Evaluation of Tropane Alkaloids in Teas and Herbal Infusions: Effect of Brewing Time and Temperature on Atropine and Scopolamine Content
Abstract
1. Introduction
2. Results and Discussion
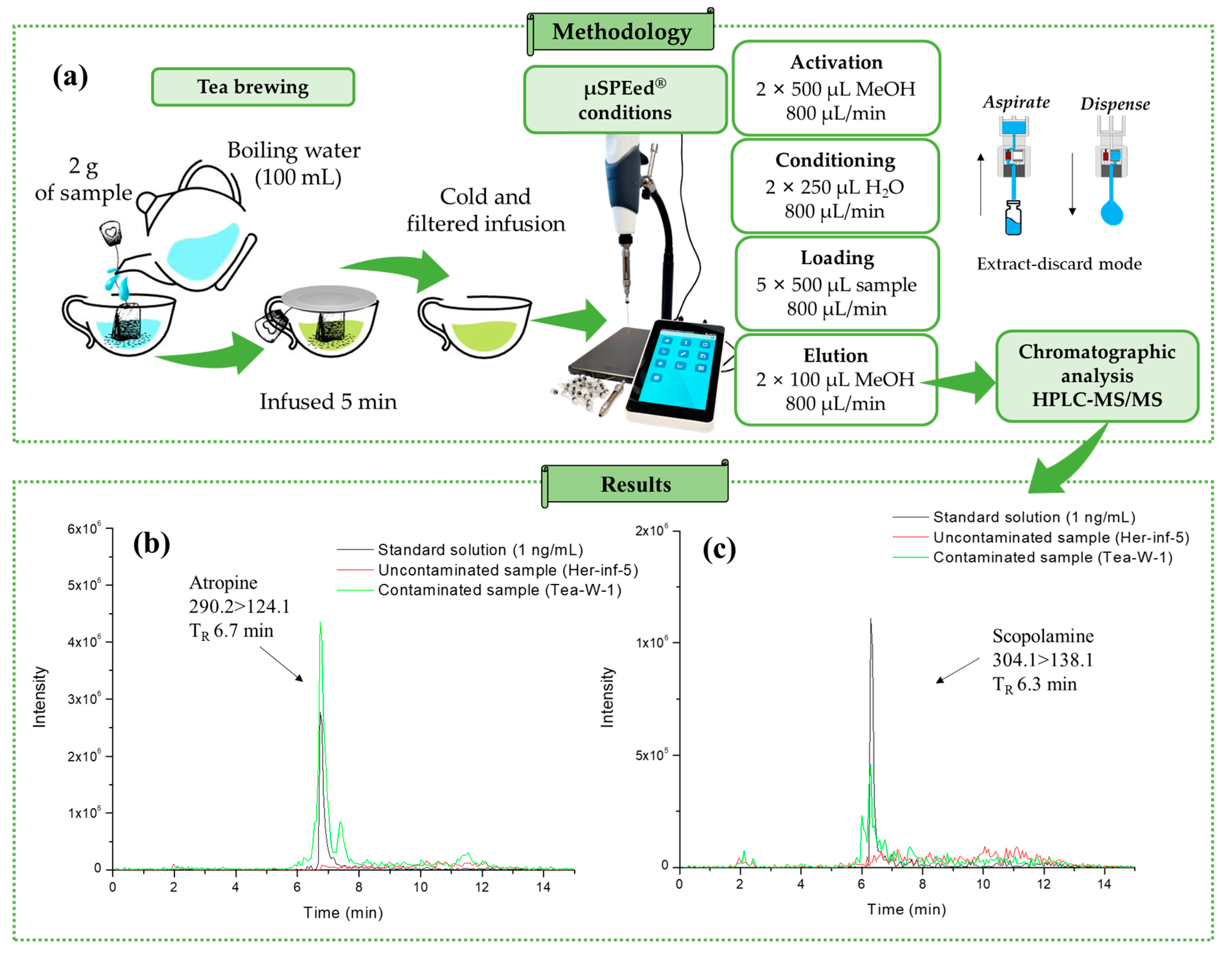
2.1. Methodology Performance
2.2. Analysis and Quantification of Atropine and Scopolamine in Different Types of Infusions
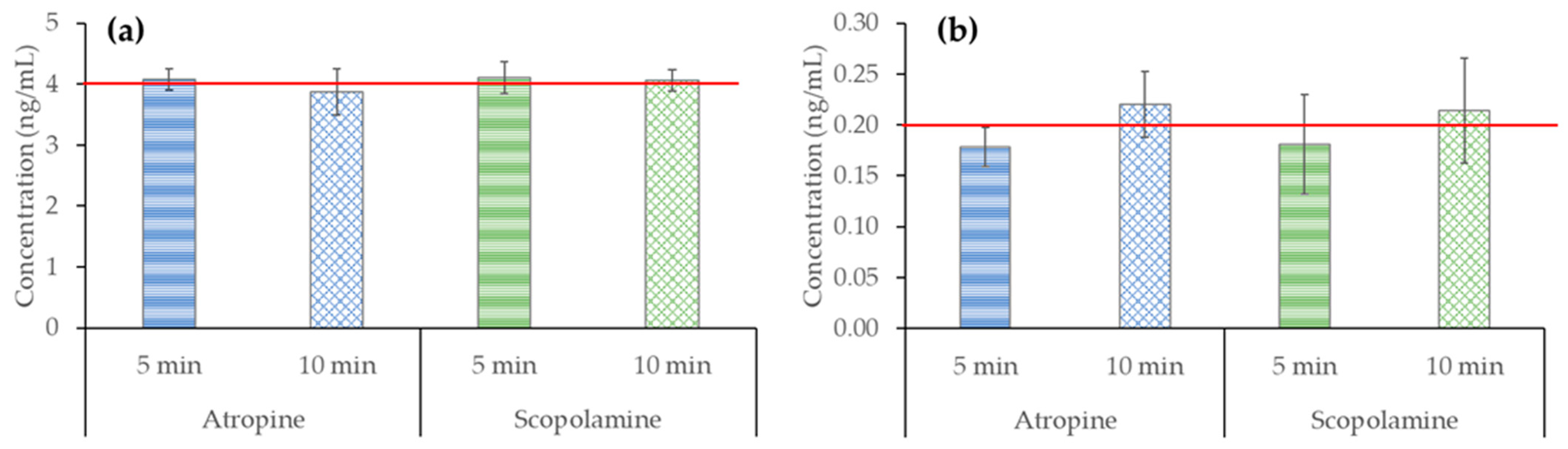
2.3. Evaluation of the Effect of Heating/Brewing Conditions on Atropine and Scopolamine
2.3.1. In Standard Solutions
2.3.2. In Contaminated Green, White, and Black Tea Samples
3. Conclusions
4. Materials and Methods
4.1. Chemicals, Reagents, and Standard Solutions
4.2. Sample and Infusion Preparations
4.3. Studies to Evaluate the Effect of Heating/Brewing Conditions on Atropine and Scopolamine
4.3.1. In Standard Solutions
4.3.2. In Contaminated Green, White, and Black Tea Samples
4.4. µSPEed® Extraction Procedure
4.5. HPLC–MS/MS Conditions
4.6. Quantification of Atropine and Scopolamine
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mulder, P.P.J.; de Nijs, M.; Castellari, M.; Hortos, M.; MacDonald, S.; Crews, C.; Hajslova, J.; Stranska, M. Occurrence of tropane alkaloids in food. EFSA Support. Publ. 2016, 13, 1140E. [Google Scholar] [CrossRef]
- Adamse, P.; van Egmond, H.P.; Noordam, M.Y.; Mulder, P.P.J.; De Nijs, M. Tropane alkaloids in food: Poisoning incidents. Qual. Assur. Saf. Crop. Foods 2014, 6, 15–24. [Google Scholar] [CrossRef]
- De Nijs, M.; Crews, C.; Dorgelo, F.; MacDonald, S.; Mulder, P.P.J. Emerging Issues on Tropane Alkaloid Contamination of Food in Europe. Toxins 2023, 15, 98. [Google Scholar] [CrossRef]
- Rivera-Pérez, A.; Romero-González, R.; Garrido Frenich, A. Determination and Occurrence of Alkenylbenzenes, Pyrrolizidine and Tropane Alkaloids in Spices, Herbs, Teas, and Other Plant-derived Food Products Using Chromatographic Methods: Review from 2010–2020. Food Rev. Int. 2021, 39, 1110–1136. [Google Scholar] [CrossRef]
