ADDovenom: Thermostable Protein-Based ADDomer Nanoparticles as New Therapeutics for Snakebite Envenoming
Abstract
:1. Introduction
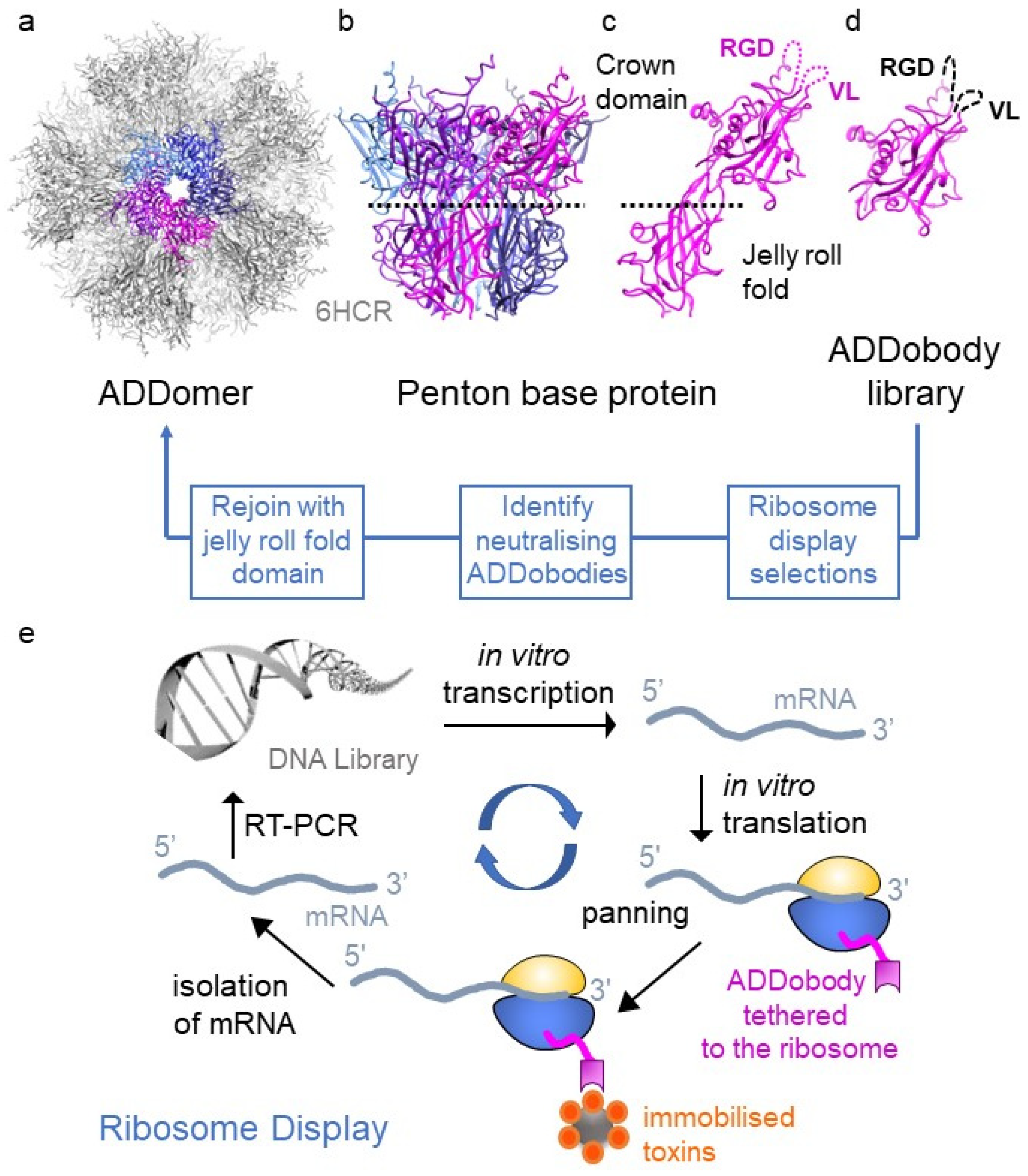
2. ADDomer—A Versatile Thermostable Nanoparticle
- Thermotolerance up to >45 °C [17,22]. It has been shown that the ADDomer nanoparticle can be stored for one month at ~20 °C; it can be frozen and thawed and/or heated to 45 °C for 1 h, virtually without losing its structural integrity [17]. Consistently, ADDomer does not require cold-chain storage, which constitutes a key logistic advance;
- ADDomers can be lyophilised [22]. This manufacturing step increases storage life, providing a commercial incentive for manufacturers;
- ADDomers can be produced as recombinant protein-based nanoparticles with exceptionally good yields using a baculovirus–insect cell expression system, allowing establishment of good manufacturing practices with stringent quality control;
- Importantly, the penton base protein is the least immunogenic of all the adenovirus capsid proteins. Therefore, ADDomer presents a low immunogenicity scaffold.
