Potential of Cry10Aa and Cyt2Ba, Two Minority δ-endotoxins Produced by Bacillus thuringiensis ser. israelensis, for the Control of Aedes aegypti Larvae
Abstract
1. Introduction
2. Results
2.1. Insecticidal Cry and Cyt Genes Identified in Bti Strain from VectoBac-12AS®
2.2. Cloning of Cyt2Ba, Cry10Aa and Cry11Aa
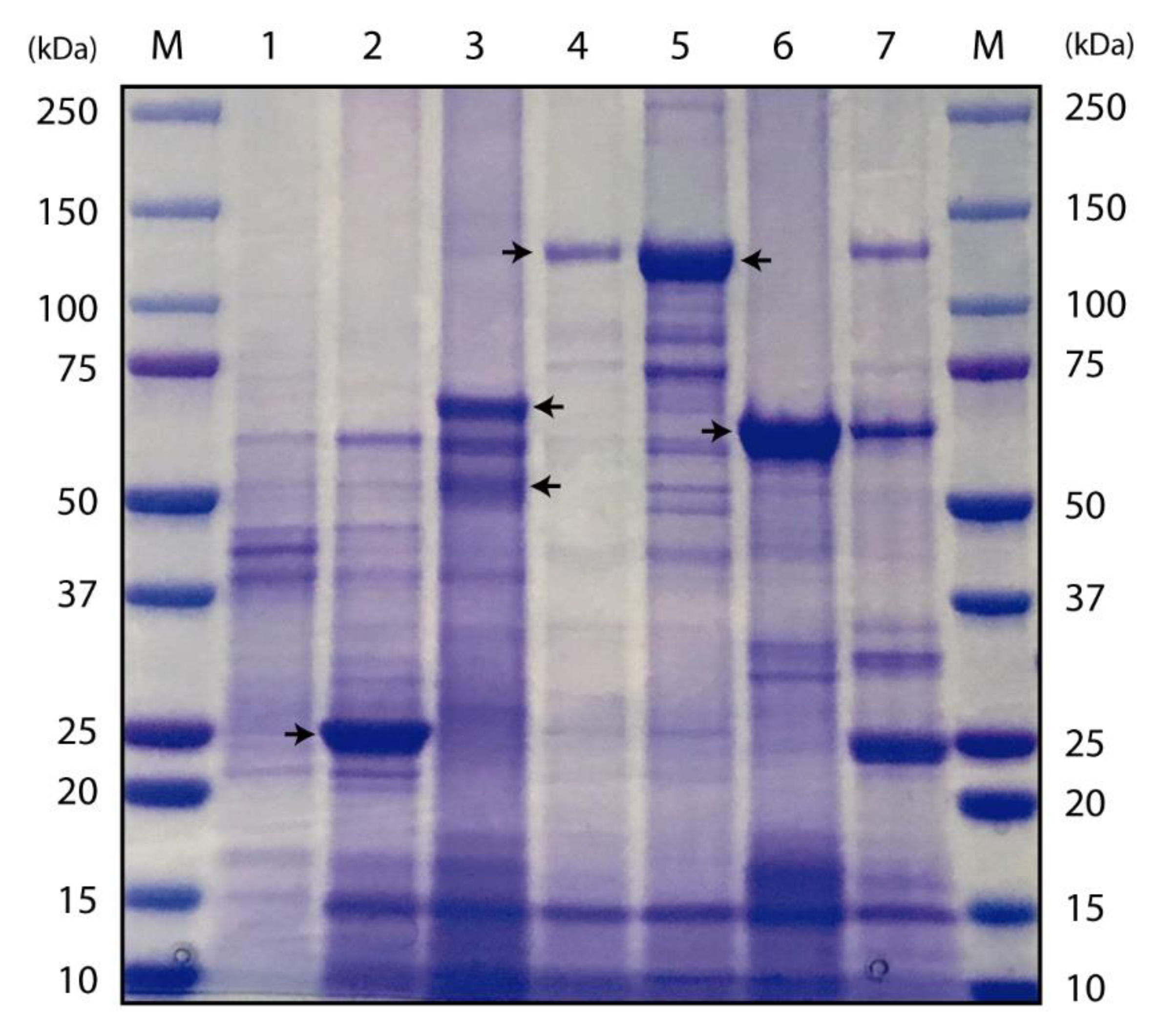
2.3. Characterization of Bt Recombinant Strains Expressing cyt2Ba, cry10Aa, cry4Aa, cry4Ba and cry11Aa
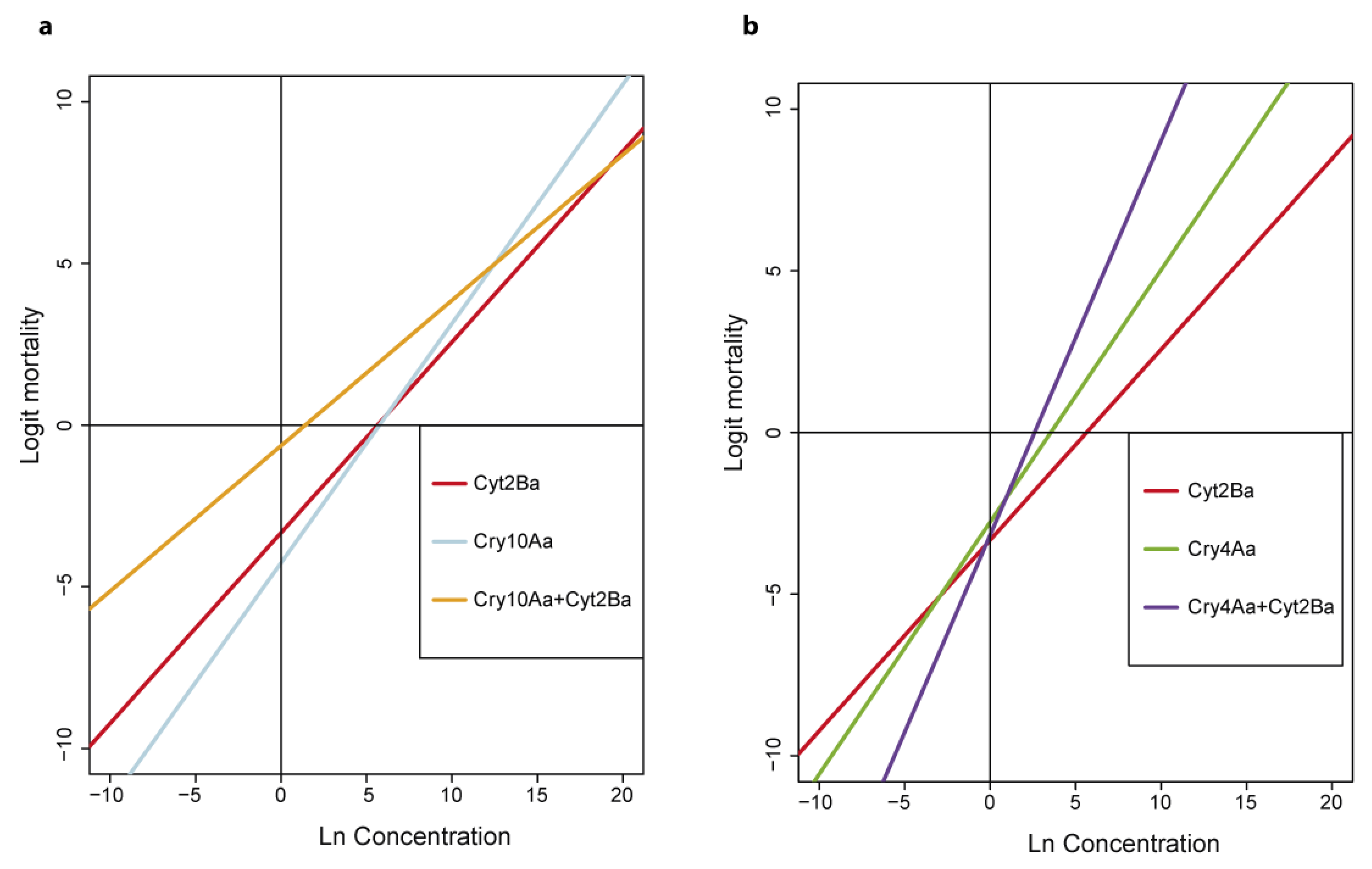
2.4. Mosquitocidal Activity of the δ-Endotoxins Produced by Bti
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Bacterial Strains and Plasmids
5.2. Insect Culture
5.3. Total DNA Extraction and Genomic Sequencing
5.4. Identification of Cry and Cyt Insecticidal Genes in VectoBac-12AS®
5.5. Amplification, Cloning and Sequencing of Cyt2Ba, Cry11Aa and Cry10Aa
5.6. Expression of Cyt2Ba, Cry10Aa, Cry4Aa, Cry4Ba and Cry11Aa Recombinant Proteins and SDS-PAGE Analysis
5.7. Mosquitocidal Activity of the δ-Endotoxins Produced by Bti
5.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Goldberg, L.J.; Margalith, J. A bacterial spore demonstrating rapid larvicidal activity against Anopheles sergentii, Uranotaenia unguiculata, Culex univitattus, Aedes aegypti and Culex pipiens. Mosq. News 1977, 1, 355–362. [Google Scholar]
- Federici, B.A.; Park, H.; Bideshi, D.K. Overview of the basic biology of Bacillus thuringiensis with emphasis on genetic engineering of bacterial larvicides for mosquito control. Open Toxinol. J. 2010, 3, 154–171. [Google Scholar] [CrossRef]
- Margalith, Y.; Ben-Dov, E. Biological control by Bacillus thuringiensis subsp. israelensis. In Insect Pest Management: Techniques for Environmental Protection; Recheigl, J.E., Recheigl, N.A., Eds.; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Lacey, L.A. Bacillus thuringiensis serovariety israelensis and Bacillus sphaericus for mosquito control. J. Am. Mosq. Control Assoc. 2007, 23, 133–163. [Google Scholar] [PubMed]
- Ben-Dov, E. Bacillus thuringiensis subsp. israelensis and its dipteran-specific toxins. Toxins 2014, 6, 1222–1243. [Google Scholar] [PubMed]
- Boyce, R.; Lenhart, A.; Kroeger, A.; Velayudhan, R.; Roberts, B.; Horstick, O. Bacillus thuringiensis israelensis (Bti) for the control of dengue vectors: Systematic literature review. Trop. Med. Int. Health 2013, 18, 564–577. [Google Scholar]
