Cyanobacterial Harmful Algal Bloom Toxin Microcystin and Increased Vibrio Occurrence as Climate-Change-Induced Biological Co-Stressors: Exposure and Disease Outcomes via Their Interaction with Gut–Liver–Brain Axis
Abstract
1. Cyanobacteria and Cyano-HABs
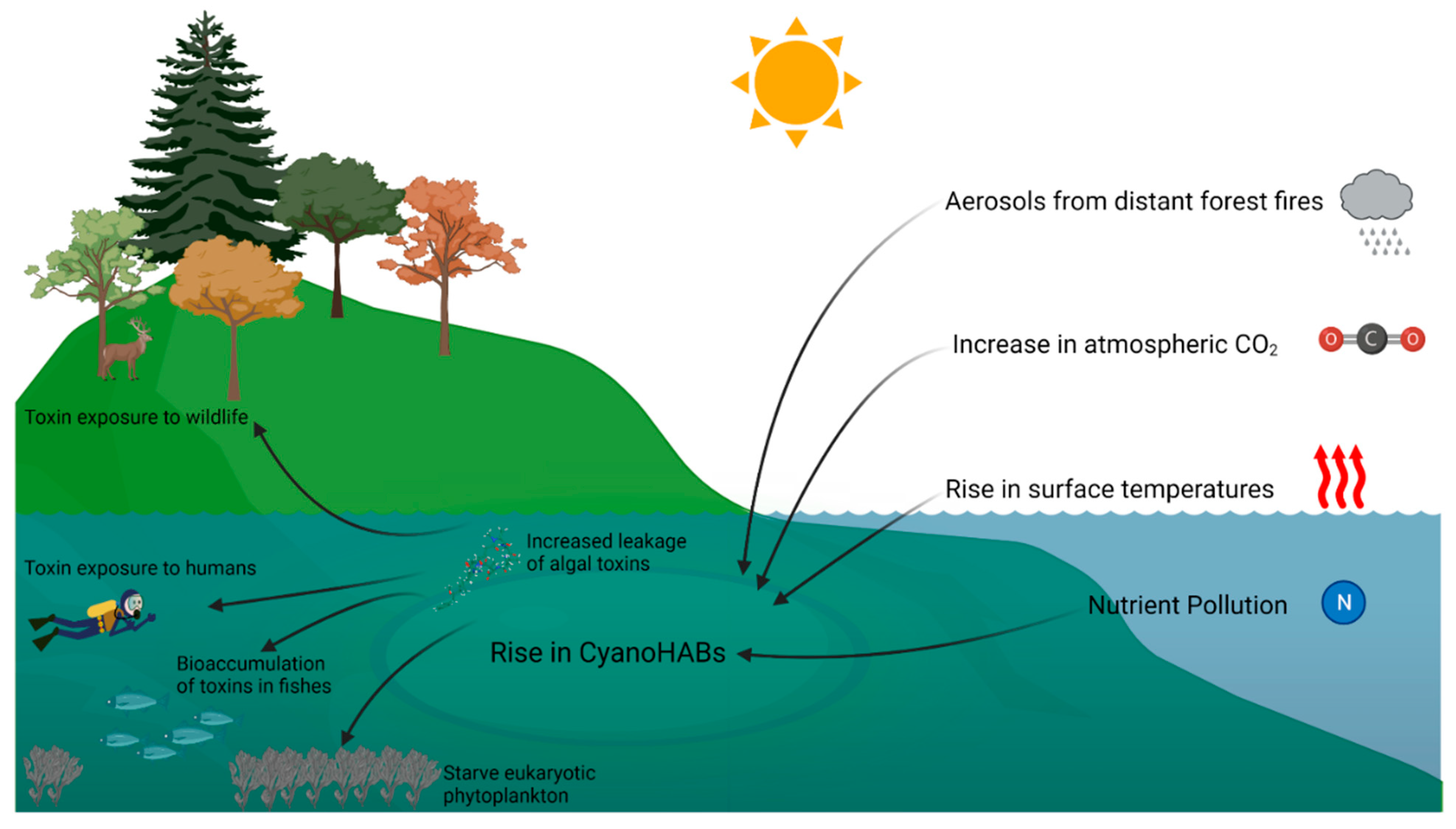
2. Climate Change and Cyanobacterial HABs
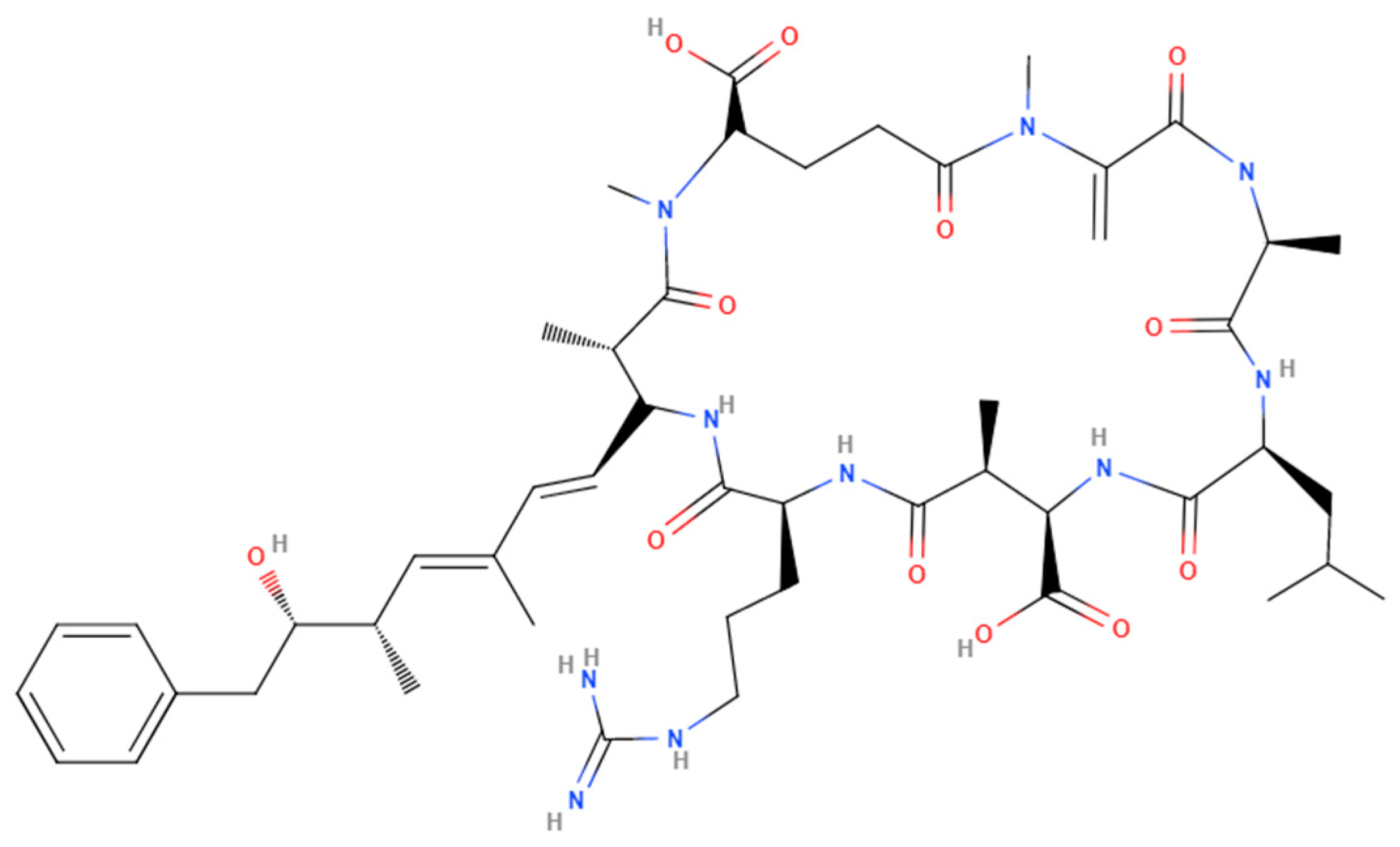
3. Increase in Cyanotoxin Exposure: Microcystin
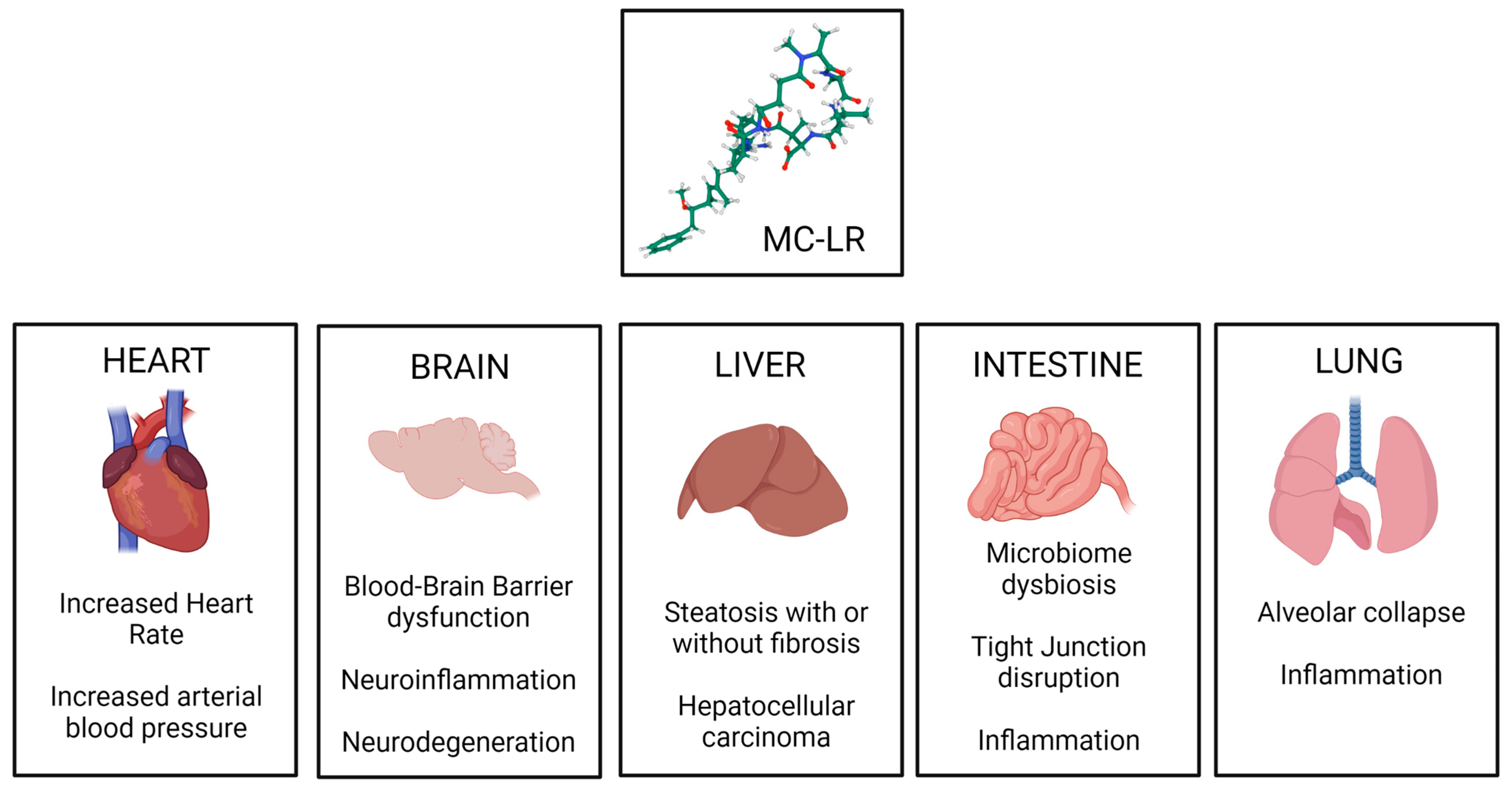
4. General Toxicity of Microcystins
5. Exposure to Microcystins Has a Potential to Complicate Pathology in Underlying Diseases and Parallel Conditions That Are Affected by Climate Change
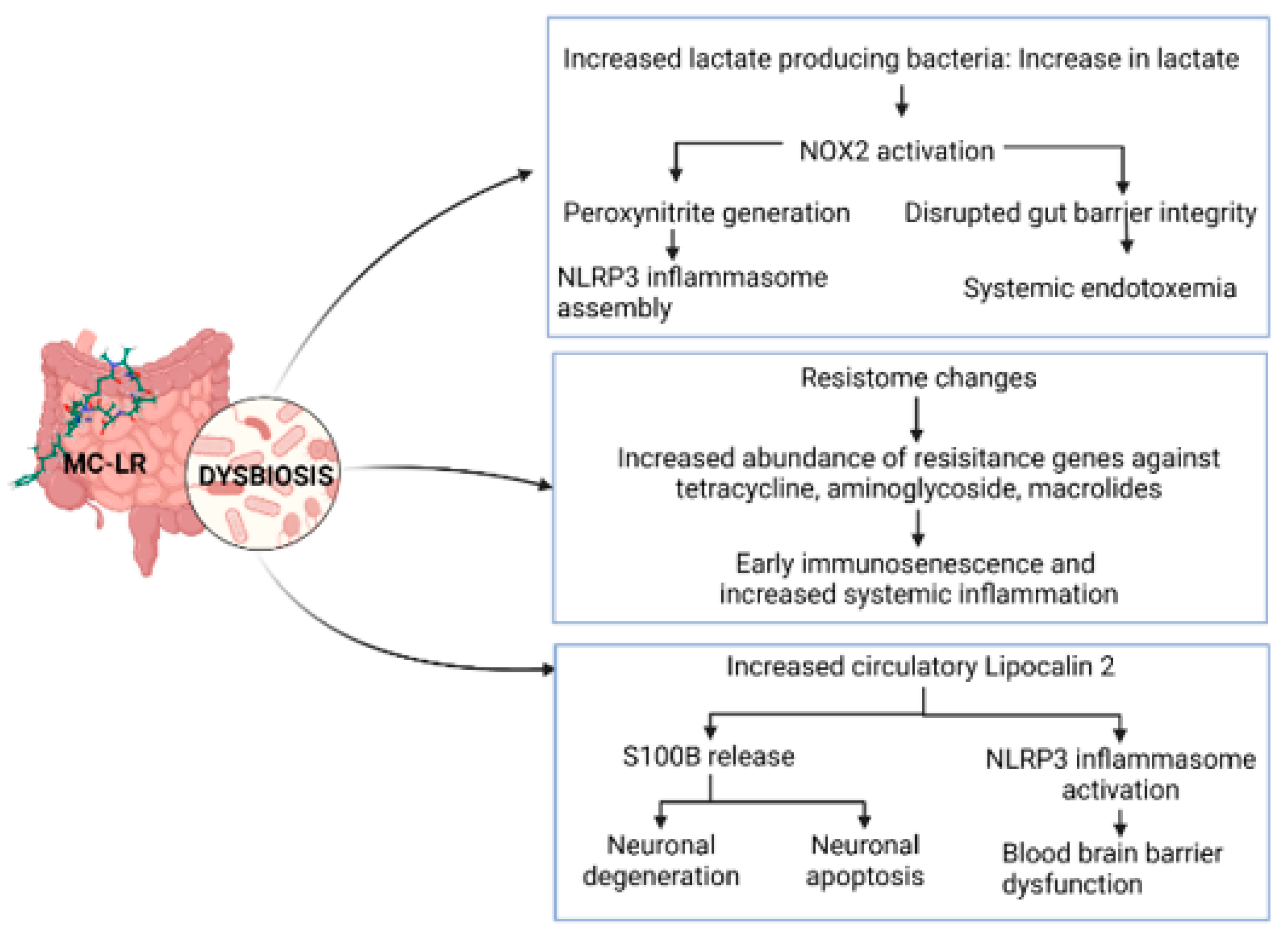
5.1. Antibiotic Resistance
5.2. Vibriosis
5.3. Heat Stress
5.4. Cardiovascular Diseases
5.5. Type II Diabetes
5.6. Non-Alcoholic Fatty Liver Disease
6. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Reynolds, C.S. The Ecology of Phytoplankton; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Demoulin, C.F.; Lara, Y.J.; Cornet, L.; Francois, C.; Baurain, D.; Wilmotte, A.; Javaux, E.J. Cyanobacteria evolution: Insight from the fossil record. Free. Radic. Biol. Med. 2019, 140, 206–223. [Google Scholar] [CrossRef] [PubMed]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M.; Fachon, E.; Pickart, R.S.; Lin, P.; Fischer, A.D.; Richlen, M.L.; Uva, V.; Brosnahan, M.L.; McRaven, L.; Bahr, F.; et al. Evidence for massive and recurrent toxic blooms of Alexandrium catenella in the Alaskan Arctic. Proc. Natl. Acad. Sci. USA 2021, 118, e2107387118. [Google Scholar] [CrossRef] [PubMed]
- Karlson, B.; Andersen, P.; Arneborg, L.; Cembella, A.; Eikrem, W.; John, U.; West, J.J.; Klemm, K.; Kobos, J.; Lehtinen, S.; et al. Harmful algal blooms and their effects in coastal seas of Northern Europe. Harmful Algae 2021, 102, 101989. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, C.H.; Bates, S.S.; Martin, J.L.; Haigh, N.; Howland, K.L.; Lewis, N.I.; Locke, A.; Peña, A.; Poulin, M.; Rochon, A.; et al. Three decades of Canadian marine harmful algal events: Phytoplankton and phycotoxins of concern to human and ecosystem health. Harmful Algae 2021, 102, 101852. [Google Scholar] [CrossRef] [PubMed]
- DeMott, P.J.; Prenni, A.J.; Liu, X.; Kreidenweis, S.M.; Petters, M.D.; Twohy, C.H.; Richardson, M.S.; Eidhammer, T.; Rogers, D.C. Predicting global atmospheric ice nuclei distributions and their impacts on climate. Proc. Natl. Acad. Sci. USA 2010, 107, 11217–11222. [Google Scholar] [CrossRef]
- Hoagland, P.; Scatasta, S. The Economic Effects of Harmful Algal Blooms; Springer: Berlin/Heidelberg, Germany, 2006; pp. 391–402. [Google Scholar]
- Scheffer, M. On the Dominance of Filamentous Cyanobacteria Inshallow, Turbid Lakes. Ecology 1997, 78, 272–282. [Google Scholar] [CrossRef]
- Lad, A.; Breidenbach, J.D.; Su, R.C.; Murray, J.; Kuang, R.; Mascarenhas, A.; Najjar, J.; Patel, S.; Hegde, P.; Youssef, M.; et al. As We Drink and Breathe: Adverse Health Effects of Microcystins and Other Harmful Algal Bloom Toxins in the Liver, Gut, Lungs and Beyond. Life 2022, 12, 418. [Google Scholar] [CrossRef]
- Sivonen, K. Cyanobacterial Toxins. In Encyclopedia of Microbiology, 3rd ed.; Schaechter, M., Ed.; Academic Press: Oxford, UK, 2009; pp. 290–307. [Google Scholar]
- Wagner, T.; Erickson, L.E. Sustainable Management of Eutrophic Lakes and Reservoirs. J. Environ. Prot. 2017, 8, 436–463. [Google Scholar] [CrossRef]
- Zanchett, G.; Oliveira-Filho, E.C. Cyanobacteria and cyanotoxins: From impacts on aquatic ecosystems and human health to anticarcinogenic effects. Toxins 2013, 5, 1896–1917. [Google Scholar] [CrossRef]
- Joyce, S. The dead zones: Oxygen-starved coastal waters. Environ. Health Perspect. 2000, 108, A120–A125. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W. Mitigating Toxic Planktonic Cyanobacterial Blooms in Aquatic Ecosystems Facing Increasing Anthropogenic and Climatic Pressures. Toxins 2018, 10, 76. [Google Scholar] [CrossRef] [PubMed]
- Giglio, S.; Jiang, J.; Saint, C.P.; Cane, D.E.; Monis, P.T. Isolation and characterization of the gene associated with geosmin production in cyanobacteria. Environ. Sci. Technol. 2008, 42, 8027–8032. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef]
- Paerl, H.W.; Paul, V.J. Climate change: Links to global expansion of harmful cyanobacteria. Water Res. 2012, 46, 1349–1363. [Google Scholar] [CrossRef]
- O’Reilly, C.M.; Sharma, S.; Gray, D.K.; Hampton, S.E.; Read, J.S.; Rowley, R.J.; Schneider, P.; Lenters, J.D.; McIntyre, P.B.; Kraemer, B.M.; et al. Rapid and highly variable warming of lake surface waters around the globe. Geophys. Res. Lett. 2015, 42, 10773–10781. [Google Scholar] [CrossRef]
- van Vliet, M.T.H.; Franssen, W.H.P.; Yearsley, J.R.; Ludwig, F.; Haddeland, I.; Lettenmaier, D.P.; Kabat, P. Global river discharge and water temperature under climate change. Glob. Environ. Chang. 2013, 23, 450–464. [Google Scholar] [CrossRef]
- Coles, J.F.; Jones, R.C. Effect of temperature on photosynthesis-light response and growth of four phytoplankton species isolated from a tidal freshwater river. J. Phycol. 2000, 36, 7–16. [Google Scholar] [CrossRef]
- De Stasio, B.T.; Hill, D.K.; Kleinhans, J.M.; Nibbelink, N.P.; Magnuson, J.J. Potential effects of global climate change on small north-temperate lakes: Physics, fish, and plankton. Limnol. Oceanogr. 1996, 41, 1136–1149. [Google Scholar] [CrossRef]
- Peeters, F.; Straile, D.; Lorke, A.; Livingstone, D.M. Earlier onset of the spring phytoplankton bloom in lakes of the temperate zone in a warmer climate. Glob. Chang. Biol. 2007, 13, 1898–1909. [Google Scholar] [CrossRef]
- Walsby, A.E.; Hayes, P.K.; Boje, R.; Stal, L.J. The selective advantage of buoyancy provided by gas vesicles for planktonic cyanobacteria in the Baltic Sea. New Phytol. 1997, 136, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Walsby, A.E.; Schanz, F.; Schmid, M. The Burgundy-blood phenomenon: A model of buoyancy change explains autumnal waterblooms by Planktothrix rubescens in Lake Zurich. New Phytol. 2006, 169, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, A.; Becker, K.; Karsten, U.; Remias, D.; Ganzera, M. Analysis of Mycosporine-Like Amino Acids in Selected Algae and Cyanobacteria by Hydrophilic Interaction Liquid Chromatography and a Novel MAA from the Red Alga Catenella repens. Mar. Drugs 2015, 13, 6291–6305. [Google Scholar] [CrossRef] [PubMed]
- Kilian, O.; Steunou, A.S.; Fazeli, F.; Bailey, S.; Bhaya, D.; Grossman, A.R. Responses of a thermophilic Synechococcus isolate from the microbial mat of Octopus Spring to light. Appl. Environ. Microbiol. 2007, 73, 4268–4278. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, M.; Hihara, Y. Acclimation to high-light conditions in cyanobacteria: From gene expression to physiological responses. J. Plant. Res. 2012, 125, 11–39. [Google Scholar] [CrossRef] [PubMed]
