Risky Business: The Function of Play in a Venomous Mammal—The Javan Slow Loris (Nycticebus javanicus)
Abstract
1. Introduction
2. Results
2.1. Play Partners
2.2. Play Patterns
2.3. Postures during Play
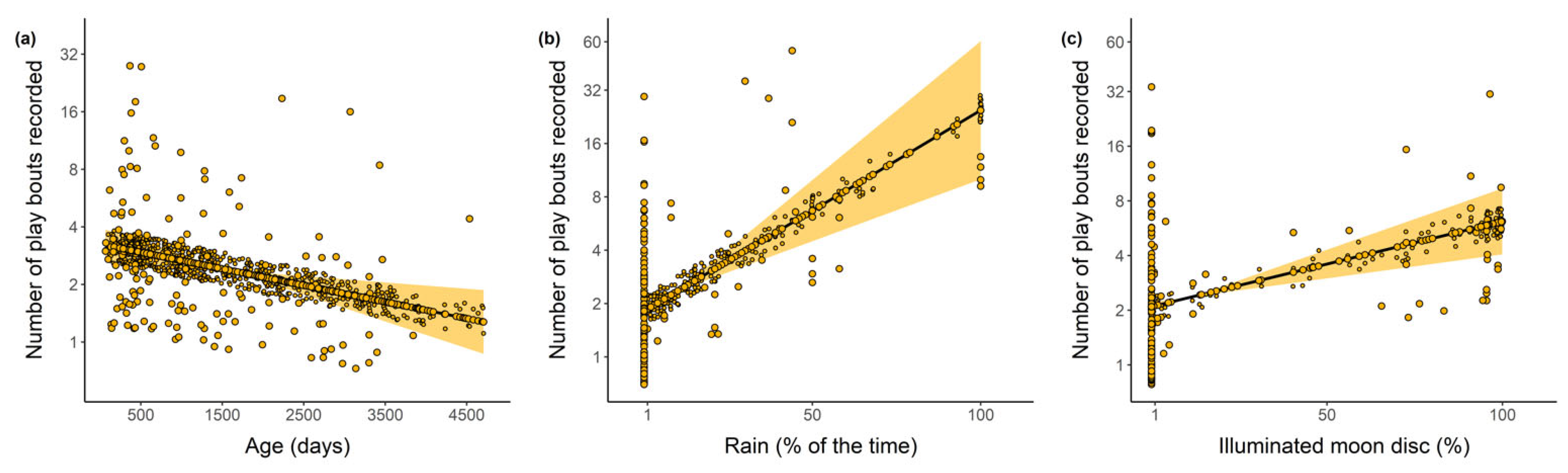
2.4. Rain and Moonlight during Play
3. Discussion
3.1. Play Partners
3.2. Play Patterns
3.3. Postural Behaviour during Play
3.4. Risky Behaviours during Play
4. Conclusions
5. Materials and Methods
5.1. Ethics Statement
5.2. Study Site
5.3. Observation Methods
5.4. Play Observations
5.5. Data Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| ID | Sex | Age (Range in Days) | Observation Time (h) | Instantaneous Sample Points of Play |
|---|---|---|---|---|
| acil | M | 1708–2593 | 143 | 17 |
| alomah | M | 731–1589 | 209 | 52 |
| azka | M | 1934–3655 | 109 | 15 |
| charlie | F | 1963–2529 | 31 | 8 |
| dali | M | 375–483 | 10 | 1 |
| dempak | M | 905–1039 | 33 | 1 |
| dindi | M | 784–1393 | 223 | 0 |
| ena | F | 2160–3644 | 98 | 9 |
| endor | F | 186 | 1 | 5 |
| fernando | M | 1280–3578 | 448 | 65 |
| galaksi | F | 131–457 | 7 | 7 |
| ghee | F | 192–297 | 29 | 5 |
| guntur | M | 904–1139 | 39 | 13 |
| indomi | F | 453–538 | 48 | 0 |
| jaimi | M | 406–896 | 226 | 2 |
| jogja | M | 1934–2004 | 10 | 2 |
| lalit | M | 133–558 | 193 | 131 |
| lava | F | 108–210 | 2 | 2 |
| ln | M | 211–770 | 261 | 39 |
| loopi | F | 172–981 | 115 | 36 |
| lucu | F | 193–3139 | 572 | 80 |
| lupak | F | 79–1585 | 316 | 99 |
| maaf | M | 361–541 | 34 | 17 |
| maya | F | 530–1411 | 110 | 26 |
| mikio | M | 67–1128 | 46 | 19 |
| mimi | F | 839–1535 | 250 | 2 |
| mo | M | 724–1277 | 34 | 2 |
| mungkin | M | 126–787 | 112 | 34 |
| oneeye | F | 1999–4013 | 154 | 14 |
| oniks | M | 274 | 1 | 1 |
| opal | F | 100–118 | 3 | 8 |
| pakbulan | M | 1151 | 2 | 1 |
| rufio | M | 654–1993 | 355 | 99 |
| sempurna | F | 290–897 | 38 | 5 |
| shakti | F | 223–616 | 109 | 78 |
| shanti | M | 286–542 | 120 | 50 |
| shirley | F | 1930–4707 | 419 | 17 |
| sibau | F | 812–2285 | 63 | 8 |
| sirious | M | 71 | 1 | 7 |
| smol | F | 352–514 | 84 | 20 |
| solo | M | 173–726 | 94 | 52 |
| sri | M | 258 | 1 | 1 |
| stan | M | 130 | 1 | 1 |
| star | F | 348–535 | 139 | 76 |
| tahini | F | 170–596 | 5 | 12 |
| tereh | F | 1461–3609 | 236 | 28 |
| timone | F | 292 | 2 | 1 |
| tombol | F | 209–943 | 215 | 70 |
| toyib | M | 927–2399 | 72 | 6 |
| tyrion | M | 188 | 2 | 1 |
| tzatziki | M | 161–538 | 53 | 3 |
| utari | M | 196–317 | 3 | 2 |
| xandie | M | 164–320 | 34 | 8 |
| xena | F | 1282–2077 | 84 | 15 |
| xerxes | M | 569–664 | 38 | 0 |
| yogi | M | 320–575 | 7 | 8 |
| TOTAL | 25 F, 31 M | 67–4707 | 6040 | 1283 |
References
- Andersson, M. Sexual Selection; Princeton University Press: Princeton, NJ, USA, 1994. [Google Scholar]
- Emlen, D.J. The evolution of animal weapons. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 387–413. [Google Scholar] [CrossRef]
- Miller, C.W. Sexual selection/male-male competition. In The Princeton Guide to Evolution; Losos, J., Ed.; Princeton University Press: Princeton, NJ, USA, 2013; pp. 641–646. [Google Scholar]
- Tinbergen, N. On aims and methods of ethology. Z. Tierpsychol. 1963, 20, 410–433. [Google Scholar] [CrossRef]
