Abstract
The development of fungal endophytes of the genus Epichloë in grasses results in the production of different groups of alkaloids, whose mechanism and biological spectrum of toxicity can differ considerably. Ergot alkaloids, when present in endophyte-infected tall fescue, are responsible for “fescue toxicosis” in livestock, whereas indole-diterpene alkaloids, when present in endophyte-infected ryegrass, are responsible for “ryegrass staggers”. In contrast, peramine and loline alkaloids are deterrent and/or toxic to insects. Other toxic effects in livestock associated with the consumption of endophyte-infected grass that contain ergot alkaloids include the “sleepy grass” and “drunken horse grass” diseases. Although ergovaline is the main ergopeptine alkaloid produced in endophyte-infected tall fescue and is recognized as responsible for fescue toxicosis, a number of questions still exist concerning the profile of alkaloid production in tall fescue and the worldwide distribution of tall fescue toxicosis. The purpose of this review is to present ergot alkaloids produced in endophyte-infected grass, the factors of variation of their level in plants, and the diseases observed in the mammalian species as relate to the profiles of alkaloid production. In the final section, interactions between ergot alkaloids and drug-metabolizing enzymes are presented as mechanisms that could contribute to toxicity.
1. Diseases in Livestock Observed after Consumption of Plants Infected by Endophytic Fungi of the Genus Epichloë
Ergot alkaloids are present in various grasses infected by endophytic fungi of the genus Epichloë (Table 1) (Note that with a recent taxonomic revision [1] of the genus Epichloë has been expanded to include the asexual Neotyphodium species known to be derived from sexual Epichloë species). The first hypotheses concerning the involvement of ergot alkaloids in livestock health problems that were not linked to the consumption of sclerotia (ergots) of Claviceps species was proposed in the 1940s for symptoms observed in cattle fed with tall fescue grass. The description of the disease incudes signs of lameness in winter, usually in the left hind foot, which can lead to loss of the foot [2]. Because the disease occurred in winter, whereas mature Claviceps sclerotia are present in the plant in late spring, and because typical signs of toxicity were observed even when the cattle were fed hay that contained no seeds, it was concluded that: “the tall fescue contained some poisonous principle which will cause lameness or shedding of feet similar in all respects to the results from feeding ergot” [2]. After this first description, “fescue foot” was reported in several locations including in Colorado in cattle fed “giant” fescue [3], and in Kentucky in cattle grazing on “Kentucky 31” pastures [4]. Next, the vasoconstrictive properties of extracts of a tall fescue grass obtained from a farm where signs of lameness occurred were demonstrated [5]. The symptoms were then experimentally reproduced in steers fed tall fescue hay [6]. Analysis of the extract of grass revealed the presence of compounds similar to those obtained with extracts of ergots formed by Claviceps purpurea on rye, but no ergots were detected by visual examination of the grass [7]. A review of toxicity of tall fescue forage describing the occurrence and severity of fescue foot in several countries pointed to high variability both in the severity and frequency of the syndrome [8].
In the 1970s, several studies were conducted to characterize fescue toxicosis. Changes in blood flow were recognized as being an important mechanism of action of the toxic compounds present in the extract of toxic tall fescue [9]. A “summer slump syndrome” occurring in summer and resulting in decreased performance, was characterized [10]. Also, “fat necrosis” was reported in beef cattle grazing fertilized Kentucky 31 tall fescue [11]. Although analysis of plant extracts provided conflicting results regarding the etiologic agent, the systemic endophyte, Epichloë coenophiala = Neotyphodium coenophialum, was isolated from tall fescue [12]. Steers fed tall fescue hay infested with E. coenophiala showed elevated temperature and decreased performances compared with steers fed hay that was not infested [13]. In 1985, the presence of ergopeptine alkaloids was demonstrated in toxic Kentucky 31 endophyte-infected tall fescue, ergovaline being the most abundant [14]. Ergopeptines extracted from infected tall fescue constricted the dorsal pedal vein of cattle, whereas loline and loline-derivative alkaloids did not [15]. Ergovaline appeared to be the most abundant and the most potent of the ergopeptide alkaloids (see below) and most studies on “fescue toxicosis” reported the ergovaline level in the feed. The toxic threshold of ergovaline was 300−500 µg/kg feed, and cattle appeared to be more sensitive than sheep [16,17].

Table 1.
Plants infected by endophytic fungi of the genus Epichloë known for their toxicity in livestock and horses, and the major alkaloids thought to be responsible.
| Grass: Common name | Grass: Latin name | Endophyte | Syndromes or symptoms in grazing livestock | Major alkaloids affecting livestock |
|---|---|---|---|---|
| Tall fescue | Lolium arundinaceum (= Schedonorus arundinaceus = Festuca arundinacea) | Epichloë coenophiala (= Neotyphodium coenophialum = Acremonium coenophialum) | Fescue toxicosis, Fescue foot, Summer slump, Fat necrosis 1 | Ergovaline 2 |
| Perennial ryegrass | L. perenne | E. festucae var. lolii (= N. lolii = A. lolii) | Ryegrass staggers | Lolitrem B 3, ergovaline |
| Perennial ryegrass | L. perenne | E. festucae var. lolii x E. typhina | Ergot alkaloid toxicity | Ergovaline |
| Fine fescues | Festuca spp. | E. festucae | Grazing deterrence | Lolitrem B 3, ergovaline |
| Drunken horse grass | Achnatherum inebrians (= Stipa inebrians) | E. gansuensis var. inebrians (= N. gansuense var. inebrians) | Stupor | Ergonovine, ergine |
| Sleepy grass | Ach. robustum (= S. robusta) | Epichloë sp. | Stupor | Ergonovine, ergine |
| Sleepy grass 4 | Ach. robustum | E. funkii (= N. funkii) | None reported | Chanoclavine I |
1: External temperatures play a role in the symptom of the disease: fescue foot is observed in cold winter whereas summer slump syndrome occurs in summer. Fat necrosis is less documented; 2: Ergovaline represents more than 80% of ergopeptides in forage grass and more than 50% in seeds. Ergotamine, ergosine, ergocryptine, ergocornine and ergocristine were reported in seeds due to contamination of plant material by Claviceps sclerotia (ergots) [18]; 3: Lolitrem B and related indole-diterpene alkaloids are recognized as responsible for staggers, whereas the toxic role of ergovaline in endophyte-infected ryegrass is less documented; 4 Though generally called sleepy grass, Ach. robustum plants with E. funkii do not have the toxic effects as those with the other (so far unnamed) Epichloë species found near Cloudcroft, New Mexico, USA [19].
Perennial ryegrass has been recognized as responsible for staggers in livestock [20], and endophytic fungi have been described in perennial ryegrass [21]. However, the sclerotia of Claviceps purpurea were considered to be causative agents of ryegrass staggers until it was demonstrated that the symptoms cannot be reproduced by feeding sclerotia present in the seed heads [22], and staggers were observed in sheep grazing the base of the ryegrass plant, whereas no signs were observed in sheep that were prevented from grazing the base of the plant [23]. Several tremorgenic mycotoxins of different fungal origin were suspected to be the causative agent of ryegrass staggers until the isolation of lolitrem B [24,25], which appeared to be the main tremorgenic mycotoxin in Epichloë festucae var. lolii = Neotyphodium lolii-infected perennial ryegrass [26]. Ergot alkaloids were also found in these plants, but symptoms of ergot alkaloid toxicity are rarely reported in livestock fed endophyte-infected ryegrass [27]. Because ergovaline often represents 10% to 15% of lolitrem B concentrations [28,29,30,31], it is generally accepted that toxic levels of lolitrem B are reached before toxic levels of ergovaline. For this reason, most studies on endophyte-infected ryegrass has focused on lolitrem B, and little information is available on ergot alkaloids [16,17,32,33], and in particular, nothing is known about possible interactions between ergovaline and lolitrem B.
Because infection of plants by the Epichloë species leads to alkaloid production and toxicity in several animal species, a simple solution to avoid toxicity could be to eliminate the endophytes from the grasses sown. However, the mutualistic relationship that exists between the fungal endophytes and their hosts also has several beneficial effects regarding resistance to insects and nematodes, and grass production under stressing conditions [34,35]. Epichloë-infected perennial ryegrasses unable to produce lolitrem B were first commercialized under the trade name “Endosafe”. Unfortunately, the use of some of these early cultivars led to ergovaline toxicity in grazing animals. Signs of ergovaline toxicity were unexpected because they had not previously been observed on pastures sown with common endophyte strains, perhaps because symptoms of ergotism had been masked by the signs of staggers [36]. Interestingly, insect feeding studies done with Epichloë-infected perennial ryegrass unable to produce ergovaline have demonstrated that this alkaloid was not necessary for deterrence properties [37]. Following these observations, the protocol for evaluation of non-toxic endophyte has changed and the absence of ergot alkaloid production was added to the selection criteria [34,35].
