Exposure Assessment for Italian Population Groups to Deoxynivalenol Deriving from Pasta Consumption
Abstract
:1. Introduction
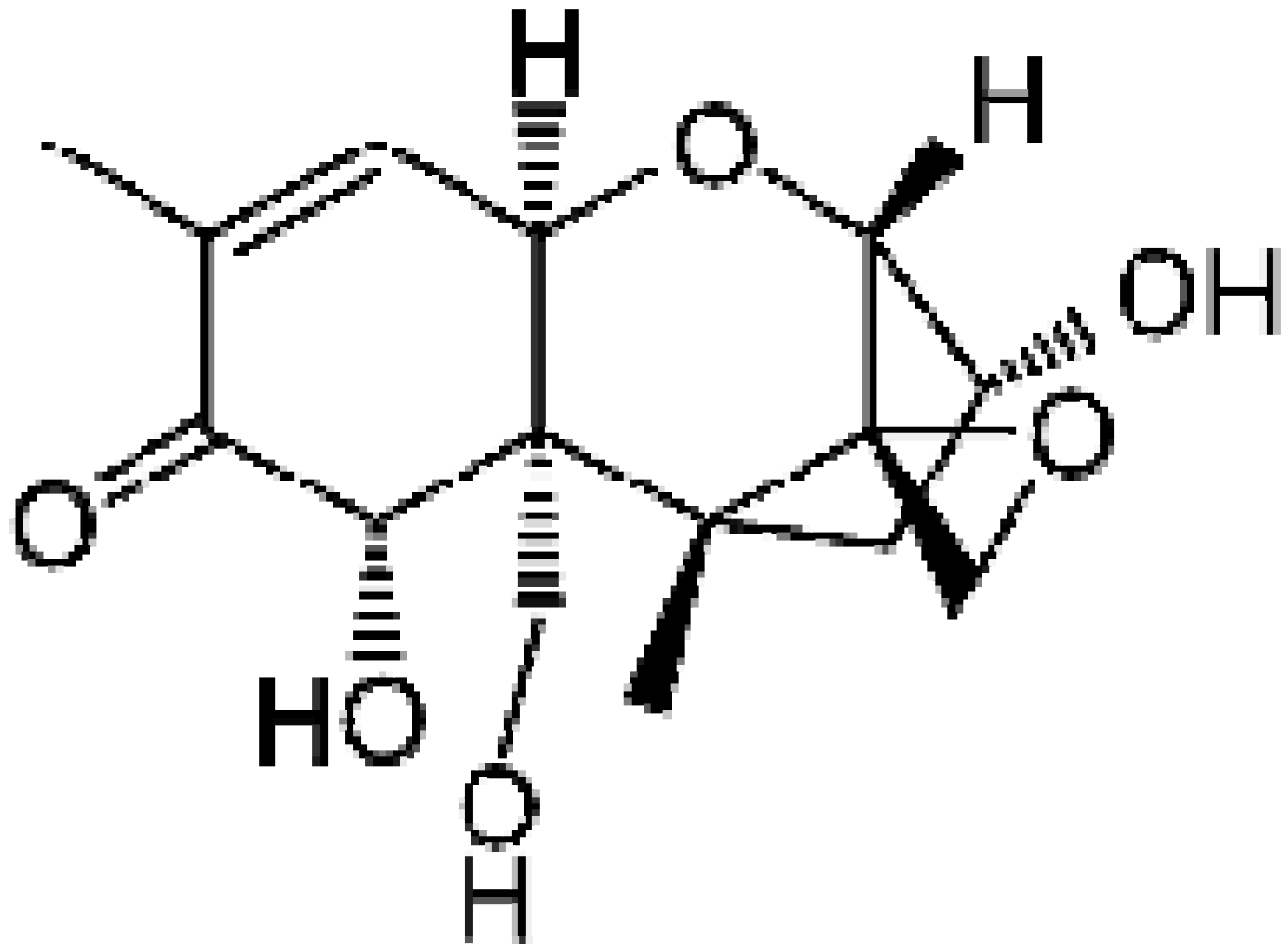
1.1. Legislation on DON
1.2. Exposure Assessment by DON in Humans
Occurrence of DON in Wheat Products
2. Results and Discussion
2.1. Exposure Assessment

2.2. Occurrence Values
| Parameter | Numerical value |
|---|---|
| Number of samples | 472 |
| Samples <LOQ | 371 (78.6%) |
| Samples ≥LOQ | 101 (21.4%) |
| Mean contamination (µg/kg) | 64.8* |
| Median contamination (µg/kg) | 35* |
| 95th percentile of contamination (µg/kg) | 239.4 |
| 99th percentile of contamination (µg/kg) | 337.0 |
| MAX contamination (µg/kg) | 385.7 |
| Parameter | Numerical value |
|---|---|
| Number of samples | 43 |
| Samples <LOQ | 26 (60.5%) |
| Samples ≥LOQ | 17 (39.5%) |
| Mean contamination (µg/kg) | 101.5* |
| Median contamination (µg/kg) | 35* |
| 95th percentile of contamination (µg/kg) | 279.6 |
| 99th percentile of contamination (µg/kg) | 320.9 |
| MAX contamination (µg/kg) | 336.4 |
2.3. Consumption Rate
| Category | Gender | Consumption (g/day) | |||
|---|---|---|---|---|---|
| Mean | 95th percentile | 99th percentile | |||
| Total population | 54.2 | 108.7 | 140.1 | ||
| Consumers only | 59.5 | 110.7 | 141.9 | ||
| Children (3–9.9 years) | Total population | 58.2 | 104.9 | 161.7 | |
| Consumers only | 59.8 | 104.9 | 161.7 | ||
| Adolescent (10–17.9 years) | Total population | M | 63.6 | 128.0 | 133.3 |
| F | 56.6 | 105.3 | 133.3 | ||
