Combined Effects of Temperature and Toxic Algal Abundance on Paralytic Shellfish Toxic Accumulation, Tissue Distribution and Elimination Dynamics in Mussels Mytilus coruscus
Abstract
1. Introduction
2. Results
2.1. Toxin of A. catenella
2.2. Effect of Temperature Increase and Toxic Algal Abundance on Mussels
2.3. Toxin Compartmentalisation
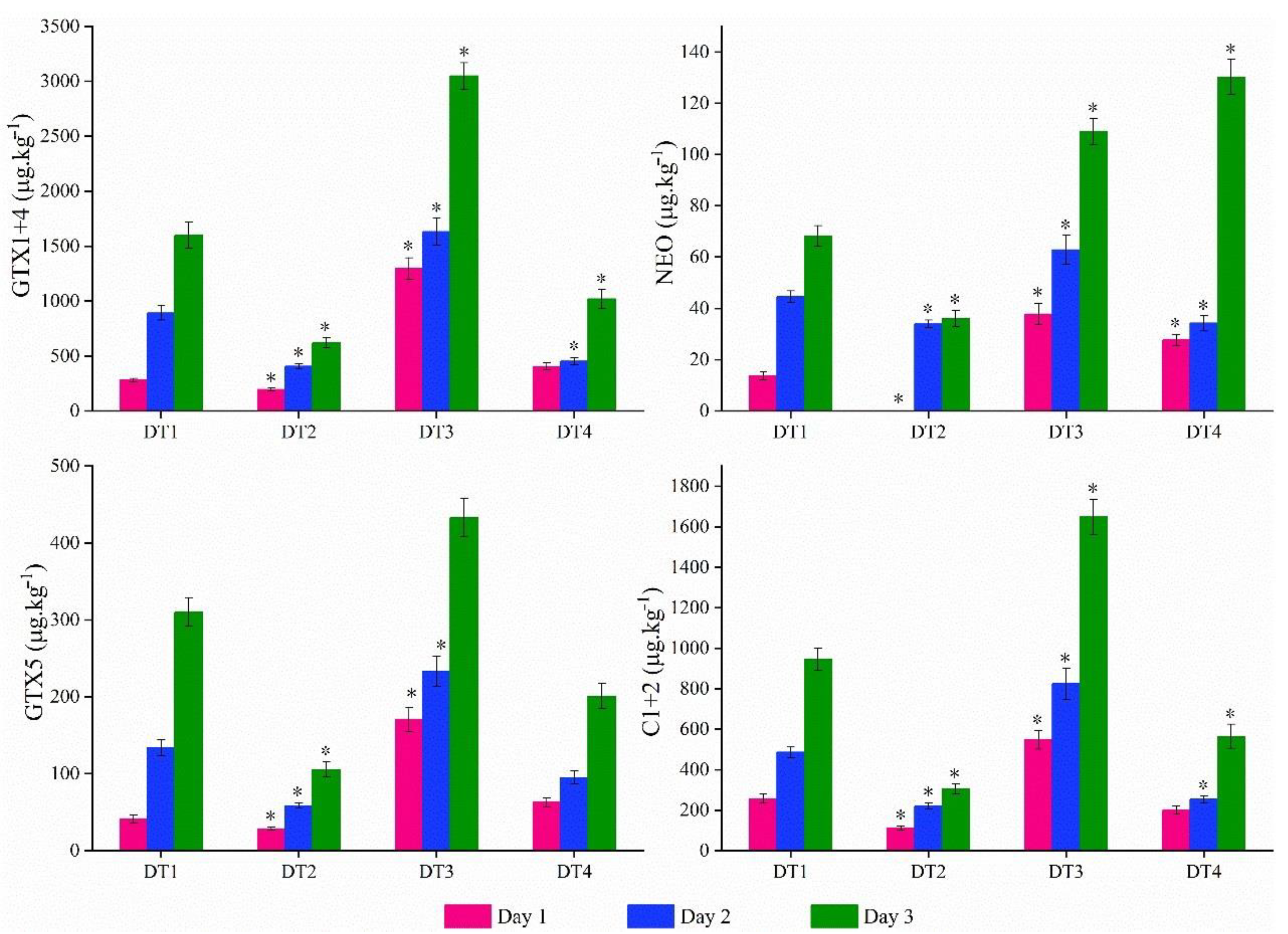
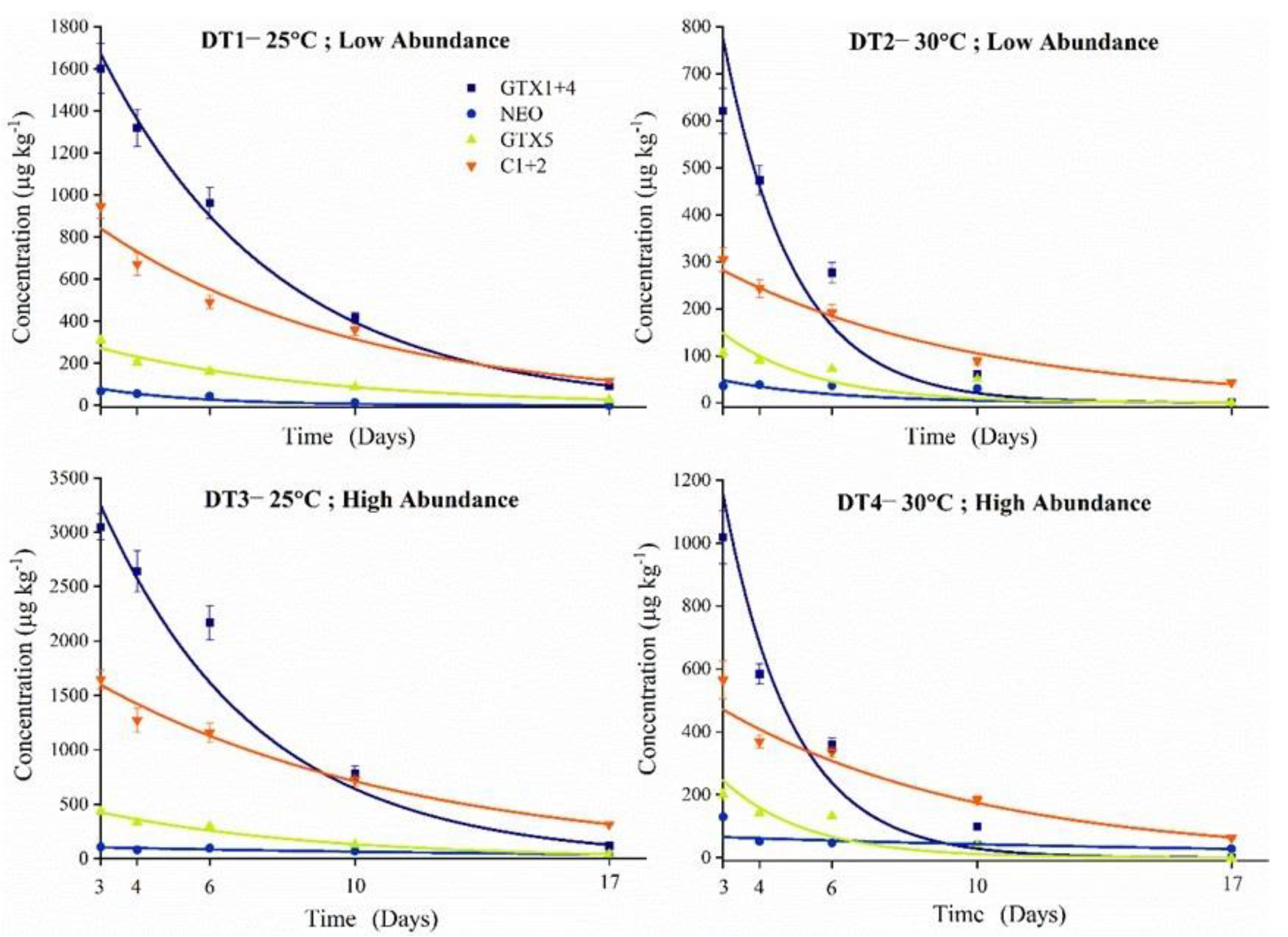
2.4. PSTs Accumulation and Elimination under High Temperature and Abundance Conditions
3. Discussion
4. Conclusions
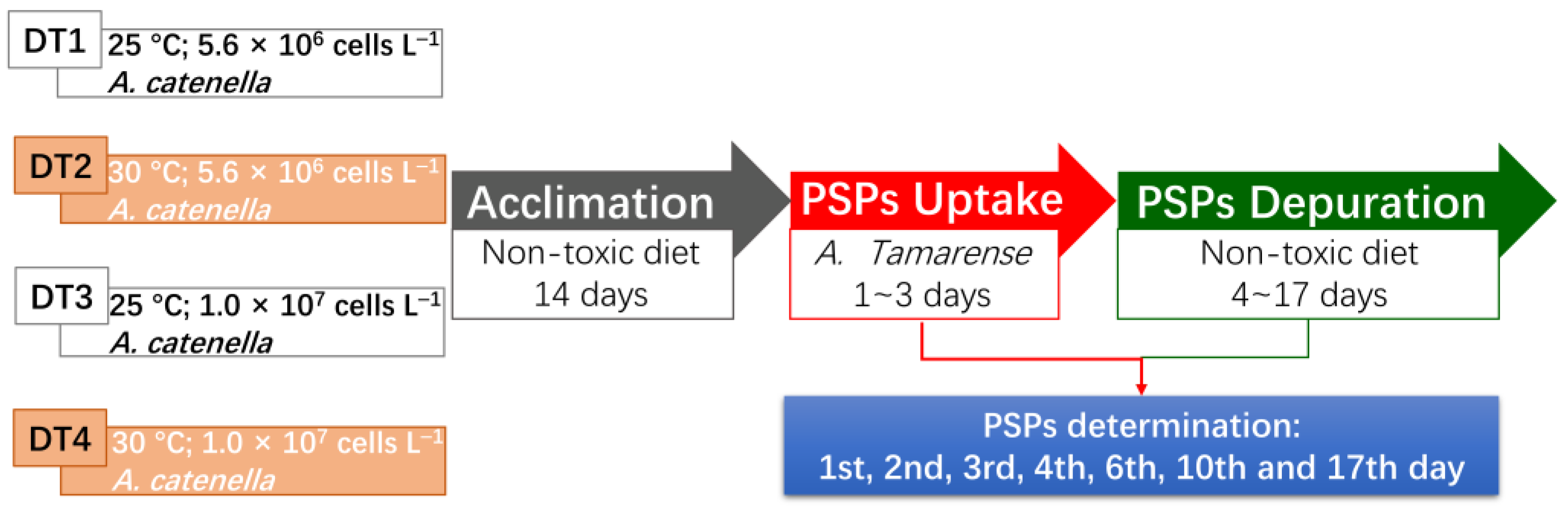
5. Materials and Methods
5.1. Reagents and Chemicals
5.2. Culture of A. catenella
5.3. Mussel Collection and Acclimation
5.4. Mussels Exposure to Toxic Dinoflagellates
5.5. Toxin Extraction
5.6. LC–MS/MS Analysis
5.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Doney, S.C.; Ruckelshaus, M.; Duffy, J.E.; Barry, J.P.; Chan, F.; English, C.A.; Galindo, H.M.; Grebmeier, J.M.; Hollowed, A.B.; Knowlton, N.; et al. Climate change impacts on marine ecosystems. Annu. Rev. Mar. Sci. 2012, 4, 11–37. [Google Scholar] [CrossRef] [PubMed]
- Hoegh-Guldberg, O.; Bruno, J.F. The impact of climate change on the world’s marine ecosystems. Science 2010, 328, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Glibert, P.M.; Icarus Allen, J.; Artioli, Y.; Beusen, A.; Bouwman, L.; Harle, J.; Holmes, R.; Holt, J. Vulnerability of coastal ecosystems to changes in harmful algal bloom distribution in response to climate change: Projections based on model analysis. Glob. Chang. Biol. 2014, 20, 3845–3858. [Google Scholar] [CrossRef]
- Gobler, C.J.; Doherty, O.M.; Hattenrath-Lehmann, T.K.; Griffith, A.W.; Kang, Y.; Litaker, R.W. Ocean warming since 1982 has expanded the niche of toxic algal blooms in the North Atlantic and North Pacific oceans. Proc. Natl. Acad. Sci. USA 2017, 114, 4975–4980. [Google Scholar] [CrossRef] [PubMed]
- Harley, C.D.; Hughes, A.R.; Hultgren, K.M.; Miner, B.G.; Sorte, C.J.; Thornber, C.S.; Rodriguez, L.F.; Tomanek, L.; Williams, S.L. The impacts of climate change in coastal marine systems. Ecol. Lett. 2006, 9, 228–241. [Google Scholar] [CrossRef]
- Duarte, C.; Navarro, J.; Acuña, K.; Torres, R.; Manríquez, P.; Lardies, M.; Vargas, C.; Lagos, N.; Aguilera, V. Combined effects of temperature and ocean acidification on the juvenile individuals of the mussel Mytilus chilensis. J. Sea Res. 2014, 85, 308–314. [Google Scholar] [CrossRef]
- Hochachka, P.W.; Lutz, P.L. Mechanism, origin, and evolution of anoxia tolerance in animals. Comp. Biochem. Phys. Part B Biochem. Mol. Biol. 2001, 130, 435–459. [Google Scholar] [CrossRef]
- Pörtner, H.O.; Farrell, A.P. Physiology and climate change. Science 2008, 322, 690–692. [Google Scholar] [CrossRef]
- Edwards, L.J.; Wilson, K.; Veitch, M.G. An outbreak of paralytic shellfish poisoning in Tasmania. Comm. Dis. Intell. 2018, 42, S2209-6051. [Google Scholar]
