1. Introduction
Diarrhetic shellfish poisoning (DSP) is a severe gastrointestinal illness caused by the consumption of shellfish contaminated with diarrhetic shellfish toxins (DSTs) [
1]. Based on their structures, DSTs were initially classified into three groups, okadaic acid (OA)/dinophysistoxin (DTX) analogues, pectenotoxins (PTXs), and yessotoxins (YTXs) [
2,
3]. OA and its analogues, dinophysistoxin-1 (DTX1) and -2 (DTX2), are the most important toxins due to their causing severe diarrhea. These toxins have been shown to be potent phosphatase inhibitors [
4], a property that can cause inflammation of the intestinal tract and diarrhea [
5], possibly leading to tumor promotion [
6]. Because PTXs and YTXs do not cause diarrhea [
7,
8,
9,
10,
11,
12], they are now not considered to be members of the DST group. Because OA/DTXs and PTXs are produced by toxic dinoflagellate
Dinophysis spp., OA/DTXs are regulated together with PTXs in European Union (EU). The regulatory level of sum of OA/DTXs and PTXs in EU is 0.16 mg/kg. The regulatory level of YTXs in EU is 3.75 mg/kg. On the other hand, the regulatory level of OA/DTXs suggested by CODEX is 0.16 mg/kg. With the change in the definition of DSTs in Japan in April 2015, the MBA as the Japanese official testing method for DSTs was replaced to instrumental methods including LC/MS/MS to detect OA analogues exclusively on the CODEX regulatory level. The regulation in Japan is the same as that of the US.
The Japanese scallop (
Patinopecten yessoensis) is an important aquaculture species in Japan in terms of the annual quantity of production and contamination with DSTs and other lipophilic toxins is a serious industrial problem. DST contamination of Japanese scallops is caused by feeding on toxic dinoflagellates (
Dinophysis spp.) that produce OA, DTX1 and PTX2 [
13,
14,
15,
16] (
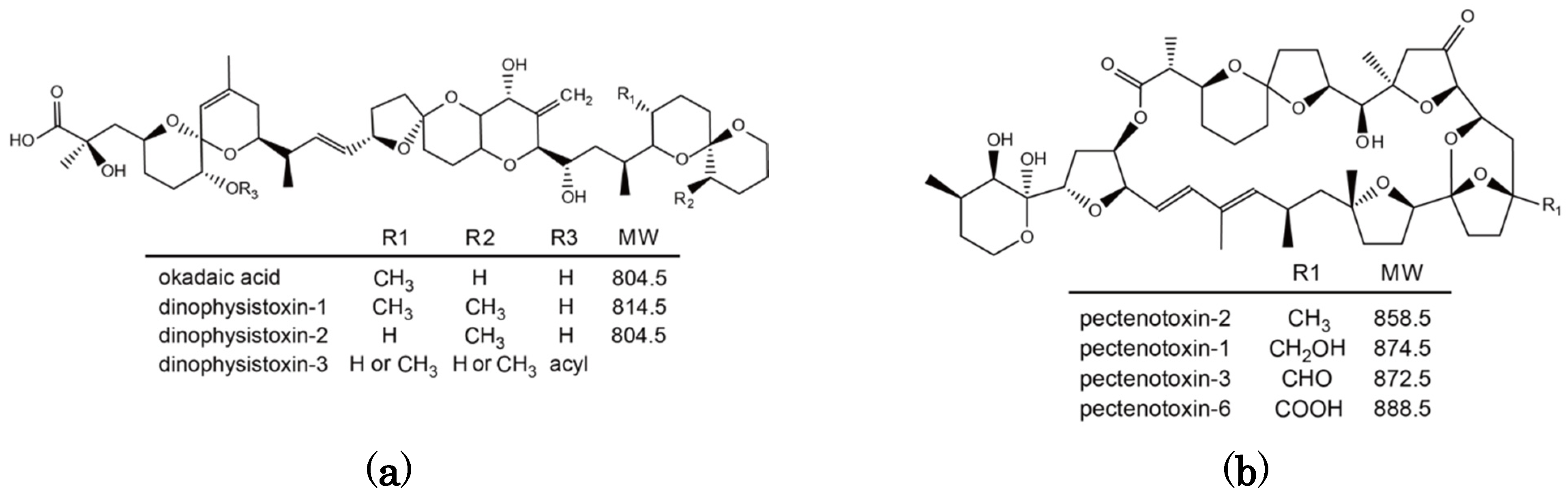
Figure 1).
