Antibodies as Snakebite Antivenoms: Past and Future
Abstract
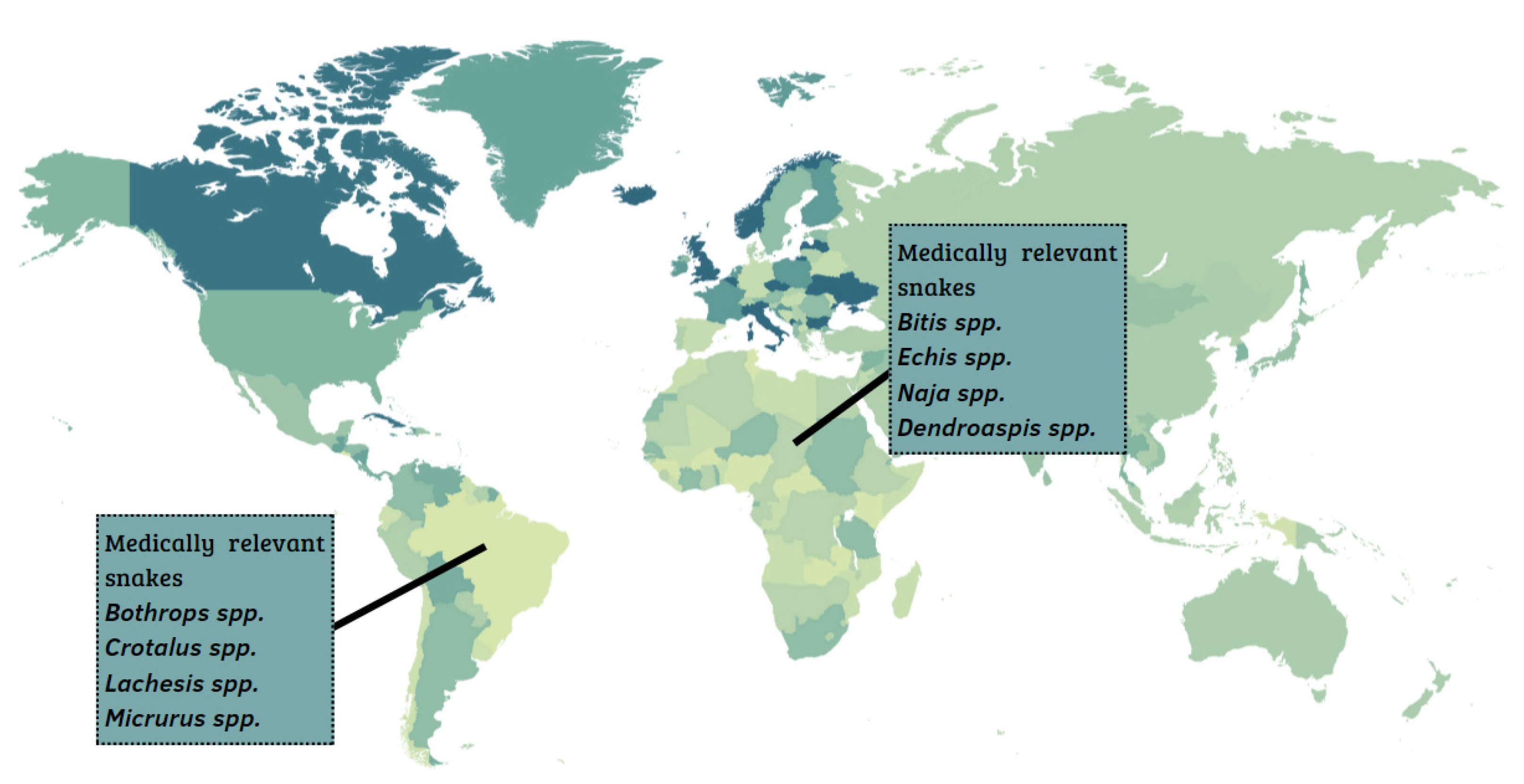
:1. Snakebites and Serum Therapy
2. Immunoglobulins for Therapeutic and Diagnostic Applications
3. Classic Methods for Improving Serum Therapy
4. Next-Generation Antivenom: Monoclonal Antibodies and Their Fragments
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kasturiratne, A.; Wickremasinghe, A.R.; de Silva, N.; Gunawardena, N.K.; Pathmeswaran, A.; Premaratna, R.; Savioli, L.; Lalloo, D.G.; de Silva, H.J. The Global Burden of Snakebite: A Literature Analysis and Modelling Based on Regional Estimates of Envenoming and Deaths. PLoS Med. 2008, 5, e218. [Google Scholar] [CrossRef]
- Chippaux, J.-P. Snakebite Envenomation Turns Again into a Neglected Tropical Disease! J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 38. [Google Scholar] [CrossRef] [PubMed]
- Seifert, S.A.; Armitage, J.O.; Sanchez, E.E. Snake Envenomation. N. Engl. J. Med. 2022, 386, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Calvete, J.J.; Habib, A.G.; Harrison, R.A.; Williams, D.J.; Warrell, D.A. Snakebite Envenoming. Nat. Rev. Dis. Primers 2017, 3, 17063. [Google Scholar] [CrossRef] [PubMed]
- WHO Snakebite: WHO Targets 50% Reduction in Deaths and Disabilities. Available online: https://www.who.int/news/item/06-05-2019-snakebite-who-targets-50-reduction-in-deaths-and-disabilities (accessed on 23 August 2022).
