Cytokine and Chemokine Retention Profile in COVID-19 Patients with Chronic Kidney Disease
Abstract
1. Introduction
2. Results
2.1. Clinical
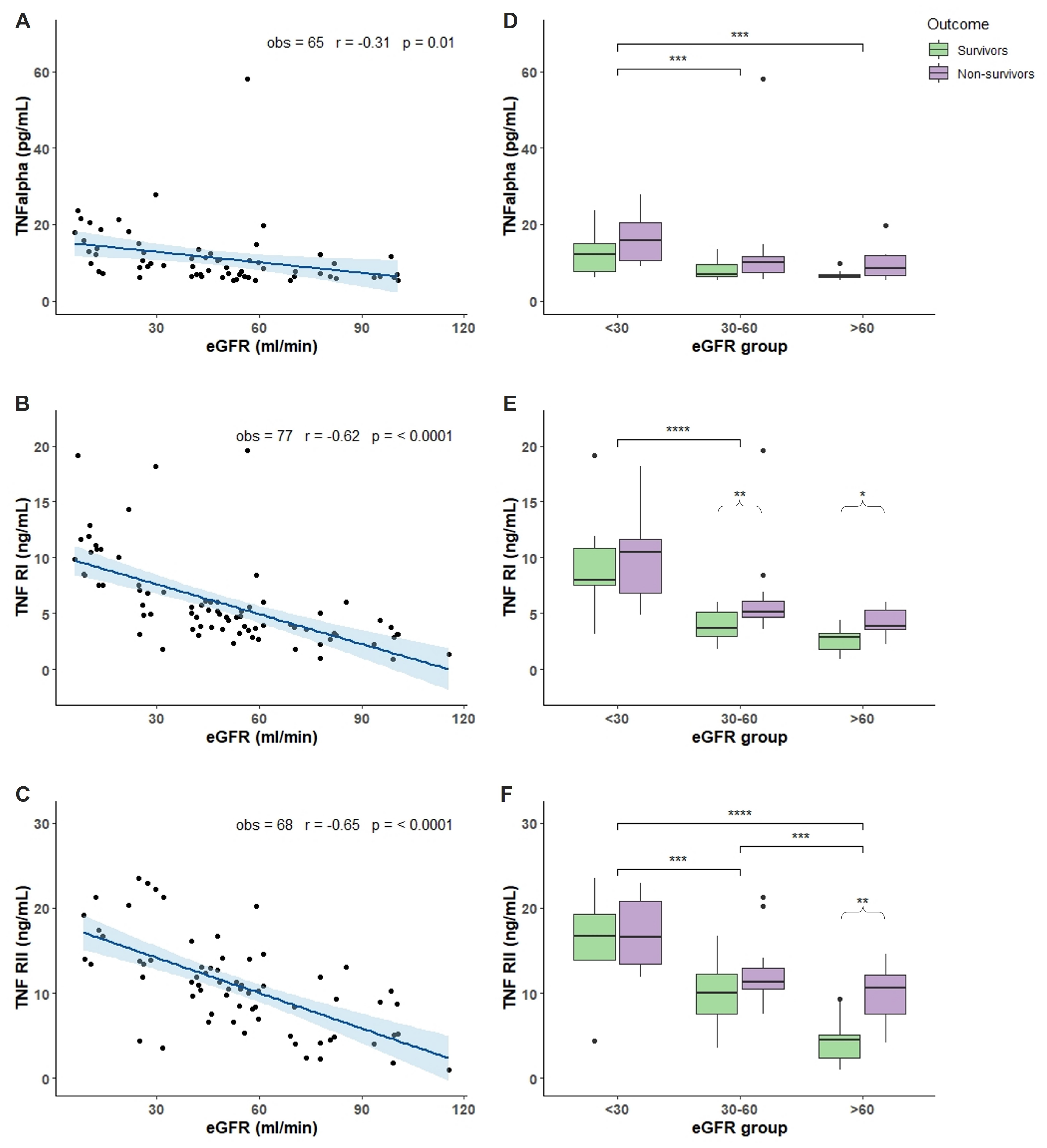
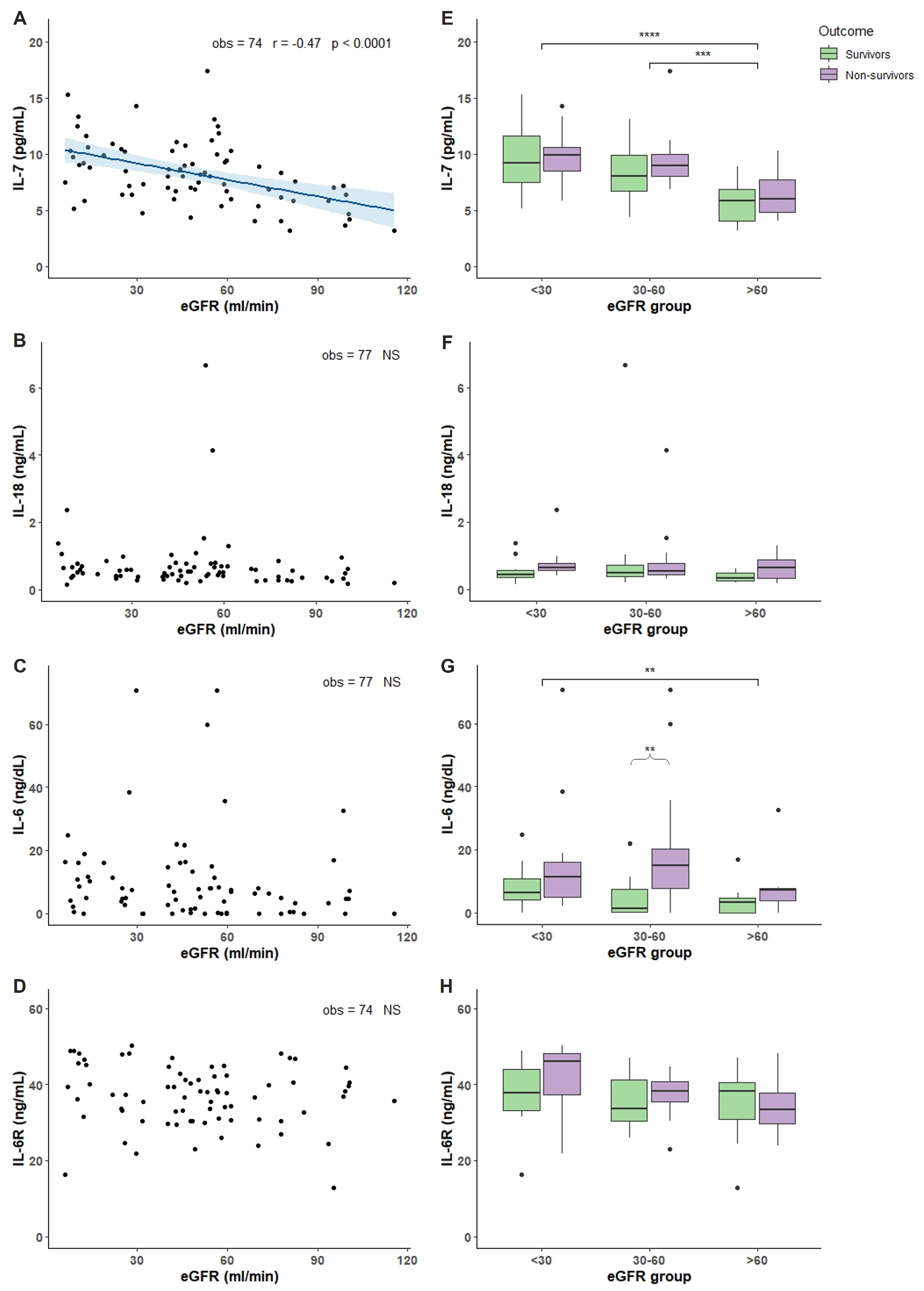
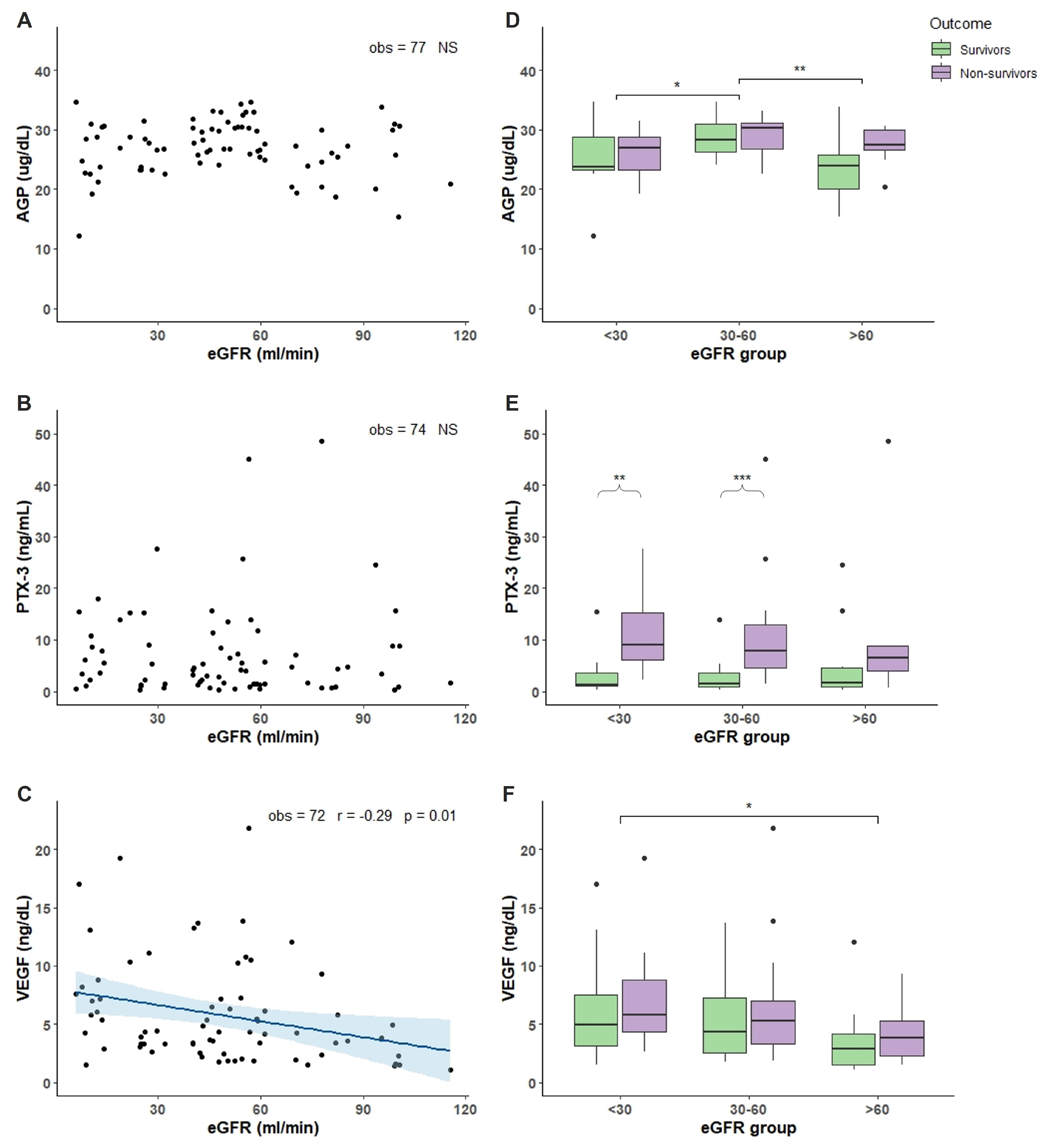
2.2. Characterization of COVID-19 Plasma with Respect of eGFR
3. Discussion
4. Materials and Methods
4.1. Plasma Cytokine Quantification
4.2. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, Regional, and National Burden of Chronic Kidney Disease, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Dalrymple, L.S.; Go, A.S. Epidemiology of Acute Infections among Patients with Chronic Kidney Disease: Figure 1. Clin. J. Am. Soc. Nephrol. 2008, 3, 1487–1493. [Google Scholar] [CrossRef]
- Ahmadian, E.; Hosseiniyan Khatibi, S.M.; Razi Soofiyani, S.; Abediazar, S.; Shoja, M.M.; Ardalan, M.; Zununi Vahed, S. COVID-19 and Kidney Injury: Pathophysiology and Molecular Mechanisms. Rev. Med. Virol. 2021, 31, e2176. [Google Scholar] [CrossRef]
- Paolini, A.; Borella, R.; De Biasi, S.; Neroni, A.; Mattioli, M.; Lo Tartaro, D.; Simonini, C.; Franceschini, L.; Cicco, G.; Piparo, A.M.; et al. Cell Death in Coronavirus Infections: Uncovering Its Role during COVID-19. Cells 2021, 10, 1585. [Google Scholar] [CrossRef]
- Clark, J. What Does Tumour Necrosis Factor Excess Do to the Immune System Long Term? Ann. Rheum. Dis. 2005, 64, iv70–iv76. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical Features of Patients Infected with 2019 Novel Coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Karki, R.; Sharma, B.R.; Tuladhar, S.; Williams, E.P.; Zalduondo, L.; Samir, P.; Zheng, M.; Sundaram, B.; Banoth, B.; Malireddi, R.K.S.; et al. Synergism of TNF-α and IFN-γ Triggers Inflammatory Cell Death, Tissue Damage, and Mortality in SARS-CoV-2 Infection and Cytokine Shock Syndromes. Cell 2021, 184, 149–168.e17. [Google Scholar] [CrossRef]
- Schrauben, S.J.; Shou, H.; Zhang, X.; Anderson, A.H.; Bonventre, J.V.; Chen, J.; Coca, S.; Furth, S.L.; Greenberg, J.H.; Gutierrez, O.M.; et al. Association of Multiple Plasma Biomarker Concentrations with Progression of Prevalent Diabetic Kidney Disease: Findings from the Chronic Renal Insufficiency Cohort (CRIC) Study. J. Am. Soc. Nephrol. 