Fungal Contamination of Building Materials and the Aerosolization of Particles and Toxins in Indoor Air and Their Associated Risks to Health: A Review
Abstract
1. Introduction: Importance of Moulds as Indoor Contaminants
2. Aerosolization of Moulds Particles from Contamination Materials
2.1. The Mechanism of Aerosolization and Characteristics of Airborne Particles
2.2. External Factors Impacting the Release of Bioaerosols
3. Microbial Species Identified on Indoor Building Materials
- Studies focused on accessible surfaces in buildings;
- The investigations of fungal development focused on surface sampling;
- Fungal identification was carried out at the species level.
3.1. Ecological Characteristics of the Most Detected Species
3.2. Sampling Methods to Analyze the Fungal Contamination of Building Materials
3.3. Analysis Methods to Investigate Fungal Diversity of Building Materials
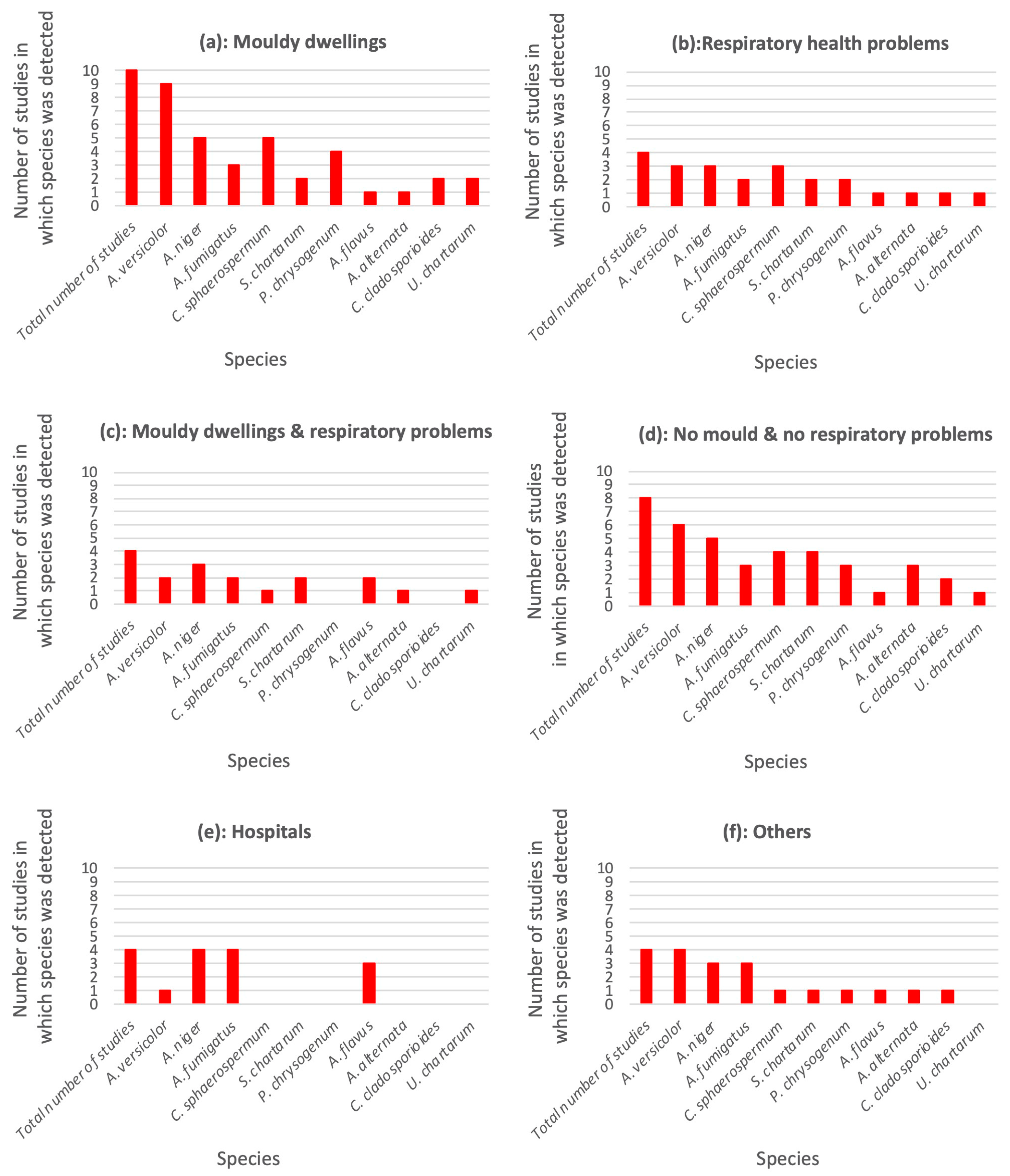
3.4. Species Identification as a Function of Building Type
3.5. Variation of Species Detection between Europe and Northern America
3.6. Quantification of Fungal Particles on Surfaces
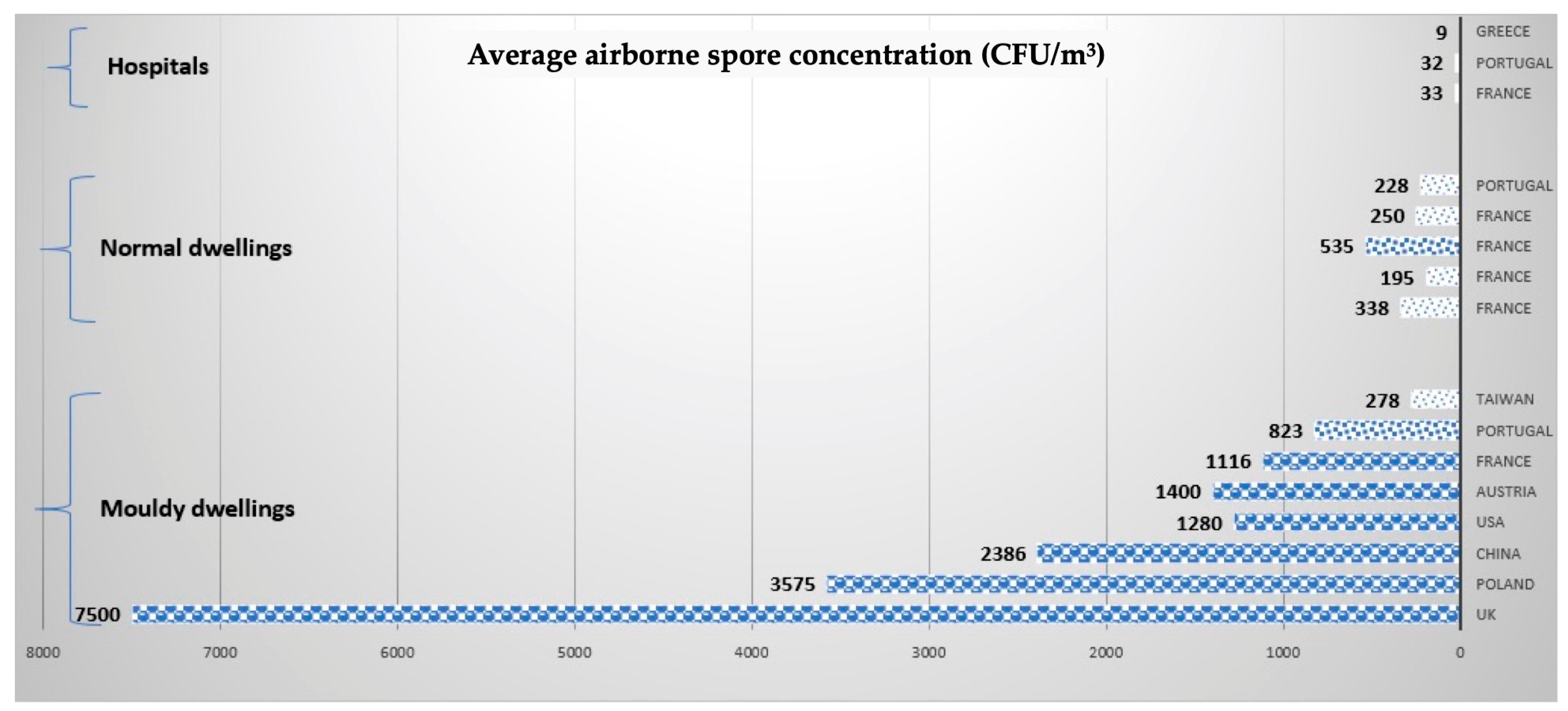
4. Comparison between Indoor Material and Indoor Air Contaminations
Quantification of Airborne Contamination by Fungal Particles
5. Possible Consequences on Human Health of Fungal Particles Release in Indoor Air
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ayanbimpe, G.M.; Danjuma, W.S.; Okolo, M.O. Relationship between Fungal Contamination of Indoor Air and Health Problems of Some Residents in Jos; IntechOpen: London, UK, 2012. [Google Scholar]
- Haleem Khan, A.A.; Mohan Karuppayil, S. Fungal Pollution of Indoor Environments and Its Management. Saudi J. Biol. Sci. 2012, 19, 405–426. [Google Scholar] [CrossRef] [PubMed]
- Verdier, T.; Coutand, M.; Bertron, A.; Roques, C. A Review of Indoor Microbial Growth across Building Materials and Sampling and Analysis Methods. Build. Environ. 2014, 80, 136–149. [Google Scholar] [CrossRef]
- Samson, R.A.; Houbraken, J.; Thrane, U.; Frisvad, J.C.; Andersen, B. Food and Indoor Fungi; CBS Laboratory Manual Series 2; CBS-Fungal Biodiversity Centre: Utrecht, The Netherlands, 2010. [Google Scholar]
- Lee, J.E.; Lee, B.U.; Bae, G.N.; Jung, J.H. Evaluation of Aerosolization Characteristics of Biocontaminated Particles from Flood-Damaged Housing Materials. J. Aerosol Sci. 2017, 106, 93–99. [Google Scholar] [CrossRef]
- Abdel Hameed, A.A.; Khoder, M.I.; Ibrahim, Y.H.; Saeed, Y.; Osman, M.E.; Ghanem, S. Study on Some Factors Affecting Survivability of Airborne Fungi. Sci. Total Environ. 2012, 414, 696–700. [Google Scholar] [CrossRef]
- Sanmartín, P.; Miller, A.Z.; Prieto, B.; Viles, H.A. Revisiting and Reanalysing the Concept of Bioreceptivity 25 Years On. Sci. Total Environ. 2021, 770, 145314. [Google Scholar] [CrossRef]
- Shirakawa, M.A.; Selmo, S.M.; Cincotto, M.A.; Gaylarde, C.C.; Brazolin, S.; Gambale, W. Susceptibility of Phosphogypsum to Fungal Growth and the Effect of Various Biocides. Int. Biodeterior. Biodegrad. 2002, 49, 293–298. [Google Scholar] [CrossRef]
- Shoemaker, R.C.; House, D.E. A Time-Series Study of Sick Building Syndrome: Chronic, Biotoxin-Associated Illness from Exposure to Water-Damaged Buildings. Neurotoxicol. Teratol. 2005, 27, 29–46. [Google Scholar] [CrossRef]
- Soulios, V.; Jan de Place Hansen, E.; Peuhkuri, R. Hygrothermal Performance of Hydrophobized and Internally Insulated Masonry Walls—Simulating the Impact of Hydrophobization Based on Experimental Results. Build. Environ. 2021, 187, 107410. [Google Scholar] [CrossRef]
- Nielsen, K.F. Mycotoxin Production by Indoor Molds. Fungal Genet. Biol. 2003, 39, 103–117. [Google Scholar] [CrossRef]
- Platt, S.D.; Martin, C.J.; Hunt, S.M.; Lewis, C.W. Damp Housing, Mould Growth, and Symptomatic Health State. BMJ 1989, 298, 1673–1678. [Google Scholar] [CrossRef] [PubMed]
- Gollogly, L. World Health Statistics 2009; World Health Organization: Geneva, Switzerland, 2009; ISBN 92-4-156381-8.
