Ecophysiological Aspects and sxt Genes Expression Underlying Induced Chemical Defense in STX-Producing Raphidiopsis raciborskii (Cyanobacteria) against the Zooplankter Daphnia gessneri
Abstract
1. Introduction
2. Results
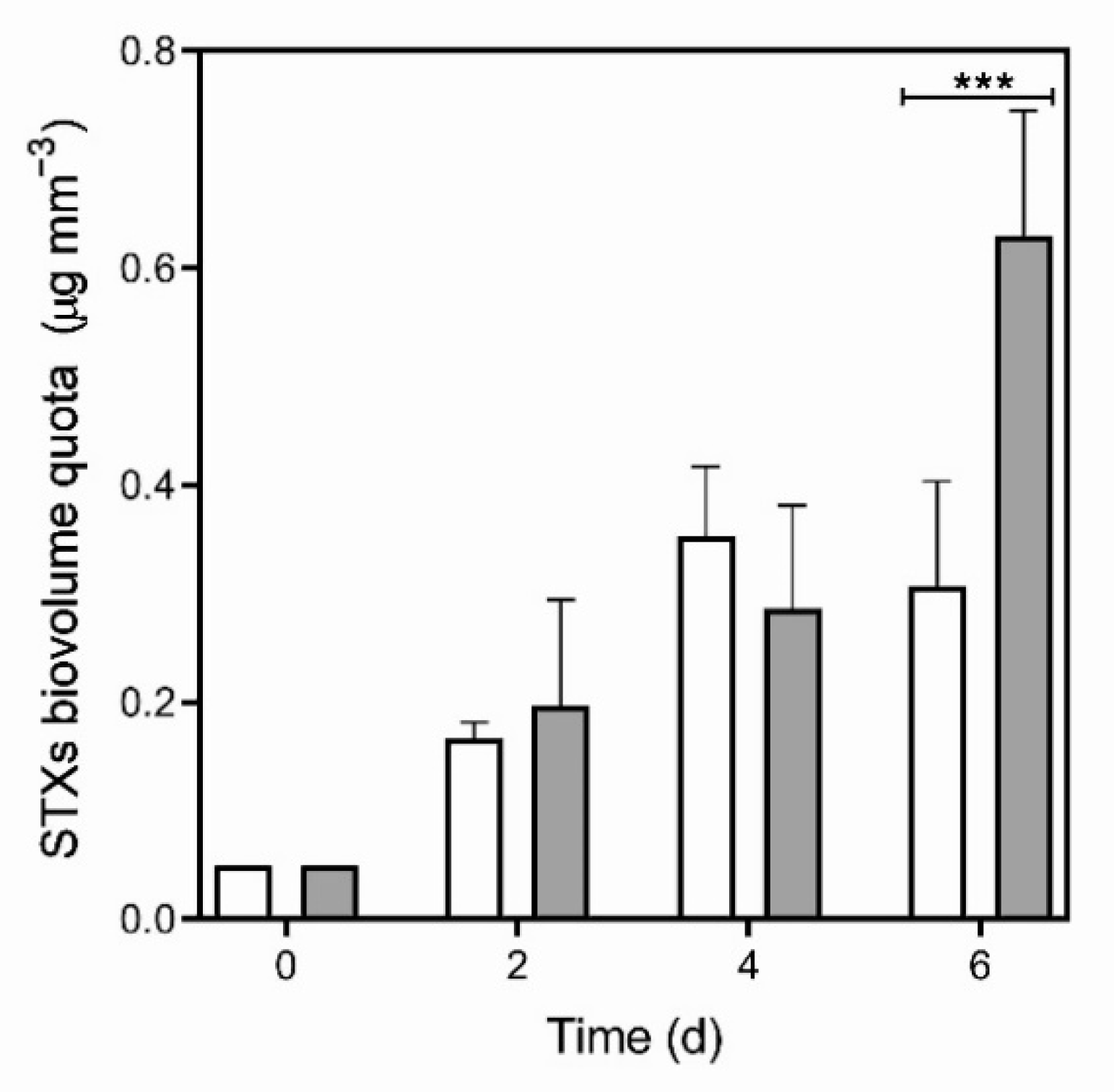
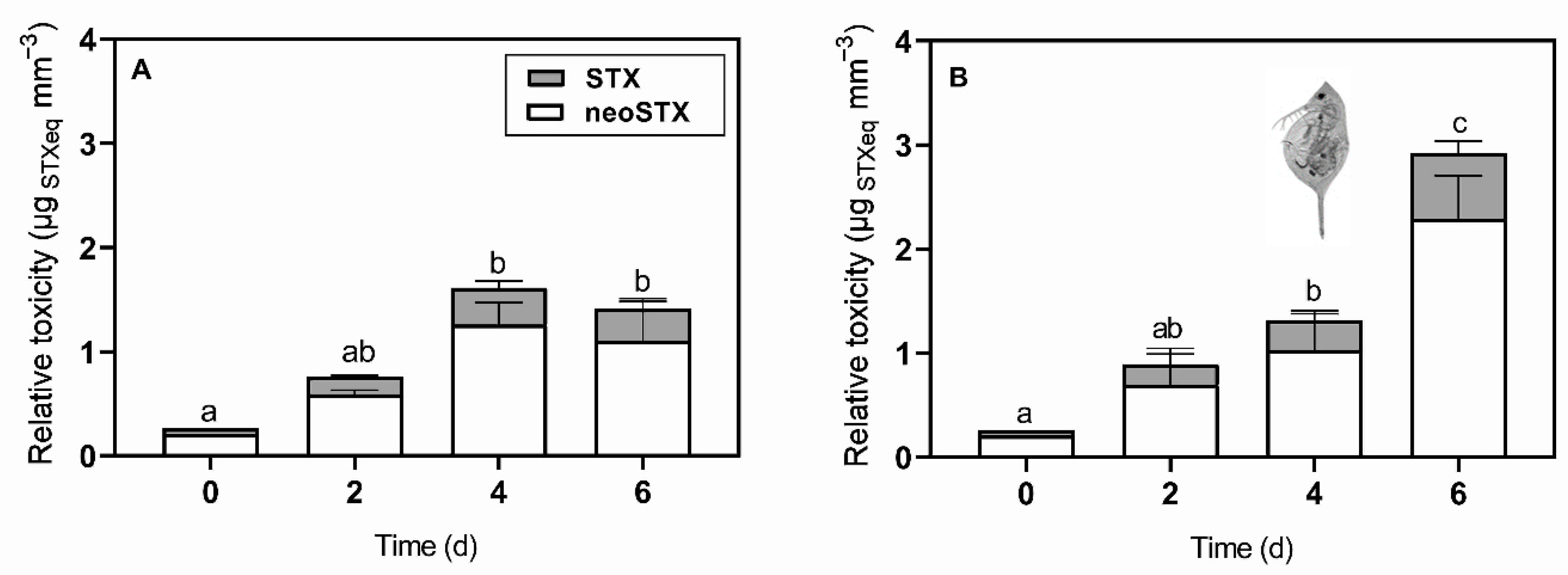
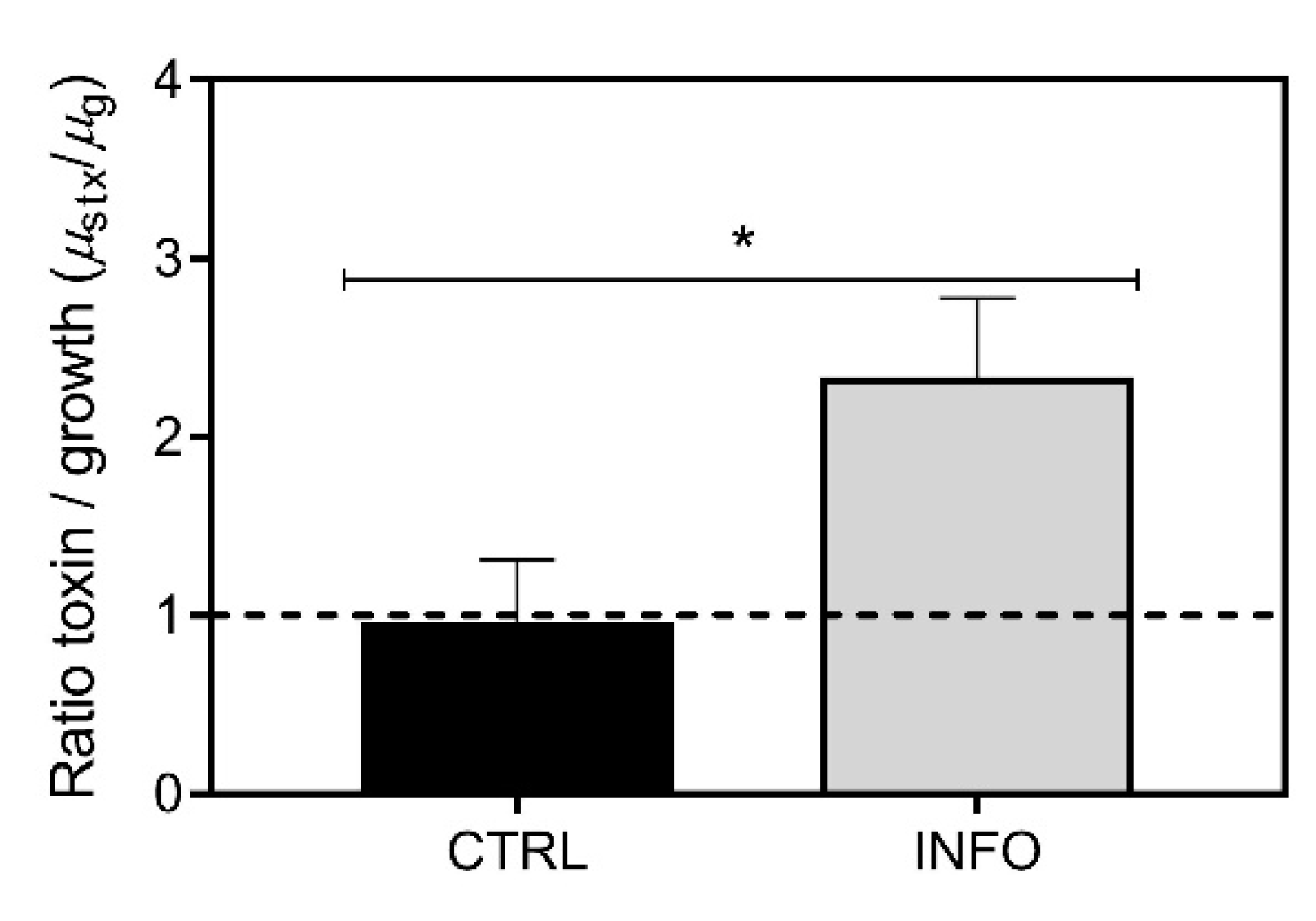
2.1. Predator-Induced Chemical Defense
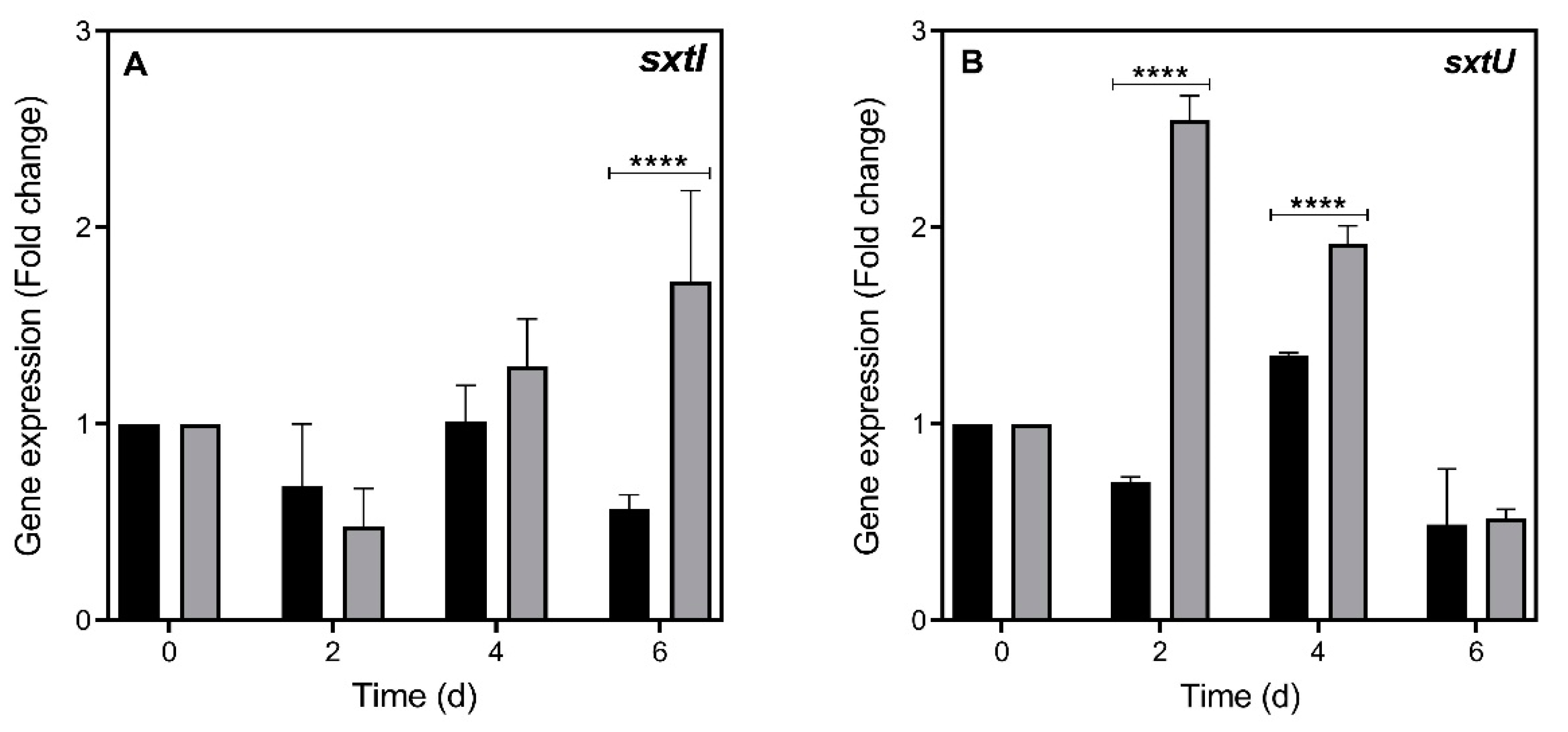
2.2. STX Gene Expression
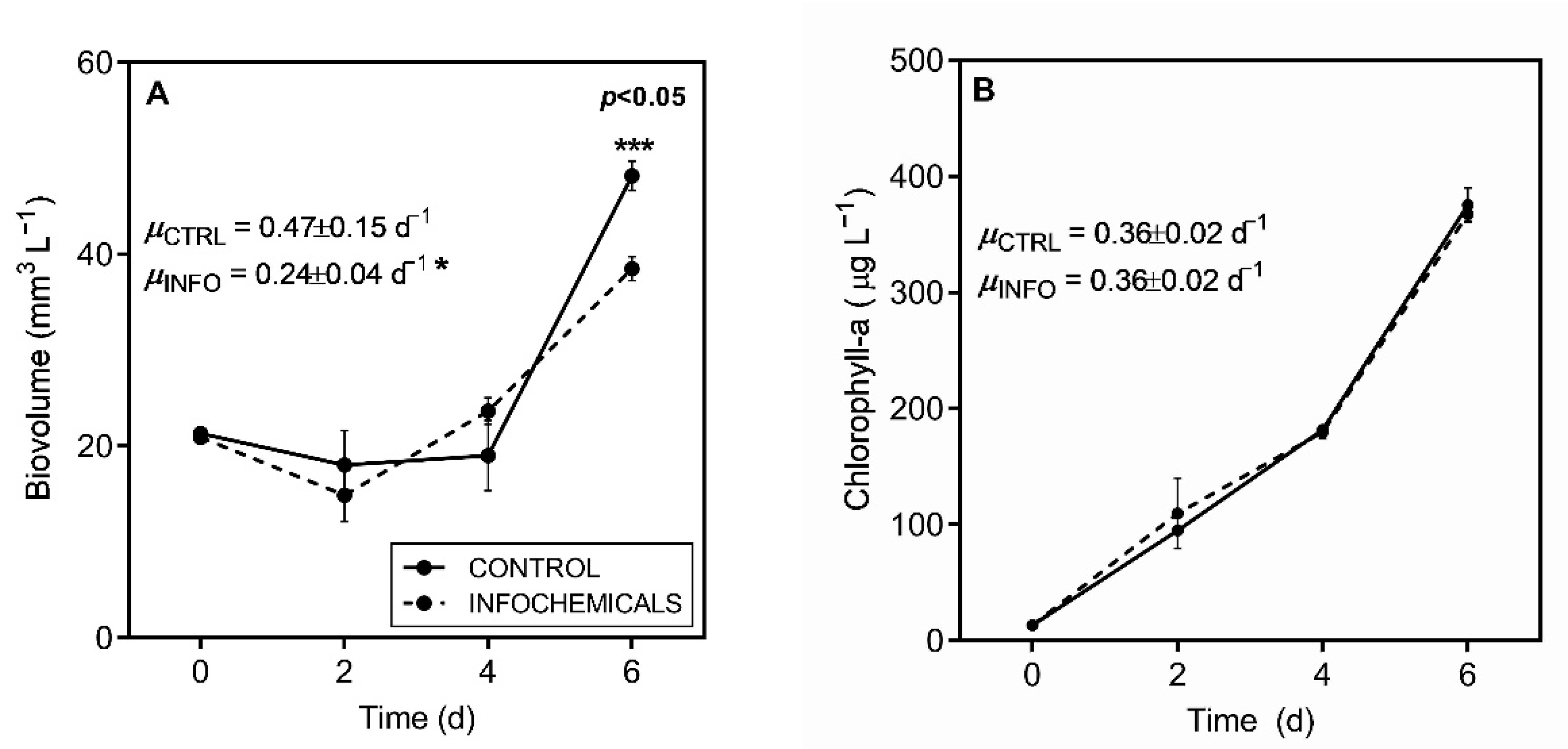
2.3. Growth, Morphology and Photosynthetic Parameters
3. Discussion
4. Materials and Methods
4.1. Phytoplankton and Zooplankton Cultures
4.2. Experimental Set-Up
4.2.1. Zooplankton Filtrate to Obtain Infochemicals
4.2.2. Predator-Induced Defenses Experiment
4.3. Growth, Morphology and Photosynthetic Measurements
4.4. STX Analyses and Toxicity Equivalency Calculation
4.5. Application of First-Order Rate Kinetics to Assess Total STX Production
4.6. Expression of Genes Involved in STX Biosynthesis
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vermeij, G.J. The evolutionary interaction among species: Selection, escalation, and coevolution. Annu. Rev. Ecol. Evol. Syst. 1994, 25, 219–236. [Google Scholar] [CrossRef]
- Van Donk, E.; Ianora, A.; Vos, M. Induced defences in marine and freshwater phytoplankton: A review. Hydrobiologia 2011, 668, 3–19. [Google Scholar] [CrossRef]
- Lürling, M. Grazing resistance in phytoplankton. Hydrobiologia 2020, 848, 237–249. [Google Scholar] [CrossRef]
- Jang, M.H.; Ha, K.; Joo, G.J.; Takamura, N. Toxin production of cyanobacteria is increased by exposure to zooplankton. Freshwa. Biol. 2003, 48, 1540–1550. [Google Scholar] [CrossRef]
- Jang, M.H.; Jung, J.M.; Takamura, N. Changes in microcystin production in cyanobacteria exposed to zooplankton at different population densities and infochemical concentrations. Limnol. Oceanogr. 2007, 52, 1454–1466. [Google Scholar] [CrossRef]
- Jang, M.; Ha, K.; Takamura, N. Microcystin production by Microcystis aeruginosa exposed to different stages of herbivorous zooplankton. Toxicon 2008, 51, 882–889. [Google Scholar] [CrossRef]
- Selander, E.; Thor, P.; Toth, G.; Pavia, H. Copepods induce paralytic shellfish toxin production in marine dinoflagellates. Proc. Royal Soc. B 2006, 273, 1673–1680. [Google Scholar] [CrossRef]
- Lürling, M. Phenotypic plasticity in the green algae Desmodesmus and Scenedesmus with special reference to the induction of defensive morphology. Ann. Limnol. 2003, 39, 85–101. [Google Scholar] [CrossRef]
- Lurling, M.; Beekman, W. Palmelloids formation in Chlamydomonas reinhardtii: Defence against rotifer predators? Ann. Limnol. 2006, 42, 65–72. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, J.; Qin, B.; Cui, G.; Yang, Z. Grazer density-dependent response of induced colony formation of Scenedesmus obliquus to grazing-associated infochemicals. Biochem. Syst. Ecol. 2013, 50, 286–292. [Google Scholar] [CrossRef]
- Wejnerowski, L.; Cerbin, S.; Wojciechowicz, M.; Jurczak, T.; Glama, M.; Meriluoto, J.; Dziuba, M. Effects of Daphnia exudates and sodium octyl sulphates on filament morphology and cell wall thickness of Aphanizomenon gracile (Nostocales), Cylindrospermopsis raciborskii (Nostocales) and Planktothrix agardhii (Oscillatoriales). Eur. J. Phycol. 2018, 53, 280–289. [Google Scholar] [CrossRef]
