Aflatoxin Reduction in Maize by Industrial-Scale Cleaning Solutions
Abstract
1. Introduction
2. Results
2.1. First Case Study
2.1.1. Trial #1 (Separator–Aspirator–Optical Sorter)
2.1.2. Trial #2 (Separator–Aspirator–Concentrator–Optical Sorter)
2.2. Second Case Study
2.2.1. Trial #3 (Separator–Aspirator)
2.2.2. Trial #4 (Concentrator)
2.2.3. Trials #5 and #6 (Optical Sorter)
2.3. Mass Balance
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Samples and Cleaning Processes
5.2. Sampling
5.3. Aflatoxin Analysis
5.4. Mass Balance
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Munkvold, G.P.; Arias, S.; Taschl, I.; Gruber-Dorninger, C. Mycotoxins in Corn: Occurrence, Impacts, and Management. In Corn: Chemistry and Technology, 3th ed.; Serna-Saldivar, S.O., Ed.; Elsevier, Woodhead Publishing: Sawston, UK, 2019; pp. 235–287. [Google Scholar] [CrossRef]
- IARC Working Group. Human exposure to aflatoxins and fumonisins. In Mycotoxin Control in Low- and Middle-Income Countries; REPORT NO. 9; Wild, C.P., Miller, J.D., Groopman, J.D., Eds.; International Agency for Research on Cancer: Lyon, France, 2015; pp. 1–5. ISBN 978-92-832-2510-2. [Google Scholar]
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Global Mycotoxin Occurrence in Feed: A Ten-Year Survey. Toxins 2019, 11, 375. [Google Scholar] [CrossRef] [PubMed]
- Doerge, D.R.; Shephard, G.S.; Adegoke, G.O.; Benford, D.; Bhatnagar, D.; Bolger, M.; Boon, P.E.; Cressey, P.; Edwards, S.; Hambridge, T.; et al. Aflatoxins. In Safety Evaluation of Certain Contaminants in Food: Prepared by the Eighty-Third Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA); WHO Food Additives Series, No. 74; FAO JECFA Monographs 19 bis; World Health Organization and Food and Agriculture Organization of the United Nations: Geneva, Switzerland, 2018; pp. 3–279. [Google Scholar]
- Strosnider, H.; Azziz-Baumgartner, E.; Banziger, M.; Bhat, R.V.; Breiman, R.; Brune, M.N.; DeCock, K.; Dilley, A.; Groopman, J.; Hell, K.; et al. Workgroup report: Public health strategies for reducing aflatoxin exposure in developing countries. Environ. Health Perspect. 2006, 114, 1898–1903. [Google Scholar] [CrossRef] [PubMed]
- Njapau, H.; Lewis, L.L. Aflatoxin and the acute aflatoxicosis outbreaks in Kenya: A review. JSM Mycotoxins 2007, 2006 (Suppl. 4), 73–85. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. IARC Monographs on the Evaluation of Carcinogenic risk to Humans; Some traditional herbal medicines, some mycotoxins, naphthalene and styrene; IARC Press: Lyon, France, 2002; Volume 82, pp. 171–300. [Google Scholar]
- The European Commission. Commission Regulation (EU) No 165/2010 of 26 February 2010 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards aflatoxins. Off. J. Eur. Union 2010, 50, 8–12. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:050:0008:0012:EN:PDF (accessed on 24 March 2020).
- Battilani, P.; Toscano, P.; Van der Fels-Klerx, H.J.; Moretti, A.; Camardo Leggieri, M.; Brera, C.; Rortais, A.; Goumperis, T.; Robinson, T. Aflatoxin B1 contamination in maize in Europe increases due to climate change. Sci. Rep. 2016, 6, 24328. [Google Scholar] [CrossRef] [PubMed]
- Udovicki, B.; Audenaert, K.; De Saeger, S.; Rajkovic, A. Overview on the Mycotoxins Incidence in Serbia in the Period 2004–2016. Toxins 2018, 10, 279. [Google Scholar] [CrossRef] [PubMed]
- CODEX CAC/RCP 51-2003 “Code of Practice for the Prevention and Reduction of Mycotoxin Contamination in Cereals”. Available online: http://www.fao.org/fao-who-codexalimentarius/codex-texts/codes-of-practice/en/ (accessed on 24 March 2020).
- The Commission of the European Communities. Commission Recommendation on prevention and reduction of Fusarium toxins in cereals. Off. J. Eur. Union 2006, 234, 35–40. [Google Scholar]
- Reyneri, A.; Bruno, G.; D’Egidio, M.G.; Balconi, C. Guidelines for the Control of Mycotoxins in Maize and Wheat, 2015, MIPAAF, Ministry of Agriculture, Food and Forestry Policies (in Italian). Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/9703 (accessed on 24 March 2020).