- Bernstein, J.A.; Alexis, N.; Bacchus, H.; Bernstein, I.L.; Fritz, P.; Horner, E.; Li, N.; Mason, S.; Nel, A.; Oullette, J.; et al. The Health Effects of Nonindustrial Indoor Air Pollution. J. Allergy Clin. Immunol. 2008, 121, 585–591. [Google Scholar] [CrossRef]
- World Health Organization. WHO Guidelines for Indoor Air Quality: Dampness and Mould; WHO Regional Office Europe: Copenhagen, Denmark, 2009; ISBN 978-92-890-4168-3.
- Srikanth, P.; Sudharsanam, S.; Steinberg, R. Bio-aerosols in indoor environment: Composition, health effects and analysis. Indian J. Med. Microbiol. 2008, 26, 302–312. [Google Scholar] [CrossRef]
- Douwes, J.; Thorne, P.; Pearce, N.; Heederick, D. Bioaerosol Health Effects and Exposure Assessment: Progress and Prospects. Ann. Occup. Hyg. 2003, 47, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Jaenicke, R. Abundance of Cellular Material and Proteins in the Atmosphere. Science 2005, 308, 73. [Google Scholar] [CrossRef] [PubMed]
- Humbal, C.; Gautam, S.; Trivedi, U. A Review on Recent Progress in Observations, and Health Effects of Bioaerosols. Environ. Int. 2018, 118, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Kabir, E.; Jahan, S.A. Airborne Bioaerosols and Their Impact on Human Health. J. Environ. Sci. 2018, 67, 23–35. [Google Scholar] [CrossRef]
- Manisalidis, I.; Stavropoulou, E.; Stavropoulos, A.; Bezirtzoglou, E. Environmental and Health Impacts of Air Pollution: A Review. Front. Public Health 2020, 8, 14. [Google Scholar] [CrossRef]
- Buzea, C.; Pacheco, I.I.; Robbie, K. Nanomaterials and Nanoparticles: Sources and Toxicity. Biointerphases 2007, 2, MR17–MR71. [Google Scholar] [CrossRef]
- Baxi, S.N.; Portnoy, J.M.; Larenas-Linnemann, D.; Phipatanakul, W.; Barnes, C.; Baxi, S.; Grimes, C.; Horner, W.E.; Kennedy, K.; Larenas-Linnemann, D.; et al. Exposure and Health Effects of Fungi on Humans. J. Allergy Clin. Immunol. Pract. 2016, 4, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Dales, R.E.; Zwanenburg, H.; Burnett, R.; Franklin, C.A. Respiratory Health Effects of Home Dampness and Molds among Canadian Children. Am. J. Epidemiol. 1991, 134, 196–203. [Google Scholar] [CrossRef]
- Cooley, J.D.; Wong, W.C.; Jumper, C.A.; Straus, D.C. Correlation between the Prevalence of Certain Fungi and Sick Building Syndrome. Occup. Environ. Med. 1998, 55, 579–584. [Google Scholar] [CrossRef]
- Bush, R.K.; Portnoy, J.M.; Saxon, A.; Terr, A.I.; Wood, R.A. The Medical Effects of Mold Exposure. J. Allergy Clin. Immunol. 2006, 117, 326–333. [Google Scholar] [CrossRef]
- ANSES. Moisissures dans le Bâti; ANSES: Paris, France, 2016.
- Mokobi, F. Aspergillus flavus—An Overview. Available online: https://microbenotes.com/aspergillus-flavus/ (accessed on 10 February 2023).
- Krishnan, S.; Manavathu, E.K.; Chandrasekar, P.H. Aspergillus flavus: An Emerging Non-Fumigatus Aspergillus Species of Significance. Mycoses 2009, 52, 206–222. [Google Scholar] [CrossRef]
- Hedayati, M.T.; Pasqualotto, A.C.; Warn, P.A.; Bowyer, P.; Denning, D.W. Aspergillus flavus: Human Pathogen, Allergen and Mycotoxin Producer. Microbiology 2007, 153, 1677–1692. [Google Scholar] [CrossRef]
- Mokobi, F. Aspergillus fumigatus—An Overview. Available online: https://microbenotes.com/aspergillus-fumigatus/ (accessed on 10 February 2023).