- Balíková, M. Collective poisoning with hallucinogenous herbal tea. Forensic Sci. Int. 2002, 128, 50–52. [Google Scholar] [CrossRef]
- European Commission Scientific Opinion on Tropane alkaloids in food and feed. EFSA J. 2013, 11, 1–113. [CrossRef]
- Kohnen-Johannsen, K.L.; Kayser, O. Tropane alkaloids: Chemistry, pharmacology, biosynthesis and production. Molecules 2019, 24, 796. [Google Scholar] [CrossRef] [PubMed]
- González-Gómez, L.; Morante-Zarcero, S.; Pérez-Quintanilla, D.; Sierra, I. Occurrence and Chemistry of Tropane Alkaloids in Foods, with a Focus on Sample Analysis Methods: A Review on Recent Trends and Technological Advances. Foods 2022, 11, 407. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.Y.K. Worldwide occurrence and investigations of contamination of herbal medicines by Tropane Alkaloids. Toxins 2017, 9, 284. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, C.; Dernovics, M.; Moreno-Gonzalez, D.; Bouten, K.; Garcia-Reyes, J.F.; Stroka, J. Occurrence and Determination of Tropane Alkaloids in Food and Feed; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2020; ISBN 9780470027318. [Google Scholar]
- Baslé, Q.; Mujahid, C.; Bessaire, T. Application of a streamlined LC-MS/MS methodology for the determination of atropine and scopolamine in cereals from Asian and African countries. Food Addit. Contam.-Part A Chem. Anal. Control. Expo. Risk Assess. 2020, 37, 1744–1754. [Google Scholar] [CrossRef]
- Marín-Sáez, J.; Romero-González, R.; Garrido Frenich, A. Reliable determination of tropane alkaloids in cereal based baby foods coupling on-line spe to mass spectrometry avoiding chromatographic step. Food Chem. 2019, 275, 746–753. [Google Scholar] [CrossRef] [PubMed]
