Aggregatibacter actinomycetemcomitans as the Aetiological Cause of Rheumatoid Arthritis: What Are the Unsolved Puzzles?
Abstract
1. Introduction
1.1. Aggregatibacter Actinomycetemcomitans Is a Pathogenic Bacterium
1.2. LtxA as a Major Virulence Factor of Aa
1.3. Ltx Operon
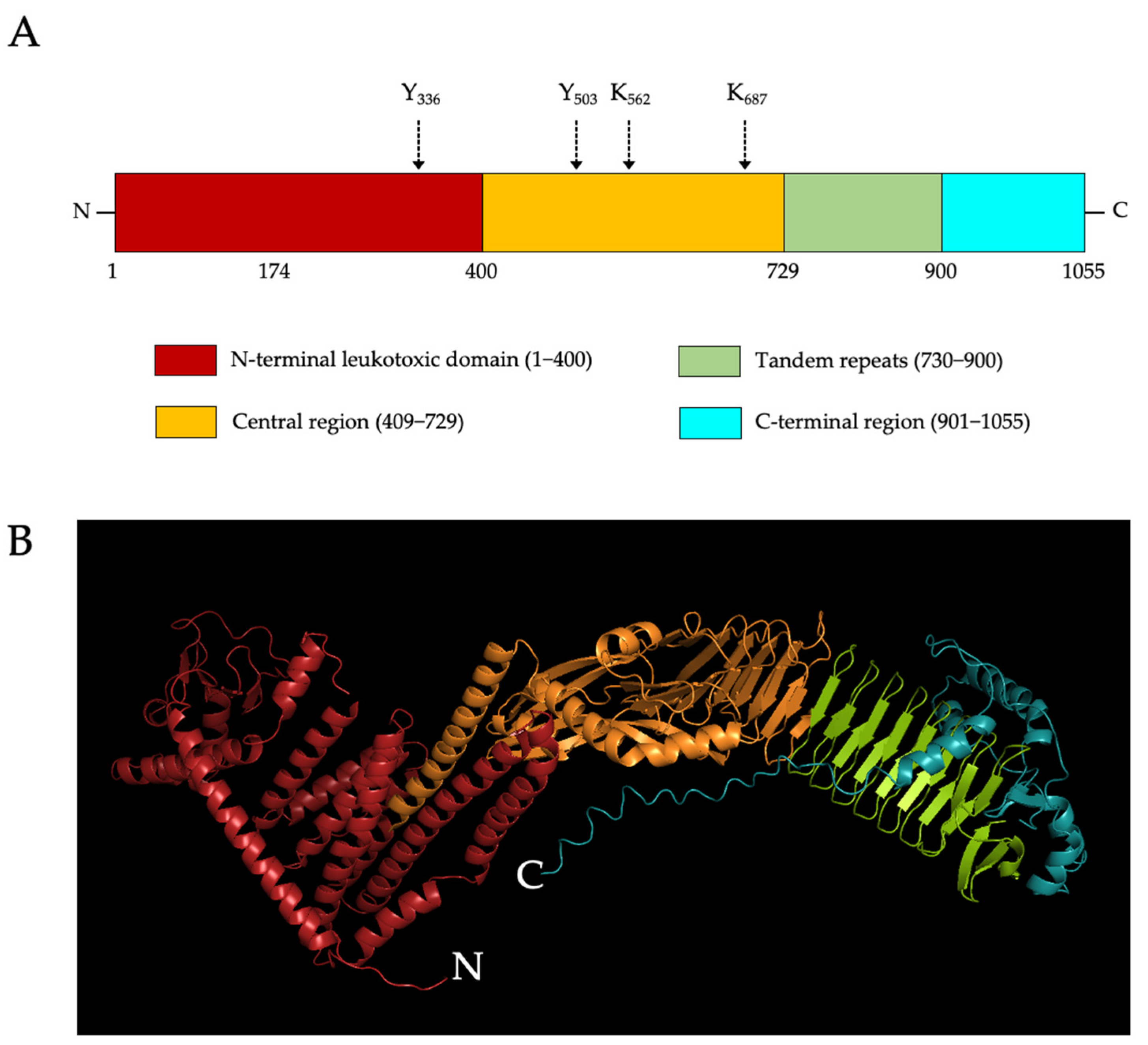
1.4. LtxA Structure
2. Hematopoietic Cells as a Target of LtxA
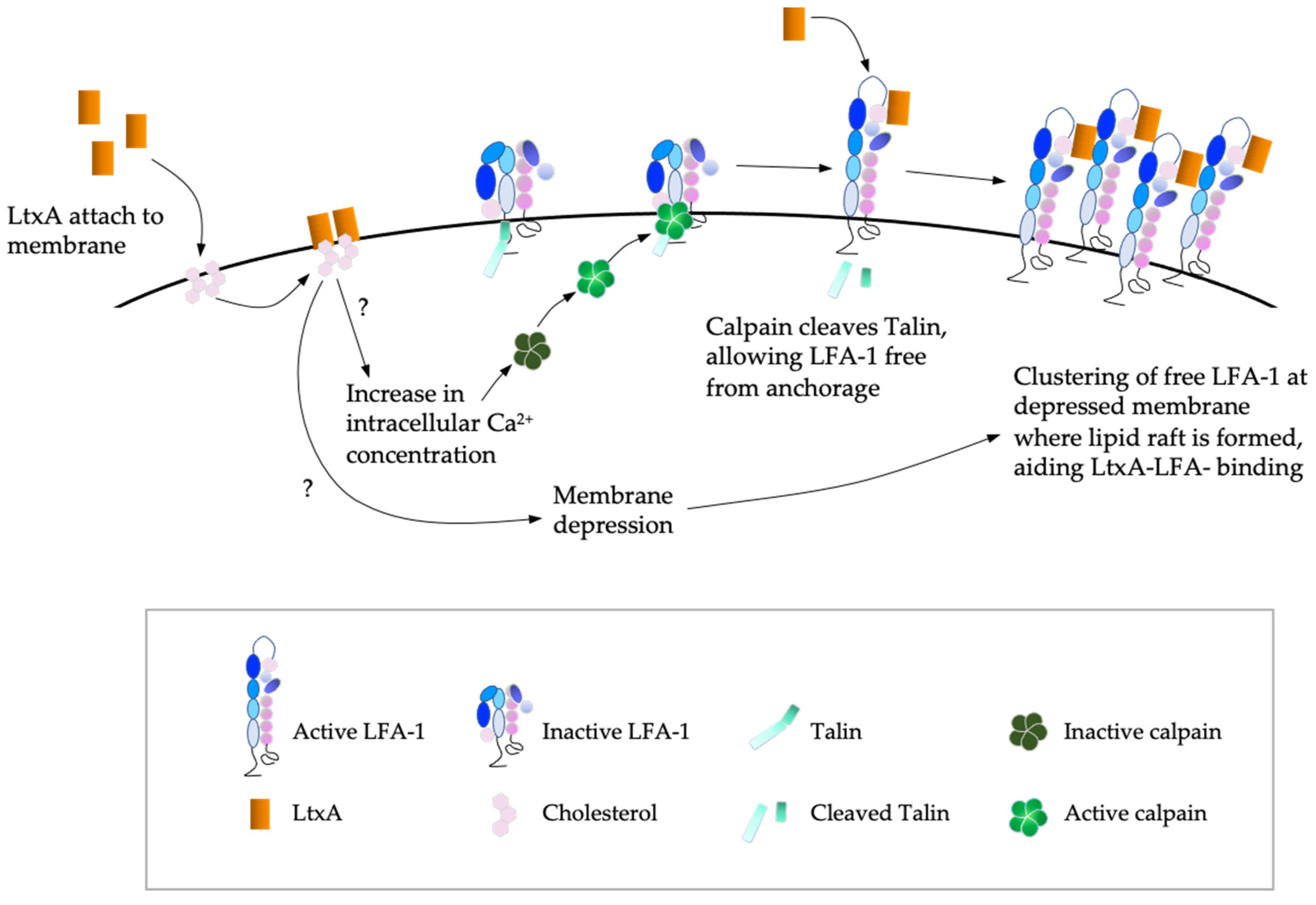
2.1. Leukocyte Function Associated Antigen-1 (LFA-1) as the Target Molecule
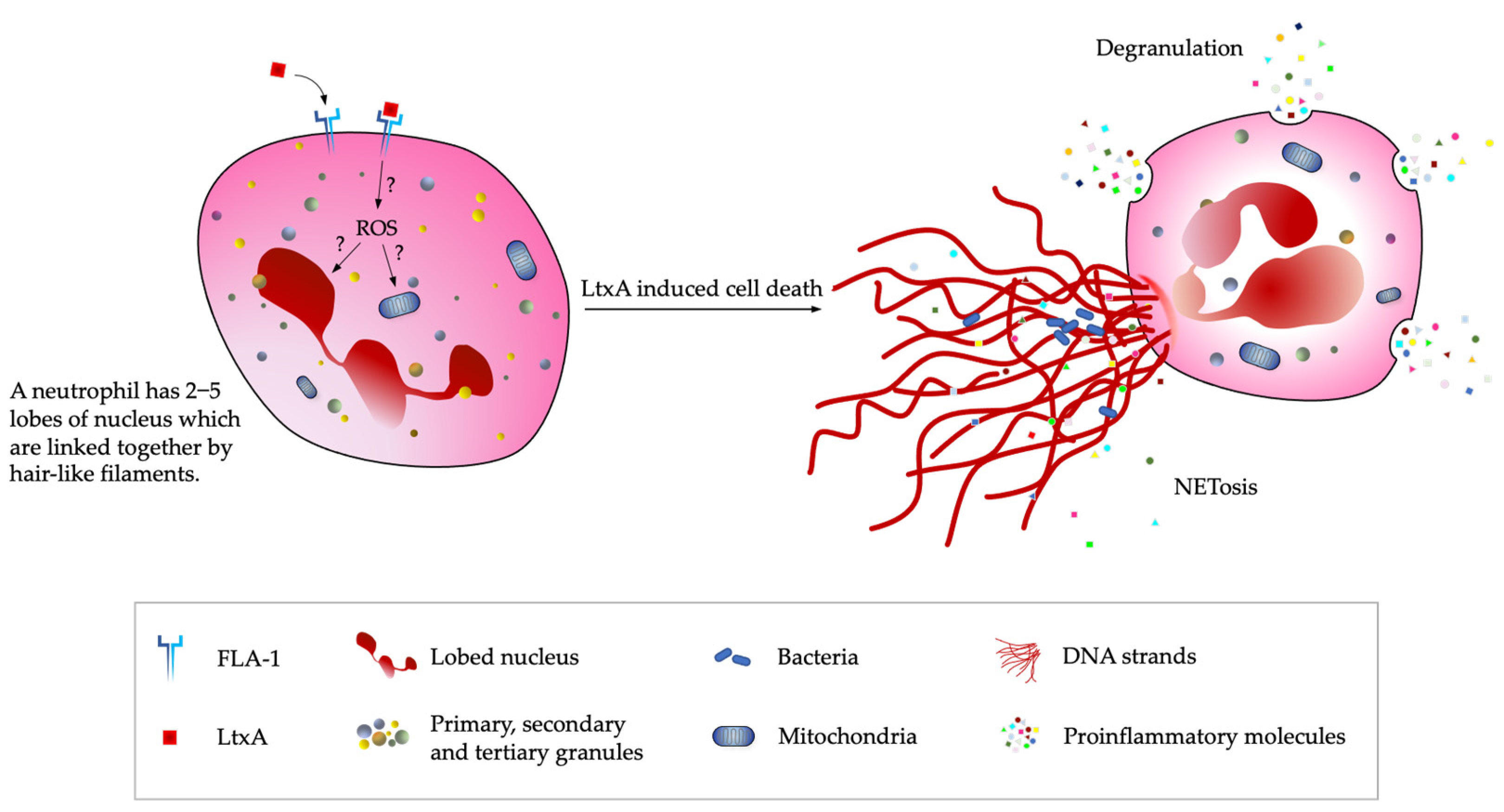
2.2. Mechanism of LtxA-Induced Cell Death
3. Insight into the Association between RA and Periodontitis
3.1. Previous Studies on the Association between RA and Periodontitis
3.2. The Effect of Periodontal Therapy on RA
3.3. The Prevalence of Oral Microbiota Dysbiosis and Various Periodontal Bacteria in RA
3.4. Protein Citrullination Bridges Periodontitis and RA
4. Aa as a Promising New Putative Periodontopathogen in RA Aetiology
5. Can Aa and LtxA Unlock the Mystery behind the Triggering of RA?
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Klinger, R. Untersuchungen uber menschliche aktinomycose. Zent. Bakteriol. 1912, 62, 191–200. [Google Scholar]
- Topley, W.W.C.; Wilson, G.S. The Principles of Bacteriology and Immunity; Edward Arnold & Co: London, UK, 1929; Volume 1. [Google Scholar]
