A Comparison of the Anti-Tumor Effects of a Chimeric versus Murine Anti-CD19 Immunotoxins on Human B Cell Lymphoma and Pre-B Acute Lymphoblastic Leukemia Cell Lines
Abstract
1. Introduction
2. Materials and Methods
2.1. Construction and Expression of the cHD37 MAb
2.2. Purification of cHD37
2.3. Preparation of ITs
2.4. Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE)
2.5. Cells
2.6. Antigen-Binding Activity of ITs and MAbs
2.7. [3H]Thymidine Incorporation
2.8. Therapy of SCID/NALM-6 Mice
3. Results
3.1. Construction, Expression and Purification of cHD37
3.2. Preparation of ITs

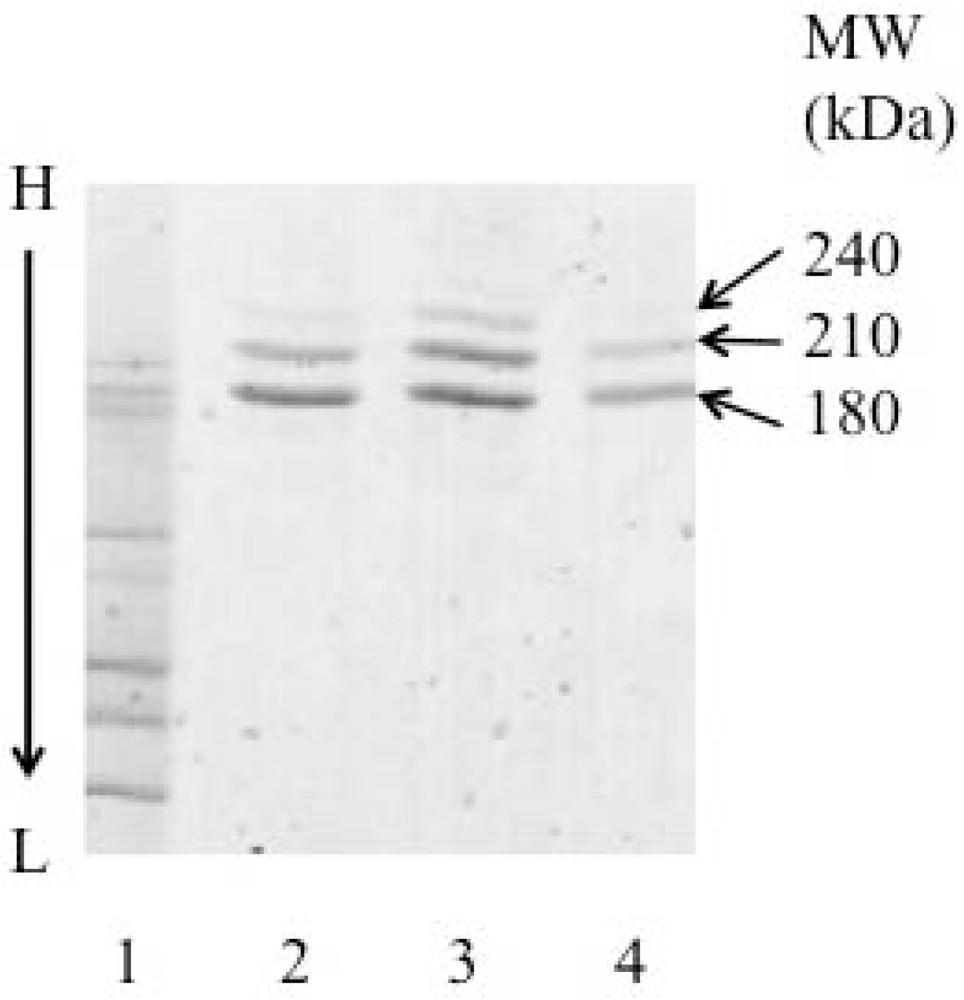
3.3. Binding of ITs and MAbs to CD19+ Cells
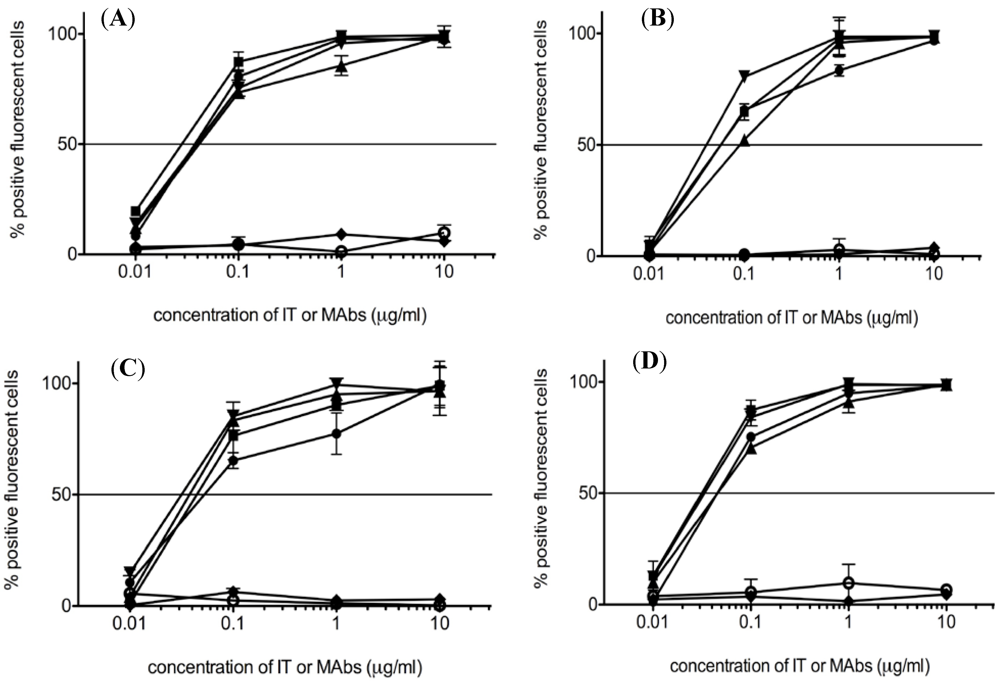
3.4. Cytotoxicity of the ITs in Vitro

3.5. Therapeutic Efficacy of the ITs in SCID/NALM-6 Mice
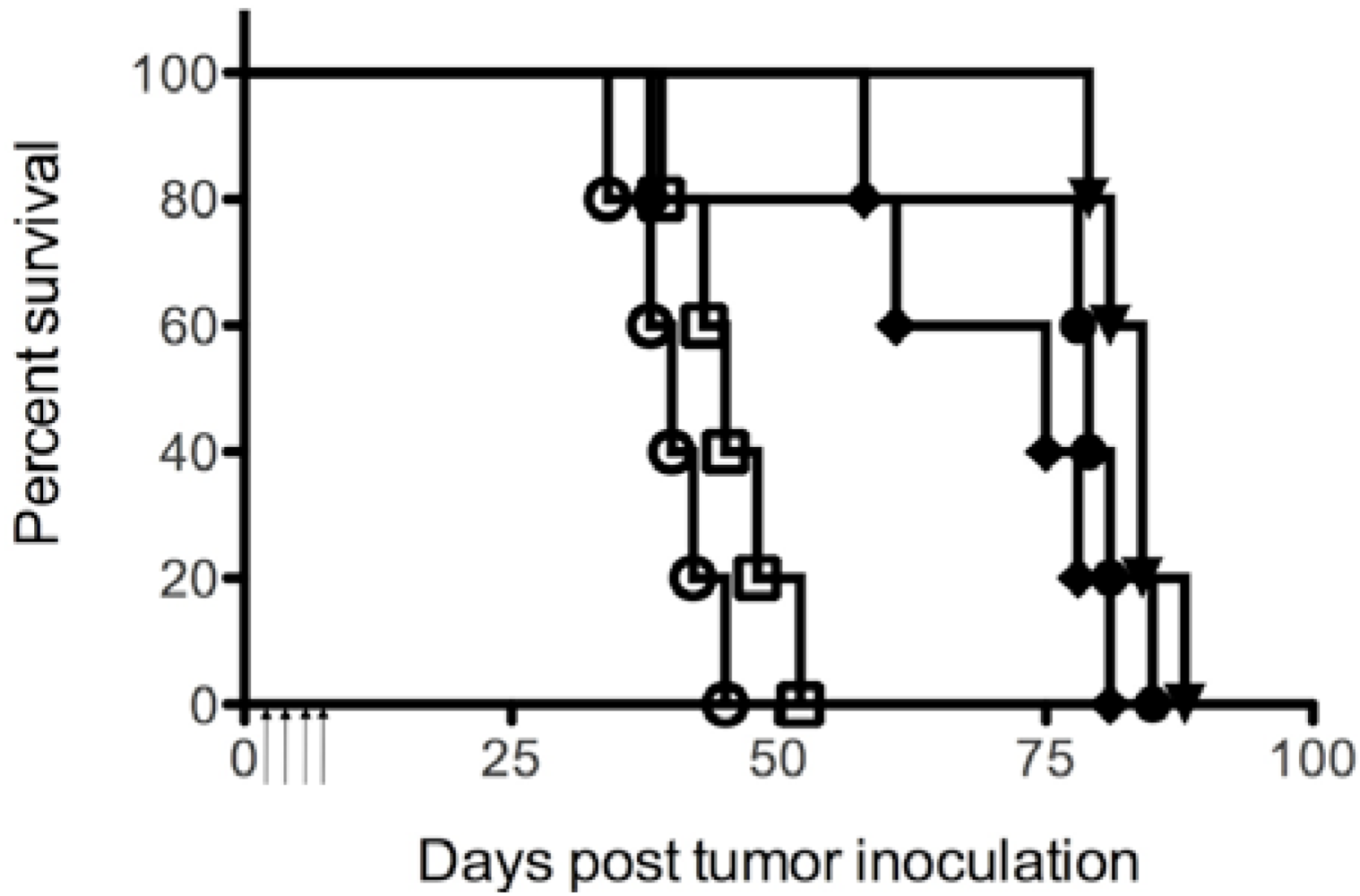
4. Discussion
Acknowledgments
References
- Xie, Y.; Davies, S.M.; Yiang, Y.; Robison, L.L.; Ross, J.A. Trends in leukemia incidence and survival in the United States (1973-1998). Cancer 2003, 97, 2229–2235. [Google Scholar] [CrossRef] [PubMed]
- Moricke, A.; Reiter, A.; Zimmermann, M.; Gadner, H.; Stanulla, M.; Dördelmann, M.; Löning, L.; Beier, R.; Ludwig, W.D.; Ratei, R.; et al. Risk-adjusted therapy of acute lymphoblastic leukemia can decrease treatment burden and improve survival: Treatment results of 2169 unselected pediatric and adolescent patients enrolled in the trial ALL-BFM 95. Blood 2008, 111, 4477–4489. [Google Scholar] [PubMed]
- Hoelzer, D.; Gökbuget, N.; Ottmann, O.; Pui, C.H.; Relling, M.V.; Appelbaum, F.R.; van Dongen, J.J.M.; Szczepanski, T. Acute lymphoblastic leukemia. Hematol. Am. Soc. Hematol. Educ. Progr. 2002, 162–192. [Google Scholar]