- Ohad, I.; Raanan, H.; Keren, N.; Tchernov, D.; Kaplan, A. Light-induced changes within photosystem II protects Microcoleus sp. in biological desert sand crusts against excess light. PLoS ONE 2010, 5, e11000. [Google Scholar] [CrossRef]
- Amorim, C.A.; Moura, A.D.N. Ecological impacts of freshwater algal blooms on water quality, plankton biodiversity, structure, and ecosystem functioning. Sci. Total Environ. 2021, 758, 143605. [Google Scholar] [CrossRef]
- Kramer, B.J.; Hem, R.; Gobler, C.J. Elevated CO(2) significantly increases N(2) fixation, growth rates, and alters microcystin, anatoxin, and saxitoxin cell quotas in strains of the bloom-forming cyanobacteria, Dolichospermum. Harmful Algae 2022, 120, 102354. [Google Scholar] [CrossRef]
- Agarwal, P.; Soni, R.; Kaur, P.; Madan, A.; Mishra, R.; Pandey, J.; Singh, S.; Singh, G. Cyanobacteria as a Promising Alternative for Sustainable Environment: Synthesis of Biofuel and Biodegradable Plastics. Front. Microbiol. 2022, 13, 939347. [Google Scholar] [CrossRef]
- Georges des Aulnois, M.; Roux, P.; Caruana, A.; Reveillon, D.; Briand, E.; Herve, F.; Savar, V.; Bormans, M.; Amzil, Z. Physiological and Metabolic Responses of Freshwater and Brackish-Water Strains of Microcystis aeruginosa Acclimated to a Salinity Gradient: Insight into Salt Tolerance. Appl. Environ. Microbiol. 2019, 85, e01614-19. [Google Scholar] [CrossRef]
- Moisander, P.H.; McClinton, E., 3rd; Paerl, H.W. Salinity effects on growth, photosynthetic parameters, and nitrogenase activity in estuarine planktonic cyanobacteria. Microb. Ecol. 2002, 43, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Kanoshina, I.; Lips, U.; Leppänen, J.-M. The influence of weather conditions (temperature and wind) on cyanobacterial bloom development in the Gulf of Finland (Baltic Sea). Harmful Algae 2003, 2, 29–41. [Google Scholar] [CrossRef]
- Chen, Y.; Qin, B.; Teubner, K.; Dokulil, M.T. Long-term dynamics of phytoplankton assemblages: Microcystis-domination in Lake Taihu, a large shallow lake in China. J. Plankton Res. 2003, 25, 445–453. [Google Scholar] [CrossRef]
- National Lakes Assessment 2017 Key Findings. 2017. Available online: https://www.epa.gov/national-aquatic-resource-surveys/national-lakes-assessment-2017-key-findings (accessed on 16 February 2023).
- Chaffin, J.D.; Bratton, J.F.; Verhamme, E.M.; Bair, H.B.; Beecher, A.A.; Binding, C.E.; Birbeck, J.A.; Bridgeman, T.B.; Chang, X.; Crossman, J.; et al. The Lake Erie HABs Grab: A binational collaboration to characterize the western basin cyanobacterial harmful algal blooms at an unprecedented high-resolution spatial scale. Harmful Algae 2021, 108, 102080. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhang, Y.; Arhonditsis, G.B.; Gao, J.; Chen, Q.; Peng, J. The magnitude and drivers of harmful algal blooms in China’s lakes and reservoirs: A national-scale characterization. Water Res. 2020, 181, 115902. [Google Scholar] [CrossRef]
- Kleinteich, J.; Wood, S.A.; Küpper, F.C.; Camacho, A.; Quesada, A.; Frickey, T.; Dietrich, D.R. Temperature-related changes in polar cyanobacterial mat diversity and toxin production. Nat. Clim. Chang. 2012, 2, 356–360. [Google Scholar] [CrossRef]
- Dziallas, C.; Grossart, H.P. Increasing oxygen radicals and water temperature select for toxic Microcystis sp. PLoS ONE 2011, 6, e25569. [Google Scholar] [CrossRef]
- Utkilen, H.; Gjolme, N. Toxin Production by Microcystis aeruginosa as a Function of Light in Continuous Cultures and Its Ecological Significance. Appl. Environ. Microbiol. 1992, 58, 1321–1325. [Google Scholar] [CrossRef]
- Kaebernick, M.; Neilan, B.A.; Borner, T.; Dittmann, E. Light and the transcriptional response of the microcystin biosynthesis gene cluster. Appl. Environ. Microbiol. 2000, 66, 3387–3392. [Google Scholar] [CrossRef]
- Wiedner, C.; Visser, P.M.; Fastner, J.; Metcalf, J.S.; Codd, G.A.; Mur, L.R. Effects of light on the microcystin content of Microcystis strain PCC 7806. Appl. Environ. Microbiol. 2003, 69, 1475–1481. [Google Scholar] [CrossRef]
- Tonk, L.; Visser, P.M.; Christiansen, G.; Dittmann, E.; Snelder, E.O.; Wiedner, C.; Mur, L.R.; Huisman, J. The microcystin composition of the cyanobacterium Planktothrix agardhii changes toward a more toxic variant with increasing light intensity. Appl. Environ. Microbiol. 2005, 71, 5177–5181. [Google Scholar] [CrossRef] [PubMed]
- Orr, P.T.; Jones, G.J. Relationship between microcystin production and cell division rates in nitrogen-limited Microcystis aeruginosa cultures. Limnol. Oceanogr. 1998, 43, 1604–1614. [Google Scholar] [CrossRef]
- Long, B.M.; Jones, G.J.; Orr, P.T. Cellular microcystin content in N-limited Microcystis aeruginosa can be predicted from growth rate. Appl. Environ. Microbiol. 2001, 67, 278–283. [Google Scholar] [CrossRef]
- Utkilen, H.; Gjolme, N. Iron-stimulated toxin production in Microcystis aeruginosa. Appl. Environ. Microbiol. 1995, 61, 797–800. [Google Scholar] [CrossRef]
- Vezie, C.; Rapala, J.; Vaitomaa, J.; Seitsonen, J.; Sivonen, K. Effect of nitrogen and phosphorus on growth of toxic and nontoxic Microcystis strains and on intracellular microcystin concentrations. Microb. Ecol. 2002, 43, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Downing, T.G.; Meyer, C.; Gehringer, M.M.; van de Venter, M. Microcystin content of Microcystis aeruginosa is modulated by nitrogen uptake rate relative to specific growth rate or carbon fixation rate. Environ. Toxicol. 2005, 20, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Downing, T.G.; Sember, C.S.; Gehringer, M.M.; Leukes, W. Medium N:P ratios and specific growth rate comodulate microcystin and protein content in Microcystis aeruginosa PCC7806 and M. aeruginosa UV027. Microb. Ecol. 2005, 49, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Van de Waal, D.B.; Verspagen, J.M.; Lurling, M.; Van Donk, E.; Visser, P.M.; Huisman, J. The ecological stoichiometry of toxins produced by harmful cyanobacteria: An experimental test of the carbon-nutrient balance hypothesis. Ecol. Lett. 2009, 12, 1326–1335. [Google Scholar] [CrossRef]
- Van de Waal, D.B.; Verspagen, J.M.; Finke, J.F.; Vournazou, V.; Immers, A.K.; Kardinaal, W.E.; Tonk, L.; Becker, S.; Van Donk, E.; Visser, P.M.; et al. Reversal in competitive dominance of a toxic versus non-toxic cyanobacterium in response to rising CO2. ISME J. 2011, 5, 1438–1450. [Google Scholar] [CrossRef] [PubMed]
- Roy-Lachapelle, A.; Solliec, M.; Sauve, S.; Gagnon, C. A Data-Independent Methodology for the Structural Characterization of Microcystins and Anabaenopeptins Leading to the Identification of Four New Congeners. Toxins 2019, 11, 619. [Google Scholar] [CrossRef]
- Malta, J.F.; Nardocci, A.C.; Razzolini, M.T.P.; Diniz, V.; Cunha, D.G.F. Exposure to microcystin-LR in tropical reservoirs for water supply poses high risks for children and adults. Environ. Monit. Assess. 2022, 194, 253. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; D’Anci, K.E.; Rosenberg, I.H. Water, hydration, and health. Nutr. Rev. 2010, 68, 439–458. [Google Scholar] [CrossRef] [PubMed]