- Mp, A.; Mamman, M.; Po, Y.; Mg, M.; Ada, G.; Io, A.; Pe, U. Overview of the Venomous Snakes in West Africa, Clinical Manifestation, Diagnosis and Treatment. J. Med. Biol. 2019, 1, 11–26. [Google Scholar]
- World Health Organization. Guidelines for the Production, Control and Regulation of Snake Antivenom Immunoglobulins; WHO Press: Geneva, Switzerland, 2016; Available online: https://extranet.who.int/pqweb/vaccines-production-control-and-regulation-snke-antivenom-immunoglobulins (accessed on 14 July 2022).
- Gutiérrez, J.M. Preclinical Assessment of the Neutralizing Efficacy of Snake Antivenoms in Latin America and the Caribbean: A Review. Toxicon 2018, 146, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Currier, R.B.; Harrison, R.A.; Rowley, P.D.; Laing, G.D.; Wagstaff, S.C. Intra-Specific Variation in Venom of the African Puff Adder (Bitis arietans): Differential Expression and Activity of Snake Venom Metalloproteinases (SVMPs). Toxicon 2010, 55, 864–873. [Google Scholar] [CrossRef]
- Warrell, D.A.; Ormerod, L.D.; Davidson, N.M. Bites by Puff-Adder (Bitis arietans) in Nigeria, and Value of Antivenom. Br. Med. J. 1975, 4, 697–700. [Google Scholar] [CrossRef]
- Bey, T.A.; Boyer, L.V.; Walter, F.G.; McNally, J.; Desai, H. Exotic Snakebite: Envenomation by an African Puff Adder (Bitis arietans). J. Emerg. Med. 1997, 15, 827–831. [Google Scholar] [CrossRef]
- Závada, J.; Valenta, J.; Kopecký, O.; Stach, Z.; Leden, P. Black Mamba Dendroaspis Polylepis Bite: A Case Report. Prague Med. Rep. 2011, 112, 298–304. [Google Scholar] [PubMed]
- Laustsen, A.H.; Lomonte, B.; Lohse, B.; Fernández, J.; Gutiérrez, J.M. Unveiling the Nature of Black Mamba (Dendroaspis polylepis) Venom through Venomics and Antivenom Immunoprofiling: Identification of Key Toxin Targets for Antivenom Development. J. Proteom. 2015, 119, 126–142. [Google Scholar] [CrossRef]
- The Reptile Database. Available online: http://reptile-database.org/ (accessed on 23 August 2022).
- Amr, Z.S.; Abu Baker, M.A.; Warrell, D.A. Terrestrial Venomous Snakes and Snakebites in the Arab Countries of the Middle East. Toxicon 2020, 177, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Stock, R.P.; Massougbodji, A.; Alagón, A.; Chippaux, J.-P. Bringing Antivenoms to Sub-Saharan Africa. Nat. Biotechnol. 2007, 25, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.-P. Estimate of the Burden of Snakebites in Sub-Saharan Africa: A Meta-Analytic Approach. Toxicon 2011, 57, 586–599. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Ciufo, S.; Domrachev, M.; Hotton, C.L.; Kannan, S.; Khovanskaya, R.; Leipe, D.; Mcveigh, R.; O’Neill, K.; Robbertse, B.; et al. NCBI Taxonomy: A Comprehensive Update on Curation, Resources and Tools. Database 2020, 1–21. [Google Scholar] [CrossRef]
- Barlow, A.; Pook, C.E.; Harrison, R.A.; Wüster, W. Coevolution of Diet and Prey-Specific Venom Activity Supports the Role of Selection in Snake Venom Evolution. Proc. R. Soc. B Biol. Sci. 2009, 276, 2443–2449. [Google Scholar] [CrossRef]
- Melgarejo, A.R.; Cardoso, J.L.C.; França, F.O.S.; Wen, F.H.; Málaque, C.M.S.; Haddad, V., Jr. Serpentes peçonhentas do Brasil. In Animais Peçonhentos no Brasil–Biologia, Clínica e Terapêutica Dos Acidentes, 2nd ed.; Sarvier: São Paulo, Brazil, 2008; pp. 42–70. [Google Scholar]
- Gren, E.C.K.; Kitano, E.S.; Andrade-Silva, D.; Iwai, L.K.; Reis, M.S.; Menezes, M.C.; Serrano, S.M.T. Comparative Analysis of the High Molecular Mass Subproteomes of Eight Bothrops Snake Venoms. Comp. Biochem. Physiol. Part D Genom. Proteom. 2019, 30, 113–121. [Google Scholar] [CrossRef]
- Gonçalves-Machado, L.; Pla, D.; Sanz, L.; Jorge, R.J.B.; Leitão-De-Araújo, M.; Alves, M.L.M.; Alvares, D.J.; de Miranda, J.; Nowatzki, J.; de Morais-Zani, K.; et al. Combined Venomics, Venom Gland Transcriptomics, Bioactivities, and Antivenomics of Two Bothrops jararaca Populations from Geographic Isolated Regions within the Brazilian Atlantic Rainforest. J. Proteom. 2016, 135, 73–89. [Google Scholar] [CrossRef]
- Nicolau, C.A.; Carvalho, P.C.; Junqueira-de-Azevedo, I.L.M.; Teixeira-Ferreira, A.; Junqueira, M.; Perales, J.; Neves-Ferreira, A.G.C.; Valente, R.H. An In-Depth Snake Venom Proteopeptidome Characterization: Benchmarking Bothrops jararaca. J. Proteom. 2017, 151, 214–231. [Google Scholar] [CrossRef]