- Anestis, A.; Lazou, A.; Pörtner, H.O.; Michaelidis, B. Behavioral, metabolic, and molecular stress responses of marine bivalve Mytilus galloprovincialis during long-term acclimation at increasing ambient temperature. Am. J. Physiol. Integr. Comp. Physiol. 2007, 293, 911–921. [Google Scholar] [CrossRef]
- Andres, J.K.; Yñiguez, A.T.; Maister, J.M.; Turner, A.D.; Olano, D.E.B.; Mendoza, J.; Salvador-Reyes, L.; Azanza, R.V. Paralytic shellfish toxin uptake, assimilation, depuration, and transformation in the Southeast Asian green-lipped mussel (Perna viridis). Toxins 2019, 11, 468. [Google Scholar] [CrossRef]
- Kaga, S.; Sato, S.; Kaga, Y.; Naiki, K.; Watanabe, S.; Yamada, Y.; Ogata, T. Prediction of on-site depuration of paralytic shellfish poisoning toxins accumulated in the scallop Patinopecten yessoensis of Ofunato Bay, Japan. Fish. Sci. 2015, 81, 635–642. [Google Scholar] [CrossRef]
- Estrada, N.A.; Lagos, N.; García, C.; Maeda-Martínez, A.N.; Ascencio, F. Effects of the toxic dinoflagellate Gymnodinium catenatum, on uptake and fate of paralytic shellfish poisons in the Pacific giant lions-paw scallop Nodipecten subnodosus. Mar. Biol. 2007, 151, 1205–1214. [Google Scholar] [CrossRef]
- Yao, J.; Jin, W.; Li, D.; Xu, D.; Wen, S.; Liu, R.; Liang, Y.; Lu, S. Geographical distribution and seasonal variation in paralytic shellfish toxins in the coastal water of the South China Sea. Toxicon 2019, 168, 67–75. [Google Scholar] [CrossRef]
- Wang, X.; Wu, H.; Cheng, Y.; Wen, H.; Liu, R.; Wang, L.; Shan, C.; Chai, C. Multi-year assessment of paralytic shellfish toxins in hard clam species along the coastline of Jiangsu Province, China. Acta Oceanol. Sin. 2019, 38, 24–33. [Google Scholar] [CrossRef]
- Ford, S.; Bricelj, V.; Lambert, C.; Paillard, C. Deleterious effects of a nonPST bioactive compound(s) from Alexandrium tamarense on bivalve hemocytes. Mar. Biol. 2008, 154, 241–253. [Google Scholar] [CrossRef]
- Xie, W.; Liu, X.; Yang, X.; Zhang, C.; Bian, Z. Accumulation and depuration of paralytic shellfish poisoning toxins in the oyster Ostrea rivularis Gould-Chitosan facilitates the toxin depuration. Food Control. 2013, 30, 446–452. [Google Scholar] [CrossRef]
- Guéguen, M.; Baron, R.; Bardouil, M.; Truquet, P.; Haberkorn, H.; Lassus, P.; Amzil, Z. Modelling of paralytic shellfish toxin biotransformations in the course of Crassostrea gigas detoxification kinetics. Ecol. Model. 2011, 222, 3394–3402. [Google Scholar] [CrossRef]
- Moore, S.; Mantua, N.; Hickey, B.; Trainer, V. The relative influences of El Niño-Southern Oscillation and Pacific Decadal Oscillation on paralytic shellfish toxin accumulation in northwest Pacific shellfish. Limnol. Oceanogr. 2010, 55, 2262–2274. [Google Scholar] [CrossRef]
- Pousse, E.; Flye-Sainte-Marie, J.; Alunno-Bruscia, M.; Hegaret, H.; Rannou, E.; Pecquerie, L.; Marques, G.; Thomas, Y.; Castrec, J.; Fabioux, C.; et al. Modelling paralytic shellfish toxins (PST) accumulation in Crassostrea gigas by using Dynamic Energy Budgets (DEB). J. Sea Res. 2019, 143, 152–164. [Google Scholar] [CrossRef]
