Paradoxical Exception to Island Tameness: Increased Defensiveness in an Insular Population of Rattlesnakes
Abstract
1. Introduction
2. Results
2.1. Snake Rattling
2.2. Snake Cloacal Gland Discharge
2.3. Snake Bite Attempts, Latency to Capture, and Venom Expulsion
2.4. Relative Head Size
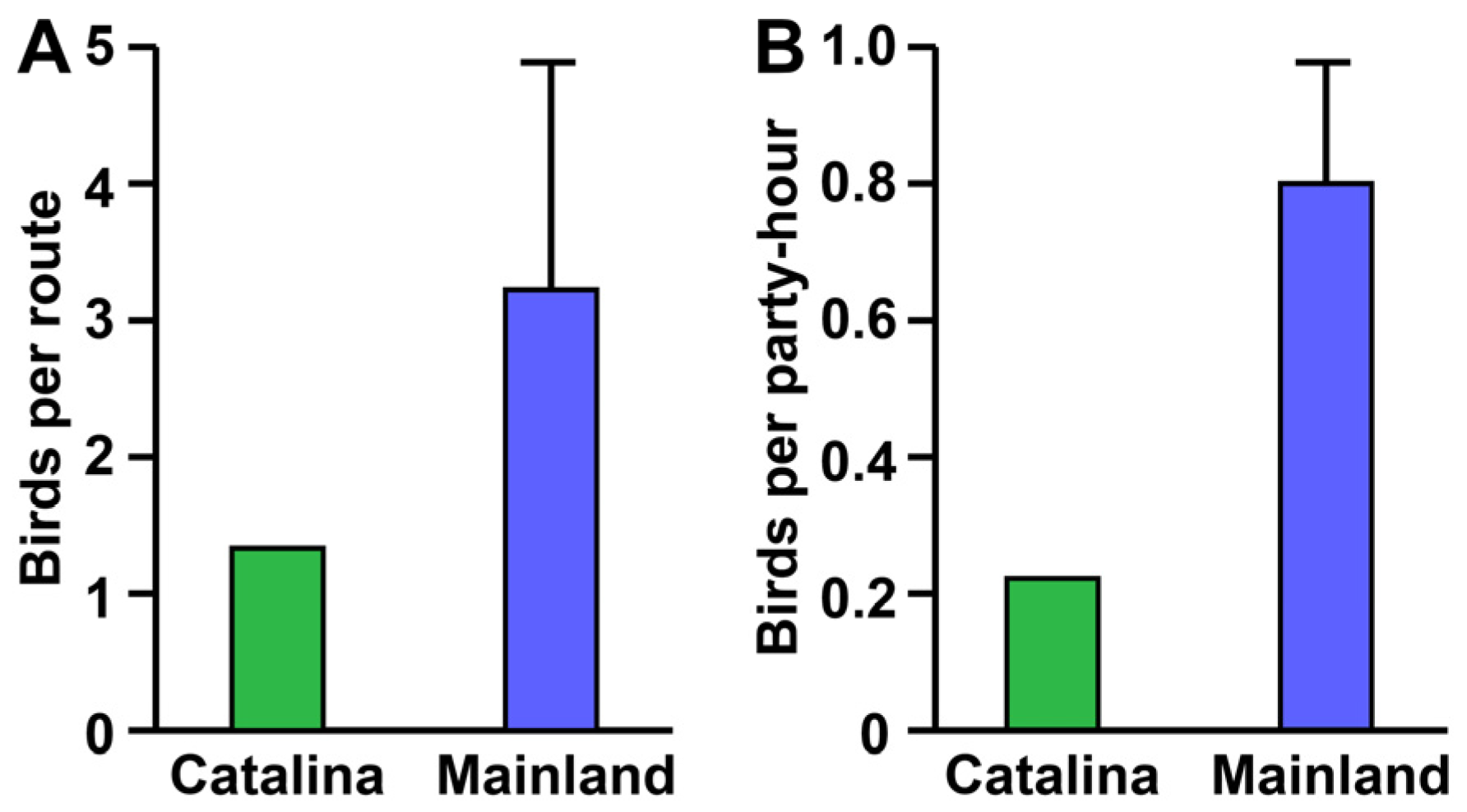
2.5. Mainland versus Island Raptor Abundance
2.6. Mainland versus Island Mammal Abundance
3. Discussion
3.1. Rattling
3.2. Cloacal Gland Discharge
3.3. Biting and Venom Expenditure
3.4. Possible Causes of Increased Defensiveness
4. Conclusions
5. Materials and Methods
5.1. Snake Subjects
5.2. Snake Behavioral Trials
5.3. Populations of Snake Predators and Antagonists
5.4. Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Institutional Animal Care and Use Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Darwin, C. Journal of Researches into the Geology and Natural History of the Various Countries Visited during the Voyage of H.M.S. Beagle Round the World; Henry Coburn: London, UK, 1839; pp. 1–615. [Google Scholar]
- Adler, G.H.; Levins, R. The island syndrome in rodent populations. Q. Rev. Biol. 1994, 69, 473–490. [Google Scholar] [CrossRef]
- McNab, B.K. Minimizing energy expenditure facilitates vertebrate persistence on oceanic islands. Ecol. Lett. 2002, 5, 693–704. [Google Scholar] [CrossRef]
- Novosolov, M.; Raia, P.; Meiri, S. The island syndrome in lizards. Global Ecol. Biogeogr. 2013, 22, 184–191. [Google Scholar] [CrossRef]
- Raia, P.; Guarino, F.M.; Turano, M.; Polese, G.; Rippa, D.; Carotenuto, F.; Monti, D.M.; Cardi, M.; Fulgione, D. The blue lizard spandrel and the island syndrome. BMC Evol. Biol. 2010, 10, 289. [Google Scholar] [CrossRef] [PubMed]
- McNab, B.K. Energy conservation and the evolution of flightlessness in birds. Am. Nat. 1994, 144, 628–642. [Google Scholar] [CrossRef]
- Blumstein, D.T.; Daniel, J.C. The loss of anti-predator behaviour following isolation on islands. Proc. R. Soc. Lond. B Biol. Sci. 2005, 272, 1663–1668. [Google Scholar] [CrossRef] [PubMed]
- Cooper, W.E., Jr.; Perez-Mellado, V. Island tameness: Reduced escape responses and morphological and physiological antipredatory adaptations related to escape in lizards. In Islands and Evolution; Perez-Mellado, V., Ramón, C., Eds.; Institut Menorquí d’Estudis: Maó-Mahón, Spain, 2010; pp. 231–253. [Google Scholar]
- Burbidge, A.A.; Manly, B.F.J. Mammal extinctions on Australian islands: Causes and conservation implications. J. Biogeogr. 2002, 29, 465–473. [Google Scholar] [CrossRef]
- Clavero, M.; García-Berthou, E. Homogenization dynamics and introduction routes of invasive freshwater fish in the Iberian Peninsula. Ecol. Appl. 2006, 16, 2313–2324. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, T.M.; Cassey, P.; Duncan, R.P.; Evans, K.L.; Gaston, K.J. Avian extinction and mammalian introductions on oceanic islands. Science 2004, 305, 1955–1958. [Google Scholar] [CrossRef]
- Duncan, R.P.; Boyer, A.G.; Blackburn, T.M. Magnitude and variation of prehistoric bird extinctions in the Pacific. Proc. Natl. Acad. Sci. USA 2013, 110, 6436–6441. [Google Scholar] [CrossRef]
- Loehle, C.; Eschenbach, W. Historical bird and terrestrial mammal extinction rates and causes. Divers. Distrib. 2012, 18, 84–91. [Google Scholar] [CrossRef]
- Case, T.J.; Bolger, D.T. The role of introduced species in shaping the distribution and abundance of island reptiles. Evol. Ecol. 1991, 5, 272–290. [Google Scholar] [CrossRef]
- Case, T.J.; Bolger, D.T.; Richman, A.D. Reptilian extinctions: The last ten thousand years. In Conservation Biology; Fiedler, P.L., Jain, S.K., Eds.; Springer: Boston, MA, USA, 1992; pp. 91–125. [Google Scholar] [CrossRef]
- Hedges, S.B.; Conn, C.E. A new skink fauna from Caribbean islands (Squamata, Mabuyidae, Mabuyinae). Zootaxa 2012, 3288, 1–244. [Google Scholar] [CrossRef]
- Martins, M.; Lillywhite, H.B. Ecology of snakes on islands. In Islands and Snakes: Isolation and Adaptive Evolution; Lillywhite, H.B., Martins, M., Eds.; Oxford University Press: New York, NY, USA, 2019; pp. 1–44. [Google Scholar]
- Delibes, M.; Blazquez, M.C. Tameness of insular lizards and loss of biological diversity. Conserv. Biol. 1998, 12, 1142–1143. [Google Scholar] [CrossRef]
- Cooper, W.E.; Pyron, R.A.; Garland, T. Island tameness: Living on islands reduces flight initiation distance. Proc. R. Soc. B-Biol. Sci. 2014, 281, 20133019. [Google Scholar] [CrossRef] [PubMed]
- Samia, D.S.M.; Blumstein, D.T.; Stankowich, T.; Cooper, W.E. Fifty years of chasing lizards: New insights advance optimal escape theory. Biol. Rev. 2015, 91, 349–366. [Google Scholar] [CrossRef]
- Brock, K.M.; Bednekoff, P.A.; Pafilis, P.; Foufopoulos, J. Evolution of antipredator behavior in an island lizard species, Podarcis erhardii (Reptilia: Lacertidae): The sum of all fears? Evolution 2015, 69, 216–231. [Google Scholar] [CrossRef] [PubMed]
