Distribution, Ecology, Chemistry and Toxicology of Plant Stinging Hairs
Abstract
1. Introduction
2. Stinging Hairs
2.1. What Are “Stinging Hairs”?
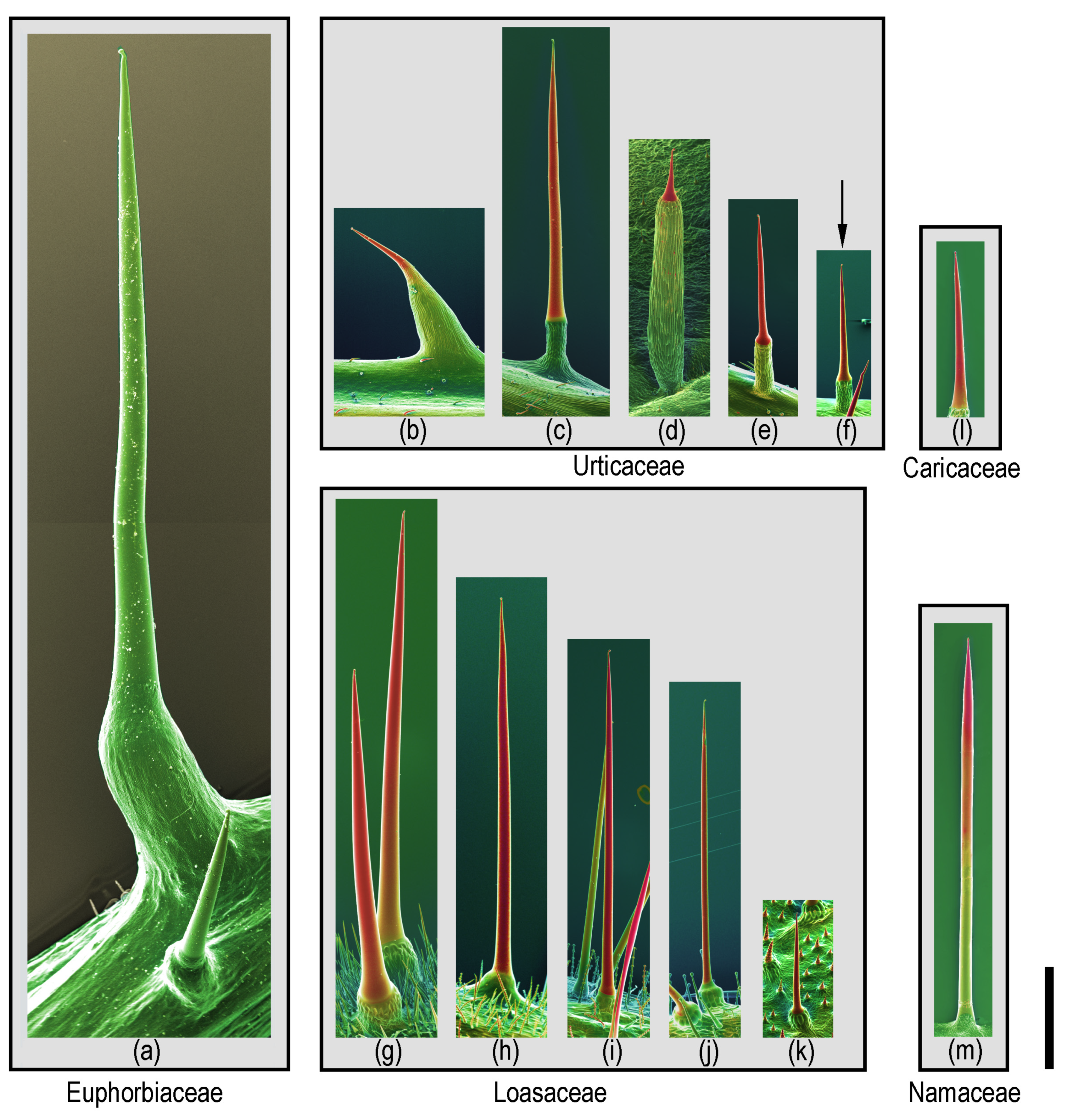
2.2. Distribution of Stinging Hairs in the Plant Kingdom
2.3. Distribution of Stinging Hairs on Plants
2.4. Homology and Ontogeny of Plant Stinging Hairs
2.5. Physiological Effects of Stinging Hairs
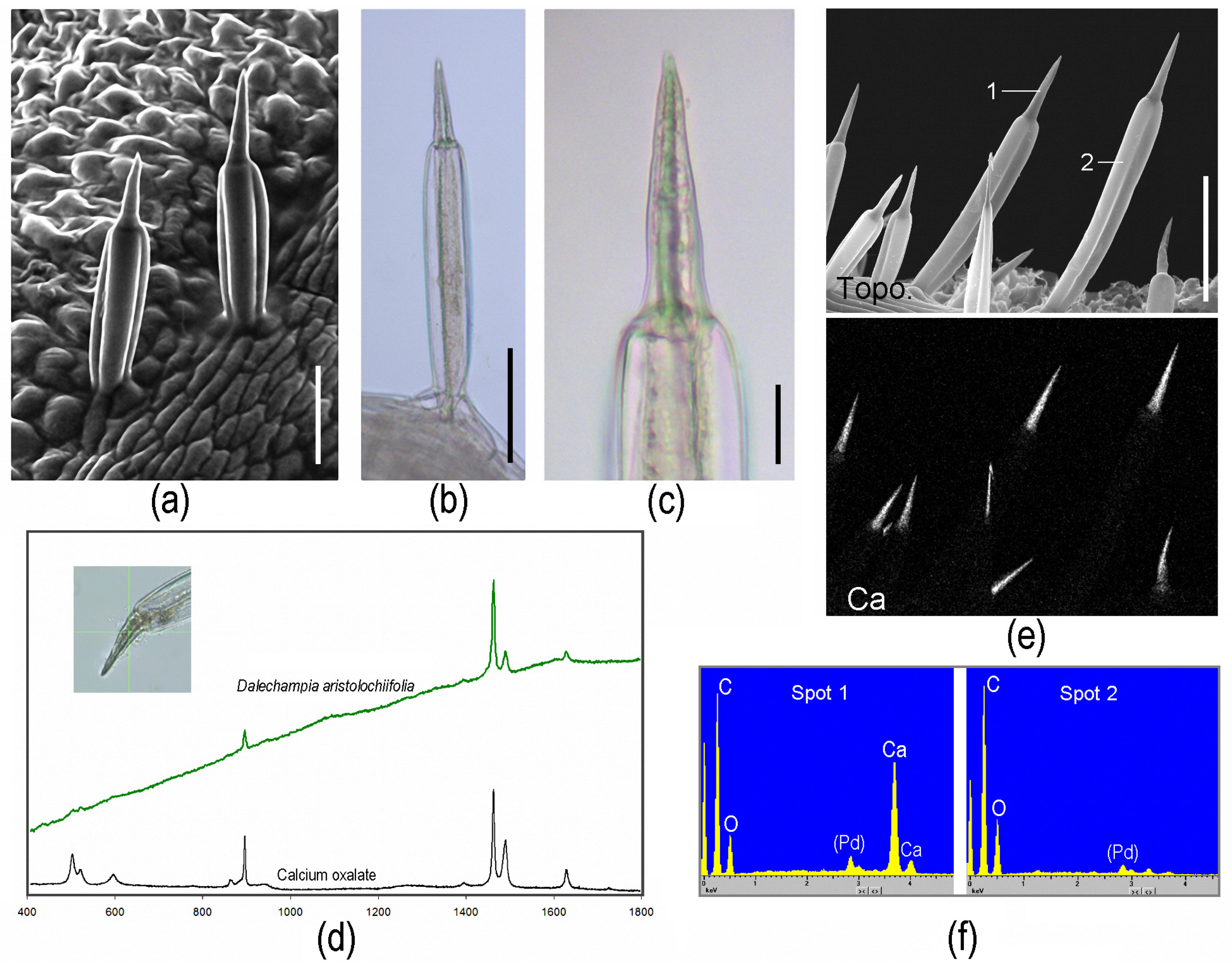
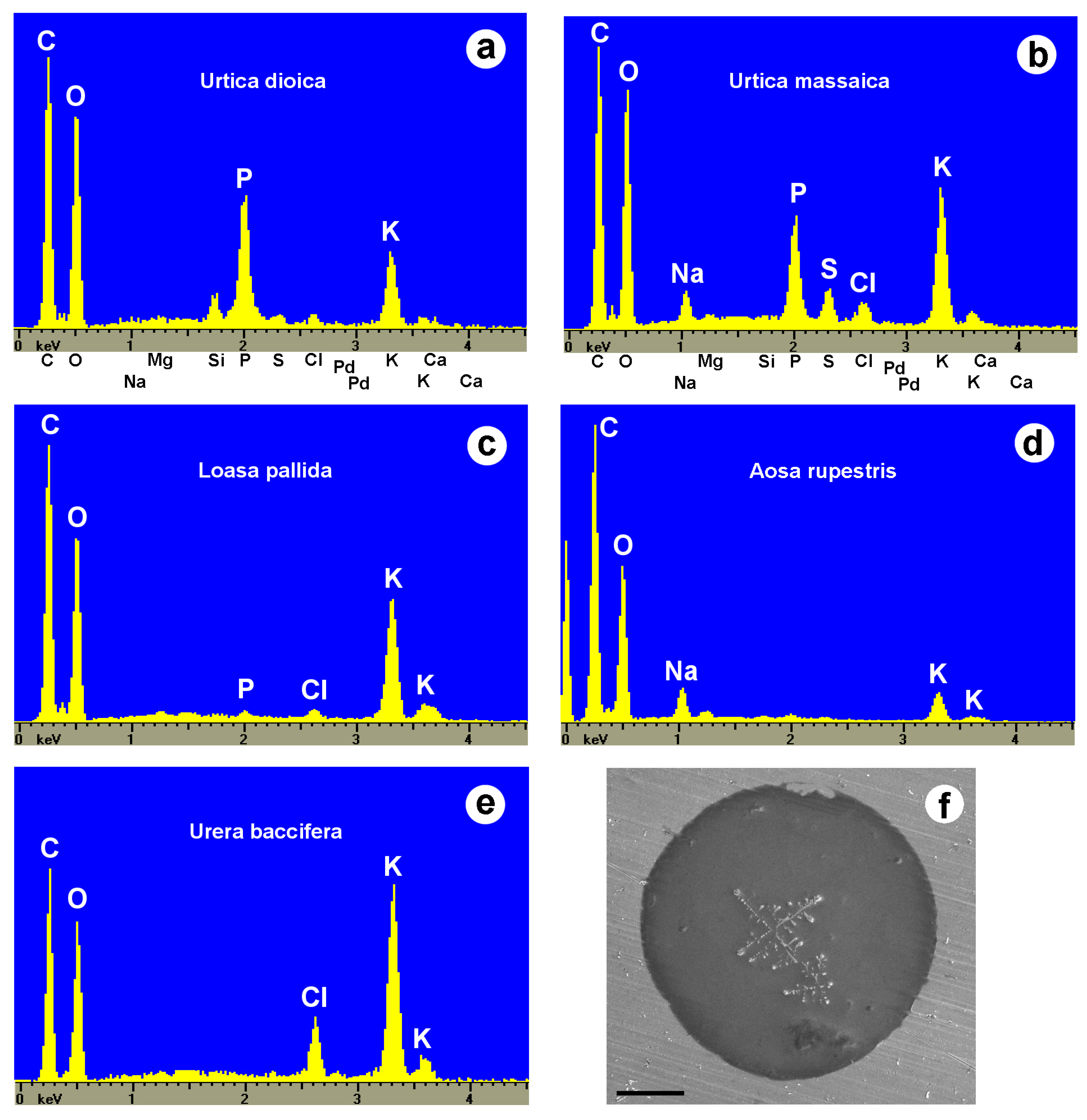
2.6. Toxins of Stinging Hairs
2.7. Ecology of Stinging Plants
3. Conclusions
4. Outlook
5. Materials and Methods
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thurston, E.L.; Lersten, N.R. The morphology and toxicology of plant stinging hairs. Bot. Rev. 1969, 35, 393–412. [Google Scholar] [CrossRef]
- Hooke, R. Micrographia: Or Some Physiological Descriptions of Minute Bodies Made by Magnifying Glasses with Observations and Inquiries thereupon. In John Martyn “Printers to the Royal Society”; Dover Pub. Co.: New York, NY, USA, 1965. [Google Scholar]
- Mustafa, A.; Ensikat, H.J.; Weigend, M. Stinging hair morphology and wall biomineralization across five plant families: Conserved morphology versus divergent cell wall composition. Am. J. Bot. 2018, 105, 1109–1122. [Google Scholar] [CrossRef]
- Gorup-Besanetz, E.F. Notiz über das Vorkommen der Ameisensäure in den Brennnesseln. J. Prakt. Chem. 1849, 48, 191–192. [Google Scholar] [CrossRef]
- Rauter, J. Zur Entwicklungsgeschichte einiger Trichomgebilde. Denkschr. Akad. Wiss. 1872, 31, 2–49. [Google Scholar]
- Bergmann, E. Untersuchungen über das Vorkommen der Ameisensäure und Essigsäure in den Pflanzen und über die physiologische Bedeutung derselben im Stoffwechsel. Bot. Zeit. 1882, 39, 730–754. [Google Scholar]
- Haberlandt, G. Zur Anatomie und Physiologie der pflanzlichen Brennhaare. Sitz. Ber. Akad. Wien 1886, 93, 122–145. [Google Scholar]
- Tassi, E. Strutt. Die pili di acl. sp. di Loasa sp. Siena. In Systematic Anatomy of the Dicotyledons; Soleder, H., Ed.; Trans. LA Boodle: Oxford, UK; Clarendon Press: Oxford, UK, 1908. [Google Scholar]
- Gibson, H.J.H.; Warham, A. Note on the stinging hairs of Urtica Dioica. Biochem. Soc. Proc. 1890, 4, 91–94. [Google Scholar]
- Ritterhausen, P. Anatomisch-Systematische Untersuchung von Blatt und Axe der Acalypheen. Master’s Thesis, University Munich, Munich, Germany, 1892. [Google Scholar]
- Giustiniani, E. Sopra alcuni costituenti dell’ortica. Gazz. Chim. Ital. 1896, 26, 1–7. [Google Scholar]
- Dragendorf, J.G. Heilpflanzen. In Biochemie der Pflanzen; Czapek, F., Ed.; G. Fischer: Jena, Germany, 1905; Volume 1, pp. 1–828. [Google Scholar]
- Knoll, F. Die Brennhaare der Euphorbiaceen-Gattungen Dalechampia und Tragia. Sitz Ber. Kaiserl. Akad. Wiss. 1905, 144, 29–50. [Google Scholar]
- Petrie, J.M. The stinging property of the giant nettle tree (Laportea gigas Wedd.). Proc. Linn. Soc. N. S. W. 1906, 31, 530–545. [Google Scholar]
- Winternitz, R. Einige Bemerkungen über Urticaria arteficialis. Arch. Syph. Dermatol. 1907, 85, 263–270. [Google Scholar] [CrossRef][Green Version]