- Kwon-Chung, K.J.; Sugui, J.A. Aspergillus fumigatus—What Makes the Species a Ubiquitous Human Fungal Pathogen? PLoS Pathog. 2013, 9, e1003743. [Google Scholar] [CrossRef] [PubMed]
- Latgé, J.-P. Aspergillus fumigatus, a Saprotrophic Pathogenic Fungus. Mycologist 2003, 17, 56–61. [Google Scholar] [CrossRef]
- Howard, D.H. Pathogenic Fungi in Humans and Animals; CRC Press: Boca Raton, FL, USA, 2002; ISBN 978-0-203-90910-2. [Google Scholar]
- Weryszko-Chmielewska, E.; Kasprzyk, I.; Nowak, M.; Sulborska, A.; Kaczmarek, J.; Szymanska, A.; Haratym, W.; Gilski, M.; Jedryczka, M. Health Hazards Related to Conidia of Cladosporium—Biological Air Pollutants in Poland, Central Europe. J. Environ. Sci. 2018, 65, 271–281. [Google Scholar] [CrossRef]
- AlMatar, M.; Makky, E.A. Cladosporium cladosporioides from the Perspectives of Medical and Biotechnological Approaches. 3 Biotech 2016, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Mok, T.; Koehler, A.P.; Yu, M.Y.; Ellis, D.H.; Johnson, P.J.; Wickham, N.W. Fatal Penicillium citrinum Pneumonia with Pericarditis in a Patient with Acute Leukemia. J. Clin. Microbiol. 1997, 35, 2654–2656. [Google Scholar] [CrossRef]
- Caillaud, D.; Annesi-Maesano, I.; Bennedjai, N.; Bex, V.; Blay, F.; Chappin, D.; Dalphin, J.; Fabre, C.; Garans, M. Contaminations Fongiques En Milieux Intérieurs Diagnostic Effets Sur La Sante Respiratoire: Conduites à Tenir; Conseil Supérieur D’hygiène Publique de France: Paris, France, 2006. [Google Scholar]
- Sánchez Espinosa, K.C.; Rojas Flores, T.I.; Davydenko, S.R.; Venero Fernández, S.J.; Almaguer, M. Fungal Populations in the Bedroom Dust of Children in Havana, Cuba, and Its Relationship with Environmental Conditions. Environ. Sci. Pollut. Res. 2021, 28, 53010–53020. [Google Scholar] [CrossRef]
- Chegini, F.M.; Baghani, A.N.; Hassanvand, M.S.; Sorooshian, A.; Golbaz, S.; Bakhtiari, R.; Ashouri, A.; Joubani, M.N.; Alimohammadi, M. Indoor and Outdoor Airborne Bacterial and Fungal Air Quality in Kindergartens: Seasonal Distribution, Genera, Levels, and Factors Influencing Their Concentration. Build. Environ. 2020, 175, 106690. [Google Scholar] [CrossRef]
- Mensah-Attipoe, J.; Toyinbo, O. Fungal Growth and Aerosolization from Various Conditions and Materials. In Fungal Infection; IntechOpen: London, UK, 2019. [Google Scholar]
- Paul, B.; Spooner, B. Soil Fungi: Diversity and Detection. Plant Soil 2001, 232, 147–154. [Google Scholar]
- Corsi, R.L.; Siegel, J.A.; Chiang, C. Particle Resuspension During the Use of Vacuum Cleaners on Residential Carpet. J. Occup. Environ. Hyg. 2008, 5, 232–238. [Google Scholar] [CrossRef]
- Aleksic, B.; Draghi, M.; Ritoux, S.; Bailly, S.; Lacroix, M.; Oswald, I.P.; Bailly, J.-D.; Robine, E. Aerosolization of Mycotoxins after Growth of Toxinogenic Fungi on Wallpaper. Appl. Environ. Microbiol. 2017, 83, e01001-17. [Google Scholar] [CrossRef] [PubMed]
- Morey, P.R. Microbiological Sampling Strategies in Indoor Environments. In Sampling and Analysis of Indoor Microorganisms; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2007; pp. 51–74. ISBN 978-0-470-11243-4. [Google Scholar]
- Santa Izabel, T.; Cruz, A.; Barbosa, F.; Leão-Ferreira, S.; Marques, M.; Gusmao, L. The Genus Stachybotrys (Anamorphic Fungi) in the Semi-Arid Region of Brazil. Rev. Bras. Bot. 2010, 33, 479–487. [Google Scholar] [CrossRef]
- Górny, R.L.; Reponen, T.; Willeke, K.; Schmechel, D.; Robine, E.; Boissier, M.; Grinshpun, S.A. Fungal Fragments as Indoor Air Biocontaminants. Appl. Environ. Microbiol. 2002, 68, 3522–3531. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.-H.; Seo, S.-C.; Schmechel, D.; Grinshpun, S.A.; Reponen, T. Aerodynamic Characteristics and Respiratory Deposition of Fungal Fragments. Atmos. Environ. 2005, 39, 5454–5465. [Google Scholar] [CrossRef]
- Reponen, T.; Seo, S.-C.; Grimsley, F.; Lee, T.; Crawford, C.; Grinshpun, S.A. Fungal Fragments in Moldy Houses: A Field Study in Homes in New Orleans and Southern Ohio. Atmos. Environ. 2007, 41, 8140–8149. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Concentration and Exposure-Response Measurement of Fine and Ultra Fine Particulate Matter for Use in Epidemiological Studies; World Health Organization: Geneva, Switzerland, 2002.
- Górny, R.L. Microbial Aerosols: Sources, Properties, Health Effects, Exposure Assessment—A Review. KONA Powder Part. J. 2020, 37, 64–84. [Google Scholar] [CrossRef]
- Chen, B.T.; Fletcher, R.A.; Cheng, Y.-S.; Kulkarni, P.; Baron, A.P.; Willeke, K. Calibration of Aerosol Instruments. In Aerosol Measurements: Principles, Techniques, and Applications, 3rd ed.; John Wiley and Sons: Hoboken, NJ, USA, 2011; pp. 449–478. [Google Scholar]
- Costa, A.; Pinheiro, M.; Magalhaes, J.; Ribeiro, R.; Seabra, V.; Reis, S.; Sarmento, B. The Formulation of Nanomedicines for Treating Tuberculosis. Adv. Drug Deliv. Rev. 2016, 102, 102–115. [Google Scholar] [CrossRef]
- Kim, K.-H.; Kabir, E.; Kabir, S. A Review on the Human Health Impact of Airborne Particulate Matter. Environ. Int. 2015, 74, 136–143. [Google Scholar] [CrossRef]
- Górny, R.; Dutkiewicz, J. Bacterial and Fungal Aerosols in Indoor Environment in Central and Eastern European Countries. Ann. Agric. Environ. Med. 2003, 9, 17–23. [Google Scholar]
- Moularat, S.; Robine, E. A Method to Determine the Transfer of Mycotoxins from Materials to Air. CLEAN Soil Air Water 2008, 36, 578–583. [Google Scholar] [CrossRef]
- Frankel, M.; Hansen, E.W.; Madsen, A.M. Effect of Relative Humidity on the Aerosolization and Total Inflammatory Potential of Fungal Particles from Dust-Inoculated Gypsum Boards. Indoor Air 2014, 24, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Money, N.P. Chapter 3—Spore Production, Discharge, and Dispersal. In The Fungi, 3rd ed.; Watkinson, S.C., Boddy, L., Money, N.P., Eds.; Academic Press: Boston, MA, USA, 2016; pp. 67–97. ISBN 978-0-12-382034-1. [Google Scholar]
- Madsen, A.M. Effects of Airflow and Changing Humidity on the Aerosolization of Respirable Fungal Fragments and Conidia of Botrytis cinerea. Appl. Environ. Microbiol. 2012, 78, 3999–4007. [Google Scholar] [CrossRef] [PubMed]
- Pasanen, A.-L.; Pasanen, P.; Jantunen, M.J.; Kalliokoski, P. Significance of Air Humidity and Air Velocity for Fungal Spore Release into the Air. Atmos. Environ. Part A Gen. Top. 1991, 25, 459–462. [Google Scholar] [CrossRef]
- Timm, M.; Hansen, E.W.; Moesby, L.; Christensen, J.D. Utilization of the Human Cell Line HL-60 for Chemiluminescence Based Detection of Microorganisms and Related Substances. Eur. J. Pharm. Sci. 2006, 27, 252–258. [Google Scholar] [CrossRef]
- Frankel, M.; Bekö, G.; Timm, M.; Gustavsen, S.; Hansen, E.W.; Madsen, A.M. Seasonal Variations of Indoor Microbial Exposures and Their Relation to Temperature, Relative Humidity, and Air Exchange Rate. Appl. Environ. Microbiol. 2012, 78, 8289–8297. [Google Scholar] [CrossRef] [PubMed]
- Viegas, C.; Pimenta, R.; Dias, M.; Gomes, B.; Brito, M.; Aranha Caetano, L.; Carolino, E.; Gomes, A.Q. Microbiological Contamination Assessment in Higher Education Institutes. Atmosphere 2021, 12, 1079. [Google Scholar] [CrossRef]
- Viegas, C.; Almeida-Silva, M.; Gomes, A.Q.; Wolterbeek, H.T.; Almeida, S.M. Fungal Contamination Assessment in Portuguese Elderly Care Centers. J. Toxicol. Environ. Health Part A 2014, 77, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Parracha, J.L.; Borsoi, G.; Flores-Colen, I.; Veiga, R.; Nunes, L.; Dionísio, A.; Gomes, M.G.; Faria, P. Performance Parameters of ETICS: Correlating Water Resistance, Bio-Susceptibility and Surface Properties. Constr. Build. Mater. 2021, 272, 121956. [Google Scholar] [CrossRef]
- Švajlenka, J.; Kozlovská, M.; Pošiváková, T. Assessment and Biomonitoring Indoor Environment of Buildings. Int. J. Environ. Health Res. 2017, 27, 427–439. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.I.; Sylvain, I.; Spilak, M.P.; Taylor, J.W.; Waring, M.S.; Mendell, M.J. Fungal Signature of Moisture Damage in Buildings: Identification by Targeted and Untargeted Approaches with Mycobiome Data. Appl. Environ. Microbiol. 2020, 86, e01047-20. [Google Scholar] [CrossRef]
- Pereira, C.; de Brito, J.; Silvestre, J.D. Contribution of Humidity to the Degradation of Façade Claddings in Current Buildings. Eng. Fail. Anal. 2018, 90, 103–115. [Google Scholar] [CrossRef]
- Heseltine, E.; Rosen, J. WHO Guidelines for Indoor Air Quality: Dampness and Mould; World Health Organization: Geneva, Switzerland, 2009.