- Gavriilidi, I.; De Meester, G.; Van Damme, R.; Baeckens, S. How to behave when marooned: The behavioural component of the island syndrome remains underexplored. Biol. Lett. 2022, 18, 20220030. [Google Scholar] [CrossRef]
- Gérard, A.; Jourdan, H.; Cugnière, C.; Millon, A.; Vidal, E. Is naïveté forever? Alien predator and aggressor recognition by two endemic island reptiles. Naturwissenschaften 2014, 101, 921–927. [Google Scholar] [CrossRef]
- Vitousek, M.N.; Romero, L.M.; Tarlow, E.; Cyr, N.E.; Wikelski, M. Island tameness: An altered cardiovascular stress response in Galápagos marine iguanas. Physiol. Behav. 2010, 99, 544–548. [Google Scholar] [CrossRef]
- Monti, D.M.; Raia, P.; Vroonen, J.; Maselli, V.; Damme, R.; Fulgione, D. Physiological change in an insular lizard population confirms the reversed island syndrome. Biol. J. Linn. Soc. 2013, 108, 144–150. [Google Scholar] [CrossRef]
- Sagonas, K.; Valakos, E.D.; Pafilis, P. The impact of insularity on the thermoregulation of a Mediterranean lizard. J. Therm. Biol. 2013, 38, 480–486. [Google Scholar] [CrossRef]
- Ladyman, M.; Bradshaw, D. The influence of dehydration on the thermal preferences of the western tiger snake, Notechis scutatus. J. Comp. Physiol. B 2003, 173, 239–246. [Google Scholar] [CrossRef]
- Allendorf, F.W.; Hard, J.J. Human-induced evolution caused by unnatural selection through harvest of wild animals. Proc. Natl. Acad. Sci. USA 2009, 106 (Suppl. 1), 9987–9994. [Google Scholar] [CrossRef]
- Delibes, M.; Blázquez, M.D.C.; Soriano, L.; Revilla, E.; Godoy, J.A. High antipredatory efficiency of insular lizards: A warning signal of excessive specimen collection? PLoS ONE 2011, 6, e29312. [Google Scholar] [CrossRef]
- Sasaki, K.; Fox, S.F.; Duvall, D. Rapid evolution in the wild: Changes in body size, life-history traits, and behavior in hunted populations of the Japanese mamushi snake. Conserv. Biol. 2008, 23, 93–102. [Google Scholar] [CrossRef]
- Stone, P.A.; Snell, H.L.; Snell, H.M. Behavioral diversity as biological diversity: Introduced cats and lava lizard wariness. Conserv. Biol. 1994, 8, 569–573. [Google Scholar] [CrossRef]
- Li, B.; Belasen, A.; Pafilis, P.; Bednekoff, P.; Foufopoulos, J. Effects of feral cats on the evolution of anti-predator behaviours in island reptiles: Insights from an ancient introduction. Proc. R. Soc. Lond. B Biol. Sci. 2014, 281, 20140339. [Google Scholar] [CrossRef]
- Freidenfelds, N.A.; Robbins, T.R.; Langkilde, T. Evading invaders: The effectiveness of a behavioral response acquired through lifetime exposure. Behav. Ecol. 2012, 23, 582–590. [Google Scholar] [CrossRef]
- Losos, J.B.; Schoener, T.W.; Langerhans, R.B.; Spiller, D.A. Rapid temporal reversal in predator-driven natural selection. Science 2006, 314, 1111. [Google Scholar] [CrossRef]
- Berger, S.; Wikelski, M.; Romero, L.M.; Kalko, E.K.V.; Rödl, T. Behavioral and physiological adjustments to new predators in an endemic island species, the Galápagos marine iguana. Horm. Behav. 2007, 52, 653–663. [Google Scholar] [CrossRef]
- Rödl, T.; Berger, S.; Romero, L.M.; Wikelski, M. Tameness and stress physiology in a predator-naive island species confronted with novel predation threat. Proc. R. Soc. Lond. B Biol. Sci. 2007, 274, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Nelsen, D.R.; Nisani, Z.; Cooper, A.M.; Fox, G.A.; Gren, E.C.K.; Corbit, A.G.; Hayes, W.K. Poisons, toxungens, and venoms: Redefining and classifying toxic biological secretions and the organisms that employ them. Biol. Rev. 2014, 89, 450–465. [Google Scholar] [CrossRef] [PubMed]
- Graves, B.M.; Duvall, D. Evidence of an alarm pheromone from the cloacal sacs of prairie rattlesnakes. Southwest. Nat. 1988, 33, 339. [Google Scholar] [CrossRef]
- Hayes, W.K.; Duvall, D. A field study of prairie rattlesnake predatory strikes. Herpetologica 1991, 47, 78–81. [Google Scholar]
- Rowe, M.P.; Owings, D.H. Probing, assessment, and management during interactions between ground squirrels and rattlesnakes. Ethology 1990, 86, 237–249. [Google Scholar] [CrossRef]
- Sisk, N.R.; Jackson, J.F. Tests of two hypotheses for the origin of the crotaline rattle. Copeia 1997, 1997, 485–495. [Google Scholar] [CrossRef]
- Tiebout, H.M. Caudal luring by a temperate colubrid snake, Elaphe obsoleta, and its implications for the evolution of the rattle among rattlesnakes. J. Herpetol. 1997, 31, 290–292. [Google Scholar] [CrossRef]
- Owings, D.H.; Rowe, M.P.; Rundus, A. The rattling sound of rattlesnakes (Crotalus viridis) as a communicative resource for ground squirrels (Spermophilus beecheyi) and burrowing owls (Athene cunicularia). J. Comp. Psychol. 2002, 116, 197–205. [Google Scholar] [CrossRef]
- Rabatsky, A. Shakin′ It Island Style: Causes and Consequences of the Evolutionary Vestigialization of the Rattling System in Insular Rattlesnakes. Unpublished. Ph.D. Thesis, University of Louisiana, Lafayette, LA, USA, 2007; pp. 1–204.