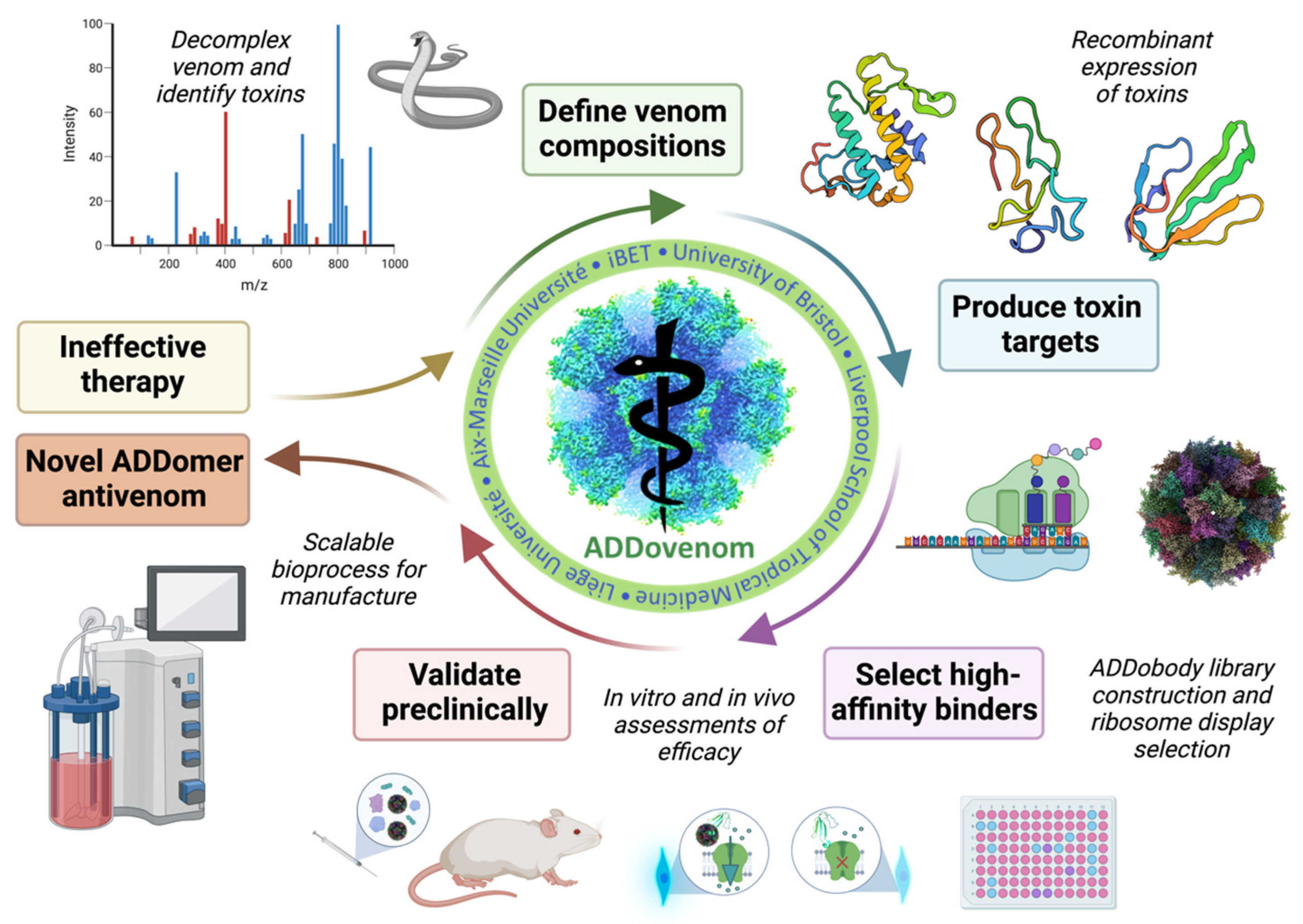
3. The EU-Funded ADDovenom Project
3.1. Venom Mass Spectrometry and Bioinformatics
3.2. Production of Recombinant Toxins as Antigens for Ribosome Display Selection
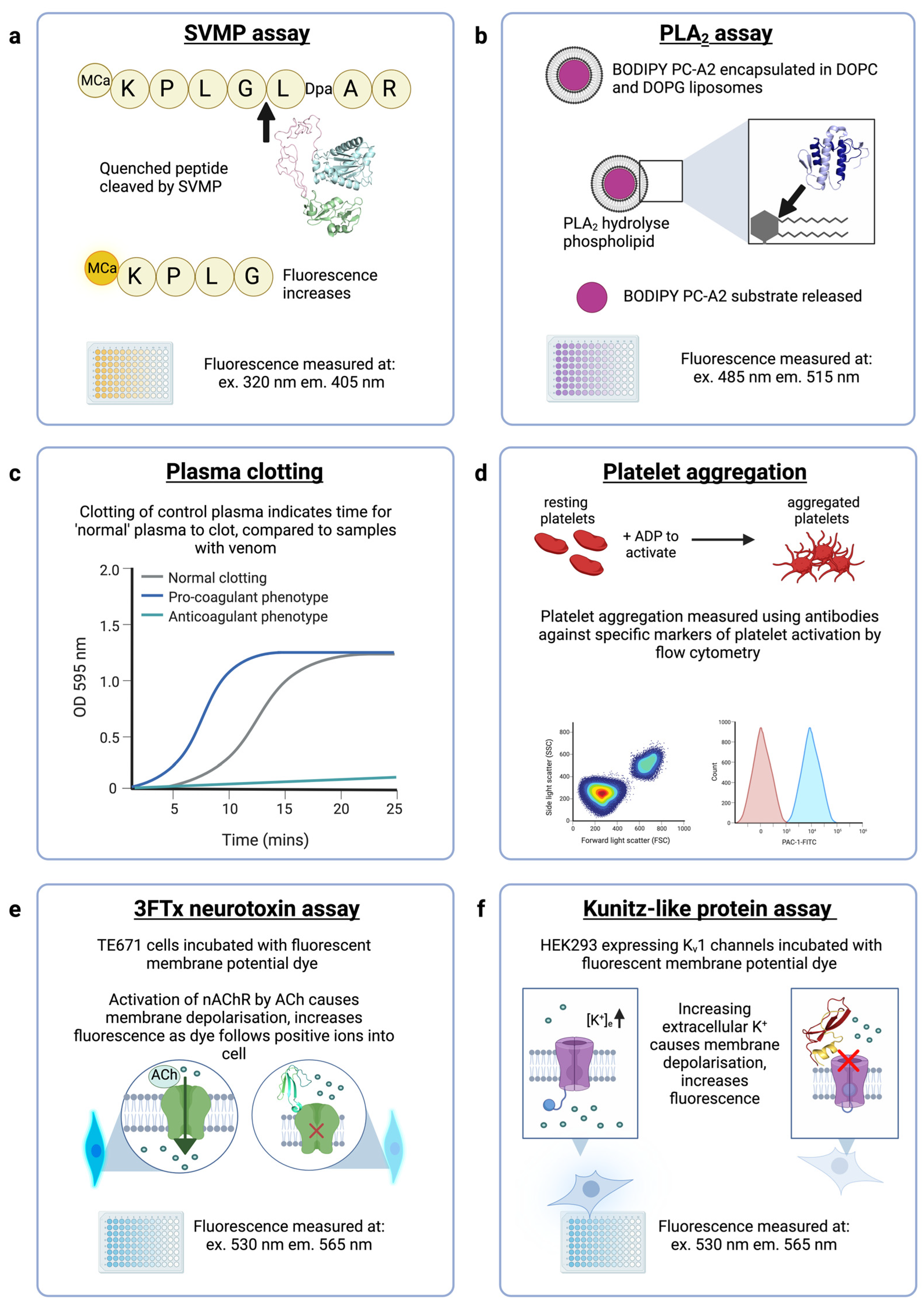
3.3. Evaluation of the Neutralising Ability of ADDobodies and ADDomers
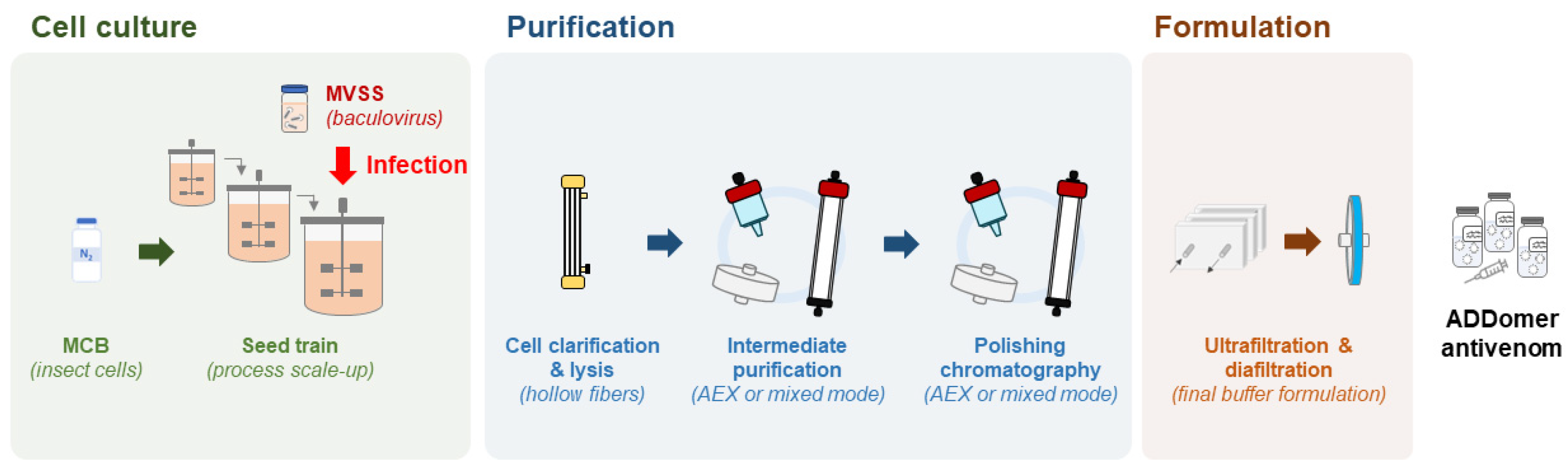
3.4. Scalable Bioprocess for ADDomer Production
4. Future Perspective and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gutiérrez, J.M.; Calvete, J.J.; Habib, A.G.; Harrison, R.A.; Williams, D.J.; Warrell, D.A. Snakebite envenoming. Nat. Rev. Dis. Primers 2017, 3, 17063. [Google Scholar] [CrossRef] [PubMed]
- Harrison, R.A.; Hargreaves, A.; Wagstaff, S.C.; Faragher, B.; Lalloo, D.G. Snake Envenoming: A Disease of Poverty. PLoS Negl. Trop. Dis. 2009, 3, e569. [Google Scholar] [CrossRef]
- Laustsen, A.H.; María Gutiérrez, J.; Knudsen, C.; Johansen, K.H.; Bermúdez-Méndez, E.; Cerni, F.A.; Jürgensen, J.A.; Ledsgaard, L.; Martos-Esteban, A.; Øhlenschlæger, M.; et al. Pros and cons of different therapeutic antibody formats for recombinant antivenom development. Toxicon 2018, 146, 151–175. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.P.; Williams, V.; White, J. Snake venom variability: Methods of study, results and interpretation. Toxicon 1991, 29, 1279–1303. [Google Scholar] [CrossRef] [PubMed]
- Harrison, R.A.; Casewell, N.R.; Ainsworth, S.A.; Lalloo, D.G. The time is now: A call for action to translate recent momentum on tackling tropical snakebite into sustained benefit for victims. Trans. R. Soc. Trop. Med. Hyg. 2019, 113, 835–838. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for the Production, Control and Regulation of Snake Antivenom Immunoglobulins. 2017. Available online: https://cdn.who.int/media/docs/default-source/biologicals/blood-products/document-migration/antivenomglrevwho_trs_1004_web_annex_5.pdf?sfvrsn=ef4b2aa5_3&download=true (accessed on 3 November 2023).