- Potts, T.V.; Zambon, J.J.; Genco, R.J. Reassignment of Actinobacillus actinomycetemcomitans to the genus Haemophilus as Haemophilus actinomycetemcomitans comb. nov. Int. J. Syst. Bacteriol. 1985, 35, 337–341. [Google Scholar] [CrossRef]
- Nørskov-Lauritsen, N.; Kilian, M. Reclassification of Actinobacillus actinomycetemcomitans, Haemophilus aphrophilus, Haemophilus paraphrophilus and Haemophilus segnis as Aggregatibacter actinomycetemcomitans gen. nov., comb. nov., Aggregatibacter aphrophilus comb. nov. and Aggregatibacter segnis comb. nov., and emended description of Aggregatibacter aphrophilus to include V factor-dependent and V factor-independent isolates. Int. J. Syst. Evol. Microbiol. 2006, 56, 2135–2146. [Google Scholar] [CrossRef]
- Xuedong, Z.; Yuqing, L. Atlas of Oral Microbiology, 2nd ed.; Springer: New York, NY, USA, 2020. [Google Scholar]
- Raja, M.; Ummer, F.; Dhivakar, C.P. Aggregatibacter actinomycetemcomitans—A tooth killer. J. Clin. Diagn. Res. 2014, 8, ZE13. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information (NCBI). Aggregatibacter actinomycetemcomitans Strain VT1169, Complete Genome—Nucleotide—NCBI. Available online: https://www.ncbi.nlm.nih.gov/nuccore/NZ_CP012958.1 (accessed on 2 December 2021).
