1. Introduction
After being discovered in 1972 [
1], the Fusarium mycotoxin deoxynivalenol (DON) is one of the most common contaminants of cereal-based food and feed. Since its discovery, a lot of research has been carried out on its occurrence [
2,
3], toxicity [
4], and reduction [
5,
6] as well as on its metabolization by plants [
7], humans [
8], and animals [
9]. Nevertheless, the current knowledge, particularly on metabolization by different animal species, is far from complete.
One metabolization type common to most studied animal species is de-epoxidation. Formation of deepoxy-DON (DOM) is predominantly achieved by the gut microbiota. Its extent varies greatly both between species and also between individuals of one species [
9,
10]. The second metabolization pathway of DON observed in most of the investigated animal species is glucuronidation. Glucuronidation is carried out by endogenous UDP-glucuronosyltransferases in liver, and possibly also in intestinal microsomes [
11]. In general, the extent of glucuronidation and the regiospecificity of the reaction are species-dependent with additional great individual differences [
11,
12,
13].
Both de-epoxidation and glucuronidation have been studied as metabolization pathways of DON for many years. In humans and pigs, de-epoxidation is of minor importance [
14,
15,
16,
17,
18], whereas glucuronidation is a major metabolization step [
8,
14,
18,
19,
20]. In ruminants [
21,
22] and in rats [
23], both de-epoxidation and glucuronidation of DON and of the formed DOM are significant metabolic processes. In addition, very recently two independent research groups discovered sulfonation as the main metabolization pathway of DON in rats [
24,
25]. Poultry, however, shows only minor metabolization of DON by glucuronidation, de-epoxidation, and sulfonation [
26]. In contrast, sulfation was recently discovered as a major metabolization pathway, with DON-3α-sulfate as the predominant metabolic product of DON in chickens and turkeys [
25,
26]. According to Wan and co-workers who used radioisotope counting radio-HPLC for quantitation, DON-3-sulfate in excreta of chickens accounted for 89% ± 7% of the administered dose of DON (2.5 mg/kg body weight (b.w.)) [
25]. Very recently, the toxicokinetic behavior of DON after oral application to turkeys and broiler chickens was investigated, and the peak areas of DON-3-sulfate and DON in plasma were compared [
26]. Five minutes after administration of DON, the DON-3-sulfate to DON ratio in plasma was already 38 for turkeys and almost 2000 for broiler chickens. Thirty minutes after treatment, ratios increased to 141 and to nearly 10,000 for turkeys and broilers, respectively. These data indicate rapid absorption as well as very rapid and extensive metabolization of DON to DON-3-sulfate in both investigated avian species.
Although DOM is a minor metabolite in poultry [
27,
28], de-epoxidation of DON-3-sulfate or rapid sulfation of DOM may still be significant processes. We therefore hypothesized that DOM-3-sulfate might equally be a natural DON metabolite in poultry. The first aim of our work was therefore to synthesize and characterize DOM-3-sulfate and DOM-15-sulfate and to investigate their toxicity on ribosomes, the molecular targets of trichothecenes. The second aim was to develop and validate the first LC-MS/MS-based method for simultaneous quantitation of DON, DOM, DON-, and DOM-sulfate as well as DON- and DOM-sulfonates (DONS and DOMS) in excreta of different poultry species. The third objective of the work was to prove the hypothesis of formation of DON-3-sulfate and DOM-3-sulfate, as well as of DONS and DOMS in poultry consuming feed contaminated with DON and/or DOM. To this end, excreta and, in one occasion, chyme samples from four previous feeding trials with turkeys, chickens, pullets [
28], and roosters [
29,
30] were re-analyzed. In three of these trials, feed consumption was recorded and the biological recoveries of orally administered DON and DOM could be assessed. In addition, the effect of species (turkey and chicken) and concomitant infection with
Ascaridia galli on DON metabolization could be investigated. The results of this work will contribute to the understanding of DON metabolization in poultry.
3. Discussion
Concluding from our results and those published in the most recent literature [
25,
26], DON-3-sulfate and DOM-3-sulfate are the missing link to quantitative biological recoveries of DON in poultry. Similar to previous studies reporting percentages of DON in excreta as being between 2% and 6% of total ingested DON [
28,
35], excreted DON was 2% and 3% of ingested DON in our turkey and chicken trial and approximately 4% in the pullet experiment. DOM was rarely detected in excreta samples in the literature. Whereas Wan
et al. and Awad and co-workers could not detect DOM in excreta extracts [
25,
35], Dänicke
et al., applying immunoaffinity clean-up as sample preparation, determined 0.5 μg DON/kg b.w./day for pullets consuming
ca. 300 μg DON/kg b.w./day, corresponding to approximately 0.2% of ingested DON [
28]. Re-analyzing these pullet excreta samples, we could not detect DOM, most likely due to higher LODs due to greater dilution factors (see below). Similarly, DOM could not be detected in any of the excreta samples of our turkey and chicken experiments. One reason might be lack of microbes capable of de-epoxidizing DON. Another reason is that our methods were not designed to detect trace concentrations of DOM, but to allow co-determination of DON- and DOM-sulfates, DONS, DOMS, DON and DOM. Separation of DONS and DOMS requires formic acid as a mobile phase additive [
24]. However, use of formic acid resulted in fivefold higher LODs for DON and DOM compared to acetic acid. Still, this compromise had to be made for proper separation of DON sulfonates. LOQ values for DON and DOM in a pure standard solution (5 ng/mL) were higher than those obtained by Devreese
et al. (0.1 and 0.5 ng/mL for DON and DOM), but similar to those obtained in our previous work on DON- and DOM sulfonates (2 ng/mL on 5500 QTrap, 5 ng/mL on 4000 QTrap). LOQ values in lyophilized pullet feces obtained by Dänicke and co-workers were 0.8 and 1.6 ng/g [
28], and as such far better than ours. The reason for this great difference is that Dänicke
et al. used multi-step sample preparation including defatting and immunoaffinity clean-up. In our work, on the contrary, great dilution factors were chosen on purpose due to the high concentrations of DON-3-sulfate and in order to minimize matrix effects.