Achnatherum inebrians (drunken horse grass; Table 1), associated with rangeland degradation in northern China, has been found to be infected by an Epichloë gansuensis var. inebrians which produces ergot alkaloids of the simple lysergic acid amide type [38]. Similar alkaloid profiles are observed in some populations of sleepy grass (Achnatherum robustum), particularly near Cloudcroft, New Mexico, in the southwestern USA. Livestock and horses that graze these grasses show a profoundly somnolent or stuporous condition [39]. Agrostis, Bromus, Elymus, Hordeum and Poa species sometimes also contain ergot alkaloids when they are infected by fungal endophytes, but little is known about their toxicity [32,33,40]. In conclusion, a large number of endophyte-infected plants contain ergot alkaloids, but only a few of these associations are known for their toxicity in livestock and horses. The signs of toxicity strongly vary, probably because of the different profiles of alkaloid production.
2. Ergots Alkaloids in Plants
Since the discovery of fescue toxicosis, several experimental studies have been conducted to reproduce the disease. In a great number of cases, supplying endophyte-infected tall fescue forage alone failed to induce signs of toxicity, so seeds were added to the feed to increase the ergovaline level [17]. However, adding large amounts of seeds to the feed introduces doubts about the alkaloids responsible for toxicity (see below). At least in some studies, the presence of sclerotia in the seeds was suspected. In addition, the respective percentages of the different alkaloids, especially ergovaline and ergotamine, are not the same in forage plants and seeds, some ergot alkaloids being found only in the seeds [33,41].
The different toxicity of forage used to reproduce fescue toxicosis can also be explained by the stage of maturity of the plant. Indeed, the level of ergovaline in the leaves, stem and inflorescence differs significantly and the stage of maturity of the plant has a major influence on the concentration of ergovaline [42,43,44,45,46]. Three peaks of ergovaline were identified in endophyte-infected tall fescue and endophyte-infected perennial ryegrass in France: the end of spring, the beginning of fall, and mid-winter. In spring, cumulative degree-days of 900 were needed before an increase in ergovaline level occurred both in tall fescue and perennial ryegrass [45,46]. A twofold increase in ergovaline content was observed in a period of only one week at the end of flowering [44,45,46].
Marked variations in the ratio between ergovaline and other alkaloids were also observed in Epichloë festucae var. lolii-infected perennial ryegrass. A study conducted in endophyte-infected ryegrass straw in Oregon has shown that the concentration of lolitrem B was around tenfold higher than that of ergovaline [29]. By contrast, studies conducted in Europe on wild endophyte-infected ryegrass revealed that the concentration of ergovaline was often higher than the concentration of lolitrem B [47,48]. Genetic factors partly explain these differences, whereby some ecotypes of Epichloë festucae var. lolii are able to synthesize ergovaline alone, whereas others synthesize lolitrem B alone, but most produce both toxins [47,48,49,50,51]. Also, the environmental conditions during plant growth seem to have a strong influence on the ratio between ergovaline and lolitrem B [46,52,53]. Comparison of New Zealand and German ecotypes have revealed that the environmental conditions during plant growth have a stronger influence on lolitrem B concentrations, than the origin of the ecotype [52]. Similarly, major differences in the concentrations of the ergot alkaloids, ergine and ergonovine, were observed in endophyte-infected drunken horse grass depending on the part of the plant analyzed and stage of maturity [54], whereas temporal variations in alkaloid level was seen in sleepy grass [55].
The levels of ergot alkaloids in plants also vary with several abiotic factors such as temperature, rainfall, fertilization and atmospheric carbon dioxide. Different results were observed depending on the conditions of experimentation and possible interactions between the factors studied. Most of the studies were conducted on endophyte-infected tall fescue and have focused on the effect of nitrogen fertilization. Some studies revealed an increase in ergovaline concentrations when nitrogen fertilization was done, but this effect varied with the dose, the kind of fertilizer and the year of the assay [42,43,44]. The effects of nitrogen fertilization also seem to vary with the part of the plant analyzed, an increase of ergovaline level being observed in the leaves but not in the inflorescence [45]. No correlation was observed between nitrogen input on pastures and ergovaline concentration in straw [56]. Also, the effect of nitrogen input on alkaloid concentrations could vary depending on the cultivar and the alkaloid assayed [57]. An input of phosphorus also increased the level of ergot alkaloids in the plants, but, again, the effect varied with the level of incorporation and the genotype of the endophyte [58]. Analysis of the effect of climate changes on ergot alkaloid contents in endophyte-infected tall fescue revealed that high CO2 level decreased ergovaline contents, whereas precipitation had no effect [59]. Also, whereas the levels of alkaloids in the plants showed strong variations depending on the year of analysis, the effect of warming was always the same: it increased ergovaline and total alkaloid levels in fall but had no effect in spring [60].
3. Worldwide Distribution
Another question concerns the worldwide distribution of diseases due to ergot alkaloids in endophyte-infected plants. Epichloë have been reported in cool season grasses on all continents except Antarctica, but the geographic distribution of cases of toxicity appears to be limited. The distribution of the endophyte-infected plants or the strains of Epichloë responsible for the infection explain in part these differences. Achnatherum robustum, is limited to southwestern USA. In New Mexico, levels of alkaloids (ergonovine, lysergic acid amide, and isolysergic amide) in sleepy grass populations declined with distance from the Cloudcroft population known for its high toxicity, although Epichloë-infection levels increased [55]. This suggests that not all endophyte-infected Achnatherum robustum have these alkaloids, as demonstrated in northern New Mexico and southern Colorado where no plants were found to contain ergot alkaloids despite 100% infection [55]. A similar toxicity is observed in animals grazing on endophyte-infected Achnatherum inebrians pastures in China. The disease is mainly distributed throughout arid, semi-arid, alpine and subalpine native grasslands of some regions in China, Mongolia and Tibet [54]. High levels of ergonovine and ergine were always measured in plants in areas where the disease was observed [38,54].
By contrast, the worldwide distribution of fescue toxicosis is more difficult to explain. Most cases of toxicity are reported in Australia, USA and New Zealand. In Europe, despite high infection rates of tall fescue by Epichloë coenophiala and high levels of ergovaline in plants, only a few cases of toxicity have been reported [61,62,63,64]. Several hypotheses have been proposed to explain these differences. The first concerns the exposure of animals, which is higher in countries where endophyte-infected tall fescue is sown as a monocrop. However, it seems unlikely that no animal is exposed to high endophyte-infected grass in Europe, as demonstrated in the two cases of fescue toxicosis reported in France [61]. The second hypothesis concerns the level of ergot alkaloid in the grass, which may be lower in some regions than in others. These differences could be due to farming practices or climatic factors, which are known to influence the level of ergot alkaloids in whole plants. Studies conducted in endophyte-infected tall fescue in France revealed that an ergovaline level of 300 µg/kg dry matter and more in the whole plant only lasted four weeks, from complete emergence of the inflorescence until the fully ripe stage [45]. The levels of ergovaline in whole plants was lower than levels reported in Georgia, Missouri and Oregon, but the difference was slight [43,44,56]. The third hypothesis concerns the animals. Most studies on tall fescue toxicity report the ergovaline level in feed, but little information is available concerning the amount ingested per kg of body weight, whereas feed intake varies considerably depending on the physiological stage and the animal strain [65]. Genetic diversity has also been proposed to explain some of the differences between strains in their sensitivity to toxicity [65,66].
4. Ergot Alkaloids, Not Only Ergovaline
Ergovaline was historically recognized as the main ergopeptide alkaloid, produced by Epichloë in tall fescue, and was thought primarily responsible for vasoconstriction and reduced plasma prolactin [67,68]. Most studies on fescue toxicosis have reported the ergovaline level in the feed [17]. However, ergovaline is not the only ergot alkaloid produced, and additive toxicity of other toxins or metabolites is suspected. In a study conducted in sheep using pure ergovaline, the toxin was administered in the feed at a level similar to the one obtained by incorporation of 10% of endophyte-infected tall fescue seeds in the diet [69]. Feed intake, skin temperature, and prolactin were measured as parameters of toxicity. The diet supplemented with the endophyte-infected seeds was more toxic than the diet supplemented with pure ergovaline, suggesting that other compounds present in the seeds have additive toxicity. In another experiment, two diets that contained similar levels of ergovaline, one with seeds of endophyte-infected tall fescue and the other with seeds of endophyte-infected perennial ryegrass, were compared [69]. Again, the toxic effects were more severe in the diet that contained seeds of tall fescue than in the diet that contained ryegrass seeds.