- Martins, M.; Arnaud, G.; Murillo-Quero, R. Exploring hypotheses about the loss of the rattle in rattlesnakes: How arboreal is the Isla Santa Catalina rattleless rattlesnake, Crotalus catalinensis? South. Am. J. Herpetol. 2008, 3, 162–167. [Google Scholar] [CrossRef]
- Klauber, L.M. Rattlesnakes Their Habits, Life Histories, and Influence on Mankind, 2nd ed.; University of California Press: Berkeley, CA, USA, 1972; pp. 1–1580. [Google Scholar]
- Duvall, D.; King, M.B.; Gutzwiller, K.J. Behavioral ecology and ethology of the prairie rattlesnake. Natl. Geogr. Res. 1985, 1, 80–111. [Google Scholar]
- Campbell, J.A.; Lamar, W.W. The Venomous Reptiles of the Western Hemisphere; Comstock Publishing Associates: Ithaca, NY, USA, 2004; pp. 1–870. [Google Scholar]
- Grismer, L.L. Amphibians and Reptiles of Baja California, Including Its Pacific Islands, and the Islands in the Sea of Corte’s; University of California Press: Berkeley, CA, USA, 2002. [Google Scholar]
- Ashton, K.G. Notes on the island population of the western rattlesnake, Crotalus viridis. Herpetol. Rev. 2000, 31, 214–217. [Google Scholar]
- Rubio, M. Rattlesnake: Portrait of A Predator, 1st ed.; Smithsonian Institution Press: Washington, DC, USA, 1998; pp. 1–272. [Google Scholar]
- Guthrie, D.A. Fossil vertebrates from Pleistocene terrestrial deposits on the northern Channel Islands, Southern California. In Contributions to the Geology of the Northern Channel Islands, Southern California; Weigand, P.W., Ed.; Pacific Section American Association of Petroleum Geologists: Bakersfield, CA, USA, 1998; pp. 187–192. [Google Scholar]
- Guthrie, D.A. New information on the prehistoric fauna of San Miguel Island, California. In Third California Islands Symposium Recent Advances in Research on the California Islands; Hochberg, F.G., Ed.; Santa Barbara Museum of Natural History: Santa Barbara, CA, USA, 1993; pp. 405–416. [Google Scholar]
- Beaman, K.R.; Hayes, W.K. Annotated checklist of the rattlesnakes (second edition). In The Biology of Rattlesnakes II; Dreslik, M.J., Hayes, W.K., Beaupre, S.J., Mackessy, S.P., Eds.; ECO Herpetological Publishing and Distribution: Rodeo, NM, USA, 2017; pp. 13–21. [Google Scholar]
- Hayes, W.K. The snake venom-metering controversy: Levels of analysis, assumptions, and evidence. In The Biology of Rattlesnakes; Hayes, W.K., Cardwell, M.D., Beaman, K.R., Bush, S.P., Eds.; Loma Linda University Press: Loma Linda, CA, USA, 2008; pp. 191–220. [Google Scholar]
- Herbert, S.S.; Hayes, W.K. Denim clothing reduces venom expenditure by rattlesnakes striking defensively at model human limbs. Ann. Emerg. Med. 2009, 54, 830–836. [Google Scholar] [CrossRef]
- Hayes, W.K.; Herbert, S.S.; Rehling, G.C.; Gennaro, J.F. Factors that influence venom expenditure in viperids and other snake species during predatory and defensive contexts. In Biology of the Vipers; Schuett, G.W., Höggren, M., Douglas, M.E., Greene, H.W., Eds.; Eagle Mountain Publishing: Eagle Mountain, UT, USA, 2002; pp. 207–233. [Google Scholar]
- McLain, D.R.; Thomas, D.H. Year-to-year fluctuations of the California countercurrent and effects on marine organisms. Calif. Coop. Oceanic Fish. Invest. Rep. 1983, 24, 165–181. [Google Scholar]
- Salerno, P.E.; Chan, L.M.; Pauly, G.B.; Funk, W.C.; Robertson, J.M. Near-shore island lizard fauna shaped by a combination of human-mediated and natural dispersal. J. Biogeogr. 2023, 50, 116–129. [Google Scholar] [CrossRef]
- Nava-Landeros, R.A. Changes in Morphology and Behavior of the Coronado Island Rattlesnake (Crotalus oreganus caliginis), an Insular Population of The Western Rattlesnake (C. oreganus). Unpublished. Master’s Thesis, San Diego State University, San Diego, CA, USA, 2022. [Google Scholar]
- Allf, B.C.; Sparkman, A.M.; Pfennig, D.W. Microevolutionary change in mimicry? Potential erosion of rattling behaviour among nonvenomous snakes on islands lacking rattlesnakes. Ethol. Ecol. Evol. 2021, 33, 125–136. [Google Scholar] [CrossRef]
- Fedriani, J.M.; Fuller, T.K.; Sauvajot, R.M. Does availability of anthropogenic food enhance densities of omnivorous mammals? An example with coyotes in southern California. Ecography 2001, 24, 325–331. [Google Scholar] [CrossRef]
- Heilbrun, R.D.; Silvy, N.J.; Peterson, M.J.; Tewes, M.E. Estimating bobcat abundance using automatically triggered cameras. Wildl. Soc. Bull. 2006, 34, 69–73. [Google Scholar] [CrossRef]
- Ordeñana, M.A.; Crooks, K.R.; Boydston, E.E.; Fisher, R.N.; Lyren, L.M.; Siudyla, S.; Haas, C.D.; Harris, S.; Hathaway, S.A.; Turschak, G.M.; et al. Effects of urbanization on carnivore species distribution and richness. J. Mammal. 2010, 91, 1322–1331. [Google Scholar] [CrossRef]
- Nowak, R.M.; Walker, E.P. Walker’s Mammals of the World, 5th ed.; Johns Hopkins University Press: Baltimore, MD, USA, 1991; pp. 1–606. [Google Scholar]
- Crooks, K.R. Relative sensitivities of mammalian carnivores to habitat fragmentation. Conserv. Biol. 2002, 16, 488–502. [Google Scholar] [CrossRef]
