Genetic Diversity, Ochratoxin A and Fumonisin Profiles of Strains of Aspergillus Section Nigri Isolated from Dried Vine Fruits
Abstract
1. Introduction
- (1)
- What is the level of OTA in the analysed dried vine fruit samples, and does the OTA content exceed the critical maximum limit of 10 µg/kg?
- (2)
- What is the overall diversity of microscopic fungi colonising the dried vine fruit samples? How many species from the Aspergillus section Nigri can be isolated and identified in the tested samples?
- (3)
- What are the individual ochratoxigenic abilities of selected isolated strains of members of Aspergillus section Nigri? Do some isolated black aspergilli strains produce fumonisins FB1 and FB2?
2. Results
2.1. Cultivations and Morphological Identifications
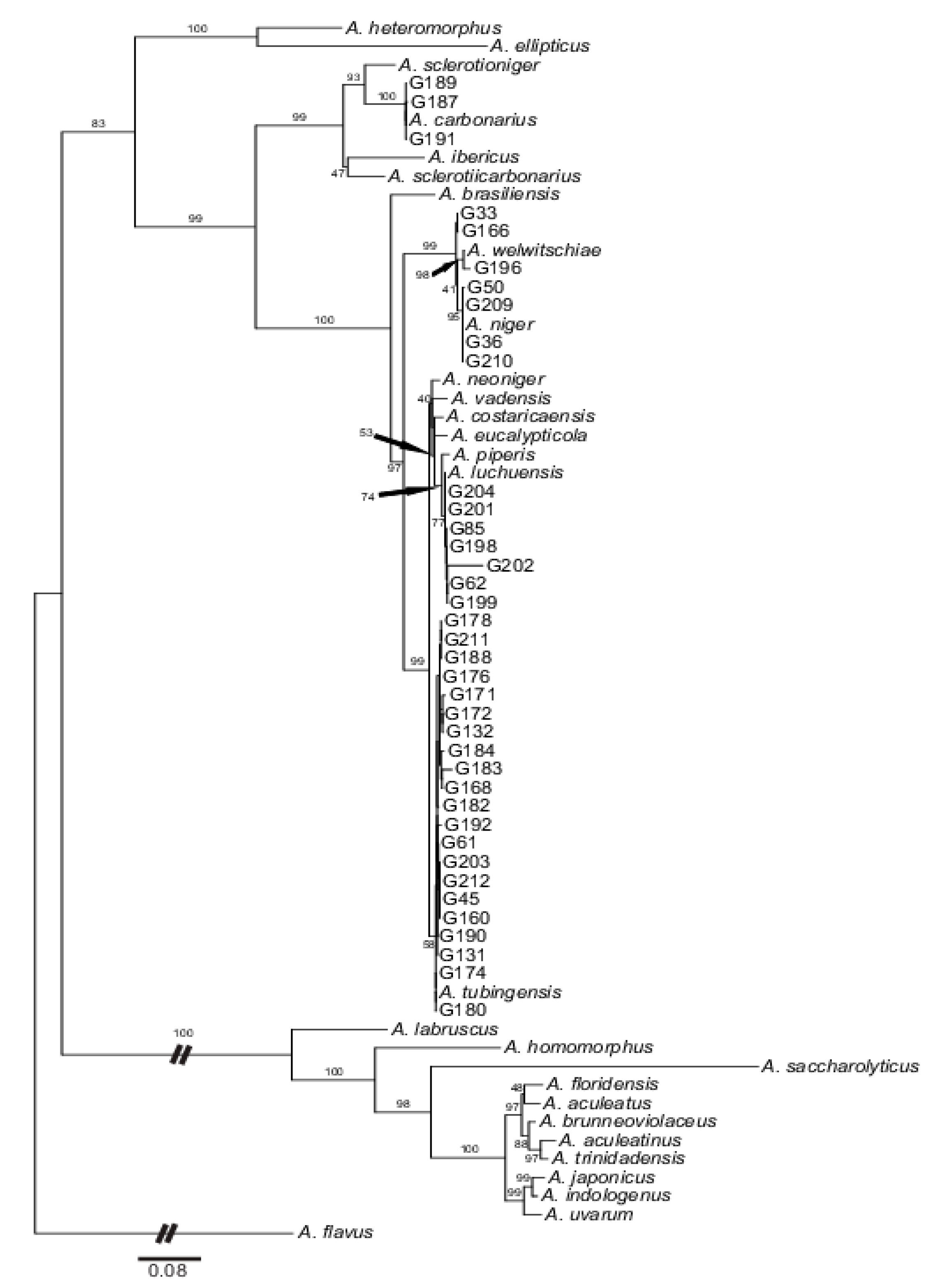
2.2. Molecular Analyses of the Black Aspergilli Strains
2.3. Determination of Ochratoxin A (OTA) Content in Analysed Raisins Samples
2.4. Toxigenic Ability of Aspergillus Isolates—Production of Ochratoxin A and Fumonisins B1 and B2
3. Discussion
3.1. Taxonomic Identity of Isolated Strains and Diversity of Black Aspergilli on Dried Vine Fruits
3.2. OTA Contamination of Analysed Dried Vine Fruits Samples
3.3. Ochratoxigenic and Fumonisins Production Potential of Aspergillus Section Nigri Isolates
4. Conclusions
5. Material and Methods
5.1. Sampling
5.2. Isolation and Morphological Identification of Retrieved Strains
5.3. Molecular Analysis
5.4. Sequence Alignments and Phylogenetic Analysis
5.5. Determination of Ochratoxin A (OTA) Level in Vine Fruits
5.6. OTA, FB1 and FB2 Production Ability of Aspergillus Isolates
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Dedication
References
- Gasghari, R.M.; Shebany, Y.M.; Gherbawy, Y.A. Molecular characterization of ochratoxigenic fungi associated with raisins. Foodborne Pathog. Dis. 2011, 8, 1221–1227. [Google Scholar] [CrossRef]
- Samson, R.A.; Visagie, C.M.; Houbraken, J.; Hong, S.B.; Hubka, V.; Klaassen, C.H.V.; Perrone, G.; Seifert, K.A.; Tanney, J.B.; Varga, J.; et al. Phylogeny, identification and nomenclature of the genus Aspergillus. Stud. Mycol. 2014, 78, 141–173. [Google Scholar] [CrossRef] [PubMed]
