Quantitative Detection of Shiga Toxins Directly from Stool Specimens of Patients Associated with an Outbreak of Enterohemorrhagic Escherichia coli in Japan—Quantitative Shiga toxin detection from stool during EHEC outbreak
Abstract
:1. Introduction
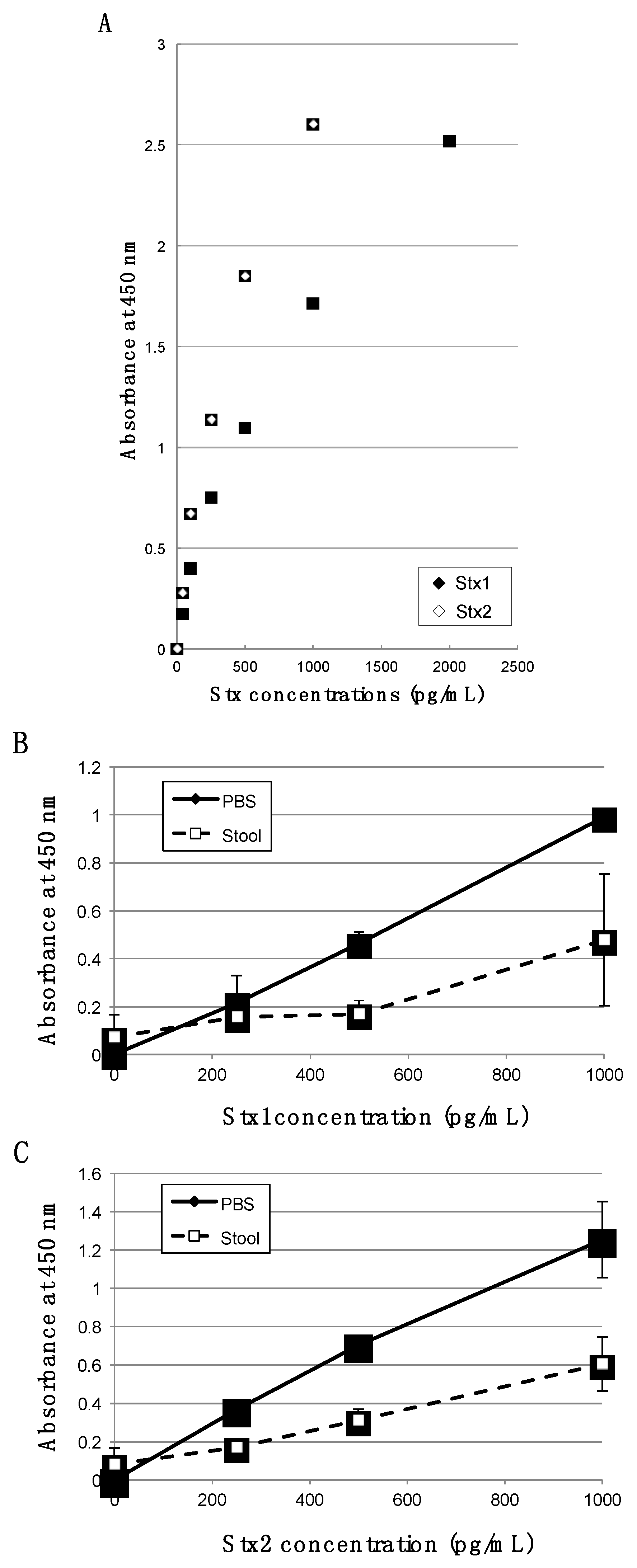
2. Results and Discussion
| Profiles of Patients | Profiles of E. coli Isolated from Stool *,1 | Stx Protein Concentration in Stool (ng/mL) | ||||||
|---|---|---|---|---|---|---|---|---|
| Patient ID | Sex | Age Group (years old) | Symptoms | Serotype | Stx1 Gene | Stx2 Gene | Stx1 | Stx2 |
| No. 1 | Female | 20–29 | Bloody diarrhea, Abdominal pains, Vomit | O157:H7 | + | - | 10.44 | 2.75 |
| O157:H7 | + | + | ||||||
| O111:H8 | - | - | ||||||
| No. 2 | Female | 40–49 | Bloody diarrhea, Abdominal pains, HUS | O111:H8 | - | - | U.D. *,2 | U.D. *,2 |
| No. 3 | Male | 10–19 | Diarrhea, Abdominal pains, HUS | O157:H7 | + | + | 1.71 | 51.61 |
| O111:H8 | - | + | ||||||
| O111:H8 | - | - | ||||||
| No. 4 *,3 | Male | 20–29 | Bloody diarrhea, Abdominal pain, Vomit | N.D. *,4 | n.d. *,5 | n.d. *,5 | 0.71 | U.D. *,2 |
| No. 5 | Female | 0–9 | Bloody diarrhea, Abdominal pains, HUS | O157:H7 | + | + | U.D. *,2 | U.D. *,2 |
| O111:H8 | - | + | ||||||
| O111:H8 | - | - | ||||||
| No. 6 | Male | 10–19 | Bloody diarrhea, Abdominal pains | O111:H8 | - | + | U.D. *,2 | 12.88 |
| No. 7 | Female | 10–19 | Bloody diarrhea, Abdominal pains, Vomit | O157:H7 | + | + | 0.71 | 3.22 |
| O111:H8 | - | + | ||||||
| O111:H8 | - | - | ||||||
| No. 8 | Male | 30–39 | Bloody diarrhea | O111:H8 | - | - | U.D. *,2 | U.D. *,2 |
| No. 9 | Female | 20–29 | Diarrhea, Abdominal pains, Vomit, HUS, AE | N.D. *,4 | n.d. *,5 | n.d. *,5 | U.D. *,2 | U.D. *,2 |
3. Experimental Section
3.1. Patient Stool Specimens
3.2. Preparation of Purified Recombinant Stx1 and Stx2
3.3. Preparation of Antisera against Purified Recombinant Stx1 and Stx2 and Construction of Bead-ELISA for Stx1 and Stx2 Detection
3.4. Quantitative Analysis of Stx1 and Stx2 by Bead-ELISA
3.5. Ethical Considerations
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yahata, Y.; Misaki, T.; Ishida, Y.; Nagira, M.; Watahiki, M.; Isobe, J.; Terajima, J.; Iyoda, S.; Mitobe, J.; Ohnishi, M.; et al. Epidemiological analysis of a large enterohaemorrhagic Escherichia coli O111 outbreak in Japan associated with haemolytic uraemic syndrome and acute encephalopathy. Epidemiol. Infect. 2015, 20, 1–12. [Google Scholar]
- Isobe, J.; Shima, T.; Kanatani, J.I.; Kimata, K.; Shimizu, M.; Kobayashi, N.; Tanaka, T.; Iyoda, S.; Ohnishi, M.; Sata, T.; et al. Serodiagnosis using microagglutination assay during the food-poisoning outbreak in Japan caused by consumption of raw beef contaminated with enterohemorrhagic Escherichia coli O111 and O157. J. Clin. Microbiol. 2014, 52, 1112–1118. [Google Scholar] [CrossRef]