- Rick, T.C.; Walker, P.L.; Willis, L.M.; Noah, A.C.; Erlandson, J.M.; Vellanoweth, R.L.; Braje, T.J.; Kennett, D.J. Dogs, humans and island ecosystems: The distribution, antiquity and ecology of domestic dogs (Canis familiaris) on California′s Channel Islands, USA. Holocene 2008, 18, 1077–1087. [Google Scholar] [CrossRef]
- Sweitzer, R.A. Conservation implications of feral pigs in island and mainland ecosystems, and a case study of feral pig expansion in California. In Proceedings of the Eighteenth Vertebrate Pest Conference, Costa Mesa, CA, USA, 2–5 March 1998; Baker, R.O., Crabb, A.C., Eds.; University of California: Davis, CA, USA, 1998; pp. 26–34. [Google Scholar]
- Guttilla, D.A.; Stapp, P. Effects of sterilization on movements of feral cats at a wildland-urban interface. J. Mammal. 2010, 91, 482–489. [Google Scholar] [CrossRef]
- King, J.L.; Duncan, C.L.; Garcelon, D.K. Status of the Santa Catalina Island fox thirteen years after its decline. Monogr. West. N. Am. Nat. 2014, 7, 382–396. [Google Scholar] [CrossRef][Green Version]
- Roemer, G.W.; Garcelon, D.K.; Coonan, T.J.; Schwemm, C.A. The use of capture-recapture methods for estimating, monitoring, and conserving island fox populations. In The Fourth California Islands Symposium Update on the Status of Resources; Halvorson, W.L., Maender, G.J., Eds.; Santa Barbara Museum of Natural History: Santa Barbara, CA, USA, 1994; pp. 388–399. [Google Scholar]
- Hofman, C.A.; Rick, T.C.; Hawkins, M.T.; Funk, W.C.; Ralls, K.; Boser, C.L.; Collins, P.W.; Coonan, T.; King, J.L.; Morrison, S.A.; et al. Mitochondrial genomes suggest rapid evolution of dwarf California Channel Islands foxes (Urocyon littoralis). PLoS ONE 2015, 10, e0118240. [Google Scholar] [CrossRef]
- Baber, D.W.; Coblentz, B.E. Density, home range, habitat use, and reproduction in feral pigs on Santa Catalina Island. J. Mammal. 1986, 67, 512–525. [Google Scholar] [CrossRef]
- Schuyler, P.T.; Garcelon, D.K.; Escover, S. Eradication of feral pigs (Sus scrofa) on Santa Catalina Island, California, USA. In Turning the Tide; Veitch, C.R., Clout, M.N., Eds.; IUCN: Gland, Switzerland, 2002; pp. 274–286. [Google Scholar]
- Garcelon, D.K.; Ryan, K.P.; Schuyler, P.T. Application of techniques for feral pig eradication on Santa Catalina Island, California. In Proceedings of the Sixth California Islands Symposium, Ventura, CA, USA, 1–3 December 2003; Garcelon, D.K., Schwemm, C.A., Eds.; Institute for Wildlife Studies: Arcata, CA, USA, 2005; pp. 331–340. [Google Scholar]
- Longhurst, W.M.; Leopold, A.S.; Dasmann, R.F. A Survey of California Deer Herds, Their Ranges, and Management Problems; State of California, Department of Fish and Game, Bureau of Game Conservation: San Diego, CA, USA, 1952; pp. 1–136.
- Lott, D.F.; Minta, S. Home ranges of American bison cows on Santa Catalina Island, California. J. Mammal. 1983, 64, 161–162. [Google Scholar] [CrossRef]
- Sweitzer, R.A.; Constible, J.M.; Van Vuren, D.H.; Schuyler, P.T.; Starkey, F.R. History, habitat use and management of bison on Catalina Island, California. In Proceedings of the Sixth California Islands Symposium, Ventura, CA, USA, 1–3 December 2003; Garcelon, D.K., Schwemm, C.A., Eds.; Institute for Wildlife Studies: Arcata, CA, USA, 2005; pp. 231–248. [Google Scholar]
- Duncan, C.L.; King, J.L.; Kirkpatrick, J.F. Romance without responsibilities: The use of the immunocontraceptive porcine zona pellucida to manage free-ranging bison (Bison bison) on Catalina Island, California, USA. J. Zoo. Wildl. Med. 2014, 44, S123–S131. [Google Scholar] [CrossRef]
- Los Angeles County Santa Catalina Island Local Coastal Plan; Los Angeles County Department of Regional Planning: Los Angeles, CA, USA, 1983. [CrossRef]
- Knapp, D.A. Ecosystem restoration on Santa Catalina Island: A synthesis of resources and threats. In Oak Ecosystem Restoration on Santa Catalina Island, California: Proceedings of An On-Island Workshop, Avalon, CA, USA, 2–4 February 2007; Knapp, D.A., Ed.; Catalina Island Conservancy: Avalon, CA, USA, 2010; pp. 135–216. [Google Scholar]
- Hein, F.J.; de La Rosa, C. Wild Catalina Island: Natural Secrets and Ecological Triumphs; Natural History Press: Charleston, SC, USA, 2013. [Google Scholar]
- O′Malley, P.G. Animal husbandry on the three southernmost Channel Islands: A preliminary overview, 1820–1950. In The Fourth California Islands Symposium Update on The Status of Resources; Halvorson, W.L., Maender, G.J., Eds.; Santa Barbara Museum of Natural History: Santa Barbera, CA, USA, 1994; pp. 157–164. [Google Scholar]
- Coblentz, B.E. Effects of feral goats on the Santa Catalina Island ecosystem. In The California Islands: Proceedings of a Multidisciplinary Symposium; Powers, D.M., Ed.; Santa Barbara Musuem of Natural History: Santa Barbara, CA, USA, 1980; pp. 167–170. [Google Scholar]
- Schuyler, P.T.; Garcelon, D.K.; Escover, S. Control of feral goats (Capra hircus) on Santa Catalina Island, California, USA. In Turning the Tide; Veitch, C.R., Clout, M.N., Eds.; IUCN: Gland, Switzerland; Cambridge, UK, 2002; pp. 412–413. [Google Scholar]