- Marín-Sáez, J.; Romero-González, R.; Garrido Frenich, A. Multi-analysis determination of tropane alkaloids in cereals and solanaceaes seeds by liquid chromatography coupled to single stage Exactive-Orbitrap. J. Chromatogr. A 2017, 1518, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Vuković, G.; Stojanović, T.; Konstantinović, B.; Bursić, V.; Puvača, N.; Popov, M.; Samardžić, N.; Petrović, A.; Marinković, D.; Roljević Nikolić, S.; et al. Atropine and Scopolamine in Maize Products from the Retail Stores in the Republic of Serbia. Toxins 2022, 14, 621. [Google Scholar] [CrossRef]
- Cirlini, M.; Demuth, T.M.; Biancardi, A.; Rychlik, M.; Dall’Asta, C.; Bruni, R. Are tropane alkaloids present in organic foods? Detection of scopolamine and atropine in organic buckwheat (Fagopyron esculentum L.) products by UHPLC–MS/MS. Food Chem. 2018, 239, 141–147. [Google Scholar] [CrossRef]
- Shimshoni, J.A.; Duebecke, A.; Mulder, P.P.J.; Cuneah, O.; Barel, S. Pyrrolizidine and tropane alkaloids in teas and the herbal teas peppermint, rooibos and chamomile in the Israeli market. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2015, 32, 2058–2067. [Google Scholar] [CrossRef] [PubMed]
- Romera-Torres, A.; Romero-González, R.; Martínez Vidal, J.L.; Garrido Frenich, A. Simultaneous analysis of tropane alkaloids in teas and herbal teas by liquid chromatography coupled to high-resolution mass spectrometry (Orbitrap). J. Sep. Sci. 2018, 41, 1938–1946. [Google Scholar] [CrossRef]
- León, N.; Miralles, P.; Yusà, V.; Coscollà, C. A green analytical method for the simultaneous determination of 30 tropane and pyrrolizidine alkaloids and their N-oxides in teas and herbs for infusions by LC-Q-Orbitrap HRMS. J. Chromatogr. A 2022, 1666, 462835. [Google Scholar] [CrossRef] [PubMed]
- Mateus, A.R.S.; Crisafulli, C.; Vilhena, M.; Barros, S.C.; Pena, A.; Sanches Silva, A. The Bright and Dark Sides of Herbal Infusions: Assessment of Antioxidant Capacity and Determination of Tropane Alkaloids. Toxins 2023, 15, 245. [Google Scholar] [CrossRef]
