Evidence for Resistance to Coagulotoxic Effects of Australian Elapid Snake Venoms by Sympatric Prey (Blue Tongue Skinks) but Not by Predators (Monitor Lizards)
Abstract
:1. Introduction
2. Results
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Venoms and Reagents
5.2. Plasma Collection
5.3. Thromboelastography Assays
5.4. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Harris, R.J.; Zdenek, C.N.; Harrich, D.; Frank, N.; Fry, B.G. An appetite for destruction: Detecting prey-selective binding of α-neurotoxins in the venom of Afro-Asian Elapids. Toxins 2020, 12, 205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lyons, K.; Dugon, M.M.; Healy, K. Diet breadth dediates the prey specificity of venom potency in snakes. Toxins 2020, 12, 74. [Google Scholar] [CrossRef] [Green Version]
- Rochelle, M.J.; Kardong, K.V. Constriction versus envenomation in prey capture by the brown tree snake, Boiga irregularis (Squamata: Colubridae). Herpetologica 1993, 49, 301–304. [Google Scholar]
- Heyborne, W.H.; Mackessy, S.P. Identification and characterization of a taxon-specific three-finger toxin from the venom of the Green Vinesnake (Oxybelis fulgidus family Colubridae). Biochimie 2013, 95, 1923–1932. [Google Scholar] [CrossRef]
- Pawlak, J.; Mackessy, S.P.; Fry, B.G.; Bhatia, M.; Mourier, G.; Fruchart-Gaillard, C.; Servent, D.; Ménez, R.; Stura, E.; Ménez, A.; et al. Denmotoxin, a three-finger toxin from the colubrid snake Boiga dendrophila (Mangrove Catsnake) with bird-specific activity. J. Biol. Chem. 2006, 281, 29030. [Google Scholar] [CrossRef] [Green Version]
- Pawlak, J.; Mackessy, S.P.; Sixberry, N.M.; Stura, E.A.; Le Du, M.H.; Ménez, R.; Foo, C.S.; Ménez, A.; Nirthanan, S.; Kini, R.M. Irditoxin, a novel covalently linked heterodimeric three-finger toxin with high taxon-specific neurotoxicity. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2009, 23, 534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Youngman, N.J.; Zdenek, C.N.; Dobson, J.S.; Bittenbinder, M.A.; Gillett, A.; Hamilton, B.; Dunstan, N.; Allen, L.; Veary, A.; Veary, E.; et al. Mud in the blood: Novel potent anticoagulant coagulotoxicity in the venoms of the Australian elapid snake genus Denisonia (mud adders) and relative antivenom efficacy. Toxicol. Lett. 2019, 302, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Asher, O.; Lupu-Meiri, M.; Jensen, B.S.; Paperna, T.; Fuchs, S.; Oron, Y. Functional characterization of mongoose nicotinic acetylcholine receptor α-subunit: Resistance to α-bungarotoxin and high sensitivity to acetylcholine. FEBS Lett. 1998, 431, 411–414. [Google Scholar] [CrossRef] [Green Version]