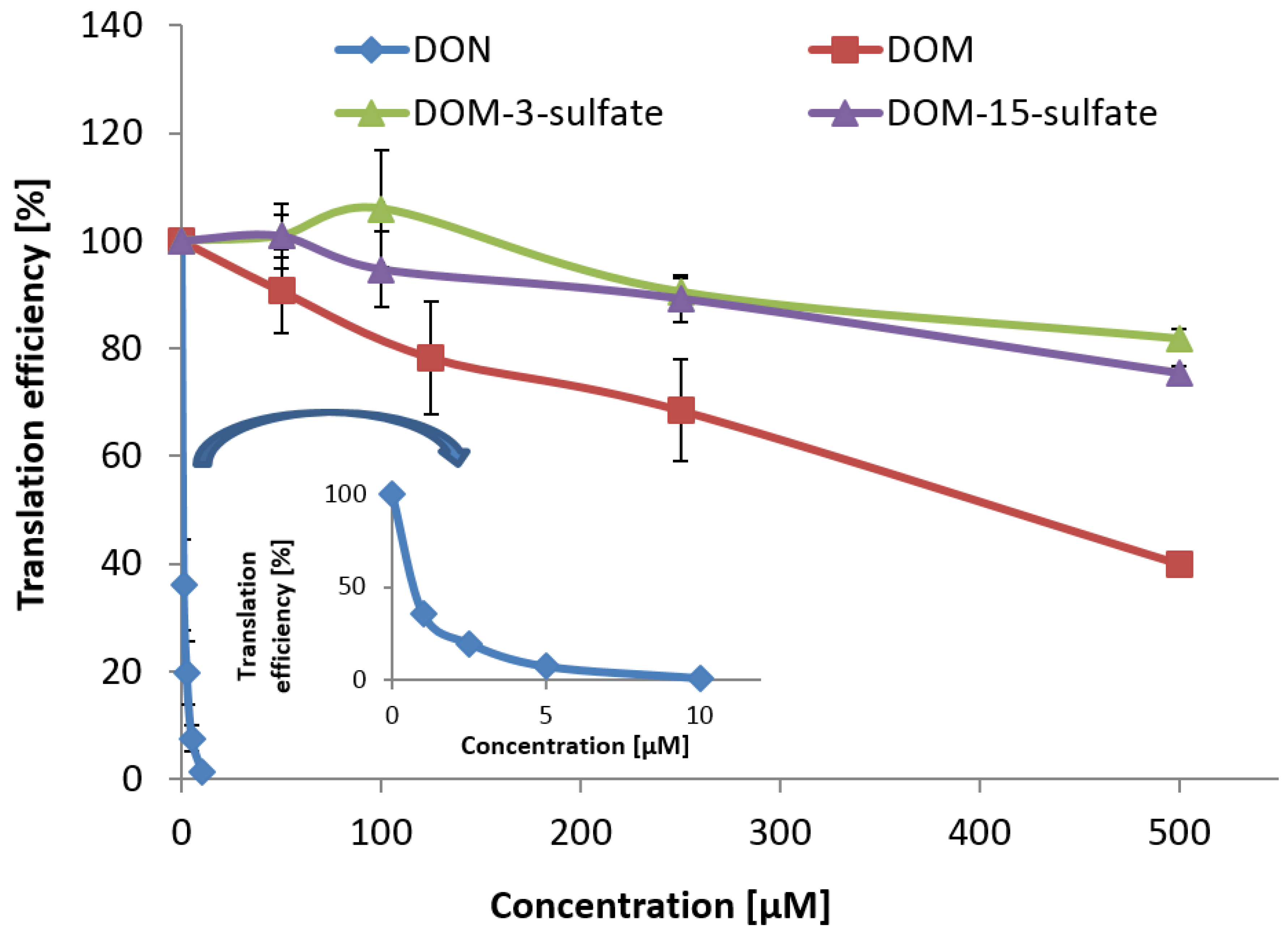
As DOM and DOM-sulfates are much less toxic than DON in an
in vitro translation assay using animal ribosomes, and the main mode of action of all trichothecenes is the suppression of translation, these metabolites can be considered as detoxification products of DON. In the following, the question about location of formation, absorption and elimination of DON-3-sulfate and DOM-3-sulfate will be tackled by putting our findings and recent literature results in context with older studies on DON metabolization in poultry. For this purpose, it is important to note that poultry excrete white pasty urine into the cloaca where it is mixed with solid excrements from the GI tract. Hence, administration of native DON to poultry and collection of excrements does not give information about the origin of excreted DON-3-sulfate and DOM-3-sulfate. Another peculiarity of poultry is that, in addition to the hepatic portal system, a renal portal system exists. Both systems drain the intestine via the
Vena mesenterica cranialis and
V. mesenterica caudalis, respectively, and might contribute to renal and/or hepatic metabolism and first pass elimination as suggested by Rotter
et al. [
36].
As early as in 1986, Prelusky and co-workers administered a single oral dose of
14C-labeled DON to white Leghorn hens by crop intubation [
37]. They observed low levels of radioactivity in plasma and reported the total body burden (excluding bile, GI tract and excreta) of radioactively-labeled DON and its metabolites to be less than 2%. The highest accumulation of radioactivity was determined in bile 6 h after treatment. Within 24 h, 79% of radioactivity was excreted. These results suggested low systemic absorption of DON and/or its metabolites and significant biliary excretion.
Two years later, Lun
et al. [
38], administering native DON to laying domestic chickens, reported that DON disappeared from the GI tract between the crop and the jejunum. However, only low levels of DON were detected in portal vein blood. Considering little decrease of DON when incubated with gastric fluid or juice from the small intestine [
38], they set up the hypothesis of rapid post-absorptive modification of DON by enterocytes and hepatic elimination into bile. In the same study,
in vitro incubation of native DON with contents of colon and caecae resulted in the reduction of the DON concentration by factors of 3, 9 and 20 after 6, 12 and 24 h of incubation, respectively.
