Interaction of Citrinin with Human Serum Albumin
Abstract
:1. Introduction
2. Results and Discussion
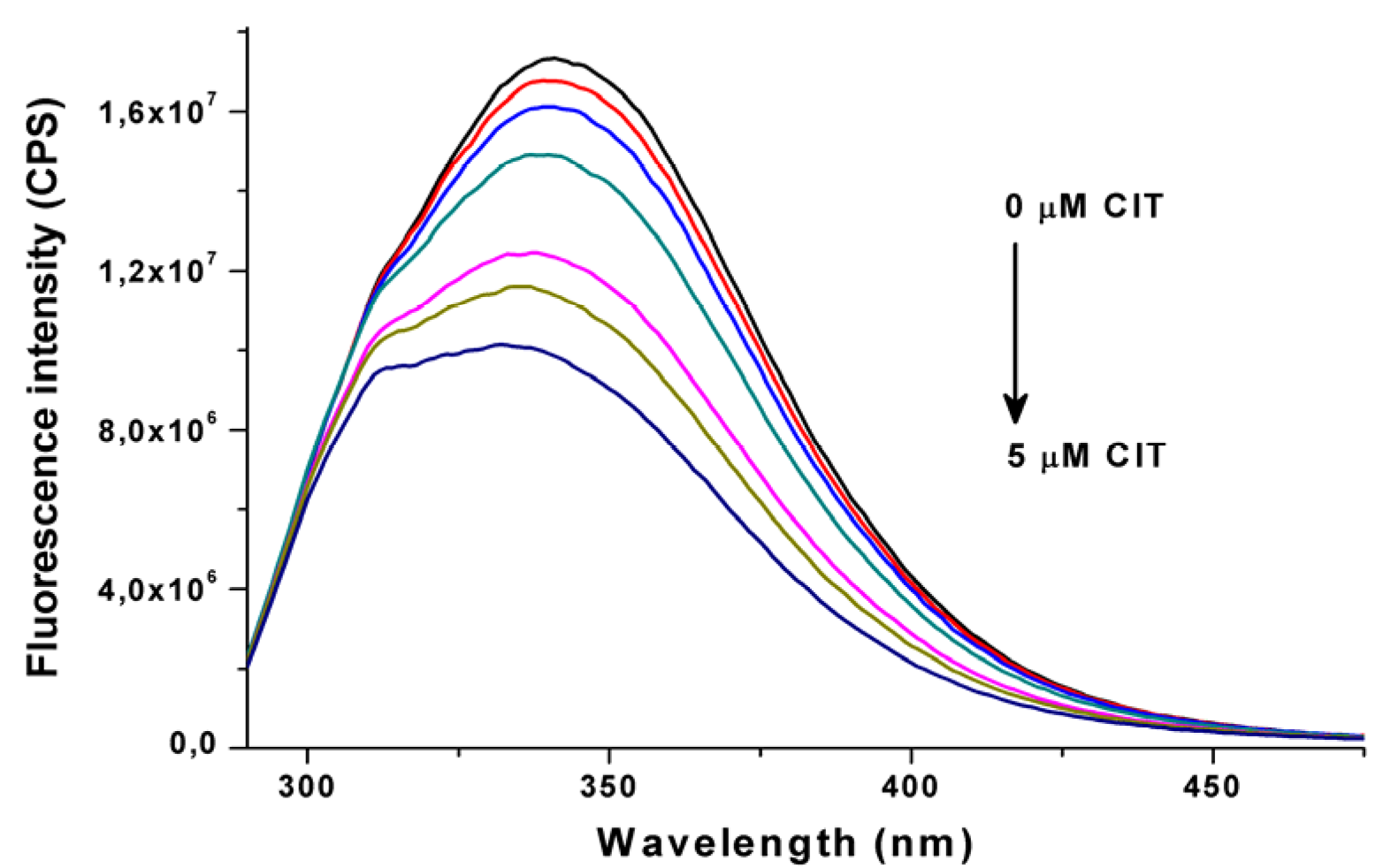
2.1. Binding Constant of Citrinin-HSA Complex
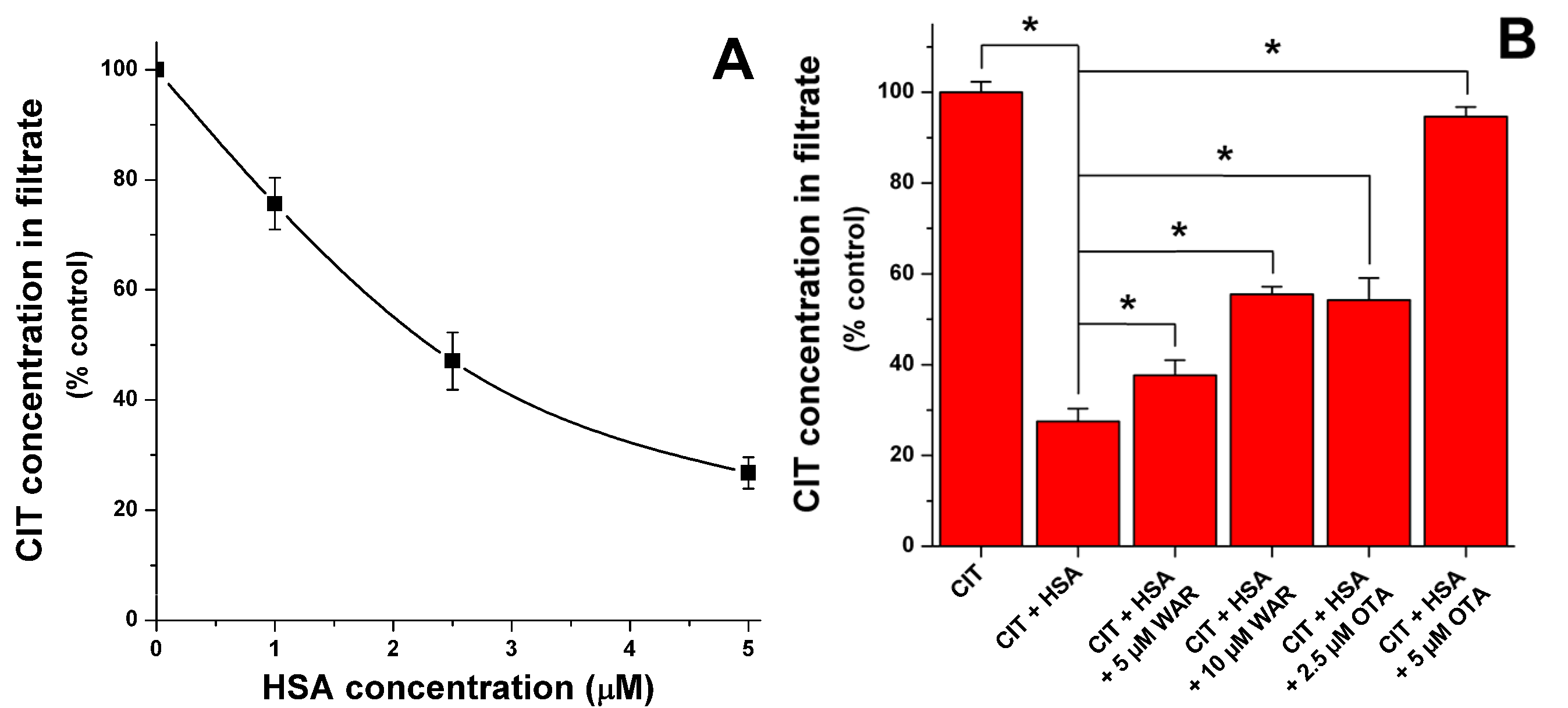
2.2. Ultrafiltration Experiments
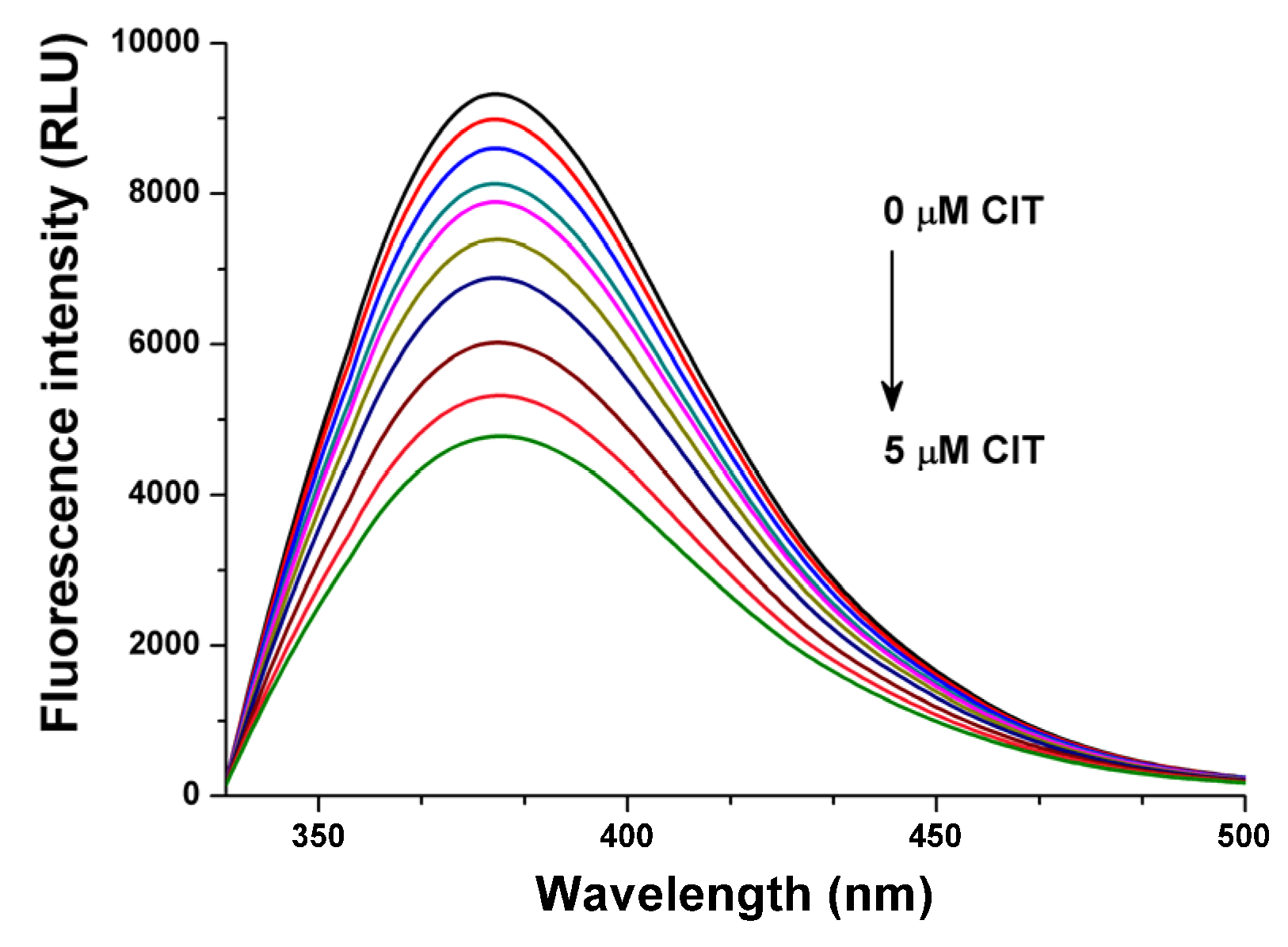
2.3. Fluorescence Investigation of Molecular Displacement of Warfarin and Ochratoxin A from HSA by Citrinin
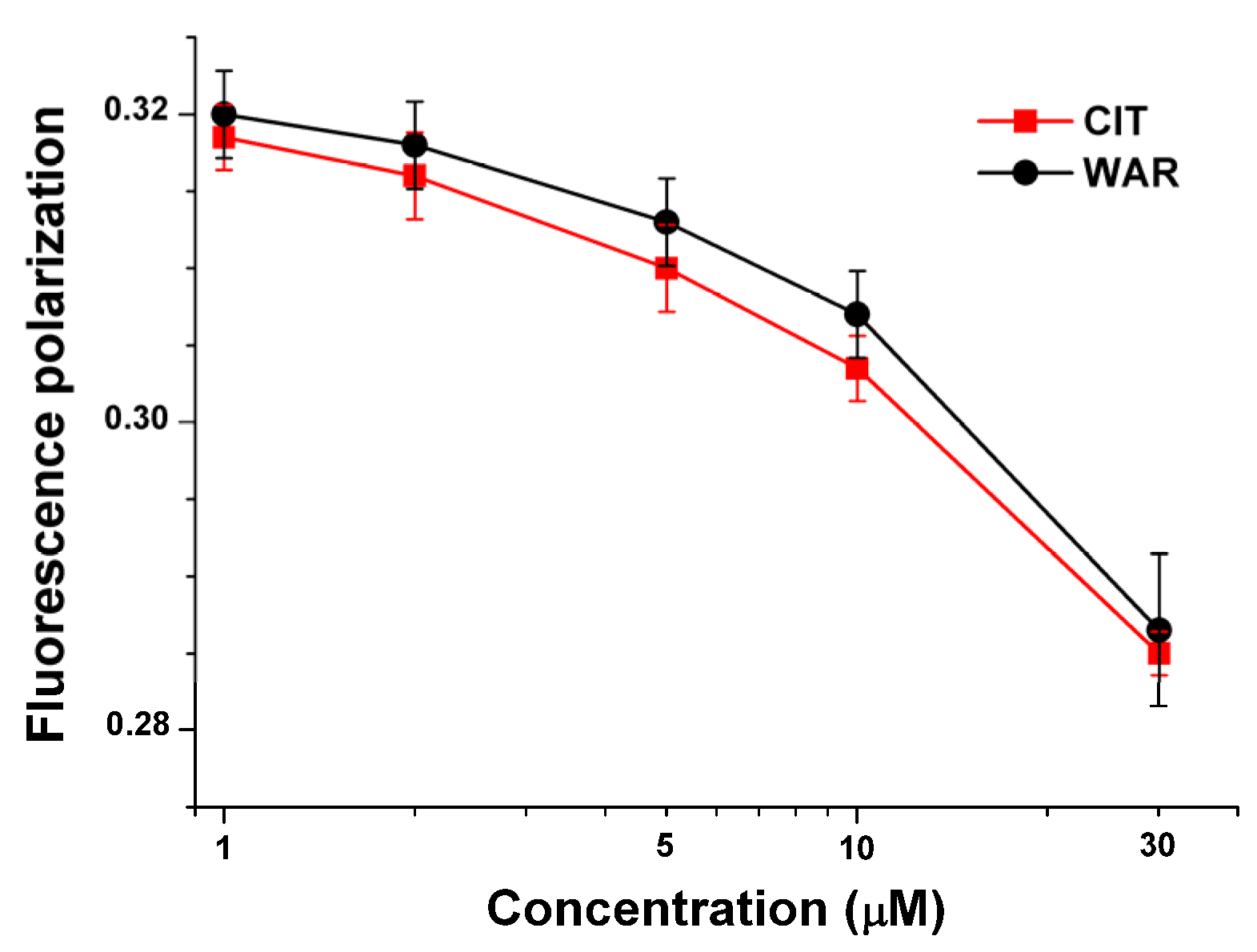
2.4. Thermodynamic Studies
| T (°C) | 25 | 30 | 35 | 40 |
| logK (±SD) (CIT-HSA) | 5.32 ± 0.01 | 5.26 ± 0.01 | 5.19 ± 0.01 | 5.11 ± 0.01 |
| T (°C) | 25 | - | 37 | 42 |
| logK (warfarin-HSA) | 5.38 | - | 5.31 | 5.28 |
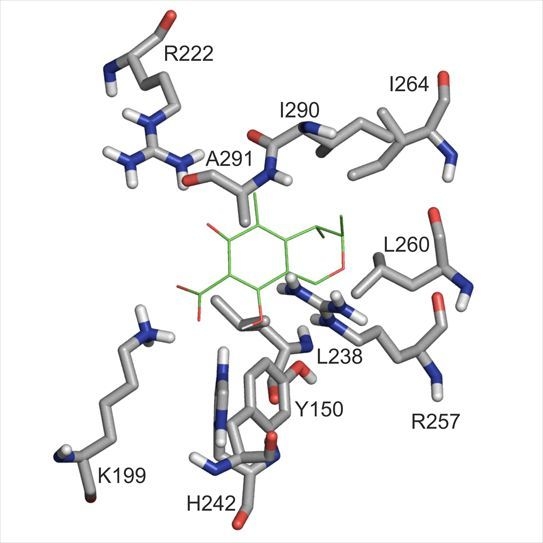
2.5. Molecular Modeling Studies

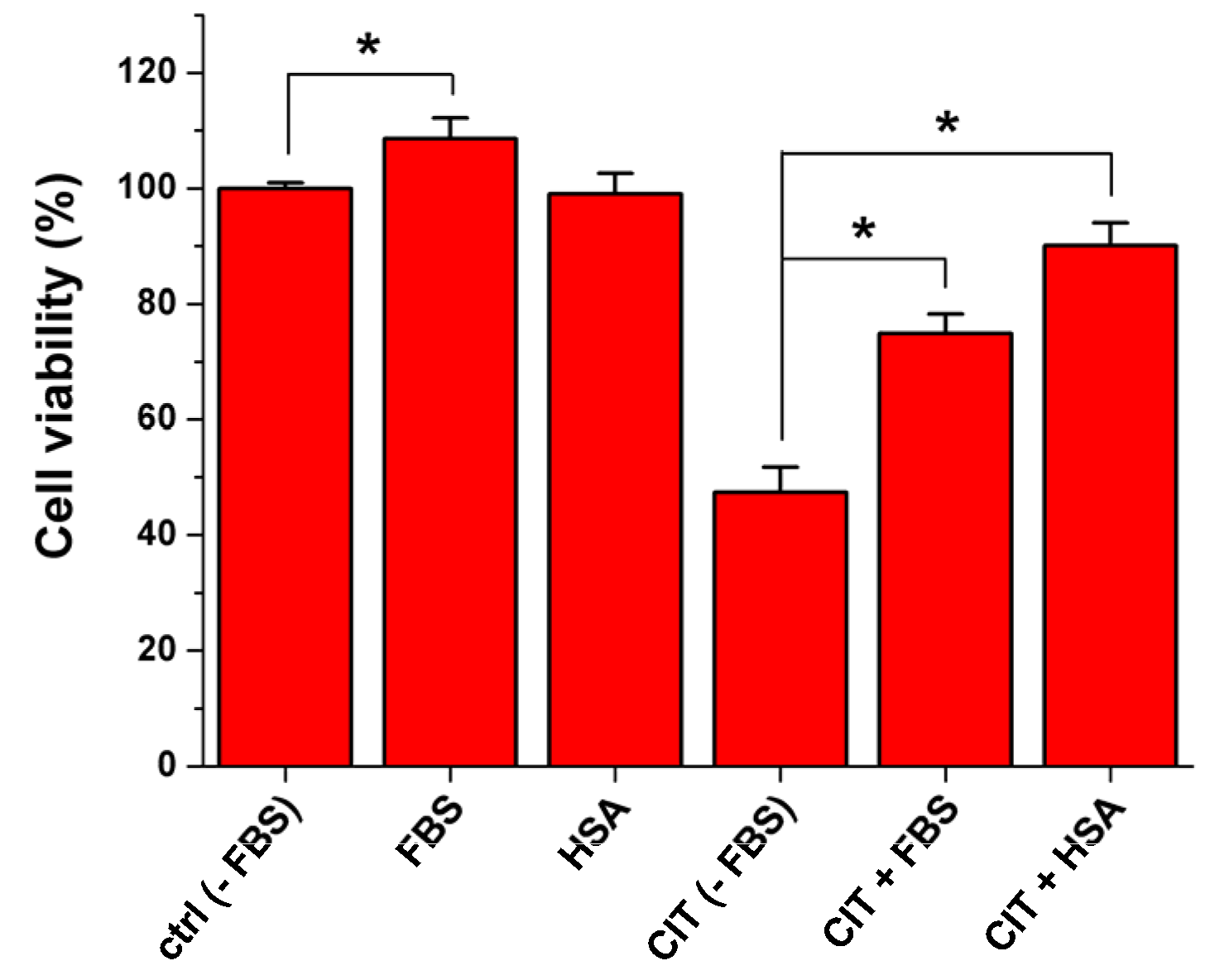
2.6. Influence of Albumin on the Citrinin-Induced Toxicity in Vitro
2.7. Interaction of Citrinin with Bovine, Porcine, and Rat Serum Albumin
| Tested albumins | HSA | BSA | PSA | RSA |
|---|---|---|---|---|
| logK (±SD) | 5.32 ± 0.01 | 5.05 ± 0.05 | 4.96 ± 0.05 | 5.50 ± 0.02 |
3. Experimental Section
3.1. Reagents
3.2. Fluorescence Spectroscopic Measurements
3.3. Thermodynamic Studies
3.4. Ultrafiltration Studies
3.5. Molecular Modeling Studies
3.6. Tissue Culture and Cell Viability Measurements