- Gams, W.; Christensen, M.; Onions, A.H.S.; Pitt, J.I.; Samson, R.A. Infrageneric taxa of Aspergillus. In Advances in Penicillium and Aspergillus Systematics. NATO ASI Series (Series A: Life Sciences); Samson, R.A., Pitt, J.I., Eds.; Springer: Boston, MA, USA, 1986; Volume 102, pp. 55–64. [Google Scholar] [CrossRef]
- Pitt, J.I.; Hocking, A.D. Fungi and Food Spoilage; Springer: Boston, MA, USA, 2009; pp. 1–519. [Google Scholar] [CrossRef]
- Magnoli, C.; Astoreca, A.; Ponsone, L.; Combina, M.; Palacio, G.; Rosa, C.; Dalcero, A. Survey of mycoflora and ochratoxin A in dried vine fruits from Argentina markets. Lett. Appl. Microbiol. 2004, 39, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, J.D.; O’Keeffe, T.L.; Vasquez, S.J.; Mahoney, N.E. Isolation and identification of ochratoxin A-producing Aspergillus section Nigri strains from California raisins. Lett. Appl. Microbiol. 2011, 52, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Pantelides, I.S.; Aristeidou, E.; Lazari, M.; Tsolakidou, M.D.; Tsaltas, D.; Christofidou, M.; Kafouris, D.; Christou, E.; Ioannou, N. Biodiversity and ochratoxin A profile of Aspergillus section Nigri populations isolated from wine grapes in Cyprus vineyards. Food Microbiol. 2017, 67, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Perrone, G.; Susca, A.; Cozzi, G.; Ehrlich, K.; Varga, J.; Frisvad, J.C.; Meijer, M.; Noonim, P.; Mahakarnchanakul, W.; Samson, R.A. Biodiversity of Aspergillus species in some important agricultural products. Stud. Mycol. 2007, 59, 53–66. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). Ochratoxin A. In IARC Monographs on the Evaluation of Carcinogenic Risk to Humans: Some Naturally is Occurring Substances, Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins. IARC Scientific Publication No. 56; International Agency for Research on Cancer: Geneva, Switzerland, 1993; pp. 26–32. [Google Scholar]
- Perrone, G.; Logrieco, A.F.; Frisvad, J.C. Comments on “Screening and Identification of Novel Ochratoxin A-Producing Fungi from Grapes. In Reporting Ochratoxin A Production from Strains of Aspergillus, Penicillium and Talaromyces. Toxins 2017, 9, 65. [Google Scholar] [CrossRef]
- Commission of the European Communities (CEC). Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L364, 5–24. Available online: http://data.europa.eu/eli/reg/2006/1881/oj (accessed on 26 June 2020).
