Aflatoxin B1 Exposure in Sheep: Insights into Hepatotoxicity Based on Oxidative Stress, Inflammatory Injury, Apoptosis, and Gut Microbiota Analysis
Abstract
1. Introduction
2. Results
2.1. Effects of AFB1 Exposure on Clinical Symptoms, Body Temperature, Respiration, Heart Rate, and Conjunctival Color
2.2. Effects of AFB1 Exposure on Serum Biochemical Indices
2.3. Effects of AFB1 Exposure on Liver Tissue Structure and Liver Tissue Function
2.4. Effects of AFB1 Exposure on Liver Oxidative Damage
2.5. Effects of AFB1 Exposure on the Expression of Inflammation-Related Factors in the Liver
2.6. Effects of AFB1 Exposure on Liver Cell Apoptosis and Apoptosis-Related Gene Expression
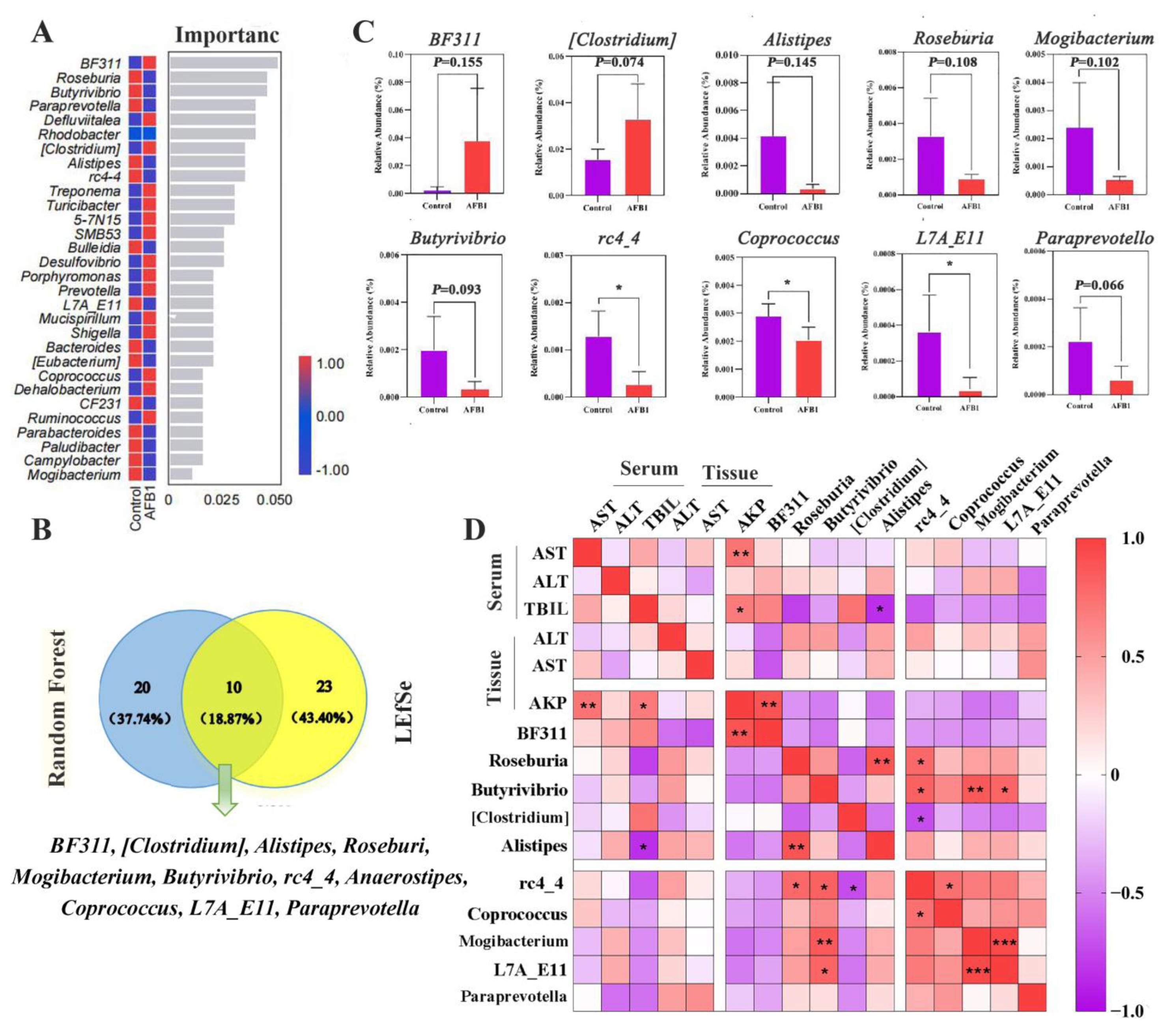
2.7. Changes in Gut Microbiota Induced by AFB1 Exposure
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Toxin
5.2. Animals, Exposure Experiment
5.3. The Color of Conjunctiva
5.4. Sample Collection
5.5. Serum Biochemistry
5.6. Liver Histopathology
5.7. Detection of Apoptosis
5.8. Liver Function of Tissue
5.9. Liver Antioxidant Abilities
5.10. Extraction of Total RNA and Quantitative Real-Time PCR Analysis
5.11. Extraction of Faecal DNA, PCR Amplification, and Illumina Sequencing
5.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- M Santhosh, N.; Shvalya, V.; Modic, M.; Hojnik, N.; Zavašnik, J.; Olenik, J.; Košiček, M.; Filipič, G.; Abdulhalim, I.; Cvelbar, U. Label-Free Mycotoxin Raman Identification by High-Performing Plasmonic Vertical Carbon Nanostructures. Small 2021, 17, e2103677. [Google Scholar] [CrossRef] [PubMed]
- Rice, L.G.; Ross, P.F. Methods for Detection and Quantitation of Fumonisins in Corn, Cereal Products and Animal Excreta. J. Food Prot. 1994, 57, 536–540. [Google Scholar] [CrossRef]
- Crudo, F.; Varga, E.; Aichinger, G.; Galaverna, G.; Marko, D.; Dall’Asta, C.; Dellafiora, L. Co-Occurrence and Combinatory Effects of Alternaria Mycotoxins and other Xenobiotics of Food Origin: Current Scenario and Future Perspectives. Toxins 2019, 11, 640. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Huang, C.; Yang, X.; Zhang, B.; He, X.; Xu, W.; Huang, K. Proteomics reveals the alleviation of zinc towards aflatoxin B1-induced cytotoxicity in human hepatocyes (HepG2 cells). Ecotoxicol. Environ. Saf. 2020, 198, 110596. [Google Scholar] [CrossRef] [PubMed]
- Solcan, C.; Gogu, M.; Floristean, V.; Oprisan, B.; Solcan, G. The hepatoprotective effect of sea buckthorn (Hippophae rhamnoides) berries on induced aflatoxin B1 poisoning in chickens 1. Poult. Sci. 2013, 92, 966–974. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, P.; Xu, F.; Huang, W.; Ji, Q.; Han, Y.; Shao, B.; Li, Y. Protective effects of lycopene against AFB(1)-induced erythrocyte dysfunction and oxidative stress in mice. Res. Vet. Sci. 2020, 129, 103–108. [Google Scholar] [CrossRef]