Biosynthetic pathways and genes for ergot alkaloid synthesis in Epichloë species have been extensively reviewed in [70,71]. Synthesis of ergot alkaloids begins by the prenylation of l-tryptophan to obtain dimethylallyltryptophan (Figure 1). N-methylation and C-oxidation of dimethylallyltryptophan followed by intramolecular rearrangement leads to chanoclavine (1), which can be measured in endophyte-infected Achnatherum robustum and has weak binding properties to the dopamine receptors in the brain and no pharmacological effects [19,39,72]. Oxidoreduction of chanoclavine is necessary to obtain agroclavine (2). This compound has antimicrobial and cytostatic properties and is a weak dopaminergic agonist [73,74]. Oxidation of agroclavine leads to the formation of elymoclavine, which is more potent than agroclavine in the prolactin reduction assay [73], and then oxidation of elymoclavine leads to d-lysergic acid (3). d-lysergic acid has been identified in different endophyte-infected plants and in animals fed with endophyte-infected plants [75,76,77]. However, in contrast to observations made on other ergot alkaloids, this compound presented low vasoconstrictor properties in the saphenous vein bioassay and in the right ruminal artery and vein bioassay [78,79,80]. Different amide derivatives of d-lysergic acid (ergopeptines) have been characterized in endophyte-infected plants. Simple amide derivatives are ergoamides whereas more complex derivatives obtained with amino acids are ergopeptines.
Ergine (4, d-lysergic acid amide) and ergonovine (5, d-lysergic acid ethanol amide, also known as ergometrine or ergobasine) are found in endophyte-infected Ach. inebrians [38]. However, E. gansuensis var. inebrians seems unable to produce ergopeptides [51]. Ergine has psychotropic properties whereas ergonovine also has vasoconstrictor properties and reduces the concentration of prolactin, but is less potent than ergotamine [74,81]. In another study, ergonovine did not reduce prolactin and was unable to cause symptoms of fescue toxicosis [82]. Comparison of the vasomotor properties of ergopeptines in the saphenous vein bioassay and in the right ruminal artery and vein ruminal bioassay generally revealed that ergoamides were less potent than ergopeptines, and that their effect was less persistent [79,80,83,84,85]. Also, ergine and ergonovine were less potent than ergovaline in binding to D2-dopamine receptors in cell culture [86]. d-lysergic acid is used for the semi-synthesis of d-lysergic acid diethylamide LSD, which is the most potent hallucinogenic substance known [38,87]. Other effects of LSD include increased arterial blood pressure and reduced prolactin.
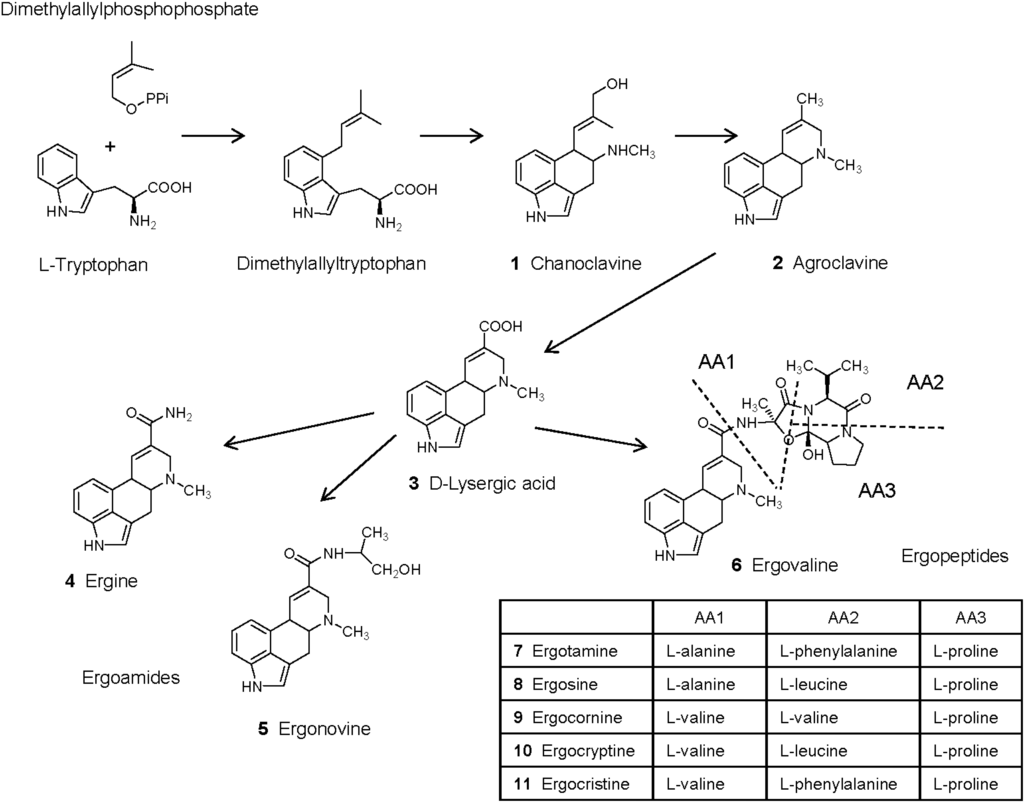
Figure 1.
Ergot alkaloids produced by endophytic fungi of the genus Epichloë in infected plants (adapted from Shardl et al. [70]. Copyright 2006, Elsevier). Ergotamine, ergosine, ergocornine, ergocryptine and ergocristine are additional ergopeptines commonly found in ergots of Claviceps purpurea but not Epichloë species.
Figure 1.
Ergot alkaloids produced by endophytic fungi of the genus Epichloë in infected plants (adapted from Shardl et al. [70]. Copyright 2006, Elsevier). Ergotamine, ergosine, ergocornine, ergocryptine and ergocristine are additional ergopeptines commonly found in ergots of Claviceps purpurea but not Epichloë species.
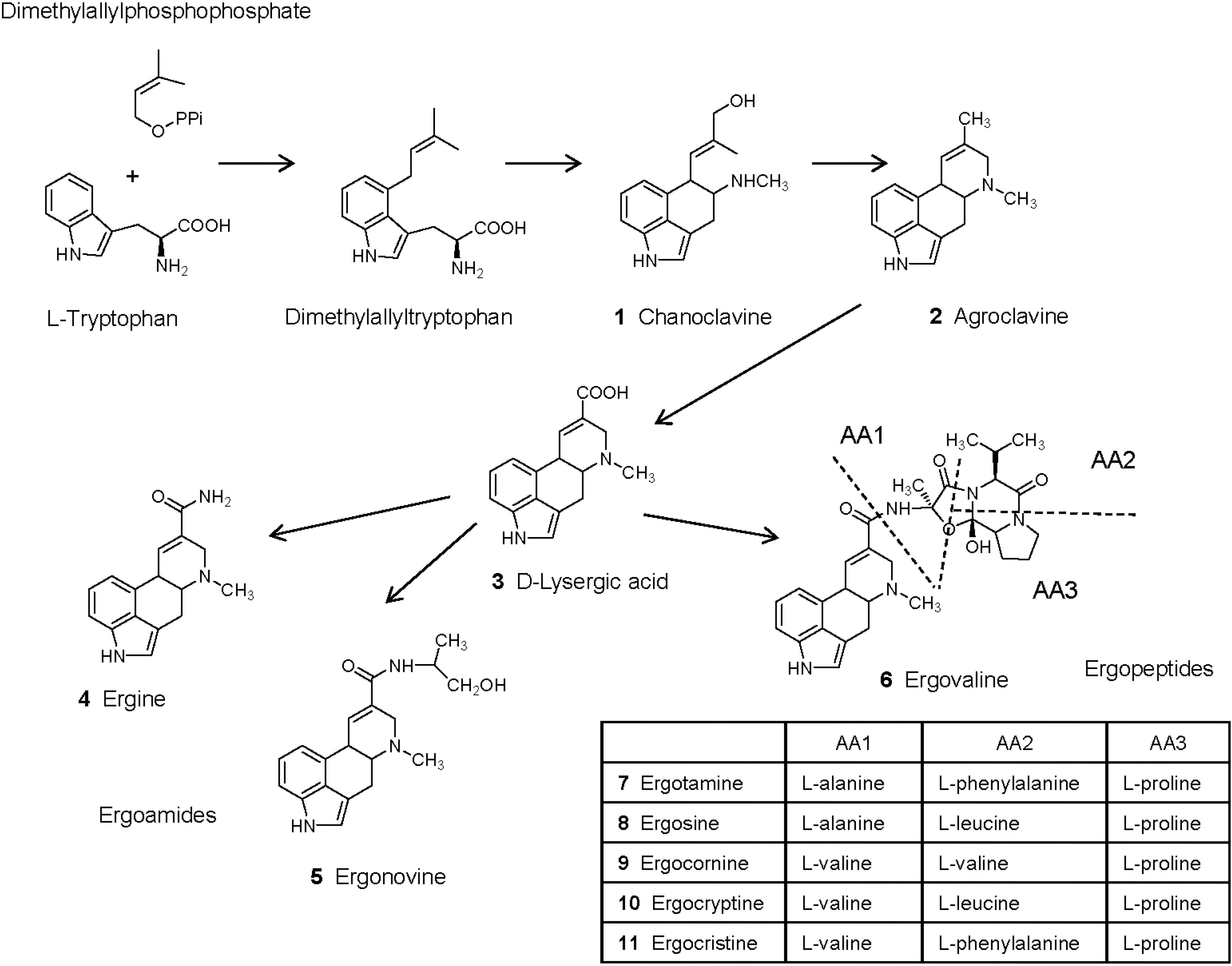
The most complex amide derivatives of d-lysergic acid are ergopeptines that are formed with amino acids. Their synthesis involves a two subunit nonribosomal peptide synthetase (lysergyl peptide synthetase subunits LPS1 and LPS2), and sequence variations in LPS1 determine which amino acids are incorporated. Condensation of d-lysergic acid activated by LPS2 with l-alanine, l-valine and l-proline (sequentially added by LPS1) leads to the formation of ergovaline (6). Other ergopeptines produced by Claviceps include ergotamine (7), ergosine (8), ergocornine (9), ergocryptine (10), ergocristine (11) [70]. These compounds may be present in some samples of endophyte-infected tall fescue [88], probably due to the contamination of plant material by Claviceps sclerotia [18]. Analysis of endophyte-infected samples of tall fescue revealed that ergovaline represented 80%–100% of ergopeptines analyzed in forage grasses and more than 50% in seeds [33]. Ergosine and ergotamine were the other ergot alkaloids found in forage, representing 0%–12% and 0%–6% of the ergopeptines analyzed, respectively [33]. Ergosine, ergotamine and ergocryptine were found in seeds, representing 15%–40% of the ergopeptines analyzed [33,41]. Ergovaline, ergotamine, ergocornine, ergocryptine, and ergocristine triggered contractile responses in the saphenous vein bioassay and in the right ruminal artery and vein bioassay. Ergovaline and ergotamine are generally the most potent, both in terms of intensity and duration of the effect [79,80,83,84,85]. Comparison of the contractile properties of extracts of endophyte-infected tall fescue with ergovaline in these models revealed differential responses to ergot alkaloids in peripheral vasculature and core vasculature [80,85]. Ergovaline, ergotamine, and ergocryptine presented similar binding properties to D2-dopamine receptors in cell culture [86].