- Flury, F. Über die Bedeutung der Ameisensäure als natürlich vorkommendes Gift. Deut. Pharm. Ges. 1919, 24, 650–673. [Google Scholar]
- Nestler, A. Zur Kenntnis der Wirkung der Brennhaare unserer Urtica-Arten. Ber. Deutsch. Bot. Ges. 1925, 43, 497–504. [Google Scholar]
- Flury, F. Über den chemischen Nachweis des Nesselgiftes. Zeitschr. Gesamte Exp. Med. 1927, 56, 402–409. [Google Scholar] [CrossRef]
- Kroeber, L. Pharmakochemische Ergebnisse der Untersuchung heimischer Arzneipflanzen. Pharm. Zeit. 1928, 73, 123–126. [Google Scholar]
- Starkenstein, E.; Wasserstrom, T. Pharmakologische und chemische Untersuchungen über die wirksamen Bestandteile der Urtica dioica und Urtica urens. Arch. Exp. Path. Pharmakol. 1933, 172, 137–148. [Google Scholar] [CrossRef]
- Emmelin, N.; Feldberg, W. The mechanism of the sting of the common nettle (Urtica dioica). J. Physiol. 1947, 106, 440–445. [Google Scholar] [CrossRef]
- Collier, H.O.J.; Chesher, G.B. Identification of 5-hydroxytryptamine in the sting of the nettle (Urtica dioica). Brit. J. Pharmacol. 1956, 11, 186–189. [Google Scholar] [CrossRef]
- Robertson, P.A.; MacFarlane, W.V. Pain producing substances from the stinging bush Laportea moroides. Aust. J. Exp. Biol. Med. Sci. 1957, 35, 381–393. [Google Scholar] [CrossRef]
- Pilgrim, R.L.C. Some properties of the sting of the New Zealand nettle, Urtica ferox. Proc. Roy. Soc. Lond. Ser. B 1959, 151, 48–56. [Google Scholar]
- Saxena, P.R.; Pant, M.C.; Kishor, K.; Bhargava, K.P. Identification of pharmacologically active substances in the Indian Stinging Nettle, Urtica parviflora (Roxb.). Canad. J. Physiol. Pharmacol. 1965, 43, 869–876. [Google Scholar] [CrossRef]
- Saxena, P.R.; Tangri, K.K.; Bhargava, K.P. Identification of acetylcholine, histamine, and 5-hydroxytryptamine in Girardinia heterophylla. Can. J. Physiol. Pharmacol. 1966, 44, 621–627. [Google Scholar] [CrossRef]
- Schildknecht, H. Irritant and defense substances of higher plants—A chemical herbarium. Angew. Chem. Int. Ed. Engl. 1981, 20, 164–184. [Google Scholar] [CrossRef]
- Fu, H.Y.; Chen, S.J.; Chen, R.F.; Kuo-Huang, L.L.; Huang, R.N. Why do nettles sting? About stinging hairs looking simple but acting complex. Funct. Plant Sci. Biotechnol. 2007, 1, 46–55. [Google Scholar]
- Moreno, M.; Giralt, E. Three valuable peptides from bee and wasp venoms for therapeutic and biotechnological use: Melittin, apamin and mastoparan. Toxins 2015, 7, 1126–1150. [Google Scholar] [CrossRef]
- Hewson, H.J. Plant Indumentum: A Handbook of Terminology; Australian Biological Resources Study, Commonwealth Department of the Environment and Energy: Canberra, Australia, 2019; 47p. [Google Scholar]
- Ensikat, H.J.; Mustafa, A.; Weigend, M. Complex patterns of multiple biomineralization in single-celled plant trichomes of the Loasaceae. Am. J. Bot. 2017, 104, 195–206. [Google Scholar] [CrossRef]
- Thurston, E.L. Morphology, fine structure and ontogeny of the stinging emergence of Tragia ramosa and T. saxicola (Euphorbiaceae). Am. J. Bot. 1976, 63, 710–718. [Google Scholar] [CrossRef]
- Wurdack, K.J.; Hoffmann, P.; Chase, M. Molecular phylogenetic analysis of uniovulate Euphorbiaceae (Euphorbiaceae sensu stricto) using plastid rbcL and trnL-F DNA sequences. Am. J. Bot. 2005, 92, 1397–1420. [Google Scholar] [CrossRef]