- Curtis, J.F. Report to headquarters district of Southern California. In War of the Rebellion: Compilation of Official Records, Union and Confederate Armies; Series I, Vol. L, 1897; U.S. Government Printing Office: Washington, DC, USA, 1864. [Google Scholar]
- Stapp, P. Status of the introduced mule deer population on Catalina Island, California, based on annual spotlight counts. In Proceedings of the 30th Vertebrate Pest Conference, Reno, NV, USA, 7–10 March 2022; Woods, D.M., Ed.; Paper No. 22. 2022; pp. 1–8. [Google Scholar]
- Stapp, P.; Guttilla, D.A.; Jones, C.A. Population density and habitat use of mule deer (Odocoileus hemionus) on Santa Catalina Island, California. Southwest. Nat. 2006, 51, 572–578. [Google Scholar] [CrossRef]
- Kie, J.G.; Boroski, B.B. Using spotlight counts to estimate mule deer population size and trends. Calif. Fish. Game 1995, 81, 55–70. [Google Scholar]
- Anderson, A.E.; Wallmo, O.C. Odocoileus hemionus. Mamm. Species 1984, 219, 1–9. [Google Scholar] [CrossRef]
- McCullough, D.R. Irruptive behavior in ungulates. In The Science of Overabundance: Deer Ecology and Population Management; Smithsonian Institution Press: Washington, DC, USA, 1997; pp. 69–98. [Google Scholar]
- Holl, S.A.; Bleich, V.C.; Torres, S.G. Population dynamics of bighorn sheep in the San Gabriel Mountains, California, 1967–2002. Wildl. Soc. Bull. 2004, 32, 412–426. [Google Scholar] [CrossRef]
- Brown, D.E.; Cancino, J.; Clark, K.B.; Smith, M. An annotated bibliography of references to historical distributions of pronghorn in southern and Baja California. Bull. South. Calif. Acad. Sci. 2006, 105, 1–16. [Google Scholar] [CrossRef][Green Version]
- Teeter, W.G.; Martinez, D.R.; Kennedy-Richardson, K.O. Cultural landscapes of Catalina Island. In California’s Channel Islands: The Archaeology of Human-Environment Interactions; Jazwa, C.S., Perry, J.E., Eds.; University of Utah Press: Salt Lake City, Utah, 2013; pp. 156–171. [Google Scholar]
- Rowe, M.P.; Farrell, T.M.; May, P.G. Rattle loss in pygmy rattlesnakes (Sistrurus miliarius): Causes, consequences, and implications for rattle function and evolution. In Biology of the Vipers; Schuett, G.W., Höggren, M., Douglas, M.E., Greene, H.W., Eds.; Eagle Mountain Publishing: Eagle Mountain, UT, USA, 2002; pp. 385–404. [Google Scholar]
- Shipman, P.A. The Evolution of Antipredator Behavior in Snakes: Ultimate and Proximate Determinants; Oklahoma State University: Stillwater, OK, USA, 2002; pp. 1–178. [Google Scholar]
- Price, A.H.; LaPointe, J.L. Structure-functional aspects of the scent gland in Lampropeltis getulus splendida. Copeia 1981, 1981, 138–146. [Google Scholar] [CrossRef]
- Martins, M. Defensive tactics in lizards and snakes: The potential contribution of the Neotropical fauna. In Anais do XIV Encontro Anual de Etologia; del Claro, K., Ed.; Universidade Federal de Uberlândia: Uberlândia, Brazil, 1996; pp. 185–199. [Google Scholar]
- Wright, J.; Weldon, P.J. Responses by domestic cats (Felis catus) to snake scent gland secretions. J. Chem. Ecol. 1990, 16, 2947–2953. [Google Scholar] [CrossRef] [PubMed]
- Weldon, P.J.; Fagre, D.B. Responses by canids to scent gland secretions of the western diamondback rattlesnake (Crotalus atrox). J. Chem. Ecol. 1989, 15, 1589–1604. [Google Scholar] [CrossRef] [PubMed]
- Gangloff, E.J.; Bertolatus, D.; Reigel, C.; Gagliardi-Seeley, J.L. Effects of sex, environment, and condition on the musking behavior of sympatric gartersnakes (Thamnophis spp.). J. North Am. Herpetol. 2014, 2014, 40–46. [Google Scholar] [CrossRef]
- Gibbons, J.W.; Dorcas, M.E. Defensive behavior of cottonmouths (Agkistrodon piscivorus) toward humans. Copeia 2002, 1, 195–198. [Google Scholar] [CrossRef]
- Glaudas, X. Do cottonmouths (Agkistrodon piscivorus) habituate to human confrontations? Southwest. Nat. 2004, 3, 129–138. [Google Scholar] [CrossRef]
- Glaudas, X.; Winne, C.T.; Fedewa, L.A. Ontogeny of anti-predator behavioral habituation in cottonmouths (Agkistrodon piscivorus). Ethology 2006, 112, 608–615. [Google Scholar] [CrossRef]
- Aubret, F.; Michniewicz, R.J.; Shine, R. Correlated geographic variation in predation risk and antipredator behaviour within a wide-ranging snake species (Notechis scutatus, Elapidae). Austral Ecol. 2011, 36, 446–452. [Google Scholar] [CrossRef]
- Glaudas, X.; Gibbons, J.W. Do thermal cues influence the defensive strike of cottonmouths (Agkistrodon piscivorus)? Amphib. Reptil. 2005, 26, 264–267. [Google Scholar] [CrossRef]
- Goode, M.; Duvall, D. Body temperature and defensive behaviour of free-ranging prairie rattlesnakes, Crotalus viridis viridis. Anim. Behav. 1989, 38, 360–362. [Google Scholar] [CrossRef]
- Graves, B.M. Defensive behavior of female prairie rattlesnakes (Crotalus viridis) changes after parturition. Copeia 1989, 1989, 791. [Google Scholar] [CrossRef]