2021, 32, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, J.; Marik, P.E.; Levine, J.S. Elevated Serum Levels of the Type I and Type II Receptors for Tumor Necrosis Factor-α as Predictive Factors for ARF in Patients with Septic Shock. Am. J. Kidney Dis. 2003, 41, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Bowman, E.R.; Cameron, C.M.A.; Avery, A.; Gabriel, J.; Kettelhut, A.; Hecker, M.; Sontich, C.U.; Tamilselvan, B.; Nichols, C.N.; Richardson, B.; et al. Levels of Soluble CD14 and Tumor Necrosis Factor Receptors 1 and 2 May Be Predictive of Death in Severe Coronavirus Disease 2019. J. Infect. Dis. 2021, 223, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Sancho Ferrando, E.; Hanslin, K.; Hultström, M.; Larsson, A.; Frithiof, R.; Lipcsey, M. Soluble TNF Receptors Predict Acute Kidney Injury and Mortality in Critically Ill COVID-19 Patients: A Prospective Observational Study. Cytokine 2022, 149, 155727. [Google Scholar] [CrossRef] [PubMed]
- ElKassar, N.; Gress, R.E. An Overview of IL-7 Biology and Its Use in Immunotherapy. J. Immunotoxicol. 2010, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Barata, J.T.; Durum, S.K.; Seddon, B. Flip the Coin: IL-7 and IL-7R in Health and Disease. Nat. Immunol. 2019, 20, 1584–1593. [Google Scholar] [CrossRef] [PubMed]
- Chi, Y.; Ge, Y.; Wu, B.; Zhang, W.; Wu, T.; Wen, T.; Liu, J.; Guo, X.; Huang, C.; Jiao, Y.; et al. Serum Cytokine and Chemokine Profile in Relation to the Severity of Coronavirus Disease 2019 in China. J. Infect. Dis. 2020, 222, 746–754. [Google Scholar] [CrossRef]
- Laterre, P.F.; François, B.; Collienne, C.; Hantson, P.; Jeannet, R.; Remy, K.E.; Hotchkiss, R.S. Association of Interleukin 7 Immunotherapy with Lymphocyte Counts among Patients with Severe Coronavirus Disease 2019 (COVID-19). JAMA Netw. Open 2020, 3, e2016485. [Google Scholar] [CrossRef]
- Bekele, Y.; Sui, Y.; Berzofsky, J.A. IL-7 in SARS-CoV-2 Infection and as a Potential Vaccine Adjuvant. Front. Immunol. 2021, 12, 737406. [Google Scholar] [CrossRef]
- Zhao, J.; Wu, Z.; Yu, Y.; Wang, L.; Cheng, L. Effects of Interleukin-7/Interleukin-7 Receptor on RANKL-mediated Osteoclast Differentiation and Ovariectomy-induced Bone Loss by Regulating C-Fos/C-Jun Pathway. J. Cell. Physiol. 2018, 233, 7182–7194. [Google Scholar] [CrossRef]
- Kaplanski, G. Interleukin-18: Biological Properties and Role in Disease Pathogenesis. Immunol. Rev. 2018, 281, 138–153. [Google Scholar] [CrossRef]