One year later, Lun and co-workers administered tritium labeled DON by crop intubation to colostomized white Leghorn hens [
39]. Interestingly, 68% of the administered radioactivity was excreted within 24 h into urine, whereas only 6% was eliminated via feces. Radioactivity in systemic blood accounted for only 7% of the administered radioactivity. Radioactivity along the GI tract of intact crop intubated hens decreased rapidly after the crop, but increased again in the large intestine six hours after administration, which is most likely due to retrograde movement of urine from cloaca into colon. However, contrary to observations by Prelusky and co-workers [
37], biliary excretion of radioactivity was <1%. These results confirmed the hypothesis set up by the same working group in 1988 that ingested DON is rapidly absorbed between crop and upper jejunum and metabolized shortly after absorption [
38]. Yet, extensive excretion of the altered DON via bile was not confirmed as most of the radioactivity was excreted into urine.
Results obtained in more recent studies where native or radiolabeled DON was administered to poultry mostly confirmed and complemented the earlier published data. Yunus
et al. [
27] recovered only 0.04% of total DON fed to broilers 1 h after oral treatment in plasma in the form of DON and DOM. Likewise, Dänicke and co-workers determined only trace levels of DON in plasma of pullets fed with poultry feed containing 4.4 mg/kg DON. However, two recent studies reported an oral bioavailability of 20% for DON administered to broiler chickens [
40], and 21% for DON fed to turkeys [
26]. Higher values than previously reported might be due to different ways of calculating the oral bioavailability. In accordance with earlier studies, rapid absorption, fast elimination, and extensive biotransformation were reported [
26,
40]. Comparison of oral and IV application of DON and inclusion of DON-3-sulfate in the analytical method for semiquantitative determination yielded precious information on the toxicokinetic behavior of DON and on the location of sulfation in turkeys and broiler chickens [
26]. Already 5 min after oral administration, DON and DON-3-sulfate were detected in plasma. Maximum values of DON were reached 10 min post dosing, and maximum values of DON-3-sulfate between 20 and 30 min after application. Only 5 min after IV administration of DON, the average peak area ratio of DON-3-sulfate to DON was 10 in turkeys and 243 in broiler chickens. This finding points to very rapid sulfation, with the liver, extrahepatic tissues, and the intestinal mucosa being potential metabolism sites. In addition, average peak area ratios of DON-3-sulfate to DON were much greater after oral, rather than after IV administration in both avian species. This observation strongly substantiates the hypothesis by Lun
et al. [
38,
39] that sulfation is already performed in enterocytes of the intestinal mucosa.
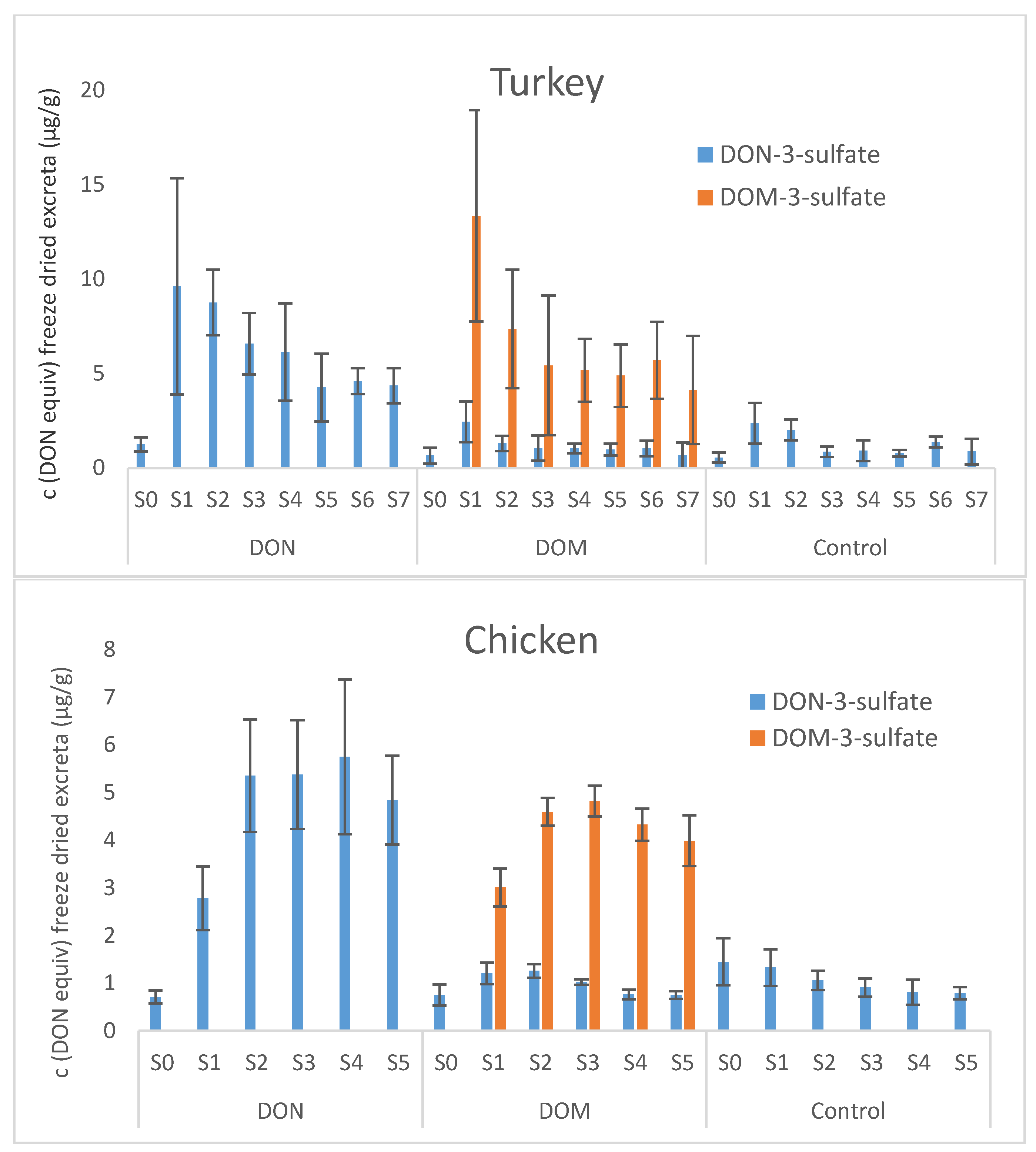
Our current turkey and chicken trial revealed that metabolization of DON and DOM was very similar in both avian species. Excretion kinetics could not be established because animals had ad libitum access to DON and DOM contaminated feed during the day. Despite large inter-individual differences between the turkeys of one feeding group, maximum metabolite concentrations in excreta between 2 and 4 h after the start of feeding and maximum feed intake in the morning suggest fast transit times in turkeys. In chickens, excreta were sampled 3 and 6 h after provision of DON or DOM contaminated feed, so that transit times can only roughly be estimated. Still, maximum concentrations obtained at the latest 6 h after start of feeding corroborate fast excretion of DON- and DOM metabolites also in chickens.
