Health-Based Cyanotoxin Guideline Values Allow for Cyanotoxin-Based Monitoring and Efficient Public Health Response to Cyanobacterial Blooms
Abstract
:1. Introduction
2. Methods
2.1. Development of Guideline Values
2.1.1. Guideline Derivation and Exposure Assumptions for Humans
| Exposure factor name | Drinking water | Recreational water | Units |
|---|---|---|---|
| Tolerable daily intake (TDI) | Cyanotoxin dependent | Cyanotoxin dependent | Micrograms cyanotoxin per kilogram body weight per day (µg/kg-day) |
| Body weight (BW) | 60 | 20 | Kilograms (kg) |
| Relative source contribution (RSC) | 1 | 1 | Unitless |
| Intake rate | 2 | 0.1 | Liters per day (L/day) |
2.1.2. Guideline Derivation and Exposure Assumptions for Dogs
2.2. Derivation of Tolerable Daily Intakes
2.2.1. Anatoxin-a
2.2.2. Cylindrospermopsin
2.2.3. Microcystins
2.2.4. Saxitoxins
2.3. Review of Guideline Values Developed by Other Jurisdictions
3. Results
3.1. Guideline Values
| Guideline value | Anatoxin-a | Cylindrospermopsin | Microcystin | Saxitoxin |
|---|---|---|---|---|
| Human TDI (µg/kg-day) | 0.1 | 0.03 | 0.05 | 0.05 |
| Dog TDI (µg/kg-day) | None—used human TDI | None—used human TDI | None—used human TDI | 0.005 |
| Drinking Water (µg/L) | 3.0 | 1.0 | 1.0 | 1.0 |
| Recreational Water (µg/L) | 20.0 | 6.0 | 10.0 | 10.0 |
| Dog-specific (µg/L) | 0.4 | 0.1 | 0.2 | 0.02 |
3.2. Toxin-Based Monitoring Results in Oregon
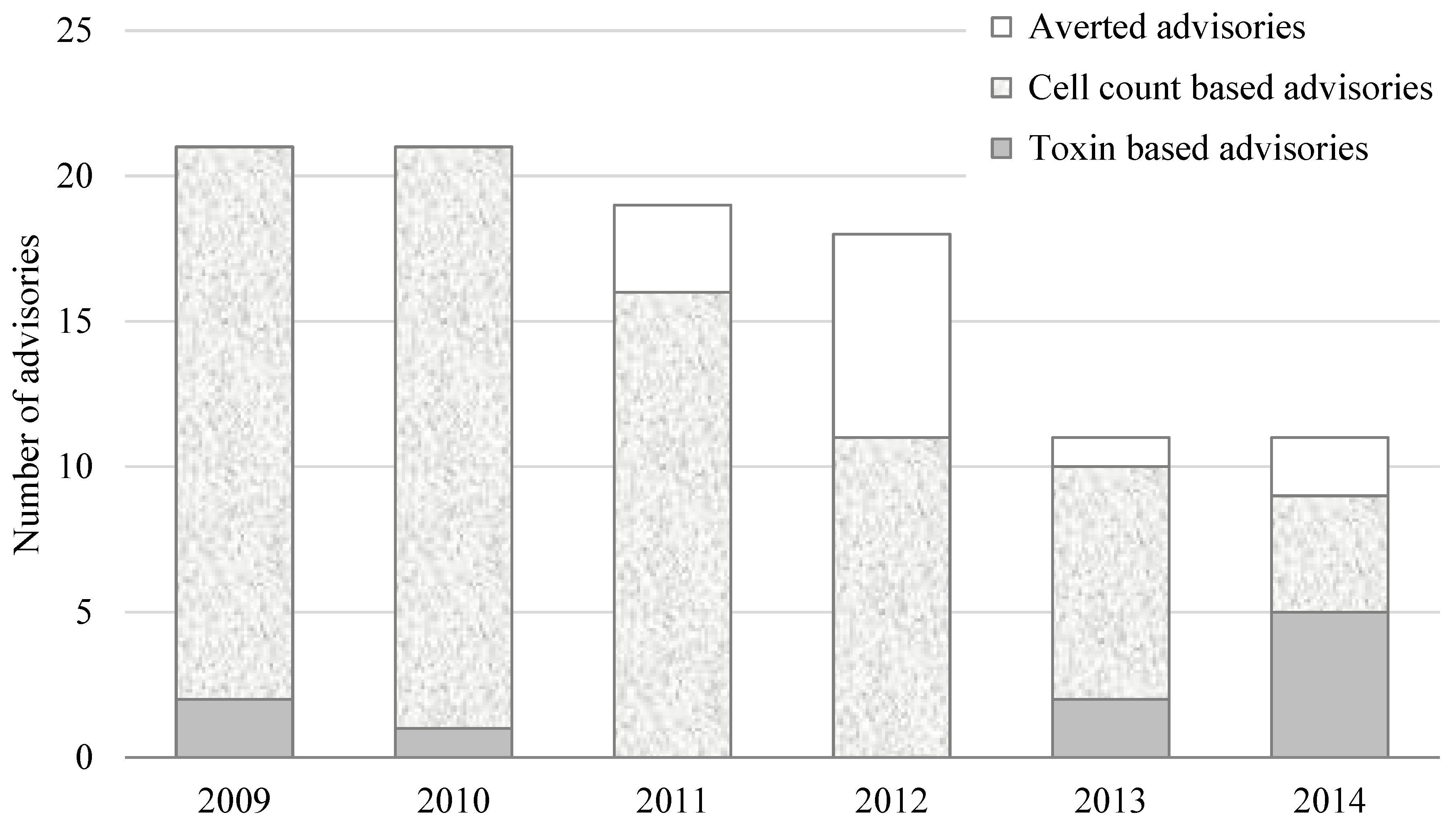
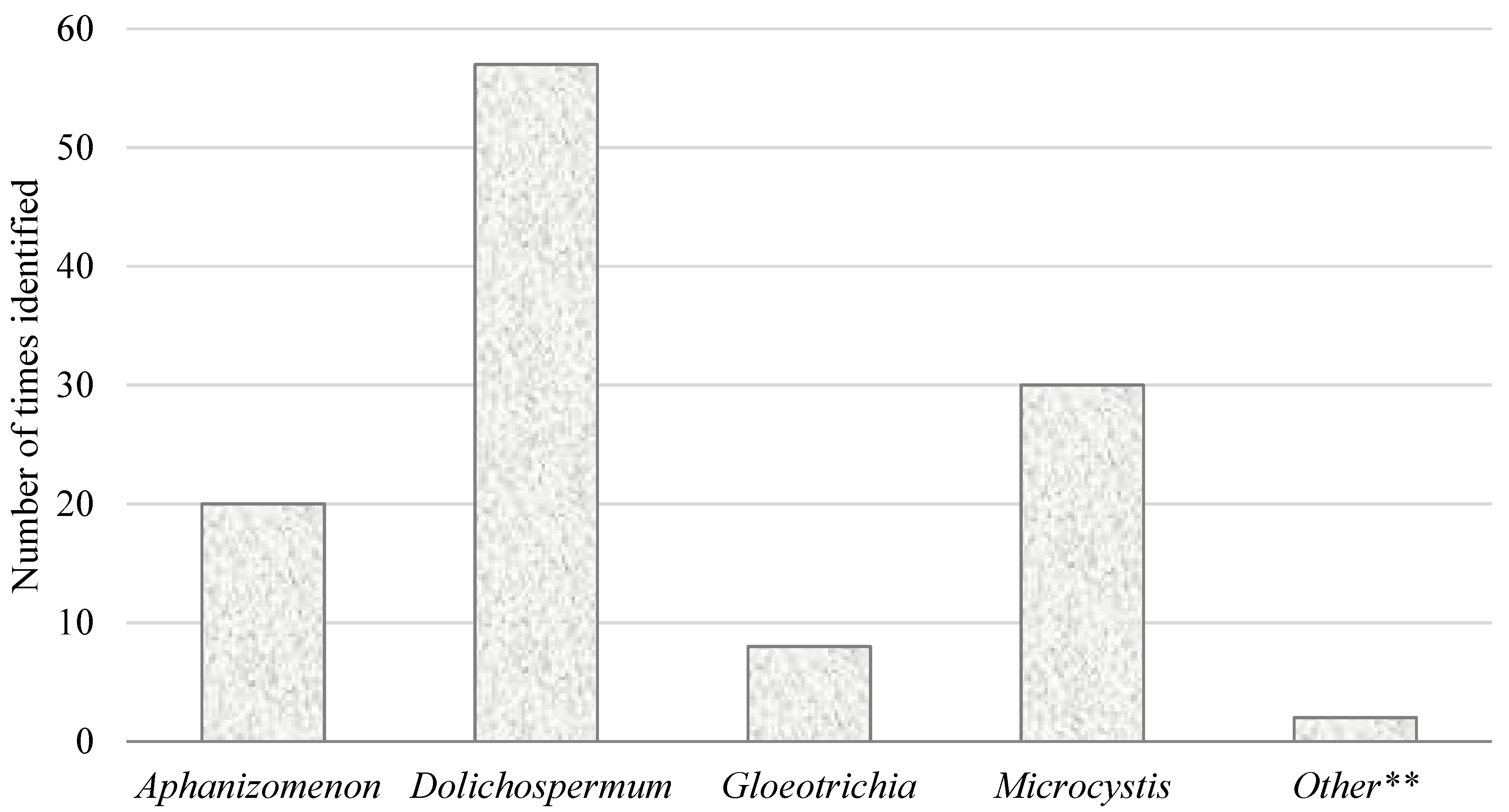
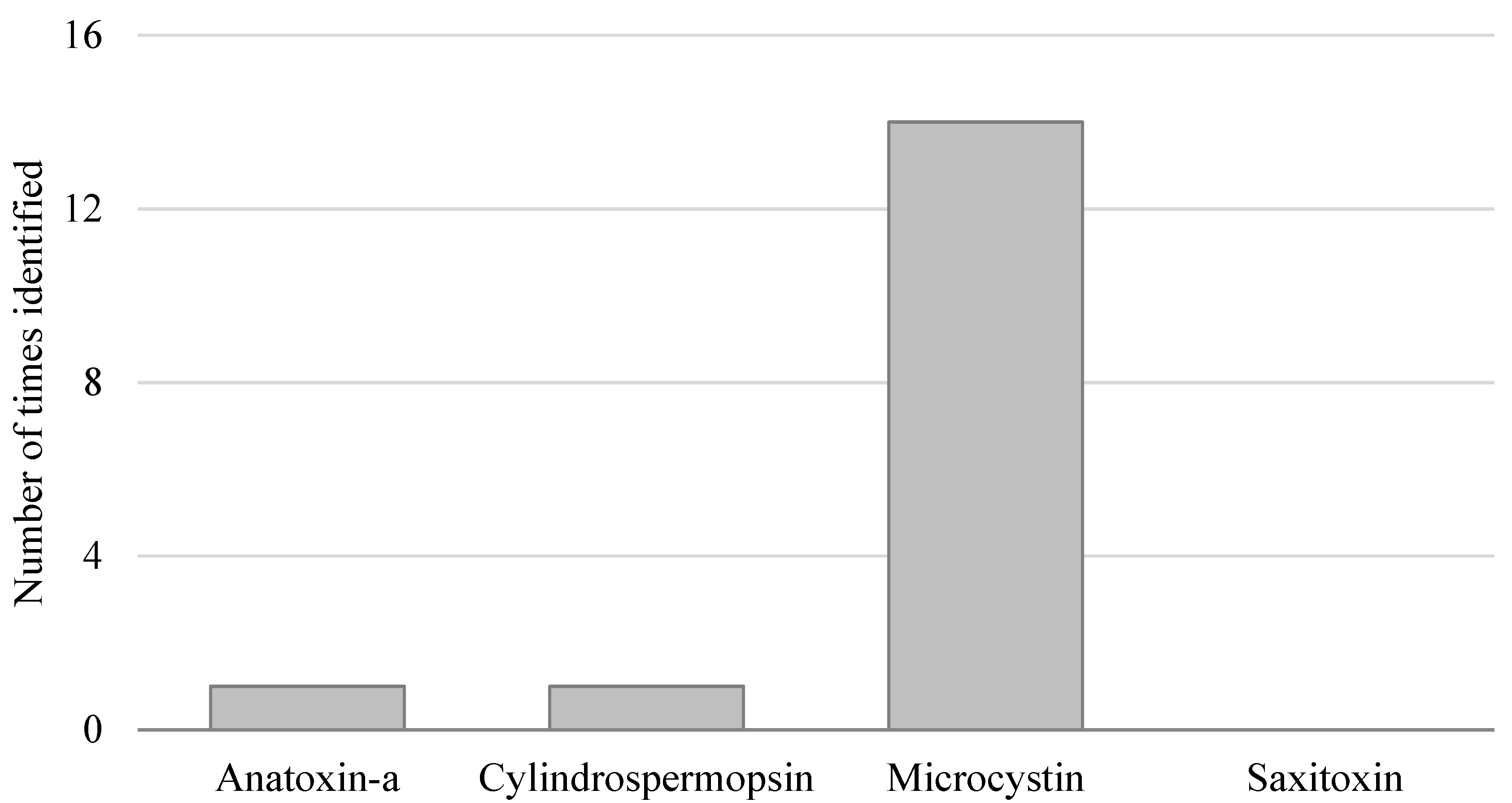
4. Discussion
4.1. Intended Application of Guideline Values
4.2. Uncertainties and Limitations Surrounding Exposure Assumptions for Calculation of Guideline Values
4.3. Limitations and Uncertainties in Selection of Critical Toxicity Studies for TDI Derivation
4.3.1. Anatoxin-a
4.3.2. Cylindrospermopsin
4.3.3. Microcystins
4.3.4. Saxitoxins