- Fardet, L.; Galicier, L.; Lambotte, O.; Marzac, C.; Aumont, C.; Chahwan, D.; Coppo, P.; Hejblum, G. Development and Validation of the HScore, a Score for the Diagnosis of Reactive Hemophagocytic Syndrome: Score for Reactive Hemophagocytic Syndrome. Arthritis Rheumatol. 2014, 66, 2613–2620. [Google Scholar] [CrossRef]
- Hayashi, N.; Yoshimoto, T.; Izuhara, K.; Matsui, K.; Tanaka, T.; Nakanishi, K. T Helper 1 Cells Stimulated with Ovalbumin and IL-18 Induce Airway Hyperresponsiveness and Lung Fibrosis by IFN-γ and IL-13 Production. Proc. Natl. Acad. Sci. USA 2007, 104, 14765–14770. [Google Scholar] [CrossRef]
- Kerget, B.; Kerget, F.; Aksakal, A.; Aşkın, S.; Sağlam, L.; Akgün, M. Evaluation of Alpha Defensin, IL-1 Receptor Antagonist, and IL-18 Levels in COVID-19 Patients with Macrophage Activation Syndrome and Acute Respiratory Distress Syndrome. J. Med. Virol. 2021, 93, 2090–2098. [Google Scholar] [CrossRef] [PubMed]
- Satış, H.; Özger, H.S.; Aysert Yıldız, P.; Hızel, K.; Gulbahar, Ö.; Erbaş, G.; Aygencel, G.; Guzel Tunccan, O.; Öztürk, M.A.; Dizbay, M.; et al. Prognostic Value of Interleukin-18 and Its Association with Other Inflammatory Markers and Disease Severity in COVID-19. Cytokine 2021, 137, 155302. [Google Scholar] [CrossRef] [PubMed]
- Scheller, J.; Rose-John, S. Interleukin-6 and Its Receptor: From Bench to Bedside. Med. Microbiol. Immunol. 2006, 195, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Coomes, E.A.; Haghbayan, H. Interleukin-6 in COVID-19: A Systematic Review and META-ANALYSIS. Rev. Med. Virol. 2020, 30, 1–9. [Google Scholar] [CrossRef]
- Meynier, S.; Rieux-Laucat, F. FAS and RAS Related Apoptosis Defects: From Autoimmunity to Leukemia. Immunol. Rev. 2019, 287, 50–61. [Google Scholar] [CrossRef]
- El-Agroudy, A.E.; El-Baz, A. Soluble Fas: A Useful Marker of Inflammation and Cardiovascular Diseases in Uremic Patients. Clin. Exp. Nephrol. 2010, 14, 152–157. [Google Scholar] [CrossRef]
- Klaric, L.; Gisby, J.S.; Papadaki, A.; Muckian, M.D.; Macdonald-Dunlop, E.; Zhao, J.H.; Tokolyi, A.; Persyn, E.; Pairo-Castineira, E.; Morris, A.P.; et al. Mendelian Randomisation Identifies Alternative Splicing of the FAS Death Receptor as a Mediator of Severe COVID-19. Infectious Diseases (except HIV/AIDS). MedRxiv 2021. [Google Scholar] [CrossRef]
- Yoshino, J.; Monkawa, T.; Tsuji, M.; Hayashi, M.; Saruta, T. Leukemia Inhibitory Factor Is Involved in Tubular Regeneration after Experimental Acute Renal Failure. J. Am. Soc. Nephrol. 2003, 14, 3090–3101. [Google Scholar] [CrossRef]