| Consumers only | M | 66.7 | 128.0 | 133.3 | |
| F | 61.0 | 105.3 | 133.3 | ||
| Adult (18–64.9 years) | Total population | M | 60.3 | 118.4 | 156.1 |
| F | 47.7 | 100.0 | 134.8 | ||
| Consumers only | M | 66.0 | 121.6 | 156.9 | |
| F | 53.8 | 102.2 | 137.8 | ||
| Elderly (≥65 years) | Total population | M | 61.1 | 109.6 | 129.8 |
| F | 50.7 | 100.6 | 117.4 | ||
| Consumers only | M | 64.3 | 116.5 | 131.2 | |
| F | 54.5 | 110.9 | 121.5 | ||
2.4. Body Weights
| AGE (years) | Boys | Girls |
|---|---|---|
| 3 | 14.3 | 13.9 |
| 4 | 16.3 | 16.1 |
| 5 | 18.3 | 18.2 |
| 9.9 | 31.2 | 31.9 |
| 10–14 | 45 | |
| 15–18 | 60 | |
| >18 | 70 |
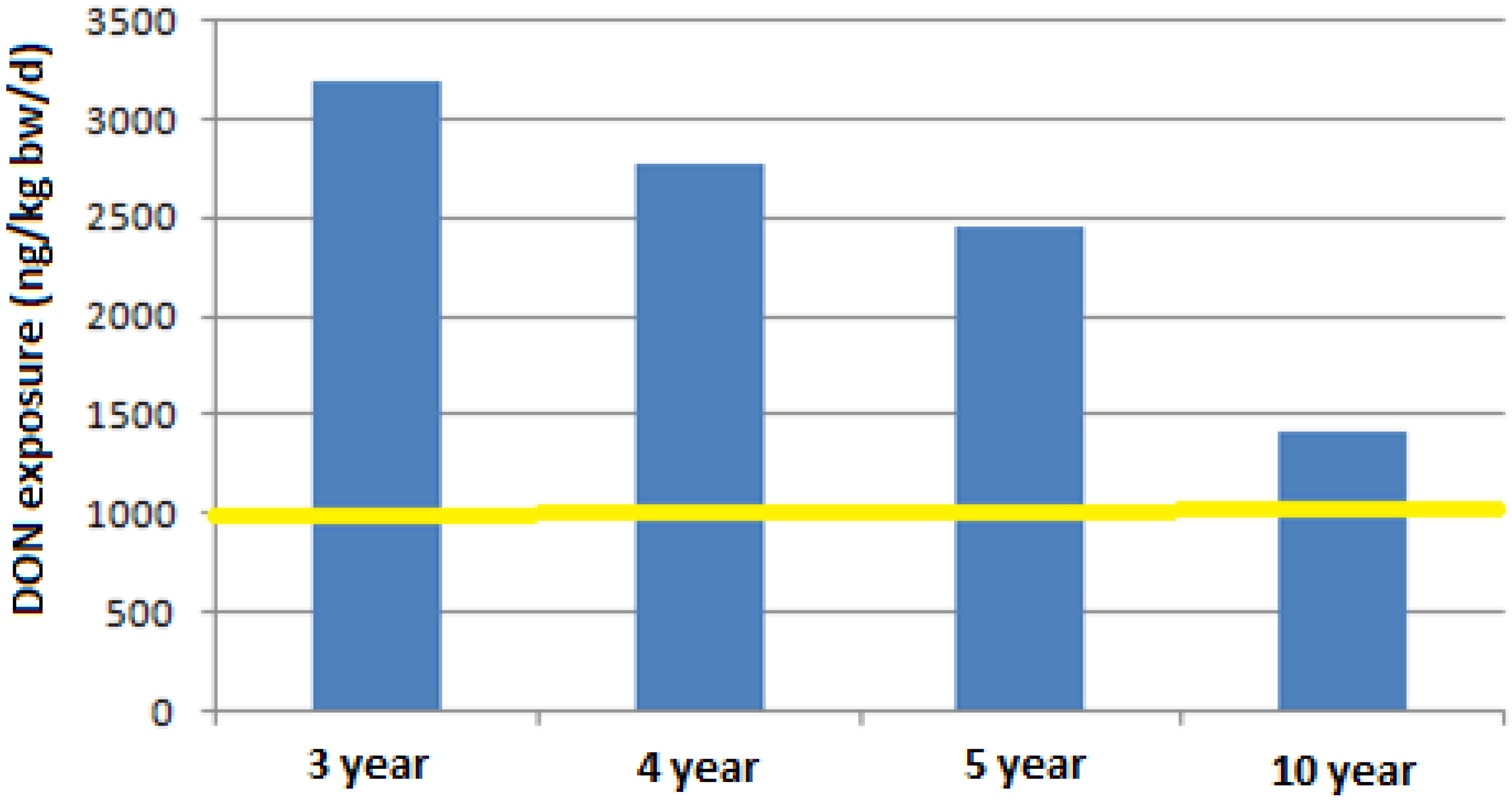
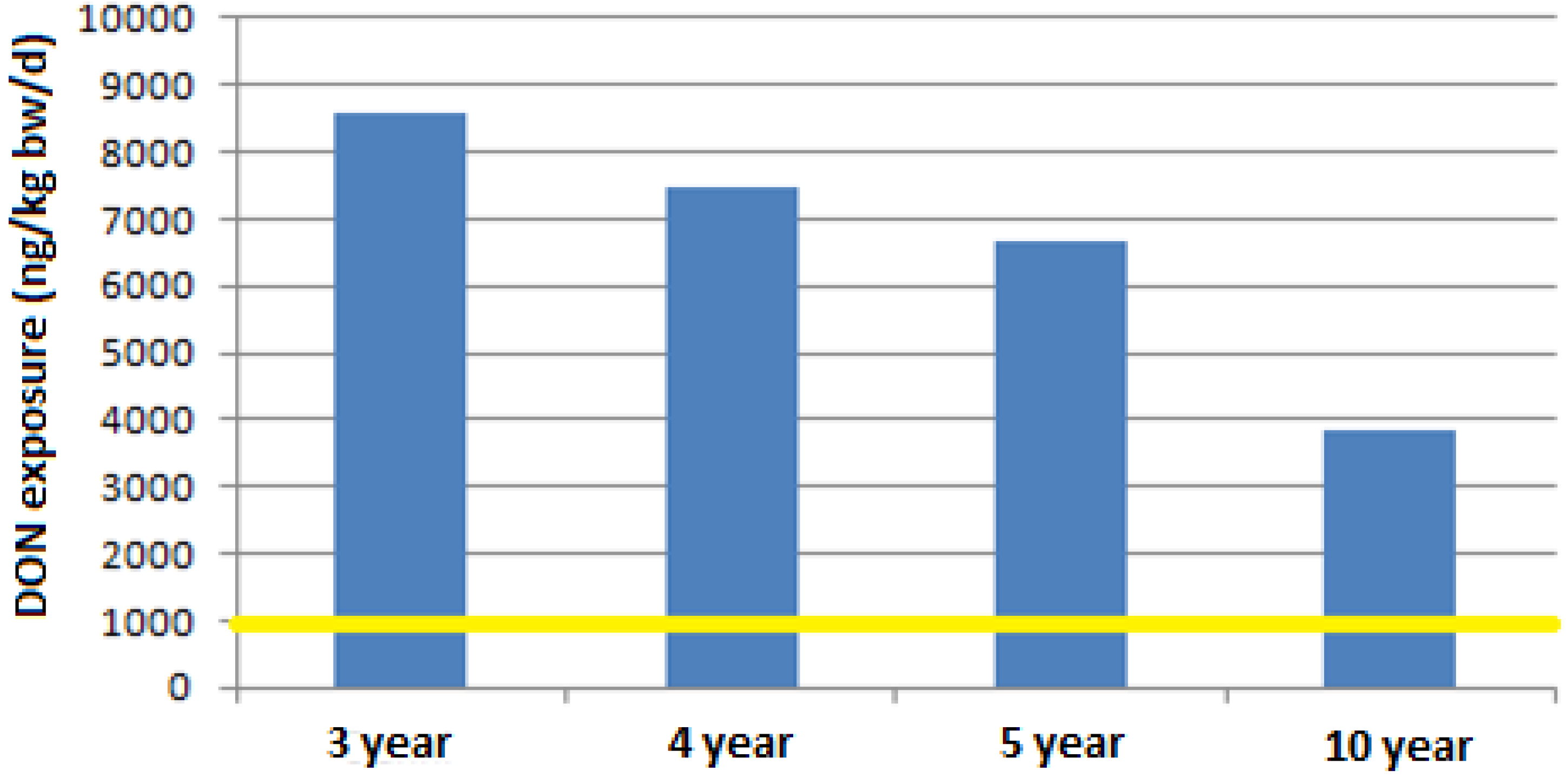
2.5. DON Exposure in Adolescents and Adults
2.6. DON Intakes in Children
| DON contamination (µg/kg) | Weight (kg) /age (years) males | Exposure of males (ng/kg bw/day) | Weight (kg) /age (years) females | Exposure of females (ng/kg bw/day) |
|---|---|---|---|---|
| 64.8* | 14.3/3 | 271 | 13.9/3 | 279 |
| 16.3/4 | 237 | 16.1/4 | 240 | |
| 18.3/5 | 211 | 18.2/5 | 213 | |
| 31.2/10 | 124 | 31.9/10 | 121 | |
| 130 | 14.3/3 | 543 | 13.9/3 | 559 |
| 16.3/4 | 476 | 16.1/4 | 482 | |
| 18.3/5 | 424 | 18.2/5 | 427 | |
| 31.2/10 | 249 | 31.9/10 | 243 | |
| 239 (M)–233 (F) | 14.3/3 | 999 | 13.9/3 | 1002 |
| 273 (M)–269 (F) | 16.3/4 | 1001 | 16.1/4 | 999 |
| 306 (M)–305 (F) | 18.3/5 | 999 | 18.2/5 | 1002 |
| 522 (M)–533 (F) | 31.2/10 | 1000 | 31.9/10 | 999 |