4.4. Comparison with Guideline Values Developed by Other Nations and States
4.4.1. Anatoxin-a
4.4.2. Cylindrospermopsin
4.4.3. Microcystins
4.4.4. Saxitoxins
4.5. Additional Cyanotoxins
4.6. Benefits of Toxin Based Monitoring
5. Conclusions
Acknowledgments
Author Contributions
Abbreviations
| ATSDR | Agency for Toxic Substances and Disease Registry |
| BMAA | α-amino-β-methylaminoproprionic acid |
| BMD | Benchmark Dose |
| BW | Body weight |
| BWIR | Body weight normalized intake rate |
| C | C-toxins |
| CALOEHHA | California EPA’s Office of Environmental Health Hazard Assessment |
| dcSTXs | 11-hydroxy-STX and decarbamoylsaxitoxins |
| EFSA | European Food Safety Authority |
| ELISA | enzyme linked immunosorbent assay |
| EPA | U.S. Environmental Protection Agency |
| GTX | Gonyautoxins |
| HAB | Harmful algae bloom |
| IR | Intake rate |
| LOAEL | Lowest observable adverse effect level |
| LPS | Lipopolysaccharide |
| neoSTX | Neosaxitoxin |
| NOAEL | No observable adverse effect level |
| OHA | Oregon Health Authority |
| POD | Point of departure |
| RfD | Oral reference dose |
| RSC | Relative source contribution |
| STX | Saxitoxin |
| STX-eq | Saxitoxin equivalent |
| TBM | Toxin based monitoring |
| TEF | Toxic equivalency factor |
| TDI | Tolerable daily intake |
| UF | Uncertainty factor |
| WHO | World Health Organization |
Conflicts of Interest
References
- Chorus, I.; Bartram, J. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring, and Management; World Health Organization: London, UK, 1999. [Google Scholar]
- Meriluoto, J.A.O.; Spoof, L.E.M. Cyanotoxins: Sampling, sample processing and toxin uptake. Adv. Exp. Med. Biol. 2008, 619, 483–499. [Google Scholar]
- ARCHIVED: Cyanobacterial Toxins-Microcystin-LR [Technical document-Chemical/Physical Parameters]. Available online: http://www.hc-sc.gc.ca/ewh-semt/pubs/water-eau/cyanobacterial_toxins/index-eng.php (accessed on 11 November 2014).
