google Scholar] [CrossRef]
- Barnes, J. Extraction of Cnidarian Venom from Living Tentacle; Animal Toxins, Pergamon Press: Oxford, UK, 1967; pp. 115–129. [Google Scholar]
- Turner, R.J.; Freeman, S.E. Effects of Chironex fleckeri toxin on the isolated perfused guinea pig heart. Toxicon 1969, 7, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Keen, T.E. Comparison of tentacle extracts from Chiropsalmus quadrigatus and Chironex fleckeri. Toxicon 1971, 9, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Endean, R.; Duchemin, C.; McColm, D.; Fraser, E.H. A study of the biological activity of toxic material derived from nematocysts of the cubomedusan Chironex fleckeri. Toxicon 1969, 6, 179–204. [Google Scholar] [CrossRef] [PubMed]
- Carrette, T.; Seymour, J. A rapid and repeatable method for venom extraction from cubozoan nematocysts. Toxicon 2004, 44, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Bloom, D.A.; Burnett, J.W.; Alderslade, P. Partial purification of box jellyfish (Chironex fleckeri) nematocyst venom isolated at the beachside. Toxicon 1998, 36, 1075–1085. [Google Scholar] [CrossRef] [PubMed]
- Hartwick, R.; Callanan, V.; Williamson, J. Disarming the box-jellyfish: Nematocyst inhibition in Chironex fleckeri. Med. J. Aust. 1980, 1, 15–20. [Google Scholar] [PubMed]
- Birsa, L.M.; Verity, P.G.; Lee, R.F. Evaluation of the effects of various chemicals on discharge of and pain caused by jellyfish nematocysts. Comp. Biochem. Physiol. C 2010, 151, 426–430. [Google Scholar]
- Rifkin, J.F.; Fenner, P.J.; Williamson, J.A. First aid treatment of the sting from the hydroid Lytocarpus philippinus: The structure of, and in vitro discharge experiments with its nematocysts. J. Wilderness Med. 1993, 4, 252–260. [Google Scholar] [CrossRef]
- Moran, Y.; Genikhovich, G.; Gordon, D.; Wienkoop, S.; Zenkert, C.; Ozbek, S.; Technau, U.; Gurevitz, M. Neurotoxin localization to ectodermal gland cells uncovers an alternative mechanism of venom delivery in sea anemones. Proc. Biol. Sci. 2012, 279, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Rifkin, J.F.; Burnett, J.W.; Fenner, P.J. Venomous and poisonous marine animals: A Medical and Biological Handbook; NewSouth Publishing: Sydney, Australia, 1996. [Google Scholar]
- Gershwin, L. Nematocysts of the Cubozoa. Zootaxa 2006, 1232, 1–57. [Google Scholar]
- Carrette, T.; Alderslade, P.; Seymour, J. Nematocyst ratio and prey in two Australian cubomedusans, Chironex fleckeri and Chiropsalmus sp. Toxicon 2002, 40, 1547–1551. [Google Scholar] [CrossRef] [PubMed]
- Morabito, R.; Marino, A.; Dossena, S.; La Spada, G. Nematocyst discharge in Pelagia noctiluca (Cnidaria, Scyphozoa) oral arms can be affected by lidocaine, ethanol, ammonia and acetic acid. Toxicon 2014, 83, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Brinkman, D.L.; Konstantakopoulos, N.; McInerney, B.V.; Mulvenna, J.; Seymour, J.E.; Isbister, G.K.; Hodgson, W.C. Chironex fleckeri (Box Jellyfish) Venom Proteins: Expansion of a cnidarian toxin family that elicits variable cytolytic and cardiovascular effects. J. Biol. Chem. 2014, 289, 4798–4812. [Google Scholar] [CrossRef]
- Rangaraju, S.; Khoo, K.K.; Feng, Z.P.; Crossley, G.; Nugent, D.; Khaytin, I.; Chi, V.; Pham, C.; Calabresi, P.; Pennington, M.W.; Norton, R.S.; Chandy, K.G. Potassium channel modulation by a toxin domain in matrix metalloprotease 23. J. Biol. Chem. 2010, 285, 9124–9136. [Google Scholar] [CrossRef] [PubMed]
- Da Silveira, R.; Wille, A.; Chaim, O.; Appel, M.; Silva, D.; Franco, C.; Toma, L.; Mangili, O.; Gremski, W.; Dietrich, C. Identification, cloning, expression and functional characterization of an astacin-like metalloprotease toxin from Loxosceles intermedia (brown spider) venom. Biochem. J 2007, 406, 355–363. [Google Scholar]
- Undheim, E.A.; Jones, A.; Clauser, K.R.; Holland, J.W.; Pineda, S.S.; King, G.F.; Fry, B.G. Clawing through evolution: toxin diversification and convergence in the ancient lineage Chilopoda (Centipedes). J. Mol. Biol. 2014. [Google Scholar] [CrossRef]