- Lin, L.; Fu, P.; Chen, N.; Gao, N.; Cao, Q.; Yue, K.; Xu, T.; Zhang, C.; Zhang, C.; Liu, F.; et al. Total flavonoids of Rhizoma Drynariae protect hepatocytes against aflatoxin B1-induced oxidative stress and apoptosis in broiler chickens. Ecotoxicol. Environ. Saf. 2022, 230, 113148. [Google Scholar] [CrossRef]
- Cao, Q.Q.; Lin, L.X.; Xu, T.T.; Lu, Y.; Zhang, C.D.; Yue, K.; Huang, S.C.; Dong, H.J.; Jian, F.C. Aflatoxin B1 alters meat quality associated with oxidative stress, inflammation, and gut-microbiota in sheep. Ecotoxicol. Environ. Saf. 2021, 225, 112754. [Google Scholar] [CrossRef]
- Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Mycotoxins: Occurrence, toxicology, and exposure assessment. Mycotoxins: Occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. 2013, 60, 218–237. [Google Scholar] [CrossRef]
- Li, S.; Muhammad, I.; Yu, H.; Sun, X.; Zhang, X. Detection of Aflatoxin adducts as potential markers and the role of curcumin in alleviating AFB1-induced liver damage in chickens. Ecotoxicol. Environ. Saf. 2019, 176, 137–145. [Google Scholar] [CrossRef]
- Budin, C.; Man, H.Y.; Al-Ayoubi, C.; Puel, S.; van Vugt-Lussenburg, B.M.A.; Brouwer, A.; Oswald, I.P.; van der Burg, B.; Soler, L. Versicolorin A enhances the genotoxicity of aflatoxin B1 in human liver cells by inducing the transactivation of the Ah-receptor. Food Chem. Toxicol. 2021, 153, 112258. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lv, Z.; Chen, J.; Nepovimova, E.; Long, M.; Wu, W.; Kuca, K. Bacillus amyloliquefaciens B10 can alleviate liver apoptosis and oxidative stress induced by aflatoxin B1. Food Chem. Toxicol. 2021, 151, 112124. [Google Scholar] [CrossRef] [PubMed]
- Pu, J.; Yuan, Q.; Yan, H.; Tian, G.; Chen, D.; He, J.; Zheng, P.; Yu, J.; Mao, X.; Huang, Z.; et al. Effects of Chronic Exposure to Low Levels of Dietary Aflatoxin B(1) on Growth Performance, Apparent Total Tract Digestibility and Intestinal Health in Pigs. Animals 2021, 11, 336. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liu, R.; Xia, S.; Wei, G.; Ishfaq, M.; Zhang, Y.; Zhang, X. Protective role of curcumin on aflatoxin B1-induced TLR4/RIPK pathway mediated-necroptosis and inflammation in chicken liver. Ecotoxicol. Environ. Saf. 2022, 233, 113319. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, M.R.; Hong, Y.C. Modification of the association of bisphenol A with abnormal liver function by polymorphisms of oxidative stress-related genes. Environ. Res. 2016, 147, 324–330. [Google Scholar] [CrossRef]