- Abubakar, S.B.; Abubakar, I.S.; Habib, A.G.; Nasidi, A.; Durfa, N.; Yusuf, P.O.; Larnyang, S.; Garnvwa, J.; Sokomba, E.; Salako, L.; et al. Pre-clinical and preliminary dose-finding and safety studies to identify candidate antivenoms for treatment of envenoming by saw-scaled or carpet vipers (Echis ocellatus) in northern Nigeria. Toxicon 2010, 55, 719–723. [Google Scholar] [CrossRef]
- Segura, Á.; Herrera, M.; Villalta, M.; Vargas, M.; Gutiérrez, J.M.; León, G. Assessment of snake antivenom purity by comparing physicochemical and immunochemical methods. Biologicals 2013, 41, 93–97. [Google Scholar] [CrossRef]
- Rawat, S.; Laing, G.; Smith, D.C.; Theakston, D.; Landon, J. A new antivenom to treat eastern coral snake (Micrurus fulvius fulvius) envenoming. Toxicon 1994, 32, 185–190. [Google Scholar] [CrossRef]
- Herrera, M.; Paiva, O.K.; Pagotto, A.H.; Segura, Á.; Serrano, S.M.T.; Vargas, M.; Villalta, M.; Jensen, S.D.; León, G.; Williams, D.J.; et al. Antivenomic Characterization of Two Antivenoms Against the Venom of the Taipan, Oxyuranus scutellatus, from Papua New Guinea and Australia. Am. J. Trop. Med. Hyg. 2014, 91, 887–894. [Google Scholar] [CrossRef]
- Harrison, R.A.; Gutiérrez, J.M. Priority Actions and Progress to Substantially and Sustainably Reduce the Mortality, Morbidity and Socioeconomic Burden of Tropical Snakebite. Toxins 2016, 8, 351. [Google Scholar] [CrossRef]
- Pucca, M.B.; Cerni, F.A.; Janke, R.; Bermúdez-Méndez, E.; Ledsgaard, L.; Barbosa, J.E.; Laustsen, A.H. History of Envenoming Therapy and Current Perspectives. Front. Immunol. 2019, 10, 1598. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Albulescu, L.-O.; Clare, R.H.; Casewell, N.R.; Abd El-Aziz, T.M.; Escalante, T.; Rucavado, A. The Search for Natural and Synthetic Inhibitors That Would Complement Antivenoms as Therapeutics for Snakebite Envenoming. Toxins 2021, 13, 451. [Google Scholar] [CrossRef]
- Jenkins, T.P.; Fryer, T.; Dehli, R.I.; Jürgensen, J.A.; Fuglsang-Madsen, A.; Føns, S.; Laustsen, A.H. Toxin Neutralization Using Alternative Binding Proteins. Toxins 2019, 11, 53. [Google Scholar] [CrossRef] [PubMed]
- Lewin, M.R.; Carter, R.W.; Matteo, I.A.; Samuel, S.P.; Rao, S.; Fry, B.G.; Bickler, P.E. Varespladib in the Treatment of Snakebite Envenoming: Development History and Preclinical Evidence Supporting Advancement to Clinical Trials in Patients Bitten by Venomous Snakes. Toxins 2022, 14, 783. [Google Scholar] [CrossRef] [PubMed]
- Abouyannis, M.; FitzGerald, R.; Ngama, M.; Mwangudzah, H.; Nyambura, Y.; Ngome, S.; Riako, D.; Babu, L.; Lewa, F.; Else, L.; et al. TRUE-1: Trial of Repurposed Unithiol for snakebite Envenoming phase 1 (safety, tolerability, pharmacokinetics and pharmacodynamics in healthy Kenyan adults). Wellcome Open Res. 2022, 7, 90. [Google Scholar] [CrossRef] [PubMed]
- Vragniau, C.; Bufton, J.C.; Garzoni, F.; Stermann, E.; Rabi, F.; Terrat, C.; Guidetti, M.; Josserand, V.; Williams, M.; Woods, C.J.; et al. Synthetic self-assembling ADDomer platform for highly efficient vaccination by genetically encoded multiepitope display. Sci. Adv. 2019, 5, eaaw2853. [Google Scholar] [CrossRef] [PubMed]
- Besson, S.; Vragniau, C.; Vassal-Stermann, E.; Dagher, M.C.; Fender, P. The Adenovirus Dodecahedron: Beyond the Platonic Story. Viruses 2020, 12, 718. [Google Scholar] [CrossRef] [PubMed]
- Chevillard, C.; Amen, A.; Besson, S.; Hannani, D.; Bally, I.; Dettling, V.; Gout, E.; Moreau, C.J.; Buisson, M.; Gallet, S.; et al. Elicitation of potent SARS-CoV-2 neutralizing antibody responses through immunization with a versatile adenovirus-inspired multimerization platform. Mol. Ther. 2022, 30, 1913–1925. [Google Scholar] [CrossRef]
- Luo, C.; Yan, Q.; Huang, J.; Liu, J.; Li, Y.; Wu, K.; Li, B.; Zhao, M.; Fan, S.; Ding, H.; et al. Using Self-Assembling ADDomer Platform to Display B and T Epitopes of Type O Foot-and-Mouth Disease Virus. Viruses 2022, 14, 1810. [Google Scholar] [CrossRef]
- Buzas, D.; Bunzel, H.A.; Staufer, O.; Milodowski, E.J.; Edmunds, G.L.; Bufton, J.C.; Mateo, B.V.V.; Yadav, S.K.N.; Gupta, K.; Fletcher, C.; et al. In vitro generated antibodies guide thermostable ADDomer nanoparticle design for nasal vaccination and passive immunization against SARS-CoV-2. Antib. Ther. 2023, tbad024. [Google Scholar] [CrossRef]