- Pui, C.H.; Evans, W.E. Treatment of acute lymphoblastic leukemia. N. Engl. J. Med. 2006, 354, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Chabner, B.A.; Roberts, T.G., Jr. Timeline: Chemotherapy and the war on cancer. Nat. Rev. Cancer 2005, 5, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.A.; Czuczman, M.S. Novel antibodies in the treatment of non-Hodgkin’s lymphoma. Neth. J. Med. 2009, 67, 311–321. [Google Scholar] [PubMed]
- Stashenko, P.; Nadler, L.M.; Hardy, R.; Schlossman, S.F. Characterization of a human B lymphocyte-specific antigen. J. Immunol. 1980, 125, 1678–1685. [Google Scholar]
- Press, O.W.; Howell-Clark, J.; Anderson, S.; Bernstein, I. Retention of B-cell-specific monoclonal antibodies by human lymphoma cells. Blood 1994, 83, 1390–1397. [Google Scholar] [PubMed][Green Version]
- Tedder, T.F.; Isaacs, C.M. Isolation of cDNAs encoding the CD19 antigen of human and mouse B lymphocytes. A new member of the immunoglobulin superfamily. J. Immunol. 1989, 143, 712–717. [Google Scholar] [PubMed]
- Scheuermann, R.H.; Racila, E. CD19 antigen in leukemia and lymphoma diagnosis and immunotherapy. Leuk. Lymphoma 1995, 18, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.C.; Bates, M.P.; Slaughenhoupt, B.L.; Pinkus, G.S.; Schlossman, S.F.; Nadler, L.M. Expression of human B cell-associated antigens on leukemias and lymphomas: A model of human B cell differentiation. Blood 1984, 63, 1424–1433. [Google Scholar] [PubMed]
- Uckun, F.M.; Jaszcz, W.; Ambrus, J.L.; Fauci, A.S.; Gajl-Peczalska, K.; Song, C.W.; Wick, M.R.; Myers, D.E.; Waddick, K.; Ledbetter, J.A. Detailed studies on expression and function of CD19 surface determinant by using B43 monoclonal antibody and the clinical potential of anti-CD19 immunotoxins. Blood 1988, 71, 13–29. [Google Scholar] [PubMed]
- Foxwell, B.M.J.; Blakey, D.C.; Brown, A.N.; Donovan, T.A.; Thorpe, P.E. The preparation of deglycosylated ricin by recombination of glycosidase-treated A- and B-chains; effects of deglycosylation on toxicity and in vivo distribution. Biochim. Biophys. Acta 1987, 923, 59–65. [Google Scholar] [PubMed]