- Cardoso, J.L.C.; França, F.O.S.; Wen, F.H.; Málaque, C.M.S.; Haddad Jr., V. Venomous Animals in Brazil: Biology, Clinic and Therapeutics of Envenomations. Rev. Inst. Med. Trop. Sao Paulo 2003, 45, 338. [Google Scholar] [CrossRef]
- Sousa, L.F.; Portes-Junior, J.A.; Nicolau, C.A.; Bernardoni, J.L.; Nishiyama-Jr, M.Y.; Amazonas, D.R.; Freitas-de-Sousa, L.A.; Mourão, R.H.; Chalkidis, H.M.; Valente, R.H.; et al. Functional Proteomic Analyses of Bothrops atrox Venom Reveals Phenotypes Associated with Habitat Variation in the Amazon. J. Proteom. 2017, 159, 32–46. [Google Scholar] [CrossRef]
- Azevedo-Marques, M.M.; Cupo, P.; Coimbra, T.M.; Hering, S.E.; Rossi, M.A.; Laure, C.J. Myonecrosis, Myoglobinuria and Acute Renal Failure Induced by South American Rattlesnake (Crotalus durissus Terrificus) Envenomation in Brazil. Toxicon 1985, 23, 631–636. [Google Scholar] [CrossRef]
- Radis-Baptista, G.; Kerkis, I. Crotamine, a Small Basic Polypeptide Myotoxin from Rattlesnake Venom with Cell-Penetrating Properties. Curr. Pharm. Des. 2011, 17, 4351–4361. [Google Scholar] [CrossRef]
- Barrabin, H.; Martiarena, J.L.; Vidal, J.C.; Barrio, A. Isolation and characterization of gyroxin from Crotalus durissus Terrificus venom. In Toxins Animal, Plant and Microbial; Rosemberg, P., Ed.; Pergamon Press: Oxford, UK, 1978; pp. 113–133. [Google Scholar]
- Barrio, A. Gyrotoxin, a New Neurotoxin of Crotales durissus terrificus Venom. In Acta Physiologica Latinoamericana; Assn Latinoamer Cienc Fisiol: Buenos Aires, Argentina, 1961; Volume 11. [Google Scholar]
- Baudou, F.G.; Rodriguez, J.P.; Fusco, L.; de Roodt, A.R.; de Marzi, M.C.; Leiva, L. South American Snake Venoms with Abundant Neurotoxic Components. Composition and Toxicological Properties. A Literature Review. Acta Trop. 2021, 224, 106119. [Google Scholar] [CrossRef]
- Walter, F.G.; Stolz, U.; Shirazi, F.; McNally, J. Epidemiology of Severe and Fatal Rattlesnake Bites Published in the American Association of Poison Control Centers’ Annual Reports. Clin. Toxicol 2009, 47, 663–669. [Google Scholar] [CrossRef]
- Tasoulis, T.; Pukala, T.L.; Isbister, G.K. Investigating Toxin Diversity and Abundance in Snake Venom Proteomes. Front Pharmacol. 2022, 12, 768015. [Google Scholar] [CrossRef]
- Behring, E.V.; Kitasato, S. Ueber das Zustandekommen der Diphtherie-Immunität und der Tetanus-Immunität bei Thieren. Mol. Immunol. 2013, 28, 1319–1320. [Google Scholar]
- Hawgood, B.J. Albert Calmette 1863–1933 Fondateur de La Sérothérapie Antivenimeuse. In Annales de l’Institut Pasteur/Actualites; Elsevier Masson: Paris, France, 1999; Volume 10, pp. 139–146. [Google Scholar] [CrossRef]
- Bochner, R.; Goyffon, M. L’oeuvre Scientifique De Césaire Phisalix (1852–1906) Découvreur Du Sérum Antivenimeux. Bulletin De La Société Herpétologique De France 2007, 123, 32. [Google Scholar]
- Lahariya, C. A Brief History of Vaccines & Vaccination in India. Indian J. Med. Res. 2014, 139, 491–511. [Google Scholar]
- Brazil, V. Memória Histórica do Instituto Butantan; Elvino Pocai: São Paulo, Brazil, 1941; pp. 105–112. [Google Scholar]
- Brazil, V.; Maibon., J. La Défense Contre L’ophidisme, 2nd ed.; Saint-Paul, Impr. Pocai-Weiss: São Paulo, Brazil, 1914. [Google Scholar]
- Cardoso, J.L.; Fan, H.W.; França, F.O.; Jorge, M.T.; Leite, R.P.; Nishioka, S.A.; Avila, A.; Sano-Martins, I.S.; Tomy, S.C.; Santoro, M.L.; et al. Randomized comparative trial of three antivenoms in the treatment of envenoming by lance-headed vipers (Bothrops jararaca) in São Paulo, Brazil. QJM Int. J. Med. 1993, 86, 315–325. [Google Scholar]
- Kang, T.H.; Jung, S.T. Boosting therapeutic potency of antibodies by taming Fc domain functions. Exp. Mol. Med. 2019, 51, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Charles, A.; Janeway, J.; Travers, P.; Walport, M.; Shlomchik, M.J. Immunobiology: The Immune System in Health and Disease, 5th ed.; Routledge: Oxfordshire, UK, 2001. [Google Scholar]
- Lu, R.-M.; Hwang, Y.-C.; Liu, I.-J.; Lee, C.-C.; Tsai, H.-Z.; Li, H.-J.; Wu, H.-C. Development of Therapeutic Antibodies for the Treatment of Diseases. J. Biomed. Sci. 2020, 27, 1. [Google Scholar] [CrossRef]
- Monoclonal Antibodies (MAbS) Market Analysis, Size and Trends Global Forecast to 2022–2030. Available online: https://www.grandviewresearch.com/industry-analysis/monoclonal-antibodies-market (accessed on 14 July 2022).