Comparisons of the toxic properties of ergot alkaloids should also take in consideration the way they reach their target. Interspecies comparisons of fescue toxicosis revealed that horses are more resistant than cattle and sheep, and that high levels of ergovaline in feed are needed to produce the disease in rodents [17]. Even if several factors explain these differences, pre-hepatic metabolism of alkaloids in the rumen likely influences their toxicity. High metabolism of ergot alkaloids was described in the rumen, and d-lysergic acid was the main metabolite formed [76,77]. However, d-lysergic acid was shown to have less vasomotor activity than ergopetines in several bioassays, and d-lysergic acid was found to be the main metabolite of ergot alkaloids in horses [75]. On the other hand, studies conducted in a parabiotic chamber revealed that the transport mechanism of ergot alkaloids across digestive barriers is an active process, and that this process is more potent via the rumen than via the abomasum [89]. Interestingly, d-lysergic acid and ergonovine had higher absorption potential than ergopeptides, suggesting toxicokinetic differences between ergot alkaloids with possible consequences for toxicity.
Finally, although several ergot alkaloids are present in Epichloë-infected plants, it appears that ergoamide derivatives are mainly involved in psychotropic effects, whereas ergopetines are responsible for vasoconstriction and the reduction in prolactin. Comparison of the properties of ergopeptines in different models suggests that ergovaline and ergotamine have the most potent vasomotor properties, and the highest affinity for D2-dopaminergic receptors. Only a few data are available concerning the precursors of d-lysergic acid. Comparison of chanoclavine and agroclavine suggested that the presence of hydroxyl or carbonyl function in the proximate environment of the N-methyl of l-tryptophan is necessary to bind to the amine receptor. Oxidation to the carboxylate derivate that leads to d-lysergic acid decreases the binding properties to the biogenic amine receptors.
5. Other Effects of Ergot Alkaloids
Because the ergot alkaloids found in Epichloë-infected grasses are related to those found in sclerotia (ergots) on florets infected with the Claviceps species, most studies of their toxic effects have focused on their pharmacological properties and binding to biogenic amine receptors. However, other mechanisms of action could explain some of the unexpected results observed in fescue toxicosis. Among these mechanisms, interaction of ergot alkaloids with drug-metabolizing enzymes has been suspected. The involvement of drug-metabolizing enzymes in ergot alkaloid metabolism was demonstrated with bromocriptine, a drug synthesized by bromination of ergocryptine. Extensive biotransformation of radiolabeled bromocriptine has been demonstrated in humans, rats, and monkeys, leading to the almost complete absence of the parent drug in urine and bile [90]. Very complex metabolite profiles were observed, with numerous radioactive components; among them, seventeen were identified. Four transformation processes were described, including hydrolytic cleavage of the amide bridge, epimerization, oxidation, and conjugation with glucuronic acid [91]. Even if the specific activity of each metabolite was unknown, these results revealed that the metabolic profile of ergot alkaloids is complex, and that any change in this profile could change toxicity. Interestingly, bromocriptine, in addition to being metabolized by the liver, appeared to be able to induce or inhibit certain drug-metabolizing processes. Studies conducted in the rat revealed that bromocriptine inhibited ethoxyresorufin O-deethylase activity, which is considered to be specific to certain cytochrome P450 iso-enzymes [92]. Likewise, bromocriptine enhanced the NADPH cytochrome c reductase activity, which is necessary for the cytochrome P450 monooxygenase system [92]. The first demonstration of the involvement of the cytochrome P450 system in fescue toxicosis was made in sheep. Endophyte-infected fescue hay increased hepatic antipyrine uptake, which was used as an indirect indicator of hepatic mixed-function oxidase activity [93]. This effect was blocked by cimetidine, a cytochrome P450 inhibitor [93]. Interestingly, increased respiration rates and rectal temperatures in animals that had consumed endophyte-infected tall fescue were linked to increased mixed-function oxidase activity, and were partially reversed by cimetidine [93]. A similar effect was proposed to explain the protective effect of an ivermectin treatment on tall fescue toxicosis in cattle [94]. Complementary studies revealed that bromocriptine and other ergopeptide alkaloids are substrates for the CYP3A subfamily of cytochrome P450 [95,96]. d-lysergic acid did not interact with CYP3A, suggesting that it is not a substrate [96]. In humans, inhibition of CYP3A activity by ritonavir resulted in increased ergotamine toxicity [97].
Investigations were conducted to measure the genomic expression of several drug-metabolizing enzymes in rodents. These studies were conducted using high toxic levels of ergovaline by incorporation of endophyte-infected seeds in the feed [98]. Some studies, conducted with animals kept under heat stress, revealed an increase in the expression of CYP2A12, 2D26, 2C13 and 2E1, whereas a decrease was observed in CYP3A25 [99,100]. Other studies conducted at ambient temperature revealed increased expression of CYP1A1, 2C9, 2E1, 3A1 but a decrease in CYP3A7 expression [101]. These effects were observed in parallel with some changes in the expression of the gene-encoding enzymes involved in the mechanisms of defense against oxidative damage [100,101]. Taken together, these results led to the hypothesis that increased expression of cytochrome P450 enzymes and decreased expression of antioxidant enzymes contribute to the toxic effect of fescue toxicosis, especially at high exposure level under heat stress [100,101,102]. Increased oxidation of glutathione in red blood cells was also demonstrated in cattle grazing infected tall fescue under heat stress [103]. The involvement of drug-metabolizing enzyme activities in the susceptibility to fescue toxicosis was also hypothesized in mice selected for their resistance to the disease. Resistant mice displayed significantly higher glutathione-S-transferase and uridine diphosphate glucuronosyltransferase activities, and these differences were correlated with toxicity [104]. Interestingly, the glutathione-S-transferase activity was correlated with the survival percentage of pups in the resistant line of mice [105]. Sleep time following sodium pentobarbital anesthesia also differed in the resistant and susceptible lines, suggesting differences in cytochrome P450 activity [106]. Differences in the microsomal metabolism of ergotamine were also measured in the two lines challenged with a diet that contained seeds of endophyte-infected tall fescue. The resistant line formed more metabolites than the sensitive line, and a gender difference in the formation of metabolites was also observed [107].