| DON contamination (µg/kg) | Weight (kg) /age (years) males | Exposure of males (ng/kg bw/day) | Weight (kg) /age (years) females | Exposure of females (ng/kg bw/day) |
|---|---|---|---|---|
| 64.8* | 14.3/3 | 732 | 13.9/3 | 753 |
| 16.3/4 | 643 | 16.1/4 | 650 | |
| 18.3/5 | 572 | 18.2/5 | 575 | |
| 31.2/10 | 335 | 31.9/10 | 328 | |
| 89 (M)–86 (F) | 14.3/3 | 1006 | 13.9/3 | 1000 |
| 101 (M)–100 (F) | 16.3/4 | 1002 | 16.1/4 | 1004 |
| 114 (M)–113 (F) | 18.3/5 | 1007 | 18.2/5 | 1003 |
| 193 (M)–198 (F) | 31.2/10 | 1000 | 31.9/10 | 1003 |
3. Experimental Section
3.1. Sampling
- -
- Typology of pasta products (dry, fresh (10%), whole (10%), not egg-pasta, not addressed to baby food chain);
- -
- Random selection among different shape of pasta products (spaghetti, medium size like rigatoni, short size like penne);
- -
- Only one 500 g package per lot, typology, shape and brand.
3.2. Principle of the Method
3.3. Sample Preparation
3.3.1. Extraction
3.3.2. Immunoaffinity Clean-Up
3.4. HPLC Analysis
HPLC Operating Conditions
- -
- Chromatographic column: Symmetry® C18 (Waters, Milford, Massachusetts, UK) reversed phase, 5 µm, 4.6 mm × 150 mm, kept at constant temperature of 40 °C;
- -
- Mobile phase: deionized water: methanol 85:15 v:v;
- -
- Flow rate: 1.0 mL/min;
- -
- UV spectrophotometer regulated at a wavelength of 220 nm;
- -
- Injection Volume: 150 µL.
3.5. Spiking Procedure
3.6. Validation Study
Limit of Quantification
3.7. Calibration Curve
4. Conclusions
Conflicts of Interest