- Yasumoto, K.; Nishigami, A.; Yasumoto, M.; Kasai, F.; Okada, Y.; Kusumi, T.; Ooi, T. Aliphatic sulfates released from Daphnia induce morphological defense of phytoplankton: Isolation and synthesis of kairomones. Tetrahedron Lett. 2005, 46, 4765–4767. [Google Scholar] [CrossRef]
- Yasumoto, K.; Nishigami, A.; Ao, H.; Tsuchihashi, C.; Kasai, F.; Kusumi, T.; Ooi, T. Isolation of new aliphatic sulfates and sulfamate as the Daphnia kairomones inducing morphological change of a phytoplankton Scenedesmus gutwinskii. Chem. Pharm. Bull. 2008, 56, 133–136. [Google Scholar] [CrossRef]
- Esterhuizen-Londt, M.; von Schnehen, M.; Kühn, S.; Pflugmacher, S. Oxidative stress responses in the animal model, Daphnia pulex exposed to a natural bloom extract versus artificial cyanotoxin mixtures. Aquat. Toxicol. 2016, 179, 151–157. [Google Scholar] [CrossRef]
- Ferrão-Filho, A.S.; Kozlowsky-Suzuki, B. Cyanotoxins: Bioaccumulation and effects on aquatic animals. Mar. Drugs 2011, 9, 2729–2772. [Google Scholar] [CrossRef]
- Ferrão-Filho, A.S.; Dias, T.M.; Pereira, U.J.; Santos, J.A.A.; Kozlowsky-Suzuki, B. Nutritional and toxicity constraints of phytoplankton from a Brazilian reservoir to the fitness of cladoceran species. Environ. Sci. Pollut. Res. 2019, 26, 12881–12893. [Google Scholar] [CrossRef]
- Santos, G.S.D.; Vilar, M.C.P.; Azevedo, S.M.F.O. Acute toxicity of neurotoxin-producing Raphidiopsis (Cylindrospermopsis) raciborskii ITEP-A1 (Cyanobacteria) on the neotropical cladoceran Macrothrix spinosa. Ecotoxicol. Environ. Contam. 2020, 15, 1–8. [Google Scholar] [CrossRef]
- Bownik, A. Harmful Algae: Effects of cyanobacterial cyclic peptides on aquatic invertebrates–a short review. Toxicon 2016, 124, 26–35. [Google Scholar] [CrossRef]
- Ferrão-Filho, A.S.; Silva, D.A.C. Saxitoxin-producing Raphidiopsis raciborskii (Cyanobacteria) inhibits swimming and physiological parameters in Daphnia similis. Sci. Total. Environ. 2020, 706, 135751. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Pearson, L.A.; Dittmann, E.; Mazmouz, R.; Ongley, S.E.; D’Agostino, P.M.; Neilan, B.A. The genetics, biosynthesis and regulation of toxic specialized metabolites of cyanobacteria. Harmful Algae 2016, 54, 98–111. [Google Scholar] [CrossRef]
- Holland, A.; Kinnear, S. Interpreting the Possible Ecological Role(s) of Cyanotoxins: Compounds for Competitive Advantage and/or Physiological Aide? Mar. Drugs 2013, 11, 2239–2258. [Google Scholar] [CrossRef]
- Kaplan, A.; Harel, M.; Kaplan-Levy, R.N.; Hadas, O.; Sukenik, A.; Dittmann, E. The languages spoken in the water body (or the biological role of cyanobacterial toxins). Front. Microbiol. 2012, 3, 138. [Google Scholar] [CrossRef]
- Omidi, A.; Esterhuizen-Londt, M.; Pflugmacher, S. Still challenging: The ecological function of the cyanobacterial toxin microcystin–What we know so far. Toxin Rev. 2018, 37, 87–105. [Google Scholar] [CrossRef]
- Aguilera, A.; Gómez, E.B.; Kaštovský, J.; Echenique, R.O.; Salerno, G.L. The polyphasic analysis of two native Raphidiopsis isolates supports the unification of the genera Raphidiopsis and Cylindrospermopsis (Nostocales, Cyanobacteria). Phycologia 2018, 57, 130–146. [Google Scholar] [CrossRef]