Effects of Epichloë-infected hay on the activity of drug-metabolizing enzymes were also measured in sheep. Feeding lactating ewes with an endophyte-infected tall fescue hay that contained 497 µg ergovaline/kg dry matter led to a general decrease in cytochrome P450 enzymes in the liver, with the exception of erythromycin N-demethylase activity, which increased by 330% [108]. Erythromycin is a substrate of the subfamily CYP3A that cross-reacts with anti-human CYP3A4 antibodies [109]. In humans, administration of erythromycin increased the bromocriptine area under the concentration−time curve by 268% [110], whereas administration of ergotamine and ritonavir, a CYP3A inhibitor, led to ergotamine overdosage and toxicity [97]. The cyclic tripeptide moiety of ergopetides was found to be essential for their metabolization by CYP3A. d-lysergic acid did not interact with cytochrome P450 [96]. Comparison of the in vitro metabolism of ergotamine performed with microsomes obtained from beef cattle and sheep revealed that both species metabolized the toxin, beef cattle being the most active [111]. Interestingly, breed, gender and species differences have been reported in CYP3A activity in livestock [112]. Taken together, these results suggest that: (i) ergopeptide metabolism by CYP3A may play an important role in tall fescue toxicosis; and (ii) differences in ergopeptine metabolism between species could contribute to the observed differences in toxicity.
In conclusion, ergot alkaloids found in endophyte-infected plants are responsible for various toxic syndromes in horses and livestock. Although several compounds may be present in endophyte-infected plants, few data are available concerning the profile of alkaloid production under field conditions. Ergoamide derivatives are commonly found in endophyte-infected Achnatherum spp. plants. The toxicological properties of ergoamides are dominated by psychotropic effects, which are responsible for the symptoms associated with consumption of sleepy grass and drunken horse grass, whereas vasomotor effects are less pronounced. The ergopeptide derivatives are mainly found in endophyte-infected Lolium spp. plants, particularly in tall fescue. The toxicological properties of ergopeptines are dominated by their vasomotor effects, which are responsible for “fescue foot” and “summer syndrome”, whose occurrence varies depending on the external temperature. Another effect that occurs before signs of toxicity is a decrease in plasma prolactin concentrations. Ergovaline, which is the most abundant endophyte-derived ergot alkaloid in grass and seeds, was shown in several bioassays to have the highest vasomotor effect. Other effects of ergopeptines include their ability to interact with drug-metabolizing enzymes. In humans, these interactions are well known, and responsible for drug interactions. Genetic differences in drug-metabolizing enzymes have been described in all the animal species. Several studies conducted in rodents and livestock suggest that interactions with drug-metabolizing enzymes occur and may play a role in the toxicity of ergot alkaloids.
Conflicts of Interest
The author declares no conflict of interest.
References
- Leuchtmann, A.; Bacon, C.W.; Schardl, C.L.; White, J.F., Jr.; Tadych, M. Nomenclatural realignment of Neotyphodium species with genus Epicholë. Mycologia 2014, 106, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, I.J. Tall fescue grass is poison for cattle. N. Z. J. Agric. Res. 1948, 77, 519. [Google Scholar]
- Goodman, A.A. Fescue foot in cattle in Colorado. J. Am. Vet. Med. Assoc. 1952, 121, 289–290. [Google Scholar] [PubMed]
- Stearns, T.J. Fescue foot or ergot-like disease in cattle in Kentucky. J. Am. Vet. Med. Assoc. 1953, 122, 388–389. [Google Scholar] [PubMed]
- Trethewie, E.R.; Gaffney, F.M.; Gladwell, P.J. Pharmacological studies of grasses obtained from a property where tall fescue lameness in cattle occurs. Aust. J. Exp. Biol. Med. Sci. 1954, 32, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Jensen, R.; Deem, A.W.; Knaus, D. Fescue lameness in cattle. I. Experimental production of disease. Am. J. Vet. Res. 1956, 17, 196–201. [Google Scholar] [PubMed]
- Maag, D.D.; Tobiska, J.W. Fescue lameness in cattle. II. Ergot alkaloids in tall fescue grass. Am. J. Vet. Res. 1956, 17, 202–204. [Google Scholar] [PubMed]
- Yates, S.G. Toxicity of tall fescue forage: A review. Econ. Bot. 1962, 16, 295–303. [Google Scholar] [CrossRef]
- Walls, J.R.; Jacobson, D.R. Skin temperature and blood flow in the tail of dairy heifers administered extracts of toxic tall fescue. J. Anim. Sci. 1970, 30, 420–423. [Google Scholar] [PubMed]
- Jacobson, D.R.; Carr, S.B.; Hatton, R.H.; Buckner, R.C.; Graden, A.P.; Dowden, D.R.; Miller, W.M. Growth, physiological responses, and evidence of toxicity in yearling dairy cattle grazing different grasses. J. Dairy Sci. 1970, 53, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Rumsey, T.S.; Stuedemann, J.A.; Wilkinson, S.R.; Williams, D.J. Chemical composition of necrotic fat lesions in beef cows grazing fertilized «Kentucky-31» tall fescue. J. Anim. Sci. 1979, 48, 673–682. [Google Scholar] [PubMed]
- Bacon, C.W.; Porter, J.K.; Robbins, J.D.; Luttrell, E.S. Epichloë typhina from toxic tall fescue grasses. Appl. Environ. Microbiol. 1977, 35, 576–581. [Google Scholar]
- Schmidt, S.P.; Hoveland, C.S.; Clark, E.M.; Davis, N.D.; Smith, L.A.; Grimes, H.W.; Holliman, J.L. Association of an endophytic fungus with fescue toxicity in steers fed Kentucky 31 tall fescue seed or hay. J. Anim. Sci. 1982, 55, 1259–1263. [Google Scholar] [PubMed]
- Yates, S.G.; Plattner, R.D.; Garner, G.B. Detection of ergopeptine alkaloids in endophyte infected, toxic Ky-31 tall fescue by mass spectrometry/mass spectrometry. J. Agric. Food Chem. 1985, 33, 719–722. [Google Scholar] [CrossRef]
- Solomons, R.N.; Oliver, J.W.; Linnabary, R.D. Reactivity of dorsal pedal vein of cattle to selected alkaloids associated with Acremonium coenophialum infected fescue grass. Am. J. Vet. Res. 1989, 50, 235–238. [Google Scholar] [PubMed]
- Tor-Agbidye, J.; Blythe, L.L.; Craig, A.M. Correlation of endophyte toxins (ergovaline and lolitrem B) with clinical disease: fescue foot and perennial ryegrass staggers. Vet. Hum. Toxicol. 2001, 43, 140–146. [Google Scholar] [PubMed]
- Zbib, N.; Repussard, C.; Tardieu, D.; Guerre, P. Toxicité des mycotoxines produites par des champignons endophytes du genre Neotyphodium. Rev. Méd. Vét. 2014, 165, 116–135. [Google Scholar]
- Shelby, R.A. Analysis of ergot alkaloids in endophyte-infected tall fescue by liquid chromatography/electrospray ionization mass spectrometry. J. Agric. Food Chem. 1997, 45, 4674–4679. [Google Scholar] [CrossRef]
- Shymanovich, T.; Saari, S.; Lovin, M.E.; Jarmusch, A.K.; Jarmusch, S.A.; Musso, A.M.; Charlton, N.D.; Young, C.A.; Cech, N.B.; Faeth, S.H. Alkaloid variation among epichloid endophytes of fleepygrass (Achnatherum robustum) and consequences for resistance to insect herbivores. J. Chem. Ecol. 2015, 41, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Hopkirk, C.S.M. Staggers in livestock. N. Z. J. Agric. Res. 1935, 51, 18–21. [Google Scholar]
- Lloyd, A.B. The endophytic fungus of perennial ryegrass. N. Z. J. Agric. Res. 1959, 2, 1187–l194. [Google Scholar]