3.7. Statistics
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Da Rocha, M.E.B.; da Chagas Oliveira Freire, F.; Maia, F.E.F.; Guedes, M.I.F.; Rondina, D. Mycotoxins and their effects on human and animal health. Food Contr. 2014, 36, 159–165. [Google Scholar] [CrossRef]
- Flajs, D.; Peraica, M. Toxicological properties of citrinin. Arh. Hig. Rada Toksikol. 2009, 60, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Ostry, V.; Malir, F.; Ruprich, J. Producers and important dietary sources of ochratoxin A and citrinin. Toxins (Basel) 2013, 5, 1574–1586. [Google Scholar] [CrossRef] [PubMed]
- Kitabatake, N.; Trivedi, A.B.; Doi, E. Thermal decomposition and detoxification of citrinin under various moisture conditions. J. Agric. Food Chem. 1991, 39, 2240–2244. [Google Scholar] [CrossRef]
- Chan, W.-H. Citrinin induces apoptosis via a mitochondria-dependent pathway and inhibition of survival signals in embryonic stem cells, and causes developmental injury in blastocysts. Biochem. J. 2007, 404, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-H.; Yu, F.-Y.; Wu, T.-S.; Wang, L.-T.; Liu, B.-H. Mycotoxin Citrinin Induced Cell Cycle G2/M arrest and numerical chromosomal aberration associated with disruption of microtubule formation in human cells. Toxicol. Sci. 2011, 119, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, K.; Ishii, Y.; Takasu, S.; Kijima, A.; Matsushita, K.; Watanabe, M.; Takahashi, H.; Sugita-Konishi, Y.; Sakai, H.; Yanai, T.; et al. Cell cycle progression, but not genotoxic activity, mainly contributes to citrinin-induced renal carcinogenesis. Toxicology 2013, 311, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Berndt, W.O.; Hayes, A.W. The Effect of Probenecid on Citrinin-Induced Nephrotoxicity. Toxicol. Appl. Pharmacol. 1982, 64, 118–124. [Google Scholar] [CrossRef]
- Berndt, W.O. Transport of citrinin by rat renal cortex. Arch. Toxicol. 1983, 54, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Föllmann, W.; Behm, C.; Degen, G.H. Toxicity of the mycotoxin citrinin and its metabolite dihydrocitrinone and of mixtures of citrinin and ochratoxin A in vitro. Arch. Toxicol. 2014, 88, 1097–1107. [Google Scholar] [CrossRef] [PubMed]
- Blaszkewicz, M.; Munoz, K.; Degen, G.H. Methods for analysis of citrinin in human blood and urine. Arch. Toxicol. 2013, 87, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Blaszkewicz, M.; Degen, G.H. Occurrence of the mycotoxin citrinin and its metabolite dihydrocitrinone in urines of German adults. Arch. Toxicol. 