- Samson, R.A.; Noonim, P.; Meijer, M.; Houbraken, J.; Frisvad, J.C.; Varga, J. Diagnostic tools to identify black aspergilli. Stud. Mycol. 2007, 59, 129–145. [Google Scholar] [CrossRef]
- Ross, P.F.; Rice, L.G.; Osweiler, G.D.; Nelson, P.E.; Richard, J.L.; Wilson, T.M. A review and update of animal toxicoses associated with fumonisin-contaminated feeds and production of fumonisins by Fusarium isolates. Mycopathologia 1992, 117, 109–114. [Google Scholar] [CrossRef]
- Thiel, P.G.; Shephard, G.S.; Sydenham, E.W.; Marasas, W.F.O.; Nelson, P.E.; Wilson, T.M. Levels of fumonisins B1 and B2 in feeds associated with confirmed cases of equine leukoencephalomalacia. J. Agric. Food Chem. 1991, 39, 109–111. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Jaskiewicz, K.; Marasas, W.F.O.; Thiel, P.G.; Horak, R.M.; Vleggaar, R.; Kriek, N.P. Fumonisins-novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl. Environ. Microbiol. 1988, 54, 1806–1811. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wei, H.; Ma, J.; Luo, X. The fumonisin B1 content in corn from North China, a high-risk area of esophageal cancer. J. Environ. Pathol. Toxicol. Oncol. 2000, 19, 139–141. [Google Scholar] [PubMed]
- Frisvad, J.C.; Smedsgaard, J.; Samson, R.A.; Larsen, T.O.; Thrane, U. Fumonisin B2 production by Aspergillus niger. J. Agric. Food Chem. 2007, 55, 9727–9732. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Larsen, T.O.; Thrane, U.; Meijer, M.; Varga, J.; Samson, R.A.; Nielsen, K.F. Fumonisin and ochratoxin production in industrial Aspergillus niger strains. PLoS ONE 2011, 6, e23496. [Google Scholar] [CrossRef]
- Mogensen, J.M.; Frisvad, J.C.; Thrane, U.; Nielsen, K.F. Production of Fumonisin B2 and B4 by Aspergillus niger on grapes and raisins. J. Agric. Food Chem. 2010, 58, 954–958. [Google Scholar] [CrossRef]
- Susca, A.; Proctor, R.H.; Morelli, M.; Haidukowski, M.; Gallo, A.; Logrieco, A.F.; Moretti, A. Variation in Fumonisin and Ochratoxin production associated with differences in biosynthetic gene content in Aspergillus niger and A. welwitschiae isolates from multiple crop and geographic origins. Front. Microbiol. 2016, 7, 1412. [Google Scholar] [CrossRef]
- Perrone, G.; Stea, G.; Epifani, F.; Varga, J.; Frisvad, J.C.; Samson, R.A. Aspergillus niger contains the cryptic phylogenetic species A. awamori. Fungal Biol. 2011, 115, 1038–1050. [Google Scholar] [CrossRef]
- Koscubé, S.; Perrone, G.; Magistà, D.; Houbraken, J.; Varga, J.; Szigeti, G.; Hubka, V.; Hong, S.B.; Frisvad, J.C.; Samson, R.A. Aspergillus is monophyletic: Evidence from multiple gene phylogenies and extrolites profiles. Stud. Mycol. 2016, 85, 199–213. [Google Scholar] [CrossRef]
- Medina, A.; Mateo, R.; López-Ocaña, L.; Valle-Algarra, F.M.; Jiménez, M. Study of Spanish grape mycobiota and ochratoxin A production by isolates of Aspergillus tubingensis and other members of Aspergillus Section Nigri. Appl. Environ. Microbiol. 2005, 71, 4696–4702. [Google Scholar] [CrossRef]
- Perrone, G.; Susca, A.; Epifani, F.; Mulè, G. AFLP characterization of Southern Europe population of Aspergillus Section Nigri from grapes. Int. J. Food Microbiol. 2006, 111, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Cabañes, F.J.; Bragulat, M.R. Ochratoxin A in profiling and speciation. In Aspergillus in the Genomic Era; Varga, J., Samson, R.A., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2008; pp. 57–70. [Google Scholar] [CrossRef]