- Grenier, B.; Loureiro-Bracarense, A.-P.; Leslie, J.F.; Oswald, I.P. Physical and Chemical Methods for mycotoxin decontamination in maize. In Mycotoxin Reduction in Grain Chains; Leslie, J.F., Logrieco, A.F., Eds.; John Wiley and Sons, Inc: Ames, IA, USA, 2004; pp. 116–129. [Google Scholar]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef]
- Peng, W.X.; Marchal, J.L.M.; van der Poel, A.F.B. Strategies to prevent and reduce mycotoxins for compound feed manufacturing. Anim. Feed Sci. Technol. 2018, 237, 129–153. [Google Scholar] [CrossRef]
- Neme, K.; Mohammed, A. Mycotoxin occurrence in grains and the role of postharvest management as a mitigation strategies. A review. Food Control 2017, 78, 412–425. [Google Scholar] [CrossRef]
- Shi, H.; Stroshine, R.L.; Ileleji, K. Differences in kernel shape, size, and density between healthy kernels and mold discolored kernels and their relationship to reduction in aflatoxin levels in a sample of shelled corn. Appl. Eng. Agric. 2017, 33, 421–431. [Google Scholar] [CrossRef]
- Sydenham, E.W.; Van der Westhuizen, L.; Stockenström, S.; Shephard, G.S.; Thiel, P.G. Fumonisin-contaminated maize: Physical treatment for the partial decontamination of bulk shipments. Food Addit. Contam. 1994, 11, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Schaarschmidt, S.; Fauhl-Hassek, C. The Fate of Mycotoxins During the Processing of Wheat for Human Consumption. Compr. Rev. Food Sci. Food Saf. 2018, 17, 556–593. [Google Scholar] [CrossRef]
- Visconti, A.; Haidukowski, M.; Pascale, M.; Silvestri, M. Reduction of deoxynivalenol during durum wheat processing and spaghetti cooking. Toxicol. Lett. 2004, 153, 181–189. [Google Scholar] [CrossRef]
- Pascale, M.; Haidukowski, M.; Lattanzio, V.M.T.; Silvestri, M.; Ranieri, R.; Visconti, A. Distribution of T-2 and HT-2 Toxins in Milling Fractions of Durum Wheat. J. Food Prot. 2011, 74, 1700–1707. [Google Scholar] [CrossRef]
- Tibola, C.S.; Fernandes, J.M.C.; Guarienti, E.M. Effect of cleaning, sorting and milling processes in wheat mycotoxin content. Food Control 2016, 60, 174–179. [Google Scholar] [CrossRef]
- Trenholm, H.L.; Charmley, L.L.; Prelusky, D.B.; Warner, R.M. Two physical methods for the decontamination of four cereals contaminated with deoxynivalenol and zearalenone. J. Agric. Food Chem. 1991, 39, 356–360. [Google Scholar] [CrossRef]
- Park, D.L. Effect of Processing on Aflatoxin. Adv. Exp. Med. Biol. 2002, 504, 173–179. [Google Scholar] [CrossRef]
- van der Westhuizen, L.; Shephard, G.S.; Rheeder, J.P.; Burger, H.M.; Gelderblom, W.C.A.; Wild, C.P.; Gong, Y.Y. Optimising sorting and washing of home-grown maize to reduce fumonisin contamination under laboratory-controlled conditions. Food Control 2011, 22, 396–400. [Google Scholar] [CrossRef]
- Matumba, L.; Van Poucke, C.; Ediage, E.N.; Jacobs, B.; De Saeger, S. Effectiveness of hand sorting, flotation/washing, dehulling and combinations thereof on the decontamination of mycotoxin-contaminated white maize. Food Addit. Contam. Part A 2015, 32, 960–969. [Google Scholar] [CrossRef]
- Ozlüoymak, O.B. Development of a UV-based imaging system for real-time detection and separation of dried figs contaminated with aflatoxins. J. Agric. Sci. 2014, 20, 302–316. Available online: https://dergipark.org.tr/download/article-file/21073 (accessed on 24 March 2020). [CrossRef]
- Whitaker, T.B.; Slate, A.; Birmingham, T.; Adams, J.; Jacobs, M.; Gray, G. Correlation between aflatoxin contamination and various USDA grade categories of shelled almonds. J. AOAC Int. 2010, 93, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.J.; Dorner, J.W.; Holbrook, C.C. Advances in mycotoxin elimination and resistance. In Advantage in Peanut Science; Pattee, H.E., Stalker, H.T., Eds.; American Peanut Research Educational Society: Stillwater, OK, USA, 1995; pp. 456–474. [Google Scholar]