- Kaplan, J.B.; Perry, M.B.; MacLean, L.L.; Furgang, D.; Wilson, M.E.; Fine, D.H. Structural and genetic analyses of O polysaccharide from Actinobacillus actinomycetemcomitans serotype f. Infect. Immun. 2001, 69, 5375–5384. [Google Scholar] [CrossRef] [PubMed]
- Nørskov-Lauritsen, N.; Claesson, R.; Jensen, A.B.; Åberg, C.H.; Haubek, D. Aggregatibacter actinomycetemcomitans: Clinical significance of a pathobiont subjected to ample changes in classification and nomenclature. Pathogens 2019, 8, 243. [Google Scholar] [CrossRef] [PubMed]
- Suprith, S.; Setty, S.; Bhat, K.; Thakur, S. Serotypes of Aggregatibacter actinomycetemcomitans in relation to periodontal status and assessment of leukotoxin in periodontal disease: A clinico-microbiological study. J. Indian Soc. Periodontol. 2018, 22, 201–208. [Google Scholar] [CrossRef]
- Christersson, L.A. Actinobacillus actinomycetemcomitans and localized juvenile periodontitis. Clinical, microbiologic and histologic studies. Swed. Dent. J. Suppl. 1993, 90, 1–46. [Google Scholar]
- Haubek, D.; Ennibi, O.K.; Poulsen, K.; Poulsen, S.; Benzarti, N.; Kilian, M. Early-onset periodontitis in morocco is associated with the highly leukotoxic clone of Actinobacillus actinomycetemcomitans. J. Dent. Res. 2001, 80, 1580–1583. [Google Scholar] [CrossRef] [PubMed]
- Fine, D.H.; Markowitz, K.; Fairlie, K.; Tischio-Bereski, D.; Ferrendiz, J.; Furgang, D.; Paster, B.J.; Dewhirs, F.E. A consortium of Aggregatibacter actinomycetemcomitans, Streptococcus parasanguinis, and Filifactor alocis is present in sites prior to bone loss in a longitudinal study of localized aggressive periodontitis. J. Clin. Microbiol. 2013, 51, 2850–2861. [Google Scholar] [CrossRef]
- Fine, D.H.; Patil, A.G.; Velusamy, S.K. Aggregatibacter actinomycetemcomitans (Aa) under the Radar: Myths and misunderstandings of Aa and its role in aggressive periodontitis. Front. Immunol. 2019, 10, 728. [Google Scholar] [CrossRef]
- Tsai, C.C.; Mcarthur, W.P.; Baehni, P.C.; Hammond, B.F.; Taichman, N.S. Extraction and partial characterization of a leukotoxin from a plaque-derived gram-negative microorganism. Infect. Immun. 1979, 25, 427–439. [Google Scholar] [CrossRef]
- Balashova, N.V.; Crosby, J.A.; Al Ghofaily, L.; Kachlany, S.C. Leukotoxin confers beta-hemolytic activity to Actinobacillus actinomycetemcomitans. Infect. Immun. 2006, 74, 2015–2021. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A. Aggregatibacter actinomycetemcomitans leukotoxin: A powerful tool with capacity to cause imbalance in the host inflammatory response. Toxins 2011, 3, 242–259. [Google Scholar] [CrossRef]
- Petersen, P.E.; Ogawa, H. The global burden of periodontal disease: Towards integration with chronic disease prevention and control. Periodontology 2000 2012, 60, 15–39. [Google Scholar] [CrossRef]
- Claesson, R.; Johansson, A.; Belibasakis, G.; Hänström, L.; Kalfas, S. Release and activation of matrix metalloproteinase 8 from human neutrophils triggered by the leukotoxin of Actinobacillus actinomycetemcomitans. J. Periodontal Res. 2002, 37, 353–359. [Google Scholar] [CrossRef]
- Kelk, P.; Abd, H.; Claesson, R.; Sandström, G.; Sjö Stedt, A.; Johansson, A. Cellular and molecular response of human macrophages exposed to Aggregatibacter actinomycetemcomitans leukotoxin. Cell Death Dis. 2011, 2, e126. [Google Scholar] [CrossRef] [PubMed]
- Hirschfeld, J.; Roberts, H.M.; Chapple, I.L.C.; Parčina, M.; Jepsen, S.; Johansson, A.; Claesson, R. Effects of Aggregatibacter actinomycetemcomitans leukotoxin on neutrophil migration and extracellular trap formation. J. Oral Microbiol. 2016, 8, 33070. [Google Scholar] [CrossRef]
- Konig, M.F.; Abusleme, L.; Reinholdt, J.; Palmer, R.J.; Teles, R.P.; Sampson, K.; Rosen, A.; Nigrovic, P.A.; Sokolove, J.; Giles, J.T.; et al. Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Sci. Transl. Med. 2016, 8, 369ra176. [Google Scholar] [CrossRef]
- Sampathkumar, V.; Velusamy, S.K.; Godboley, D.; Fine, D.H. Increased leukotoxin production: Characterization of 100 base pairs within the 530 base pair leukotoxin promoter region of Aggregatibacter actinomycetemcomitans. Sci. Rep. 2017, 7, 1887. [Google Scholar] [CrossRef] [PubMed]
- Haubek, D.; Ennibi, O.K.; Poulsen, K.; Væth, M.; Poulsen, S.; Kilian, M. Risk of aggressive periodontitis in adolescent carriers of the JP2 clone of Aggregatibacter (Actinobacillus) actinomycetemcomitans in Morocco: A prospective longitudinal cohort study. Lancet 2008, 371, 237–242. [Google Scholar] [CrossRef]
- Höglund Åberg, C.; Kwamin, F.; Claesson, R.; Dahlén, G.; Johansson, A.; Haubek, D. Progression of attachment loss is strongly associated with presence of the JP2 genotype of Aggregatibacter actinomycetemcomitans: A prospective cohort study of a young adolescent population. J. Clin. Periodontol. 2014, 41, 232–241. [Google Scholar] [CrossRef]
- Kachlany, S.C. Aggregatibacter actinomycetemcomitans leukotoxin: From threat to therapy. J. Dent. Res. 2010, 89, 561–570. [Google Scholar] [CrossRef]
- Lally, E.T.; Golub, E.E.; Kieba, I.R.; Taichman, N.S.; Decker, S.; Berthold, P.; Gibson, C.W.; Demuth, D.R.; Rosenbloom, J. Structure and function of the B and D genes of the Actinobacillus actinomycetemcomitans leukotoxin complex. Microb. Pathog. 1991, 11, 111–121. [Google Scholar] [CrossRef]
- Lally, E.T.; Kieba, I.R.; Golub, E.E.; Lear, J.D.; Tanaka, J.C. Structure/Function Aspects of Actinobacillus actinomycetemcomitans Leukotoxin. J. Periodontol. 1996, 67, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C.; Balashova, N.V.; Epand, R.M.; Epand, R.F.; Bragin, A.; Kachlany, S.C.; Walters, M.J.; Du, Y.; Boesze-Battaglia, K.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin utilizes a cholesterol recognition/amino acid consensus site for membrane association. J. Biol. Chem. 2013, 288, 23607–23621. [Google Scholar] [CrossRef] [PubMed]
- Stanley, P.; Packman, L.C.; Koronakis, V.; Hughes, C. Fatty acylation of two internal lysine residues required for the toxic activity of Escherichia coli hemolysin. Science 1994, 266, 1992–1996. [Google Scholar] [CrossRef]
- Balashova, N.V.; Shah, C.; Patel, J.K.; Megalla, S.; Kachlany, S.C. Aggregatibacter actinomycetemcomitans LtxC is required for leukotoxin activity and initial interaction between toxin and host cells. Gene 2009, 443, 42–47. [Google Scholar] [CrossRef]
- Sato, N.; Takahashi, K.; Ohta, H.; Kurihara, H.; Fukui, K.; Murayama, Y.; Taniguchi, S. Effect of Ca2+ on the binding of Actinobacillus actinomycetemcomitans leukotoxin and the cytotoxicity to promyelocytic leukemia HL-60 cells. Biochem. Mol. Biol. Int. 1993, 29, 899–905. [Google Scholar]