- Thornton, R.H. An examination of ergot in relation to ryegrass staggers. N. Z. Vet. J. 1964, 12, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Keogh, R.G. Induction and prevention of ryegrass staggers in grazing sheep. N. Z. J. Exp. Agric. 1973, 1, 55–57. [Google Scholar]
- Gallagher, R.T.; Keogh, R.G.; Latch, G.C.M.; Reid, C.S.W. The role of fungal tremorgens in ryegrass staggers. N. Z. J. Agric. Res. 1977, 20, 431–440. [Google Scholar] [CrossRef]
- Gallagher, R.T.; White, E.P.; Mortimer, P.H. Ryegrass staggers: isolation of potent neurotoxins lolitrem A and lolitrem B from staggers-producing pastures. N. Z. Vet. J. 1981, 29, 189–190. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.O.; Wilkins, A.L.; Gallagher, R.T.; Hawkes, A.D.; Munday, S.C.; Towers, N.R. Synthesis and tremorgenicity of paxitriols and lolitriol: Possible biosynthetic precursors of lolitrem B. J. Agric. Food Chem. 1992, 40, 234–238. [Google Scholar] [CrossRef]
- Easton, H.S.; Lane, G.A.; Tapper, B.A.; Keogh, R.G.; Cooper, B.M.; Blackwell, M.; Anderson, M.; Fletcher, L.R. Ryegrass endophyte-related heat stress in cattle. Proc. N. Z. Grassl. Assoc. 1996, 57, 37–41. [Google Scholar]
- Auldist, M.J.; Thom, E.R. Effects of endophyte infection of perennial ryegrass on somatic cell counts, mammary inflammation, and milk protein composition in grazing dairy cattle. N. Z. J. Agric. Res. 2000, 43, 345–349. [Google Scholar] [CrossRef]
- Hovermale, J.T.; Craig, A.M. Correlation of ergovaline and lolitrem B levels in endophyte-infected perennial ryegrass (Lolium Perenne). J. Vet. Diagn. Invest. 2001, 13, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Benkhelil, A.; Grancher, D.; Giraud, N.; Bezille, P.; Bony, S. Intoxication par des toxines de champignons endophytes chez des taureaux reproducteurs. Rev. Méd. Vét. 2004, 156, 243–247. [Google Scholar]
- Johnstone, L.K.; Mayhew, I.G.; Fletcher, L.R. Clinical expression of lolitrem B (perennial ryegrass) intoxication in horses. Equine Vet. J. 2012, 44, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Siegel, M.; Latch, G.; Bush, L.; Fannin, F.; Rowan, D.; Tapper, B.; Bacon, C.; Johnson, M. Fungal endophyte-infected grasses: Alkaloid accumulation and aphid response. J. Chem. Ecol. 1990, 16, 3301–3315. [Google Scholar] [CrossRef] [PubMed]
- Te Paske, M.; Powell, R. Analyses of selected endophyte-infected grasses for the presence of loline-type and ergot-type alkaloids. J. Agric. Food Chem. 1993, 41, 2299–2303. [Google Scholar] [CrossRef]
- Thom, E.R.; Popay, A.J.; Hume, D.E.; Fletcher, L.R. Evaluating the performance of endophytes in farm systems to improve farmer outcomes—A review. Crop Pasture Sci. 2012, 63, 927–943. [Google Scholar] [CrossRef]
- Johnson, L.J.; de Bonth, A.C.M.; Briggs, L.R.; Caradus, J.R.; Finch, S.C.; Fleetwood, D.J.; Fletcher, L.R.; Hume, D.E.; Johnson, R.D.; Popay, A.J.; et al. The exploitation of Epichloeae endophytes for agricultural benefit. Fungal Divers. 2013, 60, 171–188. [Google Scholar] [CrossRef]
- Fletcher, L.R. Novel endophytes in New Zealand grazing systems: The perfect solution or a compromise? In Epichloae, Endophytes of Cool Season Grasses: Implications, Utilization and Biology; Young, C.A., Aiken, G.E., McCulley, R.L., Strickland, J.R., Schardl, C.L., Eds.; The Samuel Roberts Noble Foundation: Ardmore, OK, USA, 2012; pp. 5–13. [Google Scholar]
- Fleetwood, D.J.; Scott, B.; Voisey, C.R.; Johnson, R.D. Insights into the molecular biology of Epichloe endophyte alkaloid biosynthesis. Proc. N. Z. Grassl. Assoc. 2008, 70, 217–220. [Google Scholar]
- Miles, C.O.; Lane, G.A.; di Menna, M.E.; Garthwaite, I.; Piper, E.L.; Ball, O.J.-P.; Latch, G.C.M.; Allen, J.M.; Hunt, M.B.; Min, F.K.; et al. High levels of ergonovine and lysergic acid amide in toxic Achnatherum inebrians accompany infection by an Acremonium-like endophytic fungus. J. Agric. Food Chem. 1996, 44, 1285–1290. [Google Scholar] [CrossRef]
- Petroski, R.J.; Powell, R.G.; Clay, K. Alkaloids of Stipa robusta (sleepygrass) infected with an Acremonium endophyte. Nat. Toxins 1992, 1, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Schardl, C.; Young, C.; Faulkner, J.; Florea, S.; Pan, J. Chemotypic diversity of Epichloae, fungal symbionts of grasses. Fungal Ecol. 2012, 5, 331–344. [Google Scholar] [CrossRef]
- Yates, S.G.; Powell, R.G. Analysis of ergopeptine alkaloids in endophyte-infected tall fescue. J. Agric. Food Chem. 1988, 36, 337–340. [Google Scholar] [CrossRef]
- Lyons, P.C.; Plattner, R.D.; Bacon, C.W. Occurrence of peptide and clavine ergot alkaloids in tall fescue grass. Science 1986, 232, 487–489. [Google Scholar] [CrossRef] [PubMed]
- Belesky, D.P.; Stuedemann, J.A.; Plattner, R.D.; Wilkinson, S.R. Ergopeptine alkaloids in grazed tall fescue. Agron. J. 1988, 80, 209–212. [Google Scholar] [CrossRef]
- Rottinghaus, G.E.; Garner, G.B.; Cornell, C.N.; Ellis, J.L. HPLC method for quantitating ergovaline in endophyte-infested tall fescue: Seasonal variation of ergovaline levels in stems with leaf sheaths, leaf blades, and seed heads. J. Agric. Food Chem. 1991, 39, 112–115. [Google Scholar] [CrossRef]
- Repussard, C.; Zbib, N.; Tardieu, D.; Guerre, P. Endophyte infection of tall fescue and the impact of climatic factors on ergovaline concentrations in field crops cultivated in southern France. J. Agric. Food Chem. 2014, 62, 9609–9614. [Google Scholar] [CrossRef] [PubMed]
- Repussard, C.; Zbib, N.; Tardieu, D.; Guerre, P. Ergovaline and lolitrem B concentrations in perennial ryegrass in field culture in southern France: Distribution in the Plant and Impact of Climatic Factors. J. Agric. Food Chem. 2014, 62, 12707–12712. [Google Scholar] [CrossRef] [PubMed]
- Bony, S.; Pichon, N.; Ravel, C.; Durix, A.; Balfourier, F.; Guillaumin, J.J. The relationship between mycotoxin synthesis and isolate morphology in fungal endophytes of Lolium perenne. New Phytol. 2001, 152, 125–137. [Google Scholar] [CrossRef]
- Oliveira, J.A.; Rottinghaus, G.E.; Prego, C.; González, A. Contenido en alcaloides en semillas de poblaciones naturales de raigrás inglés del norte de España infectadas con los hongos endofitos Neotyphodium. Invest. Agr. Prod. Prot. Veg. 2002, 17, 247–256. [Google Scholar]
- Durix, A.; Bony, S.; Ravel, C.; Balfourier, F.; Guillaumin, J.J.; Guesquière, M.; Chosson, J.F.; Charmet, G. The influence of toxic Neotyphodium endophyte on perennial ryegrass (Lolium perenne). Rev. Méd. Vet. 1998, 6, 528. [Google Scholar]
- Spiering, M.J.; Lane, G.A.; Christensen, M.J.; Schmid, J. Distribution of the fungal endophyte Neotyphodium lolii is not a major determinant of the distribution of fungal alkaloids in Lolium perenne plants. Phytochemistry 2005, 66, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Schardl, C.L.; Young, C.A.; Hesse, U.; Amyotte, S.G.; Andreeva, K.; Calie, P.J.; Fleetwood, D.J.; Haws, D.C.; Moore, N.; Oeser, B.; et al. Plant-symbiotic fungi as chemical engineers: Multi-genome analysis of the clavicipitaceae reveals dynamics of alkaloid loci. PLoS Genet. 2013, 9, e1003323. [Google Scholar] [CrossRef] [PubMed]
- Oldenburg, E. Endophytic fungi and alkaloid production in perennial ryegrass in Germany. Grass Forage Sci. 1997, 52, 425–431. [Google Scholar] [CrossRef]
- Reed, K.F.M.; Nie, Z.N.; Walker, L.V.; Kearney, G. Fluctuations in the concentration of ergovaline and lolium B produced by the wild-type endophyte (Neotyphodium lolii) in perennial ryegrass (Lolium perenne) pasture. Anim. Prod. Sci. 2011, 51, 1098–1108. [Google Scholar] [CrossRef]
