Thuringiensin: A Thermostable Secondary Metabolite from Bacillus thuringiensis with Insecticidal Activity against a Wide Range of Insects
Abstract
:1. Introduction
2. Discussion
2.1. Characteristics and Structure of Thu

2.2. Insecticidal Spectrum of Thu
2.3. Security Appraisal of Thu
2.4. Insecticide Mechanism
2.5. Separation and Purification of Thu
2.6. Genetic Determinants of Thu
2.6.1. Relationship between Plasmids and Thu Production
2.6.2. The Synthesis Genes
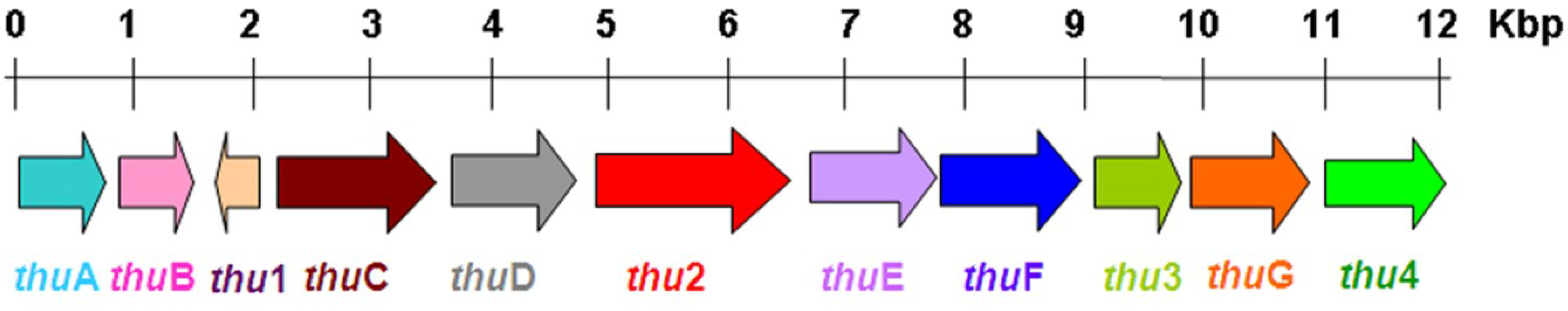
2.6.3. The Biosynthesis Pathway
2.6.4. Secretion and Immune Mechanism of Thu
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schnepf, E.; Crickmore, N.; van Rie, J.; Lereclus, D.; Baum, J.; Feitelson, J.; Zeigler, D.R.; Dean, D.H. Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 775–806. [Google Scholar]
- Sudakin, D.L. Biopesticides. Toxicol. Rev. 2003, 22, 83–90. [Google Scholar] [CrossRef]
- Ye, W.; Zhu, L.; Liu, Y.; Crickmore, N.; Peng, D.; Ruan, L.; Sun, M. Mining new crystal protein genes from Bacillus thuringiensis on the basis of mixed plasmid-enriched genome sequencing and a computational pipeline. Appl. Environ. Microbiol. 2012, 78, 4795–4801. [Google Scholar] [CrossRef]
- Estruch, J.J.; Warren, G.W.; Mullins, M.A.; Nye, G.J.; Craig, J.A.; Koziel, M.G. Vip3A, a novel Bacillus thuringiensis vegetative insecticidal protein with a wide spectrum of activities against lepidopteran insects. Proc. Natl. Acad. Sci. USA 1996, 93, 5389–5394. [Google Scholar]
- Donovan, W.P.; Engleman, J.T.; Donovan, J.C.; Baum, J.A.; Bunkers, G.J.; Chi, D.J.; Clinton, W.P.; English, L.; Heck, G.R.; Ilagan, O.M.; et al. Discovery and characterization of Sip1A: A novel secreted protein from Bacillus thuringiensis with activity against coleopteran larvae. Appl. Microbiol. Biotechnol. 2006, 72, 713–719. [Google Scholar] [CrossRef]
- Stabb, E.V.; Jacobson, L.M.; Handelsman, J. Zwittermicin A-producing strains of Bacillus cereus from diverse soils. Appl. Environ. Microbiol. 1994, 60, 4404–4412. [Google Scholar]
- Crickmore, N.; Zeigler, D.R.; Feitelson, J.; Schnepf, E.; van Rie, J.; Lereclus, D.; Baum, J.; Dean, D.H. Revision of the nomenclature for the Bacillus thuringiensis pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 807–813. [Google Scholar]
- Wirth, M.C.; Walton, W.E.; Federici, B.A. Inheritance patterns, dominance, stability, and allelism of insecticide resistance and cross-resistance in two colonies of Culex quinquefasciatus (Diptera: Culicidae) selected with cry toxins from Bacillus thuringiensis subsp, israelensis. J. Med. Entomol. 2010, 47, 814–822. [Google Scholar] [CrossRef]
- Sharma, C.B.; Prasad, S.S.; Pai, S.B.; Sharma, S. The exotoxin of Bacillus thuringiensis: A new C-mitotic agent. Experientia 1976, 32, 1465–1466. [Google Scholar] [CrossRef]