- Costa, P.; Lage, S.; Barata, M.; Pousao-Ferreira, P. Uptake, transformation, and elimination kinetics of paralytic shellfish toxins in white seabream (Diplodus sargus). Mar. Biol. 2011, 158, 2805–2811. [Google Scholar] [CrossRef]
- Seger, A.; Hallegraeff, G.; Stone, D.; Bansemer, M.; Harwood, D.; Turnbull, A. Uptake of paralytic shellfish toxins by Blacklip Abalone (Haliotis rubra rubra Leach) from direct exposure to Alexandrium catenella microalgal cells and toxic aquaculture feed. Harmful Algae 2020, 99, 101925. [Google Scholar] [CrossRef] [PubMed]
- Shumway, S.E. A review of the effects of algal blooms on shellfish and aquaculture. J. World Aquacult. Soc. 1990, 21, 65–104. [Google Scholar] [CrossRef]
- Bricelj, V.M.; Shumway, S.E. Paralytic shellfish toxins in bivalve molluscs: Occurrence, transfer kinetics, and biotransformation. Rev. Fish. Sci. 1998, 6, 315–383. [Google Scholar] [CrossRef]
- Quayle, D. Animal detoxification. In Proceedings of the Joint Sanitation Seminar on North Pacific Clams, Juneau, AK, USA, 24–25 September 1965. [Google Scholar]
- Blogoslawski, W.J.; Stewart, M.E. Paralytic shellfish poison in Spisula solidissima: Anatomical location and ozone detoxification. Mar. Biol. 1978, 45, 261–264. [Google Scholar] [CrossRef]
- Turnbull, A.; Malhi, N.; Seger, A.; Harwood, T.; Jolley, J.; Fitzgibbon, Q.; Hallegraeff, G. Paralytic shellfish toxin uptake, tissue distribution, and depuration in the Southern Rock Lobster Jasus edwardsii Hutton. Harmful Algae 2020, 95, 101818. [Google Scholar] [CrossRef] [PubMed]
- Kwong, R.; Wang, W.; Lam, P.; Yu, P.K. The uptake, distribution and elimination of paralytic shellfish toxins in mussels and fish exposed to toxic dinoflagellates. Aquat. Toxicol. 2006, 80, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Li, A.M.; Yu, P.K.; Hsieh, D.P.; Wang, W.X.; Wu, R.S.; Lam, P.K. Uptake and depuration of paralytic shellfish toxins in green-lipped mussel, Perna viridis: A dynamic model. Environ. Toxicol. Chem. 2005, 24, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Shin, I.S. Comparison of paralytic shellfish toxin profiles of Alexandrium tamarense, and blue mussel (Mytilus edulis) in Korea. Food Sci. Biotechnol. 2015, 24, 751–756. [Google Scholar] [CrossRef]
- Choi, M.; Hsieh, D.; Lam, P.; Wang, W.; Lam, K.; Wang, W. Field depuration and biotransformation of paralytic shellfish toxins in scallop Chlamys nobilis and green-lipped mussel Perna viridis. Mar. Biol. 2003, 143, 927–934. [Google Scholar] [CrossRef]
- Botelho, M.J.; Vale, C.; Mota, A.M.; de Lurdes, S.M. Depuration kinetics of paralytic shellfish toxins in Mytilus galloprovincialis exposed to Gymnodinium catenatum: Laboratory and field experiments. J. Environ. Monit. 2010, 12, 2269–2275. [Google Scholar] [CrossRef]