- Correll, K.A.; Edeen, K.E.; Zemans, R.L.; Redente, E.F.; Serban, K.A.; Curran-Everett, D.; Edelman, B.L.; Mikels-Vigdal, A.; Mason, R.J. Transitional Human Alveolar Type II Epithelial Cells Suppress Extracellular Matrix and Growth Factor Gene Expression in Lung Fibroblasts. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 317, L283–L294. [Google Scholar] [CrossRef]
- Guo, J.; Wang, S.; Xia, H.; Shi, D.; Chen, Y.; Zheng, S.; Chen, Y.; Gao, H.; Guo, F.; Ji, Z.; et al. Cytokine Signature Associated with Disease Severity in COVID-19. Front. Immunol. 2021, 12, 681516. [Google Scholar] [CrossRef]
- Metcalfe, S.M. COVID-19 Lockdown: De-Risking Exit by Protecting the Lung with Leukaemia Inhibitory Factor (LIF). Med. Drug Discov. 2020, 6, 100043. [Google Scholar] [CrossRef]
- Bonhomme, O.; André, B.; Gester, F.; de Seny, D.; Moermans, C.; Struman, I.; Louis, R.; Malaise, M.; Guiot, J. Biomarkers in Systemic Sclerosis-Associated Interstitial Lung Disease: Review of the Literature. Rheumatology 2019, 58, 1534–1546. [Google Scholar] [CrossRef] [PubMed]
- Kamle, S.; Ma, B.; He, C.H.; Akosman, B.; Zhou, Y.; Lee, C.M.; El-Deiry, W.S.; Huntington, K.; Liang, O.; Machan, J.T.; et al. Chitinase 3-like-1 Is a Therapeutic Target That Mediates the Effects of Aging in COVID-19. JCI Insight 2021, 6, e148749. [Google Scholar] [CrossRef]
- Williams, J.P.; Weiser, M.R.; Pechet, T.T.V.; Kobzik, L.; Moore, F.D.; Hechtman, H.B. α1-Acid Glycoprotein Reduces Local and Remote Injuries after Intestinal Ischemia in the Rat. Am. J. Physiol. Gastrointest. Liver Physiol. 1997, 273, G1031–G1035. [Google Scholar] [CrossRef]
- Hsiao, S.-Y.; Lai, Y.-R.; Kung, C.-T.; Tsai, N.-W.; Su, C.-M.; Huang, C.-C.; Wang, H.-C.; Cheng, B.-C.; Su, Y.-J.; Lin, W.-C.; et al. α-1-Acid Glycoprotein Concentration as an Outcome Predictor in Adult Patients with Sepsis. BioMed Res. Int. 2019, 2019, 3174896. [Google Scholar] [CrossRef] [PubMed]
- Kimhofer, T.; Lodge, S.; Whiley, L.; Gray, N.; Loo, R.L.; Lawler, N.G.; Nitschke, P.; Bong, S.-H.; Morrison, D.L.; Begum, S.; et al. Integrative Modeling of Quantitative Plasma Lipoprotein, Metabolic, and Amino Acid Data Reveals a Multiorgan Pathological Signature of SARS-CoV-2 Infection. J. Proteome Res. 2020, 19, 4442–4454. [Google Scholar] [CrossRef]
- Doni, A.; Mantovani, A.; Bottazzi, B.; Russo, R.C. PTX3 Regulation of Inflammation, Hemostatic Response, Tissue Repair, and Resolution of Fibrosis Favors a Role in Limiting Idiopathic Pulmonary Fibrosis. Front. Immunol. 2021, 12, 676702. [Google Scholar] [CrossRef]