- Paris, M.; Tetreau, G.; Laurent, F.; Lelu, M.; Despres, L.; David, J.P. Persistence of Bacillus thuringiensis israelensis (Bti) in the environment induces resistance to multiple Bti toxins in mosquitoes. Pest Manag. Sci. 2011, 67, 122–128. [Google Scholar]
- Vasquez, M.I.; Violaris, M.; Hadjivassilis, A.; Wirth, M.C. Susceptibility of Culex pipiens (Diptera: Culicidae) field populations in cyprus to conventional organic insecticides, Bacillus thuringiensis subsp. israelensis, and methoprene. J. Med. Entomol. 2009, 46, 881–887. [Google Scholar] [CrossRef]
- Cantón, P.E.; Reyes, E.Z.; De Escudero, I.R.; Bravo, A.; Soberón, M. Binding of Bacillus thuringiensis subsp. israelensis Cry4Ba to Cyt1Aa has an important role in synergism. Peptides 2011, 32, 595–600. [Google Scholar]
- Wirth, M.C.; Park, H.W.; Walton, W.E.; Federici, B.A. Cyt1A of Bacillus thuringiensis delays evolution of resistance to Cry11A in the mosquito Culex quinquefasciatus. Appl. Environ. Microbiol. 2005, 71, 185–189. [Google Scholar] [CrossRef]
- Wirth, M.C.; Georghiou, G.P.; Federici, B.A. CytA enables CryIV endotoxins of Bacillus thuringiensis to overcome high levels of CryIV resistance in the mosquito, Culex quinquefasciatus. Proc. Natl. Acad. Sci. USA 1997, 94, 10536–10540. [Google Scholar] [CrossRef]
- Ibarra, J.E.; Federici, B.A. Isolation of a relatively nontoxic 65-kilodalton protein inclusion from the parasporal body of Bacillus thuringiensis subsp. israelensis. J. Bacteriol. 1986, 165, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Eckblad, W.; Lee, A.B. Diversity of protein inclusion bodies and identification of mosquitocidal protein in Bacillus thuringiensis subsp. israelensis. Biochem. Biophys. Res. Commun. 1985, 126, 953–960. [Google Scholar] [CrossRef]
- Garduno, F.; Thorne, L.; Walfield, A.M.; Pollock, T.J. Structural relatedness between mosquitocidal endotoxins of Bacillus thuringiensis subsp. israelensis. Appl. Environ. Microbiol. 1988, 54, 277–279. [Google Scholar] [CrossRef] [PubMed]
- Berry, C.; Ben-dov, E.; Jones, A.F.; Murphy, L.; Quail, M.A.; Holden, M.T.G.; Harris, D.; Zaritsky, A.; Parkhill, J. Complete sequence and organization of pBtoxis, the toxin-coding plasmid of Bacillus thuringiensis subsp. israelensis. Appl. Environ. Microbiol. 2002, 68, 5082–5095. [Google Scholar] [CrossRef] [PubMed]