- Marques, A.; Nunes, M.L.; Moore, S.K.; Strom, M.S. Climate change and seafood safety: Human health implications. Food Res. Int. 2010, 43, 1766–1779. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- Coppola, F.; Almeida, Â.; Henriques, B.; Soares, A.M.V.M.; Figueira, E.; Pereira, E.; Freitas, R. Biochemical impacts of Hg in Mytilus galloprovincialis under present and predicted warming scenarios. Sci. Total Environ. 2017, 601, 1129–1138. [Google Scholar] [CrossRef]
- Múgica, M.; Izagirre, U.; Marigómez, I. Lysosomal responses to heat-shock of seasonal temperature extremes in Cd-exposed mussels. Aquat. Toxicol. 2015, 164, 99–107. [Google Scholar] [CrossRef]
- Van Dolah, F.M. Marine algal toxins: Origins, health effects, and their increased Occurrence. Environ. Health Perspect. 2000, 108, 133–141. [Google Scholar] [CrossRef]
- Braga, A.C.; Camacho, C.; Marques, A.; Gago-Martínez, A.; Pacheco, M.; Costa, P.R. Combined effects of warming and acidification on accumulation and elimination dynamics of paralytic shellfish toxins in mussels Mytilus galloprovincialis. Environ. Res. 2018, 164, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Farrell, H.; Seebacher, F.; O’Connor, W.; Zammit, A.; Harwood, D.T.; Murray, S. Warm temperature acclimation impacts metabolism of paralytic shellfish toxins from Alexandrium minutum in commercial oysters. Glob. Chang. Biol. 2015, 21, 3402–3413. [Google Scholar] [CrossRef] [PubMed]
- Indrasena, W.M.; Gill, T.A. Storage stability of paralytic shellfish poisoning toxins. Food Chem. 2000, 71, 71–77. [Google Scholar] [CrossRef]
- Li, S.; Wang, W.; Hsieh, D. Feeding and absorption of the toxic dinoflagellate Alexandrium tamarense by two marine bivalves from the South. China Sea. Mar. Biol. 2001, 139, 617–624. [Google Scholar] [CrossRef]
- Chen, J.H.; Yu, R.; Gao, Y.; Kong, F.; Wang, Y.; Zhang, Q.; Kang, Z.; Yan, T.; Zhou, M. Tracing the origin of paralytic shellfish toxins in scallop Patinopecten yessoensis in the northern Yellow Sea. Food Addit. Contam. A 2013, 30, 1933–1945. [Google Scholar] [CrossRef]
- Jiang, T.; Xu, Y.X.; Li, Y.; Jiang, T.J.; Wu, F.; Zhang, F. Seasonal dynamics of Alexandrium tamarense and occurrence of paralytic shellfish poisoning toxins in bivalves in Nanji Islands, East China Sea. Mar. Freshw. Res. 2014, 65, 350–358. [Google Scholar] [CrossRef]
- Qi, Y.Z.; Tian, F. Taxonomic studies on red tide causative dinoflagellates in Dapeng Bay, South China Sea. Oceanol. Limnol. Sin. 1994, 25, 206–210. (In Chinese) [Google Scholar]
- Anderson, D.M.; Kulis, D.M.; Qi, Y.Z.; Zheng, L.; Lu, S.H.; Lin, Y.T. Paralytic shellfish poisoning in southern China. Toxicon 1996, 34, 579–590. [Google Scholar] [CrossRef]
- Costa, P.R.; Robertson, A.; Quilliam, M.A. Toxin profile of Gymnodinium catenatum (Dinophyceae) from the Portuguese coast, as determined by liquid chromatography tandem mass spectrometry. Mar. Drugs 2015, 13, 2046–2062. [Google Scholar] [CrossRef] [PubMed]
- Carreto, J.I.; Carignan, M.O.; Montoya, N.G. Comparative studies on mycosporine-like amino acids, paralytic shellfish toxins and pigment profiles of the toxic dinoflagellates Alexandrium tamarense, A. catenella and A. minutum. Mar. Ecol. Prog. Ser. 2001, 223, 49–60. [Google Scholar] [CrossRef]