- Zochowska, M.; Paca, A.; Schoehn, G.; Andrieu, J.-P.; Chroboczek, J.; Dublet, B.; Szolajska, E. Adenovirus Dodecahedron, as a Drug Delivery Vector. Jagetia GC, editor. PLoS ONE 2009, 4, e5569. [Google Scholar] [CrossRef] [PubMed]
- Buzas, D.; Sun, H.; Toelzer, C.; Yadav, S.K.N.; Borucu, U.; Gautam, G.; Gupta, K.; Bufton, J.; Capin, J.; Sessions, R.B.; et al. Engineering the ADDobody protein scaffold for generation of high-avidity ADDomer super-binders. BioRxiv. 2023. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Theakston, R.D.G.; Warrell, D.A. Confronting the Neglected Problem of Snake Bite Envenoming: The Need for a Global Partnership. PLoS Med. 2006, 3, e150. [Google Scholar] [CrossRef] [PubMed]
- Warrell, D.A.; Davidson, N.M.c.D.; Greenwood, B.M.; Ormerod, L.D.; Pope, H.M.; Watkins, B.J.; Prentice, C.R.M. Poisoning by bites of the saw-scaled or carpet viper (Echis carinatus) in Nigeria. Q. J. Med. 1977, 46, 33–62. [Google Scholar] [PubMed]
- Dingwoke, E.J.; Adamude, F.A.; Mohamed, G.; Klein, A.; Salihu, A.; Abubakar, M.S.; Sallau, A.B. Venom proteomic analysis of medically important Nigerian viper Echis ocellatus and Bitis arietans snake species. Biochem. Biophys. Rep. 2021, 28, 101164. [Google Scholar] [CrossRef]
- Rowan, E.G.; Harvey, A.L. Snake toxins from mamba venoms: Unique tools for the physiologist. Acta Chim. Slov. 2011, 58, 689–692. [Google Scholar]
- Lauridsen, L.P.; Laustsen, A.H.; Lomonte, B.; Gutiérrez, J.M. Toxicovenomics and antivenom profiling of the Eastern green mamba snake (Dendroaspis angusticeps). J. Proteom. 2016, 136, 248–261. [Google Scholar] [CrossRef]
- Laustsen, A.H.; Lomonte, B.; Lohse, B.; Fernández, J.; Gutiérrez, J.M. Unveiling the nature of black mamba (Dendroaspis polylepis) venom through venomics and antivenom immunoprofiling: Identification of key toxin targets for antivenom development. J. Proteom. 2015, 119, 126–142. [Google Scholar] [CrossRef]
- Quinton, L.; Demeure, K.; Dobson, R.; Gilles, N.; Gabelica, V.; De Pauw, E. New Method for Characterizing Highly Disulfide-Bridged Peptides in Complex Mixtures: Application to Toxin Identification from Crude Venoms. J. Proteome Res. 2007, 6, 3216–3223. [Google Scholar] [CrossRef]
- Dunbar, J.P.; Fort, A.; Redureau, D.; Sulpice, R.; Dugon, M.M.; Quinton, L. Venomics Approach Reveals a High Proportion of Lactrodectus-like Toxins in the Venom of the Noble False Widow Spider Steatoda nobilis. Toxins 2020, 12, 402. [Google Scholar] [CrossRef]
- Calvete, J.J.; Sanz, L.; Angulo, Y.; Lomonte, B.; Gutiérrez, J.M. Venoms, venomics, antivenomics. FEBS Lett. 2009, 583, 1736–1743. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.; Daly, N.L. Venomics: A Mini-Review. High Throughput 2018, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.L.; Viegas, M.F.; da Silva, S.L.; Soares, A.M.; Ramos, M.J.; Fernandes, P.A. The chemistry of snake venom and its medicinal potential. Nat. Rev. Chem. 2022, 6, 451–469. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wagstaff, S.C.; Wüster, W.; Cook, D.A.N.; Bolton, F.M.S.; King, S.I.; Pla, D.; Sanz, L.; Calvete, J.J.; Harrison, R.A. Medically important differences in snake venom composition are dictated by distinct postgenomic mechanisms. Proc. Natl. Acad. Sci. USA 2014, 111, 9205–9210. [Google Scholar] [CrossRef] [PubMed]
- Morsa, D.; Baiwir, D.; La Rocca, R.; Zimmerman, T.A.; Hanozin, E.; Grifnée, E.; Longuespée, R.; Meuwis, M.-A.; Smargiasso, N.; De Pauw, E.; et al. Multi-Enzymatic Limited Digestion: The Next-Generation Sequencing for Proteomics? J. Proteome Res. 2019, 18, 2501–2513. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Zhang, K.; Hendrie, C.; Liang, C.; Li, M.; Doherty-Kirby, A.; Lajoie, G. PEAKS: Powerful software for peptide de novo sequencing by tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2003, 17, 2337–2342. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, S.; Petras, D.; Engmark, M.; Süssmuth, R.D.; Whiteley, G.; Albulescu, L.-O.; Kazandjian, T.D.; Wagstaff, S.C.; Rowley, P.; Wüster, W.; et al. The medical threat of mamba envenoming in sub-Saharan Africa revealed by genus-wide analysis of venom composition, toxicity and antivenomics profiling of available antivenoms. J. Proteom. 2018, 172, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Lomonte, B.; León, G.; Alape-Girón, A.; Flores-Díaz, M.; Sanz, L.; Angulo, Y.; Calvete, J.J. Snake venomics and antivenomics: Proteomic tools in the design and control of antivenoms for the treatment of snakebite envenoming. J. Proteom. 2009, 72, 165–182. [Google Scholar] [CrossRef]