- Borrego, S.; Molina, A. Fungal Assessment on Storerooms Indoor Environment in the National Museum of Fine Arts, Cuba. Air Qual. Atmos. Health 2019, 12, 1373–1385. [Google Scholar] [CrossRef]
- Nevalainen, A.; Täubel, M.; Hyvärinen, A. Indoor Fungi: Companions and Contaminants. Indoor Air 2015, 25, 125–156. [Google Scholar] [CrossRef]
- Reboux, G.; Rocchi, S.; Laboissière, A.; Ammari, H.; Bochaton, M.; Gardin, G.; Rame, J.-M.; Millon, L. Survey of 1012 Moldy Dwellings by Culture Fungal Analysis: Threshold Proposal for Asthmatic Patient Management. Indoor Air 2019, 29, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Boutin-Forzano, S.; Charpin-Kadouch, C.; Chabbi, S.; Bennedjai, N.; Dumon, H.; Charpin, D. Wall Relative Humidity: A Simple and Reliable Index for Predicting Stachybotrys Chartarum Infestation in Dwellings. Indoor Air 2004, 14, 196–199. [Google Scholar] [CrossRef]
- Polizzi, V.; Adams, A.; De Saeger, S.; Van Peteghem, C.; Moretti, A.; De Kimpe, N. Influence of Various Growth Parameters on Fungal Growth and Volatile Metabolite Production by Indoor Molds. Sci. Total Environ. 2012, 414, 277–286. [Google Scholar] [CrossRef]
- Bellanger, A.-P.; Reboux, G.; Roussel, S.; Grenouillet, F.; Didier-Scherer, E.; Dalphin, J.-C.; Millon, L. Indoor Fungal Contamination of Moisture-Damaged and Allergic Patient Housing Analysed Using Real-Time PCR. Lett. Appl. Microbiol. 2009, 49, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Reboux, G.; Bellanger, A.P.; Roussel, S.; Grenouillet, F.; Sornin, S.; Piarroux, R.; Dalphin, J.C.; Millon, L. Indoor Mold Concentration in Eastern France. Indoor Air 2009, 19, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Gutarowska, B.; Sulyok, M.; Krska, R. A Study of the Toxicity of Moulds Isolated from Dwellings. Indoor Built Environ. 2010, 19, 668–675. [Google Scholar] [CrossRef]
- Andersen, B.; Frisvad, J.C.; Søndergaard, I.; Rasmussen, I.S.; Larsen, L.S. Associations between Fungal Species and Water-Damaged Building Materials. Appl. Environ. Microbiol. 2011, 77, 4180–4188. [Google Scholar] [CrossRef] [PubMed]
- Jeżak, K.; Kozajda, A.; Sowiak, M.; Brzeźnicki, S.; Bonczarowska, M.; Szadkowska-Stańczyk, I. The Capability of Fungi Isolated from Moldy Dwellings to Produce Toxins. Int. J. Occup. Med. Environ. Health 2016, 29, 823–836. [Google Scholar] [CrossRef]
- Hegarty, B.; Haverinen-Shaughnessy, U.; Shaughnessy, R.J.; Peccia, J. Spatial Gradients of Fungal Abundance and Ecology throughout a Damp Building. Environ. Sci. Technol. Lett. 2019, 6, 329–333. [Google Scholar] [CrossRef]
- Hegarty, B.; Pan, A.; Haverinen-Shaughnessy, U.; Shaughnessy, R.; Peccia, J. DNA Sequence-Based Approach for Classifying the Mold Status of Buildings. Environ. Sci. Technol. 2020, 54, 15968–15975. [Google Scholar] [CrossRef] [PubMed]
- Andersen, B.; Frisvad, J.C.; Dunn, R.R.; Thrane, U. A Pilot Study on Baseline Fungi and Moisture Indicator Fungi in Danish Homes. J. Fungi 2021, 7, 71. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, V.; Schleiner, T.; Maier, U.; Fels, H.; Cramer, B.; Humpf, H.-U. Analysis of Mold and Mycotoxins in Naturally Infested Indoor Building Materials. Mycotoxin Res. 2022, 38, 205–220. [Google Scholar] [CrossRef]
- Rivier, A.; Guillaso, M.; Flabbée, J.A. Contamination fongique de l’habitat Lorrain: Enquête préliminaire au domicile des patients. Rev. Fr. Allergol. 2014, 54, 44–50. [Google Scholar] [CrossRef]
- Trout, D.; Bernstein, J.; Martinez, K.; Biagini, R.; Wallingford, K. Bioaerosol Lung Damage in a Worker with Repeated Exposure to Fungi in a Water-Damaged Building. Environ. Health Perspect. 2001, 109, 641–644. [Google Scholar] [CrossRef]
- Polizzi, V.; Adams, A.; Malysheva, S.V.; De Saeger, S.; Van Peteghem, C.; Moretti, A.; Picco, A.M.; De Kimpe, N. Identification of Volatile Markers for Indoor Fungal Growth and Chemotaxonomic Classification of Aspergillus Species. Fungal Biol. 2012, 116, 941–953. [Google Scholar] [CrossRef]
- Spurgeon, J.C. A Method for Detecting Fungal Contaminants in Wall Cavities. AIHA J. 2003, 64, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Kazemian, N.; Pakpour, S.; Milani, A.S.; Klironomos, J. Environmental Factors Influencing Fungal Growth on Gypsum Boards and Their Structural Biodeterioration: A University Campus Case Study. PLoS ONE 2019, 14, e0220556. [Google Scholar] [CrossRef] [PubMed]
- Alberti, C.; Bouakline, A.; Ribaud, P.; Lacroix, C.; Rousselot, P.; Leblanc, T.; Derouin, F. Relationship between Environmental Fungal Contamination and the Incidence of Invasive Aspergillosis in Haematology Patients. J. Hosp. Infect. 2001, 48, 198–206. [Google Scholar] [CrossRef]
- Panagopoulou, P.; Filioti, J.; Petrikkos, G.; Giakouppi, P.; Anatoliotaki, M.; Farmaki, E.; Kanta, A.; Apostolakou, H.; Avlami, A.; Samonis, G.; et al. Environmental Surveillance of Filamentous Fungi in Three Tertiary Care Hospitals in Greece. J. Hosp. Infect. 2002, 52, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Sixt, N.; Dalle, F.; Lafon, I.; Aho, S.; Couillault, G.; Valot, S.; Calinon, C.; Danaire, V.; Vagner, O.; Cuisenier, B.; et al. Reduced Fungal Contamination of the Indoor Environment with the PlasmairTM System (Airinspace). J. Hosp. Infect. 2007, 65, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Haas, D.; Galler, H.; Habib, J.; Melkes, A.; Schlacher, R.; Buzina, W.; Friedl, H.; Marth, E.; Reinthaler, F.F. Concentrations of Viable Airborne Fungal Spores and Trichloroanisole in Wine Cellars. Int. J. Food Microbiol. 2010, 144, 126–132. [Google Scholar] [CrossRef]
- Pinheiro, A.C.; Macedo, M.F.; Jurado, V.; Saiz-Jimenez, C.; Viegas, C.; Brandão, J.; Rosado, L. Mould and Yeast Identification in Archival Settings: Preliminary Results on the Use of Traditional Methods and Molecular Biology Options in Portuguese Archives. Int. Biodeterior. Biodegrad. 2011, 65, 619–627. [Google Scholar] [CrossRef]
- Gutarowska, B.; Skora, J.; Zduniak, K.; Rembisz, D. Analysis of the Sensitivity of Microorganisms Contaminating Museums and Archives to Silver Nanoparticles. Int. Biodeterior. Biodegrad. 2012, 68, 7–17. [Google Scholar] [CrossRef]
- Paiva de Carvalho, H.; Mesquita, N.; Trovão, J.; Fernández Rodríguez, S.; Pinheiro, A.C.; Gomes, V.; Alcoforado, A.; Gil, F.; Portugal, A. Fungal Contamination of Paintings and Wooden Sculptures inside the Storage Room of a Museum: Are Current Norms and Reference Values Adequate? J. Cult. Herit. 2018, 34, 268–276. [Google Scholar] [CrossRef]
- Mokobi, F. Aspergillus niger—An Overview. Available online: https://microbenotes.com/aspergillus-niger/ (accessed on 10 February 2023).
- Samson, R.A.; Hoekstra, E.S.; Frisvad, J.C. Introduction to Food- and Airborne Fungi; Centraalbureau voor Schimmelcultures: Utrecht, The Netherlands, 2004. [Google Scholar]
- Walker, G.M.; White, N.A. Introduction to Fungal Physiology. In Fungi; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 1–35. ISBN 978-1-119-37431-2. [Google Scholar]
- Jackson, S.A.; Dobson, A.D.W. Yeasts and Molds: Aspergillus flavus. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 978-0-08-100596-5. [Google Scholar]
- Fomicheva, G.M.; Vasilenko, O.V.; Marfenina, O.E. Comparative Morphological, Ecological, and Molecular Studies of Aspergillus versicolor (Vuill.) Tiraboschi Strains Isolated from Different Ecotopes. Microbiology 2006, 75, 186–191. [Google Scholar] [CrossRef]
- Pasanen, P.; Korpi, A.; Kalliokoski, P.; Pasanen, A.-L. Growth and Volatile Metabolite Production of Aspergillus versicolor in House Dust. Environ. Int. 1997, 23, 425–432. [Google Scholar] [CrossRef]
- Robert, V.; Vu, D.; Amor, A.B.H.; van de Wiele, N.; Brouwer, C.; Jabas, B.; Szoke, S.; Dridi, A.; Triki, M.; ben Daoud, S.; et al. MycoBank Gearing up for New Horizons. IMA Fungus 2013, 4, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Astoreca, A.; Magnoli, C.; Ramirez, M.; Combina, M.; Dalcero, A. Water Activity and Temperature Effects on Growth of Aspergillus niger, A. awamori and A. carbonarius Isolated from Different Substrates in Argentina. Int. J. Food Microbiol. 2007, 119, 314–318. [Google Scholar] [CrossRef]
- Alonso, V.; Cavaglieri, L.; Ramos, A.J.; Torres, A.; Marin, S. Modelling the Effect of PH and Water Activity in the Growth of Aspergillus fumigatus Isolated from Corn Silage. J. Appl. Microbiol. 2017, 122, 1048–1056. [Google Scholar] [CrossRef]
- Hocking, A.D.; Miscamble, B.F.; Pitt, J.I. Water Relations of Alternaria alternata, Cladosporium cladosporioides, Cladosporium sphaerospermum, Curvularia lunata and Curvularia pallescens. Mycol. Res. 1994, 98, 91–94. [Google Scholar] [CrossRef]
- Zalar, P.; de Hoog, G.S.; Schroers, H.-J.; Crous, P.W.; Groenewald, J.Z.; Gunde-Cimerman, N. Phylogeny and Ecology of the Ubiquitous Saprobe Cladosporium sphaerospermum, with Descriptions of Seven New Species from Hypersaline Environments. Stud. Mycol. 2007, 58, 157–183. [Google Scholar] [CrossRef]
- Sergey, N.E.; Petrunina, Y.V.; Likhachev, A.N. Growth of Stachybotrys chartarum Strains on Natural and Artificial Substrates. Bot. Lith. 2003, 9, 171–177. [Google Scholar]
- Barceloux, D.G. Medical Toxicology of Natural Substances: Foods, Fungi, Medicinal Herbs, Plants, and Venomous Animals; John Wiley & Sons: Hoboken, NJ, USA, 2012; ISBN 978-1-118-38276-9. [Google Scholar]
- Al-Doory, Y.; Domson, J.F. Mould Allergy; Lea & Febiger: Philadelphia, PA, USA, 1984; ISBN 0-8121-0897-3. [Google Scholar]
- Böhm, J.; Hoff, B.; O’Gorman, C.M.; Wolfers, S.; Klix, V.; Binger, D.; Zadra, I.; Kürnsteiner, H.; Pöggeler, S.; Dyer, P.S.; et al. Sexual Reproduction and Mating-Type–Mediated Strain Development in the Penicillin-Producing Fungus Penicillium chrysogenum. Proc. Natl. Acad. Sci. USA 2013, 110, 1476–1481. [Google Scholar] [CrossRef]
- Gibson, A.M.; Baranyi, J.; Pitt, J.I.; Eyles, M.J.; Roberts, T.A. Predicting Fungal Growth: The Effect of Water Activity on Aspergillus flavus and Related Species. Int. J. Food Microbiol. 1994, 23, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Troncoso-Rojas, R.; Tiznado-Hernández, M.E. Chapter 5—Alternaria alternata (Black Rot, Black Spot). In Postharvest Decay; Bautista-Baños, S., Ed.; Academic Press: San Diego, CA, USA, 2014; pp. 147–187. ISBN 978-0-12-411552-1. [Google Scholar]
- Institut National de Santé Publique du Québec. Alternaria Alternata. Available online: https://www.inspq.qc.ca/en/moulds/fact-sheets/alternaria-alternata (accessed on 10 February 2023).