- Friis, I. Urticaceae. In Flowering Plants—Dicotyledons. The Families and Genera of Vascular Plants; Kubitzki, K., Rohwer, J.G., Bittrich, V., Eds.; Springer: Berlin, Germany, 1993; Volume 2. [Google Scholar]
- Kariyat, R.R.; Smith, J.D.; Stephenson, A.G.; De Moraes, C.M.; Mescher, M.C. Non-glandular trichomes of Solanum carolinense deter feeding by Manduca sexta caterpillars and cause damage to the gut peritrophic matrix. Proc. R. Soc. B Biol. Sci. 2017, 284, 20162323. [Google Scholar] [CrossRef] [PubMed]
- Shelley, W.B.; Arthur, R.P. Studies on cowhage (Mucuna pruriens) and its pruritogenic proteinase, mucunain. Arch. Dermatol. 1955, 72, 399–406. [Google Scholar] [CrossRef]
- Pardo-Castello, V. Dermatitis venenata: Study of the tropical plants producing dermatitis. Arch. Derm. Syphilol. 1923, 7, 81–90. [Google Scholar] [CrossRef]
- Wu, Z.Y.; Monro, A.K.; Milne, R.I.; Yi, H.T.; Liu, J.; Li, D.Z. Molecular phylogeny of the nettle family (Urticaceae) inferred from multiple loci of three genomes and extensive generic sampling. Mol. Phylogenet. Evol. 2013, 69, 814–827. [Google Scholar] [CrossRef] [PubMed]
- Grosse-Veldmann, B.; Nürk, N.M.; Smissen, R.; Breitwieser, I.; Quandt, D.; Weigend, M. Pulling the sting out of nettle systematics—A comprehensive phylogeny of the genus Urtica (Urticaceae). Mol. Phylogenet. Evol. 2016, 102, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Leung, C.; Williams, D.H.; Barna, J.C.J.; Foti, S.; Oelrichs, P.B. Structural studies on the peptide Moroidin from Laportea moroides. Tetrahedron 1986, 42, 3333–3348. [Google Scholar] [CrossRef]
- Weigend, M. Loasaceae. In The Families and Genera of Vascular Plants; Kubitzki, K., Bayer, C., Eds.; Springer: Berlin, Germany, 2003; Volume 6, pp. 239–254. [Google Scholar]
- Maya-Lastra, C.A.; Steinmann, W. Evolution of the untouchables: Phylogenetics and classification of Cnidoscolus (Euphorbiaceae). Taxon 2019, 68, 692–713. [Google Scholar] [CrossRef]
- Solís-Montero, V.; Martínez-Natarén, D.A.; Parra-Tabla, V.; Ibarra-Cerdeña, C.; Munguía-Rosas, M.A. Herbivory and anti-herbivore defences in wild and cultivated Cnidoscolus aconitifolius: Disentangling domestication and environmental effects. AoB Plants 2020, 12, plaa023. [Google Scholar] [CrossRef] [PubMed]
- Cardinal-McTeague, W.M.; Gillespie, L.J. Molecular phylogeny and pollen evolution of Euphorbiaceae Tribe Plukenetieae. System. Botany 2016, 41, 329–347. [Google Scholar] [CrossRef]
- Lorence, D.H.; Colin, R.T. Carica cnidoscoloides (sp. nov.) and sect. Holostigma (sect. nov.) of Caricaceae from Southern Mexico. Syst. Bot. 1988, 13, 107–110. [Google Scholar]
- Ensikat, H.J.; Weigend, M. A three-dimensional perspective on multiple biomineralization in Loasaceae trichomes. Authorea 2020. [Google Scholar] [CrossRef]
- Coté, G.G. Diversity and Distribution of idioblasts producing calcium oxalate crystals in Dieffenbachia seguine (Araceae). Am. J. Bot. 2009, 96, 1245–1254. [Google Scholar] [CrossRef] [PubMed]