- Tito, J.C.R.; Luna, L.M.G.; Noppe, W.N.; Hubert, I.A. First Report on Microcystin-LR Occurrence in Water Reservoirs of Eastern Cuba, and Environmental Trigger Factors. Toxins 2022, 14, 209. [Google Scholar] [CrossRef] [PubMed]
- Uche, A.U.; Edward, A.M.; Bahram, G. Risk assessment of cyanobacteria-toxins for small drinking water treatment plants with lake water intakes. Int. J. Water Resour. Environ. Eng. 2017, 9, 121–126. [Google Scholar] [CrossRef]
- Oliveira, E.D.C.; Castelo-Branco, R.; Silva, L.; Silva, N.; Azevedo, J.; Vasconcelos, V.; Faustino, S.; Cunha, A. First Detection of Microcystin-LR in the Amazon River at the Drinking Water Treatment Plant of the Municipality of Macapa, Brazil. Toxins 2019, 11, 669. [Google Scholar] [CrossRef]
- Chatziefthimiou, A.D.; Metcalf, J.S.; Glover, W.B.; Banack, S.A.; Dargham, S.R.; Richer, R.A. Cyanobacteria and cyanotoxins are present in drinking water impoundments and groundwater wells in desert environments. Toxicon 2016, 114, 75–84. [Google Scholar] [CrossRef]
- Douma, M.; Ouahid, Y.; del Campo, F.F.; Loudiki, M.; Mouhri, K.; Oudra, B. Identification and quantification of cyanobacterial toxins (microcystins) in two Moroccan drinking-water reservoirs (Mansour Eddahbi, Almassira). Environ. Monit. Assess. 2010, 160, 439–450. [Google Scholar] [CrossRef]
- Walls, J.T.; Wyatt, K.H.; Doll, J.C.; Rubenstein, E.M.; Rober, A.R. Hot and toxic: Temperature regulates microcystin release from cyanobacteria. Sci. Total Environ. 2018, 610–611, 786–795. [Google Scholar] [CrossRef]
- Zamyadi, A.; MacLeod, S.L.; Fan, Y.; McQuaid, N.; Dorner, S.; Sauve, S.; Prévost, M. Toxic cyanobacterial breakthrough and accumulation in a drinking water plant: A monitoring and treatment challenge. Water Res. 2012, 46, 1511–1523. [Google Scholar] [CrossRef]
- Chia, M.A.; Kwaghe, M.J. Microcystins contamination of surface water supply sources in Zaria-Nigeria. Environ. Monit. Assess. 2015, 187, 606. [Google Scholar] [CrossRef]
- Svircev, Z.; Lalic, D.; Bojadzija Savic, G.; Tokodi, N.; Drobac Backovic, D.; Chen, L.; Meriluoto, J.; Codd, G.A. Global geographical and historical overview of cyanotoxin distribution and cyanobacterial poisonings. Arch. Toxicol. 2019, 93, 2429–2481. [Google Scholar] [CrossRef] [PubMed]
- Svircev, Z.; Drobac, D.; Tokodi, N.; Mijovic, B.; Codd, G.A.; Meriluoto, J. Toxicology of microcystins with reference to cases of human intoxications and epidemiological investigations of exposures to cyanobacteria and cyanotoxins. Arch. Toxicol. 2017, 91, 621–650. [Google Scholar] [CrossRef] [PubMed]
- Ueno, Y.; Nagata, S.; Tsutsumi, T.; Hasegawa, A.; Watanabe, M.F.; Park, H.D.; Chen, G.-C.; Chen, G.; Yu, S.-Z. Detection of microcystins, a blue-green algal hepatotoxin, in drinking water sampled in Haimen and Fusui, endemic areas of primary liver cancer in China, by highly sensitive immunoassay. Carcinogenesis 1996, 17, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zhao, N.; Zi, X. The relationship between cyanotoxin (microcystin, MC) in pond-ditch water and primary liver cancer in China. Zhonghua Zhong Liu Za Zhi 2001, 23, 96–99. [Google Scholar] [PubMed]
- Zhou, L.; Yu, H.; Chen, K. Relationship between microcystin in drinking water and colorectal cancer. Biomed. Environ. Sci. 2002, 15, 166–171. [Google Scholar] [PubMed]
- Sarkar, S.; Kimono, D.; Albadrani, M.; Seth, R.K.; Busbee, P.; Alghetaa, H.; Porter, D.E.; Scott, G.I.; Brooks, B.; Nagarkatti, M.; et al. Environmental microcystin targets the microbiome and increases the risk of intestinal inflammatory pathology via NOX2 in underlying murine model of Nonalcoholic Fatty Liver Disease. Sci. Rep. 2019, 9, 8742. [Google Scholar] [CrossRef] [PubMed]
- Drobac, D.; Tokodi, N.; Simeunovic, J.; Baltic, V.; Stanic, D.; Svircev, Z. Human exposure to cyanotoxins and their effects on health. Arh. Hig. Rada Toksikol. 2013, 64, 119–130. [Google Scholar] [CrossRef]
- Svircev, Z.; Drobac, D.; Tokodi, N.; Luzanin, Z.; Munjas, A.M.; Nikolin, B.; Vuleta, D.; Meriluoto, J. Epidemiology of cancers in Serbia and possible connection with cyanobacterial blooms. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2014, 32, 319–337. [Google Scholar] [CrossRef]
- Lei, F.; Lei, X.; Li, R.; Tan, H. Microcystin-LR in peripheral circulation worsens the prognosis partly through oxidative stress in patients with hepatocellular carcinoma. Clin. Exp. Med. 2019, 19, 235–243. [Google Scholar] [CrossRef]
- Feng, S.; Deng, S.; Tang, Y.; Liu, Y.; Yang, Y.; Xu, S.; Tang, P.; Lu, Y.; Duan, Y.; Wei, J.; et al. Microcystin-LR Combined with Cadmium Exposures and the Risk of Chronic Kidney Disease: A Case-Control Study in Central China. Environ. Sci. Technol. 2022, 56, 15818–15827. [Google Scholar] [CrossRef]
- Lin, H.; Liu, W.; Zeng, H.; Pu, C.; Zhang, R.; Qiu, Z.; Chen, J.-A.; Wang, L.; Tan, Y.; Zheng, C.; et al. Determination of Environmental Exposure to Microcystin and Aflatoxin as a Risk for Renal Function Based on 5493 Rural People in Southwest China. Environ. Sci. Technol. 2016, 50, 5346–5356. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, P.; Kumar Agrawal, M.; Nath Bagchi, S. Microcystin-producing and non-producing cyanobacterial blooms collected from the Central India harbor potentially pathogenic Vibrio cholerae. Ecotoxicol. Environ. Saf. 2015, 115, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Mahmuda, S.; Morshed, M.G.; Bakht, H.B.; Khan, M.N.; Sack, R.B.; A Sack, D. Role of cyanobacteria in the persistence of Vibrio cholerae O139 in saline microcosms. Can. J. Microbiol. 2004, 50, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Jesser, K.J.; Noble, R.T. Vibrio Ecology in the Neuse River Estuary, North Carolina, Characterized by Next-Generation Amplicon Sequencing of the Gene Encoding Heat Shock Protein 60 (hsp60). Appl. Environ Microbiol. 2018, 84, e00333-18. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Rahim, Z.; Alam, M.J.; Begum, S.; Moniruzzaman, S.M.; Umeda, A.; Amako, K.; Albert, M.; Sack, R.; Huq, A.; et al. Association of Vibrio cholerae O1 with the cyanobacterium, Anabaena sp., elucidated by polymerase chain reaction and transmission electron microscopy. Trans. R. Soc. Trop. Med. Hyg. 1999, 93, 36–40. [Google Scholar] [CrossRef]
- Eiler, A.; Gonzalez-Rey, C.; Allen, S.; Bertilsson, S. Growth response of Vibrio cholerae and other Vibrio spp. to cyanobacterial dissolved organic matter and temperature in brackish water. FEMS Microbiol. Ecol. 2007, 60, 411–418. [Google Scholar] [CrossRef]
- Thompson, F.L.; Austin, B.; Swings, J. Vibrio vulnificus. In The Biology of Vibrios; ASM Press: Washington, DC, USA, 2006. [Google Scholar] [CrossRef]
- Deeb, R.; Tufford, D.; Scott, G.I.; Moore, J.G.; Dow, K. Impact of Climate Change on Vibrio vulnificus Abundance and Exposure Risk. Estuaries Coast. 2018, 41, 2289–2303. [Google Scholar] [CrossRef] [PubMed]