- Cirlini, M.; Cappucci, V.; Galaverna, G.; Dall’Asta, C.; Bruni, R. A sensitive UHPLC-ESI-MS/MS method for the determination of tropane alkaloids in herbal teas and extracts. Food Control 2019, 105, 285–291. [Google Scholar] [CrossRef]
- González-Gómez, L.; Gañán, J.; Morante-Zarcero, S.; Pérez-Quintanilla, D.; Sierra, I. Atropine and scopolamine occurrence in spices and fennel infusions. Food Control 2022, 307, 109555. [Google Scholar] [CrossRef]
- Romera-Torres, A.; Romero-González, R.; Martínez Vidal, J.L.; Garrido Frenich, A. Comprehensive tropane alkaloids analysis and retrospective screening of contaminants in honey samples using liquid chromatography-high resolution mass spectrometry (Orbitrap). Food Res. Int. 2020, 133, 109130. [Google Scholar] [CrossRef]
- Martinello, M.; Borin, A.; Stella, R.; Bovo, D.; Biancotto, G.; Gallina, A.; Mutinelli, F. Development and validation of a QuEChERS method coupled to liquid chromatography and high resolution mass spectrometry to determine pyrrolizidine and tropane alkaloids in honey. Food Chem. 2017, 234, 295–302. [Google Scholar] [CrossRef]
- Castilla-Fernández, D.; Moreno-González, D.; García-Reyes, J.F.; Ballesteros, E.; Molina-Díaz, A. Determination of atropine and scopolamine in spinach-based products contaminated with genus Datura by UHPLC–MS/MS. Food Chem. 2021, 347, 129020. [Google Scholar] [CrossRef] [PubMed]
- González-Gómez, L.; Morante-Zarcero, S.; Pereira, J.A.M.; Câmara, J.S.; Sierra, I. Improved Analytical Approach for Determination of Tropane Alkaloids in Leafy Vegetables Based on µ -QuEChERS Combined with HPLC-MS/MS. Toxins 2022, 14, 650. [Google Scholar] [CrossRef] [PubMed]
- RASFF Portal. Food and Feed Safety Alerts. Available online: https://webgate.ec.europa.eu/rasff-window/portal/? (accessed on 30 January 2022).