- McConnell, E.; Richards, A.G. The production by Bacillus thuringiensis Berliner of a heat stable substance toxic for insects. Can. J. Microbiol. 1959, 5, 161–168. [Google Scholar] [CrossRef]
- Farkas, J.; Sebesta, K.; Horska, K.; Samek, Z.; Dollijs, J.; Storm, F. The structure of exotoxin of Bacillus thuringiensis var. gelechiae. Collect. Czechslov. Chem. Commun. 1969, 34, 1118–1120. [Google Scholar] [CrossRef]
- Liu, X.Y.; Ruan, L.F.; Hu, Z.F.; Peng, D.H.; Cao, S.Y.; Yu, Z.N.; Liu, Y.; Zheng, J.S.; Sun, M. Genome wide screening reveals the genetic determinants of a antibiotic insecticide in Bacillus thuringiensis. J. Biol. Chem. 2010, 285, 39191–39200. [Google Scholar]
- Toledo, J.; Liedo, P.; Williams, T.; Ibarra, J. Toxicity of Bacillus thuringiensis beta-exotoxin to three species of fruit flies (Diptera: Tephritidae). J. Econ. Entomol. 1999, 92, 1052–1056. [Google Scholar]
- Tamez-Guerra, P.; Iracheta, M.M.; Pereyra-Alférez, B.; Galán-Wong, L.J.; Gomez-Flores, R.; Tamez-Guerra, R.S.; Rodríguez-Padilla, C. Characterization of Mexican Bacillus thuringiensis strains toxic for lepidopteran and coleopteran larvae. J. Invertebr. Pathol. 2004, 86, 7–18. [Google Scholar] [CrossRef]
- Tsuchiya, S.; Kasaishi, Y.; Harada, H.; Ichimatsu, T.; Saitoh, H.; Mizuki, E.; Ohba, M. Assessment of the efficacy of Japanese Bacillus thuringiensis isolates against the cigarette beetle, Lasioderma serricorne (Coleoptera: Anobiidae). J. Invertebr. Pathol. 2002, 81, 122–126. [Google Scholar] [CrossRef]
- Zhang, J.H.; Wang, C.Z. Comparison of toxicity and deterrence among crystal, spore and thuringiensin A of Bacillus thuringiensis against Helicoverpa armigera (Hubner). Acta Entomol. Sin. 2000, 43, 85–91. [Google Scholar]
- World Health Organization. Guidelines Specification for Bacterial Larvicides for Public Health Use; Publication W. H. O./CDS/CPC/WHOPES/99.2. Report of the W. H. O. Informal Consultation; World Health Organization: Geneva, Switzerland, 1999. [Google Scholar]
- Meretoja, T.; Garlberg, G. The effect of Bacillus thuringiensis and of cell-free supernatants of some other bacteria on the mitotic activity of human lymphocytes. FEMS Microbiol. 1977, 2, 109–111. [Google Scholar] [CrossRef]
- Barjac, H.; de Dedoder, R. Purification and complementary analysis of the thermostable toxin of Bacillus thuringiensis var. thuringiensis. Bull. Soc. Chim. Biol. 1968, 50, 941–944. [Google Scholar]
- Tsai, S.F.; Liu, B.L.; Liao, J.W.; Wang, J.S.; Hwang, J.S.; Wang, S.C.; Tzeng, Y.M.; Ho, S.P. Pulmonary toxicity of thuringiensin administered intratracheally in Sprague-Dawley rats. Toxicology 2003, 186, 205–216. [Google Scholar] [CrossRef]
- Tsai, S.F.; Yang, C.; Wang, S.C.; Wang, J.S.; Hwang, J.S.; Ho, S.P. Effect of thuringiensin on adenylate cyclase in rat cerebral cortex. Toxicol. Appl. Pharmacol. 2004, 194, 34–40. [Google Scholar] [CrossRef]