- Herbert, S.S. Venom Expenditure by Viperid and Elapid Snakes: Mechanisms, Adaptation, and Application; Loma Linda University: Loma Linda, CA, USA, 2007; pp. 1–128. [Google Scholar]
- Llewelyn, J.; Webb, J.K.; Shine, R. Flexible defense: Context-dependent antipredator responses of two species of Australian elapid snakes. Herpetologica 2010, 66, 1–11. [Google Scholar] [CrossRef]
- Weinstein, S.A.; Kardong, K.V. Properties of Duvernoy′s secretions from opisthoglyphous and aglyphous colubrid snakes. Toxicon 1994, 32, 1161–1185. [Google Scholar] [CrossRef]
- Oliveira, M.E.; Santori, R.T. Predatory behavior of the opossum Didelphis albiventris on the pitviper Bothrops jararaca. Stud. Neotrop. Fauna Environ. 1999, 34, 72–75. [Google Scholar] [CrossRef]
- Heckel, J.O.; Sisson, D.C.; Quist, C.F. Apparent fatal snakebite in three hawks. J. Wildl. Dis. 1994, 30, 616–619. [Google Scholar] [CrossRef]
- Shine, R.; Sun, L.-X.; Fitzgerald, M.; Kearney, M. Accidental altruism in insular pit-vipers (Gloydius shedaoensis, Viperidae). Evol. Ecol. 2002, 16, 541–548. [Google Scholar] [CrossRef]
- Hayes, W.K. Ontogeny of striking, prey-handling and envenomation behavior of prairie rattlesnakes (Crotalus v. viridis). Toxicon 1991, 29, 867–875. [Google Scholar] [CrossRef]
- Rick, T.C.; Hofman, C.A.; Braje, T.J.; Maldonado, J.E.; Sillett, T.S.; Danchisko, K.; Erlandson, J.M. Flightless ducks, giant mice and pygmy mammoths: Late Quaternary extinctions on California′s Channel Islands. World Archaeol. 2012, 44, 3–20. [Google Scholar] [CrossRef]
- Meik, J.M.; Schaack, S.; Ingrasci, M.J.; Lawing, A.M.; Setser, K.; Mociño-Deloya, E.; Flores-Villela, O. Notes on activity, body size variation, and diet in insular speckled rattlesnakes from the western Sea of Cortés, Mexico. Herpetol. Rev. 2012, 43, 556–560. [Google Scholar]
- Alves-Nunes, J.M.; Fellone, A.; Sazima, I.; Vuolo Marques, O.A. Overcoming the phantoms of the past: Influence of predatory stimuli on the antipredator behavior of island pitvipers. PLoS ONE 2023, 18, e0288826. [Google Scholar] [CrossRef] [PubMed]
- Blumstein, D.T. The multipredator hypothesis and the evolutionary persistence of antipredator behavior. Ethology 2006, 112, 209–217. [Google Scholar] [CrossRef]
- Carpenter, C.C.; Gillingham, J.C. Postural responses to kingsnakes by crotaline snakes. Herpetologica 1975, 31, 293–302. [Google Scholar]
- Weldon, P.J.; Burghardt, G.M. The ophiophage defensive response in crotaline snakes: Extension to new taxa. J. Chem. Ecol. 1979, 5, 141–151. [Google Scholar] [CrossRef]
- Minnich, R.A. Grazing, fire, and the management of vegetation on Santa Catalina Island, California. In Dynamics and Management of Mediterranean Type Ecosystems; USDA Forest Service general technical report PSW-58; USDA Forest Service: Berkley, CA, USA, 1982; pp. 444–449. [Google Scholar]
- Van Vuren, D.; Coblentz, B.E. Some ecological effects of feral sheep on Santa Cruz Island, California, USA. Biol. Cons. 1987, 41, 253–268. [Google Scholar] [CrossRef]
- Laughrin, L.; Carroll, M.; Bromfield, A.; Carroll, J. Trends in vegetation changes with removal of feral animal grazing pressures on Santa Catalina Island. In The Fourth California Islands Symposium Update on the Status of Resources; Halvorson, W.L., Maender, G.J., Eds.; Santa Barbara Museum of Natural History: Santa Barbara, CA, USA, 1994; pp. 523–530. [Google Scholar]
- Erlandson, J.M.; Rick, T.C.; Braje, T.J.; Casperson, M.; Culleton, B.; Fulfrost, B.; Garcia, T.; Guthrie, D.A.; Jew, N.; Kennett, D.J.; et al. Paleoindian seafaring, maritime technologies, and coastal foraging on California’s Channel Islands. Science 2011, 331, 1181–1185. [Google Scholar] [CrossRef]
- Bonnet, X.; Aubret, F.; Lourdais, O.; Ladyman, M.; Bradshaw, D.; Maumelat, S. Do “quiet” places make animals placid? Island vs. mainland tiger snakes. Ethology 2005, 111, 573–592. [Google Scholar] [CrossRef]
- Placyk, J.S. The role of innate and environmental influences in shaping antipredator behavior of mainland and insular gartersnakes (Thamnophis sirtalis). J. Ethol. 2011, 30, 101–108. [Google Scholar] [CrossRef]
- Wcislo, W.T. Behavioral environments and evolutionary change. Annu. Rev. Ecol. Syst. 1989, 20, 137–169. [Google Scholar] [CrossRef]
- Cabej, N. Epigenetic Principles of Evolution; Elsevier: London, UK, 2012; pp. 1–804. [Google Scholar]
- Wolf, M.; Weissing, F.J. Animal personalities: Consequences for ecology and evolution. Trends Ecol. Evol. 2012, 27, 452–461. [Google Scholar] [CrossRef]
- Gren, E.C.K. Geographic and Ontogenetic Variation of Venom in the Rattlesnakes Crotalus oreganus oreganus and Crotalus o. helleri; Loma Linda University: Loma Linda, CA, USA, 2015; pp. 1–173. [Google Scholar]
- Grinnell, J.; Dixon, J. Natural history of the ground squirrels of California. Monthly Bull. State Comm. Hortic. 1918, 7, 597–708. [Google Scholar]
- Osgood, W.H. Revision of the Mice of the American Genus Peromyscus; North American Fauna, No.28; U.S. Government Printing Office: Washington, DC, USA, 1909; pp. 1–283.