- Antunes, J.T.; Leão, P.N.; Vasconcelos, V.M. Cylindrospermopsis raciborskii: Review of the distribution, phylogeography, and ecophysiology of a global invasive species. Front. Microbiol. 2015, 6, 1–13. [Google Scholar] [CrossRef]
- Burford, M.A.; Beardall, J.; Willis, A.; Orr, P.T.; Magalhães, V.F.; Rangel, L.M.; Azevedo, S.M.F.O.; Neilan, B.A. Understanding the winning strategies used by the bloom-forming cyanobacterium Cylindrospermopsis raciborskii. Harmful Algae 2016, 54, 44–53. [Google Scholar] [CrossRef]
- Jia, N.; Yang, Y.; Yu, G.; Wang, Y.; Qiu, P.; Li, H.; Li, R. Interspecific competition reveals Raphidiopsis raciborskii as a more successful invader than Microcystis aeruginosa. Harmful Algae 2020, 97, 101858. [Google Scholar] [CrossRef]
- Lagos, N.; Onodera, H.; Zagatto, P.A.; Andrinolo, D.; Azevedo, S.M.F.O.; Oshima, Y. The first evidence of paralytic shellfish toxins in the freshwater cyanobacterium Cylindrospermopsis raciborskii, isolated from Brazil. Toxicon 1999, 37, 1359–1373. [Google Scholar] [CrossRef]
- Mesquita, M.C.B.; Lürling, M.; Dorr, F.; Pinto, E.; Marinho, M.M. Combined effect of light and temperature on the Production of saxitoxins in Cylindrospermopsis raciborskii strains. Toxins 2019, 11, 38. [Google Scholar] [CrossRef]
- Molica, R.J.R.; Onodera, H.; García, C.; Rivas, M.; Andrinolo, D.; Nascimento, S.M.; Meguro, H.; Oshima, Y.; Azevedo, S.M.F.O.; Lagos, N. Toxins in the freshwater cyanobacterium Cylindrospermopsis raciborskii (Cyanophyceae) isolated from Tabocas reservoir in Caruaru, Brazil, including demonstration of a new saxitoxin analogue. Phycologia 2002, 41, 606–611. [Google Scholar] [CrossRef]
- Piccini, C.; Aubriot, L.; D’Alessandro, B.; Martigani, F.; Bonilla, S. Revealing Toxin Signatures in Cyanobacteria: Report of Genes Involved in Cylindrospermopsin Synthesis from Saxitoxin-Producing Cylindrospermopsis raciborskii. Adv. Microbiol. 2013, 3, 289–296. [Google Scholar] [CrossRef]
- Vilar, M.C.P.; Molica, R.J.R. Changes in pH and dissolved inorganic carbon in water affect the growth, saxitoxins production and toxicity of the cyanobacterium Raphidiopsis raciborskii ITEP-A1. Harmful Algae 2020, 97, 101870. [Google Scholar] [CrossRef]
- Lorenzi, A.S.; Cordeiro-Araújo, M.K.; Chia, M.A.; Bittencourt-Oliveira, M.C. Cyanotoxin contamination of semiarid drinking water supply reservoirs. Environ. Earth Sci. 2018, 77, 1–8. [Google Scholar] [CrossRef]
- Abreu, V.A.; Popin, R.V.; Alvarenga, D.O.; Schaker, P.D.; Hoff-Risseti, C.; Varani, A.M.; Fiore, M.F. Genomic and genotypic characterization of Cylindrospermopsis raciborskii: Toward an intraspecific phylogenetic evaluation by comparative genomics. Front. Microbiol. 2018, 9, 306. [Google Scholar] [CrossRef]
- Wiese, M.; D’Agostino, P.M.D.; Mihali, T.K.; Moffitt, M.C.; Neilan, B.A. Neurotoxic Alkaloids: Saxitoxin and its Analogs. Mar. Drugs 2010, 8, 2185–2211. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, S.; Zhang, Y.; Lin, L. Paralytic shellfish toxin biosynthesis in cyanobacteria and dinoflagellates: A molecular overview. J. Proteomics 2016, 135, 132–140. [Google Scholar] [CrossRef]
- Perez, S.; Vale, C.; Botana, A.M.; Alonso, E.; Vieytes, M.R.; Botana, L.M. Determination of Toxicity Equivalent Factors for Paralytic Shellfish Toxins by Electrophysiological Measurements in cultured neurons. Chem. Res. Toxicol. 2011, 24, 1153–1157. [Google Scholar] [CrossRef]