- Zhang, X.X.; Li, C.J.; Nan, Z.B. Effects of cutting frequency and height on alkaloid production in endophyte-infected drunken horse grass (Achnatherum inebrians). Sci. China Life Sci. 2011, 54, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Faeth, S.H.; Gardner, D.R.; Hayes, C.J.; Jani, A.; Wittlinger, S.K.; Jones, T.A. Temporal and spatial variation in alkaloid levels in Achnatherum robustum, a native grass infected with the endophyte Neotyphodium. J. Chem. Ecol. 2006, 32, 307–324. [Google Scholar] [CrossRef] [PubMed]
- Welty, R.E.; Craig, A.M.; Azevedo, M.D. Variability of ergovaline in seeds and straw and endophyte infection in seeds among endophyte-infected genotypes of tall fescue. Plant Dis. 1994, 78, 845–849. [Google Scholar] [CrossRef]
- Rasmussen, S.; Parsons, A.J.; Bassett, S.; Christensen, M.J.; Hume, D.E.; Johnson, L.J.; Johnson, R.D.; Simpson, W.R.; Stacke, C.; Voisey, C.R.; et al. High nitrogen supply and carbohydrate content reduce fungal endophyte and alkaloid concentration in Lolium perenne. New Phytol. 2007, 173, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Malinowski, D.P.; Belesky, D.P.; Hill, N.S.; Baligar, V.C.; Fedders, J.M. Influence of phosphorus on the growth and ergot alkaloid content of Neotyphodium coenophialum-infected tall fescue (Festuca arundinacea Schreb.). Plant Soil 1998, 198, 53–61. [Google Scholar] [CrossRef]
- Brosi, G.; McCulley, R.; Bush, L.; Nelson, J.; Classen, A.; Norby, R. Effects of multiple climate change factors on the tall fescue-fungal endophyte symbiosis: Infection frequency and tissue chemistry. New Phytol. 2011, 189, 797–805. [Google Scholar] [CrossRef] [PubMed]
- McCulley, R.L.; Bush, L.P.; Carlisle, A.E.; Ji, H.; Nelson, J.A. Warming reduces tall fescue abundance but stimulates toxic alkaloid concentrations in transition zone pastures of the U.S. Front Chem. 2014, 2. [Google Scholar] [CrossRef]
- Repussard, C.; Zbib, N.; Tardieu, D.; Guerre, P. Les champignons endophytes du genre Neotyphodium et leurs toxines: généralités et problématique française. Rev. Méd. Vét. 2013, 164, 583–606. [Google Scholar]
- Takach, J.; Mittal, S.; Swoboda, G.; Bright, S.; Trammell, M.; Hopkins, A.; Young, C. Genotypic and chemotypic diversity of Neotyphodium endophytes in tall fescue from Greece. Appl. Environ. Microbiol. 2012, 78, 5501–5510. [Google Scholar] [CrossRef] [PubMed]
- Clement, S.; Elberson, L.; Youssef, N.; Davitt, C.; Doss, R. Incidence and diversity of Neotyphodium fungal endophytes in tall fescue from Morocco, Tunisia, and Sardinia. Crop Sci. 2001, 41, 570–576. [Google Scholar] [CrossRef]
- Guillaumin, J.J.; Frain, M.; Pichon, N.; Ravel, C. Survey of the fungal endophytes in wild grass species of the Auvergne region (central France). In Proceedings of the Fourth International Neotyphodium/Grass Interactions Symposium; Paul, V.H., Dapprich, P.D., Eds.; Fachbereich Agrarwirtschaft: Soest, Germany, 2001; pp. 85–92. [Google Scholar]
- Gould, L.S.; Hohenboken, W.D. Differences between progeny of beef sires in susceptibility to fescue toxicosis. J. Anim. Sci. 1993, 71, 3025–3032. [Google Scholar] [PubMed]
- Miller, B.F.; Armstrong, K.L.; Wilson, L.A.; Hohenboken, W.D.; Saacke, R.G. Variation among inbred and linecross mice in response to fescue toxicosis. J. Anim. Sci. 1994, 72, 2896–2904. [Google Scholar] [PubMed]
- Oliver, J.W.; Abney, L.K. Report of isolated bovine vessel response to alkaloids of tall fescue. In Proceedings of the Tall Fescue Toxicosis Workshop, SRIEG-37, Atlanta, GA, USA, 13–14 November 1989; p. 93.
- Dyer, D.C. Evidence that ergovaline acts on serotonin receptors. Life Sci. 1993, 53, 223–228. [Google Scholar] [CrossRef]
- Gadberry, M.S.; Denard, T.M.; Spiers, D.E.; Piper, E.L. Effects of feeding ergovaline on lamb performance in a heat stress environment. J. Anim. Sci. 2003, 81, 1538–1545. [Google Scholar] [PubMed]
- Schardl, C.; Panaccione, D.; Tudzynski, P. Ergot alkaloids—Biology and molecular biology. Alkaloids Chem. Biol. 2006, 63, 45–86. [Google Scholar] [PubMed]
- Fleetwood, D.J.; Scott, B.; Lane, G.A.; Tanaka, A.; Johnson, R.D. A complex ergovaline gene cluster in Epichloë endophytes of grasses. Appl. Environ. Microbiol. 2007, 73, 2571–2579. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Somei, M.; Sekihara, S.; Nakagawa, K.; Yamada, F. Dopamine receptor stimulating effects of chanoclavine analogues, tricyclic ergot alkaloids, in the brain. Jpn. J. Pharmacol. 1987, 45, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Li, G.S.; Robinson, J.M.; Floss, H.G.; Cassady, J.M. Ergot alkaloids. Synthesis of 6-methyl-8-ergolenes as inhibitors of prolactin release. J. Med. Chem. 1975, 9, 892–895. [Google Scholar] [CrossRef]
- Meira, M.; da Silva, E.P.; David, J.M.; David, J.P. Review of the genus Ipomoea: traditional uses, chemistry and biological activities. Braz. J. Pharmacog. 2012, 22, 682–713. [Google Scholar]
- Schultz, C.; Lodge-Ivey, S.; Bush, L.; Craig, A.; Strickland, J. Effects of initial and extended exposure to an endophyte-infected tall fescue seed diet on faecal and urinary excretion of ergovaline and lysergic acid in mature geldings. N. Z. Vet. J. 2006, 54, 178–184. [Google Scholar] [CrossRef] [PubMed]
- De Lorme, M.J.M.; Lodge-Ivey, S.L.; Craig, A.M. Physiological and digestive effects of Neotyphodium coenophialum-infected tall fescue fed to lambs. J. Anim. Sci. 2007, 85, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Ayers, A.W.; Hill, N.S.; Rottinghaus, G.E.; Stuedemann, J.A.; Thompson, F.N.; Purinton, P.T.; Seman, D.H.; Dawe, D.L.; Parks, A.H.; Ensley, D. Ruminal metabolism and transport of tall fescue ergot alkaloids. Crop Sci. 2009, 49, 2309–2316. [Google Scholar] [CrossRef]
- Klotz, J.L.; Bush, L.P.; Smith, D.L.; Shafer, W.D.; Smith, L.L.; Vevoda, A.C.; Craig, A.M.; Arrington, B.C.; Strickland, J.R. Assessment of vasoconstrictive potential of d-lysergic acid using an isolated bovine lateral saphenous vein bioassay. J. Anim. Sci. 2006, 84, 3167–3175. [Google Scholar] [CrossRef] [PubMed]
- Foote, A.P.; Harmon, D.L.; Strickland, J.R.; Bush, L.P.; Klotz, J.L. Effect of ergot alkaloids on contractility of bovine right ruminal artery and vein. J. Anim. Sci. 2011, 89, 2944–2949. [Google Scholar] [CrossRef] [PubMed]
- Pesqueira, A.; Harmon, D.L.; Branco, A.F.; Klotz, J.L. Bovine lateral saphenous veins exposed to ergopeptine alkaloids do not relax. J. Anim. Sci. 2014, 92, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Browning, R., Jr.; Thompson, F.N.; Sartin, J.L.; Leite-Browning, M.L. Plasma concentrations of prolactin, growth hormone, and luteinizing hormone in steers administered ergotamine or ergonovine. J. Anim. Sci. 1977, 75, 796–802. [Google Scholar]
- Oliver, J.W.; Linnabary, R.D.; Abney, L.K.; van Manen, K.R.; Knoop, R.; Adair, H.S., 3rd. Evaluation of a dosing method for studying ergonovine effects in cattle. Am. J. Vet. Res. 1994, 55, 173–176. [Google Scholar] [PubMed]
- Klotz, J.L.; Bush, L.P.; Smith, D.L.; Shafer, W.D.; Smith, L.L.; Arrington, B.C.; Strickland, J.R. Ergovaline-induced vasoconstriction in an isolated bovine lateral saphenous vein bioassay. J. Anim. Sci. 2007, 85, 2330–2336. [Google Scholar] [CrossRef] [PubMed]