- Brunetta, E.; Folci, M.; Bottazzi, B.; De Santis, M.; Gritti, G.; Protti, A.; Mapelli, S.N.; Bonovas, S.; Piovani, D.; Leone, R.; et al. Macrophage Expression and Prognostic Significance of the Long Pentraxin PTX3 in COVID-19. Nat. Immunol. 2021, 22, 19–24. [Google Scholar] [CrossRef]
- Hansen, C.B.; Sandholdt, H.; Møller, M.E.E.; Pérez-Alós, L.; Pedersen, L.; Bastrup Israelsen, S.; Garred, P.; Benfield, T. Prediction of Respiratory Failure and Mortality in COVID-19 Patients Using Long Pentraxin PTX3. J. Innate Immun. 2022, 14, 493–501. [Google Scholar] [CrossRef]
- Ong, S.W.X.; Fong, S.-W.; Young, B.E.; Chan, Y.-H.; Lee, B.; Amrun, S.N.; Chee, R.S.-L.; Yeo, N.K.-W.; Tambyah, P.; Pada, S.; et al. Persistent Symptoms and Association With Inflammatory Cytokine Signatures in Recovered Coronavirus Disease 2019 Patients. Open Forum Infect. Dis. 2021, 8, ofab156. [Google Scholar] [CrossRef]
- Nossent, E.J.; Schuurman, A.R.; Reijnders, T.D.Y.; Saris, A.; Jongerius, I.; Blok, S.G.; de Vries, H.; Duitman, J.; Vonk Noordegraaf, A.; Meijboom, L.J.; et al. Pulmonary Procoagulant and Innate Immune Responses in Critically Ill COVID-19 Patients. Front. Immunol. 2021, 12, 664209. [Google Scholar] [CrossRef] [PubMed]
- Rosner, M.H.; Reis, T.; Husain-Syed, F.; Vanholder, R.; Hutchison, C.; Stenvinkel, P.; Blankestijn, P.J.; Cozzolino, M.; Juillard, L.; Kashani, K.; et al. Classification of Uremic Toxins and Their Role in Kidney Failure. Clin. J. Am. Soc. Nephrol. 2021, 16, 1918–1928. [Google Scholar] [CrossRef] [PubMed]
- Ciceri, P.; Cozzolino, M. Expanded Haemodialysis as a Current Strategy to Remove Uremic Toxins. Toxins 2021, 13, 380. [Google Scholar] [CrossRef] [PubMed]

| Demographical and Clinical Characteristics | Total 77 | eGFR < 30 mL/min. 23 (30%) | eGFR 30–60 mL/min. 33 (43%) | eGFR > 60 mL/min. 21 (27%) | p-Value (group 0 Vs. 1 Vs. 2) |
|---|---|---|---|---|---|
| Sex, (n, %) | |||||
| M | 37 (48%) | 11 (48%) | 20 (61%) | 9 (43%) | |
| F | 40 (52%) | 12 (52%) | 13 (39%) | 12 (57%) | p = 0.3980 |
| Age, (median, IQR) | 79 (70–86) | 79 (73–86) | 79 (73–87) | 75 (61–84) | p = 0.3513 |
| Ethnicity, (n, %) | |||||
| White/Caucasian | 72 (94%) | 21 (91%) | 32 (97%) | 19 (90%) | |
| Latin American | 3 (4%) | 1 (4%) | 0 (0%) | 2 (10%) | |
| East Asian | 1 (1%) | 0 (0%) | 0 (0%) | ||