- Buccitelli, C.; Selbach, M. mRNAs, proteins and the emerging principles of gene expression control. Nat. Rev. Genet. 2020, 21, 630–644. [Google Scholar] [CrossRef]
- Schwabe, R.F.; Tabas, I.; Pajvani, U.B. Mechanisms of Fibrosis Development in Nonalcoholic Steatohepatitis. Gastroenterology 2020, 158, 1913–1928. [Google Scholar] [CrossRef]
- Goergen, C.J.; Tweardy, M.J.; Steinhubl, S.R.; Wegerich, S.W.; Singh, K.; Mieloszyk, R.J.; Dunn, J. Detection and Monitoring of Viral Infections via Wearable Devices and Biometric Data. Annu. Rev. Biomed. Eng. 2021, 24, 1–27. [Google Scholar] [CrossRef]
- Polley, N.; Saha, S.; Singh, S.; Adhikari, A.; Das, S.; Choudhury, B.R.; Pal, S.K. Development and optimization of a noncontact optical device for online monitoring of jaundice in human subjects. J. Biomed. Opt. 2015, 20, 067001. [Google Scholar] [CrossRef]
- Bagherzadeh Kasmani, F.; Karimi Torshizi, M.A.; Allameh, A.; Shariatmadari, F. A novel aflatoxin-binding Bacillus probiotic: Performance, serum biochemistry, and immunological parameters in Japanese quail. Poult. Sci. 2012, 91, 1846–1853. [Google Scholar] [CrossRef]
- Lv, L.X.; Hu, X.J.; Qian, G.R.; Zhang, H.; Lu, H.F.; Zheng, B.W.; Jiang, L.; Li, L.J. Administration of Lactobacillus salivarius LI01 or Pediococcus pentosaceus LI05 improves acute liver injury induced by D-galactosamine in rats. Appl. Microbiol. Biotechnol. 2014, 98, 5619–5632. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Bai, W.; Mao, D.; Chen, Y.; Wang, K.; Qiu, H.; Wu, J. Detoxification II Prescription Suppresses the Th-17/IL-17 Inflammatory Axis to Improve the Liver Function of ACLF-Rats via Inactivating the P38MAPK Pathway. J. Healthc. Eng. 2021, 2021, 7563383. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, F.; Liu, M.; Zhou, X.; Wang, M.; Cao, K.; Jin, S.; Shan, A.; Feng, X. Curcumin mitigates aflatoxin B1-induced liver injury via regulating the NLRP3 inflammasome and Nrf2 signaling pathway. Food Chem. Toxicol. 2022, 161, 112823. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Fu, W.; Huo, M.; He, B.; Liu, Y.; Tian, L.; Li, W.; Zhou, Z.; Wang, B.; Xia, J.; et al. Spatial-resolved metabolomics reveals tissue-specific metabolic reprogramming in diabetic nephropathy by using mass spectrometry imaging. Acta Pharm. Sin. B 2021, 11, 3665–3677. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Hsu, L.W.; Chen, K.D.; Chiu, K.W.; Chen, C.L.; Huang, K.T. Emerging Roles of Calcium Signaling in the Development of Non-Alcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2021, 23, 256. [Google Scholar] [CrossRef]