- Abarca, M.L.; Accensi, F.; Cano, J.; Cabañes, F.J. Taxonomy and significance of black aspergilli. Antonie Van Leeuwenhoek Int. J. Gen. Mol. Microbiol. 2004, 86, 33–49. [Google Scholar] [CrossRef] [PubMed]
- Hubka, V.; Kolařík, M. β-tubulin paralogue tubC is frequently misidentified as the benA gene in Aspergillus section Nigri taxonomy: Primer specificity testing and taxonomic consequence. Persoonia 2012, 29, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kizis, D.; Natskoulis, P.; Nychas, G.J.; Panagou, E.Z. Biodiversity and ITS-RFLP characterisation of Aspergillus section Nigri isolates in grapes from four traditional grape-producing areas in Greece. PLoS ONE 2014, 9, e93923. [Google Scholar] [CrossRef]
- Palumbo, J.D.; O’Keeffe, T.L. Detection and discrimination of four Aspergillus section Nigri species by PCR. Lett. Appl. Microbiol. 2015, 60, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Dachoupakan, C.; Ratomahenina, R.; Martinez, V.; Guiraud, J.P.; Baccou, J.C.; Schorr-Galindo, S. Study of the phenotypic and genotypic biodiversity of potentially ochratoxigenic black aspergilli isolated from grapes. Int. J. Food Microbiol. 2009, 132, 14–23. [Google Scholar] [CrossRef]
- Merlera, G.G.; Muñoz, S.; Coelho, I.; Cavaglieri, L.R.; Torres, A.M.; Reynoso, M.M. Diversity of black Aspergilli isolated from raisins in Argentina: Polyphasic approach to species identification and development of SCAR markers for Aspergillus ibericus. Int. J. Food Microbiol. 2015, 210, 92–101. [Google Scholar] [CrossRef]
- Hakobyan, L.; Grigoryan, K.; Kirakosyan, A. Contamination of raisin by filamentous fungi—Potential producers of ochratoxin A. Potravinarstvo 2010, 4, 28–33. [Google Scholar] [CrossRef]
- Palumbo, J.D.; O’Keeffe, T.L.; Kattan, A.; Abbas, H.K.; Johnson, B.J. Inhibition of Aspergillus flavus in soil by antagonistic Pseudomonas strains reduces the potential for airborne spore dispersal. Phytopathology 2010, 100, 532–538. [Google Scholar] [CrossRef]
- Palumbo, J.D.; O’Keeffe, T.L.; Ho, Y.S.; Fidelibus, M.W. Population dynamics of Aspergillus section Nigri species on vineyard samples of grapes and raisins. J. Food Prot. 2016, 79, 448–453. [Google Scholar] [CrossRef]
- Serra, R.; Mendonça, C.; Venâncio, A. Fungi and ochratoxin A detected in healthy grapes for wine production. Lett. Appl. Microbiol. 2006, 42, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Perrone, G.; De Girolamo, A.; Sarigiannis, Y.; Haidukowski, M.E.; Visconti, A. Occurrence of ochratoxin A, fumonisin B2 and black aspergilli in raisins from Western Greece regions in relation to environmental and geographical factors. Food Addit. Contam. Part A 2013, 30, 1339–1347. [Google Scholar] [CrossRef] [PubMed]
- Chebil, S.; Oueslati, S.; Ben-Amar, A.; Natskoulis, P. Ochratoxigenic fungi and Ochratoxin A determination in dried grapes marketed in Tunisia. Ann. Microbiol. 2020, 70, 38. [Google Scholar] [CrossRef]
- Lucchetta, G.; Bazzo, I.; Cortivo, G.D.; Stringher, L.; Bellotto, D.; Borgo, M.; Angelini, E. Occurrence of black aspergilli and ochratoxin A on grapes in Italy. Toxins 2010, 2, 840–855. [Google Scholar] [CrossRef] [PubMed]
- Frisvad, J.C.; Nielsen, K.F.; Samson, R.A. Recommendations concerning the chronic problem of misidentification of mycotoxigenic fungi associated with foods and feeds. Adv. Exp. Med. Biol. 2006, 571, 33–46. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Thrane, U.; Samson, R.A.; Pitt, J.I. Important mycotoxins and the fungi which produce them. Adv. Exp. Med. Biol. 2006, 571, 3–31. [Google Scholar] [CrossRef] [PubMed]