- Gill, A.E. Evolutionary genetics of California islands Peromyscus. In The California Islands: Proceedings of a Multidisciplinary Symposium; Power, D.M., Ed.; The California Islands: Santa Barbara, CA, USA, 1980; pp. 719–744. [Google Scholar]
- Kardong, K.V. Predatory strike behavior of the rattlesnake, Crotalus viridis oreganus. J. Comp. Psychol. 1986, 100, 304–314. [Google Scholar] [CrossRef]
- Owings, D.H.; Coss, R.G. Hunting California ground squirrels: Constraints and opportunities for Northern Pacific rattlesnakes. In The Biology of Rattlesnakes; Hayes, W.K., Beaman, K.R., Cardwell, M.D., Bush, S.P., Eds.; Loma Linda University Press: Loma Linda, CA, USA, 2008; pp. 155–168. [Google Scholar]
- Chiszar, D.; Smith, H.M. Effects of chemical and thermal cues on striking behavior and post-strike chemosensory searching in rattlesnakes. In The Biology of Rattlesnakes; Hayes, W.K., Beaman, K.R., Cardwell, M.D., Bush, S.P., Eds.; Loma Linda University Press: Loma Linda, CA, USA, 2008; pp. 175–180. [Google Scholar]
- Kardong, K.V.; Smith, T.L. Proximate factors involved in rattlesnake predatory behavior: A review. In Biology of the Vipers; Schuett, G.W., Höggren, M., Douglas, M.E., Greene, H.W., Eds.; Eagle Mountain Publishing: Eagle Mountain, UT, USA, 2002; pp. 253–266. [Google Scholar]
- St. John, M.E.; McGirr, J.A.; Martin, C.H. The behavioral origins of novelty: Did increased aggression lead to scale-eating in pupfishes? Behav. Ecol. 2019, 30, 557–569. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.E.; Fox, G.A.; Putman, B.J.; Hayes, W.K. Social security: Can rattlesnakes reduce acute stress through social buffering? Front. Ethol. 2023, 2, 1181774. [Google Scholar] [CrossRef]
- Losos, J.B. Dynamics of range expansion by three introduced species of Anolis lizards on Bermuda. J. Herpetol. 2006, 30, 204–210. [Google Scholar] [CrossRef][Green Version]
- Jensen, P. The Ethology of Domestic Animals: An Introductory Text, 2nd ed.; Jensen, P., Ed.; CABI Publishing: Wallingford, UK, 2002; pp. 1–264. [Google Scholar]
- Houpt, K.A. Domestic Animal Behavior for Veterinarians and Animal Scientists, 5th ed.; John Wiley & Sons: Ames, IA, USA, 2011; pp. 1–576. [Google Scholar]
- Gottdenker, N.L.; Streicker, D.G.; Faust, C.L.; Carroll, C.R. Anthropogenic land use change and infectious diseases: A review of the evidence. EcoHealth 2014, 11, 619–632. [Google Scholar] [CrossRef] [PubMed]
- Quigley, H.; Herrero, S. Characterization and prevention of attacks on humans. In People and Wildlife, Conflict or Co-Existence? Woodroffe, R., Rabinowitz, A., Thirgood, S., Eds.; Cambridge University Press: Cambridge, UK, 2005; pp. 27–48. [Google Scholar]
- Johansson, T. Beasts on fields. In Human-Wildlife Conflicts in Nature-Culture Borderlands; University of Helsinki Finland: Helsinki, Finland, 2008; pp. 1–227. [Google Scholar]
- Corbit, A.G. The Dynamics of Human and Rattlesnake Conflict in Southern California; Loma Linda University: Loma Linda, CA, USA, 2015. [Google Scholar]
- Sunagar, K.; Undheim, E.A.B.; Scheib, H.; Gren, E.C.K.; Cochran, C.; Person, C.E.; Koludarov, I.; Kelln, W.; Hayes, W.K.; King, G.F.; et al. Intraspecific venom variation in the medically significant Southern Pacific Rattlesnake (Crotalus oreganus helleri): Biodiscovery, clinical and evolutionary implications. J. Proteom. 2014, 99, 68–83. [Google Scholar] [CrossRef] [PubMed]
- Gren, E.C.K.; Kelln, W.; Person, C.; McCabe, J.G.; Kornhauser, R.; Hart, A.J.; Erbas-White, K.; Pompe, L.R.; Hayes, W.K. Geographic variation of venom composition and neurotoxicity in the rattlesnakes Crotalus oreganus and C. helleri: Assessing the potential roles of selection and neutral evolutionary processes in shaping venom variation. In The Biology of Rattlesnakes II; Dreslik, M.J., Hayes, W.K., Beaupre, S.J., Mackessy, S.P., Eds.; ECO Herpetological Publishing and Distribution: Rodeo, NM, USA, 2017; pp. 228–252. [Google Scholar]
- Sahagun, L. Planned Deer Slaughter on Catalina Island Sparks Firestorm of Protest among Residents. Los Angeles Times 2023. Available online: https://www.latimes.com/environment/story/2023-11-05/deer-eradication-proposal-sparks-anger-on-catalina-island (accessed on 5 November 2023).
- Hayes, W.K.; Fox, G.A.; Nelsen, D.R. Venom collection from spiders and snakes: Voluntary and involuntary extractions (“milking”) and venom gland extractions. In Snake and Spider Toxins: Methods in Molecular Biology; Priel, A., Ed.; Humana Press: New York, NY, USA, 2020; Volume 2068, pp. 53–57. [Google Scholar] [CrossRef]
- Hayes, W.K.; Herbert, S.S.; Harrison, J.R.; Wiley, K.L. Spitting versus biting: Differential venom gland contraction regulates venom expenditure in the black-necked spitting cobra, Naja nigricollis nigricollis. J. Herpetol. 2008, 42, 453–460. [Google Scholar] [CrossRef]
- Schoenherr, A.A.; Feldmeth, C.R.; Emerson, M.J. Natural History of the Islands of California; University of California Press: Berkeley, CA, USA, 1999. [Google Scholar]
- Rick, T.C.; Erlandson, J.M.; Vellanoweth, R.L.; Braje, T.J.; Collins, P.W.; Guthrie, D.A.; Stafford, T.W., Jr. Origins and antiquity of the island fox (Urocyon littoralis) on California′s Channel Islands. Quat. Res. 2009, 71, 93–98. [Google Scholar] [CrossRef]
- Cypher, B.L.; Madrid, A.Y.; Van Horn Job, C.L.; Kelly, E.C.; Harrison, S.W.R.; Westall, T.L. Multi-population comparison of resource exploitation by island foxes: Implications for conservation. Global Ecol. Cons. 2014, 2, 255–266. [Google Scholar] [CrossRef]
- Nakagawa, S. A farewell to Bonferroni: The problems of low statistical power and publication bias. Behav. Ecol. 2004, 15, 1044–1045. [Google Scholar] [CrossRef]
- Mertler, C.A.; Vannatta, R.A. Advanced and Multivariate Statistical Methods, 4th ed.; Pyrczak Publishing: Glendale, CA, USA, 2009; pp. 1–362. [Google Scholar]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Pearson Education: New York, NY, USA, 2010; pp. 1–960. [Google Scholar]