Low concentrations of DON-3-sulfate and DON in chyme samples (content of jejunum and ileum) of pullets support the hypothesis of rapid and extensive absorption of DON from the small intestine [
39]. The origin of DON-3-sulfate in jejunum and ileum might be biliary excretion of DON-3-sulfate formed in enterocytes or in the liver. As chyme samples were collected 3 h after administration of DON, biliary excretion might not have arrived at its maximum which was reported to occur
ca. 6 h after dosing [
37].
Presence of DOM-3-sulfate in excreta of pullets and roosters might be due to formation of DOM by intestinal microbes, absorption of DOM from the GI tract, sulfation in the intestinal mucosa, liver and/or kidney, and excretion into the cloaca via urine or back into the GI tract via bile. The latter could also explain low concentrations of DOM-3-sulfate in ileum. An additional hypothesis is antiperistaltic retrograde movement of DON-3-sulfate from the cloaca to the microbial rich caecae and microbe-mediated formation of DOM-3-sulfate from DON-3-sulfate. A third possibility might be sulfation in DOM by intestinal microbes. Absence of DOM-3-sulfate in excreta samples of our turkey and chicken trial might be due to differences in the intestinal microflora, specifically due to lack of microbes capable of de-epoxidizing DON. Although infection with A. galli resulted in significantly lower formation of DON-3-sulfate and DOM-3-sulfate in pullets, the formation pattern of DON metabolites was unaltered by worm infection.
Concluding from the DON sulfonate pattern across the GI tract of pullets, DON sulfonates are formed early in the digestion process. The mechanism of DON sulfonate formation is currently unknown. Some experiments aiming at elucidating the mechanism of DON-, DOM-, and DON-3-glucoside (D3G) sulfonate formation in rats had been carried out lately [
24], but were inconclusive. However, because the major part of the sulfonates was recovered in feces of DON treated rats and the formation pattern shifted to DOM sulfonate on the second day after treatment, authors speculated that DON-, DOM-, and D3G sulfonates might be formed in a Michael addition with reactive inorganic sulfur species possibly present in intestinal chyme rather than by liver enzymes.
Biological recoveries of DON in the current study are much higher than those previously published for native DON administered to chickens, which were ≤5% [
28]. However, they compare nicely to values published for experiments with radiolabeled DON. Wan
et al. [
25], using radioisotope counting radio-HPLC, reported biological recoveries of 81%, 89% and 95% in the time span of 24, 48 and 72 h after oral administration of a single dose of radiolabeled DON. Likewise, Prelusky
et al. [
37] recovered 79%, 92% and 99% of radiolabeled DON by 24, 48 and 72 h after single bolus administration to white Leghorn hens. The finding that excretion is not complete 24 h after treatment explains incomplete biological recoveries in our feeding trials with turkeys and chickens. Lower biological recoveries in turkeys than in chickens hint at delayed excretion in turkeys compared to chickens. Recoveries greater than 100% in the pullet trial might be due to presence of DON-3-glucoside (D3G) in the Fusarium contaminated diet used in the animal experiment. As D3G was not determined in the administered feed, its presence and subsequent
in vivo metabolization to DON- and DOM-3-sulfate cannot be excluded.
To summarize, our findings and data from the recent literature substantiate, complement, and extend results and hypotheses from earlier published articles on the absorption, metabolism, and excretion of DON in poultry. DON is, to a great extent, rapidly absorbed from the GI tract between crop and jejunum. After absorption, DON is extensively metabolized to DON-3-sulfate in the intestinal mucosa, the liver, and possibly in the kidney. DON-3-sulfate is rapidly and completely eliminated into bile by the liver and/or into urine by the kidney. DOM-3-sulfate is an important natural DON metabolite in pullets and roosters and might originate from sulfation of microbe-formed DOM by intestinal mucosa, liver, and/or kidney or from microbe mediated deepoxidation of urinary DON-3-sulfate transported into caecae by retrograde movement.
4. Experimental Section
4.1. Reagents
Methanol (MeOH, LC gradient grade) and formic acid (98%–100%, p.a.) were purchased from Merck (Darmstadt, Germany). Formic acid (LC-MS gradient grade) for LC-MS/MS was obtained from Sigma Aldrich (Vienna, Austria). Reagents for synthesis of DOM-sulfates were purchased from ABCR (Karlsruhe, Germany) and Sigma Aldrich (Vienna, Austria). In all experiments, ultra-pure water (delivered by a Purelab Ultra system (ELGA LabWater, Celle, Germany)) was used.