| Maghreb/Middle East | 1 (1%) | 0 (0%) | 1 (3%) | 0 (0%) | p = 0.3435 |
| Comorbidities, (n, %) | |||||
| Hypertension | 51 (66%) | 19 (83%) | 21 (64%) | 11 (52%) | p = 0.0973 |
| CVD | 38 (49%) | 14 (61%) | 15 (45%) | 9 (43%) | p = 0.4116 |
| IMA | 17 (22%) | 6 (26%) | 8 (24%) | 3 (14%) | p = 0.5928 |
| CHF | 11 (14%) | 5 (22%) | 4 (12%) | 2 (10%) | p = 0.4587 |
| Arrhytmia | 15 (19%) | 6 (26%) | 5 (15%) | 4 (19%) | p = 0.6478 |
| Valvulopathy | 4 (5%) | 1 (4%) | 3 (9%) | 0 (0%) | p = 0.3327 |
| Cerebrovascular disease | 8 (10%) | 1 (4%) | 4 (12%) | 3 (14%) | p = 0.5090 |
| Dementia | 17 (22%) | 6 (26%) | 7 (21%) | 4 (19%) | p = 0.8431 |
| Chronic pulmonary disease | 9 (12%) | 2 (9%) | 5 (15%) | 2 (10%) | p = 0.7124 |
| Cancer | 9 (12%) | 3 (13%) | 6 (18%) | 0 (0%) | p = 0.1244 |
| CKD | 20 (26%) | 16 (70%) | 4 (12%) | 0 (0%) | p < 0.0001 |
| Dialysis | 4 (5%) | 0 (0%) | 0 (0%) | 0 (0%) | - |
| Connettivopaties | 2 (3%) | 1 (23%) | 1 (3%) | 0 (0%) | p = 0.6495 |
| Diabetes | 25 (32%) | 9 (39%) | 14 (42%) | 2 (10%) | p = 0.0302 |
| Chronic liver disease | 4 (5%) | 1 (4%) | 2 (6%) | 0 (0%) | p = 0.3327 |
| Vascular disease | 16 (21%) | 7 (30%) | 7 (21%) | 2 (10%) | p = 0.2319 |
| Age adj. charlson score | |||||
| Category 1 | 6 (8%) | 3 (13%) | 0 (0%) | 3 (14%) | p = 0.0861 |
| Category 2 | 13 (17%) | 2 (9%) | 7 (21%) | 4 (19%) | p = 0.4472 |
| Category 3 | 22 (28%) | 7 (30%) | 10 (30%) | 5 (24%) | p = 0.8517 |
| Category 4 | 36 (47%) | 11 (48%) | 16 (48%) | 9 (43%) | p = 0.0194 |
| BMI, (median, IQR) | 24.20 (22.92–29.07) | 22.86 (20.43–23.74) | 23.53 (22.15–29.30) | 27.12 (24.34–29.02) | p = 0.2683 |
| Symptoms at the admission. (n. %) | |||||
| Fever | 53 (69%) | 15 (65%) | 22 (67%) | 16 (76%) | p = 0.6900 |
| Cough | 26 (34%) | 9 (39%) | 11 (33%) | 6 (29%) | p = 0.7588 |
| Productive cough | 3 (4%) | 1 (4%) | 1 (3%) | 1 (5%) | p = 0.9415 |
| Dyspnea | 43 (56%) | 15 (65%) | 20 (61%) | 8 (38%) | p = 0.1491 |
| Fatigue | 15 (19%) | 4 (17%) | 7 (21%) | 4 (19%) | p = 0.9373 |
| Abdominal pain | 4 (5%) | 2 (9%) | 1 (3%) | 1 (5%) | p = 0.6395 |
| Nausea/vomiting | 2 (3%) | 0 (0%) | 1 (3%) | 1 (5%) | p = 0.5985 |
| Diarrhoea | 3 (4%) | 1 (4%) | 2 (6%) | 0 (0%) | p = 0.5281 |
| Chest pain | 2 (3%) | 0 (0%) | 2 (6%) | 0 (0%) | p = 0.2544 |
| Syncope | 1 (1%) | 1 (4%) | 0 (0%) | 0 (0%) | p = 0.3044 |
| Arthromyalgia | 2 (3%) | 0 (0%) | 2 (6%) | 0 (0%) | p = 0.2544 |