- United States Environmental Protection Agency. Exposure Factors Handbook; National Center for Environment Assessment Office of Research and Development: Washington, DC, USA, 2011; Document EPA/600/R-090/052F.
- Agency for Toxic Substances and Disease Registry. Public Health Assessment Guidance Manual; US Department of Health and Human Services: Atlanta, GA, USA, 2005.
- California Environmental Protection Agency (CalEPA). Toxicological Summary and Suggested Action Levels to Reduce Potential Adverse Health Effects of Six Cyanotoxins; CalEPA: Sacramento, CA, USA, 2012.
- Astrachan, N.B.; Archer, B.G. Simplified monitoring of anatoxin-a by reverse-phase high performance liquid chromatography and the sub-acute effects of anatoxin-a in rats. In The Water Environment: Algal Toxins and Health; Carmichael, W.W., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 437–446. [Google Scholar]
- Astrachan, N.B.; Archer, B.G.; Hilbelink, D.R. Evaluation of the subacute toxicity and teratogenicity of anatoxin-a. Toxicon. Off. J. Int. Soc. Toxinol. 1980, 18, 684–688. [Google Scholar] [CrossRef]
- Burch, M.D. Effective doses, guidelines & regulations. Adv. Exp. Med. Biol. 2008, 619, 831–853. [Google Scholar]
- Codd, G.A.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins: Risk management for health protection. Toxicol. Appl. Pharmacol. 2005, 203, 264–272. [Google Scholar] [CrossRef]
- Duy, T.N.; Lam, P.K.; Shaw, G.R.; Connell, D.W. Toxicology and risk assessment of freshwater cyanobacterial (blue-green algal) toxins in water. Rev. Environ. Contam. Toxicol. 2000, 163, 113–185. [Google Scholar]
- Falconer, I.R.; Humpage, A.R. Health risk assessment of cyanobacterial (blue-green algal) toxins in drinking water. Int. J. Environ. Res. Public Health 2005, 2, 43–50. [Google Scholar] [CrossRef]
- Fawell, J.F.; James, H.A. Toxins from Blue-Green Algae: Toxicological Assessment of Anatoxin-a and a Method for Its Determination in Reservoir Water; Foundation for Water Research: Marlow, UK, 1994. [Google Scholar]
- Fawell, J.K.; Mitchell, R.E.; Hill, R.E.; Everett, D.J. The toxicity of cyanobacterial toxins in the mouse: II anatoxin-a. Hum. Exp. Toxicol. 1999, 18, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Pegram, R.A.; Nichols, T.; Etheridge, S.; Humpage, A.; LeBlanc, S.; Love, A.; Neilan, B.; Pflugmacher, S.; Runnegar, M.; Thacker, R. Cyanotoxins Workgroup report. Adv. Exp. Med. Biol. 2008, 619, 317–381. [Google Scholar] [PubMed]
- Rogers, E.H.; Hunter, E.S.; Moser, V.C.; Phillips, P.M.; Herkovits, J.; Munoz, L.L.; Hall, L.; Chernoff, N. Potential developmental toxicity of anatoxin-a, a cyanobacterial toxin. J. Appl. Toxicol. 2005, 25, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Van Apeldoorn, M.E.; van Egmond, H.P.; Speijers, G.J.A.; Bakker, G.J.I. Toxins of cyanobacteria. Mol. Nutr. Food Res. 2007, 51, 7–60. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Policies and Guidelines. Available online: http://www2.epa.gov/nutrient-policy-data/policies-and-guidelines (accessed on 13 November 2014).
- Washington Department of Health. Washington State Recreational Guidance for Cylindrospermopsin (Provisional) and Saxitoxin (Provisional); Washington Department of Health: Olympia, WA, USA, 2011.