Figure 1.
Chemical structure of okadaic acid/dinophysistoxins (a) and pectenotoxins (b).
Figure 1.
Chemical structure of okadaic acid/dinophysistoxins (a) and pectenotoxins (b).
OA and DTX1 are metabolized to the esterified toxin 7-
O-acyl-OA/DTX1 in many bivalve species including Japanese scallops [
17,
18]. They are collectively called dinophysistoxin-3 (DTX3). PTX2 is enzymatically hydrolyzed to PTX2 seco acid in many bivalve species [
10,
19,
20,
21], but in
P. yessoensis PTX2 is oxidatively metabolized to pectenotoxin-1 (PTX1), pectenotoxin-3 (PTX3), and pectenotoxin-6 (PTX6) [
22,
23]. PTX6 is the dominant toxin that accumulates in
P. yessoensis and this particular mode of metabolism has only been observed in this species [
24].
Although quantification of toxicity of individual tissues of
P. yessoensis by the mouse bioassay (MBA) has demonstrated that the toxins appear to be mainly accumulated in the digestive gland [
1], a detailed investigation on the distribution of DSTs and other lipophilic toxins in individual tissues of Japanese scallops has not been carried out. In a previous study, the absorption efficiency of DTX1 by the digestive gland of
P. yessoensis was estimated at less than 3% of the total amount of DTX1 fed to the scallops in
D.
fortii cells [
17]. When a mixture of OA, DTX1, PTX6, and YTX was injected into the digestive gland, less than 20% was retained although the residual relative amount of PTX6 was slightly higher than that of OA and DTX1 [
18]. Bay scallops (
Argopecten irradians) exposed to cultured cells of
Prorocentrum lima showed a toxin-assimilation efficiency in the scallop tissues of less than 1% [
25]. Despite these investigations on the accumulation and metabolism of DSTs and other lipophilic toxins in scallops over the past twenty years, details on the accumulation kinetics of each toxin in individual tissues has not been clarified due to the inability to culture
Dinophysis species and inadequate analytical techniques.
In this study, we describe for the first time the detailed assimilation, accumulation, and metabolism of DSTs and PTXs in individual tissues of P. yessoensis. For unambiguous understanding of our research, terms in our present study are defined as follows:
Assimilation: Cells/toxins that are filtered from the water.
Accumulated toxins: Toxins and their metabolites that are incorporated into tissues.
Excretion: Toxins that are simply not retained/accumulated by the organism and pass back into the water with or without metabolism.
3. Discussion
Japanese scallops,
Patinopecten yessoensis, were fed with the toxic dinoflagellate
Dinophysis fortii to elucidate the relative magnitude of assimilation, accumulation, and metabolism of diarrhetic shellfish toxins (DSTs) and pectenotoxins (PTXs). The cellular content of PTX2 in
D. fortii was higher than those of DTX1 as reported in natural and cultured
D. fortii cells [
15,
26]. In our previous study on cultured
D. fortii, an increase in the cellular toxin content in exponential growth phase was observed [
26]. A similar trend was observed in our present study. It is interesting that
D. fortii collected in Japan only produce free DTX1 and OA in contrast that
D. acuta in New Zealand produces several OA diol esters [
27].