- Tsai, S.F.; Yang, C.; Liu, B.L.; Hwang, J.S.; Ho, S.P. Role of oxidative stress in thuringiensin-induced pulmonary toxicity. Toxicol. Appl. Pharmacol. 2006, 216, 347–353. [Google Scholar] [CrossRef]
- Beebee, T.; Korner, A.; Bond, R.P. Differential inhibition of mammalian ribonucleic acid polymerases by an exotoxin from Bacillus thuringiensis. The direct observation of nucleoplasmic ribonucleic acid polymerase activity in intact nuclei. Biochem. J. 1972, 127, 619–634. [Google Scholar]
- Sebesta, K.; Horska, K. Mechanism of inhibition of DNA-dependent RNA polymerase by exotoxin of Bacillus thuringiensis. Biochim. Biophys. Acta 1970, 209, 357–376. [Google Scholar] [CrossRef]
- Burgerjon, A.; Biache, G.; Cals, P. Teratology of the Colorado potato beetle, Leptinotarsa decemlineata, as provoked by larval administration of the thermostable toxin of Bacillus thuringiensis. J. Invertebr. Pathol. 1969, 14, 274–278. [Google Scholar] [CrossRef]
- Ignoffo, C.M.; Gregory, B.G. Effects of B. thuringiensis beta-exotoxin on larval maturation, adult longevity, fecundity, and egg viability in several species of lepidoptera. Environ. Entomol. 1972, 1, 269–272. [Google Scholar]
- Espinasse, S.; Gohar, M.; Chaufaux, J.; Buisson, C.; Perchat, S.; Sanchis, V. Correspondence of high levels of beta-exotoxin I and the presence of cry1B in Bacillus thuringiensis. Appl. Environ. Microbiol. 2002, 68, 4182–4186. [Google Scholar] [CrossRef]
- Royalty, R.N.; Hall, H.R.; Lucius, B.A. Effects of thuringiensin on tetranychus urticae (Acari: Tetranychidae) mortality, oviposition rate and feeding. Pestic. Sci. 1991, 33, 383–391. [Google Scholar] [CrossRef]
- Barjac, H.; Burgerjon, A.; Bonnefoi, A. The production of heat-stable toxin by nine serotypes of Bacillus thuringiensis. J. Invertebr. Pathol. 1966, 8, 537–538. [Google Scholar] [CrossRef]
- Kim, Y.T.; Huang, H.T. The β-exotoxin of Bacillus thuringiensis isolation and characterization. J. Invertebr. Pathol. 1970, 15, 100–108. [Google Scholar] [CrossRef]
- Tsun, H.Y.; Liu, C.M.; Tzeng, Y.M. Recovery and purification of thuringiensin from the fermentation broth of Bacillus thuringiensis. Bioseparation 1999, 7, 309–316. [Google Scholar] [CrossRef]
- Tzeng, Y.M. Method of Purifying Thuringiensin. No. U.S. Patent 6268183, 31 July 2001. [Google Scholar]
- Gohar, M.; Perchat, S. Sample preparation for β-exotoxin determination in Bacillus thuringiensis cultures by reversed-phase high-performance liquid chromatography. Anal. Biochem. 2001, 298, 112–117. [Google Scholar] [CrossRef]
- Levinson, B.L.; Kasyan, K.J.; Chiu, S.S.; Currier, T.C.; Gonzalez, J.M. Identification of beta-exotoxin production, plasmids encoding beta-exotoxin, and a new exotoxin in Bacillus thuringiensis by using high-performance liquid chromatography. J. Bacterial. 1990, 172, 3172–3179. [Google Scholar]