- Thorne, L.; Garduno, F.; Thompson, T.; Decker, D.; Zounes, M.; Wild, M.; Waldfield, A.; Pollock, T.J. Structural similarity between the Lepidoptera- and Diptera-specific insecticidal endotoxin genes of Bacillus thuringiensis subsp. “kurstaki” and “israelensis.”. J. Bacteriol. 1986, 166, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Soto, A.; Del Rincón-Castro, M.C.; Espinoza, A.M.; Ibarra, J.E. Parasporal body formation via overexpression of the Cry10Aa toxin of Bacillus thuringiensis subsp. israelensis, and Cry10Aa-Cyt1Aa synergism. Appl. Environ. Microbiol. 2009, 75, 4661–4667. [Google Scholar]
- Guerchicoff, A.; Ugalde, R.A.; Rubinstein, C.P. Identification and characterization of a previously undescribed cyt gene in Bacillus thuringiensis subsp. israelensis. Appl. Environ. Microbiol. 1997, 63, 2716–2721. [Google Scholar] [CrossRef]
- Juárez-Pérez, V.; Guerchicoff, A.; Rubinstein, C.; Delecluse, A. Characterization of Cyt2Bc toxin from Bacillus thuringiensis subsp. medellin. Appl. Environ. Microbiol. 2002, 68, 1228–1231. [Google Scholar]
- Crickmore, N.; Bone, E.J.; Williams, J.A.; Ellar, D.J. Contribution of the individual components of the δ-endotoxin crystal to the mosquitocidal activity of Bacillus thuringiensis subsp. israelensis. FEMS Microbiol. Lett. 1995, 131, 249–254. [Google Scholar] [CrossRef]
- Manasherob, R.; Itsko, M.; Sela-Baranes, N.; Ben-Dov, E.; Berry, C.; Cohen, S.; Zaritsky, A. Cyt1 Ca from Bacillus thuringiensis subsp. israelensis: Production in Escherichia coli and comparison of its biological activities with those of other Cyt-like proteins. Microbiology 2006, 152, 2651–2659. [Google Scholar] [CrossRef]
- Otieno-Ayayo, Z.N.; Zaritsky, A.; Wirth, M.C.; Manasherob, R.; Khasdan, V.; Cahan, R.; Ben-Dov, E. Variations in the mosquito larvicidal activities of toxins from Bacillus thuringiensis ssp. israelensis. Environ. Microbiol. 2008, 10, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Poncet, S.; Delecluse, A.; Klier, A.; Rapoport, G. Evaluation of synergistic interactions among CryIVA, CryIVB, and CryIVD toxic components of B. thuringiensis subsp. israelensis crystals. J. Invertebr. Pathol. 1995, 66, 131–135. [Google Scholar] [CrossRef]