- Montojo, U.; Sakamoto, S.; Cayme, M.; Gatdula, N.; Furio, E.; Relox, J.; Sato, S.; Fukuyo, Y.; Kodama, M. Remarkable difference in accumulation of paralytic shellfish poisoning toxins among bivalve species exposed to Pyrodinium bahamense var. compressum bloom in Masinloc bay, Philippines. Toxicon 2006, 48, 85–92. [Google Scholar]
- Vidal, T.; Calado, A.; Moita, M.; Cunha, M. Phytoplankton dynamics in relation to seasonal variability and upwelling and relaxation patterns at the mouth of Ria de Aveiro (West Iberian Margin) over a four-year period. PLoS ONE 2017, 12, e0177237. [Google Scholar] [CrossRef]
- Wang, Z.; Nie, X.; Jiang, S.; Jiao, J.; Cao, Y.; Zhang, Y.; Wang, D. Source and profile of paralytic shellfish poisoning toxins in shellfish in Daya Bay, South China Sea. Mar. Environ. Res. 2011, 72, 53–59. [Google Scholar] [CrossRef]
- Song, X.; Ma, J.; Liu, Y.; Liu, L.; Ma, Y.; Ren, L.; Tang, X. Evolution and formation of Alexandrium tamarense red tide in the sea area of the Nanhuangcheng Island. Trans. Oceanol Limnol. 2009, 30, 93–98. (In Chinese) [Google Scholar]
- Lischka, S.; Udenbender, J.; Boxhammer, T.; Riebesell, U. Impact of ocean acidification and elevated temperatures on early juveniles of the polar shelled pteropod Limacina helicina: Mortality, shell degradation, and shell growth. Biogeosciences 2011, 8, 919–932. [Google Scholar] [CrossRef]
- Grenier, C.; Román, R.; Duarte, C.; Navarro, J.M.; Rodriguez-Navarro, A.B.; Ramajo, L. The combined effects of salinity and pH on shell biomineralization of the edible mussel Mytilus chilensis. Environ. Pollut. 2020, 263, 114555. [Google Scholar] [CrossRef] [PubMed]
- Fernández, P.A.; Leal, P.P.; Henríquez, L.A. Co-culture in marine farms: Macroalgae can act as chemical refuge for shell-forming molluscs under an ocean acidification scenario. Phycologia 2019, 58, 542–551. [Google Scholar] [CrossRef]
- Melzner, F.; Stange, P.; Trubenbach, K.; Thomsen, J.; Casties, I.; Panknin, U.; Gorb, S.N.; Gutowska, M.A. Food supply and seawater pCO2 impact calcification and internal shell dissolution in the blue mussel Mytilus edulis. PLoS ONE 2011, 6, e24223. [Google Scholar] [CrossRef] [PubMed]
- Gobler, C.J.; DePasquale, E.L.; Griffith, A.W.; Baumann, H. Hypoxia and acidification have additive and synergistic negative effects on the growth, survival, and metamorphosis of early life stage bivalves. PLoS ONE 2014, 9, e83648. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, L.; Zheng, L.; Fu, P.; Wang, Y.; Nguyen, H.; Shen, X.; Sui, Y. Antioxidant responses of triangle sail mussel Hyriopsis cumingii exposed to harmful algae Microcystis aeruginosa and high pH. Chemosphere 2020, 243, 125241. [Google Scholar] [CrossRef]
- Jiang, H.; Lan, W.; Li, T.; Xu, Z.; Liu, W.; Pan, K. Isotopic composition reveals the impact of oyster aquaculture on pelagic nitrogen cycling in a subtropical estuary. Water Res. 2020, 187, 116431. [Google Scholar] [CrossRef]