References
- Kushiro, M. Effects of milling and cooking processes on the Deoxynivalenol content in wheat. Int. J. Mol. Sci. 2008, 9, 2127–2145. [Google Scholar] [CrossRef]
- Bretz, M.; Beyer, M.; Cramer, B.; Knecht, A.; Humpf, H.U. Thermal degradation of the Fusarium mycotoxin deoxynivalenol. J. Agric. Food Chem. 2006, 54, 6445–6451. [Google Scholar] [CrossRef]
- Hazel, C.M.; Scudamore, K.A.; Patel, S.; Scriven, F. Deoxynivalenol and other Fusarium mycotoxins in bread, cake, and biscuits produced from UK-grown wheat under commercial and pilot scale conditions. Food Addit. Contam. 2009, 26, 1191–1198. [Google Scholar] [CrossRef]
- Payne, G.A. Ear and kernel rots. In Compendium of Corn Diseases; White, D.G., Ed.; The American Phytopathology Society (APS Press): St. Paul, MN, USA, 1999; pp. 44–47. [Google Scholar]
- Lawlor, P.G.; Lynch, P.B. Mycotoxins in pig feeds. 2: Clinical aspects. Irish Vet. J. 2001, 54, 172–176. [Google Scholar]
- Cheeke, P.R.; Shull, L.R. Natural Toxicants in Feeds and Poisonous Plants; Avi Publishing Company Inc.: Westport, CT, USA, 1985; p. 492. [Google Scholar]
- Prelusky, D.; Rotter, B.; Rotter, R. Toxicology of mycotoxins. In Mycotoxins in Grain: Compounds Other than Aflatoxins; Miller, J., Trenholm, H., Eds.; Eagan Press: St. Paul, MN, USA, 1994; pp. 359–403. [Google Scholar]
- Pestka, J.J.; Smolinski, A.T. Deoxynivalenol: Toxicology and potential effects on humans. J. Toxicol. Environm. Health Part B 2005, 8, 39–69. [Google Scholar] [CrossRef]