4.2. Synthesis, Purification and Characterization of DOM-3-Sulfate and DOM-15-Sulfate
DOM as the starting material for synthesis of DOM-3-sulfate and DOM-15-sulfate was produced from DON as described earlier [
24] and purified by preparative chromatography. The preparative HPLC system and column were the same as described in [
34]. Mobile phase A consisted of methanol/water/formic acid (10/89.9/0.1, v/v/v), mobile phase B of methanol/formic acid (99.9/0.1, v/v). Gradient elution started at 0% B from 0 to 1 min and continued with linear increase to 100% B from 1 to 6 min. After 1 min at 100% B the starting conditions were re-established from 7 to 7.1 min and the column was re-equilibrated at 0% B until 10 min. The injection volume was 700 μL, the flow rate 16 mL/min and DOM was detected by measurement of UV absorption at 220 nm. The collection window of DOM was 5.66–6.03 min.
The synthesis of DOM-3-sulfate and DOM-15-sulfate was carried out as described for DON [
31] with minor modifications. In short, it included synthesis of protected intermediates (4.2.1) and subsequent deprotection (4.2.2) followed by purification by column chromatography. The progress of all reactions was monitored by thin layer chromatography (TLC) using silica gel 60 F
254 TLC plates (Merck, Germany). All chromatograms were visualized using ceric ammonium molybdate/Hanessian’s stain in EtOH/sulphuric acid. Column chromatography was performed on silica gel 60 (40–63 μm) using a Sepacore™ Flash System (Büchi, Switzerland) or small glass columns.
4.2.1. Synthesis of the Protected Intermediates
DOM (140.0 mg, 0.50 mmol, 1.0 equivalents (eq.)) was dissolved in 20 mL of dry dichloromethane (DCM), cooled to 0 °C, and 1,2-dimethylimidazole (144.0 mg, 1.50 mmol, 3.0 eq.) was added to the mixture. Finally, 2,3-dimethyl-1-((2,2,2-trichloroethoxy)sulfonyl)-1H-imidazolium trifluoromethanesulfonate (342.8 mg, 0.75 mmol, 1.5 eq.) was added and the reaction was allowed to reach room temperature overnight. TLC after 1 day showed nearly full conversion of the starting material. Hence, the reaction was directly subjected to column chromatography (hexane/ethyl acetate 3/1, v/v) upon which three protected intermediates (2,2,2-trichloroethyl-DOM-3-sulfate (71.0 mg, 29%), 2,2,2-trichloroethyl-DOM-15-sulfate (14 mg, 6%) and bis(2,2,2-trichloroethyl) DOM-3,15-disulfate (30.0 mg, 9%)) were obtained.
4.2.2. Deprotection of Protected Intermediates
Deprotection of the protected intermediates was carried out as described in [
31]. The starting material was dissolved in MeOH (1 mL/10 μmol). Ammonium formate (3 eq. for the monosulfates, 6 eq. for the disulfate) as well as Zn dust (9 and 18 eq., respectively) were added, and the reaction mixture was placed in an ultrasonic bath at room temperature. The progress of the reaction was monitored by TLC until substantial amounts of products were formed (20 to 120 min). After filtration through celite, the remaining residue was subjected to column chromatography to end up with the corresponding sulfates as ammonium salts. For this purpose, a mobile phase of DCM/MeOH/NH
4OH (10/4/1, v/v/v) was used. The final products were dissolved in water, lyophilized, and finally obtained as a white powder.
4.2.3. NMR Spectroscopy
1H and 13C spectra were recorded on a Bruker Avance DRX-400 MHz and a Bruker Avance III 600 MHz spectrometer (Bruker, Germany). Data were recorded and evaluated using TOPSPIN 1.3 and TOPSPIN 3.2 (Bruker Topspin). All chemical shifts are given in ppm relative to tetramethylsilane. The calibration was done using residual solvent signals. Multiplicities are abbreviated as s (singlet), d (doublet), t (triplet), q (quartet) and b (broad signal). Deuterated solvents were purchased from Eurisotop (Gif sur Yvette Cedex, Paris, France).
4.2.4. LC-HR-MS/MS
LC-HR-MS(/MS) spectra were recorded on a 6550 iFunnel Q-TOF instrument coupled to a 1290 Infinity UHPLC system (both Agilent Technologies, Waldbronn, Germany). Chromatographic separation was carried out on a Zorbax Eclipse Plus C18 Rapid Resolution High Definition column (2.1 × 150 mm, 1.8 μm particle size, Agilent, Waldbronn, Germany) at a flow rate of 0.25 mL/min using gradient elution (0 min: 15% B, 6 min: 100% B, 7 min: 100% B, 7.1 min: 15% B, 9 min: 15% B). Mobile phase A was water/formic acid (99.9/0.1, v/v), mobile phase B MeOH/formic acid (99.9/0.1, v/v). Compounds were ionized by electrospray ionization in the negative mode and measured first in full scan and then in targeted MS/MS mode at a collision energy of 30 eV (both in the range from m/z 40-1000). Electrospray ionization was carried out at a gas temperature of 140 °C, drying gas flow of 14 L/min, nebulizer pressure of 35 psig, sheath gas temperature of 350 °C, and sheath gas flow of 11 L/min. The capillary voltage was 4500 V, the nozzle voltage 300 V. Data acquisition was achieved in the 2 GHz extended dynamic range mode.
4.3. Standards and Standard Solutions
Solid DON (purity >95%) as well as standard solutions of DON and DOM (both 50 mg/L in acetonitrile) were supplied by Romer Labs GmbH (Tulln, Austria). DON-3-sulfate and DON-15-sulfate (both 95% purity) were synthesized according to [
31]. DOM, DOM-3-sulfate, and DOM-15-sulfate were produced as described above (4.2). DON sulfonates 1, 2, 3 and DOM sulfonate 2 were produced as described in [
24].