2015, 89, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Fanali, G.; Di Masi, A.; Trezza, V.; Marino, M.; Fasano, M.; Ascenzi, P. Human serum albumin: From bench to bedside. Mol. Aspects Med. 2012, 33, 209–290. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, K.; Chuang, V.T.; Maruyama, T.; Otagiri, M. Albumin-drug interaction and its clinical implication. Biochim. Biophys. Acta 2013, 1830, 5435–5443. [Google Scholar] [CrossRef] [PubMed]
- Poór, M.; Li, Y.; Matisz, G.; Kiss, L.; Kunsági-Máté, S.; Kőszegi, T. Quantitation of species differences in albumin-ligand interactions for bovine, human and rat serum albumins using fluorescence spectroscopy: A test case with some Sudlow’s site I ligands. J. Lumin. 2014, 145, 767–773. [Google Scholar] [CrossRef]
- Li, Y.; Czibulya, Z.; Poór, M.; Lecomte, S.; Kiss, L.; Harte, E.; Kőszegi, T.; Kunsági-Máté, S. Thermodynamic study of the effects of ethanol on the interaction of ochratoxin A with human serum albumin. J. Lumin. 2014, 148, 18–25. [Google Scholar] [CrossRef]
- Damodaran, C. In vitro binding of citrinin to serum protein. Experientia 1977, 33, 598–599. [Google Scholar] [CrossRef] [PubMed]
- Damodaran, C.; Shanmugasundaram, E. Distribution of radioactive citrinin in tissues and serum protein(s). J. Radioanal. Chem. 1978, 46, 373–377. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, J.; Dong, L.; Lu, L.; Chen, F.; Hu, D.; Wang, X. A study of fluorescence properties of citrinin in β-cyclodextrin aqueous solution and different solvents. J. Lumin. 2012, 132, 1437–1445. [Google Scholar] [CrossRef]
- Poór, M.; Li, Y.; Kunsági-Máté, S.; Petrik, J.; Vladimir-Knežević, S.; Kőszegi, T. Molecular displacement of warfarin from human serum albumin by flavonoid aglycones. J. Lumin. 2013, 142, 122–127. [Google Scholar] [CrossRef]
- Li, X.; Wang, F.; Xu, B.; Yu, X.; Yang, Y.; Zhang, L.; Li, H. Determination of the free and total concentrations of vancomycin by two-dimensional liquid chromatography and its application in elderly patients. J. Chromatogr. B 2014, 969, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.Q.; Li, Y.J.; Li, Z.; Bian, T.T.; Chen, K.; Zheng, X.X.; Yu, Y.Y.; Jiang, S.S. Study on the interaction of plasma protein binding rate between edaravone and taurine in human plasma based on HPLC analysis coupled with ultrafiltration technique. Biomed. Chromatogr. 2015, 29, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Il’ichev, Y.V.; Perry, J.L.; Rüker, F.; Dockal, M.; Simon, J.D. Interaction of ochratoxin A with human serum albumin. Binding sites localized by competitive interactions with the native protein and its recombinant fragments. Chem. Biol. Interact. 2002, 141, 275–293. [Google Scholar] [CrossRef]