- Kaplon, H.; Chenoweth, A.; Crescioli, S.; Reichert, J.M. Antibodies to Watch in 2022. In MAbs; Taylor & Francis: Oxfordshire, UK, 2022; Volume 14. [Google Scholar] [CrossRef]
- Xenaki, K.T.; Oliveira, S.; van Bergen en Henegouwen, P.M.P. Antibody or Antibody Fragments: Implications for Molecular Imaging and Targeted Therapy of Solid Tumors. Front. Immunol. 2017, 8, 1287. [Google Scholar] [CrossRef] [Green Version]
- Nelson, A.L.; Reichert, J.M. Development Trends for Therapeutic Antibody Fragments. Nat. Biotechnol. 2009, 27, 331–337. [Google Scholar] [CrossRef]
- Steeland, S.; Vandenbroucke, R.E.; Libert, C. Nanobodies as Therapeutics: Big Opportunities for Small Antibodies. Drug Discov. Today 2016, 21, 1076–1113. [Google Scholar] [CrossRef]
- Yusakul, G.; Sakamoto, S.; Pongkitwitoon, B.; Tanaka, H.; Morimoto, S. Effect of Linker Length between Variable Domains of Single Chain Variable Fragment Antibody against Daidzin on Its Reactivity. Biosci. Biotechnol. Biochem. 2016, 80, 1306–1312. [Google Scholar] [CrossRef]
- The Antibody Society Antibody Therapeutics Approved or in Regulatory Review in the EU or US. Available online: https://www.antibodysociety.org/resources/approved-antibodies/ (accessed on 23 August 2022).
- Charpentier, I.; Pillet, L.; Karlsson, E.; Couderc, J.; Ménez, A. Recognition of the Acetylcholine Receptor Binding Site of a Long-Chain Neurotoxin by Toxin-Specific Monoclonal Antibodies. J. Mol. Recognit. 1990, 3, 74–81. [Google Scholar] [CrossRef]
- Engmark, M.; Lomonte, B.; Gutiérrez, J.M.; Laustsen, A.H.; de Masi, F.; Andersen, M.R.; Lund, O. Cross-Recognition of a Pit Viper (Crotalinae) Polyspecific Antivenom Explored through High-Density Peptide Microarray Epitope Mapping. PLoS Negl. Trop. Dis. 2017, 11, e0005768. [Google Scholar] [CrossRef]
- Lafaye, P.; Choumet, V.; Demangel, C.; Bon, C.; Mazié, J.-C. Biologically Active Human Anti-Crotoxin ScFv Isolated from a Semi-Synthetic Phage Library. Immunotechnology 1997, 3, 117–125. [Google Scholar] [CrossRef]
- Laustsen, A.H.; María Gutiérrez, J.; Knudsen, C.; Johansen, K.H.; Bermúdez-Méndez, E.; Cerni, F.A.; Jürgensen, J.A.; Ledsgaard, L.; Martos-Esteban, A.; Øhlenschlæger, M.; et al. Pros and Cons of Different Therapeutic Antibody Formats for Recombinant Antivenom Development. Toxicon 2018, 146, 151–175. [Google Scholar] [CrossRef]
- Visser, L.E.; Kyei-Faried, S.; Belcher, D.W.; Geelhoed, D.W.; van Leeuwen, J.S.; van Roosmalen, J. Failure of a New Antivenom to Treat Echis ocellatus Snake Bite in Rural Ghana: The Importance of Quality Surveillance. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 445–450. [Google Scholar] [CrossRef]
- Habib, A.G.; Onayade, A.A.; Yakubu, A.; Nasidi, A.; Theakston, R.D.G.; Smith, D.C.; Daudu, I.J.; Warrell, D.A.; Meyer, W.P. First Clinical Experiences with a New Ovine Fab Echis ocellatus Snake Bite Antivenom in Nigeria: Randomized Comparative Trial with Institute Pasteur Serum (Ipser) Africa Antivenom. Am. J. Trop. Med. Hyg. 1997, 56, 291–300. [Google Scholar] [CrossRef]
- León, G.; Herrera, M.; Segura, Á.; Villalta, M.; Vargas, M.; Gutiérrez, J.M. Pathogenic Mechanisms Underlying Adverse Reactions Induced by Intravenous Administration of Snake Antivenoms. Toxicon 2013, 76, 63–76. [Google Scholar] [CrossRef]
- Squaiella-Baptistão, C.C.; Gutiérrez, J.M.; Tambourgi, D.V.; Marcelino, J.R.; Ribeiro da Cunha, L.E. Anticomplementary Activity of Horse IgG and F(Ab’)2 Antivenoms. Am. J. Trop. Med. Hyg. 2014, 90, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Battellino, C.; Piazza, R.; da Silva, A.M.M.; Cury, Y.; Farsky, S.H.P. Assessment of Efficacy of Bothropic Antivenom Therapy on Microcirculatory Effects Induced by Bothrops jararaca Snake Venom. Toxicon 2003, 41, 583–593. [Google Scholar] [CrossRef]