- Li, R.; Yu, H.; Xue, W.; Yue, Y.; Liu, S.; Xing, R.; Li, P. Jellyfish venomics and venom gland transcriptomics analysis of Stomolophus meleagris to reveal the toxins associated with sting. J. Proteomics 2014, 106, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Moran, Y.; Praher, D.; Schlesinger, A.; Ayalon, A.; Tal, Y.; Technau, U. Analysis of soluble protein contents from the nematocysts of a model sea anemone sheds light on venom evolution. Mar. Biotechnol. 2013, 15, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Gómez, F.; Ortíz-Pineda, P.A.; Rojas-Cartagena, C.; Suárez-Castillo, E.C.; García-Ararrás, J.E. Immune-related genes associated with intestinal tissue in the sea cucumber Holothuria glaberrima. Immunogenetics 2008, 60, 57–71. [Google Scholar] [CrossRef] [PubMed]
- Garvey, W. Modified elastic tissue-Masson trichrome stain. Stain Technol. 1984, 59, 213–216. [Google Scholar] [PubMed]
- Archer, J.; Whiteley, G.; Casewell, N.R.; Harrison, R.A.; Wagstaff, S.C. VTBuilder: A tool for the assembly of multi isoform transcriptomes. Bioinformatics 2014, 15, 389. [Google Scholar] [PubMed]
- Gotz, S.; Garcia-Gomez, J.M.; Terol, J.; Williams, T.D.; Nagaraj, S.H.; Nueda, M.J.; Robles, M.; Talon, M.; Dopazo, J.; Conesa, A. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res. 2008, 36, 3420–3435. [Google Scholar] [CrossRef] [PubMed]
- Conesa, A.; Gotz, S.; Garcia-Gomez, J.M.; Terol, J.; Talon, M.; Robles, M. Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 2005, 21, 3674–3676. [Google Scholar] [CrossRef] [PubMed]
- Emanuelsson, O.; Brunak, S.; von Heijne, G.; Nielsen, H. Locating proteins in the cell using TargetP, SignalP and related tools. Nat. Protoc. 2007, 2, 953–971. [Google Scholar] [CrossRef] [PubMed]
- Zdobnov, E.M.; Apweiler, R. InterProScan–an integration platform for the signature-recognition methods in InterPro. Bioinformatics 2001, 17, 847–848. [Google Scholar] [CrossRef] [PubMed]
- Kappler, U.; Nouwens, A.S. The molybdoproteome of Starkeya novella–insights into the diversity and functions of molybdenum containing proteins in response to changing growth conditions. Metallomics 2013, 5, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Sousa, L.F.; Nicolau, C.A.; Peixoto, P.S.; Bernardoni, J.L.; Oliveira, S.S.; Portes-Junior, J.A.; Mourao, R.H.; Lima-dos-Santos, I.; Sano-Martins, I.S.; Chalkidis, H.M.; et al. Comparison of phylogeny, venom composition and neutralization by antivenom in diverse species of bothrops complex. PLoS Negl. Trop. Dis. 2013, 7, e2442. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jouiaei, M.; Casewell, N.R.; Yanagihara, A.A.; Nouwens, A.; Cribb, B.W.; Whitehead, D.; Jackson, T.N.W.; Ali, S.A.; Wagstaff, S.C.; Koludarov, I.; et al. Firing the Sting: Chemically Induced Discharge of Cnidae Reveals Novel Proteins and Peptides from Box Jellyfish (Chironex fleckeri) Venom. Toxins 2015, 7, 936-950. https://doi.org/10.3390/toxins7030936
Jouiaei M, Casewell NR, Yanagihara AA, Nouwens A, Cribb BW, Whitehead D, Jackson TNW, Ali SA, Wagstaff SC, Koludarov I, et al. Firing the Sting: Chemically Induced Discharge of Cnidae Reveals Novel Proteins and Peptides from Box Jellyfish (Chironex fleckeri) Venom. Toxins. 2015; 7(3):936-950. https://doi.org/10.3390/toxins7030936
Chicago/Turabian StyleJouiaei, Mahdokht, Nicholas R. Casewell, Angel A. Yanagihara, Amanda Nouwens, Bronwen W. Cribb, Darryl Whitehead, Timothy N. W. Jackson, Syed A. Ali, Simon C. Wagstaff, Ivan Koludarov, and et al. 2015. "Firing the Sting: Chemically Induced Discharge of Cnidae Reveals Novel Proteins and Peptides from Box Jellyfish (Chironex fleckeri) Venom" Toxins 7, no. 3: 936-950. https://doi.org/10.3390/toxins7030936
APA StyleJouiaei, M., Casewell, N. R., Yanagihara, A. A., Nouwens, A., Cribb, B. W., Whitehead, D., Jackson, T. N. W., Ali, S. A., Wagstaff, S. C., Koludarov, I., Alewood, P., Hansen, J., & Fry, B. G. (2015). Firing the Sting: Chemically Induced Discharge of Cnidae Reveals Novel Proteins and Peptides from Box Jellyfish (Chironex fleckeri) Venom. Toxins, 7(3), 936-950. https://doi.org/10.3390/toxins7030936