- O'Hare, M.; Roberts, L.M.; Thorpe, P.E.; Watson, G.J.; Prior, B.; Lord, J.M. Expression of ricin A chain in Escherichia coli. FEBS Lett. 1987, 216, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Smallshaw, J.E.; Ghetie, V.; Rizo, J.; Fulmer, J.R.; Trahan, L.L.; Ghetie, M.A.; Vitetta, E.S. Genetic engineering of an immunotoxin to eliminate pulmonary vascular leak in mice. Nat. Biotechnol. 2003, 21, 387–391. [Google Scholar] [PubMed]
- Ghetie, M.A.; May, R.D.; Till, M.; Uhr, J.W.; Ghetie, V.; Knowles, P.P.; Relf, M.; Brown, A.; Wallace, P.M.; Janossy, G.; et al. Evaluation of ricin A chain-containing immunotoxins directed against CD19 and CD22 antigens on normal and malignant human B-cells as potential reagents for in vivo therapy. Cancer Res. 1988, 48, 2610–2617. [Google Scholar] [PubMed]
- Herrera, L.; Farah, R.A.; Pellegrini, V.A.; Aquino, D.B.; Sandler, E.S.; Buchanan, G.R.; Vitetta, E.S. Immunotoxins against CD19 and CD22 are effective in killing precursor-B acute lymphoblastic leukemia cells in vitro. Leukemia 2000, 14, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Herrera, L.; Stanciu-Herrera, C.; Morgan, C.; Ghetie, V.; Vitetta, E.S. Anti-CD19 immunotoxin enhances the activity of chemotherapy in severe combined immunodeficient mice with human pre-B acute lymphoblastic leukemia. Leuk. Lymphoma 2006, 47, 2380–2387. [Google Scholar] [CrossRef] [PubMed]
- Herrera, L.; Yarbrough, S.; Ghetie, V.; Aquino, D.B.; Vitetta, E.S. Treatment of SCID/human B cell precursor ALL with anti-CD19 and anti-CD22 immunotoxins. Leukemia 2003, 17, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Ghetie, M.A.; Tucker, K.; Richardson, J.; Uhr, J.W.; Vitetta, E.S. Eradication of minimal disease in severe combined immunodeficient mice with disseminated Daudi lymphoma using chemotherapy and an immunotoxin cocktail. Blood 1994, 84, 702–707. [Google Scholar] [PubMed][Green Version]