- Il’ichev, Y.V.; Perry, J.L.; Simon, J.D. Interaction of Ochratoxin A with Human Serum Albumin. A Common Binding Site of Ochratoxin A and Warfarin in Subdomain IIA. J. Phys. Chem. B 2002, 106, 460–465. [Google Scholar] [CrossRef]
- Poór, M.; Kunsági-Máté, S.; Bencsik, T.; Petrik, J.; Vladimir-Knežević, S.; Kőszegi, T. Flavonoid aglycones can compete with Ochratoxin A for human serum albumin: A new possible mode of action. Int. J. Biol. Macromol. 2012, 51, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Poór, M.; Kunsági-Máté, S.; Czibulya, Z.; Li, Y.; Peles-Lemli, B.; Petrik, J.; Vladimir-Knežević, S.; Kőszegi, T. Fluorescence spectroscopic investigation of competitive interactions between ochratoxin A and 13 drug molecules for binding to human serum albumin. Luminescence 2013, 28, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Ross, P.D.; Subramanian, S. Thermodynamics of protein association reactions: Forces contributing to stability. Biochemistry 1981, 20, 3096–3102. [Google Scholar] [CrossRef] [PubMed]
- Oester, Y.T.; Keresztes-Nagy, S.; Mais, R.F.; Becktel, J.; Zaroslinski, J.F. Effect of temperature on binding of warfarin by human serum albumin. J. Pharm. Sci. 1976, 65, 1673–1677. [Google Scholar] [CrossRef] [PubMed]