- Bekoff, M. The development of social interaction, play, and metacommunication in mammals: An ethological perspective. Q. Rev. Biol. 1972, 47, 412–434. [Google Scholar] [CrossRef]
- Poirier, F.E.; Smith, E.O. Socializing functions of primate play. Am. Zool. 1974, 14, 275–287. [Google Scholar] [CrossRef]
- Byers, J.A. Play partner preferences in Siberian ibex, Capra ibex sibirica. Z. Tierpsychol. 1980, 53, 23–40. [Google Scholar] [CrossRef]
- Graham, K.L. Play. In The International Encyclopaedia of Primatology; Fuentes, A., Ed.; John Wiley & Sons Inc.: Chicester, UK, 2017; pp. 1–8. [Google Scholar] [CrossRef]
- Shadle, A.R. The play of American porcupines (Erethizon d. dorsatum and E. epixanthum). J. Comp. Psychol. 1944, 37, 145–150. [Google Scholar] [CrossRef]
- Lee, P.C.; Moss, C.J. African elephant play, competence and social complexity. Anim. Behav. Cogn. 2014, 1, 144–156. [Google Scholar] [CrossRef]
- Nekaris, K.A.I.; Campera, M.; Nijman, V.; Birot, H.; Rode-Margono, E.J.; Fry, B.G.; Weldon, A.; Wirdateti, W.; Imron, M.A. Slow lorises use venom as a weapon in intraspecific competition. Curr. Biol. 2020, 30, R1252–R1253. [Google Scholar] [CrossRef]
- Ligabue-Braun, R.; Verli, H.; Carlini, C.R. Venomous mammals: A review. Toxicon 2012, 59, 680–695. [Google Scholar] [CrossRef] [PubMed]
- Burghardt, G.M. The Genesis of Animal Play; MIT Press: Cambridge, MA, USA, 2005. [Google Scholar]
- Park, S.R. Observation on the behavioral development of the common vampire bat Desmodus rotundus-Play Behaviors. J. Mammal. Soc. Jpn. 1990, 15, 25–32. [Google Scholar] [CrossRef]
- Graham, K.L.; Burghardt, G.M. Current perspectives on the biological study of play: Signs of progress. Q. Rev. Biol. 2010, 85, 393–418. [Google Scholar] [CrossRef]
- Cordoni, G.; Palagi, E. Ontogenetic trajectories of chimpanzee social play: Similarities with humans. PLoS ONE 2011, 6, e27344. [Google Scholar] [CrossRef] [PubMed]
- Cheney, D.L. The play partners of immature baboons. Anim. Behav. 1978, 26, 1038–1050. [Google Scholar] [CrossRef]
- Paquette, D. Fighting and playfighting in captive adolescent chimpanzees. Aggress. Behav. 1994, 20, 49–65. [Google Scholar] [CrossRef]
- Owens, N. Social play behaviour in free-living baboons. Papio anubis. Anim. Behav. 1975, 23, 387–408. [Google Scholar] [CrossRef]
- Byers, J.A.; Walker, C. Refining the motor training hypothesis for the evolution of play. Am. Nat. 1995, 146, 25–40. [Google Scholar] [CrossRef]
- Pfeifer, S. Sex differences in social play of Scimitar-horned oryx calves (Oryx dammah). Z. Tierpsychol. 1985, 69, 281–292. [Google Scholar] [CrossRef]
- Gomendio, M. The development of different types of play in gazelles: Implications for the nature and functions of play. Anim. Behav. 1988, 36, 825–836. [Google Scholar] [CrossRef]
- Hass, C.C.; Jenni, D.A. Social play among juvenile bighorn sheep: Structure, development, and relationship to adult behavior. Ethology 1993, 93, 105–116. [Google Scholar] [CrossRef]
- Smith, P.K. Does play matter? Functional and evolutionary aspects of animal and human play. Behav. Brain Sci. 1982, 5, 139–155. [Google Scholar] [CrossRef]