- Harris, R.J.; Fry, B.G. Electrostatic resistance to alpha-neurotoxins conferred by charge reversal mutations in nicotinic acetylcholine receptors. Proc. R. Soc. B 2021, 288, 20202703. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Dashevsky, D.; Kerkkamp, H.; Kordiš, D.; de Bakker, M.A.; Wouters, R.; van Thiel, J.; Vonk, F.; Kini, R.M.; Nazir, J. Widespread evolution of molecular resistance to snake venom α-neurotoxins in vertebrates. Toxins 2020, 12, 638. [Google Scholar] [CrossRef] [PubMed]
- Takacs, Z.; Wilhelmsen, K.C.; Sorota, S. Snake α-neurotoxin binding site on the Egyptian cobra (Naja haje) nicotinic acetylcholine receptor is conserved. Mol. Biol. Evol. 2001, 18, 1800–1809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drabeck, D.H.; Rucavado, A.; Hingst-Zaher, E.; Cruz, Y.P.; Dean, A.M.; Jansa, S.A. Resistance of South American opossums to vWF-binding venom C-type lectins. Toxicon 2020, 178, 92–99. [Google Scholar] [CrossRef]
- Nahas, L.; Kamiguti, A.S.; Sousa E Silva, M.C.C.; Ribeiro de Barros, M.A.A.; Morena, P. The inactivating effect of Bothrops jararaca and Waglerophis merremii snake plasma on the coagulant activity of various snake venoms. Toxicon 1983, 21, 239–246. [Google Scholar] [CrossRef]
- Vieira, C.O.; Tanaka, A.S.; Sano-Martins, I.S.; Morais, K.B.; Santoro, M.L.; Tanaka-Azevedo, A.M. Bothrops jararaca fibrinogen and its resistance to hydrolysis evoked by snake venoms. Comp. Biochem. Physiol. Part B 2008, 151, 428–432. [Google Scholar] [CrossRef]
- Gibbs, H.L.; Sanz, L.; Pérez, A.; Ochoa, A.; Hassinger, A.T.; Holding, M.L.; Calvete, J.J. The molecular basis of venom resistance in a rattlesnake-squirrel predator-prey system. Mol. Ecol. 2020, 29, 2871–2888. [Google Scholar] [CrossRef]
- Cogger, H.G. Reptiles and Amphibians of Australia; CSIRO Publishing: Clayton, Australia, 2018. [Google Scholar]
- Shine, R. Habitats, diets, and sympatry in snakes: A study from Australia. Can. J. Zool. 1977, 55, 1118–1128. [Google Scholar] [CrossRef]
- Shine, R. Ecology of eastern Australian whipsnakes of the genus Demansia. J. Herpetol. 1980, 14, 381–389. [Google Scholar] [CrossRef] [Green Version]