| Anosmia/dysgeusia | 3 (4%) | 1 (4%) | 1 (3%) | 1 (5%) | p = 0.9415 |
| Duration of symptoms before the hospitalization. days, (median, IQR) | 4 (2–8) | 4 (2–6) | 5 (3–9) | 5 (2–8) | p = 0.5657 |
| Radiological pulmonary infiltrates upon admission, (n, %) | 67 (87%) | 20 (87%) | 30 (91%) | 17 (81%) | p = 0.5695 |
| Respiratory setting upon admission, (n, %) | |||||
| Room air | 60 (78%) | 16 (70%) | 29 (88%) | 15 (71%) | |
| O2-therapy | 17 (22%) | 7 (30%) | 4 (12%) | 6 (29%) | p = 0.1873 |
| Respiratory parameters upon admission, (median, IQR) | |||||
| pO2 | 72 (62–88) | 63 (70–78) | 70 (63–78) | 71 (61–87) | p = 0.5435 |
| pO2/FiO2 | 251 (307–357) | 286 (305–351) | 205 (286–351) | 296 (209–343) | p = 0.2812 |
| SpO2 | 96 (91–97) | 95 (93–97) | 95 (93–97) | 93 (89–96) | p = 0.0140 |
| Blood examinations upon admission. (median. IRQ) | |||||
| Hemoglobin, g/dL | 12.0 (11.0–13.30) | 11.6 (11.0–12.5) | 12.0 (11.0–13.5) | 12.5 (11.7–13.4) | p = 0.2495 |
| WBC count, 103/μL | 7.00 (5.42–9.71) | 7.27 (5.64–10.19) | 7.33 (5.83–9.71) | 6.45 (5.26–7.66) | p = 0.3568 |
| Neutrophils, 103/μL | 4.93 (3.93–7.51) | 5.15 (4.03–8.45) | 5.82 (4.10–7.57) | 4.13 (3.43–5.94) | p = 0.1097 |
| Lymphocytes, 103/μL | 1.04 (0.64–1.34) | 1.01 (0.64–1.21) | 0.83 (0.63–1.45) | 1.10 (0.73–1.59) | p = 0.3656 |
| NL ratio | 5.16 (3.12–9.81) | 6.73 (3.48–12.75) | 6.14 (3.77–11.13) | 3.66 (2.60–5.29) | p = 0.0454 |
| Monocytes, 103/μL | 0.54 (0.34–0.77) | 0.53 (0.35–0.89) | 0.57 (0.34–0.89) | 0.49 (0.27–0.65) | p = 0.4117 |
| Platelets 103/μL | 204 (162–304) | 177 (145–259) | 206 (167–312) | 230 (176–293) | p = 0.4868 |
| C-reactive protein, mg/L | 68.9 (27.3–99.1) | 75.8 (32.7–98.1) | 67.6 (27.8–132.3) | 60.0 (21.4–80.2) | p = 0.4752 |
| LDH, U/L | 288 (211–390) | 307 (226–429) | 279 (209–398) | 293 (210–374) | p = 0.8047 |
| Creatine-P-kinase, U/L | 92 (45–189) | 97 (44–175) | 126 (67–203) | 61 (39–181) | p = 0.2437 |
| D-dimer, ng/mL | 580 (310–1448) | 980 (487–3189) | 600 (268–899) | 494 (260–908) | p = 0.1572 |
| ALT, U/L | 21 (16–29) | 19 (15–24) | 24 (18–34) | 21 (16–29) | p = 0.0404 |
| AST, U/L | 37 (28–49) | 31 (23–47) | 39 (30–51) | 34 (31–46) | p = 0.1101 |
| Creatinin, mg/dL | 1.3 (1.0–2.2) | 3.4 (2.3–4.2) | 1.2 (1.2–1.4) | 0.7 (0.6–0.9) | p < 0.0001 |
| Procalcitonin, ng/mL | 0.13 (0.07–1.21) | 0.26 (0.13–5.00) | 0.11 (0.07–0.47) | 0.09 (0.04–0.56) | p = 0.0889 |
| Ferritin, ng/mL | 436 (231–829) | 401 (231–865) | 478 (179–827) | 436 (310–567) | p = 0.9435 |