- Pellis, S.M.; Pellis, V.C.; Bell, H.C. The function of play in the development of the social brain. Am. J. Play 2010, 2, 278–296. [Google Scholar]
- Thompson, K.V. Play-partner preferences and the function of social play in infant sable antelope. Hippotragus niger. Anim. Behav. 1996, 52, 1143–1155. [Google Scholar] [CrossRef]
- Pellis, S.M.; Pellis, S.M. What is play fighting and what is it good for? Learn. Behav. 2017, 45, 355–366. [Google Scholar] [CrossRef]
- Špinka, M.; Newberry, R.C.; Bekoff, M. Mammalian play: Training for the unexpected. Q. Rev. Biol. 2001, 76, 141–168. [Google Scholar] [CrossRef] [PubMed]
- Petrů, M.; Špinka, M.; Charvátová, V.; Lhota, S. Revisiting play elements and self-handicapping in play: A comparative ethogram of five old world monkey species. J. Comp. Psychol. 2009, 123, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Carter, R.N.; Romanow, C.A.; Pellis, S.M.; Lingle, S. Play for prey: Do deer fawns play to develop species-typical antipredator tactics or to prepare for the unexpected? Anim. Behav. 2019, 156, 31–40. [Google Scholar] [CrossRef]
- Byers, J.A. Terrain preferences in the play behaviour of Siberian ibex kids (Capra ibex sibirica). Z. Tierpsychol. 1977, 45, 199–209. [Google Scholar] [CrossRef]
- Nekaris, K.A.I.; Weldon, A.; Imron, M.A.; Maynard, K.Q.; Nijman, V.; Poindexter, S.A.; Morcatty, T.Q. Venom in furs: Facial masks as aposematic signals in a venomous mammal. Toxins 2019, 11, 93. [Google Scholar] [CrossRef]
- Nekaris, K.A.I.; Moore, R.S.; Rose, J.; Fry, B.G. Mad, bad and dangerous to know: The biochemistry, ecology and evolution of slow loris venom. J. Venom. Anim. Toxins Incl. Trop. Dis. 2013, 19, 21. [Google Scholar] [CrossRef]
- Madani, G.; Nekaris, K.A.I. Anaphylactic shock following the bite of a wild Kayan slow loris (Nycticebus kayan): Implications for slow loris conservation. J. Venom. Anim. Toxins Incl. Trop. Dis. 2014, 20, 43. [Google Scholar] [CrossRef]
- Campera, M.; Brown, E.; Imron, M.A.; Nekaris, K.A.I. Unmonitored releases of small animals? The importance of considering natural dispersal, health, and human habituation when releasing a territorial mammal threatened by wildlife trade. Biol. Conserv. 2020, 242, 108404. [Google Scholar] [CrossRef]
- Rotundo, M.; Fernandez-Duque, E.; Dixson, A.F. Infant development and parental care in free-ranging Aotus azarai azarai in Argentina. Int. J. Primatol. 2005, 26, 1459–1473. [Google Scholar] [CrossRef]
- Nekaris, K.A.I. Social lives of adult Mysore slender lorises (Loris lydekkerianus lydekkerianus). Am. J. Primatol. 2006, 68, 1171–1182. [Google Scholar] [CrossRef]
- Řeháková, M. Preliminary observations of infant ontogeny in the Philippine tarsier (Tarsius syrichta) and the first description of play behaviour and its ontogeny in tarsiers. In Primates; Burke, M., Ptito, M., Eds.; IntechOpen: Rijeka, Croatia, 2018; pp. 29–44. [Google Scholar]
- Ehrlich, A.; Musicant, A. Social and individual behaviors in captive slow lorises. Behaviour 1977, 60, 195–219. [Google Scholar] [CrossRef]