- Watahiki, M.; Isobe, J.; Kimata, K.; Shima, T.; Kanatani, J.; Shimizu, M.; Nagata, A.; Kawakami, K.; Yamada, M.; Izumiya, H.; et al. Characterization of enterohemorrhagic Escherichia coli O111 and O157 strains isolated from outbreak patients in Japan. J. Clin. Microbiol. 2014, 52, 2757–2763. [Google Scholar] [CrossRef] [PubMed]
- Karch, H.; Meyer, T.; Rüssmann, H.; Heesemann, J. Frequent loss of Shiga-like toxin genes in clinical isolates of Escherichia coli upon subcultivation. Infect. Immun. 1992, 60, 3464–3467. [Google Scholar] [PubMed]
- Gould, L.H.; Bopp, C.; Strockbine, N.; Atkinson, R.; Baselski, V.; Body, B.; Carey, R.; Crandall, C.; Hurd, S.; Kaplan, R.; et al. Recommendations for diagnosis of shiga toxin-producing Escherichia coli infections by clinical laboratories. MMWR. Recomm. Rep. 2009, 58, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Cornick, N.A.; Jelacic, S.; Ciol, M.A.; Tarr, P.I. Escherichia coli O157:H7 infections: Discordance between filterable fecal shiga toxin and disease outcome. J. Infect. Dis. 2002, 186, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Oku, Y.; Uesaka, Y.; Hirayama, T.; Takeda, Y. Development to detect of a highly sensitive bead-ELISA to detect bacterial protein toxin. Microbiol. Immunol. 1988, 32, 807–816. [Google Scholar] [CrossRef] [PubMed]
- Uesaka, Y.; Otsuka, Y.; Kashida, M.; Oku, Y.; Horigome, K.; Nair, G.B.; Pal, S.C.; Yamasaki, S.; Takeda, Y. Detection of cholera toxin by a highly sensitive linked immunosorbent assay. Microbiol. Immunol. 1992, 36, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Ramamurthy, T.; Bhattacharya, S.K.; Uesaka, Y.; Horigome, K.; Paul, M.; Sen, D.; Pal, S.C.; Takeda, T.; Takeda, Y.; Nair, G.B. Evaluation of the bead enzyme-linked immunosorbent assay for detection of cholera toxin directly from stool specimens. J. Clin. Microbiol. 1992, 30, 1783–1786. [Google Scholar] [PubMed]
- Caprioli, A.; Luzzi, I.; Rosmini, F.; Pasquini, P.; Cirrincione, R.; Gianviti, A.; Matteucci, M.C.; Rizzoni, G. Hemolytic-uremic syndrome and Vero cytotoxin-producing Escherichia coli infection in Italy. J. Infect. Dis. 1992, 166, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Karmali, M.A.; Petric, M.; Lim, C.; Fleming, P.C.; GArbus, G.S.; Lior, H. The association between idiopathic hemolytic uremic syndrome and infection by verotoxin-producing Escherichia coli. J. Infect. Dis. 1985, 151, 775–782. [Google Scholar] [CrossRef] [PubMed]
- Brigotti, M.; Tazzari, P.L.; Ravanelli, E.; Carnicelli, D.; Rocchi, L.; Arfilli, V.; Scavia, G.; Minelli, F.; Ricci, F.; Pagliaro, P.; et al. Clinical relevance of shiga toxin concentrations in the blood of patients with hemolytic uremic syndrome. Pediatr. Infect. Dis. J. 2011, 30, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Kehl, K.S.; Havens, P.; Behnke, C.E.; Acheson, D.W.K. Evaluation of the premier EHEC assay for detection of Shiga toxin-producing Escherichia coli. J. Clin. Microbiol. 1997, 35, 2051–2054. [Google Scholar] [PubMed]