- Jin, S.; Yang, H.; Wang, Y.; Pang, Q.; Jiao, Y.; Shan, A.; Feng, X. Dietary Curcumin Alleviated Aflatoxin B1-Induced Acute Liver Damage in Ducks by Regulating NLRP3-Caspase-1 Signaling Pathways. Foods 2021, 10, 3086. [Google Scholar] [CrossRef]
- Wang, C.; Ma, C.; Fu, K.; Gong, L.H.; Zhang, Y.F.; Zhou, H.L.; Li, Y.X. Phillygenin Attenuates Carbon Tetrachloride-Induced Liver Fibrosis via Modulating Inflammation and Gut Microbiota. Front. Pharmacol. 2021, 12, 756924. [Google Scholar] [CrossRef]
- Bai, K.; Huang, Q.; Zhang, J.; He, J.; Zhang, L.; Wang, T. Supplemental effects of probiotic Bacillus subtilis fmbJ on growth performance, antioxidant capacity, and meat quality of broiler chickens. Poult. Sci. 2017, 96, 74–82. [Google Scholar] [CrossRef]
- Fouad, A.M.; Ruan, D.; El-Senousey, H.K.; Chen, W.; Jiang, S.; Zheng, C. Harmful Effects and Control Strategies of Aflatoxin B1 Produced by Aspergillus flavus and Aspergillus parasiticus Strains on Poultry: Review. Toxins 2019, 11, 176. [Google Scholar] [CrossRef]
- Yang, R.L.; Li, W.; Shi, Y.H.; Le, G.W. Lipoic acid prevents high-fat diet–induced dyslipidemia and oxidative stress: A microarray analysis. Nutrition 2008, 24, 582–588. [Google Scholar] [CrossRef]
- Ding, Z.; Zhang, Y.; Ye, J.; Du, Z.; Kong, Y. An evaluation of replacing fish meal with fermented soybean meal in the diet of Macrobrachium nipponense: Growth, nonspecific immunity, and resistance to Aeromonas hydrophila. Fish Shellfish Immunol. 2015, 44, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Yin, H.; Zuo, Z.; Yang, Z.; Yang, Y.; Wei, L.; Cui, H.; Deng, H.; Chen, X.; Zhu, Y.; et al. Oxidative stress-mediated apoptosis and autophagy involved in Ni-induced nephrotoxicity in the mice. Ecotoxicol. Environ. Saf. 2021, 228, 112954. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, N.; Xu, Y.; Tan, H.Y.; Li, S.; Feng, Y. Molecular Mechanisms Involved in Oxidative Stress-Associated Liver Injury Induced by Chinese Herbal Medicine: An Experimental Evidence-Based Literature Review and Network Pharmacology Study. Int. J. Mol. Sci. 2018, 19, 2745. [Google Scholar] [CrossRef] [PubMed]
- Herrington, J.D.; Figueroa, J.A.; Kirstein, M.N.; Zamboni, W.C.; Stewart, C.F. Effect of hemodialysis on topotecan disposition in a patient with severe renal dysfunction. Cancer Chemother. Pharmacol. 2001, 47, 89–93. [Google Scholar] [CrossRef]