- Wu, D.; Johnson, J.J.; Federici, B.A. Synergism of mosquitocidal toxicity between CytA and CryIVD proteins using inclusions produced from cloned genes of Bacillus thuringiensis. Mol. Microbiol. 1994, 13, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Wirth, M.C.; Georghiou, G.P.; Malik, J.I.; Abro, G.H. Laboratory selection for resistance to Bacillus sphaericus in Culex quinquefasciatus (Diptera: Culicidae) from California, USA. J. Med. Entomol. 2000, 37, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Donovan, W.P.; Dankocsik, C.; Gilbert, M.P. Molecular characterization of a gene encoding a 72-kilodalton mosquito-toxic crystal protein from Bacillus thuringiensis subsp. israelensis. J. Bacteriol. 1988, 170, 4732–4738. [Google Scholar] [CrossRef] [PubMed]
- Anderson, I.; Sorokin, A.; Kapatral, V.; Reznik, G.; Bhattacharya, A.; Mikhailova, N.; Burd, H.; Joukov, V.; Kaznadzey, D.; Walunas, T.; et al. Comparative genome analysis of Bacillus cereus group genomes with Bacillus subtilis. FEMS Microbiol. Lett. 2005, 250, 175–184. [Google Scholar] [CrossRef]
- Waalwijlc, C.; Dullemans, A.M.; Van Workum, M.E.S.; Visser, B. Molecular cloning and the nucleotide sequence of the Mr 28 000 crystal protein gene of Bacillus thuringiensis subsp. israelensis. Nucleic Acids Res. 1985, 13, 5753–5763. [Google Scholar]
- Wu, D.; Federici, B.A. Improved production of the insecticidal CryIVD protein in Bacillus thuringiensis using cryIA(c) promoters to express the gene for an associated 20 kDa protein. Appl. Microbiol. Biotechnol. 1995, 42, 697–702. [Google Scholar] [CrossRef]
- Delecluse, A.; Poncet, S.; Klier, A.; Rapoport, G. Expression of cryIVA and cryIVB genes, independently or in combination, in a crystal-negative strain of Bacillus thuningiensis subsp. israelensis. Appl. Environ. Microbiol. 1993, 59, 3922–3927. [Google Scholar] [CrossRef]
- Tabashnik, B.E. Evaluation of synergism among Bacillus thuringiensis toxins. Appl. Environ. Microbiol. 1992, 58, 3343–3346. [Google Scholar] [CrossRef]
- Stiles, B.; Paschke, J.D. Midgut pH in different instars of three Aedes mosquito species and the relation between pH and susceptibility of larvae to a nuclear polyhedrosis virus. J. Invertebr. Pathol. 1980, 35, 58–64. [Google Scholar] [CrossRef]
- McLaughlin, R.E.; Dulmage, H.T.; Alls, R.; Couch, T.L.; Dame, D.A.; Hall, I.M.; Rose, R.I.; Versoi, P.L. U.S. standard bioassay for the potency assessment of Bacillus thuringiensis serotype H-14 against mosquito larvae. Bull. Entomol. Soc. Am. 1984, 30, 26–29. [Google Scholar] [CrossRef]
- Zehnder, G.W.; Gelernter, W.D. Activity of the M-ONE formulation of a new strain of Bacillus thuringiensis against the colorado potato beetle (Coleoptera: Chrysomelidae): Relationship between susceptibility and insect life stage. J. Econ. Entomol. 1989, 82, 756–761. [Google Scholar] [CrossRef]