- Selmar, D.; Wittke, C.; Beck-von Wolffersdorff, I.; Klier, B.; Lewerenz, L.; Kleinwächter, M.; Nowak, M. Transfer of pyrrolizidine alkaloids between living plants: A disregarded source of contaminations. Environ. Pollut. 2019, 248, 456–461. [Google Scholar] [CrossRef]
- Letsyo, E.; Adams, Z.S.; Dzikunoo, J.; Asante-Donyinah, D. Uptake and accumulation of pyrrolizidine alkaloids in the tissues of maize (Zea mays L.) plants from the soil of a 4-year-old Chromolaena odorata dominated fallow farmland. Chemosphere 2021, 270, 128669. [Google Scholar] [CrossRef]
- European Union. Commission regulation (EU) 2021/1408 of 27 August 2021 amending Regulation (EC) No 1881/2006 as regards maximum levels of tropane alkaloids in certain foodstuffs. Off. J. Eur. Union 2021, 304, 1–4. [Google Scholar]
- González-Gómez, L.; Pereira, J.A.M.; Morante-Zarcero, S.; Câmara, J.S.; Sierra, I. Green extraction approach based on μSPEed® followed by HPLC-MS/MS for the determination of atropine and scopolamine in tea and herbal tea infusions. Food Chem. 2022, 394, 133512. [Google Scholar] [CrossRef]
- Martinello, M.; Manzinello, C.; Gallina, A.; Mutinelli, F. In-house validation and application of UHPLC-MS/MS method for the quantification of pyrrolizidine and tropane alkaloids in commercial honey bee-collected pollen, teas and herbal infusions purchased on Italian market in 2019–2020 referring to recent Europe. Int. J. Food Sci. Technol. 2022, 57, 7505–7516. [Google Scholar] [CrossRef]
- Marín-Sáez, J.; Romero-González, R.; Garrido Frenich, A. Effect of tea making and boiling processes on the degradation of tropane alkaloids in tea and pasta samples contaminated with Solanaceae seeds and coca leaf. Food Chem. 2019, 287, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Casado, N.; Casado-Hidalgo, G.; González-Gómez, L.; Morante-Zarcero, S.; Sierra, I. Insight into the Impact of Food Processing and Culinary Preparations on the Stability and Content of Plant Alkaloids Considered as Natural Food Contaminants. Appl. Sci. 2023, 13, 1704. [Google Scholar] [CrossRef]
- European Union. Analytical Quality Control and Method Validation Procedures for Pesticide Residues Analysis in Food and Feed; European Comission Document No SANTE 11312/2021; European Union: Brussels, Belgium, 2021; pp. 1–52. [Google Scholar]
- Kumar, K.R.; Dashora, K.; Kumar, S.; Dharmaraja, S.; Sanyal, S.; Aditya, K.; Kumar, R. A review of drying technology in tea sector of industrial, non-conventional and renewable energy based drying systems. Appl. Therm. Eng. 2023, 224, 120118. [Google Scholar] [CrossRef]
- Hilal, Y.; Engelhardt, U. Characterisation of white tea—Comparison to green and black tea. J. Fur Verbrauch. Und Leb. 2007, 2, 414–421. [Google Scholar] [CrossRef]
- Liu, Z.; Vincken, J.P.; de Bruijn, W.J.C. Tea phenolics as prebiotics. Trends Food Sci. Technol. 2022, 127, 156–168. [Google Scholar] [CrossRef]
- Jiao, W.; Shen, T.; Wang, L.; Zhu, L.; Li, Q.X.; Wang, C.; Chen, H.; Hua, R.; Wu, X. Source and Route of Pyrrolizidine Alkaloid Contamination in Tea Samples. J. Vis. Exp. 2022, 187, e64375. [Google Scholar] [CrossRef]
- European Union. Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the provision of food information to consumers, amending Regulations (EC) No 1924/2006 and (EC) No 1925/2006 of the European Parliament and of the Council, an. Off. J. Eur. Union 2011, 020, 18–63. [Google Scholar]
- Fernández-Pintor, B.; Casado, N.; Morante-zarcero, S.; Sierra, I. Evaluation of the thermal stability and transfer rate of pyrrolizidine alkaloids during the of pyrrolizidine alkaloids during the brewing of herbal infusions contaminated with Echium vulgare and Senecio vulgaris weeds. Food Control 2023, in press. [Google Scholar]
- ISO. ISO 3103; Tea—Preparation of Liquor for Use in Sensory Tests. ISO: Geneva, Switzerland, 2019.