- Williams, D.J.; Gutiérrez, J.-M.; Calvete, J.J.; Wüster, W.; Ratanabanangkoon, K.; Paiva, O.; Brown, N.I.; Casewell, N.R.; Harrison, R.A.; Rowley, P.D.; et al. Ending the Drought: New Strategies for Improving the Flow of Affordable, Effective Antivenoms in Asia and Africa. J. Proteom. 2011, 74, 1735–1767. [Google Scholar] [CrossRef]
- Fasoli, E.; Sanz, L.; Wagstaff, S.; Harrison, R.A.; Righetti, P.G.; Calvete, J.J. Exploring the Venom Proteome of the African Puff Adder, Bitis arietans, Using a Combinatorial Peptide Ligand Library Approach at Different PHs. J. Proteom. 2010, 73, 932–942. [Google Scholar] [CrossRef]
- Ferreira, L.A.F.; Henriques, O.B.; Andreoni, A.A.S.; Vital, G.R.F.; Campos, M.M.C.; Habermehl, G.G.; de Moraes, V.L.G. Antivenom and Biological Effects of Ar-Turmerone Isolated from Curcuma Longa (Zingiberaceae). Toxicon 1992, 30, 1211–1218. [Google Scholar] [CrossRef]
- Silva, W.D.D.; Guidolin, R.; Raw, I.; Higashi, H.G.; Caricati, C.P.; Morais, J.F.; Lima, M.L.S.; Yamaguchi, I.K.; Nishikawa, A.K.; Stephano, M.A.; et al. Cross-reactivity of horse monovalent antivenoms to venoms of ten Bothrops species. Mem. Inst. Butantan 1989, 51, 153–168. [Google Scholar]
- Sousa, L.F.; Nicolau, C.A.; Peixoto, P.S.; Bernardoni, J.L.; Oliveira, S.S.; Portes-Junior, J.A.; Mourão, R.H.V.; Lima-dos-Santos, I.; Sano-Martins, I.S.; Chalkidis, H.M.; et al. Comparison of Phylogeny, Venom Composition and Neutralization by Antivenom in Diverse Species of Bothrops Complex. PLoS Negl. Trop. Dis. 2013, 7, e2442. [Google Scholar] [CrossRef]
- Guidolin, F.R.; Caricati, C.P.; Marcelino, J.R.; da Silva, W.D. Development of Equine IgG Antivenoms against Major Snake Groups in Mozambique. PLoS Negl. Trop. Dis. 2016, 10, e0004325. [Google Scholar] [CrossRef] [PubMed]
- Tahis, L.D.S.; Magnóli, F.C.; Dias da Silva, W. Characterization of a Hemorrhage-Inducing Component Present in Bitis arietans Venom. Afr. J. Biotechnol. 2015, 14, 999–1008. [Google Scholar] [CrossRef]
- Freitas, T.V.; Fortes-Dias, C.L.; Diniz, C.R.; Velarde, D.T.; Freitas, C.F. Immunization of Horses with Crotalus durissus Terrificus (South American Rattlesnake) Venom. A Comparison of Four Different Procedures. Braz. J. Med. Biol. Res. 1991, 24, 281–290. [Google Scholar] [PubMed]
- Almeida, C.M.C.; Kanashiro, M.M.; Rangel Filho, F.B.; Mata, M.F.R.; Kipnis, T.L.; Dias da Silva, W. Development of Snake Antivenom Antibodies in Chickens and Their Purification from Yolk. Vet. Rec. 1998, 143, 579–584. [Google Scholar] [CrossRef]
- Guidlolin, R.G.; Marcelino, R.M.; Gondo, H.H.; Morais, J.F.; Ferreira, R.A.; Silva, C.L.; Kipnis, T.L.; Silva, J.A.; Fafetine, J.; da Silva, W.D. Polyvalent horse F(Ab’)2 snake antivenom: Development of process to produce polyvalent horse F(Ab’)2 antibodies anti-african snake venom. Afr. J. Biotechnol. 2010, 9, 2446–2455. [Google Scholar]
- Alvarenga, L.; Zahid, M.; Tommaso, A.; Juste, M.; Aubrey, N.; Billiald, P.; Muzard, J. Engineering Venom’s Toxin-Neutralizing Antibody Fragments and Its Therapeutic Potential. Toxins 2014, 6, 2541–2567. [Google Scholar] [CrossRef]
- Guidolin, F.R.; Tambourgi, D.V.; Guidolin, R.; Marcelino, J.R.; Okamoto, C.K.; Magnoli, F.C.; Queiroz, G.P.; Dias da Silva, W. Characterization of Anti-Crotalic Antibodies. Toxicon 2013, 66, 7–17. [Google Scholar] [CrossRef]
- Kuniyoshi, A.K.; Kodama, R.T.; Cajado-Carvalho, D.; Iwai, L.K.; Kitano, E.; da Silva, C.C.F.; Duzzi, B.; Dias da Silva, W.; Portaro, F.C. Experimental Antivenom against Serine Proteases from the Bothrops jararaca Venom Obtained in Mice, and Its Comparison with the Antibothropic Serum from the Butantan Institute. Toxicon 2019, 169, 59–67. [Google Scholar] [CrossRef]