- Shine, R. Comparative ecology of three Australian snake species of the genus Cacophis (Serpentes: Elapidae). Copeia. 1980, 1980, 831–838. [Google Scholar] [CrossRef]
- Shine, R. Ecology of Australian elapid snakes of the genera Furina and Glyphodon. J. Herpetol. 1981, 15, 219–224. [Google Scholar] [CrossRef] [Green Version]
- Zdenek, C.N.; Den Brouw, B.O.; Dashevsky, D.; Gloria, A.; Youngman, N.J.; Watson, E.; Green, P.; Hay, C.; Dunstan, N.; Allen, L.; et al. Clinical implications of convergent procoagulant toxicity and differential antivenom efficacy in Australian elapid snake venoms. Toxicol. Lett. 2019, 316, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Zdenek, C.N.; Hay, C.; Arbuckle, K.; Jackson, T.N.W.; Bos, M.H.A.; Op Den Brouw, B.; Debono, J.; Allen, L.; Dunstan, N.; Morley, T.; et al. Coagulotoxic effects by brown snake (Pseudonaja) and taipan (Oxyuranus) venoms, and the efficacy of a new antivenom. Toxicol. Vitr. 2019, 58, 97–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lister, C.; Arbuckle, K.; Jackson, T.N.W.; Debono, J.; Zdenek, C.N.; Dashevsky, D.; Dunstan, N.; Allen, L.; Hay, C.; Bush, B.; et al. Catch a tiger snake by its tail: Differential toxicity, co-factor dependence and antivenom efficacy in a procoagulant clade of Australian venomous snakes. Comp. Biochem. Physiol. Part C 2017, 202, 39–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isbister, G.K. Procoagulant snake toxins: Laboratory studies, diagnosis, and understanding snakebite coagulopathy. Semin. Thromb. Hemost. 2009, 35, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.; O’leary, M.A.; Isbister, G.K. Coagulant effects of black snake (Pseudechis spp.) venoms and in vitro efficacy of commercial antivenom. Toxicon 2011, 58, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Maduwage, K.P.; Scorgie, F.E.; Lincz, L.F.; O’Leary, M.A.; Isbister, G.K. Procoagulant snake venoms have differential effects in animal plasmas: Implications for antivenom testing in animal models. Thromb. Res. 2016, 137, 174–177. [Google Scholar] [CrossRef] [PubMed]
- Minton, S.A., Jr.; Minton, M.R. Toxicity of some Australian snake venoms for potential prey species of reptiles and amphibians. Toxicon 1981, 19, 749–755. [Google Scholar] [CrossRef]