- Lewis, T. The Blood Vessels of the Human Skin and Their Responses; Shaw and Sons: London, UK, 1927. [Google Scholar]
- Starr, I. Change in the reaction of the skin to histamine as evidence of deficient circulation in the lower extremities. JAMA 1928, 90, 2092–2094. [Google Scholar] [CrossRef]
- Lahti, A. Non-Immunologic Contact Urticaria. In Handbook of Occupational Dermatology; Kanerva, L., Wahlberg, J.E., Elsner, P., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2000. [Google Scholar]
- Edwards, W.C.; Remer, J.C. Nettle poisoning in dogs. Vet. Med. 1983, 78, 347–350. [Google Scholar]
- Coile, N. Urtica chamaedryoides Pursh: A stinging nettle, or fireweed and some related species. Bot. Circ. 1999, 34, 1–6. [Google Scholar]
- Caliskaner, Z.; Karaayvaz, M.; Ozturk, S. Misuse of a herb: Stinging nettle (Urtica urens) induced severe tongue oedema. Complementary Ther. Med. 2004, 12, 57–58. [Google Scholar] [CrossRef] [PubMed]
- MacFarlane, W.V. The Stinging Properties of Laportea. Econ. Bot. 1963, 17, 303–311. [Google Scholar] [CrossRef]
- Hurley, M. Selective stingers. Ecos 2000, 105, 20–23. [Google Scholar]
- Hurley, M. The Stinging Tree, Dendrocnide Moroides. Available online: https://theconversation.com/the-worst-kind-of-pain-you-can-imagine-what-its-like-to-be-stung-by-a-stinging-tree-103220 (accessed on 21 August 2020).
- Hammond-Tooke, G.D.; Taylor, P.; Punchihewa, S.; Beasley, M. Urtica ferox Neuropathy. Muscle Nerve 2007, 35, 804–807. [Google Scholar] [CrossRef] [PubMed]
- Connor, H.E. The Poisonous Plants in New Zealand; Government Printer: Wellington, New Zealand, 1977.
- Puig, J.; Griffin, A.S.; Montoliu, P. Neurological signs following suspected exposure to stinging nettles in two dogs. Vet. Rec. Case Rep. 2019, 7, e000672. [Google Scholar] [CrossRef]
- Willox, S. An investigation of the New Zealand stinging nettle Urtica ferox. Ph.D. Thesis, University of Otago, Dunedin, New Zealand, 1969. [Google Scholar]
- Clark, F.P. Tree nettle (Urtica ferox) poisoning. N. Z. Med. J. 1993, 106, 234. [Google Scholar]
- Reynolds, G.W.; Epstein, W.L.; Rodriguez, E. Unusual contact allergens from plants in the family Hydrophyllaceae. Contact Dermatitis 1986, 14, 39–44. [Google Scholar] [CrossRef]
- Scheman, A.J.; Conde, A. Contact Dermatitis from Cnidoscolus angustidens. Contact Dermatitis 2001, 45, 39. [Google Scholar] [CrossRef]
- Kregiel, D.; Pawlikowska, E.; Antolak, H. Urtica spp.: Ordinary plants with extraordinary properties. Molecules 2018, 23, 1664. [Google Scholar] [CrossRef] [PubMed]
- Cordeior, R.S.; Aragão, J.B.; Morhy, L. The presence of histamine in Cnidosculus oligandrus (Euphorbiaceae). An. Acad. Bras. Cienc. 1983, 55, 123–128. [Google Scholar] [PubMed]
- Willis, C.L. Toxic Constituents of the Stinging Nettle. Master’s Thesis, Iowa State University, Ames, IA, USA, 1969. [Google Scholar]
- Wessely, H. Mass spectrometric investigation of Urtica stinging trichomes juice. Bachelor’s Thesis, Rheinische Friedrich-Wilhelms-Universität Bonn, Bonn, Germany, 2020. [Google Scholar]
- Roshchina, V.V. Neurotransmitters in Plant Live; Science Publishers, Inc.: Enfield, NH, USA, 2001. [Google Scholar]