| Calibration (Sample) | Linearity for At 1.25–313 ng/mL (R2) a | Linearity for Sc 1.25–313 ng/mL (R2) a | MDL b (ng/mL) | MQL c (ng/mL) | ME d (%) | |||
|---|---|---|---|---|---|---|---|---|
| At | Sc | At | Sc | At | Sc | |||
| Matrix-matched calibration (Tea-B-3) | 2.2 × 106x + 4.1 × 106 (0.995) | 8.6 × 105x − 1.1 × 106 (0.996) | 0.04 | 0.05 | 0.14 | 0.18 | 10 | 23 |
| Matrix-matched calibration (Her-Inf-4) | 1.7 × 106x + 2.9 × 106 (0.998) | 4.7 × 105x + 2.0 × 106 (0.996) | 0.02 | 0.05 | 0.06 | 0.18 | −15 | −33 |
| Infusion Code | (±)-Atropine-D3 (Recovery % ± SD) | (−)-Scopolamine-D3 (Recovery % ± SD) |
|---|---|---|
| Tea-W-1 | 101 ± 3 | 98 ± 8 |
| Tea-W-2 | 103 ± 4 | 101 ± 5 |
| Tea-W-3 | 89 ± 9 | 81 ± 9 |
| Tea-G-1 | 102 ± 1 | 95 ± 2 |
| Tea-G-2 | 102 ± 1 | 105 ± 3 |
| Tea-G-3 | 97 ± 6 | 103 ± 1 |
| Tea-G-4 | 106 ± 5 | 102 ± 2 |
| Tea-G-5 | 97 ± 1 | 102 ± 2 |
| Tea-B-1 | 97 ± 1 | 100 ± 1 |
| Tea-B-2 | 104 ± 1 | 106 ± 4 |
| Tea-B-3 | 107 ± 1 | 103 ± 5 |
| Tea-B-4 | 101 ± 3 | 99 ± 7 |
| Tea-B-5 | 96 ± 5 | 89 ± 3 |
| Tea-B-6 | 102 ± 3 | 94 ± 9 |
| Tea-B-7 | 99 ± 1 | 90 ± 1 |
| Tea-B-8 | 105 ± 4 | 92 ± 2 |
| Her-Inf-1 | 103 ± 8 | 73 ± 3 |
| Her-Inf-2 | 102 ± 5 | 99 ± 3 |
| Her-Inf-3 | 97 ± 4 | 104 ± 2 |
| Her-Inf-4 | 98 ± 4 | 86 ± 9 |
| Her-Inf-5 | 106 ± 1 | 97 ± 4 |
| Her-Inf-6 | 104 ± 9 | 103 ± 11 |
| Herb-Inf-7 | 108 ± 8 | 114 ± 2 |
| Herb-Inf-8 | 106 ± 5 | 102 ± 4 |
| Herb-Inf-9 | 103 ± 1 | 73 ± 7 |
| Herb-Inf-10 | 101 ± 1 | 87 ± 1 |
| Herb-Inf-11 | 102 ± 1 | 98 ± 1 |
| Herb-Inf-12 | 103 ± 6 | 106 ± 4 |
| Herb-Inf-13 | 98 ± 1 | 85 ± 3 |
| Herb-Inf-14 | 105 ± 1 | 92 ± 6 |
| Herb-Inf-15 | 109 ± 9 | 96 ± 1 |
| Herb-Inf-16 | 92 ± 8 | 93 ± 8 |
| Herb-Inf-17 | 93 ± 3 | 96 ± 3 |
| Infusion Code | Sample Description (Code) | Atropine (ng/mL ± SD) | Scopolamine (ng/mL ± SD) |
|---|---|---|---|
| Tea-W-1 a | White tea (W-1) | 2.57 ± 0.28 | 0.33 ± 0.02 |
| Tea-W-2 a | White tea (W-2) | 1.24 ± 0.14 | 0.33 ± 0.04 |
| Tea-W-3 a | White tea (W-3) | 2.86 ± 0.12 | ND |
| Tea-G-1 a | Green tea (G-1) | 3.65 ± 0.10 | 0.34 ± 0.02 |
| Tea-G-2 a | Green tea (G-2) | 4.65 ± 0.57 | ND |
| Tea-G-3 a | Green tea (G-3) | 4.94 ± 0.74 | ≤MQL |
| Tea-G-4 a | Green tea (G-4) | 4.15 ± 0.24 | ND |
| Tea-G-5 a | Kukicha green tea (G-5) | ≤MQL | ND |
| Tea-B-1 a | Black tea (B-1) | 1.81 ± 0.14 | ND |
| Tea-B-2 a | Black tea (B-2) | 1.65 ± 0.07 | ND |
| Tea-B-3 a | Black tea (B-3) | 1.30 ± 0.16 | ND |