- Kraig, E.; Dailey, T.; Kolodrubetz, D. Nucleotide sequence of the leukotoxin gene from Actinobacillus actinomycetemcomitans: Homology to the alpha-hemolysin/leukotoxin gene family. Infect. Immun. 1990, 58, 920–929. [Google Scholar] [CrossRef] [PubMed]
- Hogg, N.; Smith, A.; McDowall, A.; Giles, K.; Stanley, P.; Laschinger, M.; Henderson, R. How T cells use LFA-1 to attach and migrate. Immunol. Lett. 2004, 92, 51–54. [Google Scholar] [CrossRef]
- Walling, B.L.; Kim, M. LFA-1 in T cell migration and differentiation. Front. Immunol. 2018, 9, 952. [Google Scholar] [CrossRef] [PubMed]
- Dileepan, T.; Kachlany, S.C.; Balashova, N.V.; Patel, J.; Maheswaran, S.K. Human CD18 is the functional receptor for Aggregatibacter actinomycetemcomitans leukotoxin. Infect. Immun. 2007, 75, 4851–4856. [Google Scholar] [CrossRef] [PubMed]
- Kieba, I.R.; Fong, K.P.; Tang, H.Y.; Hoffman, K.E.; Speicher, D.W.; Klickstein, L.B.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin requires β-sheets 1 and 2 of the human CD11a β-propeller for cytotoxicity. Cell. Microbiol. 2007, 9, 2689–2699. [Google Scholar] [CrossRef] [PubMed]
- Shimaoka, M.; Xiao, T.; Liu, J.H.; Yang, Y.; Dong, Y.; Jun, C.D.; McCormack, A.; Zhang, R.; Joachimiak, A.; Takagi, J.; et al. Structures of the αL I domain and its complex with ICAM-1 reveal a shape-shifting pathway for integrin regulation. Cell 2003, 112, 99–111. [Google Scholar] [CrossRef]
- Kachlany, S.C.; Schwartz, A.B.; Balashova, N.V.; Hioe, C.E.; Tuen, M.; Le, A.; Kaur, M.; Mei, Y.; Rao, J. Anti-leukemia activity of a bacterial toxin with natural specificity for LFA-1 on white blood cells. Leuk. Res. 2010, 34, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Nygren, P.; Balashova, N.; Brown, A.C.; Kieba, I.; Dhingra, A.; Boesze-Battaglia, K.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin causes activation of lymphocyte function-associated antigen 1. Cell. Microbiol. 2019, 21, e12967. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C.; Boesze-Battaglia, K.; Du, Y.; Stefano, F.P.; Kieba, I.R.; Epand, R.F.; Kakalis, L.; Yeagle, P.L.; Epand, R.M.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin cytotoxicity occurs through bilayer destabilization. Cell. Microbiol. 2012, 14, 869–881. [Google Scholar] [CrossRef]
- Bouillot, S.; Reboud, E.; Huber, P. Functional consequences of calcium influx promoted by bacterial pore-forming toxins. Toxins 2018, 10, 387. [Google Scholar] [CrossRef]
- Fong, K.P.; Pacheco, C.M.F.; Otis, L.L.; Baranwal, S.; Kieba, I.R.; Harrison, G.; Hersh, E.V.; Boesze-Battaglia, K.; Lally, E.T. Actinobacillus actinomycetemcomitans leukotoxin requires lipid microdomains for target cell cytotoxicity. Cell. Microbiol. 2006, 8, 1753–1767. [Google Scholar] [CrossRef]
- Fong, K.P.; Tang, H.Y.; Brown, A.C.; Kieba, I.R.; Speicher, D.W.; Boesze-Battaglia, K.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin is post-translationally modified by addition of either saturated or hydroxylated fatty acyl chains. Mol. Oral Microbiol. 2011, 26, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Korostoff, J.; Yamaguchi, N.; Miller, M.; Kieba, I.; Lally, E.T. Perturbation of mitochondrial structure and function plays a central role in Actinobacillus actinomycetemcomitans leukotoxin-induced apoptosis. Microb. Pathog. 2000, 29, 267–278. [Google Scholar] [CrossRef]
- Vega, B.A.; Schober, L.T.; Kim, T.; Belinka, B.A.; Kachlany, S.C. Aggregatibacter actinomycetemcomitans Leukotoxin (LtxA) requires death receptor Fas, in addition to LFA-1, to trigger cell death in T lymphocytes. Infect. Immun. 2019, 87, 85–105. [Google Scholar] [CrossRef]
- Vega, B.A.; Belinka, B.A., Jr.; Kachlany, S.C. Aggregatibacter actinomycetemcomitans leukotoxin (LtxA.; Leukothera®): Mechanisms of action and therapeutic applications. Toxins 2019, 11, 489. [Google Scholar] [CrossRef]
- Johansson, A.; Hänström, L.; Kalfas, S. Inhibition of Actinobacillus actinomycetemcomitans leukotoxicity by bacteria from the subgingival flora. Oral Microbiol. Immunol. 2000, 15, 218–225. [Google Scholar] [CrossRef]
- Johansson, A.; Claesson, R.; Hänström, L.; Sandström, G.; Kalfas, S. Polymorphonuclear leukocyte degranulation induced by leukotoxin from Actinobacillus actinomycetemcomitans. J. Periodontal Res. 2000, 35, 85–92. [Google Scholar] [CrossRef]
- Furugen, R.; Hayashida, H.; Yoshii, Y.; Saito, T. Neutrophil-derived resistin release induced by Aggregatibacter actinomycetemcomitans. FEMS Microbiol. Lett. 2011, 321, 175–182. [Google Scholar] [CrossRef]
- Aulik, N.A.; Hellenbrand, K.M.; Klos, H.; Czuprynski, C.J. Mannheimia haemolytica and its leukotoxin cause neutrophil extracellular trap formation by bovine neutrophils. Infect. Immun. 2010, 78, 4454–4466. [Google Scholar] [CrossRef]
- Palmer, L.J.; Cooper, P.R.; Ling, M.R.; Wright, H.J.; Huissoon, A.; Chapple, I.L.C. Hypochlorous acid regulates neutrophil extracellular trap release in humans. Clin. Exp. Immunol. 2012, 167, 261–268. [Google Scholar] [CrossRef]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018, 18, 134–147. [Google Scholar] [CrossRef]
- Winning, L.; Linden, G.J. Periodontitis and systemic disease: Association or causality? Curr. Oral Health Rep. 2017, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Demmer, R.T.; Molitor, J.A.; Jacobs, D.R.; Michalowicz, B.S. Periodontal disease, tooth loss and incident rheumatoid arthritis: Results from the First National Health and Nutrition Examination Survey and its epidemiological follow-up study. J. Clin. Periodontol. 2011, 38, 998–1006. [Google Scholar] [CrossRef]
- Almutairi, K.; Nossent, J.; Preen, D.; Keen, H.; Inderjeeth, C. The global prevalence of rheumatoid arthritis: A meta-analysis based on a systematic review. Rheumatol. Int. 2021, 41, 863–877. [Google Scholar] [CrossRef]
- Xu, B.; Lin, J. Characteristics and risk factors of rheumatoid arthritis in the United States: An NHANES analysis. PeerJ 2017, 5, e4035. [Google Scholar] [CrossRef]
- Pradhan-Palikhe, P.; Mäntylä, P.; Paju, S.; Buhlin, K.; Persson, G.R.; Nieminen, M.S.; Sinisalo, J.; Pussinen, P.J. Subgingival bacterial burden in relation to clinical and radiographic periodontal parameters. J. Periodontol. 2013, 84, 1809–1817. [Google Scholar] [CrossRef]
- Rannou, F.; François, M.; Corvol, M.T.; Berenbaum, F. Cartilage breakdown in rheumatoid arthritis. Joint Bone Spine 2006, 73, 29–36. [Google Scholar] [CrossRef]