- Ramakrishna, A.; Roshchina, V.V. Neurotransmitters in Plants—Perspectives and Applications. CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2019. [Google Scholar]
- Ramakrishna, A.; Giridhar, P.; Ravishankar, G.A. Phytoserotonin. Plant Signal. Behav. 2011, 6, 800–809. [Google Scholar] [PubMed]
- Erland, L.; Murch, S.J.; Reiter, R.J.; Saxena, P.K. A new balancing act: The many roles of melatonin and serotonin in plant growth and development. Plant Signal. Behav. 2015, 10, e1096469. [Google Scholar] [CrossRef]
- Tretyn, A.; Kendrick, R.E. Acetylcholine in plants: Presence, metabolism and mechanism of action. Bot. Rev. 1991, 57, 33–73. [Google Scholar] [CrossRef]
- Czarnetzki, B.M.; Thiele, T.; Rosenbach, T. Immunoreactive leukotrienes in nettle plants (Urtica urens). Int. Arch. Allergy Immunol. 1990, 91, 43–46. [Google Scholar] [CrossRef]
- Juhlin, L.; Hammerström, S. Wheal reactions in human skin after injection of leukotrienes B4, C4, D4 and E4. Prostaglandins Leukot. Med. 1983, 14, 381–383. [Google Scholar] [CrossRef]
- Kahn, S.D.; Booth, P.M.; Waltho, J.P.; Williams, D.H. Computer-assisted structure determination. Structure of the peptide moroidin from Laportea moroides. J. Org. Chem. 2000, 65, 8406. [Google Scholar] [CrossRef]
- Gilding, E.K.; Jami, S.; Deuis, J.R.; Israel, M.R.; Harvey, P.J.; Poth, A.G.; Rehm, F.B.H.; Stow, J.L.; Robinson, S.D.; Yap, K.; et al. Neurotoxic peptides from the venom of the giant Australian stinging tree. Sci. Adv. 2020, 6, eabb8828. [Google Scholar] [CrossRef]
- Armstrong, D.; Dry, R.M.L.; Keele, C.A.; Markham, J.W. Method for studying chemical excitants of cutaneous pain in man. J. Physiol. 1951, 115, 59–61. [Google Scholar]
- Armstrong, D.; Dry, R.M.L.; Keele, C.A.; Markham, J.W. Observations on chemical excitants of cutaneous pain in man. J. Physiol. 1953, 120, 326–351. [Google Scholar] [CrossRef]
- Oliver, F.; Amon, E.U.; Breathnach, A.; Francis, D.M.; Saracthchandra, P.; Kobza, A.; Greaves, M.W. Contact urticaria due to the common stinging nettle (Urtica dioica)—Histological, ultrastructural and pharmacological studies. Clin. Exp. Dermatol. 1991, 16, 1–7. [Google Scholar] [CrossRef]
- Ramírez-Bravo, O.E.; Hernández-Santin, L. Plant diversity along a disturbance gradient in a semi-arid ecosystem in Central Mexico. Act. Bot. Mex. 2016, 117, 11–25. [Google Scholar] [CrossRef]
- Marchi, S.R.; Silva, H.M.; Ferreira, C.F.; Marques, R.F.; Moraes, J.B. Interference of noxious shrubs on grazing behaviour by bovines. Planta Daninha 2019, 37, e019185644. [Google Scholar] [CrossRef]
- Eycott, A.E.; Watkinson, A.R.; Hemami, M.R.; Dolman, P.M. The dispersal of vascular plants in a forest mosaic by a guild of mammalian herbivores. Oecologia 2007, 154, 107–118. [Google Scholar] [CrossRef]
- Pakeman, R.J.; Digneffe, G.; Small, J.L. Ecological correlates of endozoochory by herbivores. Funct. Ecol. 2002, 16, 296–304. [Google Scholar] [CrossRef]
- Jaroszewicz, B.; Piroźnikow, E.; Sagehorn, R. Endozoochory by European bison (Bison bonasus) in Bialowiez. a Primeval Forest across a management gradient. For. Ecol. Manag. 2009, 258, 11–17. [Google Scholar] [CrossRef]