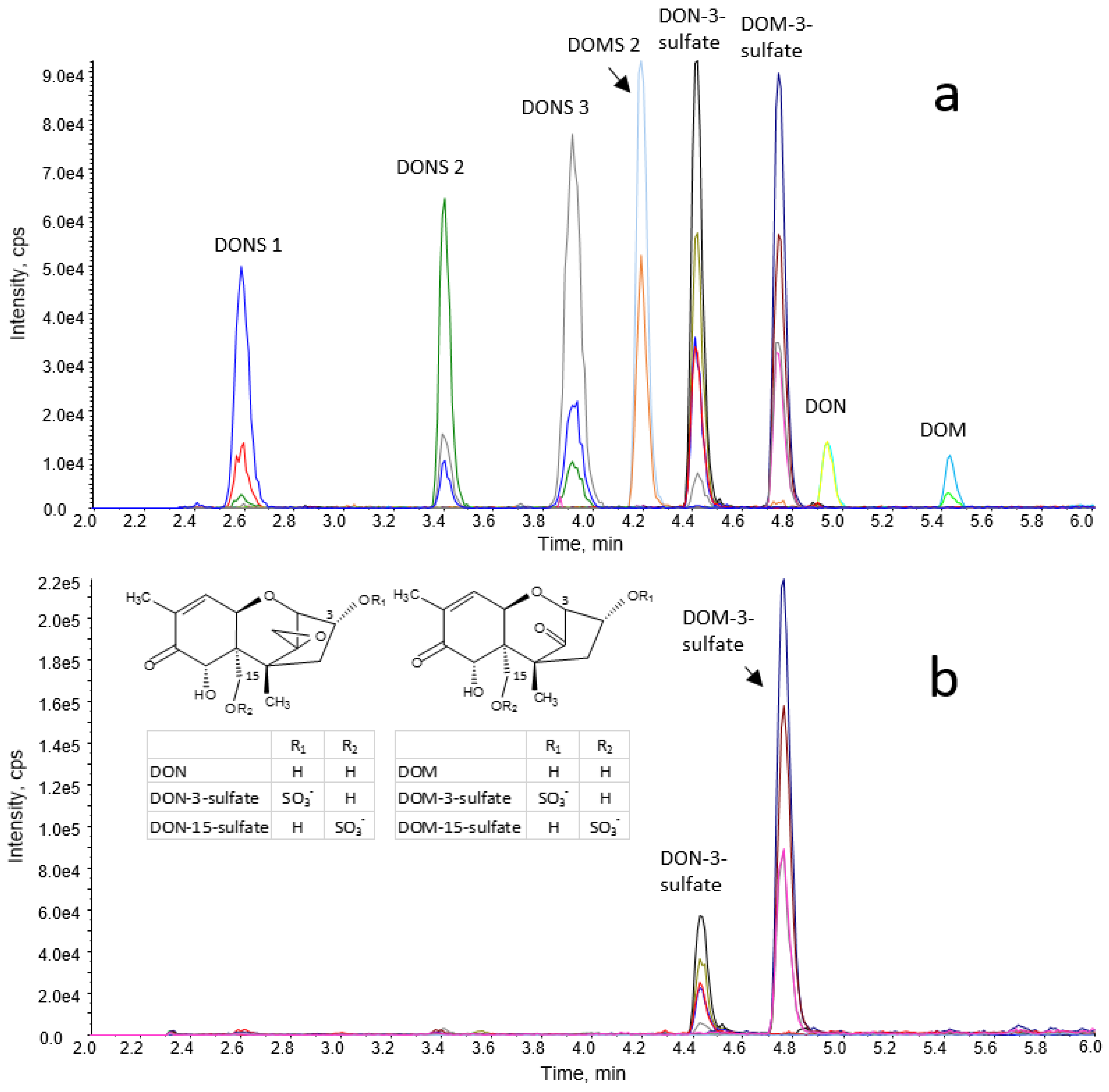
A mixed standard solution containing 300 mg/L of DON-3-sulfate, DOM-3-sulfate, DON, DOM, DONS 1, DONS 2, DONS 3, and DOMS 2 was prepared in methanol/water/formic acid (20/79.9/0.1, v/v/v). This solution and several dilutions thereof were used for spiking experiments and the establishment of pure solvent and matrix-matched calibration functions.
4.4. Sample Preparation Methods for Determination of DON, DOM, and Their Sulfates in Excreta and GI Samples of Poultry
Extraction of DON, DOM and their sulfate- and sulfonate metabolites from excreta samples was carried out according to a procedure previously optimized for the extraction of DON- and DOM sulfonates from rat feces [
24]. A 300 mg aliquot of homogenized lyophilized excreta or GI content sample was consecutively extracted with 4, 3 and 3 mL of methanol/water/formic acid (49.5/49.5/1, v/v/v) by shaking for 30, 20 and 10 min in 15 mL polypropylene tubes. Prior to LC-MS/MS analysis, aliquots of the pooled extracts were diluted with water (turkeys, chickens: 1 + 2; pullets: jejunum and ileum: 1 + 1, excreta: 1 + 5; roosters: 1 + 9) and centrifuged at 14,000 ×
g.
4.5. LC-MS/MS Analysis of DON, DOM, Their Sulfates and Their Sulfonates in Excreta of Poultry
LC-MS/MS analyses were carried out on an Agilent 1290 series UHPLC system coupled to a 6500 QTrap mass spectrometer equipped with an IonDrive Turbo V® source (Sciex, Foster City, CA, USA). Chromatographic separation was achieved on a Kinetex Biphenyl column (150 × 3 mm, 2.6 μm) protected by a SecurityGuard ULTRA pre-column of the same stationary phase (both Phenomenex, Aschaffenburg, Germany) at 30 °C and at a flow rate of 0.4 mL/min. Mobile phase A consisted of water/formic acid, mobile phase B of methanol/formic acid (both 99.9/0.1, v/v). Two different gradient methods were used, a short one for routine measurements, and a long one for separation of the 3/15 isomers of DON- and DOM-sulfate. The gradient of the short routine method was: 0.0–0.5 min: 10% B, 6.0 min: 90% B, 6.1–7.5 min: 100% B, 7.6–10.0 min: 10% B. Time segments of the gradient of the long method were: 0.0–0.5 min: 10% B, 4.5 min: 40% B, 8.0 min: 45% B, 8.5–10.9 min: 100% B, 11.0–13.5 min: 10% B. The injection volume was 3 μL and the LC eluent was diverted to the MS between 2.3 and 6.0 min (8.0 min for the long method).