- Hanes, J.; Schaffitzel, C.; Knappik, A.; Plückthun, A. Picomolar affinity antibodies from a fully synthetic naive library selected and evolved by ribosome display. Nat. Biotechnol. 2000, 18, 1287–1292. [Google Scholar] [CrossRef]
- Dreier, B.; Plückthun, A. Rapid Selection of High-Affinity Antibody scFv Fragments Using Ribosome Display. Methods Mol. Biol. 2018, 1827, 235–268. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, I.; Egloff, P.; Hutter, C.A.J.; Kuhn, B.T.; Bräuer, P.; Newstead, S.; Dawson, R.J.P.; Geertsma, E.R.; Seeger, M.A. Generation of synthetic nanobodies against delicate proteins. Nat. Protoc. 2020, 15, 1707–1741. [Google Scholar] [CrossRef] [PubMed]
- Binz, H.K.; Amstutz, P.; Kohl, A.; Stumpp, M.T.; Briand, C.; Forrer, P.; Grütter, M.G.; Plückthun, A. High-affinity binders selected from designed ankyrin repeat protein libraries. Nat. Biotechnol. 2004, 22, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Zahnd, C.; Spinelli, S.; Luginbühl, B.; Amstutz, P.; Cambillau, C.; Plückthun, A. Directed in Vitro Evolution and Crystallographic Analysis of a Peptide-binding Single Chain Antibody Fragment (scFv) with Low Picomolar Affinity. J. Biol. Chem. 2004, 279, 18870–18877. [Google Scholar] [CrossRef] [PubMed]
- Rivera-de-Torre, E.; Rimbault, C.; Jenkins, T.P.; Sørensen, C.V.; Damsbo, A.; Saez, N.J.; Duhoo, Y.; Hackney, C.M.; Ellgaard, L.; Laustsen, A.H. Strategies for Heterologous Expression, Synthesis, and Purification of Animal Venom Toxins. Front. Bioeng. Biotechnol. 2022, 9, 811905. [Google Scholar] [CrossRef] [PubMed]
- Simonato, M.; Morbiato, L.; Zorzi, V.; Caccin, P.; Fernández, J.; Massimino, M.L.; de Laureto, P.P.; Tonello, F. Production in Escherichia coli, folding, purification and characterization of notexin with wild type sequence and with N-terminal and catalytic site mutations. Toxicon 2014, 88, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Chiou, Y.-L.; Lin, S.-R.; Chang, L.-S. Mutations on N-terminal region of Taiwan cobra phospholipase A2 result in structurally distorted effects. J. Pept. Sci. 2008, 14, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Turchetto, J.; Sequeira, A.F.; Ramond, L.; Peysson, F.; Brás, J.L.A.; Saez, N.J.; Duhoo, Y.; Blémont, M.; Guerreiro, C.I.P.D.; Quinton, L.; et al. High-throughput expression of animal venom toxins in Escherichia coli to generate a large library of oxidized disulphide-reticulated peptides for drug discovery. Microb. Cell Fact. 2017, 16, 6. [Google Scholar] [CrossRef]
- Sequeira, A.F.; Turchetto, J.; Saez, N.J.; Peysson, F.; Ramond, L.; Duhoo, Y.; Blémont, M.; Fernandes, V.O.; Gama, L.T.; Ferreira, L.M.A.; et al. Gene design, fusion technology and TEV cleavage conditions influence the purification of oxidized disulphide-rich venom peptides in Escherichia coli. Microb. Cell Fact. 2017, 16, 4. [Google Scholar] [CrossRef]
- Fairhead, M.; Howarth, M. Site-specific biotinylation of purified proteins using BirA. Methods Mol. Biol. 2015, 1266, 171–184. [Google Scholar] [CrossRef]
- Nguyen, V.D.; Hatahet, F.; Salo, K.E.; Enlund, E.; Zhang, C.; Ruddock, L.W. Pre-expression of a sulfhydryl oxidase significantly increases the yields of eukaryotic disulfide bond containing proteins expressed in the cytoplasm of E. coli. Microb. Cell Fact. 2011, 10, 1. [Google Scholar] [CrossRef]
- Fitzgerald, D.J.; Berger, P.; Schaffitzel, C.; Yamada, K.; Richmond, T.J.; Berger, I. Protein complex expression by using multigene baculoviral vectors. Nat. Methods 2006, 3, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Tessier, D.C.; Thomas, D.Y.; Khouri, H.E.; Laliberté, F.; Vernet, T. Enhanced secretion from insect cells of a foreign protein fused to the honeybee melittin signal peptide. Gene 1991, 98, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Wagstaff, S.C.; Laing, G.D.; Theakston, R.D.G.; Papaspyridis, C.; Harrison, R.A. Bioinformatics and Multiepitope DNA Immunization to Design Rational Snake Antivenom. PLoS Med. 2006, 3, e184. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Vargas, M.; Segura, Á.; Herrera, M.; Villalta, M.; Solano, G.; Sánchez, A.; Herrera, C.; León, G. In Vitro Tests for Assessing the Neutralizing Ability of Snake Antivenoms: Toward the 3Rs Principles. Front. Immunol. 2021, 11, 617429. Available online: https://www.frontiersin.org/articles/10.3389/fimmu.2020.617429 (accessed on 13 December 2022). [CrossRef] [PubMed]