- Huygens, O.C.; Miyashita, T.; Dahle, B.; Carr, M.; Izumiyama, S.; Sugawara, T.; Hayashi, H. Diet and habits of Asiatic black bears feeding in the Northern Japanese Alps. Ursus 2003, 14, 236–245. [Google Scholar]
- Everitt, J.H.; Alaniz, M.A. Fall and winter diets of feral pigs in South Texas. J. Range Manage. 1908, 33, 126–129. [Google Scholar] [CrossRef]
- Wood, J.R.; Richardson, S.J.; McGlone, M.S.; Wilmshurst, J.M. The diets of moa (Aves: Dinornithiformes). N. Z. J. Ecol. 2020, 44. [Google Scholar] [CrossRef]
- Kirillova, I.V.; Argant, J.; Lapteva, E.G.; Korona, O.M.; van der Plicht, J.; Zinovyev, E.V.; Kotov, A.A.; Chernova, O.F.; Fadeeva, E.O.; Baturina, O.A.; et al. The diet and environment of mammoths in North-East Russia reconstructed from the contents of their feces. Quat. Int. 2016, 406, 147–161. [Google Scholar] [CrossRef]
- Rothman, J.M.; Chapman, C.A.; Hansen, J.L.; Cherney, D.J.R.; Pell, A.N. Rapid assessment of the nutritional value of foods eaten by Mountain Gorillas: Applying near-infrared reflectance spectroscopy to primatology. Int. J. Primatol. 2009, 30, 729–742. [Google Scholar] [CrossRef]
- Wasserman, M.D.; Taylor-Gutt, A.; Rothman, J.M.; Chapman, C.A.; Milton, K.; Leitman, D.C. Estrogenic plant foods of Red Colobus Monkeys and Mountain Gorillas in Uganda. Am. J. Phys. Anthropol. 2012, 48, 8897. [Google Scholar] [CrossRef]
- Matthews, J.K.; Ridley, A.; Niyigaba, P.; Kaplin, B.A.; Grueter, C.C. Chimpanzee feeding ecology and fallback food use in the montane forest of Nyungwe National Park, Rwanda. Am. J. Primatol. 2019, 81, e22971. [Google Scholar] [CrossRef]
- Pullin, A.S.; Gilbert, J.E. The Stinging Nettle, Urtica dioica, increases trichome density after herbivore and mechanical damage. Oikos 1989, 54, 275–280. [Google Scholar] [CrossRef]
- Kato, T.; Ishida, K.; Kikuchi, J.; Torii, H. Induced response to herbivory in stinging hair traits of Japanese nettle (Urtica thunbergiana) seedling in two subpopulations with different browsing pressures by sika deer. Plant Spec. Biol. 2017, 32, 340–347. [Google Scholar] [CrossRef]
- Abdala-Roberts, L.; Parra-Tabla, V. Artificial defoliation induces trichome production in the tropical shrub Cnidoscolus aconitifolius (Euphorbiaceae). Biotropica 2005, 37, 251–257. [Google Scholar] [CrossRef]
- Kato, T.; Ishida, K.; Sato, H. The evolution of nettle resistance to heavy deer browsing. Ecol. Res. 2008, 23, 339–345. [Google Scholar] [CrossRef]
- Pollard, A.J. Variation in Cnidoscolus texanus in relation to herbivory. Oecologia 1986, 70, 411–413. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.N.K. The European distribution of insects on the stinging nettles, Urtica dioica L.: A field survey. Boll. Zool. 1989, 56, 321–326. [Google Scholar] [CrossRef]
- Cates, R.G.; Orians, G.H. Successional status and the palatability of plants to generalized herbivores. Ecology 1975, 56, 410–418. [Google Scholar] [CrossRef]
- Janz, N.; Nyblom, K.; Nylin, S. Evolutionary dynamics of host-plant specialization: A case study of the tribe Nymphalini. Evolution 2001, 55, 783–796. [Google Scholar] [CrossRef]