- Nisnevitch, M.; Cohen, S.; Ben-Dov, E.; Zaritsky, A.; Sofer, Y.; Cahan, R. Cyt2Ba of Bacillus thuringiensis israelensis: Activation by putative endogenous protease. Biochem. Biophys. Res. Commun. 2006, 344, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Wirth, M.C.; Delecluse, A.; Walton, W.E. Cyt1Ab1 and Cyt2Ba1 from Bacillus thuringiensis subsp. medellin and B. thuringiensis subsp. israelensis synergize Bacillus sphaericus against Aedes aegypti and resistant Culex quinquefasciatus (Diptera: Culicidae). Appl. Environ. Microbiol. 2001, 67, 3280–3284. [Google Scholar] [CrossRef]
- Delécluse, A.; Bourgouin, C.; Klier, A.; Rapoport, G. Specificity of action on mosquito larvae of Bacillus thuringiensis israelensis toxins encoded by two different genes. MGG Mol. Gen. Genet. 1988, 214, 42–47. [Google Scholar] [CrossRef]
- Khasdan, V.; Ben-Dov, E.; Manasherob, R.; Boussiba, S.; Zaritsky, A. Toxicity and synergism in transgenic Escherichia coli expressing four genes from Bacillus thuringiensis subsp. israelensis. Environ. Microbiol. 2001, 3, 798–806. [Google Scholar] [CrossRef]
- Promdonkoy, B.; Promdonkoy, P.; Panyim, S. Co-expression of Bacillus thuringiensis Cry4Ba and Cyt2Aa2 in Escherichia coli revealed high synergism against Aedes aegypti and Culex quinquefasciatus larvae. FEMS Microbiol. Lett. 2005, 252, 121–126. [Google Scholar] [CrossRef]
- Lailak, C.; Khaokhiew, T.; Promptmas, C.; Promdonkoy, B.; Pootanakit, K.; Angsuthanasombat, C. Bacillus thuringiensis Cry4Ba toxin employs two receptor-binding loops for synergistic interactions with Cyt2Aa2. Biochem. Biophys. Res. Commun. 2013, 435, 216–221. [Google Scholar] [CrossRef]
- López-Diaz, J.A.; Cantón, P.E.; Gill, S.S.; Soberón, M.; Bravo, A. Oligomerization is a key step in Cyt1Aa membrane insertion and toxicity but not necessary to synergize Cry11Aa toxicity in Aedes aegypti larvae. Environ. Microbiol. 2013, 15, 3030–3039. [Google Scholar] [CrossRef]
- Perez, C.; Fernandez, L.E.; Sun, J.; Folch, J.L.; Gill, S.S.; Soberon, M.; Bravo, A. Bacillus thuringiensis subsp. israelensis Cyt1Aa synergizes Cry11Aa toxin by functioning as a membrane-bound receptor. Proc. Natl. Acad. Sci. USA 2005, 102, 18303–18308. [Google Scholar] [PubMed]