- Kellman, R.; Mihali, T.K.; Jeon, Y.J.; Pickford, R.; Pomati, F.; Neilan, B.A. Biosynthetic intermediate analysis and functional homology reveal a saxitoxin gene cluster in Cyanobacteria. Appl. Environ. Microbiol. 2008, 74, 4044–4053. [Google Scholar] [CrossRef]
- Panosso, R.; Lürling, M. Daphnia magna feeding on Cylindrospermopsis raciborskii: The role of food composition, filament length and body size. J. Plankton Res. 2010, 32, 1393–1404. [Google Scholar] [CrossRef]
- Ferrão-Filho, A.S.; Silva, D.A.S.; Oliveira, T.A.; Magalhães, V.F.; Pflugmacher, S.; Silva, E.M. Single and combined effects of microcystin-and saxitoxin-producing cyanobacteria on the fitness and antioxidant defenses of cladocerans. Environ. Toxicol. Chem. 2017, 36, 2689–2697. [Google Scholar] [CrossRef] [PubMed]
- Ferrão-Filho, A.S.; Soares, M.C.S.; Magalhães, V.F.; Azevedo, S.M.F.O. A rapid bioassay for detecting saxitoxins using a Daphnia acute toxicity test. Environ. Pollut. 2010, 158, 2084–2093. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.M.; Ferrão-Filho, A.S.; Azevedo, S.M.F.O. Effects of saxitoxin-and non-saxitoxin-producing strains of the cyanobacterium Cylindrospermopsis raciborskii on the fitness of temperate and tropical cladocerans. Harmful Algae 2013, 28, 55–63. [Google Scholar] [CrossRef]
- Soares, M.C.S.; Lürling, M.; Panosso, R.; Huszar, V. Effects of the cyanobacterium Cylindrospermopsis raciborskii on feeding and life history characteristics of the grazer Daphnia magna. Ecotoxicol. Environ. Saf. 2009, 72, 1183–1189. [Google Scholar] [CrossRef]
- Harke, M.J.; Jankowiak, J.G.; Morrell, B.K.; Gobler, C.J. Transcriptomic responses in the bloom-forming cyanobacterium Microcystis induced during exposure to zooplankton. Appl. Environ. Microbiol. 2017, 83, e02832-16. [Google Scholar] [CrossRef]
- Agrawal, A.A. Phenotypic plasticity in the interactions and evolution of species. Science 2001, 294, 321–326. [Google Scholar] [CrossRef]
- Pavia, H.; Toth, G.B.; Åberg, P. Optimal defense theory: Elasticity analysis as a tool to predict intraplant variation in defenses. Ecology 2002, 83, 891–897. [Google Scholar] [CrossRef]
- Murray, S.A.; Mihali, T.K.; Neilan, B.A. Extraordinary conservation, gene loss, and positive selection in the evolution of an ancient neurotoxin. Mol. Biol. Evol. 2010, 28, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- McKey, D. Adaptive patterns in alkaloid physiology. Am. Nat. 1974, 108, 305–320. [Google Scholar] [CrossRef]
- Van Donk, E.; Lürling, M.; Lampert, W. Consumer induced changes in phytoplankton: Inducibility, costs, benefits, and the impact on grazers. In The Ecology, Evolution of Inducible Defenses; Tollrian, R., Harvell, C.D., Eds.; Princeton University Press: Princeton, NJ, USA, 1999; pp. 89–103. [Google Scholar]
- Akbar, S.; Du, J.; Lin, H.; Kong, X.; Sun, S.; Tian, X. Understanding interactive inducible defenses of Daphnia and its phytoplankton prey. Harmful Algae 2017, 66, 47–56. [Google Scholar] [CrossRef]
- Princiotta, S.D.; Hendricks, S.P.; White, D.S. Production of cyanotoxins by Microcystis aeruginosa mediates interactions with the mixotrophic flagellate Cryptomonas. Toxins 2019, 11, 223. [Google Scholar] [CrossRef]