- Lopez, S.E.; Cabral, D. Alternaria . In Encyclopedia of Food Microbiology; Robinson, R.K., Ed.; Elsevier: Oxford, UK, 1999; pp. 42–49. ISBN 978-0-12-227070-3. [Google Scholar]
- Deshmukh, S.K.; Rai, M.K. Biodiversity of Fungi; Science Publishers, Inc.: Enfield, UK, 2005. [Google Scholar]
- Samson, R.A.; Houbraken, J.; Summerbell, R.C.; Flannigan, B.; Miller, J.D. Common and Important Species of Fungi and Actinomycetes in Indoor Environments. In Microorganisms in Home and Indoor Work Environments: Diversity, Health Impacts, Investigation and Control; CRC Press: London, UK, 2002; pp. 285–473. [Google Scholar]
- Flannigan, B.; Samson, R.A.; Miller, J.D. Microorganisms in Home and Indoor Work Environments: Diversity, Health Impacts, Investigation and Control; CRC Press: London, UK, 2002; ISBN 0-429-21953-9. [Google Scholar]
- Yang, P.; Burson, K.; Feder, D.; Macdonald, F. Swab Sampling for Cleaning Validation. Pharm. Technol. 2005, 1, 84–94. [Google Scholar]
- Silva, N.C.; Pintado, M.; Moreira, P.R. Sampling Methods for Outdoor Sculptures: Comparison of Swabs and Cryogels by Flow Cytometry as Novel Alternatives for Assessment and Quantification of Microbial Contamination. J. Cult. Herit. 2022, 54, 94–102. [Google Scholar] [CrossRef]
- Mouchtouri, V.A.; Koureas, M.; Kyritsi, M.; Vontas, A.; Kourentis, L.; Sapounas, S.; Rigakos, G.; Petinaki, E.; Tsiodras, S.; Hadjichristodoulou, C. Environmental Contamination of SARS-CoV-2 on Surfaces, Air-Conditioner and Ventilation Systems. Int. J. Hyg. Environ. Health 2020, 230, 113599. [Google Scholar] [CrossRef]
- Bain, R.M.; Fedick, P.W.; Dilger, J.M.; Cooks, R.G. Analysis of Residual Explosives by Swab Touch Spray Ionization Mass Spectrometry. Propellants Explos. Pyrotech. 2018, 43, 1139–1144. [Google Scholar] [CrossRef]
- Samson, R.A.; Van Reenen-Hoekstra, E.S. Introduction to Food-Borne Fungi; Centraalbureau voor Schimmelcultures: Baarn, The Netherlands, 1988. [Google Scholar]
- Antecka, A.; Bizukojc, M.; Ledakowicz, S. Modern Morphological Engineering Techniques for Improving Productivity of Filamentous Fungi in Submerged Cultures. World J. Microbiol. Biotechnol. 2016, 32, 193. [Google Scholar] [CrossRef]
- Driouch, H.; Roth, A.; Dersch, P.; Wittmann, C. Optimized Bioprocess for Production of Fructofuranosidase by Recombinant Aspergillus niger. Appl. Microbiol. Biotechnol. 2010, 87, 2011–2024. [Google Scholar] [CrossRef]
- Hospodsky, D.; Yamamoto, N.; Peccia, J. Accuracy, Precision, and Method Detection Limits of Quantitative PCR for Airborne Bacteria and Fungi. Appl. Environ. Microbiol. 2010, 76, 7004–7012. [Google Scholar] [CrossRef] [PubMed]
- Verdier, T.; Bertron, A.; Johansson, P. Overview of Indoor Microbial Growth on Building Materials. In Proceedings of the International RILEM Symposium on the Microorganisms-Cementitious Materials Interactions (TC253-MCI), Delft, The Netherlands, 23 June 2016; Volume 23. [Google Scholar]
- Mousavi, B.; Hedayati, M.; Hedayati, N.; Ilkit, M.; Syedmousavi, S. Aspergillus Species in Indoor Environments and Their Possible Occupational and Public Health Hazards. Curr. Med. Mycol. 2016, 2, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Cesuroglu, O.; Colakoglu, G.T. A Study on the Fungi Isolated from the Carpeting, Walls and Holly Qurans from the Blue Mosque and Little Hagia Sophia Mosque siuated in the Province of Istanbul. J. Yeast Fungal Res. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- Egbuta, M.A.; Mwanza, M.; Babalola, O.O. Health Risks Associated with Exposure to Filamentous Fungi. Int. J. Environ. Res. Public Health 2017, 14, 719. [Google Scholar] [CrossRef]
- Engelhart, S.; Loock, A.; Skutlarek, D.; Sagunski, H.; Lommel, A.; Färber, H.; Exner, M. Occurrence of Toxigenic Aspergillus versicolor Isolates and Sterigmatocystin in Carpet Dust from Damp Indoor Environments. Appl. Environ. Microbiol. 2002, 68, 3886–3890. [Google Scholar] [CrossRef] [PubMed]
- Gaüzère, C.; Moletta-Denat, M.; Blanquart, H.; Ferreira, S.; Moularat, S.; Godon, J.J.; Robine, E. Stablility of Airborne Microbes in the Louvre Museum Over Time. Indoor Air 2014, 24, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.-C.; Huang, N.; Hsieh, C.-J.; Hung, C.-C.; Guo, Y.-L.L. Contribution of Visible Surface Mold to Airborne Fungal Concentration as Assessed by Digital Image Quantification. Pathogens 2021, 10, 1032. [Google Scholar] [CrossRef] [PubMed]
- Bjurman, J.; Kristensson, J. Volatile Production by Aspergillus versicolor as a Possible Cause of Odor in Houses Affected by Fungi. Mycopathologia 1992, 118, 173–178. [Google Scholar] [CrossRef]
- Reijula, K.; Tuomi, T. Mycotoxins of Aspergilli; Exposure and Health Effects. Front. Biosci. Landmark 2003, 8, 232–235. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Larsen, T.O.; Thrane, U.; Meijer, M.; Varga, J.; Samson, R.A.; Nielsen, K.F. Fumonisin and Ochratoxin Production in Industrial Aspergillus niger Strains. PLoS ONE 2011, 6, e23496. [Google Scholar] [CrossRef]
- Nielsen, K.F.; Mogensen, J.M.; Johansen, M.; Larsen, T.O.; Frisvad, J.C. Review of Secondary Metabolites and Mycotoxins from the Aspergillus niger Group. Anal. Bioanal. Chem. 2009, 395, 1225–1242. [Google Scholar] [CrossRef]
- Matsuda, Y.; Awakawa, T.; Abe, I. Reconstituted Biosynthesis of Fungal Meroterpenoid Andrastin A. Tetrahedron 2013, 69, 8199–8204. [Google Scholar] [CrossRef]