Tandem mass spectrometric detection was performed in negative selected reaction monitoring (SRM) mode after electrospray ionization. The following ion source settings were used: Temperature 400 °C, ion spray voltage -4500 V, curtain gas 35 psi, ion source gas 1 80 psi, ion source gas 2 90 psi, collision gas (N
2) high. SRM parameters (declustering potential, collision energy, collision cell exit potential) were optimized for the individual analytes by software-controlled compound optimization and are listed in
Supplementary Table S3. Analyst
® software version 1.6.2 (Sciex) was used for instrument control and data evaluation.
4.6. Method Validation
Validation of the short routine method included determination of the apparent recovery (RA), recovery of extraction (RE), and mass spectrometric matrix effects (SSE) for excreta of turkeys and chickens as well as assessment of SSE for extracts of excreta and GI content samples of pullets, and excreta extracts of roosters. In addition, intra- and inter-day repeatability of sample work-up and analysis, limits of detection and quantitation, and linear range of calibration functions were evaluated. The long gradient method was validated with respect to SSE in excreta extracts of turkeys and chickens.
For assessment of R
A, R
E, and SSE in excreta samples of turkeys and chickens, 300 mg aliquots of freeze dried excreta collected before administration of toxins were spiked with 60 μL aliquots of spiking solutions containing between 0.5 and 300 mg/L of DON-3-sulfate, DOM-3-sulfate, DON, DOM, DONS 1, DONS 2, DONS 3, and DOMS 2, resulting in 0.1 to 60 mg/kg of these analytes in the freeze dried sample aliquots. Spiking was performed at seven concentration levels (0.1/0.3/1/3/10/30/60 mg/kg) in triplicate. One hour after spiking, 21 spiked and three unspiked samples were worked-up and diluted as described above, and measured in the same run as matrix-matched and pure solvent calibration functions. Pooled diluted extracts of unspiked samples were used to prepare matrix-matched calibration functions as described in detail in [
24]. Both matrix-matched and pure solvent calibration functions were established at seven concentration levels (1.1/3.4/11/34/112/337/674 ng/mL), which corresponded to the theoretical analyte concentrations in measurement solutions of samples spiked prior to work-up in the case of 100% apparent recovery. R
A, R
E, and SSE were calculated by comparing the slopes of the standard addition curve (k
SA), matrix-matched calibration curve (k
MM), and pure solvent calibration curve (k
SOL) as described in [
41]. The following equations were used: R
A = k
SA/k
SOL × 100; R
E = k
SA/k
MM × 100; SSE = k
MM/k
SOL × 100. Matrix-matched calibration functions were established between 3 and 600 ng/mL in diluted extracts of chyme and excreta samples of the negative control group of pullets and in excreta sample extracts of roosters, and used for calculation of SSE in these matrices. Apparent recoveries in the same matrices were estimated by multiplying the SSE of each individual analyte by the average R
E of the same analyte determined in excreta of turkeys and chickens (see above).
The intra-day repeatability was determined by triplicate work-up and measurement of blank turkey and chicken excreta samples spiked at four concentration levels (1, 3, 10, 30 ng/g) on one day. The inter-day repeatability was assessed by work-up and analysis of two randomly selected excreta samples (one of turkey, one of chicken) on each day sample preparation and analysis was carried out.
Limits of detection (LOD, signal to noise ratio (S/N) 3/1) and quantitation (LOQ, S/N 10/1) were determined in pure solvent standard solutions and in matrix-matched standard solutions. LODs and LOQs in freeze-dried samples were calculated by dividing LODs and LOQs in matrix-matched solutions by the recovery of extraction and by multiplying by the dilution factor. The linear range in matrix-matched calibration curves defined the upper end of the working range.
4.7. Design of the Feeding Trials
In total, excreta samples from four different feeding trials with poultry were analyzed. The parameters of the individual feeding trials are summarized in
Table 5. Turkeys, chickens, and pullets of the negative control groups were fed with basal poultry feed naturally contaminated with 0.2–0.3 mg/kg DON. Turkeys and chickens of the DON group received basal poultry feed enriched with DON from culture material to a concentration of 1.5–1.7 mg/kg. Similarly, turkeys and chickens of the DOM group were fed with basal poultry feed artificially contaminated with the equimolar concentration of DOM. Pullets were exposed to 4.4 mg/kg DON in feed. As this feed was prepared from wheat contaminated with different Fusarium toxins [
28], it also contained traces of 3-acetyl-DON (0.13 mg/kg) and 15-acetyl-DON (0.03 mg/kg). Roosters received diet containing 11 mg/kg DON.
All feeding trials and animal experiments were conducted following the European Guidelines for the Care and Use of Animals for Research Purpose [
42]. The feeding trials with turkeys and chickens lasted for one day during which animals had unlimited access to feed and water until feed was removed in the evening. Animals were housed under a light/dark cycle of 18/6 h. Feed consumption of the individual animals was recorded. Excreta samples were collected at regular intervals during the day and in the morning of the following day. After the experiment, animals received basal poultry feed for two weeks before they were reintegrated into the meat production process. In the animal experiment with pullets, pullets received 65 g feed/day in two equal portions for two weeks and excreta samples were collected twice a day in the second week [
28]. Pooled excreta samples were used for determination of biological recoveries. On the last day of the trial, pullets were slaughtered 3 h after the last feeding and chyme samples (contents of jejunum and ileum) were taken. Roosters were fed the DON contaminated feed at amounts of 90 g/day and excreta were collected for seven days [
30]. Samples taken on different days were pooled for each animal prior to analysis. All excreta and chyme samples were stored frozen until lyophilization. Freeze-dried samples were stored at −20 °C.
Table 5.
Overview of four DON/DOM feeding trials with poultry.
Table 5.