- Salari, M.H.; Kadkhoda, Z. Rate of cultivable subgingival periodontopathogenic bacteria in chronic periodontitis. J. Oral Sci. 2004, 46, 157–161. [Google Scholar] [CrossRef]
- Hirsch, R.; Deng, H.; Laohachai, M.N. Azithromycin in periodontal treatment: More than an antibiotic. J. Periodontol. Res. 2012, 47, 137–148. [Google Scholar] [CrossRef]
- Teughels, W.; Dhondt, R.; Dekeyser, C.; Quirynen, M. Treatment of aggressive periodontitis. Periodontol. 2000 2014, 65, 107–133. [Google Scholar] [CrossRef]
- Prakasam, A.; Elavarasu, S.S.; Natarajan, R.K. Antibiotics in the management of aggressive periodontitis. J. Pharm. Bioallied Sci. 2012, 4, S252–S255. [Google Scholar] [CrossRef]
- Martinez-Martinez, R.E.; Abud-Mendoza, C.; Patiño-Marin, N.; Rizo-Rodríguez, J.C.; Little, J.W.; Loyola-Rodríguez, J.P. Detection of periodontal bacterial DNA in serum and synovial fluid in refractory rheumatoid arthritis patients. J. Clin. Periodontol. 2009, 36, 1004–1010. [Google Scholar] [CrossRef]
- Ogrendik, M. Rheumatoid arthritis is linked to oral bacteria: Etiological association. Mod. Rheumatol. 2009, 19, 453–456. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, B.; Li, S.; Yang, L.; Zhu, D.; Wang, Y.; Wang, H.; Wang, T.; Shi, B.; Gai, Z.; et al. Detection and characterization of bacterial nucleic acids in culture-negative synovial tissue and fluid samples from rheumatoid arthritis or osteoarthritis patients. Sci. Rep. 2018, 8, 14305. [Google Scholar] [CrossRef]
- Mukherjee, A.; Jantsch, V.; Khan, R.; Hartung, W.; Fischer, R.; Jantsch, J.; Ehrenstein, B.; Konig, M.F.; Andrade, F. Rheumatoid arthritis-associated autoimmunity due to Aggregatibacter actinomycetemcomitans and its resolution with antibiotic therapy. Front. Immunol. 2018, 9, 2352. [Google Scholar] [CrossRef] [PubMed]
- Araújo, V.M.A.; Melo, I.M.; Lima, V. Relationship between periodontitis and rheumatoid arthritis: Review of the literature. Mediators Inflamm. 2015, 2015, 259074. [Google Scholar] [CrossRef] [PubMed]
- Baltacioǧlu, E.; Akalin, F.A.; Alver, A.; Deǧer, O.; Karabulut, E. Protein carbonyl levels in serum and gingival crevicular fluid in patients with chronic periodontitis. Arch. Oral Biol. 2008, 53, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Raghavendra, N.M.; Prasad, M.V.R.; Kathariya, R.; Patel, S.P.; Sharma, A. Gingival crevicular fluid and serum visfatin concentration: Their relationship in periodontal health and disease. J. Periodontol. 2011, 82, 1314–1319. [Google Scholar] [CrossRef]
- Thorat, M.K.; Pradeep, A.R.; Garg, G. Correlation of levels of oncostatin M cytokine in crevicular fluid and serum in periodontal disease. Int. J. Oral Sci. 2010, 2, 198–207. [Google Scholar] [CrossRef]
- Kalburgi, V.; Sravya, L.; Warad, S.; Vijayalaxmi, K.; Sejal, P.; Hazeil, D. Role of systemic markers in periodontal diseases: A possible inflammatory burden and risk factor for cardiovascular diseases? Ann. Med. Health Sci. Res. 2014, 4, 388–392. [Google Scholar] [CrossRef]
- Shrivastava, A.K.; Singh, H.V.; Raizada, A.; Singh, S.K.; Pandey, A.; Singh, N.; Yadav, D.S.; Sharma, H. Inflammatory markers in patients with rheumatoid arthritis. Allergol. Immunopathol. 2015, 43, 81–87. [Google Scholar] [CrossRef]
- Brennan, F.M.; McInnes, I.B. Evidence that cytokines play a role in rheumatoid arthritis. J. Clin. Investig. 2008, 118, 3537–3545. [Google Scholar] [CrossRef]
- Nesse, W.; Westra, J.; van der Wal, J.E.; Abbas, F.; Nicholas, A.P.; Vissink, A.; Brouwer, E. The periodontium of periodontitis patients contains citrullinated proteins which may play a role in ACPA (anti-citrullinated protein antibody) formation. J. Clin. Periodontol. 2012, 39, 599–607. [Google Scholar] [CrossRef]
- de Smit, M.J.; Rahajoe, P.S.; Schuurmans, G.J.; Eelsing, E.; Kertia, N.; Vissink, A.; Westra, J. Rheumatoid arthritis (RA)-associated autoantibodies are present in the periodontal exudate of patients with and without RA. Ann. Rheum. Dis. 2018, 77, 1253–1254. [Google Scholar] [CrossRef]
- Terao, C.; Asai, K.; Hashimoto, M.; Yamazaki, T.; Ohmura, K.; Yamaguchi, A.; Takahashi, K.; Takei, N.; Ishii, T.; Kawaguchi, T.; et al. Significant association of periodontal disease with anti-citrullinated peptide antibody in a Japanese healthy population—The Nagahama study. J. Autoimmun. 2015, 59, 85–90. [Google Scholar] [CrossRef]
- de Pablo, P.; Chapple, I.L.C.; Buckley, C.D.; Dietrich, T. Periodontitis in systemic rheumatic diseases. Nat. Rev. Rheumatol. 2009, 5, 218–224. [Google Scholar] [CrossRef]
- Wegner, N.; Lundberg, K.; Kinloch, A.; Fisher, B.; Malmström, V.; Feldmann, M.; Venables, P.J. Autoimmunity to specific citrullinated proteins gives the first clues to the etiology of rheumatoid arthritis. Immunol. Rev. 2010, 233, 34–54. [Google Scholar] [CrossRef]
- Gonzalez, S.M.; Payne, J.B.; Yu, F.; Thiele, G.M.; Erickson, A.R.; Johnson, P.G.; Schmid, M.J.; Cannon, G.W.; Kerr, G.S.; Reimold, A.M.; et al. Alveolar bone loss is associated with circulating anti-citrullinated protein antibody (ACPA) in patients with rheumatoid arthritis. J. Periodontol. 2015, 86, 222–231. [Google Scholar] [CrossRef]
- Lappin, D.F.; Apatzidou, D.; Quirke, A.-M.; Oliver-Bell, J.; Butcher, J.P.; Kinane, D.F.; Riggio, M.P.; Venables, P.; McInnes, I.B.; Culshaw, S. Influence of periodontal disease, Porphyromonas gingivalis and cigarette smoking on systemic anti-citrullinated peptide antibody titres. J. Clin. Periodontol. 2013, 40, 907–915. [Google Scholar] [CrossRef] [PubMed]
- van der Helm-van Mil, A.H.M.; Verpoort, K.N.; Breedveld, F.C.; Toes, R.E.M.; Huizinga, T.W.J. Antibodies to citrullinated proteins and differences in clinical progression of rheumatoid arthritis. Arthritis Res. Ther. 2005, 7, R949. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Knevel, R.; Suwannalai, P.; van der Linden, M.P.; Janssen, G.M.C.; van Veelen, P.A.; Levarht, N.E.W.; van der Helm-van Mil, A.H.M.; Cerami, A.; Huizinga, T.W.J.; et al. Autoantibodies recognizing carbamylated proteins are present in sera of patients with rheumatoid arthritis and predict joint damage. Proc. Natl. Acad. Sci. USA 2011, 108, 17372–17377. [Google Scholar] [CrossRef]
- Juarez, M.; Bang, H.; Hammar, F.; Reimer, U.; Dyke, B.; Sahbudin, I.; Buckley, C.D.; Fisher, B.; Filer, A.; Raza, K. Identification of novel antiacetylated vimentin antibodies in patients with early inflammatory arthritis. Ann. Rheum. Dis. 2016, 75, 1099–1107. [Google Scholar] [CrossRef]