- Laustsen, A.H. Toxin Synergism in Snake Venoms. Toxin Rev. 2016, 35, 165–170. [Google Scholar] [CrossRef]
- Calvete, J.J. Venomics: Integrative Venom Proteomics and Beyond. Biochem. J. 2017, 474, 611–634. [Google Scholar] [CrossRef]
- Boulain, J.C.; Menez, A.; Couderc, J.; Faure, G.; Liacopoulos, P.; Fromageot, P. Neutralizing Monoclonal Antibody Specific for Naja nigricollis Toxin.Alpha.: Preparation, Characterization and Localization of the Antigenic Binding Site. Biochemistry 1982, 21, 2910–2915. [Google Scholar] [CrossRef]
- Jianxin, M.; John, T.R.; Kaiser, I.I. Specificity and Binding Affinity of an Anti-Crotoxin Combinatorial Antibody Selected from a Phage-Displayed Library. Biochem. Pharm. 1995, 50, 1969–1977. [Google Scholar] [CrossRef]
- Hansel, T.T.; Kropshofer, H.; Singer, T.; Mitchell, J.A.; George, A.J.T. The Safety and Side Effects of Monoclonal Antibodies. Nat. Rev. Drug Discov. 2010, 9, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Morine, N.; Matsuda, S.; Terada, K.; Eto, A.; Ishida, I.; Oku, H. Neutralization of Hemorrhagic Snake Venom Metalloproteinase HR1a from Protobothrops flavoviridis by Human Monoclonal Antibody. Toxicon 2008, 51, 345–352. [Google Scholar] [CrossRef]
- Iddon, D.; Hommel, M.; Theakston, R.D.G. Characterisation of a Monoclonal Antibody Capable of Neutralising the Haemorrhagic Activity of West African Echis carinatus (Carpet Viper) Venom. Toxicon 1988, 26, 167–179. [Google Scholar] [CrossRef]
- Perez, J.C.; Garcia, V.E.; Huang, S.Y. Production of a Monoclonal Antibody against Hemorrhagic Activity of Crotalus atrox (Western Diamondback Rattlesnake) Venom. Toxicon 1984, 22, 967–973. [Google Scholar] [CrossRef]
- Ledsgaard, L.; Kilstrup, M.; Karatt-Vellatt, A.; McCafferty, J.; Laustsen, A. Basics of Antibody Phage Display Technology. Toxins 2018, 10, 236. [Google Scholar] [CrossRef]
- Frauches, T.S.; Petretski, J.H.; Arnholdt, A.C.V.; Lasunskaia, E.B.; de Carvalho, E.C.Q.; Kipnis, T.L.; da Silva, W.D.; Kanashiro, M.M. Bothropic Antivenom Based on Monoclonal Antibodies, Is It Possible? Toxicon 2013, 71, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Schneider, F.S.; Nguyen, D.L.; Castro, K.L.; Cobo, S.; Machado de Avila, R.A.; Ferreira, N. de A.; Sanchez, E.F.; Nguyen, C.; Granier, C.; Galéa, P.; et al. Use of a Synthetic Biosensor for Neutralizing Activity-Biased Selection of Monoclonal Antibodies against Atroxlysin-I, an Hemorrhagic Metalloproteinase from Bothrops atrox Snake Venom. PLoS Negl. Trop. Dis. 2014, 8, e2826. [Google Scholar] [CrossRef]
- Lomonte, B.; Kahan, L. Production and Partial Characterization of Monoclonal Antibodies to Bothrops asper (Terciopelo) Myotoxin. Toxicon 1988, 26, 675–689. [Google Scholar] [CrossRef]
- Fernandes, I.; Assumpção, G.G.; Silveira, C.R.F.; Faquim-Mauro, E.L.; Tanjoni, I.; Carmona, A.K.; Alves, M.F.M.; Takehara, H.A.; Rucavado, A.; Ramos, O.H.P.; et al. Immunochemical and Biological Characterization of Monoclonal Antibodies against BaP1, a Metalloproteinase from Bothrops asper Snake Venom. Toxicon 2010, 56, 1059–1065. [Google Scholar] [CrossRef] [PubMed]
- Guillon, V.; Alzari, P.M.; Grognet, J.-M. Preliminary Crystallographic Study of the Fab Fragment of a Monoclonal Antibody Directed against a Cobra Cardiotoxin. J. Mol. Biol. 1986, 189, 723–724. [Google Scholar] [CrossRef]
- Yang, C.C.; Chan, H.L. Preparation and Characterization of Β1-Bungarotoxin Bispecific Monoclonal Antibody. IUBMB Life 1999, 47, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, N.; England, F.; Vaughan, M.; Lafaye, P. Neutralizing Human Anti Crotoxin ScFv Isolated from a Nonimmunized Phage Library. Scand. J. Immunol. 2000, 51, 337–344. [Google Scholar] [CrossRef]