- Prelusky, D.B.; Veira, D.M.; Trenholm, H.L. Plasma pharmacokinetics of the mycotoxin Deoxynivalenol following oral and intravenous administration to sheep. J. Environ. Sci. Health B 1985, 20, 603–624. [Google Scholar]
- Prelusky, D.B.; Hartin, K.E.; Trenholm, H.L.; Miller, J.D. Pharmacokinetic fate of carbon-14-labeled Deoxynivalenol in swine. Fundam. Appl. Toxicol. 1988, 10, 276–286. [Google Scholar] [CrossRef]
- Pestka, J.J.; Lin, W.S.; Miller, E.R. Emetic activity of the trichothecene 15 acetyldeoxynivalenol in pigs. Food Chem. Toxicol. 1987, 25, 855–858. [Google Scholar] [CrossRef]
- Prelusky, D.B.; Trenholm, H.L. The efficacy of various classes of anti-emetics in preventing Deoxynivalenol-induced vomiting in swine. Nat. Toxins 1993, 1, 296–302. [Google Scholar] [CrossRef]
- Canady, R.A.; Coker, R.D.; Egan, S.K.; Krska, R.; Kuiper-Goodman, T.; Olsen, M.; Pestka, J.; Resnik, S.; Schlatter, J. Deoxynivalenol. In Safety Evaluation of Certain Mycotoxins in Food; WHO Food Additive Series 47; World Health Organization: Geneva, Switzerland, 2001; pp. 419–555. [Google Scholar]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef]
- Maresca, M. From the gut to the brain: Journey and pathophysiological effects of the food-associated trichothecene mycotoxin Deoxynivalenol. Toxins 2013, 23, 784–820. [Google Scholar] [CrossRef]
- Visconti, A. Problems associated with Fusarium mycotoxins in cereals. Bull. Inst. Compr. Agric. Sci. 2001, 9, 39–55. [Google Scholar]