- Abou-Zied, O.K.; Al-Shihi, O.I.K. Characterization of subdomain IIA binding site of human serum albumin in its native, unfolded, and refolded states using small molecular probes. J. Am. Chem. Soc. 2008, 130, 10793–10801. [Google Scholar] [CrossRef] [PubMed]
- Petitpas, I.; Bhattacharya, A.A.; Twine, S.; East, M.; Curry, S. Crystal structure analysis of warfarin binding to human serum albumin anatomy of drug site I. J. Biol. Chem. 2001, 276, 22804–22809. [Google Scholar] [CrossRef] [PubMed]
- Destro, R.; Marsh, R.E. Temperature dependence of tautomeric equilibria in the solid state: The case of citrinin. J. Am. Chem. Soc. 1984, 106, 7269–7271. [Google Scholar] [CrossRef]
- Hetényi, C.; van der Spoel, D. Efficient docking of peptides to proteins without prior knowledge of the binding site. Protein Sci. 2002, 11, 1729–1737. [Google Scholar] [CrossRef] [PubMed]
- Hetényi, C.; van der Spoel, D. Blind docking of drug-sized compounds to proteins with up to a thousand residues. FEBS Lett. 2006, 580, 1447–1450. [Google Scholar] [CrossRef] [PubMed]
- Hetényi, C.; van der Spoel, D. Toward prediction of functional protein pockets using blind docking and pocket search algorithms. Protein Sci. 2011, 20, 880–893. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [PubMed]