It was reported that the toxicity of scallops contaminated with DSTs determined by mouse bioassay (MBA) was exclusively detected in the digestive gland and the toxicity of gills, mantles, gonads, and adductor muscles were less than detectable levels [
1]. Although the higher toxin content of the digestive gland in comparison with other tissues obtained by LC/MS/MS was consistent with the previous study [
1], contamination of toxins in other Japanese scallop tissues was confirmed for the first time.
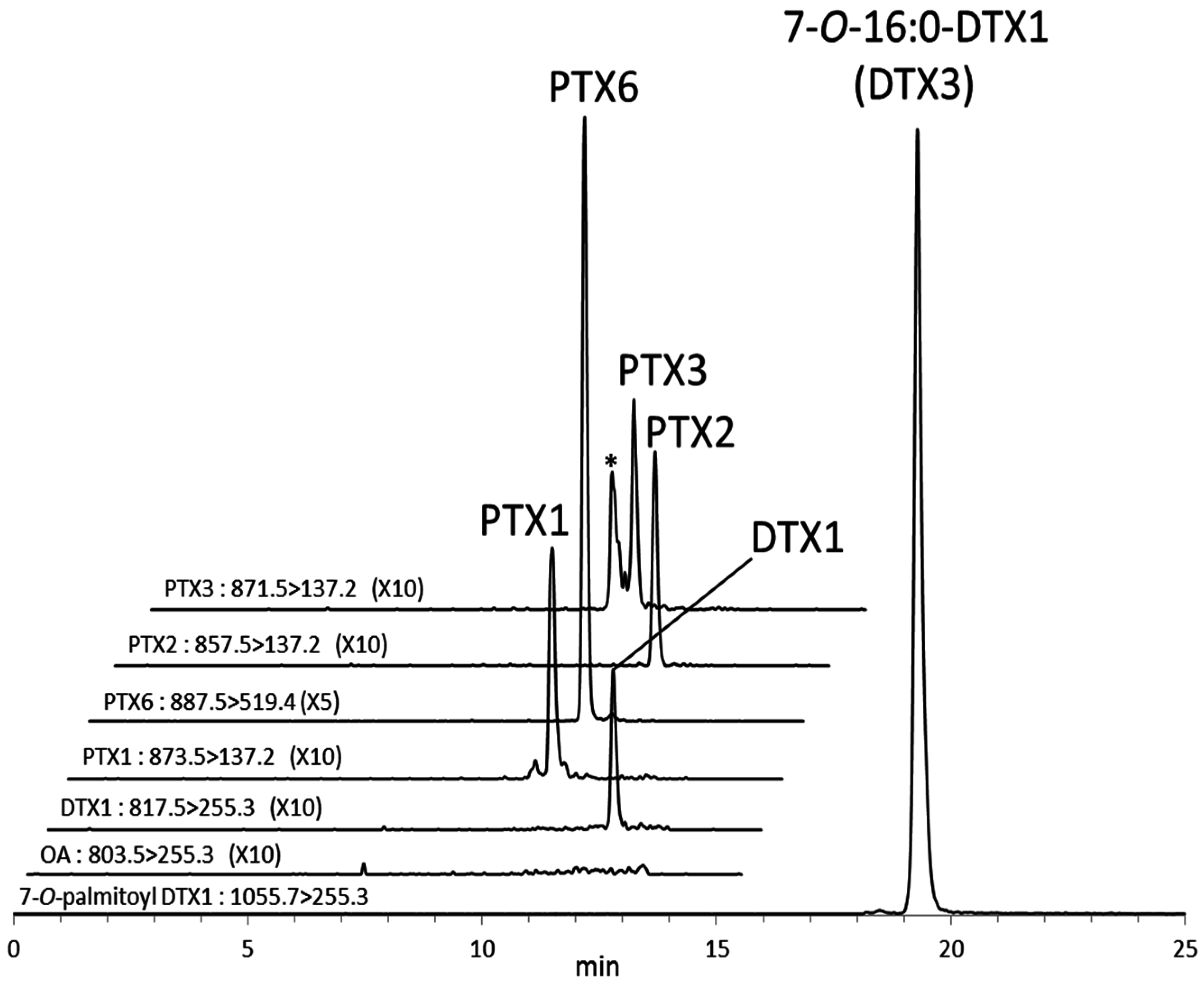
The increase of the DTX1 concentration in the digestive gland samples subjected to alkaline hydrolysis was higher than that obtained by direct quantification of DTX3 (7-
O-16:0-DTX1) (
Figure 4F). This suggests the presence of other DTX3 homologues esterified with other fatty acyl groups such as 18:0 and 20:5 [
28]. PTX6 and DTX3 were dominant toxins in the digestive gland of scallops. These results confirm the rapid biotransformation of PTX2 and DTX1 to their respective metabolites and accumulation in the digestive gland of Japanese scallops as reported in our previous studies [
17,
23]. This is assumed to be a detoxification mechanism within the scallop tissues because the major metabolites (PTX6 and DTX3) have lower toxicities than their parent compounds (PTX2 and DTX1) [
1,
22,
24,
29].