- Käber, U.R.; Michel, A.; Bolten, W.W.; Gleissner, C.; Dehne, F.; Willershausen-Zönnchen, B. Risk for periodontal disease in patients with longstanding rheumatoid arthritis. Arthritis Rheum. 1997, 40, 2248–2251. [Google Scholar] [CrossRef]
- de Pablo, P.; Dietrich, T.; McAlindon, T.E. Association of periodontal disease and tooth loss with rheumatoid arthritis in the US population. J. Rheumatol. 2008, 35, 70–76. [Google Scholar]
- Havemose-Poulsen, A.; Westergaard, J.; Stoltze, K.; Skjødt, H.; Danneskiold-Samsøe, B.; Locht, H.; Bendtzen, K.; Holmstrup, P. Periodontal and hematological characteristics associated with aggressive periodontitis, juvenile idiopathic arthritis, and rheumatoid arthritis. J. Periodontol. 2006, 77, 280–288. [Google Scholar] [CrossRef]
- Pischon, N.; Pischo, T.; Kröger, J.; Gülmez, E.; Kleber, B.-M.; Bernimoulin, J.-P.; Landau, H.; Brinkmann, P.-G.; Schlattmann, P.; Zernicke, J.; et al. Association among rheumatoid arthritis, oral hygiene, and periodontitis. J. Periodontol. 2008, 79, 979–986. [Google Scholar] [CrossRef]
- Mercado, F.; Marshall, R.I.; Klestov, A.C.; Bartold, P.M. Is there a relationship between rheumatoid arthritis and periodontal disease? J. Clin, Periodontol. 2000, 27, 267–272. [Google Scholar] [CrossRef]
- Mercado, F.B.; Marshall, R.I.; Klestov, A.C.; Bartold, P.M. Relationship between rheumatoid arthritis and periodontitis. J. Periodontol. 2001, 72, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Yokoyama, T.; Ishida, K.; Abe, A.; Yamamoto, K.; Yoshie, H. Serum cytokine and periodontal profiles in relation to disease activity of rheumatoid arthritis in Japanese adults. J. Periodontol. 2010, 81, 650–657. [Google Scholar] [CrossRef]
- Marotte, H.; Farge, P.; Gaudin, P.; Alexandre, C.; Mougin, B.; Miossec, P. The association between periodontal disease and joint destruction in rheumatoid arthritis extends the link between the HLA-DR shared epitope and severity of bone destruction. Ann. Rheum. Dis. 2006, 65, 905–909. [Google Scholar] [CrossRef]
- Dissick, A.; Redman, R.S.; Jones, M.; Rangan, B.V.; Reimold, A.; Griffiths, G.R.; Mikuls, T.R.; Amdur, R.L.; Richards, J.S.; Kerr, G.S. Association of periodontitis with rheumatoid arthritis: A pilot study. J. Periodontol. 2010, 81, 223–230. [Google Scholar] [CrossRef]
- Rodríguez-Lozano, B.; González-Febles, J.; Garnier-Rodríguez, J.L.; Dadlani, S.; Bustabad-Reyes, S.; Sanz, M.; Sánchez-Alonso, F.; Sánchez-Piedra, C.; González-Dávila, E.; Díaz-González, F. Association between severity of periodontitis and clinical activity in rheumatoid arthritis patients: A case-control study. Arthritis Res. Ther. 2019, 21, 27. [Google Scholar] [CrossRef]
- Rajkarnikar, J.; Thomas, B.S.; Rao, S.K. Inter-relationship between rheumatoid arthritis and periodontitis. Kathmandu Univ. Med. J. 2013, 11, 22–26. [Google Scholar] [CrossRef]
- Joseph, R.; Rajappan, S.; Nath, S.G.; Paul, B.J. Association between chronic periodontitis and rheumatoid arthritis: A hospital-based case-control study. Rheumatol. Int. 2013, 33, 103–109. [Google Scholar] [CrossRef]
- Ortiz, P.; Bissada, N.F.; Palomo, L.; Han, Y.W.; Al-Zahrani, M.S.; Panneerselvam, A.; Askari, A. Periodontal therapy reduces the severity of active rheumatoid arthritis in patients treated with or without tumor necrosis factor inhibitors. J. Periodontol. 2009, 80, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Erciyas, K.; Sezer, U.; Üstün, K.; Pehlivan, Y.; Kısacık, B.; Şenyurt, S.; Tarakçıoğlu, M.; Onat, A. Effects of periodontal therapy on disease activity and systemic inflammation in rheumatoid arthritis patients. Oral Dis. 2013, 19, 394–400. [Google Scholar] [CrossRef]
- Salemi, S.; Biondo, M.I.; Fiorentino, C.; Argento, G.; Paolantonio, M.; di Murro, C.; Malagnino, V.A.; Canzoni, M.; Diamanti, A.P.; D’Amelio, R. Could early rheumatoid arthritis resolve after periodontitis treatment only? Case report and review of the literature. Medicine 2014, 93, e195. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Liu, Z.; Shu, D.; Xiong, Y.; He, M.; Xu, S.; Si, S.; Guo, B. Association of periodontitis with rheumatoid arthritis and the effect of non-surgical periodontal treatment on disease activity in patients with rheumatoid arthritis. Med. Sci. Monit. 2018, 24, 5802–5810. [Google Scholar] [CrossRef]
- De Pinho, N.M.; Oliveira, R.D.R.; Novaes, A.B.; Voltarelli, J.C. Relationship between periodontitis and rheumatoid arthritis and the effect of non-surgical periodontal treatment. Braz. Dent. J. 2009, 20, 355–364. [Google Scholar] [CrossRef]
- Kaushal, S.; Singh, A.K.; Lal, N.; Das, S.K.; Mahdi, A.A. Effect of periodontal therapy on disease activity in patients of rheumatoid arthritis with chronic periodontitis. J. Oral Biol. Craniofac. Res. 2019, 9, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Cosgarea, R.; Tristiu, R.; Dumitru, R.B.; Arweiler, N.B.; Rednic, S.; Sirbu, C.I.; Lascu, L.; Sculean, A.; Eick, S. Effects of non-surgical periodontal therapy on periodontal laboratory and clinical data as well as on disease activity in patients with rheumatoid arthritis. Clin. Oral Investig. 2019, 23, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Scher, J.U.; Ubeda, C.; Equinda, M.; Khanin, R.; Buischi, Y.; Viale, A.; Lipuma, L.; Attur, M.; Pillinger, M.H.; Weissmann, G.; et al. Periodontal disease and the oral microbiota in new-onset rheumatoid arthritis. Arthritis Rheum. 2012, 64, 3083–3094. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, D.; Jia, H.; Feng, Q.; Wang, D.; Liang, D.; Wu, X.; Li, J.; Tang, L.; Li, Y.; et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 2015, 21, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Oliva, I.; Paropkari, A.D.; Saraswat, S.; Serban, S.; Yonel, Z.; Sharma, P.; de Pablo, P.; Raza, K.; Filer, A.; Chapple, I.; et al. Dysbiotic subgingival microbial communities in periodontally healthy patients with rheumatoid arthritis. Arthritis Rheumatol. 2018, 70, 1008–1013. [Google Scholar] [CrossRef]
- Corrêa, J.D.; Fernandes, G.R.; Calderaro, D.C.; Mendonça, S.M.S.; Silva, J.M.; Albiero, M.L.; Cunha, F.Q.; Xiao, E.; Ferreira, G.A.; Teixeira, A.L.; et al. Oral microbial dysbiosis linked to worsened periodontal condition in rheumatoid arthritis patients. Sci. Rep. 2019, 9, 8379. [Google Scholar] [CrossRef]
- Ogrendik, M.; Kokino, S.; Ozdemir, F.; Bird, P.S.; Hamlet, S. Serum antibodies to oral anaerobic bacteria in patients with rheumatoid arthritis. MedGenMed 2005, 7, 2. [Google Scholar]