- Pérez, C.; Muñoz-Garay, C.; Portugal, L.C.; Sánchez, J.; Gill, S.S.; Soberón, M.; Bravo, A. Bacillus thuringiensis ssp. israelensis Cyt1Aa enhances activity of Cry11Aa toxin by facilitating the formation of a pre-pore oligomeric structure. Cell. Microbiol. 2007, 9, 2931–2937. [Google Scholar]
- Anaya, P.; Onofre, J.; Torres-Quintero, M.C.; Sánchez, J.; Gill, S.S.; Bravo, A.; Soberón, M. Oligomerization is a key step for Bacillus thuringiensis Cyt1Aa insecticidal activity but not for toxicity against red blood cells. Insect Biochem. Mol. Biol. 2020, 119, 103317. [Google Scholar] [CrossRef] [PubMed]
- Park, H.W.; Ge, B.; Bauer, L.S.; Federici, B.A. Optimization of Cry3A yields in Bacillus thuringiensis by use of sporulation-dependent promoters in combination with the STAB-SD mRNA sequence. Appl. Environ. Microbiol. 1998, 64, 3932–3938. [Google Scholar] [PubMed]
- Li, L.; Yang, C.; Liu, Z.; Li, F.; Yu, Z. Screening of acrystalliferous mutants from Bacillus thuringiensis and their transformation properties. Wei Sheng Wu Xue Bao. 2000, 40, 85–90. [Google Scholar]
- Stewart, S.D.; Adamczyk, J.J.; Knighten, K.S.; Davis, F.M. Impact of Bt cottons expressing one or two insecticidal proteins of Bacillus thuringiensis Berliner on growth and survival of Noctuid (Lepidoptera) larvae. J. Econ. Entomol. 2001, 94, 752–760. [Google Scholar] [CrossRef]
- Borodovsky, M.; McIninch, J. GENMARK: Parallel gene recognition for both DNA strands. Comput. Chem. 1993, 17, 123–133. [Google Scholar]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Crickmore, N.; Zeigler, D.R.; Feitelson, J.; Schnepf, E.; Van Rie, J.; Lereclus, D.; Baum, J.; Dean, D.H. Revision of the nomenclature for the Bacillus thuringiensis pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 807–813. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D. Molecular Cloning: A laboratory Manual, 3rd ed.; Harbor, C.S., Ed.; Cold Spring Harbor Laboratory: New York, NY, USA, 2001. [Google Scholar]
- Dominguez-Arrizabalaga, M.; Villanueva, M.; Fernandez, A.B.; Caballero, P. A strain of Bacillus thuringiensis containing a novel cry7Aa2 gene that is toxic to Leptinotarsa desemlineata (Say) (Coleoptera: Chrysomelidae). Insects 2019, 10, 259. [Google Scholar] [CrossRef]
- Lee, J.C. Electrotransformation of Staphylococci. Electroporation Protoc. Microorg. 1995, 47, 209–216. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of Bacteriophage T4. Nature 1970, 225, 680–685. [Google Scholar] [CrossRef] [PubMed]