- Ghetie, M.A.; Tucker, K.; Richardson, J.; Uhr, J.W.; Vitetta, E.S. The antitumor activity of an anti-CD22 immunotoxin in SCID mice with disseminated Daudi lymphoma is enhanced by either an anti-CD19 antibody or an anti-CD19 immunotoxin. Blood 1992, 80, 2315–2320. [Google Scholar] [PubMed]
- Conry, R.M.; Khazaeli, M.B.; Saleh, M.N.; Ghetie, V.; Vitetta, E.S.; Liu, T.; LoBuglio, A.F. Phase I trial of an anti-CD19 deglycosylated ricin A chain immunotoxin in non-Hodgkin’s lymphoma: Effect of an intensive schedule of administration. J. Immunother. Emphas. Tumor Immunol. 1995, 18, 231–241. [Google Scholar]
- Stone, M.J.; Sausville, E.A.; Fay, J.W.; Headlee, D.; Collins, R.H.; Figg, W.D.; Stetler-Stevenson, M.; Jain, V.; Jaffe, E.S.; Solomon, D.; et al. A phase I study of bolus versus continuous infusion of the anti-CD19 immunotoxin, IgG-HD37-dgA, in patients with B-cell lymphoma. Blood 1996, 88, 1188–1197. [Google Scholar] [PubMed]
- Messmann, R.A.; Vitetta, E.S.; Headlee, D.; Senderowicz, A.M.; Figg, W.D.; Schindler, J.; Michiel, D.F.; Creekmore, S.; Steinberg, S.M.; Kohler, D.; et al. A phase I study of combination therapy with immunotoxins IgG-HD37-deglycosylated ricin A chain (dgA) and IgG-RFB4-dgA (Combotox) in patients with refractory CD19(+), CD22(+) B cell lymphoma. Clin. Cancer Res. 2000, 6, 1302–1313. [Google Scholar] [PubMed]
- Coloma, M.J.; Hastings, A.; Wims, L.A.; Morrison, S.L. Novel vectors for the expression of antibody molecules using variable regions generated by polymerase chain reaction. J. Immunol. Methods 1992, 152, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Ghetie, V.; Till, M.A.; Ghetie, M.A.; Tucker, T.; Porter, J.; Patzer, E.J.; Richardson, J.A.; Uhr, J.W.; Vitetta, E.S. Preparation and characterization