- Viggiano, A.; Salo, O.; Ali, H.; Szymanski, W.; Lankhorst, P.P.; Nygård, Y.; Bovenberg, R.A.L.; Driessen, A.J.M. Pathway for the Biosynthesis of the Pigment Chrysogine by Penicillium chrysogenum. Appl. Environ. Microbiol. 2018, 84, e02246-17. [Google Scholar] [CrossRef]
- Khalil, N.M.; Abd El-Ghany, M.N.; Rodríguez-Couto, S. Antifungal and Anti-Mycotoxin Efficacy of Biogenic Silver Nanoparticles Produced by Fusarium chlamydosporum and Penicillium chrysogenum at Non-Cytotoxic Doses. Chemosphere 2019, 218, 477–486. [Google Scholar] [CrossRef]
- Ali, H.; Ries, M.I.; Lankhorst, P.P.; van der Hoeven, R.A.M.; Schouten, O.L.; Noga, M.; Hankemeier, T.; van Peij, N.N.M.E.; Bovenberg, R.A.L.; Vreeken, R.J.; et al. A Non-Canonical NRPS Is Involved in the Synthesis of Fungisporin and Related Hydrophobic Cyclic Tetrapeptides in Penicillium chrysogenum. PLoS ONE 2014, 9, e98212. [Google Scholar] [CrossRef]
- Ali, H.; Ries, M.I.; Nijland, J.G.; Lankhorst, P.P.; Hankemeier, T.; Bovenberg, R.A.L.; Vreeken, R.J.; Driessen, A.J.M. A Branched Biosynthetic Pathway Is Involved in Production of Roquefortine and Related Compounds in Penicillium chrysogenum. PLoS ONE 2013, 8, e65328. [Google Scholar] [CrossRef]
- Reiss, J. Mycotoxins in Foodstuffs. x. Production of Citrinin by Penicillium chrysogenum in Bread. Food Cosmet. Toxicol. 1977, 15, 303–307. [Google Scholar] [CrossRef]
- Nielsen, K.F.; Gravesen, S.; Nielsen, P.A.; Andersen, B.; Thrane, U.; Frisvad, J.C. Production of Mycotoxins on Artificially and Naturally Infested Building Materials. Mycopathologia 1999, 145, 43–56. [Google Scholar] [CrossRef]
- International Agency for Research against Cancer. Monographs on the Evaluation of Carcinogenic Risks to Humans. In Some Industrial Chemicals; IARC Publications: Geneva, Switzerland, 1994; Volume 60, pp. 389–433. [Google Scholar]
- Chari, S.; Xie, K.; Kale, I.; Khow, K.; Nair, R.; Hanna, N.M. Detection and Control of Spoilage Fungi in Refrigerated Vegetables and Fruits. J. Emerg. Investig. 2021, 3, 1. [Google Scholar]
- Alwatban, M.A.; Hadi, S.; Moslem, M.A. Mycotoxin Production in Cladosporium Species Influenced by Temperature Regimes. J. Pure Appl. Microbiol. 2014, 8, 4061–4069. [Google Scholar]
- Andersen, B.; Hollensted, M. Metabolite Production by Different Ulocladium Species. Int. J. Food Microbiol. 2008, 126, 172–179. [Google Scholar] [CrossRef]
- Adan, O.C.; Samson, R.A. Fundamentals of Mold Growth in Indoor Environments and Strategies for Healthy Living; Springer: Berlin/Heidelberg, Germany, 2011; ISBN 90-8686-722-7. [Google Scholar]
- Torres-Rodríguez, J.M.; Madrenys-Brunet, N.; Siddat, M.; López-Jodra, O.; Jimenez, T. Aspergillus versicolor as Cause of Onychomycosis: Report of 12 Cases and Susceptibility Testing to Antifungal Drugs. J. Eur. Acad. Dermatol. Venereol. 1998, 11, 25–31. [Google Scholar] [CrossRef]
- Charles, M.P.; Noyal, M.J.; Easow, J.M. Invasive Pulmonary Aspergillosis Caused by Aspergillus versicolor in a Patient on Mechanical Ventilation. Australas. Med. J. 2011, 4, 632. [Google Scholar] [CrossRef]
- Person, A.K.; Chudgar, S.M.; Norton, B.L.; Tong, B.C.; Stout, J.E. Aspergillus niger: An Unusual Cause of Invasive Pulmonary Aspergillosis. J. Med. Microbiol. 2010, 59, 834. [Google Scholar] [CrossRef]
- Georgiadou, S.P.; Kontoyiannis, D.P. Concurrent Lung Infections in Patients with Hematological Malignancies and Invasive Pulmonary Aspergillosis: How Firm Is the Aspergillus Diagnosis? J. Infect. 2012, 65, 262–268. [Google Scholar] [CrossRef]
- Kamei, K.; Watanabe, A. Aspergillus Mycotoxins and Their Effect on the Host. Med. Mycol. 2005, 43 (Suppl. S1), S95–S99. [Google Scholar] [CrossRef] [PubMed]
- Fischer, G.; Müller, T.; Schwalbe, R.; Ostrowski, R.; Dott, W. Exposure to Airborne Fungi, MVOC and Mycotoxins in Biowaste-Handling Facilities. Int. J. Hyg. Environ. Health 2000, 203, 97–104. [Google Scholar] [CrossRef]
- Kupfahl, C.; Michalka, A.; Lass-Flörl, C.; Fischer, G.; Haase, G.; Ruppert, T.; Geginat, G.; Hof, H. Gliotoxin Production by Clinical and Environmental Aspergillus fumigatus Strains. Int. J. Med. Microbiol. 2008, 298, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Segers, F.J.J.; Meijer, M.; Houbraken, J.; Samson, R.A.; Wösten, H.A.B.; Dijksterhuis, J. Xerotolerant Cladosporium sphaerospermum Are Predominant on Indoor Surfaces Compared to Other Cladosporium Species. PLoS ONE 2015, 10, e0145415. [Google Scholar] [CrossRef] [PubMed]
- Yew, S.M.; Chan, C.L.; Ngeow, Y.F.; Toh, Y.F.; Na, S.L.; Lee, K.W.; Hoh, C.-C.; Yee, W.-Y.; Ng, K.P.; Kuan, C.S. Insight into Different Environmental Niches Adaptation and Allergenicity from the Cladosporium sphaerospermum Genome, a Common Human Allergy-Eliciting Dothideomycetes. Sci. Rep. 2016, 6, 27008. [Google Scholar] [CrossRef]
- Pestka, J.J.; Yike, I.; Dearborn, D.G.; Ward, M.D.W.; Harkema, J.R. Stachybotrys chartarum, Trichothecene Mycotoxins, and Damp Building-Related Illness: New Insights into a Public Health Enigma. Toxicol. Sci. 2008, 104, 4–26. [Google Scholar] [CrossRef]
- Ulrich, S.; Schäfer, C. Toxin Production by Stachybotrys chartarum Genotype S on Different Culture Media. J. Fungi 2020, 6, 159. [Google Scholar] [CrossRef]
- Frazer, S. Characterisation of Stachybotrys chartarum from Water Damaged Buildings. Ph.D. Thesis, Cranfield University, Cranfield, UK, 2011. [Google Scholar]