- Paz, S.; Bisharat, N.; Paz, E.; Kidar, O.; Cohen, D. Climate change and the emergence of Vibrio vulnificus disease in Israel. Environ. Res. 2007, 103, 390–396. [Google Scholar] [CrossRef]
- Greenfield, D.I.; Gooch Moore, J.; Stewart, J.R.; Hilborn, E.D.; George, B.J.; Li, Q.; Dickerson, J.; Keppler, C.K.; Sandifer, P.A. Temporal and Environmental Factors Driving Vibrio Vulnificus and V. Parahaemolyticus Populations and Their Associations with Harmful Algal Blooms in South Carolina Detention Ponds and Receiving Tidal Creeks. Geohealth 2017, 1, 306–317. [Google Scholar] [CrossRef]
- Falcone-Dias, M.F.; Rodrigues, M.V.; Nielsen, J.L.; de Jonge, N.; Jorgensen, N.O.G.; Alonso, D.P.; David, G.S.; da Silva, R.J.; Júnior, J.P.A. Occurrence of Cyanobacteria and microcystins in hydroelectric reservoirs used for fish farming. J. Water Health 2020, 18, 983–994. [Google Scholar] [CrossRef]
- Dai, L.; Liu, C.; Peng, L.; Song, C.; Li, X.; Tao, L.; Li, G. Different distribution patterns of microorganisms between aquaculture pond sediment and water. J. Microbiol. 2021, 59, 376–388. [Google Scholar] [CrossRef] [PubMed]
- Stabili, L.; Di Salvo, M.; Alifano, P.; Tala, A. An Integrative, Multiparametric Approach for the Comprehensive Assessment of Microbial Quality and Pollution in Aquaculture Systems. Microb. Ecol. 2022, 83, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Dieng, M.T.; Niang, S.O.; Ly, F.; Bathily, T.; Ndiaye, B. Necrotizing dermatitis due to Vibrio Vulnificus. Ann. Dermatol. Venereol. 2001, 128, 653–655. [Google Scholar] [PubMed]
- Rodrigues, S.M.; Goncalves, E.G.; Mello, D.M.; Oliveira, E.G.; Hofer, E. Identification of Vibrio spp bacteria on skin lesions of fisherman in the county of Raposa-MA. Rev. Soc. Bras. Med. Trop. 2001, 34, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Xing, C.; Hou, D.; Zeng, S.; Zhou, R.; Yu, L.; Wang, H.; Deng, Z.; Weng, S.; He, J.; et al. Distinct bacterial communities in the environmental water, sediment and intestine between two crayfish-plant coculture ecosystems. Appl. Microbiol. Biotechnol. 2021, 105, 5087–5101. [Google Scholar] [CrossRef] [PubMed]
- Chithira, M.S.; Aishwarya, P.V.; Mohan, A.S.; Antony, S.P. Metagenomic analysis of microbial communities in the sediments of a semi-intensive penaeid shrimp culture system. J. Genet. Eng. Biotechnol. 2021, 19, 136. [Google Scholar] [CrossRef]
- Bouaicha, N.; Miles, C.O.; Beach, D.G.; Labidi, Z.; Djabri, A.; Benayache, N.Y.; Nguyen-Quang, T. Structural Diversity, Characterization and Toxicology of Microcystins. Toxins 2019, 11, 714. [Google Scholar] [CrossRef]
- Peng, L.; Tang, Q.; Gu, J.; Lei, L.; Chen, W.; Song, L. Seasonal variation of microcystins and their accumulation in fish in two large shallow lakes of China. Ecotoxicology 2020, 29, 790–800. [Google Scholar] [CrossRef]
- Codd, G.; Bell, S.; Kaya, K.; Ward, C.; Beattie, K.; Metcalf, J. Cyanobacterial toxins, exposure routes and human health. Eur. J. Phycol. 1999, 34, 405–415. [Google Scholar] [CrossRef]
- Plaas, H.E.; Paerl, H.W. Toxic Cyanobacteria: A Growing Threat to Water and Air Quality. Environ. Sci. Technol. 2021, 55, 44–64. [Google Scholar] [CrossRef]
- Greer, B.; Maul, R.; Campbell, K.; Elliott, C.T. Detection of freshwater cyanotoxins and measurement of masked microcystins in tilapia from Southeast Asian aquaculture farms. Anal. Bioanal. Chem. 2017, 409, 4057–4069. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Li, Y.W.; Liu, B.L.; Zhao, H.M.; Li, H.; Cai, Q.Y.; Mo, C.-H.; Wong, M.-H.; Li, Q.X. High ecological and human health risks from microcystins in vegetable fields in southern China. Environ. Int. 2019, 133 Pt A, 105142. [Google Scholar] [CrossRef]
- Poste, A.E.; Hecky, R.E.; Guildford, S.J. Evaluating microcystin exposure risk through fish consumption. Environ. Sci. Technol. 2011, 45, 5806–5811. [Google Scholar] [CrossRef] [PubMed]
- Ito, E.; Kondo, F.; Harada, K. First report on the distribution of orally administered microcystin-LR in mouse tissue using an immunostaining method. Toxicon 2000, 38, 37–48. [Google Scholar] [CrossRef]
- Guo, X.; Chen, L.; Chen, J.; Xie, P.; Li, S.; He, J.; Li, W.; Fan, H.; Yu, D.; Zeng, C. Quantitatively evaluating detoxification of the hepatotoxic microcystin-LR through the glutathione (GSH) pathway in SD rats. Environ. Sci. Pollut. Res. Int. 2015, 22, 19273–19284. [Google Scholar] [CrossRef] [PubMed]
- Snyder, H. Microcystin Accumulation in Fish Muscle Tissue: Exploring the Safety of Fish Consumption in Several Michigan Water Bodies; Grand Valley State University: Allendale, MI, USA, 2015. [Google Scholar]
- Zegura, B. An Overview of the Mechanisms of Microcystin-LR Genotoxicity and Potential Carcinogenicity. Mini Rev. Med. Chem. 2016, 16, 1042–1062. [Google Scholar] [CrossRef]
- Hinojosa, M.G.; Gutierrez-Praena, D.; Prieto, A.I.; Guzman-Guillen, R.; Jos, A.; Camean, A.M. Neurotoxicity induced by microcystins and cylindrospermopsin: A review. Sci. Total Environ. 2019, 668, 547–565. [Google Scholar] [CrossRef]
- Sitprija, V.; Sitprija, S. Marine toxins and nephrotoxicity: Mechanism of injury. Toxicon 2019, 161, 44–49. [Google Scholar] [CrossRef]
- Zhang, S.; Du, X.; Liu, H.; Losiewic, M.D.; Chen, X.; Ma, Y.; Wang, R.; Tian, Z.; Shi, L.; Guo, H.; et al. The latest advances in the reproductive toxicity of microcystin-LR. Environ. Res. 2021, 192, 110254. [Google Scholar] [CrossRef]
- Runnegar, M.; Berndt, N.; Kong, S.M.; Lee, E.Y.; Zhang, L. In vivo and in vitro binding of microcystin to protein phosphatases 1 and 2A. Biochem. Biophys. Res. Commun. 1995, 216, 162–169. [Google Scholar] [CrossRef]
- Liu, J.; Sun, Y. The role of PP2A-associated proteins and signal pathways in microcystin-LR toxicity. Toxicol. Lett. 2015, 236, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.X.; Nam Ong, C. Role of oxidative stress and mitochondrial changes in cyanobacteria-induced apoptosis and hepatotoxicity. FEMS Microbiol. Lett. 2003, 220, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Christen, V.; Meili, N.; Fent, K. Microcystin-LR induces endoplasmatic reticulum stress and leads to induction of NFkappaB, interferon-alpha, and tumor necrosis factor-alpha. Environ. Sci. Technol. 2013, 47, 3378–3385. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xie, P.; Fan, H. Genomic profiling of microRNAs and proteomics reveals an early molecular alteration associated with tumorigenesis induced by MC-LR in mice. Environ. Sci. Technol. 2012, 46, 34–41. [Google Scholar] [CrossRef]
- Saha, P.; Bose, D.; Stebliankin, V.; Cickovski, T.; Seth, R.K.; Porter, D.E.; Brooks, B.W.; Mathee, K.; Narasimhan, G.; Colwell, R.; et al. Prior exposure to microcystin alters host gut resistome and is associated with dysregulated immune homeostasis in translatable mouse models. Sci Rep. 2022, 12, 11516, PMID: 35799048; PMCID: PMC9262933. [Google Scholar] [CrossRef]