| Treatment 1 | Concentration (ng/mL) | Mortality (% ± SD) |
|---|---|---|
| Cry10Aa | 40 | 26 ± 5 |
| Cyt2Ba | 40 | 31 ± 8 |
| Cry10Aa+Cyt2Ba (1:1) | 80 | 93 ± 6 |
| Cry4Aa | 10 | 31 ± 10 |
| Cyt2Ba | 10 | 28 ± 23 |
| Cry4Aa+Cyt2Ba (1:1) | 20 | 100 ± 0 |
| Cry4Ba | 0.02 | 32 ± 18 |
| Cyt2Ba | 0.02 | 10 ± 9 |
| Cry4Ba+Cyt2Ba (1:1) | 0.04 | 43 ± 19 |
| Cry11Aa | 1.5 | 40 ± 16 |
| Cyt2Ba | 1.5 | 21 ± 14 |
| Cry11Aa+Cyt2Ba (1:1) | 3 | 46 ± 21 |
| Cry10Aa | Cyt2Ba | Cry10Aa+Cyt2Ba | ||||||
|---|---|---|---|---|---|---|---|---|
| ng/mL | Dead/Total | Mortality (% ± SD) | ng/mL | Dead/Total | Mortality (% ± SD) | ng/mL | Dead/Total | Mortality (% ± SD) |
| 2000 | 39/50 | 78 ± 2% | 4000 | 84/92 | 91.3 ± 10% | 300 | 115/124 | 92.7 ± 9% |
| 666 | 30/50 | 60 ± 10% | 1333 | 64/85 | 75.3 ± 17% | 60 | 86/114 | 75.4 ± 9% |
| 222 | 23/41 | 56.1 ± 2% | 444 | 39/94 | 41.5 ± 9% | 12 | 75/135 | 55.6 ± 11% |
| 74 | 12/44 | 27.3 ± 12% | 148 | 28/78 | 35.9 ± 7% | 2.4 | 53/131 | 40.5 ± 6% |
| 24.7 | 6/46 | 13 ± 17% | 49.4 | 22/86 | 25.6 ± 6% | 0.48 | 34/116 | 29.3 ± 6% |
| 8.2 | 1/42 | 2.4 ± 3% | 16.4 | 21/84 | 25 ± 13% | 0.096 | 23/120 | 19.2 ± 14% |
| Cry4Aa | Cyt2Ba | Cry4Aa+Cyt2Ba | ||||||
|---|---|---|---|---|---|---|---|---|
| ng/mL | Dead/Total | Mortality (% ± SD) | ng/mL | Dead/Total | Mortality (% ± SD) | ng/mL | Dead/Total | Mortality (% ± SD) |
| 486 | 63/70 | 90 ± 11% | 4000 | 84/92 | 91.3 ± 10% | 54 | 139/163 | 85.3 ± 13% |
| 162 | 53/71 | 74.6 ± 17% | 1333 | 64/85 | 75.3 ± 17% | 27 | 111/155 | 71.6 ± 17% |
| 54 | 42/70 | 60 ± 13% | 444 | 39/94 | 41.5 ± 9% | 13.5 | 87/200 | 43.5 ± 14% |
| 18 | 27/73 | 37 ± 11% | 148 | 28/78 | 35.9 ± 7% | 6.74 | 58/142 | 40.8 ± 24% |
| 6 | 13/64 | 20.3 ± 10% | 49.4 | 22/86 | 25.6 ± 6% | 3.36 | 11/102 | 10.8 ± 16% |
| 2 | 7/71 | 9.9 ± 7% | 16.4 | 21/84 | 25 ± 13% | 1.68 | 5/73 | 6.8 ± 8% |
| Treatment (a) | Regression | LC50 Observed | FL (95%) (b) | LC50 Expected (c) | FL (95%) (b) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Slope ± SE | Intercept ± SE | (ng/mL) | Lower | Upper | (ng/mL) | Synergistic Factor (d) | Potency | Lower | Upper | |
| Cyt2Ba | 0.59 ± 0.13 | −3.33 ± 0.79 | 279.37 | 190.20 | 410.38 | - | - | 1 | - | - |
| Cry10Aa | 0.74 ± 0.09 | −4.26 ± 0.53 | 299.62 | 245.06 | 366.34 | - | - | 0.93 | 0.78 | 1.12 |
| Cry4Aa | 0.78 ± 0.02 | −2.77 ± 0.09 | 34.63 | 29.73 | 40.34 | - | - | 8.07 | 6.40 | 10.17 |