- Pestka, J.J. Deoxynivalenol: Mechanisms of action, human exposure, and toxicological relevance. Arch. Toxicol. 2010, 84, 663–679. [Google Scholar] [CrossRef]
- Pestka, J.J. Deoxynivalenol-induced proinflammatory gene expression: Mechanisms and pathological sequelae. Toxins 2010, 2, 1300–1317. [Google Scholar] [CrossRef]
- Rocha, O.; Ansari, K.; Doohan, F.M. Effects of trichothecene mycotoxins on eukaryotic cells: A review. Food Addit. Contam. 2005, 22, 369–378. [Google Scholar] [CrossRef]
- Sobrova, P.; Adam, V.; Vasatkova, A.; Beklova, M.; Zeman, L. Deoxynivalenol and its toxicity. Interdiscip. Toxicol. 2010, 3, 94–99. [Google Scholar]
- Rotter, B.A.; Prelusky, D.B.; Pestka, J.J. Toxicology of deoxynivalenol (vomitoxin). J. Toxicol. Environm. Health 1996, 48, 1–34. [Google Scholar]
- Sergent, T.; Parys, M.; Garsou, S.; Pussemier, L.; Schneider, Y.J.; Larondelle, Y. Deoxynivalenol transport across human intestinal Caco-2 cells and its effects on cellular metabolism at realistic intestinal concentrations. Toxicol. Lett. 2006, 164, 167–176. [Google Scholar] [CrossRef]
- FAO. Worldwide Regulations for Mycotoxins in Food and Feed in 2003; Food and Nutrition Paper 81; Food and Agriculture Organization of the United Nations: Rome, Italy, 2003. [Google Scholar]
- Proposed draft maximum levels for Deoxynivalenol in cereals and cereal-based products and associated sampling plans (CX/CF 13/7/7). In Proceedings of Codex Committee on Contaminants in Food 7th Session, Moscow, Russian Federation, 8–12 April 2013.