- United States Environmental Protection Agency. Toxicological Reviews of Cyanobacterial Toxins: Anatoxin-a (External Review Draft); US Environmental Protection Agency: Washington, DC, USA, 2006; Document NCEA-C-1743.
- New Zealand Ministry of Health. Drinking-Water Standards for New Zealand 2005 (Revised 2008); Ministry of Health: Wellington, New Zealand, 2008.
- Ohio Environmental Protection Agency. Public Water System Harmful Algal Bloom Response Strategy; Ohio EPA: Columbus, OH, USA, 2014.
- United States Environmental Protection Agency. Toxicological Reviews of Cyanobacterial Toxins: Cylindrospermopsin; US Environmental Protection Agency: Washington, DC, USA, 2006; Document NCEA-C-1763.
- Humpage, A.R.; Falconer, I.R. Oral toxicity of the cyanobacterial toxin cylindrospermopsin in male swiss albino mice: Determination of no observable adverse effect level for deriving a drinking water guideline value. Environ. Toxicol. 2003, 18, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Heinze, R. Toxicity of the cyanobacterial toxin microcystin-LR to rats after 28 days intake with the drinking water. Environ. Toxicol. 1999, 14, 57–60. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific Opinion: Marine biotoxins in shellfish-Saxitoxin group. EFSA J. 2009, 1019, 1–76. [Google Scholar]
- Sukenik, A.; Reisner, M.; Carmeli, S.; Werman, M. Oral toxicity of the cyanobacterial toxin cylindrospermopsin in mice: Long-term exposure to low doses. Envirion. Toxicol. 2006, 21, 575–582. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Toxicological Reviews of Cyanobacterial Toxins: Microcystins LR, RR, YR and LA; US Environmental Protection Agency: Washington, DC, USA, 2006; Document NCEA-C-1765.
- Gupta, N.; Pant, S.C.; Vijayaraghavan, R.; Lakshmana Rao, P.V. Comparative toxicity evaluation of cyanobacterial cyclic peptide toxin microcystin variants (LR, RR, YR) in Mice. Toxicology 2003, 188, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Vesterkvist, P.S.M.; Misiorek, J.O.; Spoof, L.E.M.; Toivola, D.M.; Meriluoto, J.A.O. Comparative Cellular Toxicity of Hydrophilic and Hydrophobic Microcystins on Caco-2 Cells. Toxins 2012, 4, 1008–1023. [Google Scholar] [CrossRef] [PubMed]
- Berry, J.P.; Lee, E.; Walton, K.; Wilson, A.E.; Bernal-Brooks, F. Bioaccumulation of microcystins by fish associated with a persistent cyanobacterial bloom in Lago De Patzcuaro (Michoacan, Mexico). Environ. Toxicol. Chem. 2011, 30, 1621–1628. [Google Scholar] [CrossRef] [PubMed]
- Saqrane, S.; Oudra, B. CyanoHAB occurrence and water irrigation cyanotoxin contamination: ecological impacts and potential health risks. Toxins 2009, 1, 113–122. [Google Scholar] [CrossRef] [PubMed]
- De la Cruz, A.A.; Hiskia, A.; Kaloudis, T.; Chernoff, N.; Hill, D.; Antoniou, M.G.; He, X.; Loftin, K.; O’Shea, K.; Zhao, C.; et al. A review on cylindrospermopsin: The global occurrence, detection, toxicity, and degradation of a potent cyanotoxin. Environ. Sci. Process. Impacts 2013, 13, 49–58. [Google Scholar]
- Fawell, J.K.; Mitchell, R.E.; Everett, D.J.; Hill, R.E. The toxicity of cyanobacterial toxins in the mouse: I microcystin-LR. Hum. Exp. Toxicol. 1999, 18, 162–167. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Benchmark Dose Technical Guidance; US Environmental Protection Agency: Washington, DC, USA, 2012; Document EPA/100/R-12/001.