Overview of four DON/DOM feeding trials with poultry.
| | Turkeys | Chickens | Pullets | Roosters |
|---|
| Reference | - | - | [28] | [29,30] |
| Animals | Hybrid Converter | Ross 308 | Lohmann LSL | New Hampshire hybrids |
| Age | 11 weeks | 5 weeks | 12 weeks | adult |
| No. of animals/group | 4 | 4 | 9 | 8 |
| c (DON) in feed (DON group) (mg/kg) | 1.5 | 1.7 | 4.4 | 11 |
| c (DON, DOM) in feed (DOM group) (mg/kg) | DON: 0.3
DOM: 1.4 | DON: 0.2
DOM: 1.6 | - | - |
| c (DON) in feed negative control group (mg/kg) | 0.3 | 0.2 | 0.2 | No NC |
| Feeding | Ad libitum access from 7:00 to 22:00 | Ad libitum access from 7:00 to 19:00 | 65 g feed/day (2 equal portions) | 90 g feed /day |
| Duration | 1 day | 1 day | 2 weeks | 9 days * |
| Sampling times | Every 2 h until 15:00; 18:00; 22:00; 7:00 next day | Every 3 h until 19 h; 7:00 next day | Excreta: morning and afternoon for 1 week; chyme: 3 h after administration | Morning and afternoon for 1 week |
| Samples taken | Excreta | Excreta | Excreta, content of jejunum and ileum | Excreta |
4.8. Analysis of Samples and Data Evaluation
Excreta and GI content samples were worked-up in duplicate, each pooled and diluted extract was measured once. Analyte concentrations in freeze-dried samples were determined on the basis of pure solvent calibration functions (peak area versus analyte concentration) established routinely between 1 and 300 ng/mL under consideration of the apparent recoveries. In case the concentrations determined by duplicate sample work-up and analysis differed by more than 20%, work-up and analysis was repeated in duplicate and the new values were taken for further data processing.
For determination of the biological recoveries of ingested DON and DOM in the trials with turkeys, chickens, and pullets, DON equivalent amounts of all DON- and DOM metabolites quantified in excreta were calculated and divided by the DON equivalent amounts of DON or DOM ingested in the corresponding time period (one day for turkeys and chickens, one week for pullets). Statistical evaluation of differences in metabolite concentrations in excreta samples of pullets with and without A. galli infection was performed by one-way analysis of variance in MS Excel 2013.
4.9. Toxicity Assessment of DOM-3-Sulfate and DOM-15-Sulfate
To determine the
in vitro toxicities of DON, DOM, DOM-3-sulfate, and DOM-15-sulfate, an
in vitro transcription/translation assay with rabbit reticulocyte lysate (Promega, Madison, WI, USA) was employed. Transcription/translation reactions were carried out according to the manufacturer’s instructions as described in [
33] for wheat germ extract with two modifications. Firstly, animal ribosomes (rabbit reticulocyte lysate) were used instead of wheat ribosomes. Secondly, the reactions were stopped after 20 min instead of 30 min. All substances tested were dissolved in water prior to use. The concentrations of DON in the assay were between 0 and 10 μM, while the concentration range of DOM and DOM-sulfates was 0–500 μM. For each compound, three independent assays were performed on three different days using the same batch of the reticulocyte lysate. For determining the translation efficiency at different inhibitor concentrations, one single test reaction was performed for each inhibitor at each concentration level. In addition, a control reaction was carried out at each concentration level where water was used instead of the test substance. The luciferase activity determined for the control reaction was set to 100% (uninhibited reaction) and the readouts from the other reactions were related to this control. Statistical evaluation was performed with IBM SPSS Statistics software. Comparison of mean values was performed with a two-sided
T-test with Welch correction (equal variances not assumed). Results were considered significant at
p < 0.05.
5. Conclusions
An LC-MS/MS based method for quantitative determination of DON-3-sulfate, DOM-3-sulfate, DON, DOM, DON sulfonates 1, 2, 3, and DOM sulfonate 2 in excreta and chyme samples of poultry was developed and validated. Application of the method to excreta and chyme samples from four different feeding trials with turkeys, broiler chickens, pullets, and roosters confirmed DON-3-sulfate as major DON metabolite in all investigated poultry species. Orally administered DOM was equally extensively metabolized to its 3-sulfate metabolite. In addition, DOM-3-sulfate was shown to be an important natural DON metabolite in pullets and roosters, where it amounted to ca. 12% and 33%, respectively, of all the detected metabolites. Interestingly, pullets not infected with A. galli excreted significantly greater concentrations and greater amounts of DON-3-sulfate and DOM-3-sulfate than pullets infected with worms. DON sulfonates of the series 1, 2 and 3 were detected in chyme samples of broilers, albeit only at trace levels. Biological recoveries of orally administered DON in the form of DON-3-sulfate and DOM-3-sulfate in chickens were close to 100%, supporting the hypothesis of nearly quantitative conversion of DON to its 3-sulfate metabolites in chickens. Although turkeys showed very similar metabolization of DON and DOM as chickens, biological recoveries of orally administered DON and DOM were only between 50% and 70%, suggesting faster excretion of DON and DOM metabolites in chickens than in turkeys.
Similar to DON-3-sulfate, DOM-3-sulfate was much less toxic than DON on the ribosome. Although in vitro transcription/translation assays cannot predict all aspects of in vivo toxicity, greatly reduced toxicity on the ribosome, the molecular target of trichothecenes, and low susceptibility of poultry to DON indicates that sulfation serves as detoxification mechanism for DON in poultry. Concluding from literature reports and our current study, locations of formation of DON-3-sulfate are most likely the intestinal mucosa, liver, and possibly the kidney. Elimination into excreta is probably achieved via bile and via urine. DOM-3-sulfate might also be formed by microbial conversion of DON-3-sulfate after retrograde movement of urinary DON-3-sulfate into the microbial rich caecae. In order to confirm this hypothesis, more complex animal experiments with separate collection of blood (hepatic and renal portal vein blood, systemic blood), bile, urine, contents of jejunum, ileum, caecae, and cloaca samples are required which demands highly sophisticated methods of sampling.