- Albulescu, L.-O.; Xie, C.; Ainsworth, S.; Alsolaiss, J.; Crittenden, E.; Dawson, C.A.; Softley, R.; Bartlett, K.E.; Harrison, R.A.; Kool, J.; et al. A therapeutic combination of two small molecule toxin inhibitors provides broad preclinical efficacy against viper snakebite. Nat. Commun. 2020, 11, 6094. [Google Scholar] [CrossRef]
- Macêdo, J.K.A.; Fox, J.W. Biological Activities and Assays of the Snake Venom Metalloproteinases (SVMPs). In Venom Genomics and Proteomics; Gopalakrishnakone, P., Calvete, J.J., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 211–238. [Google Scholar] [CrossRef]
- Hiu, J.J.; Yap, M.K.K. Cytotoxicity of snake venom enzymatic toxins: Phospholipase A2 and l-amino acid oxidase. Biochem. Soc. Trans. 2020, 48, 719–731. [Google Scholar] [CrossRef]
- Conlon, J.M.; Attoub, S.; Arafat, H.; Mechkarska, M.; Casewell, N.R.; Harrison, R.A.; Calvete, J.J. Cytotoxic activities of [Ser49]phospholipase A2 from the venom of the saw-scaled vipers Echis ocellatus, Echis pyramidum leakeyi, Echis carinatus sochureki, and Echis coloratus. Toxicon 2013, 71, 96–104. [Google Scholar] [CrossRef]
- Still, K.B.M.; Nandlal, R.S.S.; Slagboom, J.; Somsen, G.W.; Casewell, N.R.; Kool, J. Multipurpose HTS Coagulation Analysis: Assay Development and Assessment of Coagulopathic Snake Venoms. Toxins 2017, 9, 382. [Google Scholar] [CrossRef]
- Navred, K.; Martin, M.; Ekdahl, L.; Zetterberg, E.; Andersson, N.G.; Strandberg, K.; Norstrom, E. A simplified flow cytometric method for detection of inherited platelet disorders—A comparison to the gold standard light transmission aggregometry. PLoS ONE 2019, 14, e0211130. [Google Scholar] [CrossRef]
- Kolvekar, N.; Bhattacharya, N.; Sarkar, A.; Chakrabarty, D. How snake venom disintegrins affect platelet aggregation and cancer proliferation. Toxicon 2023, 221, 106982. [Google Scholar] [CrossRef]
- Patel, R.N.; Clare, R.H.; Ledsgaard, L.; Nys, M.; Kool, J.; Laustsen, A.H.; Ulens, C.; Casewell, N.R. An in vitro assay to investigate venom neurotoxin activity on muscle-type nicotinic acetylcholine receptor activation and for the discovery of toxin-inhibitory molecules. Biochem. Pharmacol. 2023, 216, 115758. [Google Scholar] [CrossRef] [PubMed]
- Slagboom, J.; Otvos, R.A.; Cardoso, F.C.; Iyer, J.; Visser, J.C.; van Doodewaerd, B.R.; McCleary, R.J.; Niessen, W.M.; Somsen, G.W.; Lewis, R.J.; et al. Neurotoxicity fingerprinting of venoms using on-line microfluidic AChBP profiling. Toxicon 2018, 148, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Servent, D.; Winckler-Dietrich, V.; Hu, H.-Y.; Kessler, P.; Drevet, P.; Bertrand, D.; Ménez, A. Only Snake Curaremimetic Toxins with a Fifth Disulfide Bond Have High Affinity for the Neuronal α7 Nicotinic Receptor. J. Biol. Chem. 1997, 272, 24279–24286. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L. Twenty years of dendrotoxins. Toxicon 2001, 39, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Yajuan, X.; Xin, L.; Zhiyuan, L. A Comparison of the Performance and Application Differences between Manual and Automated Patch-Clamp Techniques. Curr. Chem. Genom. 2012, 6, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Theakston, R.D.; Reid, H.A. Development of simple standard assay procedures for the characterization of snake venom. Bull. World Health Organ. 1983, 61, 949–956. [Google Scholar] [PubMed]
- Knudsen, C.; Casewell, N.R.; Lomonte, B.; Gutiérrez, J.M.; Vaiyapuri, S.; Laustsen, A.H. Novel Snakebite Therapeutics Must Be Tested in Appropriate Rescue Models to Robustly Assess Their Preclinical Efficacy. Toxins 2020, 12, 528. [Google Scholar] [CrossRef]
- Ikonomou, L.; Schneider, Y.-J.; Agathos, S.N. Insect cell culture for industrial production of recombinant proteins. Appl. Microbiol. Biotechnol. 2003, 62, 1–20. [Google Scholar] [CrossRef]
- Yee, C.M.; Zak, A.J.; Hill, B.D.; Wen, F. The Coming Age of Insect Cells for Manufacturing and Development of Protein Therapeutics. Ind. Eng. Chem. Res. 2018, 57, 10061–10070. [Google Scholar] [CrossRef]
- Maghodia, A.B.; Geisler, C.; Jarvis, D.L. Characterization of an Sf-rhabdovirus-negative Spodoptera frugiperda cell line as an alternative host for recombinant protein production in the baculovirus-insect cell system. Protein Expr. Purif. 2016, 122, 45–55. [Google Scholar] [CrossRef]