- Van Gremberghe, I.; Vanormelingen, P.; Van der Gucht, K.; Mancheva, A.; D’hondt, S.; De Meester, L.; Vyverman, W. Influence of Daphnia infochemicals on functional traits of Microcystis strains (Cyanobacteria). Hydrobiologia 2009, 635, 147–155. [Google Scholar] [CrossRef]
- Pineda-Mendoza, R.M.; Zúñiga, G.; Martínez-Jerónimo, F. Infochemicals released by Daphnia magna fed on Microcystis aeruginosa affect mcyA gene expression. Toxicon 2014, 80, 78–86. [Google Scholar] [CrossRef]
- Selander, E.; Fagerberg, T.; Wohlrab, S.; Pavia, H. Fight and flight in dinoflagellates? Kinetics of simultaneous grazer-induced responses in Alexandrium tamarense. Limnol. Oceanogr. 2012, 57, 58–64. [Google Scholar] [CrossRef]
- Senft-Batoh, C.D.; Dam, H.G.; Shumway, S.E.; Wikfors, G.H.; Schlichting, C.D. Influence of predator–prey evolutionary history, chemical alarm-cues, and feeding selection on induction of toxin production in a marine dinoflagellate. Limnol. Oceanogr. 2015, 60, 318–328. [Google Scholar] [CrossRef]
- Senft-Batoh, C.D.; Dam, H.G.; Shumway, S.E.; Wikfors, G.H. A multi-phylum study of grazer-induced paralytic shellfish toxin production in the dinoflagellate Alexandrium fundyense: A new perspective on control of algal toxicity. Harmful Algae 2015, 44, 20–31. [Google Scholar] [CrossRef]
- Van de Waal, D.B.; Smith, V.H.; Declerck, S.A.; Stam, E.C.; Elser, J.J. Stoichiometric regulation of phytoplankton toxins. Ecol. Lett. 2014, 17, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Griffin, J.E.; Park, G.; Dam, H.G. Relative importance of nitrogen sources, algal alarm cues and grazer exposure to toxin production of the marine dinoflagellate Alexandrium catenella. Harmful Algae 2019, 84, 181–187. [Google Scholar] [CrossRef]
- Mihali, T.K.; Kellmann, R.; Neilan, B.A. Characterization of the paralytic shellfish toxin biosynthesis gene clusters in Anabaena circinalis AWQC131C and Aphanizomenon sp. NH-5. BMC Biochem. 2009, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Ibelings, B.W.; Havens, K.E. Cyanobacterial toxins: A qualitative meta–analysis of concentrations, dosage and effects in freshwater, estuarine and marine biota. In Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs; Springer: New York, NY, USA, 2008; pp. 675–732. [Google Scholar]
- Ferrão-Filho, A.S.; Soares, M.C.S.; Lima, R.S.; Magalhães, V.F. Effects of Cylindrospermopsis raciborskii (Cyanobacteria) on the swimming behavior of Daphnia (Cladocera). Environ. Toxicol. Chem. 2014, 31, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Freitas, E.C.; Pinheiro, C.; Rocha, O.; Loureiro, S. Can mixtures of cyanotoxins represent a risk to the zooplankton? The case study of Daphnia magna Straus exposed to hepatotoxic and neurotoxic cyanobacterial extracts. Harmful Algae 2014, 31, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.; Selander, E.; Pavia, H.; John, U. Grazer-induced toxin formation in dinoflagellates: A transcriptomic model study. Eur. J. Phycol. 2011, 46, 66–73. [Google Scholar] [CrossRef]
- Beamud, G.; Vico, P.; Haakonsson, S.; Martínez de la Escalera, G.; Piccini, C.; Brena, B.; Pírez, M.; Bonilla, S. Influence of UV-B radiation on the fitness and toxin expression of the cyanobacterium Cylindrospermopsis raciborskii. Hydrobiologia 2016, 763, 161–172. [Google Scholar] [CrossRef]