The higher proportion of PTX2 in the gills in comparison with other tissues is noteworthy. This suggests that PTX2 is selectively retained on the gills and this tissue may take part in some metabolic activities in conversion of PTX2 to other analogues. PTXs may be distributed to other organs for metabolism, while DTX1 appears to be accumulated and metabolized mainly in the digestive gland. PTXs and DTXs are both lipophilic constituents but it is suggested they pass through different metabolic pathways.
Accumulation rates of DTXs in the digestive gland reported in our previous study of scallops fed with natural
D. fortii was less than 3% [
17]. The accumulation of DTXs obtained in our present study was higher than that reported previously (
Table 3). The difference in the feeding regimes between the previous and the present studies was the planktonic compositions fed to scallops. In our previous study,
D. fortii collected from natural seawater was fed to scallops with other phytoplankton although the planktonic composition was dominated by
D. fortii. In the present study, scallops were fed with uni-algal cultured
D. fortii. Feeding on other plankton species besides
Dinophysis may affect accumulation rate of toxins in scallops.
The marked increase in specific toxins in the waters used to house the scallop in the evening of fourth day could be explained by relatively high cell numbers of
D. fortii fed to the scallops (
Table 1 and
Table 4). An interesting finding of our present study is that 54%–75% of PTX2 and 52%–70% of DTX1 assimilated by the scallops was directly excreted to the environmental seawater in the form of DTX1 and PTX2 mainly without metabolic processing.
5. Conclusions
Detailed DSTs and PTXs accumulation kinetics in Japanese scallops fed with the toxic dinoflagellate D. fortii was clarified in our present study. Toxins were mainly accumulated in the digestive gland although low levels of toxins were detected in the gills, mantles, gonads, and adductor muscles. The toxin concentrations in tissues were in the order of: digestive gland > gill > mantle > gonad > adductor muscle. Because the amount of toxin in the adductor muscle was extremely low, the validity of the practise of evisceration of Japanese scallops contaminated with DSTs (implemented since 1980 in Japan) was confirmed. The accumulation efficiencies of PTXs and DTXs in the scallops, (accumulated toxins versus assimilated toxins) were 21%–39% and 7%–23%, respectively. The higher the accumulation efficiency of PTXs in comparison to OA/DTXs could partly contribute to the higher concentrations of PTXs in toxin profiles of Japanese scallops. An interesting finding of our present study is that 54%–75% of PTX2 and 52%–70% of DTX1 assimilated by the scallops was directly excreted to the environmental seawater in the form of DTX1 and PTX2 mainly without metabolic processing.
There is a concern about the limited numbers of scallops (n = 3) used. While the patterns do seem quite consistent between these, using a larger sample would provide a stronger basis for analysis and conclusions.