- European Commission. Commission Regulation (EC) No 1126/2007 of 28 September 2007 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards Fusarium toxins in maize and maize products. Off. J. Eur. Union 2007, L 255/14. [Google Scholar]
- European Commission. Commission Recommendation of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding. Off. J. Eur. Union 2006, L 229/7. [Google Scholar]
- Swanson, S.P.; Rood, H.D.; Behrens, J.C.; Sanders, P.E. Preparation and characterization of the deepoxy trichothecenes: deepoxy HT-2, deepoxy T-2 triol, deepoxy T-2 tetraol, deepoxy 15-monoacetoxyscirpenol, and deepoxy scirpentriol. Appl. Environm. Microb. 1987, 53, 2821–2826. [Google Scholar]
- Maresca, M.; Fantini, J. Some food-associated mycotoxins as potential risk factors in humans predisposed to chronic intestinal inflammatory diseases. Toxicon 2010, 56, 282–294. [Google Scholar] [CrossRef]
- Mezzelani, A.; Landini, M.; Facchiano, F.; Raggi, M.E.; Villa, L.; Molteni, M.; De Santis, B.; Brera, C.; Caroli, A.M.; Milanesi, L.; et al. Environment, dysbiosis, immunity, and sex-specific susceptibility: An evidence-based translational hypothesis for regressive autism pathogenesis. Nutr. Neurosci. 2013. submitted for publication. [Google Scholar]
- Ueno, Y. The toxicology of mycotoxins. CRC Crit. Rev. Toxicol. 1985, 14, 99–132. [Google Scholar] [CrossRef]
- Ueno, Y. Toxicology of trichothecene mycotoxins. ISI Atlas Sci. Pharm. 1988, 2, 121–124. [Google Scholar]
- Meky, F.A.; Turner, P.C.; Ashcroft, A.E.; Miller, J.D.; Qiao, Y.L.; Roth, M.J.; Wild, C.P. Development of a urinary biomarker of human exposure to Deoxynivalenol. Food Chem. Toxicol. 2003, 41, 265–273. [Google Scholar]
- Pieters, M.N.; Freijer, J.L.; Baars, A.J.; Fiolet, D.C.M.; Van Klaveren, J.; Slob, W. Risk assessment of Deoxynivalenol in food. Concentration limits, exposure and effects. Adv. Exp. Med. Biol. 2002, 504, 235–248. [Google Scholar] [CrossRef]
- Turner, P.C.; Rothwell, J.A.; White, K.L.M.; Cade, J.E.; Wild, C.P. Urinary Deoxynivalenol is correlated with cereal intake in individuals from the United Kingdom. Environ. Health Persp. 2008, 116, 21–25. [Google Scholar]
- Hepworth, S.J.; Hardie, L.J.; Fraser, L.K.; Burley, V.J.; Mijal, R.S.; Wild, C.P.; Azad, R.; McKinney, P.A.; Turner, P.C. Deoxynivalenol exposure assessment in a cohort of pregnant women from Bradford, UK. Food Addit. Contam. Part A 2012, 29, 269–276. [Google Scholar]
- Collection of OCCURRENCE DATA of Fusarium TOXIns in FOod and ASSESSment of DIETARY INTake by the POpulation of EU Member States; Scientific Cooperation (SCOOP) 3.2.10.; Directorate General Health and Consumer Protection: Brussels, Belgium, 2003.