- Cano-Santana, Z.; Oyama, K. Variation in leaf trichomes and nutrients of Wigandia urens (Hydrophyllaceae) and its implications for herbivory. Oecologia 1992, 92, 405–409. [Google Scholar] [CrossRef]
- Dillon, P.M.; Lowrie, S.; McKey, D. Disarming the “Evil Woman”: Petiole constriction by a sphingid larva circumvents mechanical defenses of its host plant, Cnidoscolus urens (Euphorbiaceae). Biotropica 1983, 15, 112–116. [Google Scholar] [CrossRef]
- Hawkeswood, T.J. Observations on a Diphucephala species (Coleoptera: Scarabaeidae) and its feeding relationships with the giant stinging tree, Dendrocnide excelsa (Wedd.) Chew (Urticaceae) in Queensland, Australia. Entomologist 1991, 110, 170–177. [Google Scholar]
- Weigend, M.; Kufer, J.K.; Mueller, A.A. Phytochemistry and the systematics and ecology of Loasaceae & Gronoviaceae (Loasales). Am. J. Bot. 2000, 87, 1002–1011. [Google Scholar]
- Machado, S.R.; Morellato, L.P.C.; Sajo, M.G.; Oliveira, P.S. Morphological patterns of extrafloral nectaries in woody plant species of the Brazilian cerrado. Plant Biol. 2008, 10, 660–672. [Google Scholar] [CrossRef]
- Jones, R.S.; Stutte, C.A. 1985 Chromatographic analysis of choline and acetylcholine by UV visualization. J. Chromatogr. 1985, 319, 454–460. [Google Scholar] [CrossRef]
- Ho, C.S.; Lam, C.W.K.; Chan, M.H.M.; Cheung, R.C.K.; Law, L.K.; Lit, L.C.W.; Ng, K.F.; Suen, M.W.M.; Tai, H.L. Electrospray ionisation mass spectrometry: Principles and clinical applications. Clin. Biochem. Rev. 2003, 24, 3–12. [Google Scholar] [PubMed]
- Weigend, M.; Mustafa, A.; Ensikat, H.J. Calcium phosphate in plant trichomes: The overlooked biomineral. Planta 2018, 247, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Ensikat, H.J.; Weigend, M. EDX and Raman spectroscopy reveal the unexpected complexity of plant biomineralisation. Microsc. Anal. 2019, 45, 20–23. [Google Scholar]









| Authors (Listed Chronologically); Ref. | Formic Acid | Acetylcholine | Histamine | 5-hydroxytryptamine | Alkaloid | Acetic Acid | Enzyme | Glucoside | Protein | Tartaric Acid | Resin Acid | Calcium | Salt | Calcium-Sensitive Factor |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hooke (1665); [2] | Urt | |||||||||||||
| Gorup-Besanez (1849); [4] | Urt | |||||||||||||
| Rauter (1872); [5] | Urt | |||||||||||||
| Bergmann (1882); [6] | Urt | |||||||||||||
| Haberlandt (1886); [7] | Urt | |||||||||||||
| Tassi (1886); [8] | Loa | |||||||||||||
| Gibson & Warham (1890); [9] | Urt | |||||||||||||
| Ritterhausen (1892); [10] | Tra | |||||||||||||
| Giustiniani (1896); [11] | Urt | |||||||||||||
| Dragendorf (1905); [12] | Urt | |||||||||||||
| Knoll (1905); [13] | Tra | |||||||||||||
| Petrie (1906); [14] | Lap | Lap | ||||||||||||
| Winternitz (1907); [15] | Urt | Urt | Urt | |||||||||||
| Flury (1919); [16] | Urt | |||||||||||||
| Nestler (1925); [17] | Urt | Urt | ||||||||||||
| Flury (1927); [18] | Urt, Lap | |||||||||||||
| Kroeber (1928); [19] | Urt | |||||||||||||
| Starkenstein & Wasserstrom (1933); [20] | Urt | Urt | ||||||||||||
| Emmelin & Feldberg (1947); [21] | Urt | Urt | ||||||||||||
| Collier & Chesher (1956); [22] | Urt | Urt | Urt | |||||||||||