- Codex Alimentarius Commission. Codex Standard 292-2008 Standard for Live and Raw Bivalve Molluscs; Codex Alimentarius Commisssion: Rome, Italy, 2008. [Google Scholar]
- Mak, K.; Li, A.; Hsieh, D.; Wong, P.; Lam, M.; Wu, R.; Richardson, B.; Lam, P. Paralytic shellfish toxins in green-lipped mussels, Perna viridis, in Hong Kong. Mar. Pollut. Bull. 2003, 46, 258–268. [Google Scholar] [CrossRef]
- Yu, K.; Choi, M.; Shen, X.; Wu, R.; Wang, W.; Lam, P. Modeling of depuration of paralytic shellfish toxins in Chlamys nobilis and Perna viridis. Mar. Pollut. Bull. 2005, 50, 463–484. [Google Scholar] [CrossRef]
- Anna, M.B.; Monica, C.; Alessandro, G.M.; Giuseppa, L.; Igor, A.; Giovanna, S.; Fiorella, P.; Anna, M.; Sebastiano, V. Recent findings of paralytic shellfish toxins linked to the genus Alexandrium Halim in Mediterranean mollusc production areas. Toxicon 2020, 174, 48–56. [Google Scholar]
- Ding, L.; Qiu, J.; Li, A. Proposed biotransformation pathways of new metabolites of paralytic shellfish toxins based in field and experimental mussel samples. J. Agric. Food Chem. 2017, 65, 5494–5502. [Google Scholar] [CrossRef]
- Xu, Y.; He, X.; Li, H.; Zhang, T.; Lei, F.; Gu, H.; Anderson, D. Molecular identification and toxin analysis of Alexandrium spp. in the Beibu Gulf: First report of toxic A. tamiyavanichii in Chinese coastal waters. Toxins 2021, 13, 161. [Google Scholar] [CrossRef]
- Asakawa, M.; Beppu, R.; Ito, K.; Tsubota, M.; Takayama, H.; Miyazawa, K. Accumulation of paralytic shellfish poison (PSP) and biotransformation of its components in oysters Crassostrea gigas fed with the toxic dinoflagellate Alexandrium tamarense. Shokuhin Eiseigaku Zasshi 2005, 47, 28–32. [Google Scholar] [CrossRef]
- Ichimi, K.; Suzuki, T.; Yamasaki, M. Non-selective retention of PSP toxins by the mussel Mytilus galloprovincialis fed with the toxic dinoflagellate Alexandrium tamarense. Toxicon 2001, 39, 1917–1921. [Google Scholar] [CrossRef]
- Wiese, M.; D’Agostino, P.M.; Mihali, T.K.; Moffitt, M.C.; Neilan, B.A. Neurotoxic Alkaloids: Saxitoxin and Its Analogs. Mar. Drugs 2010, 8, 2185–2211. [Google Scholar] [CrossRef] [PubMed]
- Anestis, A.; Portner, H.O.; Karagiannis, D.; Angelidis, P.; Staikou, A.; Michaelidis, B. Response of Mytilus galloprovincialis (L.) to increasing seawater temperature and to marteliosis: Metabolic and physiological parameters. Comp. Biochem. Phys. Part A Mol. Integr. Physiol. 2010, 156, 57–66. [Google Scholar] [CrossRef]
- Donovan, C.J.; Ku, J.C.; Quilliam, M.A.; Gill, T.A. Bacterial degradation of paralytic shellfish toxins. Toxicon 2008, 52, 91–100. [Google Scholar] [CrossRef]
- Fernández-Reiriz, M.; Range, P.; Álvarez-Salgado, X.; Espinosa, J.; Labarta, U. Tolerance of juvenile Mytilus galloprovincialis to experimental seawater acidification. Mar. Ecol. Prog. Ser. 2012, 454, 65–74. [Google Scholar] [CrossRef]