- Larsen, J.C.; Hunt, J.; Perrin, I.; Ruckenbauer, P. Workshop on trichothecenes with a focus on DON: Summary report. Toxicol. Lett. 2004, 153, 1–22. [Google Scholar] [CrossRef]
- Pietri, A.; Bertuzzi, T.; Zanetti, M.; Rastelli, S. Presenza di tricoteceni e di ocratossina A in baby-foods e prodotti dietetici ricchi di crusca. In Rapporti ISTISAN 05/42; Miraglia, M., Carlo, B., Eds.; Istituto Superiore di Sanità: Rome, Italy, 2005; pp. 39–42. [Google Scholar]
- Brera, C.; Peduto, A.; Debegnach, F.; Pannunzi, E.; Prantera, E.; Gregori, E.; De Giacomo, M.; De Santis, B. Study of the influence of the milling process on the distribution of Deoxynivalenol content from the caryopsis to cooked pasta. Food Control 2012, 32, 309–312. [Google Scholar]
- Visconti , A.; Haidukowski, M.; Pascale, M.; Silvestri, M. Reduction of Deoxynivalenol during durum wheat processing and spaghetti cooking. Toxicol. Lett. 2004, 153, 181–189. [Google Scholar] [CrossRef]
- Raiola, A.; Meca, G.; Mañes, J.; Ritieni, A. Bioaccessibility of Deoxynivalenol and its natural co-occurrence with Ochratoxin A and Aflatoxin B1 in Italian commercial pasta. Food Chem. Toxicol. 2012, 50, 280–287. [Google Scholar] [CrossRef]
- González-Osnaya, L.; Cortés, C.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Occurrence of Deoxynivalenol and T-2 toxin in bread and pasta commercialised in Spain. Food Chem. 2011, 124, 156–161. [Google Scholar] [CrossRef]
- Bockhorn, I.; Bockhorn, A.; Pohler, S. Deoxynivalenol (DON) in raw and cooked pasta. Mycotoxin Res. 2001, 17 (Suppl. 1), 67–70. [Google Scholar] [CrossRef]
- SCF (Scientific Committee on Food). Opinion of the Scientific Committee on Food on Fusarium toxins. Part 6: Group Evaluation of T-2 Toxin, HT-2 Toxin, Nivalenol and Deoxynivalenol: SCF/CS/CNTM/MYC/27 Final. 2002. Available online: http://europa.eu.int/comm/food/fs/sc/scf/out123_en.pdf (accessed on 15 June 2007).
- Leclercq, C.; Arcella, D.; Piccinelli, R.; Sette, S.; Le Donne, C.; Turrini, A. The Italian National Food Consumption Survey INRAN-SCAI 2005–06: Main results in terms of food consumption. Public Health Nutr. 2009, 12, 2504–2532. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Child Growth Standards. Available online: http://www.who.int/childgrowth/standards/en/ (accessed on 21 November 2013).
- European Food Safety Authority (EFSA). Scientific opinion—Guidance on selected default values to be used by the EFSA Scientific Committee, Scientific Panels and Units in the absence of actual measured data. EFSA J. 2012, 10, 2579–2611. [Google Scholar]
- Thompson, M.; Ellison, S.L.R.; Wood, R. Harmonized guidelines for single-laboratory validation of methods of analysis (IUPAC Technical Report). Pure Appl. Chem. 2002, 74, 835–855. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 401/2006 of 23 February 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. Eur. Union 2006, L70/1. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Brera, C.; Bertazzoni, V.; Debegnach, F.; Gregori, E.; Prantera, E.; De Santis, B. Exposure Assessment for Italian Population Groups to Deoxynivalenol Deriving from Pasta Consumption. Toxins 2013, 5, 2293-2309. https://doi.org/10.3390/toxins5122293
Brera C, Bertazzoni V, Debegnach F, Gregori E, Prantera E, De Santis B. Exposure Assessment for Italian Population Groups to Deoxynivalenol Deriving from Pasta Consumption. Toxins. 2013; 5(12):2293-2309. https://doi.org/10.3390/toxins5122293
Chicago/Turabian StyleBrera, Carlo, Valentina Bertazzoni, Francesca Debegnach, Emanuela Gregori, Elisabetta Prantera, and Barbara De Santis. 2013. "Exposure Assessment for Italian Population Groups to Deoxynivalenol Deriving from Pasta Consumption" Toxins 5, no. 12: 2293-2309. https://doi.org/10.3390/toxins5122293
APA StyleBrera, C., Bertazzoni, V., Debegnach, F., Gregori, E., Prantera, E., & De Santis, B. (2013). Exposure Assessment for Italian Population Groups to Deoxynivalenol Deriving from Pasta Consumption. Toxins, 5(12), 2293-2309. https://doi.org/10.3390/toxins5122293