- Reference Dose (RfD): Description and Use in Health Risk Assessments. Available online: http://www.epa.gov/iris/rfd.htm (accessed on 17 September 2012).
- Ueno, Y.; Makita, Y.; Nagata, S.; Tsutsumi, T.; Yoshida, F.; Tamura, S.I.; Sekijima, M.; Tashiro, F.; Harada, T.; Yoshida, T. No chronic oral toxicity of a low dose of microcystin-LR, a cyanobacterial hepatotoxin, in female BALB/c mice. Environ. Toxicol. 1999, 14, 45–55. [Google Scholar] [CrossRef]
- Pilotto, L.; Hobson, P.; Burch, M.D.; Ranmuthugala, G.; Attewell, R.; Weightman, W. Acute skin irritant effects of cyanobacteria (Blue-Green Algae) in healthy volunteers. Aust. N. Z. J. Public Health 2004, 28, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Chiu, A.S.; Gehringer, M.M.; Welch, J.H.; Neilan, B.A. Does α-Amino-β-Methylaminopropionic Acid (BMAA) play a role in neurodegeneration? Int. J. Environ. Res. Public Health 2011, 8, 3728–3746. [Google Scholar] [CrossRef] [PubMed]
- Kisby, G.E.; Spencer, P.S. Is neurodegenerative disease a long-latency response to early-life genotoxin exposure? Int. J. Environ. Res. Public Health 2011, 8, 3889–3921. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, R.A.; Cox, P.A.; Banack, S.A.; Rodgers, K.J. The non-protein amino acid BMAA is misincorporated into human proteins in place of l-serine causing protein misfolding and aggregation. PLoS One 2013, 8, e75376. [Google Scholar] [CrossRef] [PubMed]
- Larkin, S.L.; Adams, C.M. Harmful algal blooms and coastal business: Economic consequences in Florida. Soc. Nat. Resour. 2007, 20, 849–859. [Google Scholar] [CrossRef]
- Hudnell, H.K. The state of U.S. freshwater harmful algal blooms assessments, policy and legislation. Toxicon 2010, 55, 1024–1034. [Google Scholar]
- Hoagland, P.; Anderson, D.; Kaoru, M.Y.; White, A.W. The economic effects of harmful algal blooms in the United States: Estimates, assessment issues, and information needs. Estuaries 2002, 25, 819–837. [Google Scholar] [CrossRef]
- Dodds, W.K.; Bouska, W.W.; Eitzmann, J.L.; Pilger, T.J.; Pitts, K.L.; Riley, A.J.; Schloesser, J.T.; Thornbrugh, D.J. Eutrophication of U.S. freshwaters: Analysis of potential economic damages. Environ. Sci. Technol. 2009, 43, 12–19. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farrer, D.; Counter, M.; Hillwig, R.; Cude, C. Health-Based Cyanotoxin Guideline Values Allow for Cyanotoxin-Based Monitoring and Efficient Public Health Response to Cyanobacterial Blooms. Toxins 2015, 7, 457-477. https://doi.org/10.3390/toxins7020457
Farrer D, Counter M, Hillwig R, Cude C. Health-Based Cyanotoxin Guideline Values Allow for Cyanotoxin-Based Monitoring and Efficient Public Health Response to Cyanobacterial Blooms. Toxins. 2015; 7(2):457-477. https://doi.org/10.3390/toxins7020457
Chicago/Turabian StyleFarrer, David, Marina Counter, Rebecca Hillwig, and Curtis Cude. 2015. "Health-Based Cyanotoxin Guideline Values Allow for Cyanotoxin-Based Monitoring and Efficient Public Health Response to Cyanobacterial Blooms" Toxins 7, no. 2: 457-477. https://doi.org/10.3390/toxins7020457
APA StyleFarrer, D., Counter, M., Hillwig, R., & Cude, C. (2015). Health-Based Cyanotoxin Guideline Values Allow for Cyanotoxin-Based Monitoring and Efficient Public Health Response to Cyanobacterial Blooms. Toxins, 7(2), 457-477. https://doi.org/10.3390/toxins7020457