- Albadrani, M.; Seth, R.K.; Sarkar, S.; Kimono, D.; Mondal, A.; Bose, D.; Porter, D.E.; Scott, G.I.; Brooks, B.; Raychoudhury, S.; et al. Exogenous PP2A inhibitor exacerbates the progression of nonalcoholic fatty liver disease via NOX2-dependent activation of miR21. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 317, G408–G428. [Google Scholar] [CrossRef]
- Mondal, A.; Saha, P.; Bose, D.; Chatterjee, S.; Seth, R.K.; Xiao, S.; Porter, D.E.; Brooks, B.W.; Scott, G.I.; Nagarkatti, M.; et al. Environmental Microcystin exposure in underlying NAFLD-induced exacerbation of neuroinflammation, blood-brain barrier dysfunction, and neurodegeneration are NLRP3 and S100B dependent. Toxicology 2021, 461, 152901, PMID: 34416350; PMCID: PMC8503918. [Google Scholar] [CrossRef]
- Sarkar, S.; Alhasson, F.; Kimono, D.; Albadrani, M.; Seth, R.K.; Xiao, S.; Porter, D.E.; Scott, G.I.; Brooks, B.; Nagarkatti, M.; et al. Microcystin exposure worsens nonalcoholic fatty liver disease associated ectopic glomerular toxicity via NOX-2-MIR21 axis. Environ. Toxicol. Pharmacol. 2020, 73, 103281, PMID: 31706246; PMCID: PMC7100051. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.; Song, L.; Liu, J. Responses of antioxidant systems in the hepatocytes of common carp (Cyprinus carpio L.) to the toxicity of microcystin-LR. Toxicon 2003, 42, 85–89. [Google Scholar] [CrossRef]
- Ghosh, S.; Khan, S.A.; Wickstrom, M.; Beasley, V. Effects of microcystin-LR on actin and the actin-associated proteins alpha-actinin and talin in hepatocytes. Nat. Toxins 1995, 3, 405–414. [Google Scholar] [CrossRef]
- Mereish, K.A.; Bunner, D.L.; Ragland, D.R.; Creasia, D.A. Protection against microcystin-LR-induced hepatotoxicity by Silymarin: Biochemistry, histopathology, and lethality. Pharm. Res. 1991, 8, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Ito, E.; Kondo, F.; Terao, K.; Harada, K. Neoplastic nodular formation in mouse liver induced by repeated intraperitoneal injections of microcystin-LR. Toxicon 1997, 35, 1453–1457. [Google Scholar] [CrossRef] [PubMed]
- Milutinovic, A.; Zivin, M.; Zorc-Pleskovic, R.; Sedmak, B.; Suput, D. Nephrotoxic effects of chronic administration of microcystins -LR and -YR. Toxicon 2003, 42, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, C.; Zhu, J.; Ding, J.; Chen, Y.; Han, X. Blood-brain barrier disruption and inflammation reaction in mice after chronic exposure to Microcystin-LR. Sci. Total Environ. 2019, 689, 662–678. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Gu, S.; Yin, X.; Yuan, M.; Xiang, Z.; Li, Z.; Cao, H.; Meng, X.; Hu, K.; Han, X. The toxic effects of microcystin-LR on mouse lungs and alveolar type II epithelial cells. Toxicon 2016, 115, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Milutinovic, A.; Zorc-Pleskovic, R.; Petrovic, D.; Zorc, M.; Suput, D. Microcystin-LR induces alterations in heart muscle. Folia Biol. 2006, 52, 116–118. [Google Scholar]
- Burnham, J.P. Climate change and antibiotic resistance: A deadly combination. Ther. Adv. Infect. Dis. 2021, 8, 2049936121991374. [Google Scholar] [CrossRef]
- Sarkar, S.; Saha, P.; Seth, R.K.; Mondal, A.; Bose, D.; Kimono, D.; Albadrani, M.; Mukherjee, A.; Porter, D.E.; Scott, G.I.; et al. Higher intestinal and circulatory lactate associated NOX2 activation leads to an ectopic fibrotic pathology following microcystin co-exposure in murine fatty liver disease. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2020, 238, 108854, PMID: 32781293; PMCID: PMC7541568. [Google Scholar] [CrossRef]
- Al-Badrani, M.; Saha, P.; Mondal, A.; Seth, R.K.; Sarkar, S.; Kimono, D.; Bose, D.; Porter, D.E.; Scott, G.I.; Brooks, B.; et al. Early microcystin-LR exposure-linked inflammasome activation in mice causes development of fatty liver disease and insulin resistance. Environ. Toxicol. Pharmacol. 2020, 80, 103457, PMID: 32687983; PMC7609636. [Google Scholar] [CrossRef]
- Aslam, B.; Wang, W.; Arshad, M.I.; Khurshid, M.; Muzammil, S.; Nisar, M.A.; Alvi, R.F.; Aslam, M.A.; Qamar, M.U.; Salamat, M.K.F.; et al. Antibiotic resistance: A rundown of a global crisis. Infect. Drug Resist. 2018, 11, 1645–1658. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Amabile-Cuevas, C.F.; Cars, O.; Evans, T.; Heymann, D.L.; Hoffman, S.; Holmes, A.; Mendelson, M.; Sridhar, D.; Woolhouse, M.; et al. UN High-Level Meeting on antimicrobials—What do we need? Lancet 2016, 388, 218–220. [Google Scholar] [CrossRef] [PubMed]
- Wright, G.D. The antibiotic resistome: The nexus of chemical and genetic diversity. Nat. Rev. Microbiol. 2007, 5, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.A.; Westman, E.L.; Wright, G.D. The antibiotic resistome: What’s new? Curr. Opin. Microbiol. 2014, 21, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.A.; Wright, G.D. The antibiotic resistance “mobilome”: Searching for the link between environment and clinic. Front. Microbiol. 2013, 4, 138. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, K.J.; Reyes, A.; Wang, B.; Selleck, E.M.; Sommer, M.O.; Dantas, G. The shared antibiotic resistome of soil bacteria and human pathogens. Science 2012, 337, 1107–1111. [Google Scholar] [CrossRef]
- Guo, X.; Yan, Z.; Zhang, Y.; Xu, W.; Kong, D.; Shan, Z.; Wang, N. Behavior of antibiotic resistance genes under extremely high-level antibiotic selection pressures in pharmaceutical wastewater treatment plants. Sci. Total Environ. 2018, 612, 119–128. [Google Scholar] [CrossRef]
- Singh, S.; Verma, N.; Taneja, N. The human gut resistome: Current concepts & future prospects. Indian J. Med. Res. 2019, 150, 345–358. [Google Scholar] [CrossRef]
- Wright, G.D. Antibiotic resistance in the environment: A link to the clinic? Curr. Opin. Microbiol. 2010, 13, 589–594. [Google Scholar] [CrossRef]
- Centurion, V.B.; Silva, J.B.; Duarte, A.; Rosa, L.H.; Oliveira, V.M. Comparing resistome profiles from anthropogenically impacted and non-impacted areas of two South Shetland Islands—Maritime Antarctica. Environ. Pollut. 2022, 304, 119219. [Google Scholar] [CrossRef]
- Guan, Y.; Jia, J.; Fan, X.; Li, K.; Wang, Z. Anthropogenic impacts on antibiotic resistance genes and their hosts from pristine to urban river using metagenomic and binning approaches. Aquat. Toxicol. 2022, 249, 106221. [Google Scholar] [CrossRef]
- Matviichuk, O.; Mondamert, L.; Geffroy, C.; Gaschet, M.; Dagot, C.; Labanowski, J. River Biofilms Microbiome and Resistome Responses to Wastewater Treatment Plant Effluents Containing Antibiotics. Front. Microbiol. 2022, 13, 795206. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, K.; McNamara, P.; McDonald, W. Antibiotic resistance genes in an urban stream before and after a state fair. J. Water Health 2021, 19, 885–894. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, Q.; Wang, T.; Xu, N.; Lu, T.; Hong, W.; Penuelas, J.; Gillings, M.; Wang, M.; Gao, W.; et al. Assessment of global health risk of antibiotic resistance genes. Nat. Commun. 2022, 13, 1553. [Google Scholar] [CrossRef] [PubMed]