- Raposo, M.I.C.; Gomes, M.T.S.R.; Botelho, M.J.; Rudnitskaya, A. Paralytic shellfish toxins (PST)-transforming enzymes: A review. Toxins 2020, 12, 344. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, S.; Sato, S.; Ogata, T.; Kodama, M. Formation of intermediate conjugates in the reductive transformation of gonyautoxins to saxitoxins by thiol compounds. Fish. Sci. 2000, 66, 136–141. [Google Scholar] [CrossRef]
- Barbosa, V.; Santos, M.; Anacleto, P.; Maulvault, A.L.; Pousão-Ferreira, P.; Costa, P.R.; Marques, A. Paralytic shellfish toxins and ocean warming: Bioaccumulation and ecotoxicological responses in Juvenile Gilthead Seabream (Sparus aurata). Toxins 2019, 11, 408. [Google Scholar] [CrossRef]
- Blanco, J.; Reyero, M.I.; Franco, J. Kinetics of accumulation and transformation of paralytic shellfish toxins in the blue mussel Mytilus galloprovincialis. Toxicon 2003, 42, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Guillard, R.R.; Ryther, J.H. Studies of marine planktonic diatoms: I. Cyclotella nana Hustedt, and Detonula confervacea (Cleve) Gran. Can. J. Microbiol. 1962, 8, 229–239. [Google Scholar] [CrossRef]
- Silva, T.; Caeiro, M.F.; Costa, P.R.; Amorim, A. Gymnodinium catenatum Graham isolated from the Portuguese coast: Toxin content and genetic characterization. Harmful Algae 2015, 48, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, J.; Menard, C. Determination of marine toxins by liquid chromatography. J. Anal. Chem. 1991, 339, 494–498. [Google Scholar] [CrossRef]
- Turner, A.; McNabb, P.; Harwood, T.; Selwood, A.; Boundy, M. Single-laboratory validation of a multitoxin ultra-performance LC-hydrophilic interaction LC-MS/MS method for quantitation of paralytic shellfish toxins in bivalve shellfish. J. AOAC Int. 2015, 98, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations (FAO); Intergovernmental Oceanographic Commission (IOC); World Health Organization (WHO). Joint FAO/IOC/WHO ad hoc Expert Consultation on Biotoxins in Bivalve Molluscs—Background Document. In Proceedings of the Report of the Joint FAO/IOC/WHO ad hoc Expert Consultation on Biotoxins in Bivalve Molluscs, Oslo, Norway, 26–30 September 2004. [Google Scholar]
- Azanza, R.V.; Cruz, L.J.; CariñO, F.A.; Blanco, A.G.; Butardo, V.M., Jr. Paralytic shellfish toxin concentration and cell density changes in Pyrodinium bahamense—Noctiluca scintillans feeding experiments. Toxicon 2010, 55, 1017–1023. [Google Scholar] [CrossRef]


| Toxins | Toxin Concentration (pg/cell) | % of Total Toxin |
|---|---|---|
| C1+2 | 6.69 | 38.9 |
| GTX1+4 | 8.86 | 51.6 |
| GTX2+3 | 0.053 | 0.3 |
| GTX5 | 1.15 | 6.7 |
| dcGTX2+3 | 0.055 | 0.4 |
| STX | 0.018 | 0.1 |
| NEO | 0.319 | 1.9 |
| dcSTX | 0.019 | 0.1 |
| Days | % of Toxin Distribution | % of the PSTs | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Hepatopancreas | Gill | Mantle | Muscle | Gonad | GTX1+4 | NEO | GTX5 | C1+2 | |
| 1 | 88 | 3 | 4 | 3 | 2 | 63 | 2 | 8 | 27 |
| 2 | 86 | 4 | 5 | 3 | 2 | 60 | 2 | 8 | 30 |