- Joseph, J.S.; Chung, M.C.; Mirtschin, P.J.; Kini, R.M. Effect of snake venom procoagulants on snake plasma: Implications for the coagulation cascade of snakes. Toxicon 2002, 40, 175–183. [Google Scholar] [CrossRef]
- Jones, L.; Harris, R.J.; Fry, B.G. Not goanna get me: Mutations in the savannah monitor lizard (Varanus exanthematicus) nicotinic acetylcholine receptor confer reduced susceptibility to sympatric cobra venoms. Neurotox. Res. 2021, 39, 1116–1122. [Google Scholar] [CrossRef]
- Norval, G.; Clayton, J.; Sharrad, R.D.; Gardner, M.G. Notes on the stomach contents of a juvenile Sleepy Lizard, Tiliqua rugosa (Gray 1825), killed by an Eastern Brown Snake, Pseudonaja textilis (Duméril, Bibron, and Duméril 1854) in South Australia. Reptiles Amphib. 2018, 25, 200–203. [Google Scholar] [CrossRef]
- Shine, R. Constraints, allometry, and adaptation: Food habits and reproductive biology of Australian brownsnakes (Pseudonaja: Elapidae). Herpetologica 1989, 45, 195–207. [Google Scholar]
- Mayes, P.; Thompson, G.; Withers, P. Diet and foraging behaviour of the semi-aquatic Varanus mertensi (Reptilia: Varanidae). Wildl. Res. 2005, 32, 67–74. [Google Scholar] [CrossRef]
- Shine, R. Food habits, habitats and reproductive biology of four sympatric species of varanid lizards in tropical Australia. Herpetologica 1986, 42, 346–360. [Google Scholar]
- Vidal, N.; Hedges, S.B. The molecular evolutionary tree of lizards, snakes, and amphisbaenians. Comptes rendus biologies. 2009, 332, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Brennan, I.G.; Lemmon, A.R.; Lemmon, E.M.; Portik, D.M.; Weijola, V.; Welton, L.; Donnellan, S.C.; Keogh, J.S. Phylogenomics of monitor lizards and the role of competition in dictating body size disparity. Syst. Biol. 2021, 70, 120–132. [Google Scholar] [CrossRef]
- Grashof, D.; Zdenek, C.N.; Dobson, J.S.; Youngman, N.J.; Coimbra, F.; Benard-Valle, M.; Alagon, A.; Fry, B.G. A web of coagulotoxicity: Failure of antivenom to neutralize the destructive (non-clotting) fibrinogenolytic activity of Loxosceles and Sicarius spider venoms. Toxins 2020, 12, 91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Youngman, N.J.; Chowdhury, A.; Zdenek, C.N.; Coster, K.; Sundman, E.; Braun, R.; Fry, B.G. Utilising venom activity to infer dietary composition of the Kenyan horned viper (Bitis worthingtoni). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021, 240, 108921. [Google Scholar] [CrossRef]