- Ohore, O.E.; Wei, Y.; Wang, Y.; Nwankwegu, A.S.; Wang, Z. Tracking the influence of antibiotics, antibiotic resistomes, and salinity gradient in modulating microbial community assemblage of surface water and the ecological consequences. Chemosphere 2022, 305, 135428. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yang, Y.; Jiang, X.; Ke, Y.; He, T.; Xie, S. Metagenomic insights into the profile of antibiotic resistomes in sediments of aquaculture wastewater treatment system. J. Environ. Sci. 2022, 113, 345–355. [Google Scholar] [CrossRef]
- Brown, L.P.; Murray, R.; Scott, A.; Tien, Y.C.; Lau, C.H.; Tai, V.; Topp, E. Responses of the Soil Bacterial Community, Resistome, and Mobilome to a Decade of Annual Exposure to Macrolide Antibiotics. Appl. Environ. Microbiol. 2022, 88, e0031622. [Google Scholar] [CrossRef]
- Seyoum, M.M.; Lichtenberg, R.; Orlofsky, E.; Bernstein, N.; Gillor, O. Antibiotic resistance in soil and tomato crop irrigated with freshwater and two types of treated wastewater. Environ. Res. 2022, 211, 113021. [Google Scholar] [CrossRef]
- Marathe, N.P.; Janzon, A.; Kotsakis, S.D.; Flach, C.F.; Razavi, M.; Berglund, F.; Kristiansson, E.; Larsson, D.G.J. Functional metagenomics reveals a novel carbapenem-hydrolyzing mobile beta-lactamase from Indian river sediments contaminated with antibiotic production waste. Environ. Int. 2018, 112, 279–286. [Google Scholar] [CrossRef]
- Tang, Z.P.; Xiao, S.S.; Duan, Y.; Liu, Y.J.; Gao, Y.Y.; Wu, Y.Y.; Chen, Y.-W.; Zhou, S. Distribution and Removal of Antibiotic-Resistant Bacteria and Antibiotic Resistance Genes in Petrochemical Wastewater Treatment Plants. Huan Jing Ke Xue 2021, 42, 3375–3384. [Google Scholar] [CrossRef]
- Pal, M.; Yadav, S.; Kapley, A.; Qureshi, A. Impact of cyanobacterial bloom on microbiomes of freshwater lakes. J. Biosci. 2021, 46, 96. [Google Scholar] [CrossRef]
- Valitalo, P.; Kruglova, A.; Mikola, A.; Vahala, R. Toxicological impacts of antibiotics on aquatic micro-organisms: A mini-review. Int. J. Hyg. Environ. Health 2017, 220, 558–569. [Google Scholar] [CrossRef] [PubMed]
- Cohen, Y.; Gurevitz, M. The Cyanobacteria—Ecology, Physiology and Molecular Genetics; Springer: New York, NY, USA, 2006; pp. 1074–1098. [Google Scholar]
- Tyagi, R.; Kaushik, B.D.; Kumar, J. Antimicrobial Activity of Some Cyanobacteria; Springer: Delhi, India, 2014; pp. 463–470. [Google Scholar]
- Cameron, J.C.; Pakrasi, H.B. Glutathione facilitates antibiotic resistance and photosystem I stability during exposure to gentamicin in cyanobacteria. Appl. Environ. Microbiol. 2011, 77, 3547–3550. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Chen, Q.; Zhang, J.; Guan, T.; Chen, Y.; Shi, W. Critical roles of cyanobacteria as reservoir and source for antibiotic resistance genes. Environ. Int. 2020, 144, 106034. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Haas, S.; Zemojtel, T.; Xiao, P.; Vingron, M.; Li, R. Genome-wide comparison of cyanobacterial transposable elements, potential genetic diversity indicators. Gene 2011, 473, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Mao, D.; Luo, Y. Ionic Liquid Facilitates the Conjugative Transfer of Antibiotic Resistance Genes Mediated by Plasmid RP4. Environ. Sci. Technol. 2015, 49, 8731–8740. [Google Scholar] [CrossRef]
- Bhat, P.; Bhaskar, M.; Sistla, S.; Kadhiravan, T. Fatal case of necrotising fasciitis due to Vibrio vulnificus in a patient with alcoholic liver disease and diabetes mellitus. BMJ Case Rep. 2019, 12, bcr-2018. [Google Scholar] [CrossRef]
- Froelich, B.A.; Daines, D.A. In hot water: Effects of climate change on Vibrio-human interactions. Environ. Microbiol. 2020, 22, 4101–4111. [Google Scholar] [CrossRef]
- Nelson, E.J.; Harris, J.B.; Morris, J.G., Jr.; Calderwood, S.B.; Camilli, A. Cholera transmission: The host, pathogen and bacteriophage dynamic. Nat. Rev. Microbiol. 2009, 7, 693–702. [Google Scholar] [CrossRef]
- Oliver, J.D. Vibrio vulnificus: Death on the half shell. A personal journey with the pathogen and its ecology. Microb. Ecol. 2013, 65, 793–799. [Google Scholar] [CrossRef]
- Newton, A.; Kendall, M.; Vugia, D.J.; Henao, O.L.; Mahon, B.E. Increasing rates of vibriosis in the United States, 1996–2010: Review of surveillance data from 2 systems. Clin. Infect. Dis. 2012, 54 (Suppl. S5), S391–S395. [Google Scholar] [CrossRef]
- Kelly, M.T. Effect of temperature and salinity on Vibrio (Beneckea) vulnificus occurrence in a Gulf Coast environment. Appl. Environ. Microbiol. 1982, 44, 820–824. [Google Scholar] [CrossRef] [PubMed]
- Miles, D.W.; Ross, T.; Olley, J.; McMeekin, T.A. Development and evaluation of a predictive model for the effect of temperature and water activity on the growth rate of Vibrio parahaemolyticus. Int. J. Food Microbiol. 1997, 38, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Ulitzur, S. Vibrio parahaemolyticus andVibrio alginolyticus: Short generation-time marine bacteria. Microb. Ecol. 1974, 1, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, D.; Hasan, N.A.; Huq, A.; Colwell, R.R. Distribution and dynamics of epidemic and pandemic Vibrio parahaemolyticus virulence factors. Front. Cell. Infect. Microbiol. 2013, 3, 97. [Google Scholar] [CrossRef] [PubMed]
- Strom, M.S.; Paranjpye, R.N. Epidemiology and pathogenesis of Vibrio vulnificus. Microbes Infect. 2000, 2, 177–188. [Google Scholar] [CrossRef]
- Hlady, W.G.; Klontz, K.C. The epidemiology of Vibrio infections in Florida, 1981–1993. J. Infect Dis. 1996, 173, 1176–1183. [Google Scholar] [CrossRef]
- Klontz, K.C.; Lieb, S.; Schreiber, M.; Janowski, H.T.; Baldy, L.M.; Gunn, R.A. Syndromes of Vibrio vulnificus infections. Clinical and epidemiologic features in Florida cases, 1981–1987. Ann. Intern. Med. 1988, 109, 318–323. [Google Scholar] [CrossRef]
- Wright, A.C.; Morris, J.G., Jr. The extracellular cytolysin of Vibrio vulnificus: Inactivation and relationship to virulence in mice. Infect. Immun. 1991, 59, 192–197. [Google Scholar] [CrossRef]
- Gray, L.D.; Kreger, A.S. Mouse skin damage caused by cytolysin from Vibrio vulnificus and by V. vulnificus infection. J. Infect. Dis. 1987, 155, 236–241. [Google Scholar] [CrossRef]
- Miyoshi, N.; Miyoshi, S.; Sugiyama, K.; Suzuki, Y.; Furuta, H.; Shinoda, S. Activation of the plasma kallikrein-kinin system by Vibrio vulnificus protease. Infect. Immun. 1987, 55, 1936–1939. [Google Scholar] [CrossRef]
- Gulig, P.A.; Bourdage, K.L.; Starks, A.M. Molecular Pathogenesis of Vibrio vulnificus. J. Microbiol. 2005, 43, 118–131. [Google Scholar] [PubMed]
- Kim, Y.R.; Lee, S.E.; Kook, H.; Yeom, J.A.; Na, H.S.; Kim, S.Y.; Chung, S.S.; Choy, H.E.; Rhee, J.H. Vibrio vulnificus RTX toxin kills host cells only after contact of the bacteria with host cells. Cell. Microbiol. 2008, 10, 848–862. [Google Scholar] [CrossRef] [PubMed]
- Baker-Austin, C.; Oliver, J.D. Vibrio vulnificus. Trends Microbiol. 2020, 28, 81–82. [Google Scholar] [CrossRef] [PubMed]