| 3 | 90 | 3 | 3 | 2 | 2 | 59 | 2 | 8 | 31 |
| 4 | 89 | 4 | 3 | 2 | 2 | 61 | 2 | 8 | 29 |
| 6 | 93 | 2 | 2 | 2 | 1 | 59 | 2 | 8 | 31 |
| 10 | 91 | 2 | 3 | 2 | 2 | 46 | 4 | 8 | 42 |
| 17 | 92 | 0.2 | 7.8 | - | - | 23 | 7 | 8 | 62 |
| Treatment | Toxin | Elimination Rate | R2 | |
|---|---|---|---|---|
| DT1 25 °C and Low abundance | GTX1+4 | 0.206 | (±0.004) | 0.9967 |
| NEO | 0.351 | (±0.064) | 0.8762 | |
| GTX5 | 0.163 | (±0.007) | 0.9865 | |
| C1+2 | 0.140 | (±0.012) | 0.9765 | |
| DT2 30 °C and Low abundance | GTX1+4 | 0.518 | (±0.082) | 0.8264 |
| NEO | 0.322 | (±0.116) | 0.6666 | |
| GTX5 | 0.393 | (±0.099) | 0.7592 | |
| C1+2 | 0.141 | (±0.011) | 0.9766 | |
| DT3 25 °C and High abundance | GTX1+4 | 0.232 | (±0.014) | 0.9791 |
| NEO | 0.069 | (±0.011) | 0.9377 | |
| GTX5 | 0.167 | (±0.006) | 0.9912 | |
| C1+2 | 0.116 | (±0.004) | 0.9948 | |
| DT4 30 °C and High abundance | GTX1+4 | 0.526 | (±0.095) | 0.7638 |
| NEO | 0.060 | (±0.026) | 0.6236 | |
| GTX5 | 0.432 | (±0.098) | 0.8189 | |
| C1+2 | 0.142 | (±0.009) | 0.9828 | |
| ESI Mode | Toxin | Precursor Ion (m/z) | Product Ion (m/z) | Fragmentor (v) | Collision Energy (v) |
|---|---|---|---|---|---|
| ESI− | GTX2,3 | 394.0 | 333.1 | 80 | −22 |
| 351.1 | −16 | ||||
| GTX1,4 | 410.1 | 367.1 | 80 | −15 | |
| 349.1 | −22 | ||||
| dcGTX2,3 | 351.1 | 333.1 | 100 | −17 | |
| 164.0 | −30 | ||||
| C1/2 | 474.1 | 351.1 | 90 | −25 | |
| 122.0 | −30 | ||||
| GTX5 | 378.1 | 122.1 | 100 | −22 | |
| 360.1 | −16 | ||||
| ESI+ | STX | 300.2 | 221.0 | 120 | 35 |
| 204.0 | 30 | ||||
| NEO | 316.1 | 298.2 | 120 | 34 | |
| 126.1 | 34 | ||||
| dcSTX | 257.1 | 239.1 | 120 | 22 | |
| 126.1 | 30 | ||||
| dcNEO | 273.1 | 225.2 | 120 | 35 | |
| 126.1 | 35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, Y.; Zhang, H.; Wang, Y.; Fan, C.; Shen, X. Combined Effects of Temperature and Toxic Algal Abundance on Paralytic Shellfish Toxic Accumulation, Tissue Distribution and Elimination Dynamics in Mussels Mytilus coruscus. Toxins 2021, 13, 425. https://doi.org/10.3390/toxins13060425
Tang Y, Zhang H, Wang Y, Fan C, Shen X. Combined Effects of Temperature and Toxic Algal Abundance on Paralytic Shellfish Toxic Accumulation, Tissue Distribution and Elimination Dynamics in Mussels Mytilus coruscus. Toxins. 2021; 13(6):425. https://doi.org/10.3390/toxins13060425
Chicago/Turabian StyleTang, Yunyu, Haiyan Zhang, Yu Wang, Chengqi Fan, and Xiaosheng Shen. 2021. "Combined Effects of Temperature and Toxic Algal Abundance on Paralytic Shellfish Toxic Accumulation, Tissue Distribution and Elimination Dynamics in Mussels Mytilus coruscus" Toxins 13, no. 6: 425. https://doi.org/10.3390/toxins13060425
APA StyleTang, Y., Zhang, H., Wang, Y., Fan, C., & Shen, X. (2021). Combined Effects of Temperature and Toxic Algal Abundance on Paralytic Shellfish Toxic Accumulation, Tissue Distribution and Elimination Dynamics in Mussels Mytilus coruscus. Toxins, 13(6), 425. https://doi.org/10.3390/toxins13060425