- Vico, P.; Aubriot, L.; Martigani, F.; Rigamonti, N.; Bonilla, S.; Piccini, C. Influence of nitrogen availability on the expression of genes involved in the biosynthesis of saxitoxin and analogs in Cylindrospermopsis raciborskii. Harmful Algae 2016, 56, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Wohlrab, S.; Selander, E.; John, U. Predator cues reduce intraspecific trait variability in a marine dinoflagellate. BMC Ecol. 2017, 17, 8. [Google Scholar] [CrossRef] [PubMed]
- Rantala, A.; Fewer, D.P.; Hisbergues, M.; Rouhiainen, L.; Vaitomaa, J.; Börner, T.; Sivonen, K. Phylogenetic evidence for the early evolution of microcystin synthesis. Proc. Natl. Acad. Sci. USA 2004, 101, 568–573. [Google Scholar] [CrossRef]
- Rzymski, P.; Klimaszyk, P.; Jurczak, T.; Poniedziałek, B. Oxidative stress, programmed cell death and microcystin release in Microcystis aeruginosa in response to Daphnia grazers. Front. Microbiol. 2020, 11, 1201. [Google Scholar] [CrossRef]
- Kenesi, G.; Shafik, H.; Kovacs, A.; Herodek, S.; Presing, M. Effect of nitrogen forms on growth, cell composition and N2 fixation of Cylindrospermopsis raciborskii in phosphorus-limited chemostat cultures. Hydrobiologia 2009, 623, 191–202. [Google Scholar] [CrossRef]
- Nor, N.H.M.; Te, S.H.; Mowe, M.A.D.; Gin, K.Y.H. Environmental factors influence cylindrospermopsin production of Cylindrospermopsis raciborskii (CR12). J. Plankton Res. 2019, 41, 114–126. [Google Scholar]
- Stucken, K.; John, U.; Cembella, A.; Soto-Liebe, K.; Vásquez, M. Impact of nitrogen sources on gene expression and toxin production in the diazotroph Cylindrospermopsis raciborskii CS-505 and non-diazotroph Raphidiopsis brookii D9. Toxins 2014, 6, 1896–1915. [Google Scholar] [CrossRef]
- Blossom, H.E.; Markussen, B.; Daugbjerg, N.; Krock, B.; Norlin, A.; Hansen, P.J. The cost of toxicity in microalgae: Direct evidence from the dinoflagellate Alexandrium spp. Front. Microbiol. 2019, 10, 1065. [Google Scholar] [CrossRef] [PubMed]
- Orr, P.T.; Jones, G.J. Relationship between microcystin production and cell division rates in nitrogen-limited Microcystis aeruginosa cultures. Limnol. Oceanogr. 1998, 43, 1604–1614. [Google Scholar] [CrossRef]
- Orr, P.T.; Willis, A.; Burford, M.A. Application of first order rate kinetics to explain changes in bloom toxicity—the importance of understanding cell toxin quotas. J. Ocenanol. Limnol. 2018, 36, 1063–1074. [Google Scholar] [CrossRef]
- Jähnichen, S.; Ihle, T.; Petzoldt, T. Variability of microcystin cell quota: A small model explains dynamics and equilibria. Limnologica 2008, 38, 339–349. [Google Scholar] [CrossRef]
- Bojadzija Savic, G.; Edwards, C.; Briand, E.; Lawton, L.; Wiegand, C.; Bormans, M. Daphnia magna Exudates Impact Physiological and Metabolic Changes in Microcystis aeruginosa. Toxins 2019, 11, 421. [Google Scholar] [CrossRef] [PubMed]
- Bojadzija Savic, G.; Bormans, M.; Edwards, C.; Lawton, L.; Briand, E.; Wiegand, C. Cross talk: Two-way allelopathic interactions between toxic Microcystis and Daphnia. Harmful Algae 2020, 94, 101803. [Google Scholar] [CrossRef] [PubMed]
- Cerbin, S.; Wejnerowski, Ł.; Dziuba, M. Aphanizomenon gracile increases in width in the presence of Daphnia. A defence mechanism against grazing? J. Limnol. 2013, 72, e41. [Google Scholar] [CrossRef]