| eGFR | 48.4 (26.2–61.3) | 12.3 (8.7–24.9) | 49.1 (43.0–55.7) | 82.0 (73.6–98.4) | p < 0.0001 |
| Medical therapy, (n, %) | |||||
| Lopinavir/darunavir | 10 (13%) | 3 (13%) | 2 (6%) | 5 (24%) | p = 0.1672 |
| Hydroxychloroquine | 57 (74%) | 16 (70%) | 26 (79%) | 15 (71%) | p = 0.7044 |
| Steroids | 17 (22%) | 8 (35%) | 5 (15%) | 4 (19%) | p = 0.2029 |
| Heparin | 58 (75%) | 16 (70%) | 29 (88%) | 13 (62%) | p = 0.0727 |
| Biological drug | 10 (13%) | 5 (22%) | 4 (12%) | 1 (5%) | p = 0.2419 |
| Maximum respiratory support, (n, %) | |||||
| Room air | 10 (13%) | 1 (4%) | 4 (12%) | 5 (24%) | p = 0.1559 |
| O2-therapy | 32 (41%) | 8 (35%) | 16 (49%) | 8 (38%) | p = 0.5515 |
| C-pap | 26 (34%) | 11 (48%) | 9 (27%) | 6 (29%) | p = 0.2812 |
| NIMV | 7 (9%) | 2 (9%) | 4 (12%) | 1 (5%) | p = 0.6547 |
| Oro-tracheal intubation | 2 (3%) | 1 (4%) | 0 (0%) | 1 (5%) | - |
| Outcome, (n, %) | |||||
| Discharge | 42 (55%) | 8 (43%) | 19 (57%) | 13 (62%) | |
| Death | 35 (45%) | 13 (57%) | 14 (43%) | 8 (38%) | p = 0.4238 |
| Time from symptoms onset to outcome, days, (median, IQR) | 18 (11–35) | 17 (10–50) | 18 (12–35) | 19 (14–26) | p = 0.9860 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciceri, P.; Bono, V.; Magagnoli, L.; Sala, M.; d’Arminio Monforte, A.; Galassi, A.; Barassi, A.; Marchetti, G.; Cozzolino, M. Cytokine and Chemokine Retention Profile in COVID-19 Patients with Chronic Kidney Disease. Toxins 2022, 14, 673. https://doi.org/10.3390/toxins14100673
Ciceri P, Bono V, Magagnoli L, Sala M, d’Arminio Monforte A, Galassi A, Barassi A, Marchetti G, Cozzolino M. Cytokine and Chemokine Retention Profile in COVID-19 Patients with Chronic Kidney Disease. Toxins. 2022; 14(10):673. https://doi.org/10.3390/toxins14100673
Chicago/Turabian StyleCiceri, Paola, Valeria Bono, Lorenza Magagnoli, Matteo Sala, Antonella d’Arminio Monforte, Andrea Galassi, Alessandra Barassi, Giulia Marchetti, and Mario Cozzolino. 2022. "Cytokine and Chemokine Retention Profile in COVID-19 Patients with Chronic Kidney Disease" Toxins 14, no. 10: 673. https://doi.org/10.3390/toxins14100673
APA StyleCiceri, P., Bono, V., Magagnoli, L., Sala, M., d’Arminio Monforte, A., Galassi, A., Barassi, A., Marchetti, G., & Cozzolino, M. (2022). Cytokine and Chemokine Retention Profile in COVID-19 Patients with Chronic Kidney Disease. Toxins, 14(10), 673. https://doi.org/10.3390/toxins14100673