- Pauletto, M.; Giantin, M.; Tolosi, R.; Bassan, I.; Barbarossa, A.; Zaghini, A.; Dacasto, M. Curcumin Mitigates AFB1-Induced Hepatic Toxicity by Triggering Cattle Antioxidant and Anti-inflammatory Pathways: A Whole Transcriptomic In Vitro Study. Antioxidants 2020, 9, 1059. [Google Scholar] [CrossRef]
- Li, H.; Huang, K.; Liu, X.; Liu, J.; Lu, X.; Tao, K.; Wang, G.; Wang, J. Lithium chloride suppresses colorectal cancer cell survival and proliferation through ROS/GSK-3β/NF-κB signaling pathway. Oxidative Med. Cell. Longev. 2014, 2014, 241864. [Google Scholar] [CrossRef]
- Rajendran, P.; Chen, Y.F.; Chen, Y.F.; Chung, L.C.; Tamilselvi, S.; Shen, C.Y.; Day, C.H.; Chen, R.J.; Viswanadha, V.P.; Kuo, W.W.; et al. The multifaceted link between inflammation and human diseases. J. Cell. Physiol. 2018, 233, 6458–6471. [Google Scholar] [CrossRef]
- Guo, Y.; Balasubramanian, B.; Zhao, Z.H.; Liu, W.C. Marine algal polysaccharides alleviate aflatoxin B1-induced bursa of Fabricius injury by regulating redox and apoptotic signaling pathway in broilers. Poult. Sci. 2021, 100, 844–857. [Google Scholar] [CrossRef]
- Yasin, M.; Mazdak, R.; Mino, I. Aflatoxin B1 impairs spermatogenesis: An experimental study for crosslink between oxidative stress and mitochondria-dependent apoptosis. Environ. Toxicol. 2018, 33, 1204–1213. [Google Scholar] [CrossRef]
- Chen, Y.; Feng, X.; Hu, X.; Sha, J.; Li, B.; Zhang, H.; Fan, H. Dexmedetomidine Ameliorates Acute Stress-Induced Kidney Injury by Attenuating Oxidative Stress and Apoptosis through Inhibition of the ROS/JNK Signaling Pathway. Oxidative Med. Cell. Longev. 2018, 2018, 4035310. [Google Scholar] [CrossRef]
- Xu, F.; Li, Y.; Cao, Z.; Zhang, J.; Huang, W. AFB(1)-induced mice liver injury involves mitochondrial dysfunction mediated by mitochondrial biogenesis inhibition. Ecotoxicol. Environ. Saf. 2021, 216, 112213. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shao, D.; Wu, Y.; Cai, C.; Hu, C.; Shou, X.; Dai, B.; Ye, B.; Wang, M.; Jia, X. Apoptotic responses of Carassius auratus lymphocytes to nodularin exposure In Vitro. Fish Shellfish Immunol. 2012, 33, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Han, B.; Xing, X.; Li, Y.; Zhao, D.; Liu, M.; Wang, S. A Protein from Dioscorea polystachya (Chinese Yam) Improves Hydrocortisone-Induced Testicular Dysfunction by Alleviating Leydig Cell Injury via Upregulation of the Nrf2 Pathway. Oxidative Med. Cell. Longev. 2021, 2021, 3575016. [Google Scholar] [CrossRef] [PubMed]