- Okada, M.; Kobayashi, T.; Ito, S.; Yokoyama, T.; Komatsu, Y.; Abe, A.; Murasawa, A.; Yoshie, H. Antibody responses to periodontopathic bacteria in relation to rheumatoid arthritis in Japanese adults. J. Periodontol. 2011, 82, 1433–1441. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Yoshida, S.; Takeuchi, T.; Kimura, M.; Yoshikawa, A.; Hiramatsu, Y.; Ishida, T.; Makino, S.; Takasugi, Y.; Hanafusa, T. Periodontal pathogens participate in synovitis in patients with rheumatoid arthritis in clinical remission: A retrospective case–control study. Rheumatology 2015, 54, 2257–2263. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moen, K.; Brun, J.G.; Valen, M.; Skartveit, L.; Ribs Eribe, E.K.; Olsen, I.; Jonsson, R. Synovial inflammation in active rheumatoid arthritis and psoriatic arthritis facilitates trapping of a variety of oral bacterial DNAs. Clin. Exp. Rheumatol. 2006, 24, 656–663. [Google Scholar]
- Ziebolz, D.; Pabel, S.O.; Lange, K.; Krohn-Grimberghe, B.; Hornecker, E.; Mausberg, R.F. Clinical periodontal and microbiologic parameters in patients with rheumatoid arthritis. J. Periodontol. 2011, 82, 1424–1432. [Google Scholar] [CrossRef]
- Reichert, S.; Haffner, M.; Keyßer, G.; Schäfer, C.; Stein, J.M.; Schaller, H.-G.; Wienke, A.; Strauss, H.; Heide, S.; Schulz, S. Detection of oral bacterial DNA in synovial fluid. J. Clin. Periodontol. 2013, 40, 591–598. [Google Scholar] [CrossRef]
- Ebbers, M.; Lübcke, P.M.; Volzke, J.; Kriebel, K.; Hieke, C.; Engelmann, R.; Lang, H.; Kreikemeyer, B.; Müller-Hilke, B. Interplay between P. gingivalis, F. nucleatum and A. actinomycetemcomitans in murine alveolar bone loss, arthritis onset and progression. Sci. Rep. 2018, 8, 15129. [Google Scholar] [CrossRef]
- Nienhuis, R.L.; Mandema, E. A new serum factor in patients with rheumatoid arthritis; the antiperinuclear factor. Ann. Rheum. Dis. 1964, 23, 302–305. [Google Scholar] [CrossRef]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef]
- Nielen, M.M.J.; van Schaardenburg, D.; Reesink, H.W.; van de Stadt, R.J.; van der Horst-Bruinsma, I.E.; de Koning, M.H.M.T.; Habibuw, M.R.; Vandenbroucke, J.P.; Dijkmans, B.A.C. Specific autoantibodies precede the symptoms of rheumatoid arthritis: A study of serial measurements in blood donors. Arthritis Rheum. 2004, 50, 380–386. [Google Scholar] [CrossRef]
- Van de Stadt, L.A.; de Koning, M.H.M.T.; van de Stadt, R.J.; Wolbink, G.; Dijkmans, B.A.C.; Hamann, D.; van Schaardenburg, D. Development of the anti-citrullinated protein antibody repertoire prior to the onset of rheumatoid arthritis. Arthritis Rheum. 2011, 63, 3226–3233. [Google Scholar] [CrossRef]
- Van der Woude, D.; Syversen, S.W.; van der Voort, E.I.H.; Verpoort, K.N.; Goll, G.L.; van der Linden, M.P.M.; van Mil, A.H.M.; van der Heijde, D.M.F.M.; Huizinga, T.W.J.; Kvien, T.K.; et al. The ACPA isotype profile reflects long-term radiographic progression in rheumatoid arthritis. Ann. Rheum. Dis. 2010, 69, 1110–1116. [Google Scholar] [CrossRef] [PubMed]
- Kleyer, A.; Finzel, S.; Rech, J.; Manger, B.; Krieter, M.; Faustini, F.; Araujo, E.; Hueber, A.J.; Harre, U.; Engelke, K.; et al. Bone loss before the clinical onset of rheumatoid arthritis in subjects with anticitrullinated protein antibodies. Ann. Rheum. Dis. 2014, 73, 854–860. [Google Scholar] [CrossRef]
- Loutan, L.; Alpizar-Rodriguez, D.; Courvoisier, D.S.; Finckh, A.; Mombelli, A.; Giannopoulou, C. Periodontal status correlates with anti-citrullinated protein antibodies in first-degree relatives of individuals with rheumatoid arthritis. J. Clin. Periodontol. 2019, 46, 690–698. [Google Scholar] [CrossRef] [PubMed]
- González-Febles, J.; Rodríguez-Lozano, B.; Sánchez-Piedra, C.; Garnier-Rodríguez, J.; Bustabad, S.; Hernández-González, M.; González-Dávila, E.; Sanz, M.; Diáz-González, F. Association between periodontitis and anti-citrullinated protein antibodies in rheumatoid arthritis patients: A cross-sectional study. Arthritis Res. Ther. 2020, 22, 27. [Google Scholar] [CrossRef]
- Kinloch, A.; Lundberg, K.; Wait, R.; Wegner, N.; Lim, N.H.; Zendman, A.J.W.; Saxne, T.; Malmstr, V.; Venables, P.J. Synovial fluid is a site of citrullination of autoantigens in inflammatory arthritis. Arthritis Rheum. 2008, 58, 2287–2295. [Google Scholar] [CrossRef] [PubMed]
- Laugisch, O.; Wong, A.; Sroka, A.; Kantyka, T.; Koziel, J.; Neuhaus, K.; Sculean, A.; Venables, P.J.; Potempa, J.; Möller, B.; et al. Citrullination in the periodontium—A possible link between periodontitis and rheumatoid arthritis. Clin. Oral Investig. 2016, 20, 675–683. [Google Scholar] [CrossRef]
- Suzuki, A.; Yamada, R.; Chang, X.; Tokuhiro, S.; Sawada, T.; Suzuki, M.; Nagasaki, M.; Nakayama-Hamada, M.; Kawaida, R.; Ono, M.; et al. Functional haplotypes of PADI4, encoding citrullinating enzyme peptidylarginine deiminase 4, are associated with rheumatoid arthritis. Nat. Genet. 2003, 34, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, A.; Nakano, Y.; Yamashita, Y.; Oho, T.; Ito, H.; Kondo, M.; Ohishi, M.; Koga, T. Immunodominant region of Actinobacillus actinomycetemcomitans 40-kilodalton heat shock protein in patients with rheumatoid arthritis. J. Dent. Res. 2001, 80, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Romero, V.; Fert-Bober, J.; Nigrovic, P.A.; Darrah, E.; Haque, U.J.; Lee, D.M.; van Eyk, J.; Rosen, A.; Andrade, F. Immune-mediated pore-forming pathways induce cellular hypercitrullination and generate citrullinated autoantigens in rheumatoid arthritis. Sci. Transl. Med. 2013, 5, 209ra150. [Google Scholar] [CrossRef]
- Konig, M.F.; Andrade, F. A critical reappraisal of neutrophil extracellular traps and NETosis mimics based on differential requirements for protein citrullination. Front. Immunol. 2016, 7, 461. [Google Scholar] [CrossRef] [PubMed]
- Volkov, M.; Dekkers, J.; Loos, B.G.; Bizzarro, S.; Huizinga, T.W.J.; Praetorius, H.A.; Toes, R.E.M.; van der Woude, D. Comment on “Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis”. Sci. Transl. Med. 2018, 10, 8349. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Bañuelos, E.; Johansson, L.; Konig, M.; Lundquist, A.; Paz, M.; Buhlin, K.; Johansson, A.; Rantapää-Dahlqvist, S.; Andrade, F. Exposure to Aggregatibacter actinomycetemcomitans before Symptom Onset and the Risk of Evolving to Rheumatoid Arthritis. J. Clin. Med. 2020, 9, 1906. [Google Scholar] [CrossRef]
- Konig, M.F.; Giles, J.T.; Teles, R.P.; Moutsopoulos, N.M.; Andrade, F. Response to comment on “Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis”. Sci. Transl. Med. 2018, 10, eaao3031. [Google Scholar] [CrossRef]