- Lad, A.; Su, R.C.; Breidenbach, J.D.; Stemmer, P.M.; Carruthers, N.J.; Sanchez, N.K.; Khalaf, F.K.; Zhang, S.; Kleinhenz, A.L.; Dube, P.; et al. Chronic Low Dose Oral Exposure to Microcystin-LR Exacerbates Hepatic Injury in a Murine Model of Non-Alcoholic Fatty Liver Disease. Toxins 2019, 11, 486. [Google Scholar] [CrossRef] [PubMed]
- Moreira, C.; Gomes, C.; Vasconcelos, V.; Antunes, A. Risk assessment of cyanobacteria toxic metabolites on freshwater ecosystems applying molecular methods. Environ. Sci. Pollut. Res. Int. 2023, 30, 219–227. [Google Scholar] [CrossRef]
- Weinberger, K.R.; Harris, D.; Spangler, K.R.; Zanobetti, A.; Wellenius, G.A. Estimating the number of excess deaths attributable to heat in 297 United States counties. Environ. Epidemiol. 2020, 4, e096. [Google Scholar] [CrossRef]
- IPCC-AR6 ASRCC. AR6 Synthesis Report: Climate Change 2023. 2021. Available online: https://www.ipcc.ch/2021/08/09/ar6-wg1-20210809-pr/ (accessed on 27 February 2023).
- Presbitero, A.; Melnikov, V.R.; Krzhizhanovskaya, V.V.; Sloot, P.M.A. A unifying model to estimate the effect of heat stress in the human innate immunity during physical activities. Sci. Rep. 2021, 11, 16688. [Google Scholar] [CrossRef]
- Riezman, H. Why do cells require heat shock proteins to survive heat stress? Cell. Cycle 2004, 3, 61–63. [Google Scholar] [CrossRef]
- Qu, Q.; Li, H.; Bai, L.; Zhang, S.; Sun, J.; Lv, W.; Ye, C.; Liu, C.; Shi, D. Effects of Heat Stress on Gut Microbiome in Rats. Indian J. Microbiol. 2021, 61, 338–347. [Google Scholar] [CrossRef]
- Shi, D.; Bai, L.; Qu, Q.; Zhou, S.; Yang, M.; Guo, S.; Li, Q.; Liu, C. Impact of gut microbiota structure in heat-stressed broilers. Poult. Sci. 2019, 98, 2405–2413. [Google Scholar] [CrossRef]
- Xiong, Y.; Yi, H.; Wu, Q.; Jiang, Z.; Wang, L. Effects of acute heat stress on intestinal microbiota in grow-finishing pigs, and associations with feed intake and serum profile. J. Appl. Microbiol. 2020, 128, 840–852. [Google Scholar] [CrossRef] [PubMed]
- Dokladny, K.; Moseley, P.L.; Ma, T.Y. Physiologically relevant increase in temperature causes an increase in intestinal epithelial tight junction permeability. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, G204–G212. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.C.; He, S.H.; Zheng, P.Y. Investigation into the signal transduction pathway via which heat stress impairs intestinal epithelial barrier function. J. Gastroenterol. Hepatol. 2007, 22, 1823–1831. [Google Scholar] [CrossRef] [PubMed]
- Karhausen, J.; Furuta, G.T.; Tomaszewski, J.E.; Johnson, R.S.; Colgan, S.P.; Haase, V.H. Epithelial hypoxia-inducible factor-1 is protective in murine experimental colitis. J. Clin. Investig. 2004, 114, 1098–1106. [Google Scholar] [CrossRef]
- Gobler, C.J. Climate Change and Harmful Algal Blooms: Insights and perspective. Harmful Algae 2020, 91, 101731. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Stegeman, J.J.; Fleming, L.E.; Allemand, D.; Anderson, D.M.; Backer, L.C.; Brucker-Davis, F.; Chevalier, N.; Corra, L.; Czerucka, D.; et al. Human Health and Ocean Pollution. Ann. Glob. Health 2020, 86, 151. [Google Scholar] [CrossRef]
- Cao, L.; Massey, I.Y.; Feng, H.; Yang, F. A Review of Cardiovascular Toxicity of Microcystins. Toxins 2019, 11, 507. [Google Scholar] [CrossRef]
- LeClaire, R.D.; Parker, G.W.; Franz, D.R. Hemodynamic and calorimetric changes induced by microcystin-LR in the rat. J. Appl. Toxicol. 1995, 15, 303–311. [Google Scholar] [CrossRef]
- Beasley, V.R.; Lovell, R.A.; Holmes, K.R.; Walcott, H.E.; Schaeffer, D.J.; Hoffmann, W.E.; Carmichael, W.W. Microcystin-LR decreases hepatic and renal perfusion, and causes circulatory shock, severe hypoglycemia, and terminal hyperkalemia in intravascularly dosed swine. J. Toxicol. Environ. Health A 2000, 61, 281–303. [Google Scholar] [CrossRef]
- Qiu, T.; Xie, P.; Liu, Y.; Li, G.; Xiong, Q.; Hao, L.; Li, H. The profound effects of microcystin on cardiac antioxidant enzymes, mitochondrial function and cardiac toxicity in rat. Toxicology 2009, 257, 86–94. [Google Scholar] [CrossRef]
- Zhang, Z.; Kang, S.; Chen, C.; Wei, G.; Yu, S. The acute toxic effects of microcystin LR in SD rats. Zhonghua Yu Fang Yi Xue Za Zhi 2002, 36, 295–297. [Google Scholar]
- Sozen, E.; Karademir, B.; Ozer, N.K. Basic mechanisms in endoplasmic reticulum stress and relation to cardiovascular diseases. Free Radic. Biol. Med. 2015, 78, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Ait Abderrahim, L.; Taibi, K.; Boussaid, M.; Al-Shara, B.; Ait Abderrahim, N.; Ait Abderrahim, S. Allium sativum mitigates oxidative damages induced by Microcystin-LR in heart and liver tissues of mice. Toxicon 2021, 200, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Hall, D.D.; Feekes, J.A.; Arachchige Don, A.S.; Shi, M.; Hamid, J.; Chen, L.; Strack, S.; Zamponi, G.W.; Horne, M.C.; Hell, J.W. Binding of protein phosphatase 2A to the L-type calcium channel Cav1.2 next to Ser1928, its main PKA site, is critical for Ser1928 dephosphorylation. Biochemistry 2006, 45, 3448–3459. [Google Scholar] [CrossRef]
- Kimura, T.; Han, W.; Pagel, P.; Nairn, A.C.; Caplan, M.J. Protein phosphatase 2A interacts with the Na,K-ATPase and modulates its trafficking by inhibition of its association with arrestin. PLoS ONE 2011, 6, e29269. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Zhou, J.; Zhang, M. Microcystins Induces Vascular Inflammation in Human Umbilical Vein Endothelial Cells via Activation of NF-kappaB. Mediat. Inflamm. 2015, 2015, 942159. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Deng, H.; Pan, H.; Xu, Y.; Zhang, M. Epigallocatechin-3-gallate attenuates microcystin-LR induced oxidative stress and inflammation in human umbilical vein endothelial cells. Chemosphere 2017, 168, 25–31. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2019. Diabetes Care 2018, 42 (Suppl. S1), S13–S28. [Google Scholar] [CrossRef]
- Belkina, A.C.; Denis, G.V. Obesity genes and insulin resistance. Curr. Opin. Endocrinol. Diabetes Obes. 2010, 17, 472–477. [Google Scholar] [CrossRef]
- Zhang, D.; Xie, P.; Liu, Y.; Qiu, T. Transfer, distribution and bioaccumulation of microcystins in the aquatic food web in Lake Taihu, China, with potential risks to human health. Sci. Total Environ. 2009, 407, 2191–2199. [Google Scholar] [CrossRef]
- Zhang, Q.; Qin, W.; Yang, L.; An, J.; Zhang, X.; Hong, H.; Xu, L.; Wang, Y. Microcystis bloom containing microcystin-LR induces type 2 diabetes mellitus. Toxicol. Lett. 2018, 294, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhao, Q.; Zhou, W.; Xu, L.; Wang, Y. Effects of chronic exposure to microcystin-LR on hepatocyte mitochondrial DNA replication in mice. Environ. Sci. Technol. 2015, 49, 4665–4672. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Cao, N.; Chen, K.; Wang, H.; Xu, L.; Xu, C.; Huang, P. Microcystin-LR exposure disrupts the insulin signaling pathway in C2C12 mice muscle cell line. Environ. Toxicol. 2020, 35, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Benedict, M.; Zhang, X. Non-alcoholic fatty liver disease: An expanded review. World J. Hepatol. 2017, 9, 715–732. [Google Scholar] [CrossRef] [PubMed]