- Schrödinger. Release, S. 2: Maestro, Schrödinger. LLC: New York, NY, USA, 2013.
- Appell, M.; Moravec, D.; Bosma, W.B. Quantum chemical study of the structure and properties of citrinin. Mol. Simul. 2012, 38, 284–292. [Google Scholar] [CrossRef]
- Stewart, J.J.P. MOPAC 2012, Stewart Computational Chemistry: Colorado Springs, CO, USA, 2012; Version 14.243L.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poór, M.; Lemli, B.; Bálint, M.; Hetényi, C.; Sali, N.; Kőszegi, T.; Kunsági-Máté, S. Interaction of Citrinin with Human Serum Albumin. Toxins 2015, 7, 5155-5166. https://doi.org/10.3390/toxins7124871
Poór M, Lemli B, Bálint M, Hetényi C, Sali N, Kőszegi T, Kunsági-Máté S. Interaction of Citrinin with Human Serum Albumin. Toxins. 2015; 7(12):5155-5166. https://doi.org/10.3390/toxins7124871
Chicago/Turabian StylePoór, Miklós, Beáta Lemli, Mónika Bálint, Csaba Hetényi, Nikolett Sali, Tamás Kőszegi, and Sándor Kunsági-Máté. 2015. "Interaction of Citrinin with Human Serum Albumin" Toxins 7, no. 12: 5155-5166. https://doi.org/10.3390/toxins7124871
APA StylePoór, M., Lemli, B., Bálint, M., Hetényi, C., Sali, N., Kőszegi, T., & Kunsági-Máté, S. (2015). Interaction of Citrinin with Human Serum Albumin. Toxins, 7(12), 5155-5166. https://doi.org/10.3390/toxins7124871