- Klotz, J.L.; Kirch, B.H.; Aiken, G.E.; Bush, L.P.; Strickland, J.R. Contractile response of fescue-naïve bovine lateral saphenous veins to increasing concentrations of tall fescue alkaloids. J. Anim. Sci. 2010, 88, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Foote, A.P.; Harmon, D.L.; Brown, K.R.; Strickland, J.R.; McLeod, K.R.; Bush, L.P.; Klotz, J.L. Constriction of bovine vasculature caused by endophyte-infected tall fescue seed extract is similar to pure ergovaline. J. Anim. Sci. 2012, 90, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Larson, B.T.; Harmon, D.L.; Piper, E.L.; Griffis, L.M.; Bush, L.P. Alkaloid binding and activation of D2 dopamine receptors in cell culture. J. Anim. Sci. 1999, 77, 942–947. [Google Scholar] [PubMed]
- Passie, T.; Halpern, J.H.; Stichtenoth, D.O.; Emrich, H.M.; Hintzen, A. The pharmacology of lysergic acid diethylamide: A review. CNS Neurosci. Ther. 2008, 14, 295–314. [Google Scholar] [CrossRef] [PubMed]
- Lehner, A.F.; Craig, M.; Fannin, N.; Bush, L.; Tobin, T. Electrospray[+] tandem quadrupole mass spectrometry in the elucidation of ergot alkaloids chromatographed by HPLC: Screening of grass or forage samples for novel toxic compounds. J. Mass Spectrom. 2005, 11, 1484–1502. [Google Scholar] [CrossRef]
- Hill, N.S.; Thompson, F.N.; Stuedemann, J.A.; Rottinghaus, G.W.; Ju, H.J.; Dawe, D.L.; Hiatt, E.E., 3rd. Ergot alkaloid transport across ruminant gastric tissues. J. Anim. Sci. 2001, 79, 542–549. [Google Scholar] [PubMed]
- Maurer, G.; Schreier, E.; Delaborde, S.; Nufer, R.; Shukla, A.P. Fate and disposition of bromocriptine in animals and man. II: Absorption, elimination and metabolism. Eur. J. Drug Metab. Pharmacokinet. 1983, 8, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Maurer, G.; Schreier, E.; Delaborde, S.; Loosli, H.R.; Nufer, R.; Shukla, A.P. Fate and disposition of bromocriptine in animals and man. I: Structure elucidation of the metabolites. Eur. J. Drug Metab. Pharmacokinet. 1983, 7, 281–292. [Google Scholar] [CrossRef]
- Moochhala, S.M.; Lee, E.J.D.; Hu, G.T.M.; Koh, O.S.; Becket, G. Effects of bromocriptine on hepatic cytochrome P-450 monooxygenase system. Jpn. J. Pharmacol. 1989, 49, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Zanzalari, K.P.; Heitmann, R.N.; McLaren, J.B.; Fribourg, H.A. Effects of endophyte-infected fescue and cimetidine on respiration rates, rectal temperatures and hepatic mixed function oxidase activity as measured by hepatic antipyrine metabolism in sheep. J. Anim. Sci. 1989, 67, 3370–3378. [Google Scholar] [PubMed]
- Bransby, D.L. Steer weight gain responses to ivermectin when grazing fescue. Large Anim. Pract. 1997, 18, 16–19. [Google Scholar]
- Ball, S.E.; Maurer, G.; Zollinger, M.; Ladona, M.; Vickers, A.E. Characterization of the cytochrome P-450 gene family responsible for the N-dealkylation of the ergot alkaloid CQA 206–291 in humans. Drug Metab. Dispos. 1992, 20, 56–63. [Google Scholar] [PubMed]
- Peyronneau, M.A.; Delaforge, M.; Riviere, R.C.; Renaud, J.-P.; Mansuy, D. High affinity of ergopeptides for cytochromes P450 3A Importance of their peptide moiety for P450 recognition and hydroxylation of bromocriptine. Eur. J. Biochem. 1994, 223, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Liaudet, L. Severe ergotism associated with interaction between ritonavir and ergotamine. BMJ 1999, 318, 771. [Google Scholar] [CrossRef] [PubMed]
- Spiers, D.E.; Evans, T.J.; Rottinghaus, G.E. Interaction between thermal stress and fescue toxicosis: animal models and new perspectives. In Neotyphodium in Cool Season Grasses; Roberts, C.A., West, C.P., Spiers, D.E., Eds.; Blackwell: Ames, IA, USA, 2005; pp. 243–270. [Google Scholar]
- Bhusari, S.; Liu, Z.; Hearne, L.B.; Spiers, D.E.; Lamberson, W.R.; Antoniou, E. Expression profiling of heat stress effects on mice fed ergot alkaloids. Toxicol. Sci. 2006, 95, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Settivari, R.S.; Bhusari, S.; Evans, T.; Eichen, P.A.; Hearne, L.B.; Antoniou, E.; Spiers, D.E. Genomic analysis of the impact of fescue toxicosis on hepatic function. J. Anim. Sci. 2006, 84, 1279–1294. [Google Scholar] [PubMed]
- Settivari, R.S.; Evans, T.J.; Rucker, E.; Rottinghaus, G.E.; Spiers, D.E. Effect of ergot alkaloids associated with fescue toxicosis on hepatic cytochrome P450 and antioxidant proteins. Toxicol. Appl. Pharmacol. 2008, 227, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Settivari, R.S.; Evans, T.J.; Yarru, L.P.; Eichen, P.A.; Sutovsky, P.; Rottinghaus, G.E.; Antoniou, E.; Spiers, D.E. Effects of short-term heat stress on endophytic ergot alkaloid-induced alterations in rat hepatic gene expression. J. Anim. Sci. 2009, 87, 3142–3155. [Google Scholar] [CrossRef] [PubMed]
- Lakritz, J.; Leonard, M.J.; Eichen, P.A.; Rottinghaus, G.E.; Johnson, G.C.; Spiers, D.E. Whole-blood concentrations of glutathione in cattle exposed to heat stress or a combination of heat stress and endophyte-infected tall fescue toxins in controlled environmental conditions. Am. J. Vet. Res. 2002, 63, 799–803. [Google Scholar] [CrossRef] [PubMed]
- Hohenboken, W.D.; Blodgett, D.J. Growth and physiological responses to toxicosis in lines of mice selected for resistance or susceptibility to endophyte-infected tall fescue in the diet. J. Anim. Sci. 1997, 75, 2165–2173. [Google Scholar] [PubMed]
- Wagner, C.R.; Howell, T.M.; Hohenboken, W.D.; Blodgett, D.J. Impacts of an endophyte-infected fescue seed diet on traits of mouse lines divergently selected for response to that same diet. J. Anim. Sci. 2000, 78, 1191–1198. [Google Scholar] [PubMed]
- Arthur, K.A.; Kuehn, L.A.; Hohenboken, W.D. Sleep time following anesthesia in mouse lines selected for resistance or susceptibility to fescue toxicosis. J. Anim. Sci. 2003, 81, 2562–2567. [Google Scholar] [PubMed]
- Duringer, J.M.; Lewis, R.; Kuehn, L.; Fleischmann, T.; Craig, A.M. Growth and hepatic in vitro metabolism of ergotamine in mice divergently selected for response to endophyte toxicity. Xenobiotica 2005, 35, 531–548. [Google Scholar] [CrossRef] [PubMed]
- Zbib, N.; Repussard, C.; Tardieu, D.; Priymenko, N.; Domange, C.; Guerre, P. Ergovaline in tall fescue and its effect on health, milk quality, biochemical parameters, oxidative status, and drug metabolizing enzymes of lactating ewes. J. Anim. Sci. 2014, 92, 5112–5123. [Google Scholar] [CrossRef] [PubMed]
- Szotáková, B.; Baliharová, V.; Lamka, J.; Nožinová, E.; Wsól, V.; Velı́k, J.; Machala, M.; Neča, J.; Souček, P.; Šusová, S.; Skálová, L. Comparison of in vitro activities of biotransformation enzymes in pig, cattle, goat and sheep. Res. Vet. Sci. 2004, 76, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.V.; Berchou, R.C.; Kareti, D.; LeWitt, P.A. Pharmacokinetic evaluation of erythromycin and caffeine administered with bromocriptine. Clin. Pharmacol. Ther. 1990, 47, 694–697. [Google Scholar] [CrossRef] [PubMed]
- Moubarak, A.; Cannon, S.M.; Rosenkrans, C.F. Cattle vs. Sheep in their responses to fescue toxins. Available online: http://arkansasagnews.uark.edu/545-33.pdf (accessed on 5 March 2015).
- Dacasto, M.; Eeckhoutte, C.; Capolongoa, F.; Dupuy, J.; Carletti, M.; Calléja, C.; Nebbia, C.; Alvinerie, M.; Galtier, P. Effect of breed and gender on bovine liver cytochrome P450 3A (CYP3A) expression and inter-species comparison with other domestic ruminants. Vet. Res. 2005, 36, 179–190. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