- Castro, J.M.A.; Oliveira, T.S.; Silveira, C.R.F.; Caporrino, M.C.; Rodriguez, D.; Moura-da-Silva, A.M.; Ramos, O.H.P.; Rucavado, A.; Gutiérrez, J.M.; Magalhães, G.S.; et al. A Neutralizing Recombinant Single Chain Antibody, ScFv, against BaP1, A P-I Hemorrhagic Metalloproteinase from Bothrops asper Snake Venom. Toxicon 2014, 87, 81–91. [Google Scholar] [CrossRef]
- Kulkeaw, K.; Sakolvaree, Y.; Srimanote, P.; Tongtawe, P.; Maneewatch, S.; Sookrung, N.; Tungtrongchitr, A.; Tapchaisri, P.; Kurazono, H.; Chaicumpa, W. Human Monoclonal ScFv Neutralize Lethal Thai Cobra, Naja kaouthia, Neurotoxin. J. Proteom. 2009, 72, 270–282. [Google Scholar] [CrossRef]
- Lee, C.-H.; Lee, Y.-C.; Liang, M.-H.; Leu, S.-J.; Lin, L.-T.; Chiang, J.-R.; Yang, Y.-Y. Antibodies against Venom of the Snake Deinagkistrodon acutus. Appl. Environ. Microbiol. 2016, 82, 71–80. [Google Scholar] [CrossRef]
- Oliveira, J.G.; Soares, S.G.; Soares, A.M.; Giglio, J.R.; Teixeira, J.E.; Barbosa, J.E. Expression of Human Recombinant Antibody Fragments Capable of Partially Inhibiting the Phospholypase Activity of Crotalus Durissus Terrificus Venom. Basic Clin. Pharm. Toxicol. 2009, 105, 84–91. [Google Scholar] [CrossRef]
- Roncolato, E.C.; Pucca, M.B.; Funayama, J.C.; Bertolini, T.B.; Campos, L.B.; Barbosa, J.E. Human Antibody Fragments Specific for Bothrops Jararacussu Venom Reduce the Toxicity of Other Bothrops sp. Venoms. J. Immunotoxicol 2013, 10, 160–168. [Google Scholar] [CrossRef]
- Silva, L.C.; Pucca, M.B.; Pessenda, G.; Campos, L.B.; Martinez, E.Z.; Cerni, F.A.; Barbosa, J.E. Discovery of Human ScFvs That Cross-Neutralize the Toxic Effects of B. jararacussu and C. d. Terrificus Venoms. Acta Trop. 2018, 177, 66–73. [Google Scholar] [CrossRef]
- Gomes, M.; Alvarez, M.A.; Quellis, L.R.; Becher, M.L.; Castro, J.M.D.A.; Gameiro, J.; Caporrino, M.C.; Moura-da-Silva, A.M.; de Oliveira Santos, M. Expression of an ScFv Antibody Fragment in Nicotiana Benthamiana and in Vitro Assessment of Its Neutralizing Potential against the Snake Venom Metalloproteinase BaP1 from Bothrops asper. Toxicon 2019, 160, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.B.; Cardoso, R.; Almeida-Souza, H.O.; Carvalho, C.P.; Correia, L.I.V.; Faria, P.C.B.; Araujo, G.R.; Mendes, M.M.; Rodrigues, R.S.; Rodrigues, V.M.; et al. Generation and In-Planta Expression of a Recombinant Single Chain Antibody with Broad Neutralization Activity on Bothrops pauloensis Snake Venom. Int. J. Biol. Macromol. 2020, 149, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Julve Parreño, J.M.; Huet, E.; Fernández-del-Carmen, A.; Segura, A.; Venturi, M.; Gandía, A.; Pan, W.; Albaladejo, I.; Forment, J.; Pla, D.; et al. A Synthetic Biology Approach for Consistent Production of Plant-Made Recombinant Polyclonal Antibodies against Snake Venom Toxins. Plant Biotechnol. J. 2018, 16, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Luiz, M.; Pereira, S.; Gonçalves, N.; Prado, N.; Dill, L.; Soares, A.; Zanchi, F.; Zuliani, J.; Stábeli, R.; Fernandes, C. Inhibition of the Myotoxicity Induced by Crotoxin B, from Crotalus durissus Terrificus Venom, by Camelid Nanobodies. In Proceedings of the Anais do III Simpósio Internacional de Imunobiológicos; Instituto de Tecnologia em Imunobiológicos; Bio-Manguinhos: Rio de Janeiro, Brazil, 2016; pp. 80–81. [Google Scholar]