- Kurowska, W.; Kuca-Warnawin, E.H.; Radzikowska, A.; Maslinski, W. The role of anti-citrullinated protein antibodies (ACPA) in the pathogenesis of rheumatoid arthritis. Cent. Eur. J. Immunol. 2017, 42, 390–398. [Google Scholar] [CrossRef]
- Alexiou, I.; Germenis, A.; Ziogas, A.; Theodoridou, K.; Sakkas, L.I. Diagnostic value of anti-cyclic citrullinated peptide antibodies in Greek patients with rheumatoid arthritis. BMC Musculoskelet. Disord. 2007, 8, 37. [Google Scholar] [CrossRef]
- Bizzaro, N. Antibodies to citrullinated peptides: A significant step forward in the early diagnosis of rheumatoid arthritis. Clin. Chem. Lab. Med. 2007, 45, 150–157. [Google Scholar] [CrossRef] [PubMed]
- van den Broek, M.; Dirven, L.; Klarenbeek, N.B.; Molenaar, T.H.; Han, K.H.; Kerstens, P.J.; Huizinga, T.W.; Dijkmans, B.A.; Allaart, C.F. The association of treatment response and joint damage with ACPA-status in recent-onset RA: A subanalysis of the 8-year follow-up of the BeSt study. Ann. Rheum. Dis. 2012, 71, 245–248. [Google Scholar] [CrossRef]
- Zhang, L.; Yuan, Y.; Xu, Q.; Jiang, Z.; Chu, C.Q. Contribution of neutrophils in the pathogenesis of rheumatoid arthritis. J. Biomed. Res. 2020, 34, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Eyre, S.; Bowes, J.; Diogo, D.; Lee, A.; Barton, A.; Martin, P.; Zhernakova, A.; Stahl, E.; Viatte, S.; McAllister, K.; et al. High-density genetic mapping identifies new susceptibility loci for rheumatoid arthritis. Nat. Genet. 2012, 44, 1336–1340. [Google Scholar] [CrossRef] [PubMed]
| No | Features | Periodontitis | Rheumatoid Arthritis |
|---|---|---|---|
| 1 | Prevalence | Affects about 20–50% of the global population [55], while its severe form affects about 10–15% of the global population [18] | Affects about 0.1–2.0 of the global population [56] |
| 2 | Modifiable/Environmental risk factors | Cigarette smoking; alcohol consumption; diabetes; obesity; stress; microorganisms | Regular smoking; diabetes; obesity; dietary; microbes at mucosal surfaces such as oral cavity, lung and gut [57] |
| 3 | Bone and tissue destruction | Alveolar bone resorption and bone loss in periodontitis patients [58] | Bone and cartilage break down [59] |
| 4 | Evidences supporting bacteria aetiology | High prevalence of anaerobic bacteria was found in the gingival tissues of patients with periodontitis [60]. Antibiotic treatment frequently results in improved clinical outcome [61,62,63] | Bacterial (including periodontopathogens) DNA and peptidoglycans are frequently detected in the synovial fluid and serum of RA patients [64,65,66]. Antibiotic treatment has been shown to effectively reduce RA disease activity [65,67] |
| 5 | Inflammatory markers | C-reactive protein (CRP), IL-1b, IL-6, TNF-a, visfatin, VEGF, oncostatin M, protein carbonyl, RANKL, IL-17, IL-36γ, MMPs and PGE2 [68,69,70,71,72] | CRP, IL-1b, IL-6, TNF-a, IL-23, IL-17A, IL-18, IFN-γ, PGE2, MMPs, RANKL and granulocyte macrophage colony-stimulating factor [73,74] |
| 6 | Autoantibodies | RA-associated autoantibodies eg. rheumatoid factors (RF) and ACPAs were present in the gingival crevicular fluid of non-RA periodontitis patients [75,76]. The positivity and titre of ACPAs are significantly associated with periodontitis [77,78,79] and alveolar bone loss [80]. Non-surgical periodontal treatment appears to reduce ACPA levels in periodontitis patients [81] | RF and ACPAs are prominently detected in the serum of RA patients. They are also associated with increased radiographic progression and joint damage [82]. Recently, anti-carbamylated protein antibodies (anti-CarP) and anti-acetylated protein antibodies were discovered in the serum of RA patients [83,84] |
| 7 | Citrullinated autoantigens | Histone H1x, adenylyl cyclase-associated protein 1, actin (cytoplasmic ½), apolipoprotein A-1 preproprotein, elongation factor 1-alpha, heterogeneous nuclear ribonucleoprotein A2/B1, histone H2A, vimentin, myeloid cell nuclear differentiation antigen, histone H2B were found in the gingival crevicular fluid of periodontitis patients [22] | Histone H1x, actin (cytoplasmic ½), apolipoprotein A-1 preproprotein, elongation factor 1-alpha, heterogeneous nuclear ribonucleoprotein A2/B1, histone H2A, vimentin, myeloid cell nuclear differentiation antigen, histone H2B were found in the synovial fluid of RA patients [22] |
| Socransky’s Classification a | Example | Remark |
|---|---|---|
| Yellow | Streptococci sanguinis and Streptococci oralis | Early colonisers essential for colonization of other bacteria associated with periodontal disease |
| Green | Capnocytophaga spp., Campylobacter concisus, Eikenella corrodens, and Actinobacillus actinomycetemcomitans | |
| Violet | Actinomyces odontolyticus and Veillonella parvula | |
| Orange | Prevotella intermedia, Prevotella nigrescens, Micromonas micros, Fusobacterium periodonticum, Eubacterium nodatum and Streptococcus constellatus | Bridging colonizers that aggregate with early and later colonizing bacteria |
| Red | Porphyromonas gingivalis, Treponema denticola and Tannerella forsythia | Late-colonizing bacteria, which are strongly associated with periodontitis and co-exist with the orange complex. |
| Ungrouped | Aggregatibacter actinomycetemcomitans, Selenomonas | More research is needed to group these bacteria properly |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Looh, S.C.; Soo, Z.M.P.; Wong, J.J.; Yam, H.C.; Chow, S.K.; Hwang, J.S. Aggregatibacter actinomycetemcomitans as the Aetiological Cause of Rheumatoid Arthritis: What Are the Unsolved Puzzles? Toxins 2022, 14, 50. https://doi.org/10.3390/toxins14010050
Looh SC, Soo ZMP, Wong JJ, Yam HC, Chow SK, Hwang JS. Aggregatibacter actinomycetemcomitans as the Aetiological Cause of Rheumatoid Arthritis: What Are the Unsolved Puzzles? Toxins. 2022; 14(1):50. https://doi.org/10.3390/toxins14010050
Chicago/Turabian StyleLooh, Sung Cheng, Zoey May Pheng Soo, Jia Jia Wong, Hok Chai Yam, Sook Khuan Chow, and Jung Shan Hwang. 2022. "Aggregatibacter actinomycetemcomitans as the Aetiological Cause of Rheumatoid Arthritis: What Are the Unsolved Puzzles?" Toxins 14, no. 1: 50. https://doi.org/10.3390/toxins14010050
APA StyleLooh, S. C., Soo, Z. M. P., Wong, J. J., Yam, H. C., Chow, S. K., & Hwang, J. S. (2022). Aggregatibacter actinomycetemcomitans as the Aetiological Cause of Rheumatoid Arthritis: What Are the Unsolved Puzzles? Toxins, 14(1), 50. https://doi.org/10.3390/toxins14010050