- Maghodia, A.B.; Geisler, C.; Jarvis, D.L. A new nodavirus-negative Trichoplusia ni cell line for baculovirus-mediated protein production. Biotechnol. Bioeng. 2020, 117, 3248–3264. [Google Scholar] [CrossRef] [PubMed]
- Fuenmayor, J.; Gòdia, F.; Cervera, L. Production of virus-like particles for vaccines. New Biotechnol. 2017, 39, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Roldão, A.; Mellado, M.C.M.; Castilho, L.R.; Carrondo, M.J.; Alves, P.M. Virus-like particles in vaccine development. Expert Rev. Vaccines 2010, 9, 1149–1176. [Google Scholar] [CrossRef] [PubMed]
- Junne, S.; Neubauer, P. How scalable and suitable are single-use bioreactors? Curr. Opin. Biotechnol. 2018, 53, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Gerstweiler, L.; Bi, J.; Middelberg, A.P.J. Continuous downstream bioprocessing for intensified manufacture of biopharmaceuticals and antibodies. Chem. Eng. Sci. 2021, 231, 116272. [Google Scholar] [CrossRef]
- Gerstweiler, L.; Billakanti, J.; Bi, J.; Middelberg, A.P.J. An integrated and continuous downstream process for microbial virus-like particle vaccine biomanufacture. Biotech. Bioeng. 2022, 119, 2122–2133. [Google Scholar] [CrossRef]
- Mvundura, M.; Kien, V.D.; Nga, N.T.; Robertson, J.; Van Cuong, N.; Tung, H.T.; Hong, D.T.; Levin, C. How much does it cost to get a dose of vaccine to the service delivery location? Empirical evidence from Vietnam’s Expanded Program on Immunization. Vaccine 2014, 32, 834–838. [Google Scholar] [CrossRef]
- Mvundura, M.; Lorenson, K.; Chweya, A.; Kigadye, R.; Bartholomew, K.; Makame, M.; Lennon, T.P.; Mwangi, S.; Kirika, L.; Kamau, P.; et al. Estimating the costs of the vaccine supply chain and service delivery for selected districts in Kenya and Tanzania. Vaccine 2015, 33, 2697–2703. [Google Scholar] [CrossRef]
- Kumar, R.; Srivastava, V.; Baindara, P.; Ahmad, A. Thermostable vaccines: An innovative concept in vaccine development. Expert Rev. Vaccines 2022, 21, 811–824. [Google Scholar] [CrossRef]
- Daniell, H.; Rai, V.; Xiao, Y. Cold chain and virus-free oral polio booster vaccine made in lettuce chloroplasts confers protection against all three poliovirus serotypes. Plant Biotechnol. J. 2019, 17, 1357–1368. [Google Scholar] [CrossRef]
- Guo, M.; Li, J.; Teng, Z.; Ren, M.; Dong, H.; Zhang, Y.; Ru, J.; Du, P.; Sun, S.; Guo, H. Four Simple Biomimetic Mineralization Methods to Improve the Thermostability and Immunogenicity of Virus-like Particles as a Vaccine against Foot-and-Mouth Disease. Vaccines 2021, 9, 891. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, F.M.C.; Petrovajová, D.; Horňáková, T. Viral vaccine stabilizers: Status and trends. Acta Virol. 2017, 61, 231–239. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menzies, S.K.; Arinto-Garcia, R.; Amorim, F.G.; Cardoso, I.A.; Abada, C.; Crasset, T.; Durbesson, F.; Edge, R.J.; El-Kazzi, P.; Hall, S.; et al. ADDovenom: Thermostable Protein-Based ADDomer Nanoparticles as New Therapeutics for Snakebite Envenoming. Toxins 2023, 15, 673. https://doi.org/10.3390/toxins15120673
Menzies SK, Arinto-Garcia R, Amorim FG, Cardoso IA, Abada C, Crasset T, Durbesson F, Edge RJ, El-Kazzi P, Hall S, et al. ADDovenom: Thermostable Protein-Based ADDomer Nanoparticles as New Therapeutics for Snakebite Envenoming. Toxins. 2023; 15(12):673. https://doi.org/10.3390/toxins15120673
Chicago/Turabian StyleMenzies, Stefanie K., Raquel Arinto-Garcia, Fernanda Gobbi Amorim, Iara Aimê Cardoso, Camille Abada, Thomas Crasset, Fabien Durbesson, Rebecca J. Edge, Priscila El-Kazzi, Sophie Hall, and et al. 2023. "ADDovenom: Thermostable Protein-Based ADDomer Nanoparticles as New Therapeutics for Snakebite Envenoming" Toxins 15, no. 12: 673. https://doi.org/10.3390/toxins15120673
APA StyleMenzies, S. K., Arinto-Garcia, R., Amorim, F. G., Cardoso, I. A., Abada, C., Crasset, T., Durbesson, F., Edge, R. J., El-Kazzi, P., Hall, S., Redureau, D., Stenner, R., Boldrini-França, J., Sun, H., Roldão, A., Alves, P. M., Harrison, R. A., Vincentelli, R., Berger, I., ... Schaffitzel, C. (2023). ADDovenom: Thermostable Protein-Based ADDomer Nanoparticles as New Therapeutics for Snakebite Envenoming. Toxins, 15(12), 673. https://doi.org/10.3390/toxins15120673