- Fitch-Synder, H.; Ehrlich, A. Mother-infant interactions in slow lorises (Nycticebus bengalensis) and pygmy lorises (N. pygmaeus). Folia Primatol. 2003, 74, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Lonsdorf, E.V. Sex differences in nonhuman primate behavioral development. J. Neurosci. Res. 2017, 95, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Prugh, L.R.; Golden, C.D. Does moonlight increase predation risk? Meta-analysis divergent responses of nocturnal mammals to lunar cycles. J. Anim. Ecol. 2014, 83, 504–514. [Google Scholar] [CrossRef]
- Rode-Margono, E.J.; Nekaris, K.A.I. Impact of climate and moonlight on a venomous mammal, the Javan slow loris (Nycticebus javanicus Geoffroy, 1812). Contrib. Zool. 2014, 83, 217–225. [Google Scholar] [CrossRef]
- Dapporto, L.; Turillazzi, S.; Palagi, E. Dominance interactions in young adult paper wasp (Polistes dominulus) foundresses: A playlike behavior? J. Comp. Psychol. 2006, 120, 394. [Google Scholar] [CrossRef]
- Paquette, D. Theorizing the father-child relationship: Mechanisms and developmental outcomes. Hum. Dev. 2004, 47, 193–219. [Google Scholar] [CrossRef]
- Henry, J.D.; Herrero, S.M. Social play in the American black bear: Its similarity to canid social play and an examination of its identifying characteristics. Am. Zool. 1974, 14, 371–389. [Google Scholar] [CrossRef]
- Poindexter, S.A.; Nekaris, K.A.I. Vertical clingers and gougers: Rapid acquisition of adult limb proportions facilitates feeding behaviours in young Javan slow lorises (Nycticebus javanicus). Mamm. Biol. 2017, 87, 40–49. [Google Scholar] [CrossRef]
- Bezanson, M. Primate positional behavior development and evolution. Annu. Rev. Anthropol. 2017, 46, 279–298. [Google Scholar] [CrossRef]
- Bezanson, M. The Ontogeny of Prehensile-Tail Use in Cebus capucinus and Alouatta palliata. Am. J. Primatol. 2012, 74, 770–782. [Google Scholar] [CrossRef]
- Fontaine, R.P. Play as physical flexibility training in five ceboid primates. J. Comp. Psychol. 1994, 108, 203–212. [Google Scholar] [CrossRef]
- Lammers, A.R. The effects of substrate texture on the mechanics of quadrupedal arboreal locomotion in the gray short-tailed opossum (Monodelphis domestica). J. Exp. Zool. 2009, 311, 813–823. [Google Scholar] [CrossRef]
- Starr, C.; Nekaris, K.A.I.; Leung, L. Hiding from the moonlight: Luminosity and temperature affect activity of Asian nocturnal primates in a highly seasonal forest. PLoS ONE 2012, 7, e36396. [Google Scholar] [CrossRef]
- Rode-Margono, E.J.; Nijman, V.; Wirdateti; Nekaris, K.A.I. Ethology of the Critically Endangered Javan slow loris Nycticebus javanicus E. Geoffroy Saint-Hilaire in West Java. Asian Primates J. 2014, 4, 27–41. [Google Scholar]
- Altmann, J. Observational study of behavior: Sampling methods. Behaviour 1974, 49, 227–266. [Google Scholar] [CrossRef] [PubMed]
- Bastian, M.; Heymann, S.; Jacomy, M. Gephi: An open source software for exploring and manipulating networks. ICWSM 2009, 8, 361–362. [Google Scholar]
- Farine, D.R.; Whitehead, H. Constructing, conducting and interpreting animal social network analysis. J. Anim. Ecol. 2015, 84, 1144–1163. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.N. Generalized Additive Models: An Introduction with R, 2nd ed.; Chapman and Hall/CRC: Boca Raton, FL, USA, 2017. [Google Scholar]