- Walsh, T.J.; Groll, A.; Hiemenz, J.; Fleming, R.; Roilides, E.; Anaissie, E. Infections Due to Emerging and Uncommon Medically Important Fungal Pathogens. Clin. Microbiol. Infect. 2004, 10, 48–66. [Google Scholar] [CrossRef]
- Sporik, R.B.; Arruda, L.K.; Woodfolk, J.; Chapman, M.D.; Platts-Mills, T.A.E. Environmental Exposure to Aspergillus fumigatus Allergen (Asp f I). Clin. Exp. Allergy 1993, 23, 326–331. [Google Scholar] [CrossRef]
- Pose, G.; Patriarca, A.; Kyanko, V.; Pardo, A.; Fernández Pinto, V. Water Activity and Temperature Effects on Mycotoxin Production by Alternaria alternata on a Synthetic Tomato Medium. Int. J. Food Microbiol. 2010, 142, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, D.; Yuan, S.; Feng, X.; Wang, M. Transcriptomic Insights into the Antifungal Effects of Magnolol on the Growth and Mycotoxin Production of Alternaria alternata. Toxins 2020, 12, 665. [Google Scholar] [CrossRef] [PubMed]
- Magan, N.; Cayley, G.R.; Lacey, J. Effect of Water Activity and Temperature on Mycotoxin Production by Alternaria alternata in Culture and on Wheat Grain. Appl. Environ. Microbiol. 1984, 47, 1113–1117. [Google Scholar] [CrossRef] [PubMed]
- Bensassi, F.; Gallerne, C.; Sharaf el dein, O.; Rabeh Hajlaoui, M.; Bacha, H.; Lemaire, C. Combined Effects of Alternariols Mixture on Human Colon Carcinoma Cells. Toxicol. Mech. Methods 2015, 25, 56–62. [Google Scholar] [CrossRef]
- Wang, T.; Li, Y.; Bi, Y.; Zhang, M.; Zhang, T.; Zheng, X.; Dong, Y.; Huang, Y. Benzyl Isothiocyanate Fumigation Inhibits Growth, Membrane Integrity and Mycotoxin Production in Alternaria alternata. RSC Adv. 2020, 10, 1829–1837. [Google Scholar] [CrossRef]
- Ostry, V. Alternaria Mycotoxins: An Overview of Chemical Characterization, Producers, Toxicity, Analysis and Occurrence in Foodstuffs. World Mycotoxin J. 2008, 1, 175–188. [Google Scholar] [CrossRef]
- Pfeiffer, S.; Sandler, P.; Raith, M.; Pascal, M.; Munoz-Cano, R.M.; San Bartolome, C.; Nöbauer, K.; Quirce, S.; Razzazi-Fazeli, E.; Focke-Tejkl, M.; et al. Identification of Ulocladium chartarum as an Important Indoor Allergen Source. Allergy 2021, 76, 3202–3206. [Google Scholar] [CrossRef]
- Alassane-Kpembi, I.; Puel, O.; Pinton, P.; Cossalter, A.M.; Chou, T.C. Co-exposure to low doses of the food contaminants deoxynivalenol and nivalenol has a synergic inflammatory effect on intestinal explants. Arch. Toxicol. 2017, 91, 2677–2687. [Google Scholar] [CrossRef]
- Brasel, T.L.; Martin, J.M.; Carriker, C.G.; Wilson, S.C.; Straus, D.C. Detection of airborne Stachybotrys chartarum macrocyclic trichothecene mycotoxins in the indoor environment. Appl. Environ. Microbiol. 2005, 71, 7376–7388. [Google Scholar] [CrossRef]
- Polizzi, V.; Delmulle, B.; Adams, A.; Moretti, A.; Susca, A.; Picco, A.M.; Rosseel, Y.; Kindt, R.; Van Bocxlaer, J.; De Kimpe, N.; et al. Fungi, mycotoxins and microbial volatil organic compounds in mouldy interiors from water-damaged buildings. J. Environ. Monit. 2009, 11, 1849–1858. [Google Scholar] [CrossRef]
- Charpin-Kadouch, C.; Maurel, G.; Felipo, R.; Queralt, J.; Ramadour, M.; Dumon, H.; Garans, M.; Botta, A.; Charpin, D. Mycotoxin identification in moldy dwellings. J. Appl. Toxicol. 2006, 26, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, C.; Bauer, J.; Meyer, K. Detection of satratoxin G and H in indoor air from a water-damaged building. Mycopathologia 2008, 166, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Bloom, E.; Nyman, E.; Must, A.; Pehrson, C.; Larsson, L. Molds and mycotoxins in indoor environments: A survey in water-damaged buildings. J. Occup. Environ. Hyg. 2009, 6, 671–678. [Google Scholar] [CrossRef] [PubMed]
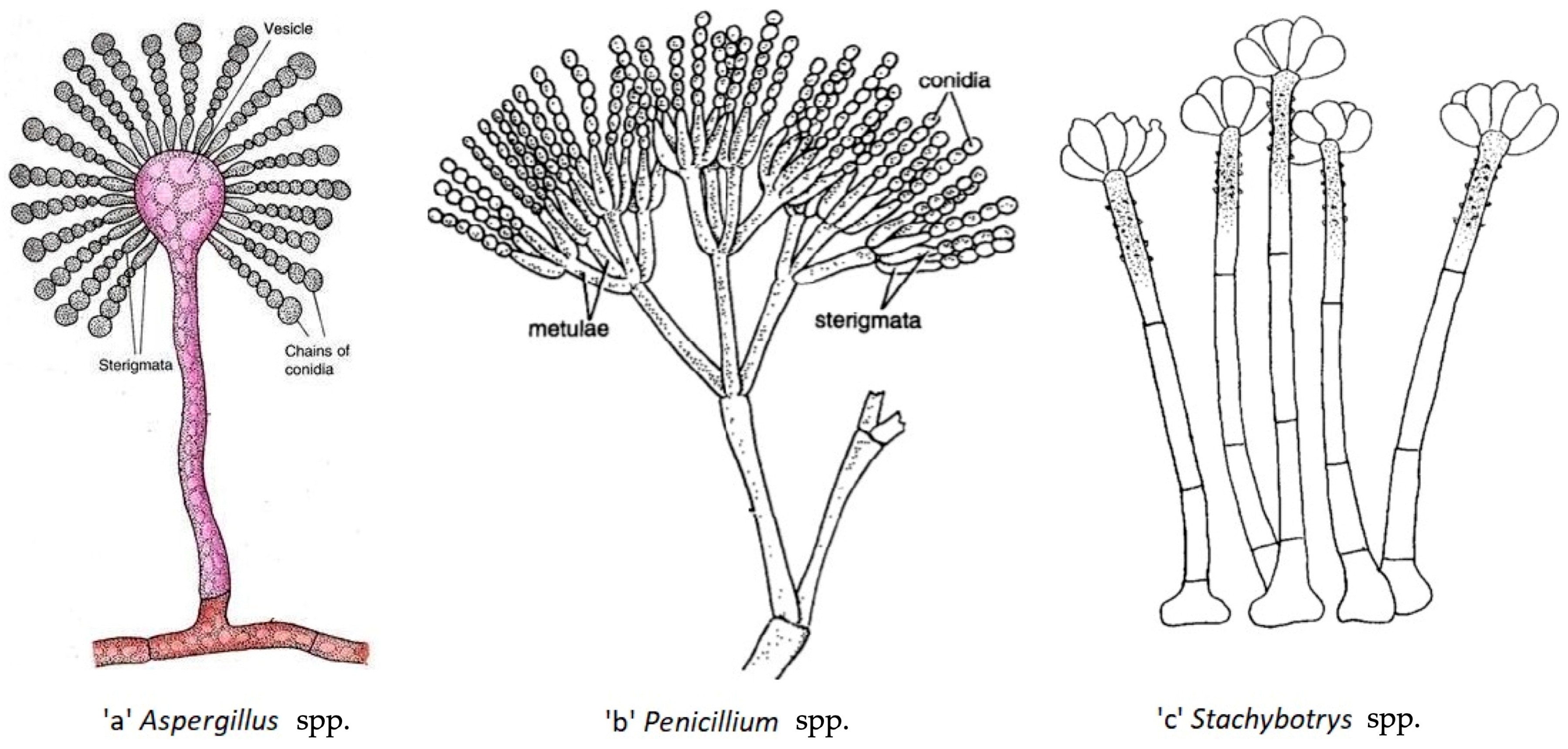
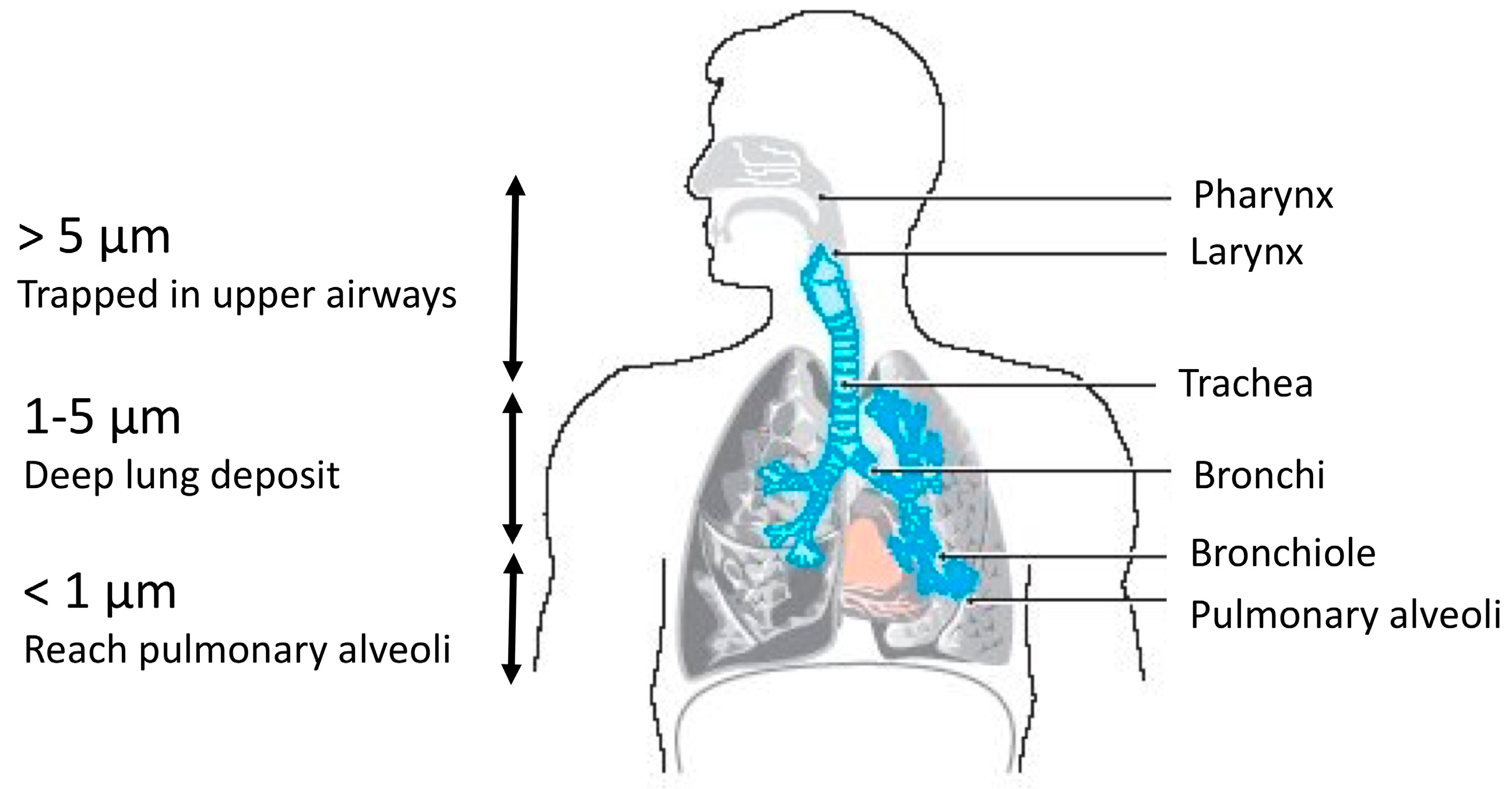
| Species | Temperature for Development | Minimal Water Activity | Colour of the Colony | References | |
|---|---|---|---|---|---|
| Optimal | Range | ||||
| Aspergillus versicolor | 22–26 °C | 4–40 °C | 0.75–0.81 | Variable according to environmental conditions: white, pinkish, from yellow to green | [100,101,102] |
| Aspergillus niger | 27 °C | 25–40 °C | 0.85 | Turns to black during sporulation | [96,103] |
| Aspergillus fumigatus | 37 °C | Up to 55 °C Survive at 70 °C | >0.9 | Blue-green | [31,32,104] |
| Cladosporium sphaerospermum | 25 °C | −5–35 °C | >0.82 | Black | [105,106] |
| Stachybotrys chartarum | 23–27 °C | 2–40 °C | >0.94 | Black | [107,108] |
| Penicillium chrysogenum | 20–30 °C | 5–37 °C | >0.78 | Blue-green | [109,110] |
| Aspergillus flavus | 32–36 °C | 10–50 °C | >0.78 | Bright yellow-green | [99,104,111] |
| Alternaria alternata | 25–29 °C | 2–32 °C | >0.85 | Black | [97,112,113,114] |
| Cladosporium cladosporioides | 18–28 °C | −10–35°C | >0.88 | Black | [36,97,115] |
| Ulocladium chartarum | 25 °C | 5–34 °C | >0.9 | Golden brown to blackish brown | [97,116,117] |
| Species | Any location [n = 34; (%)] | Europe [n = 26; (%)] | Northern America [n = 8; (%)] |
|---|---|---|---|
| Aspergillus versicolor | 25 (74%) | 20 (77%) | 5 (63%) |
| Aspergillus niger | 23 (68%) | 17 (65%) | 6 (75%) |
| Aspergillus fumigatus | 18 (53%) | 17 (65%) | 1 (13%) |
| Cladosporium sphaerospermum | 14 (41%) | 12 (46%) | 2 (25%) |
| Stachybotrys chartarum | 11 (3%) | 7 (27%) | 4 (50%) |
| Penicillium chrysogenum | 10 (29%) | 10 (38%) | 0 (0%) |
| Aspergillus flavus | 9 (26%) | 9 (35%) | 0 (0%) |
| Alternaria alternata | 7 (21%) | 6 (23%) | 1 (13%) |