| Cry10Aa+Cyt2Ba (e) | 0.45 ± 0.05 | −0.64 ± 0.14 | 4.22 | 3.25 | 5.50 | 289.27 | 68.55 | 66.20 | 58.52 | 74.61 |
| Cry4Aa+Cyt2Ba (e) | 1.22 ± 0.17 | −3.17 ± 0.46 | 13.41 | 12.55 | 14.33 | 61.62 | 4.60 | 20.83 | 15.15 | 28.63 |
| VectoBac-12AS® | 1.51 ± 0.29 | 3.44 ± 0.73 | 1.02 × 10−1 | 9.34 × 10−2 | 1.11 × 10−1 | - | - | 2.73 × 103 | 2.03 × 103 | 3.69 × 103 |
| Primer Name | Primer Sequence | Reference |
|---|---|---|
| Cyt2B-Fw-XbaI | 5′-TTCTAGAGATAATGAAGGAGGGGAGTC-3′ | This study |
| Cyt2B-Rv-PstI | 5′-CCTGCAGCAAAATTAAATTGCTGAGTTACTATAATAAC-3′ | This study |
| Cry10A-Fw-SalI | 5′-ATGTCGACTTGCAACAGAAAAGAGTTGTGTC-3′ | [17] |
| Cry10A-Rv-PaeI | 5′-GAGCATGCACATTTCCCCACAATTTTCA-3′ | [17] |
| Cry10A-test-Fw | 5′-CGAAATTGTCAGACATAGAGAG-3′ | This study |
| Cry10A-test-Rv | 5′-GAATTACCAAGTCTCCACCTG-3′ | This study |
| p20-Fw-PstI | 5′-CCTGCAGGGATAAAATTGGAGGATAATTGATG-3′ | This study |
| p20-Rv-PaeI | 5′-GGCATGCGTTTCCAGTGCATTCAATTTAC-3′ | This study |
| p19-Fw-SalI | 5′-GTGTCGACGTTTTTTAAAATTGCATAGAAGGG-3′ | This study |
| Cry11A-Rv-PstI | 5′-CTCTGCAGGTGCTAACATGACTTCTACTTTAG-3′ | This study |
| Cry11A-test | 5′-GGTCATAATTTATGAATAAAAATATGAC-3′ | This study |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valtierra-de-Luis, D.; Villanueva, M.; Lai, L.; Williams, T.; Caballero, P. Potential of Cry10Aa and Cyt2Ba, Two Minority δ-endotoxins Produced by Bacillus thuringiensis ser. israelensis, for the Control of Aedes aegypti Larvae. Toxins 2020, 12, 355. https://doi.org/10.3390/toxins12060355
Valtierra-de-Luis D, Villanueva M, Lai L, Williams T, Caballero P. Potential of Cry10Aa and Cyt2Ba, Two Minority δ-endotoxins Produced by Bacillus thuringiensis ser. israelensis, for the Control of Aedes aegypti Larvae. Toxins. 2020; 12(6):355. https://doi.org/10.3390/toxins12060355
Chicago/Turabian StyleValtierra-de-Luis, Daniel, Maite Villanueva, Liliana Lai, Trevor Williams, and Primitivo Caballero. 2020. "Potential of Cry10Aa and Cyt2Ba, Two Minority δ-endotoxins Produced by Bacillus thuringiensis ser. israelensis, for the Control of Aedes aegypti Larvae" Toxins 12, no. 6: 355. https://doi.org/10.3390/toxins12060355
APA StyleValtierra-de-Luis, D., Villanueva, M., Lai, L., Williams, T., & Caballero, P. (2020). Potential of Cry10Aa and Cyt2Ba, Two Minority δ-endotoxins Produced by Bacillus thuringiensis ser. israelensis, for the Control of Aedes aegypti Larvae. Toxins, 12(6), 355. https://doi.org/10.3390/toxins12060355