- Wood, S.N. Low rank scale invariant tensor product smooths for generalized additive mixed models. Biometrics 2006, 62, 1025–1036. [Google Scholar] [CrossRef] [PubMed]
- Burnham, K.P.; Anderson, D.R. Multimodel inference: Understanding AIC and BIC in model selection. Sociol. Methods Res. 2004, 33, 261–304. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: https://www.R-project.org/ (accessed on 10 December 2020).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, 2016. [Google Scholar]


| Response Variable | Predictor | Estimate | Std. Error | t-Value | p | Smooth Term | p | |
|---|---|---|---|---|---|---|---|---|
| Edf | F-Value | |||||||
| Horizontal suspension | Intercept | 1.467 | 0.219 | 6.7 | <0.001 * | |||
| Adult male—offspring | −0.177 | 0.254 | −2.5 | 0.485 | ||||
| Mother—offspring | −0.785 | 0.261 | −3.0 | 0.003 * | ||||
| Other | −0.923 | 0.319 | −2.9 | 0.004* | ||||
| (Age) | 1.000 | 11.4 | <0.001 * | |||||
| Non-suspensory | Intercept | −2.041 | 0.271 | −7.5 | <0.001 * | |||
| Adult male—offspring | −0.245 | 0.331 | −0.7 | 0.460 | ||||
| Mother—offspring | 0.479 | 0.328 | 1.5 | 0.144 | ||||
| Other | 1.407 | 0.368 | 3.8 | <0.001 * | ||||
| (Age) | 1.748 | 13.3 | <0.001 * | |||||
| Vertical suspension | Intercept | −2.897 | 0.347 | −8.3 | <0.001 * | |||
| Adult male ‒offspring | 0.608 | 0.368 | 1.7 | 0.099 | ||||
| Mother—offspring | 1.021 | 0.382 | 2.7 | 0.008 * | ||||
| Other | −1.954 | 1.080 | −1.8 | 0.071 | ||||
| (Age) | 1.943 | 2.5 | 0.081 | |||||
| Response Variable a | Predictor Variables | Estimate | Std. Error | t-Value | p |
|---|---|---|---|---|---|
| Frequency of play | Intercept | 1.091 | 0.109 | 10.1 | <0.001 * |
| N observations (effort control) | 0.005 | 0.002 | −2.3 | 0.03 * | |
| Rain | 0.026 | 0.006 | 4.2 | <0.001 * | |
| Moonlight | 0.012 | 0.003 | 3.9 | <0.001 * | |
| Age | −0.0002 | 0.00001 | 96.6 | <0.001 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barrett, M.; Campera, M.; Morcatty, T.Q.; Weldon, A.V.; Hedger, K.; Maynard, K.Q.; Imron, M.A.; Nekaris, K.A.I. Risky Business: The Function of Play in a Venomous Mammal—The Javan Slow Loris (Nycticebus javanicus). Toxins 2021, 13, 318. https://doi.org/10.3390/toxins13050318
Barrett M, Campera M, Morcatty TQ, Weldon AV, Hedger K, Maynard KQ, Imron MA, Nekaris KAI. Risky Business: The Function of Play in a Venomous Mammal—The Javan Slow Loris (Nycticebus javanicus). Toxins. 2021; 13(5):318. https://doi.org/10.3390/toxins13050318
Chicago/Turabian StyleBarrett, Meg, Marco Campera, Thais Q. Morcatty, Ariana V. Weldon, Katherine Hedger, Keely Q. Maynard, Muhammad Ali Imron, and K. A. I. Nekaris. 2021. "Risky Business: The Function of Play in a Venomous Mammal—The Javan Slow Loris (Nycticebus javanicus)" Toxins 13, no. 5: 318. https://doi.org/10.3390/toxins13050318
APA StyleBarrett, M., Campera, M., Morcatty, T. Q., Weldon, A. V., Hedger, K., Maynard, K. Q., Imron, M. A., & Nekaris, K. A. I. (2021). Risky Business: The Function of Play in a Venomous Mammal—The Javan Slow Loris (Nycticebus javanicus). Toxins, 13(5), 318. https://doi.org/10.3390/toxins13050318