- Bekheit, H.K.; Lucas, A.D.; Gee, S.J. Development of an enzymelinked immunosorbent assay for the β-exotoxin of Bacillus thuringiensis. J. Agric. Food Chem. 1993, 41, 1530–1534. [Google Scholar] [CrossRef]
- Liu, C.M.; Tzeng, Y.M. Quantiative analysis of thuringiensin by HPLC using AMP as internal standard. J. Chromatogr. Sci. 1998, 36, 340–344. [Google Scholar] [CrossRef]
- Campbell, D.P.; Dieball, D.E.; Brackett, J.M. Rapid HPLC assay for the beta-exotoxin of Bacillus thuringiensis. J. Agric. Food Chem. 1987, 35, 156–158. [Google Scholar] [CrossRef]
- De rijk, T.C.; van Dam, R.C.; Zomer, P.; Boers, E.A.; de Waard, P.; Mol, H.G. Development and validation of a confirmative LC-MS/MS method for the determination of β-exotoxin thuringiensin in plant protection products and selected greenhouse crops. Anal. Bioanal. Chem. 2013, 405, 1631–1639. [Google Scholar] [CrossRef]
- Perani, M.; Bishop, A.H.; Vaid, A. Prevalence of β-exotoxin, diarrhoeal toxin and specific δ-endotoxin in natural isolates of Bacillus thuringiensis. FEMS Microbiol. Lett. 1998, 160, 55–60. [Google Scholar]
- Espinasse, S.; Gohar, M.; Lereclus, D.; Sanchis, V. An ABC transporter from Bacillus thuringiensis is essential for beta-exotoxin I production. J. Bacteriol. 2002, 184, 5848–5854. [Google Scholar] [CrossRef]
- Espinasse, S.; Gohar, M.; Lereclus, D.; Sanchis, V. An extracytoplasmic-function sigma factor is involved in a pathway controlling beta-exotoxin I production in Bacillus thuringiensis subsp. thuringiensis strain 407-1. J. Bacteriol. 2004, 186, 3108–3116. [Google Scholar] [CrossRef]
- He, J.; Wang, J.; Yin, W.; Shao, X.; Zheng, H.; Li, M.; Zhao, Y.; Sun, M.; Wang, S.; Yu, Z. Complete genome sequence of Bacillus thuringiensis subsp. chinensis strain CT-43. J. Bacteriol. 2011, 193, 3407–3408. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, X.; Ruan, L.; Peng, D.; Li, L.; Sun, M.; Yu, Z. Thuringiensin: A Thermostable Secondary Metabolite from Bacillus thuringiensis with Insecticidal Activity against a Wide Range of Insects. Toxins 2014, 6, 2229-2238. https://doi.org/10.3390/toxins6082229
Liu X, Ruan L, Peng D, Li L, Sun M, Yu Z. Thuringiensin: A Thermostable Secondary Metabolite from Bacillus thuringiensis with Insecticidal Activity against a Wide Range of Insects. Toxins. 2014; 6(8):2229-2238. https://doi.org/10.3390/toxins6082229
Chicago/Turabian StyleLiu, Xiaoyan, Lifang Ruan, Donghai Peng, Lin Li, Ming Sun, and Ziniu Yu. 2014. "Thuringiensin: A Thermostable Secondary Metabolite from Bacillus thuringiensis with Insecticidal Activity against a Wide Range of Insects" Toxins 6, no. 8: 2229-2238. https://doi.org/10.3390/toxins6082229
APA StyleLiu, X., Ruan, L., Peng, D., Li, L., Sun, M., & Yu, Z. (2014). Thuringiensin: A Thermostable Secondary Metabolite from Bacillus thuringiensis with Insecticidal Activity against a Wide Range of Insects. Toxins, 6(8), 2229-2238. https://doi.org/10.3390/toxins6082229