- Mackenzie, A.M.R.; Lebel, P.; Orrbine, E.; Rowe, P.C.; Hyde, L.; Chan, F.; Johnson, W.; McLaine, P.N. Sensitivities and specificities of premier E. coli O157 and premier EHEC enzyme immunoassays for diagnosis of infection with verotxin (Shiga-like toxin)-producing Escherichia coli. J. Clin. Microbiol. 1998, 36, 1608–1611. [Google Scholar] [PubMed]
- Yamasaki, S.; Furutani, M.; Ito, K.; Igarashi, K.; Nishibuchi, M.; Takeda, Y. Importance of arginine at position 170 of the A subunit of Vero toxin 1 produced by enterohemorrhagic Escherichia coli for toxin activity. Microb. Pathog. 1991, 11, 1–9. [Google Scholar] [CrossRef]
- Ohmura, M.; Cao, C.; Karasawa, T.; Okuda, J.; Kurazono, H.; Gannon, V.P.J.; Gyles, C.L.; Takeda, Y. Purification and some properties of a Vero toxin 2 (Shiga-like toxin II) variant (SLT-IIva) of Escherichia coli isolated from infantile diarrhea. Microb. Phathog. 1993, 15, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, E.; Sakamoto, R.; Matsumoto, T.; Morimatsu, F.; Kurazono, T.; Hiroi, T.; Nair, G.B.; Kurazono, H. Development of an immunochromatographic test strip for detection of cholera toxin. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamasaki, E.; Watahiki, M.; Isobe, J.; Sata, T.; Nair, G.B.; Kurazono, H. Quantitative Detection of Shiga Toxins Directly from Stool Specimens of Patients Associated with an Outbreak of Enterohemorrhagic Escherichia coli in Japan—Quantitative Shiga toxin detection from stool during EHEC outbreak. Toxins 2015, 7, 4381-4389. https://doi.org/10.3390/toxins7104381
Yamasaki E, Watahiki M, Isobe J, Sata T, Nair GB, Kurazono H. Quantitative Detection of Shiga Toxins Directly from Stool Specimens of Patients Associated with an Outbreak of Enterohemorrhagic Escherichia coli in Japan—Quantitative Shiga toxin detection from stool during EHEC outbreak. Toxins. 2015; 7(10):4381-4389. https://doi.org/10.3390/toxins7104381
Chicago/Turabian StyleYamasaki, Eiki, Masanori Watahiki, Junko Isobe, Tetsutaro Sata, G. Balakrish Nair, and Hisao Kurazono. 2015. "Quantitative Detection of Shiga Toxins Directly from Stool Specimens of Patients Associated with an Outbreak of Enterohemorrhagic Escherichia coli in Japan—Quantitative Shiga toxin detection from stool during EHEC outbreak" Toxins 7, no. 10: 4381-4389. https://doi.org/10.3390/toxins7104381
APA StyleYamasaki, E., Watahiki, M., Isobe, J., Sata, T., Nair, G. B., & Kurazono, H. (2015). Quantitative Detection of Shiga Toxins Directly from Stool Specimens of Patients Associated with an Outbreak of Enterohemorrhagic Escherichia coli in Japan—Quantitative Shiga toxin detection from stool during EHEC outbreak. Toxins, 7(10), 4381-4389. https://doi.org/10.3390/toxins7104381