| Cladosporium cladosporioides | 6 (18%) | 6 (23%) | 0 (0%) |
| Ulocladium chartarum | 5 (15%) | 4 (15%) | 1 (13%) |
| Species | Studies That Carried Out Both Air and Surface Sampling | Studies That Carried Out Only Surface Sampling | |
|---|---|---|---|
| Number (%) of Airborne Detections [n = 21] | Number (%) of Surface Detections [n = 21] | Number (%) of Surface Detections [n = 34] | |
| Aspergillus versicolor | 18 (86%) | 18 (86%) | 25 (74%) |
| Aspergillus niger | 15 (71%) | 14 (67%) | 23 (68%) |
| Aspergillus fumigatus | 9 (43%) | 9 (43%) | 18 (53%) |
| Cladosporium sphaerospermum | 9 (43%) | 9 (43%) | 14 (41%) |
| Stachybotrys chartarum | 6 (29%) | 5 (24%) | 11 (32%) |
| Penicillium chrysogenum | 7 (33%) | 7 (33%) | 10 (29%) |
| Aspergillus flavus | 7 (33%) | 5 (24%) | 9 (26%) |
| Alternaria alternata | 4 (19%) | 5 (24%) | 7 (21%) |
| Cladosporium cladosporioides | 5 (24%) | 4 (19%) | 6 (18%) |
| Ulocladium chartarum | 3 (14%) | 4 (19%) | 5 (15%) |
| Species | Mycotoxins Produced | Associated Diseases | References |
|---|---|---|---|
| Aspergillus versicolor | Sterigmatocystin, nidulotoxins, Aspergillomarasmine A, Aspergillomarasmine B, xanthones, fellutamides and anthraquinones | Allergic diseases, aspergilloses, onychomycosis, immunosuppression | [129,132,133,134] |
| Aspergillus niger | Ochratoxin A (OTA), fumonisin B2 (FB2), fumonisin B4 (FB4) | Aspergillosis, otomycosis kidney failure (OTA) | [135,136] |
| Aspergillus fumigatus | Gliotoxin, fumagillin, fumigaclavines, helvolic acid, fumitremorgin A and Asphemolysin, β-1,3 glucans | Pulmonary infections, modulation of the immune system, abortions in farm animals | [62,63,64,65] |
| Cladosporium sphaerospermum | Atenuisol, calphostin A, C, D, cladosporol, emodin | Inhibition of protein kinase C | [76] |
| Stachybotrys chartarum | Atranones, macrocyclic trichothecenes | Pulmonary inflammation, Protein synthesis inhibition | [66,67,68] |
| Penicillium chrysogenum | Citrinin, roquefortine C, meleagrin, chrysogine, fungisporin, andrastin A | Necrotizing esophagitis, and asthma | [137,138,139,140,141,142,143] |
| Aspergillus flavus | Aflatoxins, sterigmatocystin, cyclopiazonic acid, kojic acid and aflatrem | Hepatocarcinoma (aflatoxins = group I of the IARC), immunosuppression, invasive aspergillosis, allergies | [30,111,139,144] |
| Alternaria alternata | Alternariol (AOH), Altenuene (AE), Alternariol mono-methyl ether (AME), tentoxin (TEN) and Tenuazonic acid (TeA) | Cytotoxicity for animal cells. Fetotoxic and teratogenic to mice and hamsters. | [65,66,67,68,69] |
| Cladosporium cladosporioides | Atenuisol, calphostin A, C, D, cladosporol, emodin, | Allergic reactions, Inhibition of protein kinase C | [145,146] |
| Ulocladium chartarum | Infectopyrones and derivatives of altertoxin I | Allergic diseases | [147,148] |
| Country | Number of Locations | Detected Toxins | Toxin Concentration | References |
|---|---|---|---|---|
| USA | 7 water-damaged homes with Stachybotrys contamination: 40 air samples from 16 rooms | Macrocyclic trichothecenes | <10 to >1300 pg/m3 of sampled air | [171] |
| Belgium | 7 water-damaged homes: 20 air samples | Roquefortine C (in 1 sample) Chaetoglobosin A (in 3 samples) Sterigmatocystin (in 3 samples) Aflatoxin B1 (in 5 samples) Aflatoxin B2 (in 4 samples) Roridin E (in 3 samples) Ochratoxin A (in 3 samples) | Roquefortine C: 1–4 ng/m3 Chaetoglobosin A: 0.0067–3.4 ng/m3 Sterigmatocystin: 0.0034–1.77 ng/m3 Aflatoxin B1: 0.0024–0.15 ng/m3 Aflatoxin B2: 0.0003–0.02 ng/m3 Roridin E: 0.0031–0.08 ng/m3 Ochratoxin A: 0.01–0.23 ng/m3 | [172] |
| France | 15 flooded dwellings with Stachybotrys contamination + 9 control dwellings | Macrocyclic trichothecenes | 0.29–0.62 ng/m3 | [173] |
| Germany | 1 air sample form water-damaged building | Satratoxin G and H | Satratoxin G: 0.25 ng/m3 Satratoxin H: 0.43 ng/m3 | [174] |
| Sweden | 37 samples from 22 water-damaged building | Trichodermol (in 26% of samples) Verrucarol (in 21.6%) Sterigmatocystin (in 0.5%) Gliotoxin (in 2.7%) Satratoxin G (in 5.4%) Satratoxin H (in 5.4%) Aflatoxin B1 (in 5.4%) | [175] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Hallak, M.; Verdier, T.; Bertron, A.; Roques, C.; Bailly, J.-D. Fungal Contamination of Building Materials and the Aerosolization of Particles and Toxins in Indoor Air and Their Associated Risks to Health: A Review. Toxins 2023, 15, 175. https://doi.org/10.3390/toxins15030175
Al Hallak M, Verdier T, Bertron A, Roques C, Bailly J-D. Fungal Contamination of Building Materials and the Aerosolization of Particles and Toxins in Indoor Air and Their Associated Risks to Health: A Review. Toxins. 2023; 15(3):175. https://doi.org/10.3390/toxins15030175
Chicago/Turabian StyleAl Hallak, Mohamad, Thomas Verdier, Alexandra Bertron, Christine Roques, and Jean-Denis Bailly. 2023. "Fungal Contamination of Building Materials and the Aerosolization of Particles and Toxins in Indoor Air and Their Associated Risks to Health: A Review" Toxins 15, no. 3: 175. https://doi.org/10.3390/toxins15030175
APA StyleAl Hallak, M., Verdier, T., Bertron, A., Roques, C., & Bailly, J.-D. (2023). Fungal Contamination of Building Materials and the Aerosolization of Particles and Toxins in Indoor Air and Their Associated Risks to Health: A Review. Toxins, 15(3), 175. https://doi.org/10.3390/toxins15030175