- Bailon Calderon, H.; Yaniro Coronel, V.O.; Cáceres Rey, O.A.; Colque Alave, E.G.; Leiva Duran, W.J.; Padilla Rojas, C.; Montejo Arevalo, H.; García Neyra, D.; Galarza Pérez, M.; Bonilla, C.; et al. Development of Nanobodies Against Hemorrhagic and Myotoxic Components of Bothrops atrox Snake Venom. Front. Immunol. 2020, 11, 655. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.S.; MacKenzie, C.R.; Christopher Hall, J. Isolation, Characterization and Pentamerization of α-Cobrotoxin Specific Single-Domain Antibodies from a Naïve Phage Display Library: Preliminary Findings for Antivenom Development. Toxicon 2007, 49, 699–709. [Google Scholar] [CrossRef]
- Chavanayarn, C.; Thanongsaksrikul, J.; Thueng-in, K.; Bangphoomi, K.; Sookrung, N.; Chaicumpa, W. Humanized-Single Domain Antibodies (VH/VHH) That Bound Specifically to Naja kaouthia Phospholipase A2 and Neutralized the Enzymatic Activity. Toxins 2012, 4, 554–567. [Google Scholar] [CrossRef]
- Richard, G.; Meyers, A.J.; McLean, M.D.; Arbabi-Ghahroudi, M.; MacKenzie, R.; Hall, J.C. In Vivo Neutralization of α-Cobratoxin with High-Affinity Llama Single-Domain Antibodies (VHHs) and a VHH-Fc Antibody. PLoS ONE 2013, 8, e69495. [Google Scholar] [CrossRef]
- Prado, N.D.R.; Pereira, S.S.; da Silva, M.P.; Morais, M.S.S.; Kayano, A.M.; Moreira-Dill, L.S.; Luiz, M.B.; Zanchi, F.B.; Fuly, A.L.; EF Huacca, M.; et al. Inhibition of the Myotoxicity Induced by Bothrops jararacussu Venom and Isolated Phospholipases A2 by Specific Camelid Single-Domain Antibody Fragments. PLoS ONE 2016, 11, e0151363. [Google Scholar] [CrossRef]
- Jenkins, T.P.; Laustsen, A.H. Cost of Manufacturing for Recombinant Snakebite Antivenoms. Front. Bioeng. Biotechnol. 2020, 8, 703. [Google Scholar] [CrossRef]
- Kris, A. Wetterstrand DNA Sequencing Costs: Data. Available online: https://www.genome.gov/about-genomics/fact-sheets/DNA-Sequencing-Costs-Data (accessed on 23 August 2022).
- Gray, A.C.; Sidhu, S.S.; Chandrasekera, P.C.; Hendriksen, C.F.M.; Borrebaeck, C.A.K. Animal-Friendly Affinity Reagents: Replacing the Needless in the Haystack. Trends Biotechnol. 2016, 34, 960–969. [Google Scholar] [CrossRef]
- Laustsen, A.H.; Greiff, V.; Karatt-Vellatt, A.; Muyldermans, S.; Jenkins, T.P. Animal Immunization, in Vitro Display Technologies, and Machine Learning for Antibody Discovery. Trends Biotechnol. 2021, 39, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.H.; Ainsworth, S.; Lomonte, B.; Kini, R.M.; Chávez-Olórtegui, C. Editorial: Novel Immunotherapies Against Envenomings by Snakes and Other Venomous Animals. Front. Immunol. 2020, 11, 1004. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias da Silva, W.; De Andrade, S.A.; Megale, Â.A.A.; De Souza, D.A.; Sant’Anna, O.A.; Magnoli, F.C.; Guidolin, F.R.; Godoi, K.S.; Saladini, L.Y.; Spencer, P.J.; et al. Antibodies as Snakebite Antivenoms: Past and Future. Toxins 2022, 14, 606. https://doi.org/10.3390/toxins14090606
Dias da Silva W, De Andrade SA, Megale ÂAA, De Souza DA, Sant’Anna OA, Magnoli FC, Guidolin FR, Godoi KS, Saladini LY, Spencer PJ, et al. Antibodies as Snakebite Antivenoms: Past and Future. Toxins. 2022; 14(9):606. https://doi.org/10.3390/toxins14090606
Chicago/Turabian StyleDias da Silva, Wilmar, Sonia A. De Andrade, Ângela Alice Amadeu Megale, Daniel Alexandre De Souza, Osvaldo Augusto Sant’Anna, Fábio Carlos Magnoli, Felipe Raimondi Guidolin, Kemily Stephanie Godoi, Lucas Yuri Saladini, Patrick Jack Spencer, and et al. 2022. "Antibodies as Snakebite Antivenoms: Past and Future" Toxins 14, no. 9: 606. https://doi.org/10.3390/toxins14090606
APA StyleDias da Silva, W., De Andrade, S. A., Megale, Â. A. A., De Souza, D. A., Sant’Anna, O. A., Magnoli, F. C., Guidolin, F. R., Godoi, K. S., Saladini, L. Y., Spencer, P. J., & Portaro, F. C. V. (2022). Antibodies as Snakebite Antivenoms: Past and Future. Toxins, 14(9), 606. https://doi.org/10.3390/toxins14090606