- Wejnerowski, L.; Cerbin, S.; Dziuba, M.K. Thicker filaments of Aphanizomenon gracile are more harmful to Daphnia than thinner Cylindrospermopsis raciborskii. Zool. Stud. 2015, 54, 2. [Google Scholar] [CrossRef]
- Bergkvist, J.; Thor, P.; Jakobsen, H.H.; Wängberg, S.Å.; Selander, E. Grazer-induced chain length plasticity reduces grazing risk in a marine diatom. Limnol. Oceanogr. 2012, 57, 318–324. [Google Scholar] [CrossRef]
- Bjærke, O.; Jonsson, P.R.; Alam, A.; Selander, E. Is chain length in phytoplankton regulated to evade predation? J. Plankton Res. 2015, 37, 1110–1119. [Google Scholar] [CrossRef]
- Selander, E.; Jakobsen, H.H.; Lombard, F.; Kiørboe, T. Grazer cues induce stealth behavior in marine dinoflagellates. Proc. Natl. Acad. Sci. USA 2011, 108, 4030–4034. [Google Scholar] [CrossRef]
- Selander, E.; Berglund, E.C.; Engström, P.; Berggren, F.; Eklund, J.; Harðardóttir, S.; Lundholm, N.; Grebner, W.; Andersson, M.X. Copepods drive large-scale trait-mediated effects in marine plankton. Sci. Adv. 2019, 5, eaat5096. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.A. Induced responses to herbivory and increased plant performance. Science 1998, 279, 1201–1202. [Google Scholar] [CrossRef]
- Agrawal, A.A.; Laforsch, C.; Tollrian, R. Transgenerational induction of defences in animals and plants. Nature 1999, 401, 2. [Google Scholar] [CrossRef]
- Pančić, M.; Kiørboe, T. Phytoplankton defence mechanisms: Traits and trade-offs. Biol. Rev. 2018, 93, 1269–1303. [Google Scholar] [CrossRef]
- Cadotte, M.W.; Tucker, C.M. Should environmental filtering be abandoned? Trends Ecol. Evol. 2017, 32, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Fileto, C.; Arcifa, M.S.; Marchetti, J.M.; Turati, I.C.; Lopes, N.P. Influence of biochemical, mineral and morphological features of natural food on tropical cladocerans. Aquatic Ecol. 2007, 41, 557–568. [Google Scholar] [CrossRef]
- Soares, M.C.S.; Rocha, M.I.D.A.; Marinho, M.M.; Azevedo, S.M.; Branco, C.W.; Huszar, V.L. Changes in species composition during annual cyanobacterial dominance in a tropical reservoir: Physical factors, nutrients and grazing effects. Aquat. Microb. Ecol. 2009, 57, 137–149. [Google Scholar] [CrossRef]
- Zamora-Barrios, C.A.; Nandini, S.; Sarma, S.S.S. Bioaccumulation of microcystins in seston, zooplankton and fish: A case study in Lake Zumpango, Mexico. Environ. Pollut. 2019, 249, 267–276. [Google Scholar] [CrossRef]
- D’Agostino, P.M.; Boundy, M.J.; Harwood, T.D.; Carmichael, W.W.; Neilan, B.A.; Wood, S.A. Re-evaluation of paralytic shellfish toxin profiles in cyanobacteria using hydrophilic interaction liquid chromatography-tandem mass spectrometry. Toxicon 2019, 158, 1–7. [Google Scholar] [CrossRef]
- Gorham, P.R.; McLachlan, J.; Hammer, U.T.; Kim, W.K. Isolation and culture of toxic strains of Anabaena flos-aquae (Lyngb.) de Breäb. Verh. Int. Ver. Theor. Angew. Limnol. 1964, 15, 796. [Google Scholar]
- Tollrian, R. Neckteeth formation in Daphnia pulex as an example of continuous phenotypic plasticity: Morphological effects of Chaoborus kairomone concentration and their quantification. J. Plankton Res. 1993, 15, 1309–1318. [Google Scholar] [CrossRef]
- Hillebrand, H.; Dürselen, C.D.; Kirshtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- Sun, J.; Liu, D. Geometric models for calculating cell biovolume and surface area for phytoplankton. J. Plankton Res. 2003, 25, 1331–1346. [Google Scholar] [CrossRef]