- Nakagawa, S.; Cuthill, I.C. Effect size, confidence interval and statistical significance: A practical guide for biologists. Biol. Rev. 2007, 82, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988; pp. 1–567. [Google Scholar]
- Pierce, C.A.; Block, R.A.; Aguinis, H. Cautionary note on reporting eta-squared values from multifactor ANOVA designs. Educ. Psychol. Meas. 2004, 64, 916–924. [Google Scholar] [CrossRef]
- Revell, T.K.; Hayes, W.K. Desert iguanas (Dipsosaurus dorsalis) sleep less when in close proximity to a rattlesnake predator (Crotalus cerastes). J. Herpetol. 2009, 43, 29–37. [Google Scholar] [CrossRef]

| Measure | Mainland (N = 20) | Catalina Island (N = 10) |
|---|---|---|
| Snout–vent length (cm) | 91 ± 4 | 72 ± 3 |
| Rattle before grasping (%) | 50.0 | 50.0 |
| Rattle during grasping (%) | 90.0 | 80.0 |
| Rattle during extraction (%) | 80.0 | 70.0 |
| Cloacal gland discharge (%) | 15.0 | 10.0 |
| Attempted to bite (%) | 15.0 | 70.0 |
| Latency to capture (s) | 7.7 ± 1.0 | 15.8 ± 2.2 |
| Venom expended (mg wet mass) a,b | 0.13 ± 0.02 | 0.27 ± 0.03 |
| Bilateral venom pulses (number) a,c | 2.0 ± 0.3 | 2.5 ± 0.2 |
| Venom per bite (mg) a,b | 0.07 ± 0.01 | 0.15 ± 0.02 |
| Predictors a | B | SE | Wald | p-Value | Odds Ratio (95% C.I.) |
|---|---|---|---|---|---|
| Rattle before grasping (χ23 = 2.55, p = 0.47, Nagelkerke R2 = 0.11, 56.7% predicted correctly) | |||||
| Population | 1.08 | 1.24 | 0.93 | 0.336 | 2.95 (0.33–26.70) |
| Snake length | −0.09 | 0.07 | 2.05 | 0.153 | 0.91 (0.80–1.04) |
| Duration captivity | 0.002 | 0.05 | 0.00 | 0.961 | 1.00 (0.91–1.10) |
| Rattle during grasping (χ23 = 2.51, p = 0.47, Nagelkerke R2 = 0.15, 86.7% predicted correctly) | |||||
| Population | 2.50 | 2.16 | 1.33 | 0.248 | 12.12 (0.18–838.97) |
| Snake length | −0.12 | 0.12 | 0.94 | 0.333 | 0.89 (0.70–1.13) |
| Duration captivity | −0.05 | 0.08 | 0.42 | 0.518 | 0.95 (0.82–1.11) |
| Rattle during extraction (χ23 = 1.11, p = 0.78, Nagelkerke R2 = 0.06, 76.7% predicted correctly) | |||||
| Population | 0.30 | 1.18 | 0.06 | 0.800 | 1.35 (0.13–13.51) |
| Snake length | 0.02 | 0.07 | 0.10 | 0.754 | 1.02 (0.89–1.17) |
| Duration captivity | −0.05 | 0.06 | 0.70 | 0.402 | 0.95 (0.85–1.07) |
| Cloacal gland discharge (χ23 = 1.33, p = 0.72, Nagelkerke R2 = 0.08, 86.7% predicted correctly) | |||||
| Population | −0.60 | 1.74 | 0.12 | 0.729 | 0.55 (0.18–16.65) |
| Snake length | 0.08 | 010 | 0.79 | 0.374 | 1.09 (0.90–1.31) |
| Duration captivity | −0.05 | 0.07 | 0.56 | 0.456 | 0.95 (0.83–1.09) |
| Attempted to bite (χ23 = 10.55, p = 0.014, Nagelkerke R2 = 0.41, 80.0% predicted correctly) | |||||
| Population | −4.06 | 1.81 | 5.06 | 0.024 | 0.02 (0.00–0.59) |
| Snake length | 0.11 | 0.10 | 1.21 | 0.272 | 1.12 (0.92–1.35) |
| Duration captivity | −0.01 | 0.06 | 0.04 | 0.837 | 0.99 (0.88–1.11) |
| Variables a | F-Value b | p-Value | Partial η2 |
|---|---|---|---|
| Latency to capture (s) | |||
| Population | 16.62 | <0.001 | 0.39 |
| Snake length | 0.88 | 0.358 | 0.03 |
| Duration captivity | 0.33 | 0.573 | 0.01 |
| Venom expended (mg) | |||
| Population | 11.64 | 0.002 | 0.33 |
| Snake length | 13.62 | 0.001 | 0.36 |
| Duration captivity | 0.54 | 0.472 | 0.02 |
| Bilateral venom pulses (number) | |||
| Population | 1.99 | 0.172 | 0.08 |
| Snake length | 0.04 | 0.843 | 0.00 |
| Duration captivity | 0.00 | 0.994 | 0.00 |
| Venom per bite (mg) | |||
| Population | 7.31 | 0.012 | 0.23 |
| Snake length | 12.07 | 0.002 | 0.34 |
| Duration captivity | 0.01 | 0.922 | 0.00 |
| Species | Introduction | Removal | Highest N | Highest Density | Distribution | Source |
|---|---|---|---|---|---|---|
| Cat (Felis catus) | 1800s | extant | 600–750 | 3.1–3.9 | island-wide | [69] |
| Island fox (Urocyon littoralis catalinae) | 7100–9200 years ago | extant | 1115–1342 | 5.7–6.9 | island-wide | [70,71,72] |
| Pig (Sus scrofa) | early 1930s | 2004 | 1260–5000 | 6.5–25.8 | island-wide | [73,74,75,76] |
| American bison (Bison bison) | 1924 | extant | 400–524 | 2.1–2.7 | currently east end | [76,77,78,79] |
| Black buck (Antilope cervicapra) | 1967–1973 (uncertain) | 2014 | <25 | ~0.1 | east end | [80,81,82] |
| Cattle (Bos taurus) | 1800s | 1950s | >5225 | 26.9 | island-wide | [76,78,83] |
| Goat (Capra hircus) | early 1800s | 2003 | 30,000–50,000 | 154.6–257.7 | island-wide | [76,84,85,86] |
| Horse (Equus caballus) | 1800s | unknown | 240 | 1.2 | unknown | [76] |
| Mule deer (Odocoileus hemionus) | 1928 | extant | 2000–3285 | 5.4–16.9 | island-wide | [76,87,88] |
| Sheep (Ovis aries) | 1800s | 1920s | 22,000 | 113.4 | island-wide | [76,78,86] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayes, W.K.; Person, C.E.; Fox, G.A.; King, J.L.; Briggs, E.; Gren, E.C.K. Paradoxical Exception to Island Tameness: Increased Defensiveness in an Insular Population of Rattlesnakes. Toxins 2024, 16, 157. https://doi.org/10.3390/toxins16030157
Hayes WK, Person CE, Fox GA, King JL, Briggs E, Gren ECK. Paradoxical Exception to Island Tameness: Increased Defensiveness in an Insular Population of Rattlesnakes. Toxins. 2024; 16(3):157. https://doi.org/10.3390/toxins16030157
Chicago/Turabian StyleHayes, William K., Carl E. Person, Gerad A. Fox, Julie L. King, Erick Briggs, and Eric C. K. Gren. 2024. "Paradoxical Exception to Island Tameness: Increased Defensiveness in an Insular Population of Rattlesnakes" Toxins 16, no. 3: 157. https://doi.org/10.3390/toxins16030157
APA StyleHayes, W. K., Person, C. E., Fox, G. A., King, J. L., Briggs, E., & Gren, E. C. K. (2024). Paradoxical Exception to Island Tameness: Increased Defensiveness in an Insular Population of Rattlesnakes. Toxins, 16(3), 157. https://doi.org/10.3390/toxins16030157