| Robertson and MacFarlane (1957); [23] | Lap | Lap | Lap | |||||||||||
| Pilgrim (1959); [24] | Urt | |||||||||||||
| Saxena et al. (1965, 1966); [25,26] | Urt, Gir | Urt, Gir | Urt, Gir |
| Species | Length (mm) | Lumen (nL) |
|---|---|---|
| Urticaceae | ||
| Urtica dioica | 1.38 | 3.85 |
| Urtica atrovirens | 2.34 | 13.1 |
| Urtica mairei | 2.79 | 38.5 |
| Urera baccifera | 2 | 58.5 |
| Loasaceae | ||
| Aosa rupestris | 2.6 | 10.4 |
| Caiophora sp. | 2.12 | 8 |
| Caiophora deserticola | 3.47 | 34.4 |
| Chichicaste grandis | 1.22 | 4.1 |
| Loasa pallida | 2.9 | 19.1 |
| Nasa amaluzensis | 2.86 | 31.3 |
| Euphorbiaceae | ||
| Cnidoscolus aconitifolius | 4.25 | 140.6 |
| Namaceae | ||
| Wigandia spec. | 5.4 | 44.4 |
| Plant Family | Species | C | P | Cl | Na | K | S |
|---|---|---|---|---|---|---|---|
| Urticaceae | |||||||
| Laportea perrieri | 100 | 11 | 0 | 0 | 1.8 | 2.8 | |
| Urera baccifera | 110 | 0 | 30.4 | 0 | 100 | 0 | |
| “ | 257 | 4.3 | 0 | 0 | 100 | 0 | |
| “ | 189 | 42 | 0 | 0 | 100 | 0 | |
| Urtica dioica | 1133 | 110 | 0 | 0 | 100 | 10.4 | |
| “ | 791 | 70 | 15.2 | 0 | 100 | 15.2 | |
| “ | 167 | 42 | 11.1 | 0 | 100 | 0 | |
| “ | 370 | 173 | 14 | 0 | 100 | 9.4 | |
| “ | 663 | 128 | 18.8 | 0 | 100 | 15 | |
| Urtica gracilis | 735 | 97 | 0 | 0 | 100 | 19 | |
| Urtica massaica | 600 | 114 | 23 | 128 | 100 | 43 | |
| “ | 218 | 77 | 15 | 22 | 100 | 22 | |
| Urtica pilulifera | 280 | 0 | 0 | 0 | 100 | 0 | |
| Loasaceae | |||||||
| Aosa rupestris | 747 | 0 | 0 | 132 | 100 | 0 | |
| Blumenbachia hieronymi | 300 | 0 | 0 | 0 | 100 | 0 | |
| Caiophora lateritia | 467 | 45 | 40 | 38 | 100 | 0 | |
| Loasa elongata | 657 | 0 | 0 | 0 | 100 | 0 | |
| Loasa insons | 294 | 0 | 16 | 0 | 100 | 0 | |
| Loasa pallida | 249 | 4.8 | 7.1 | 0 | 100 | 0 | |
| “ | 485 | 0 | 20 | 0 | 100 | 0 | |
| “ | 166 | 0 | 23 | 0 | 100 | 0 | |
| Loasa tricolor | 279 | 17 | 0 | 0 | 100 | 0 | |
| Loasa triloba | 162 | 0 | 17 | 3 | 100 | 0 | |
| Nasa amaluzensis | 150 | 0 | 0 | 0 | 100 | 0 | |
| “ | 309 | 0 | 0 | 0 | 100 | 0 | |
| Nasa poissoniana | 393 | 0 | 0 | 0 | 100 | 0 | |
| Euphorbiaceae | |||||||
| Cnidoscolus aconitifolius | 177 | 0 | 0 | 0 | 100 | 0 | |
| “ | 170 | 0 | 0 | 0 | 100 | 0 | |
| Namaceae | |||||||
| Wigandia caracasana | 101 | 0 | 0 | 0 | 100 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ensikat, H.-J.; Wessely, H.; Engeser, M.; Weigend, M. Distribution, Ecology, Chemistry and Toxicology of Plant Stinging Hairs. Toxins 2021, 13, 141. https://doi.org/10.3390/toxins13020141
Ensikat H-J, Wessely H, Engeser M, Weigend M. Distribution, Ecology, Chemistry and Toxicology of Plant Stinging Hairs. Toxins. 2021; 13(2):141. https://doi.org/10.3390/toxins13020141
Chicago/Turabian StyleEnsikat, Hans-Jürgen, Hannah Wessely, Marianne Engeser, and Maximilian Weigend. 2021. "Distribution, Ecology, Chemistry and Toxicology of Plant Stinging Hairs" Toxins 13, no. 2: 141. https://doi.org/10.3390/toxins13020141
APA StyleEnsikat, H.-J., Wessely, H., Engeser, M., & Weigend, M. (2021). Distribution, Ecology, Chemistry and Toxicology of Plant Stinging Hairs. Toxins, 13(2), 141. https://doi.org/10.3390/toxins13020141