- Oshima, Y. Postcolumn derivatization liquid chromatographic method for paralytic shellfish toxins. J. AOAC Int. 1995, 78, 528–532. [Google Scholar] [CrossRef]
- FAO/WHO. Technical Paper on Toxicity Equivalency Factors for Marine Biotoxins Associated with Bivalve Molluscs; Food and Agriculture Organization/World Health Organization: Rome, Italy, 2016; 108p. [Google Scholar]
- Martínez De La Escalera, G.; Antoniades, D.; Bonilla, S.; Piccini, C. Application of ancient DNA to the reconstruction of past microbial assemblages and for the detection of toxic cyanobacteria in subtropical freshwater ecosystems. Mol. Ecol. 2014, 23, 5791–5802. [Google Scholar] [CrossRef] [PubMed]
- Willis, A.; Chuang, A.; Dyrhman, S.; Burford, M.A. Differential expression of phosphorus acquisition genes in responses to phosphorus stress in two Raphidiopsis raciborskii strains. Harmful Algae 2019, 82, 19–25. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]

| day | Yield | ETRmax | Ik | Alpha | ||||
|---|---|---|---|---|---|---|---|---|
| (Relative Fv’/Fm’) | (µmol e m−2 s−1) | (µmol photon m−2 s−1) | (µmol photon m−2 s−1) | |||||
| CTRL 1 | INFO 1 | CTRL | INFO | CTRL | INFO | CTRL | INFO | |
| 0 | 0.48 ± 0.0 a | 0.48 ± 0.0 a | 81.80 ± 0.0 a | 81.80 ± 0.0 a | 353.10 ± 0.0 a | 353.10 ± 0.0 a | 0.23 ± 0.0 a | 0.22 ± 0.0 a |
| 2 | 0.59 ± 0.0 a | 0.48 ± 0.1 a | 104.07 ± 16.5 a | 77.70 ± 31.4 a | 429.23 ± 66.7 a | 359.10 ± 69.3 a | 0.23 ± 0.0 a | 0.23 ± 0.0 a |
| 4 | 0.55 ± 0.0 a | 0.53 ± 0.0 a | 103.27 ± 15.3 a | 102.47 ± 8.1 a | 448.03 ± 56.9 a | 440.23 ± 36.2 a | 0.24 ± 0.0 a | 0.23 ± 0.0 a |
| 6 | 0.52 ± 0.0 a | 0.51 ± 0.0 a | 118.27 ± 5.7 a | 109.77 ± 6.7 a | 531.03 ± 41.9 a | 514.60 ± 41.9 a | 0.22 ± 0.0 a | 0.23 ± 0.0 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilar, M.C.P.; Rodrigues, T.F.C.P.; Silva, L.O.; Pacheco, A.B.F.; Ferrão-Filho, A.S.; Azevedo, S.M.F.O. Ecophysiological Aspects and sxt Genes Expression Underlying Induced Chemical Defense in STX-Producing Raphidiopsis raciborskii (Cyanobacteria) against the Zooplankter Daphnia gessneri. Toxins 2021, 13, 406. https://doi.org/10.3390/toxins13060406
Vilar MCP, Rodrigues TFCP, Silva LO, Pacheco ABF, Ferrão-Filho AS, Azevedo SMFO. Ecophysiological Aspects and sxt Genes Expression Underlying Induced Chemical Defense in STX-Producing Raphidiopsis raciborskii (Cyanobacteria) against the Zooplankter Daphnia gessneri. Toxins. 2021; 13(6):406. https://doi.org/10.3390/toxins13060406
Chicago/Turabian StyleVilar, Mauro C. P., Thiago F. C. P. Rodrigues, Luan O. Silva, Ana Beatriz F. Pacheco, Aloysio S. Ferrão-Filho, and Sandra M. F. O. Azevedo. 2021. "Ecophysiological Aspects and sxt Genes Expression Underlying Induced Chemical Defense in STX-Producing Raphidiopsis raciborskii (Cyanobacteria) against the Zooplankter Daphnia gessneri" Toxins 13, no. 6: 406. https://doi.org/10.3390/toxins13060406
APA StyleVilar, M. C. P., Rodrigues, T. F. C. P., Silva, L. O., Pacheco, A. B. F., Ferrão-Filho, A. S., & Azevedo, S. M. F. O. (2021). Ecophysiological Aspects and sxt Genes Expression Underlying Induced Chemical Defense in STX-Producing Raphidiopsis raciborskii (Cyanobacteria) against the Zooplankter Daphnia gessneri. Toxins, 13(6), 406. https://doi.org/10.3390/toxins13060406