- Ozer, H.; Oktay Basegmez, H.I.; Ozay, G. Mycotoxin risks and toxigenic fungi in date, prune and dried apricot among Mediterranean crops. Phytopathol. Mediterr. 2012, 51, 148–157. [Google Scholar] [CrossRef]
- Van der Merwe, K.J.; Steyn, P.S.; Fourie, L. 1304. Mycotoxins. Part II. The constitution of ochratoxins A, B, and C, metabolites of Aspergillus ochraceus Wilh. J. Chem. Soc. Perkin. Trans. 1965, 11, 7083–7088. [Google Scholar] [CrossRef]
- Fernández-Cruz, M.L.; Mansilla, M.L.; Tadeo, J.L. Mycotoxins in fruits and their processed products: Analysis, occurrence and health implications. J. Adv. Res. 2010, 1, 113–122. [Google Scholar] [CrossRef]
- O’Brien, E.; Dietrich, D.R. Ochratoxin A: The continuing enigma. Crit. Rev. Toxicol. 2005, 35, 33–60. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Manderville, R.A. Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef] [PubMed]
- Samson, R.A.; Hoekstra, E.S.; Frisvad, J.C. Introduction to Food and Airborne Fungi; Centraalbureau voor Schimmelcultures: Utrecht, The Netherlands, 2004; pp. 1–389. [Google Scholar]
- Varga, J.; Kozakiewicz, Z. Ochratoxin A in grapes and grape-derived products. Trends Food Sci. Technol. 2006, 17, 72–81. [Google Scholar] [CrossRef]
- RASFF Portal. Available online: https://webgate.ec.europa.eu/rasff-window/portal/?event=SearchForm&cleanSearch=1 (accessed on 19 December 2017).
- Asghar, M.A.; Ahmed, A.; Iqbal, J. Aflatoxins and ochratoxin A in export quality raisins collected from different areas of Pakistan. Food Addit. Contam. Part B 2016, 9, 51–58. [Google Scholar] [CrossRef]
- Heshmati, A.; Mozaffari Nejad, A.S. Ochratoxin A in dried grapes in Hamadan province, Iran. Food Addit. Contam. Part B 2015, 8, 255–259. [Google Scholar] [CrossRef]
- Kollia, E.; Kanapitsas, A.; Markaki, P. Occurrence of aflatoxin B1 and ochratoxin A in dried vine fruits from Greek market. Food Addit. Contam. Part B 2014, 7, 11–16. [Google Scholar] [CrossRef]
- Aksoy, U.; Eltem, R.; Meyvaci, K.B.; Altindisli, A.; Karabat, S. Five-year survey of ochratoxin A in processed sultanas from Turkey. Food Addit. Contam. 2007, 24, 292–296. [Google Scholar] [CrossRef]
- Blesa, J.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Factors affecting the presence of ochratoxin A in wines. Crit. Rev. Food Sci. Nutr. 2006, 46, 473–478. [Google Scholar] [CrossRef]
- Fanelli, F.; Cozzi, G.; Raiola, A.; Dini, I.; Mule, G.; Logrieco, A.F.; Ritieni, A. Raisins and currants as conventional nutraceuticals in Italian market: Natural occurrence of Ochratoxin A. J. Food Sci. 2017, 82, 2306–2312. [Google Scholar] [CrossRef]
- Imperato, R.; Campone, L.; Piccinelli, A.L.; Veneziano, A.; Rastrelli, L. Survey of aflatoxins and ochratoxin A contamination in food products imported in Italy. Food Control 2011, 22, 1905–1910. [Google Scholar] [CrossRef]
- Miraglia, M.; Brera, C. Assessment of dietary intake of ochratoxin A by the population of EU Member States — report of experts participating in Task 3.2.7. In Reports on Tasks for Scientific Cooperation; Directorate-General Health and Consumer Protection of the European Commission: Brussels, Belgium, 2002; pp. 69–86. Available online: http://ec.europa.eu/food/fs/scoop/index_en.html (accessed on 26 June 2020).