- Youngman, N.J.; Debono, J.; Dobson, J.S.; Zdenek, C.N.; Harris, R.J.; Brouw, B.O.D.; Coimbra, F.C.P.; Naude, A.; Coster, K.; Sundman, E.; et al. Venomous landmines: Clinical implications of extreme coagulotoxic diversification and differential neutralization by antivenom of venoms within the Viperid snake genus Bitis. Toxins 2019, 11, 422. [Google Scholar] [CrossRef] [Green Version]
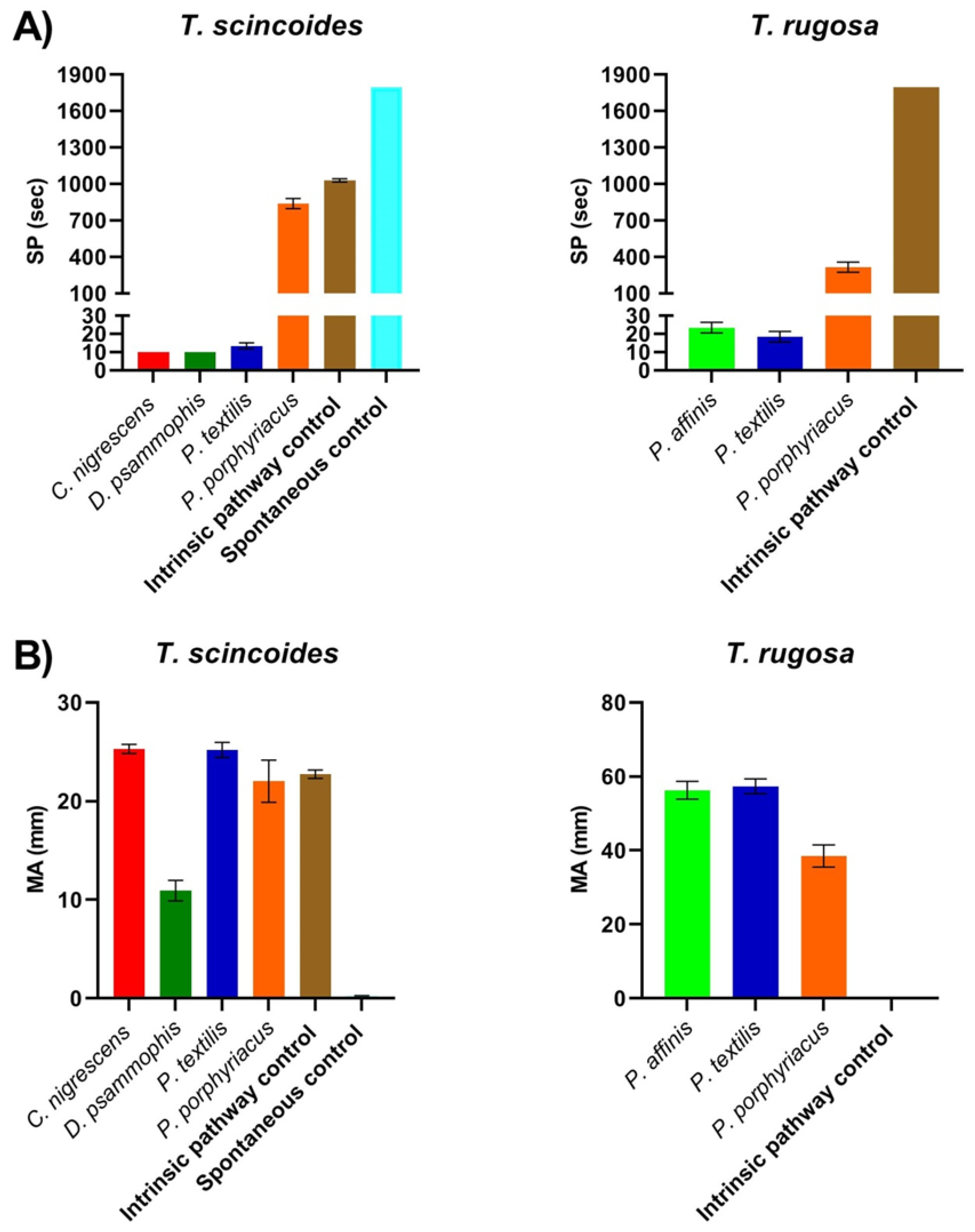
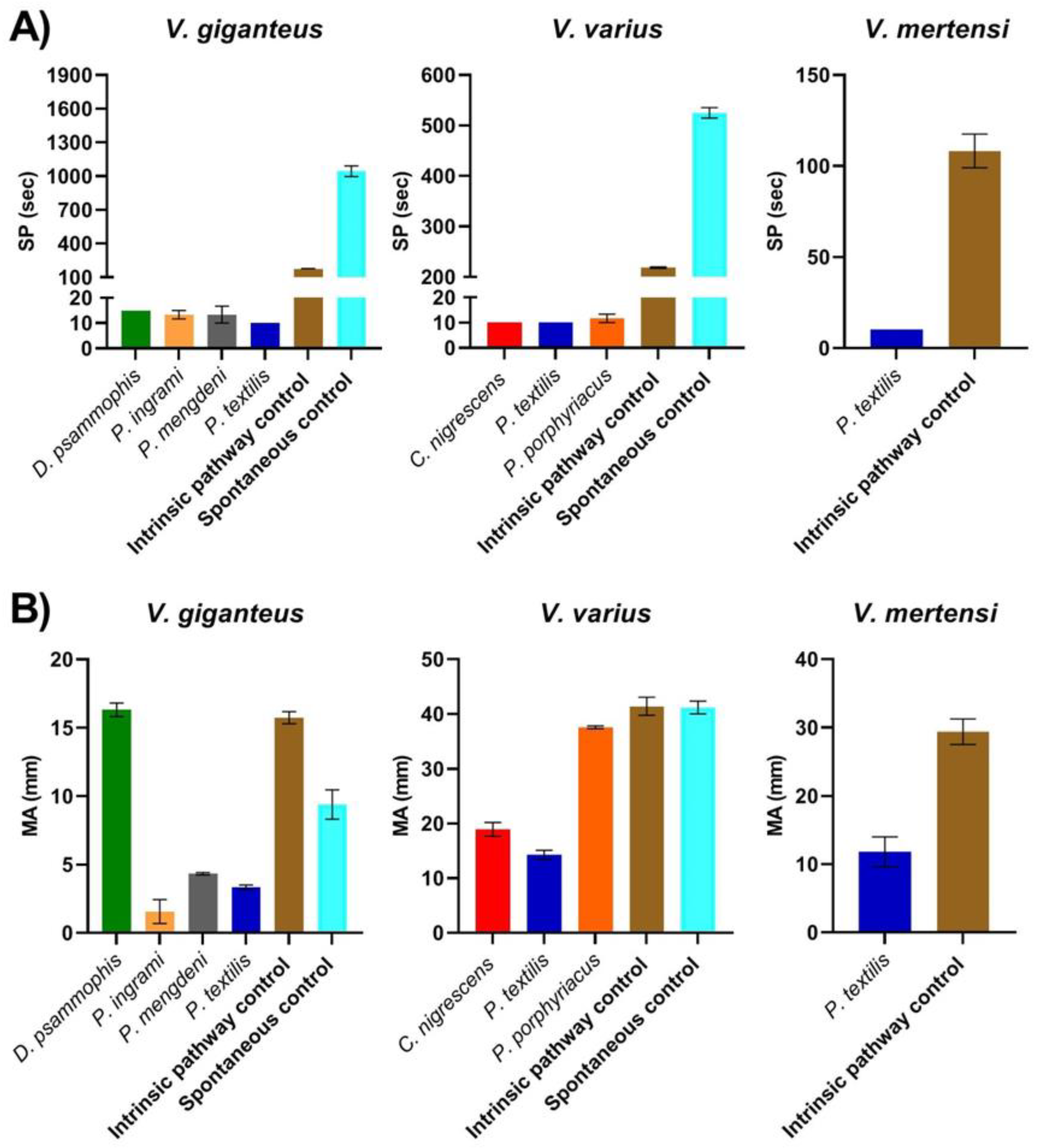
| Coagulation via Intrinsic Pathway Activation | Coagulation via Spontaneous Clotting | |||
|---|---|---|---|---|
| Species | SP (s) | MA (mm) | SP (s) | MA (mm) |
| Tiliqua scincoides | 1028.33 ± 13.02 | 22.73 ± 0.42 | * | * |
| Tiliqua rugosa | * | * | NA | NA |
| Varanus giganteus | 171.67 ± 6.67 | 15.73 ± 0.44 | 1045 ± 48.05 | 9.4 ± 1.07 |
| Varanus mertensi | 108.33 ± 9.28 | 29.4 ± 1.86 | NA | NA |
| Varanus varius | 218.33 ± 1.67 | 41.4 ± 1.65 | 525 ± 10.41 | 41.2 ± 1.16 |
| Rhinella marina | 65 ± 2.89 | 7.4 ± 0.12 | * | * |
| Gallus gallus | 166.67 ± 3.33 | 44.2 ± 1.36 | 188.33 ± 4.41 | 39.03 ± 0.45 |
| Rattus norvegicus | 48.33 ± 3.33 | 19.03 ± 1.3 | 135 ± 5 | 22.57 ± 2.55 |
| Homo sapiens | 173.33 ± 1.67 | 17.67 ± 0.35 | 646.67 ± 24.55 | 20.2 ± 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Youngman, N.J.; Llinas, J.; Fry, B.G. Evidence for Resistance to Coagulotoxic Effects of Australian Elapid Snake Venoms by Sympatric Prey (Blue Tongue Skinks) but Not by Predators (Monitor Lizards). Toxins 2021, 13, 590. https://doi.org/10.3390/toxins13090590
Youngman NJ, Llinas J, Fry BG. Evidence for Resistance to Coagulotoxic Effects of Australian Elapid Snake Venoms by Sympatric Prey (Blue Tongue Skinks) but Not by Predators (Monitor Lizards). Toxins. 2021; 13(9):590. https://doi.org/10.3390/toxins13090590
Chicago/Turabian StyleYoungman, Nicholas J., Joshua Llinas, and Bryan G. Fry. 2021. "Evidence for Resistance to Coagulotoxic Effects of Australian Elapid Snake Venoms by Sympatric Prey (Blue Tongue Skinks) but Not by Predators (Monitor Lizards)" Toxins 13, no. 9: 590. https://doi.org/10.3390/toxins13090590
APA StyleYoungman, N. J., Llinas, J., & Fry, B. G. (2021). Evidence for Resistance to Coagulotoxic Effects of Australian Elapid Snake Venoms by Sympatric Prey (Blue Tongue Skinks) but Not by Predators (Monitor Lizards). Toxins, 13(9), 590. https://doi.org/10.3390/toxins13090590