- Delli Bovi, A.P.; Marciano, F.; Mandato, C.; Siano, M.A.; Savoia, M.; Vajro, P. Oxidative Stress in Non-alcoholic Fatty Liver Disease. An Updated Mini Review. Front. Med. 2021, 8, 595371. [Google Scholar] [CrossRef]
- Testart, J.; Lassalle, B.; Belaisch-Allart, J.; Forman, R.; Hazout, A.; Volante, M.; Frydman, R. Human embryo viability related to freezing and thawing procedures. Am. J. Obstet. Gynecol. 1987, 157, 168–171. [Google Scholar] [CrossRef]
- Carvalho-Gontijo, R.; Han, C.; Zhang, L.; Zhang, V.; Hosseini, M.; Mekeel, K.; Schnabl, B.; Loomba, R.; Karin, M.; Brenner, D.A.; et al. Metabolic Injury of Hepatocytes Promotes Progression of NAFLD and AALD. Semin. Liver Dis. 2022, 42, 233–249. [Google Scholar] [CrossRef]
- Nagata, K.; Suzuki, H.; Sakaguchi, S. Common pathogenic mechanism in development progression of liver injury caused by non-alcoholic or alcoholic steatohepatitis. J. Toxicol. Sci. 2007, 32, 453–468. [Google Scholar] [CrossRef]
- Seth, R.K.; Kumar, A.; Das, S.; Kadiiska, M.B.; Michelotti, G.; Diehl, A.M.; Chatterjee, S. Environmental toxin-linked nonalcoholic steatohepatitis and hepatic metabolic reprogramming in obese mice. Toxicol. Sci. 2013, 134, 291–303. [Google Scholar] [CrossRef]
- Marce, R.; George, G.; Buscarinu, P.; Deidda, M.; Dunalska, J.; de Eyto, E.; Flaim, G.; Grossart, H.-P.; Istvanovics, V.; Lenhardt, M.; et al. Automatic High Frequency Monitoring for Improved Lake and Reservoir Management. Environ. Sci. Technol. 2016, 50, 10780–10794. [Google Scholar] [CrossRef]
- Mishra, D.R.; Kumar, A.; Ramaswamy, L.; Boddula, V.K.; Das, M.C.; Page, B.P.; Weber, S.J. CyanoTRACKER: A cloud-based integrated multi-platform architecture for global observation of cyanobacterial harmful algal blooms. Harmful Algae 2020, 96, 101828. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Holbach, A.; Wilhelms, A.; Qin, Y.; Zheng, B.; Zou, H.; Qin, B.; Zhu, G.; Norra, S. Highly time-resolved analysis of seasonal water dynamics and algal kinetics based on in-situ multi-sensor-system monitoring data in Lake Taihu, China. Sci. Total Environ. 2019, 660, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Mozo, A.; Moron-Lopez, J.; Vakaruk, S.; Pompa-Pernia, A.G.; Gonzalez-Prieto, A.; Aguilar, J.A.P.; Gómez-Canaval, S.; Ortiz, J.M. Chlorophyll soft-sensor based on machine learning models for algal bloom predictions. Sci. Rep. 2022, 12, 13529. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Wang, S.; Zhan, Y.; Kai, T.; Ding, P. Sensitive Identification of Microcystin-LR via a Reagent-Free and Reusable Electrochemical Biosensor Using a Methylene Blue-Labeled Aptamer. Biosensors 2022, 12, 556. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.; Zhu, H.; Wang, J.; Hou, S.; Shutes, B.; Yan, B. Removal of microcystin (MC-LR) in constructed wetlands integrated with microbial fuel cells: Efficiency, bioelectricity generation and microbial response. J. Environ. Manag. 2022, 309, 114669. [Google Scholar] [CrossRef] [PubMed]
- Meriluoto, J.; Gueimonde, M.; Haskard, C.A.; Spoof, L.; Sjovall, O.; Salminen, S. Removal of the cyanobacterial toxin microcystin-LR by human probiotics. Toxicon 2005, 46, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Nybom, S.M.; Salminen, S.J.; Meriluoto, J.A. Specific strains of probiotic bacteria are efficient in removal of several different cyanobacterial toxins from solution. Toxicon 2008, 52, 214–220. [Google Scholar] [CrossRef]
| Country | Water Body | MC-LR Concentration | Reference |
|---|---|---|---|
| Brazil | Cascata water reservoir | ~3 µg/L | [55] |
| Guarapiranga water reservoir | ~1 µg/L | [55] | |
| Amazon River | ~1.4 µg/L | [59] | |
| Canada | Quebec water reservoir | 2.47 µg/L | [63] |
| Nigeria | Kubanni | 2.8 µg/L | [64] |
| Bomo | 1.8 µg/L | [64] | |
| Eastern Cuba | 24 water reservoir | 3.06 µg/L (in 5 lakes that exceeded WHO limits) | [57] |
| Morocco | Mansour Eddahbi lake | 64.4 µg/g dry biomass weight | [61] |
| Almassira lake | 9.9 µg/g dry biomass weight | [61] |
| Target Organ | Model | Exposure | Observed Changes | Reference |
|---|---|---|---|---|
| Liver | Various | Various | Hepatic inflammation; activation of Kupffer cells and hepatic stellate cells; centrilobular apoptosis and necrosis; cytoplasmic vacuolization; cytoskeletal reorganization; hepatocyte blebbing; hepatocarcinogenesis. | [115,116,117], https://doi.org/10.1293/tox.14.259 (accessed on 10 April 2023), [118] |
| Kidney | Adult male Wistar rats | Chronic, sub-lethal dose, administered intraperitoneally | Glomerular collapse with thickening of the basement membrane; renal tubules showed actin condensation, appeared dilated and filled with proteinaceous casts; increased apoptotic cells in renal cortex and medulla. | [119] |
| Brain | Adult male BALB/c mice | Chronic sub-lethal dose, administered orally via drinking water | Disruption of blood–brain barrier function; neuroinflammation; microglial and astrocyte activation. | [120] |
| Lung | Specific-pathogen-free female BALB/C mice | Chronic low dose, administered orally in drinking water | Alveolar collapse and thickening of alveolar septum; slight increase cellular apoptosis at a higher dose; loss of epithelial barrier function. | [121] |
| Heart | Adult Wistar rats | Chronic | Increased size of cardiomyocytes; reduced myofibril volume fraction; fibrosis; mononuclear infiltration in interstitial spaces. | [122] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chatterjee, S.; More, M. Cyanobacterial Harmful Algal Bloom Toxin Microcystin and Increased Vibrio Occurrence as Climate-Change-Induced Biological Co-Stressors: Exposure and Disease Outcomes via Their Interaction with Gut–Liver–Brain Axis. Toxins 2023, 15, 289. https://doi.org/10.3390/toxins15040289
Chatterjee S, More M. Cyanobacterial Harmful Algal Bloom Toxin Microcystin and Increased Vibrio Occurrence as Climate-Change-Induced Biological Co-Stressors: Exposure and Disease Outcomes via Their Interaction with Gut–Liver–Brain Axis. Toxins. 2023; 15(4):289. https://doi.org/10.3390/toxins15040289
Chicago/Turabian StyleChatterjee, Saurabh, and Madhura More. 2023. "Cyanobacterial Harmful Algal Bloom Toxin Microcystin and Increased Vibrio Occurrence as Climate-Change-Induced Biological Co-Stressors: Exposure and Disease Outcomes via Their Interaction with Gut–Liver–Brain Axis" Toxins 15, no. 4: 289. https://doi.org/10.3390/toxins15040289
APA StyleChatterjee, S., & More, M. (2023). Cyanobacterial Harmful Algal Bloom Toxin Microcystin and Increased Vibrio Occurrence as Climate-Change-Induced Biological Co-Stressors: Exposure and Disease Outcomes via Their Interaction with Gut–Liver–Brain Axis. Toxins, 15(4), 289. https://doi.org/10.3390/toxins15040289