- Battilani, P.; Giorni, P.; Bertuzzi, T.; Formenti, S.; Pietri, A. Black aspergilli and ochratoxin A in grapes in Italy. Int. J. Food Microbiol. 2006, 111, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Battilani, P.; Magan, N.; Logrieco, A. European research on ochratoxin A in grapes and wine. Int. J. Food Microbiol. 2006, 111, 2–4. [Google Scholar] [CrossRef]
- Youssef, M.S.; Abo-Dahab, N.F.; Abou-Seidah, A.A. Mycobiota and mycotoxin contamination of dried raisins in Egypt. Afr. J. Mycol. Biotechnol. 2000, 8, 69–86. [Google Scholar]
- Somma, S.; Perrone, G.; Logrieco, A. Diversity of black Aspergilli and mycotoxin risks in grape, wine and dried vine fruits. Phytopathol. Mediterr. 2012, 51, 131–147. [Google Scholar] [CrossRef]
- Zimmerli, B.; Dick, R. Ochratoxin A in the table wine and grape-juice: Occurrence and risk assessment. Food Addit. Contam. 1996, 13, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.B.; Lee, M.; Kim, D.H.; Varga, J.; Frisvad, J.C.; Perrone, G.; Gomi, K.; Yamada, O.; Machida, M.; Houbraken, J.; et al. Aspergillus luchuensis, an industrially important black Aspergillus in East Asia. PLoS ONE 2013, 8, e63769. [Google Scholar] [CrossRef] [PubMed]
- Yamada, O.; Machida, M.; Hosoyama, A.; Goto, M.; Takahashi, T.; Futagami, T.; Yamagata, Y.; Takeuchi, M.; Kobayashi, T.; Koike, H.; et al. Genome sequence of Aspergillus luchuensis NBRC 4314. DNA Res. 2016, 23, 507–515. [Google Scholar] [CrossRef][Green Version]
- Cabañes, F.J.; Bragulat, M.R.; Castellá, G. Ochratoxin A producing species in the genus Penicillium. Toxins 2010, 2, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Chiotta, M.L.; Ponsone, M.L.; Combina, M.; Torres, A.M.; Chulze, S.N. Aspergillus section Nigri species isolated from different wine-grape growing regions in Argentina. Int. J. Food Microbiol. 2010, 136, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Cabañes, F.J.; Accensi, F.; Bragulat, M.L.; Abarca, M.L.; Castella, G.; Minguez, S.; Pons, A. What is the source of ochratoxin A in wine? Int. J. Food Microbiol. 2002, 79, 213–215. [Google Scholar] [CrossRef]
- Da Rocha Rosa, C.A.; Palacios, V.; Combina, M.; Fraga, M.E.; De Oliveira Rekson, A.; Magnoli, C.E.; Dalcero, A.M. Potential ochratoxin A producers from wine grapes in Argentina and Brazil. Food Addit. Contam. 2002, 19, 408–414. [Google Scholar] [CrossRef]
- Magnoli, C.; Violante, M.; Combina, M.; Palacio, G.; Dalcero, A. Mycoflora and ochratoxin-producing strains of Aspergillus section Nigri in wine grapes in Argentina. Lett. Appl. Microbiol. 2003, 37, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Serra, R.; Abrunhosa, L.; Kozakiewicz, Z.; Venancio, A. Black Aspergillus species as ochratoxin A producers in Portuguese wine grapes. Int. J. Food Microbiol. 2003, 88, 63–68. [Google Scholar] [CrossRef]
- Tjamos, S.E.; Antoniou, P.P.; Kazantzidou, A.; Antonopoulos, D.F.; Papageorgiou, I.; Tjamos, E.C. Aspergillus niger and Aspergillus carbonarius in Corinth raisin and wine-producing vineyards in Greece: Population composition, ochratoxin A production and chemical control. J. Phytopathol. 2004, 152, 250–255. [Google Scholar] [CrossRef]
- Abarca, M.L.; Accensi, F.; Bragulat, M.R.; Castellá, G.; Cabañes, F.J. Aspergillus carbonarius as the main source of ochratoxin A contamination in dried vine fruits from the Spanish market. J. Food Prot. 2000, 66, 504–506. [Google Scholar] [CrossRef] [PubMed]