- Nwachukwu, C.U.; Woad, K.J.; Barnes, N.; Gardner, D.S.; Robinson, R.S. Maternal protein restriction affects fetal ovary development in sheep. Reprod. Fertil. 2021, 2, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liu, L.; Chen, J.; Xiao, A. Response of Intestinal Bacterial Flora to the Long-term Feeding of Aflatoxin B1 (AFB1) in Mice. Toxins 2017, 9, 317. [Google Scholar] [CrossRef]
- Young, J.C.; Zhou, T.; Yu, H.; Zhu, H.; Gong, J. Degradation of trichothecene mycotoxins by chicken intestinal microbes. Food Chem. Toxicol. 2007, 45, 136–143. [Google Scholar] [CrossRef]
- Wang, J.; Tang, L.; Glenn, T.C.; Wang, J.S. Aflatoxin B1 Induced Compositional Changes in Gut Microbial Communities of Male F344 Rats. Toxicol. Sci. 2016, 150, 54–63. [Google Scholar] [CrossRef]
- Pan, D.; Yu, Z. Intestinal microbiome of poultry and its interaction with host and diet. Gut Microbes 2014, 5, 108–119. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, N.; Shen, W.; Zhao, S.; Wang, J. Synchrony Degree of Dietary Energy and Nitrogen Release Influences Microbial Community, Fermentation, and Protein Synthesis in a Rumen Simulation System. Microorganisms 2020, 8, 231. [Google Scholar] [CrossRef]
- Parker, B.J.; Wearsch, P.A.; Veloo, A.C.M.; Rodriguez-Palacios, A. The Genus Alistipes: Gut Bacteria with Emerging Implications to Inflammation, Cancer, and Mental Health. Front. Immunol. 2020, 11, 906. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sui, Y.; Lu, Y.; Zuo, S.; Wang, H.; Bian, X.; Chen, G.; Huang, S.; Dai, H.; Liu, F.; Dong, H. Aflatoxin B1 Exposure in Sheep: Insights into Hepatotoxicity Based on Oxidative Stress, Inflammatory Injury, Apoptosis, and Gut Microbiota Analysis. Toxins 2022, 14, 840. https://doi.org/10.3390/toxins14120840
Sui Y, Lu Y, Zuo S, Wang H, Bian X, Chen G, Huang S, Dai H, Liu F, Dong H. Aflatoxin B1 Exposure in Sheep: Insights into Hepatotoxicity Based on Oxidative Stress, Inflammatory Injury, Apoptosis, and Gut Microbiota Analysis. Toxins. 2022; 14(12):840. https://doi.org/10.3390/toxins14120840
Chicago/Turabian StyleSui, Yuzhen, Ying Lu, Shoujun Zuo, Haidong Wang, Xiaokun Bian, Guizhen Chen, Shucheng Huang, Hongyu Dai, Fang Liu, and Haiju Dong. 2022. "Aflatoxin B1 Exposure in Sheep: Insights into Hepatotoxicity Based on Oxidative Stress, Inflammatory Injury, Apoptosis, and Gut Microbiota Analysis" Toxins 14, no. 12: 840. https://doi.org/10.3390/toxins14120840
APA StyleSui, Y., Lu, Y., Zuo, S., Wang, H., Bian, X., Chen, G., Huang, S., Dai, H., Liu, F., & Dong, H. (2022). Aflatoxin B1 Exposure in Sheep: Insights into Hepatotoxicity Based on Oxidative Stress, Inflammatory Injury, Apoptosis, and Gut Microbiota Analysis. Toxins, 14(12), 840. https://doi.org/10.3390/toxins14120840