| Tea-B-4 a | Black tea with bergamot (B-4) | 1.95 ± 0.08 | ND |
| Tea-B-5 a | Black tea (B-5) | 2.00 ± 0.16 | ND |
| Tea-B-6 a | Black tea (B-6) | 0.44 ± 0.05 | ND |
| Tea-B-7 a | Pakistani black tea (B-7) | 1.74 ± 0.08 | ND |
| Tea-B-8 a | Black tea (B-8) | 0.75 ± 0.03 | ND |
| Her-Inf-1 b | Pink lapacho bark tea (Her-1) | ND | ND |
| Her-Inf-2 b | Lemon grass tea (Her-2) | ND | ND |
| Her-Inf-3 b | Rosemary (Her-3) | ND | ND |
| Her-Inf-4 b | Valerian (Her-4) | ≤MQL | ND |
| Her-Inf-5 b | Echinacea (Her-5) | ND | ND |
| Her-Inf-6 b | Star anise (Her-6) | ND | ND |
| Herb-Inf-7 b | Flavored yerba mate (Her-7) | 0.14 ± 0.01 | ND |
| Herb-Inf-8 b | Flavored yerba mate (Her-8) | ≤MQL | ND |
| Herb-Inf-9 b | Mixed herbal tea (Her-9) | <MQL | ND |
| Herb-Inf-10 b | Mixed herbal tea (Her-10) | ND | ND |
| Herb-Inf-11 b | Mixed herbal tea (Her-11) | <MQL | ND |
| Herb-Inf-12 b | Mixed herbal tea (Her-12) | ND | ND |
| Herb-Inf-13 b | Mixed herbal tea (Her-13) | ND | ND |
| Herb-Inf-14 b | Mixed herbal tea (Her-14) | ND | ND |
| Herb-Inf-15 b | Mixed herbal tea (Her-15) | ND | ND |
| Herb-Inf-16 b | Mixed herbal tea (Her-16) | ND | ND |
| Herb-Inf-17 b | Mixed herbal tea (Her-17) | ND | ND |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Gómez, L.; Morante-Zarcero, S.; Pereira, J.A.M.; Câmara, J.S.; Sierra, I. Evaluation of Tropane Alkaloids in Teas and Herbal Infusions: Effect of Brewing Time and Temperature on Atropine and Scopolamine Content. Toxins 2023, 15, 362. https://doi.org/10.3390/toxins15060362
González-Gómez L, Morante-Zarcero S, Pereira JAM, Câmara JS, Sierra I. Evaluation of Tropane Alkaloids in Teas and Herbal Infusions: Effect of Brewing Time and Temperature on Atropine and Scopolamine Content. Toxins. 2023; 15(6):362. https://doi.org/10.3390/toxins15060362
Chicago/Turabian StyleGonzález-Gómez, Lorena, Sonia Morante-Zarcero, Jorge A. M. Pereira, José S. Câmara, and Isabel Sierra. 2023. "Evaluation of Tropane Alkaloids in Teas and Herbal Infusions: Effect of Brewing Time and Temperature on Atropine and Scopolamine Content" Toxins 15, no. 6: 362. https://doi.org/10.3390/toxins15060362
APA StyleGonzález-Gómez, L., Morante-Zarcero, S., Pereira, J. A. M., Câmara, J. S., & Sierra, I. (2023). Evaluation of Tropane Alkaloids in Teas and Herbal Infusions: Effect of Brewing Time and Temperature on Atropine and Scopolamine Content. Toxins, 15(6), 362. https://doi.org/10.3390/toxins15060362