- Chiotta, M.L.; Susca, A.; Stea, G.; Mule, G.; Perrone, G.; Logrieco, A.; Chulze, S.N. Phylogenetic characterization and ochratoxin A-fumonisin profile of black Aspergillus isolated from grapes in Argentina. Int. J. Food Microbiol. 2011, 15, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Choque, E.; Klopp, C.; Valiere, S.; Raynal, J.; Mathieu, F. Whole-genome sequencing of Aspergillus tubingensis G131 and overview of its secondary metabolism potential. BMC Genom. 2018, 19, 200. [Google Scholar] [CrossRef]
- Fotso, J.; Leslie, J.F.; Smith, S. Production of beauvericin, moniliformin, fusaproliferin, and fumonisins B1, B2, and B3 by fifteen ex-type strains of Fusarium species. Appl. Environ. Microbiol. 2002, 68, 5195–5197. [Google Scholar] [CrossRef]
- Han, X.; Jiang, H.; Xu, J.; Zhang, J.; Li, F. Dynamic Fumonisin B2 production by Aspergillus niger intented used in food industry in China. Toxins 2017, 9, 217. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Shinsky, T.J., White, T.J., Eds.; Academic Press Inc.: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Hong, S.B.; Go, S.J.; Shin, H.D. Polyphasic taxonomy of Aspergillus fumigatus and related species. Mycologia 2005, 97, 1316–1329. [Google Scholar] [CrossRef]
- Glass, L.; Donaldson, G.C. Development of premier sets designed for use with the PCR to amplify conserved genes from filamentous Ascomycetes. Appl. Environ. Microb. 1995, 61, 1323–1330. [Google Scholar] [CrossRef]
- Katoh, K.; Misawa, K.; Kuma, K.; Miyata, T. MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef] [PubMed]
- Talavera, G.; Castresana, J. Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst. Biol. 2007, 56, 564–577. [Google Scholar] [CrossRef] [PubMed]
- Huson, D.H.; Bryant, D. Application of phylogenetic networks in evolutionary studies. Mol. Biol. Evol. 2006, 23, 254–267. [Google Scholar] [CrossRef] [PubMed]
- Lanfear, R.; Frandsen, P.B.; Wright, A.M.; Senfeld, T.; Calcott, B. PartitionFinder 2: New methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol. Biol. Evol. 2016, 34, 772–773. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and postanalysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In Proceedings of the Gateway Computing Environments Workshop (GCE), New Orleans, LA, USA, 14 November 2010; pp. 1–8. [Google Scholar] [CrossRef]
- Bragulat, M.R.; Abarca, M.L.; Cabañes, F.J. An easy screening method for fungi producing ochratoxin A in pure culture. Int. J. Food Microbiol. 2001, 71, 139–144. [Google Scholar] [CrossRef]
- da Cruz, C.L.; Delgado, J.; Patriarca, A.; Rodríguez, A. Differential response to synthetic and natural antifungals by Alternaria tenuissima in wheat simulating media: Growth, mycotoxin production and expression of a gene related to cell wall integrity. Int. J. Food Microbiol. 2019, 292, 48–55. [Google Scholar] [CrossRef]
- Castaldo, L.; Graziani, G.; Gaspari, A.; Izzo, L.; Tolosa, J.; Rodríguez-Carrasco, Y.; Ritieni, A. Target analysis and retrospective screening of multiple mycotoxins in pet food using UHPLC-Q-Orbitrap HRMS. Toxins 2019, 11, 434. [Google Scholar] [CrossRef]
| Sample ID | Country of Origin | Level of OTA | Colony Forming Units | Occurrence of Aspergillus spp. within CFU | Aspergillus Sect. Nigri | Strain Code | Another Fungal Taxa Isolated |
|---|---|---|---|---|---|---|---|
| (µg/kg) | (CFU)/g | ||||||
| DVF_01/2016 | Chile | b.d.l. | 2.2 × 103 | 84% | A. niger | G_209 | Penicillium sp. |
| A. tubingensis | G_45 | Rhizopus sp. | |||||
| G_178 | |||||||