of conjugates of recombinant CD4 and deglycosylated ricin A chain using different cross-linkers. Bioconjug. Chem. 1990, 1, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Ghetie, V.; Thorpe, P.; Ghetie, M.A.; Knowles, P.; Uhr, J.W.; Vitetta, E.S. The GLP large scale preparation of immunotoxins containing deglycosylated ricin A chain and a hindered disulfide bond. J. Immunol. Methods 1991, 142, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Knowles, P.P.; Thorpe, P.E. Purification of immunotoxins containing ricin A-chain and abrin A-chain using blue sepharose CL-6B. Anal. Biochem. 1987, 160, 440–443. [Google Scholar] [CrossRef] [PubMed]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tsai, L.K.; Pop, L.M.; Liu, X.; Vitetta, E.S. A Comparison of the Anti-Tumor Effects of a Chimeric versus Murine Anti-CD19 Immunotoxins on Human B Cell Lymphoma and Pre-B Acute Lymphoblastic Leukemia Cell Lines. Toxins 2011, 3, 409-419. https://doi.org/10.3390/toxins3040409
Tsai LK, Pop LM, Liu X, Vitetta ES. A Comparison of the Anti-Tumor Effects of a Chimeric versus Murine Anti-CD19 Immunotoxins on Human B Cell Lymphoma and Pre-B Acute Lymphoblastic Leukemia Cell Lines. Toxins. 2011; 3(4):409-419. https://doi.org/10.3390/toxins3040409
Chicago/Turabian StyleTsai, Lydia K., Laurentiu M. Pop, Xiaoyun Liu, and Ellen S. Vitetta. 2011. "A Comparison of the Anti-Tumor Effects of a Chimeric versus Murine Anti-CD19 Immunotoxins on Human B Cell Lymphoma and Pre-B Acute Lymphoblastic Leukemia Cell Lines" Toxins 3, no. 4: 409-419. https://doi.org/10.3390/toxins3040409
APA StyleTsai, L. K., Pop, L. M., Liu, X., & Vitetta, E. S. (2011). A Comparison of the Anti-Tumor Effects of a Chimeric versus Murine Anti-CD19 Immunotoxins on Human B Cell Lymphoma and Pre-B Acute Lymphoblastic Leukemia Cell Lines. Toxins, 3(4), 409-419. https://doi.org/10.3390/toxins3040409