| G_211 | |||||||
| DVF_02/2016 | Iran | b.d.l. | 5.4 × 102 | 100% | A. tubingensis | G_180 | - |
| G_182 | |||||||
| DVF_03/2016 | Iran | 1.7 | 2.2 × 103 | 88% | A. niger | G_33 | Aspergillus flavus |
| A. welwitschiae | G_36 | Rhizopus sp. | |||||
| DVF_04/2016 | Chile | 1.6 | 9 × 102 | 80% | A. luchuensis | G_202 | Rhizopus sp. |
| G_201 | |||||||
| G_204 | |||||||
| A. tubingensis | G_203 | ||||||
| DVF_05/2016 | Czech Republic | 1.6 | 1.4 × 103 | 88% | A. luchuensis | G_198 | Cladosporium cladosporioides |
| G_199 | Penicillium sp. | ||||||
| A. welwitschiae | G_196 | ||||||
| DVF_06/2016 | Slovak Republic | b.d.l. | 9.9 × 102 | 100% | A. luchuensis | G_62 | - |
| A. niger | G_50 | ||||||
| DVF_07/2016 | Turkey | b.d.l. | 1.1 × 103 | 77% | A. tubingensis | G_132 | Penicillium sp. |
| Rhizopus sp. | |||||||
| Saccharomyces sp. | |||||||
| DVF_08/2016 | Turkey | 1.6 | 2.7 × 102 | 0% | - | - | Aspergillus flavus |
| Cladosporium cladosporioides | |||||||
| Trichothecium roseum | |||||||
| DVF_09/2016 | Chile | 10.5 | 6.3 × 102 | 14% | A. tubingensis | G_160 | Alternaria alternata |
| Paecilomyces sp. | |||||||
| Penicillium chrysogenum | |||||||
| Saccharomyces sp. | |||||||
| DVF_10/2016 | Chile | 1.8 | 1.2 × 103 | 42% | A. welwitschiae | G_166 | Rhizopus sp. |
| Saccharomyces sp. | |||||||
| DVF_11/2016 | unknown | 0.8 | 0 | 0% | - | - | - |
| DVF_12/2016 | Iran | 1.8 | 3.6 × 103 | 10% | A. tubingensis | G_168 | - |
| DVF_13/2016 | Chile | 10.6 | 4.5 × 102 | 40% | A. tubingensis | G_171 | Penicillium sp. |
| G_172 | Saccharomyces sp. | ||||||
| DVF_14/2016 | RSA | 2.5 | 6.2 × 102 | 71% | A. tubingensis | G_183 | Saccharomyces sp. |
| G_184 | |||||||
| DVF_15/2016 | Iran | 1.2 | 1.9 × 103 | 91% | A. carbonarius | G_187 | Rhizopus sp. |
| A. tubingensis | G_188 | ||||||
| DVF_16/2016 | Turkey | 1.8 | 1.9 × 103 | 91% | A. carbonarius | G_191 | Rhizopus sp. |
| A. tubingensis | G_190 | ||||||
| G_192 | |||||||
| DVF_17/2016 | Czech Republic | b.d.l. | 0 | 0% | - | - | - |
| DVF_18/2016 | Uzbekistan | 2.1 | 1.1 × 103 | 100% | A. tubingensis | G_174 | - |
| DVF_19/2016 | Chile | 3.9 | 9.9 × 102 | 100% | A. niger | G_210 | - |
| A. tubingensis | G_61 | ||||||
| DVF_20/2016 | unknown | 1.3 | 4.5 × 12 | 100% | A. tubingensis | G_176 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mikušová, P.; Caboň, M.; Melichárková, A.; Urík, M.; Ritieni, A.; Slovák, M. Genetic Diversity, Ochratoxin A and Fumonisin Profiles of Strains of Aspergillus Section Nigri Isolated from Dried Vine Fruits. Toxins 2020, 12, 592. https://doi.org/10.3390/toxins12090592
Mikušová P, Caboň M, Melichárková A, Urík M, Ritieni A, Slovák M. Genetic Diversity, Ochratoxin A and Fumonisin Profiles of Strains of Aspergillus Section Nigri Isolated from Dried Vine Fruits. Toxins. 2020; 12(9):592. https://doi.org/10.3390/toxins12090592
Chicago/Turabian StyleMikušová, Petra, Miroslav Caboň, Andrea Melichárková, Martin Urík, Alberto Ritieni, and Marek Slovák. 2020. "Genetic Diversity, Ochratoxin A and Fumonisin Profiles of Strains of Aspergillus Section Nigri Isolated from Dried Vine Fruits" Toxins 12, no. 9: 592. https://doi.org/10.3390/toxins12090592
APA StyleMikušová, P., Caboň, M., Melichárková, A., Urík, M., Ritieni, A., & Slovák, M. (2020). Genetic Diversity, Ochratoxin A and Fumonisin Profiles of Strains of Aspergillus Section Nigri Isolated from Dried Vine Fruits. Toxins, 12(9), 592. https://doi.org/10.3390/toxins12090592