- Whitaker, T.B.; Dorner, J.W.; Lamb, M.; Slate, A.B. The Effect of Sorting Farmers’ Stock Peanuts by Size and Color on Partitioning Aflatoxin into Various Shelled Peanut Grade Sizes. Peanut Sci. 2005, 32, 103–118. [Google Scholar] [CrossRef]
- Pearson, T.C.; Wicklow, D.T.; Pasikatan, M.C. Reduction of aflatoxin and fumonisin contamination in yellow corn by high-speed dual-wavelength sorting. Cereal Chem. 2004, 81, 490–498. [Google Scholar] [CrossRef]
- Pearson, T.C.; Wicklow, D.T.; Brabec, D.L. Characteristics and sorting of white food corn contaminated with mycotoxins. Appl. Eng. Agric. 2010, 26, 109–113. [Google Scholar] [CrossRef]
- Stasiewicz, M.J.; Falade, T.D.O.; Mutuma, M.; Mutiga, S.K.; Harvey, J.J.W.; Fox, G.; Pearson, T.C.; Muthomi, J.W.; Nelson, R.J. Multi-spectral kernel sorting to reduce aflatoxins and fumonisins in Kenyan maize. Food Control 2017, 78, 203–214. [Google Scholar] [CrossRef]
- Shi, H.; Stroshine, R.; Ileleji, K. Aflatoxin reduction in corn by cleaning and sorting. In Annual International Meeting 2014; American Society of Agricultural and Biological Engineers (ASABE): St. Joseph, MI, USA, 2014; Volume 1, pp. 311–321. [Google Scholar]
- Pietri, A.; Zanetti, M.; Bertuzzi, T. Distribution of aflatoxins and fumonisins in dry-milled maize fractions. Food Addit. Contam. Part A 2009, 26, 372–380. [Google Scholar] [CrossRef]
- Vanara, F.; Scarpino, V.; Blandino, M. Fumonisin Distribution in Maize Dry-Milling Products and By-Products: Impact of Two Industrial Degermination Systems. Toxins 2018, 10, 357. [Google Scholar] [CrossRef]
- Brera, C.; Catano, C.; De Santis, B.; Debegnach, F.; De Giacomo, M.; Pannunzi, E.; Miraglia, M. Effect of industrial processing on the distribution of aflatoxins and zearalenone in corn-milling fractions. J. Agric. Food Chem. 2006, 54, 5014–5019. [Google Scholar] [CrossRef]
- Cheli, F.; Pinotti, L.; Rossi, L.; Dell’Orto, V. Effect of milling procedures on mycotoxin distribution in wheat fractions: A review. LWT Food Sci. Technol. 2013, 54, 307–314. [Google Scholar] [CrossRef]
- Bullerman, L.B.; Bianchini, A. Good food-processing techniques: Stability of mycotoxins in processed maize-based foods. In Mycotoxin Reduction in Grain Chains; Leslie, J.F., Logrieco, A.F., Eds.; John Wiley and Sons, Inc.: Ames, IA, USA, 2004; pp. 89–100. [Google Scholar]
- Oluwafemi, F. Fate of aflatoxin levels in cereals and cereal products during processing. J. Food Technol. 2004, 2, 299–303. Available online: http://medwelljournals.com/abstract/?doi=jftech.2004.299.303 (accessed on 15 May 2020).
- Directive 2002/32/EC of the European Parliament and of the Council of 7 May 2002 on undesirable substances in animal feed. Off. J. Eur. Communities 2002, 140, 10–21.
- Innovative Sorting Solutions for Food and Plastics. Available online: https://www.buhlergroup.com/content/buhlergroup/global/en/product-families/optical-sorters.html (accessed on 7 May 2020).
- Electronic Sorting. Available online: https://www.cimbria.com/products/electronic-sorting (accessed on 7 May 2020).
- Multifunction color sorter. Available online: https://www.grotechcolorsorter.com/ (accessed on 7 May 2020).
- Color Sorting Solutions. Available online: https://www.sortexgroup.com/ (accessed on 7 May 2020).
- Optical Sorting Solutions for Agricultural and Industrial Markets. Available online: http://www.satake-usa.com/optical-sorting-solutions.html (accessed on 7 May 2020).
- Commission Regulation (EU) No 401/2006 of 23 February 2006 Laying down the Method of Sampling and Analysis for the Official Control of the Levels of Mycotoxins in Foodstuffs. Off. J. Eur. Union 2006, 70, 12–34.
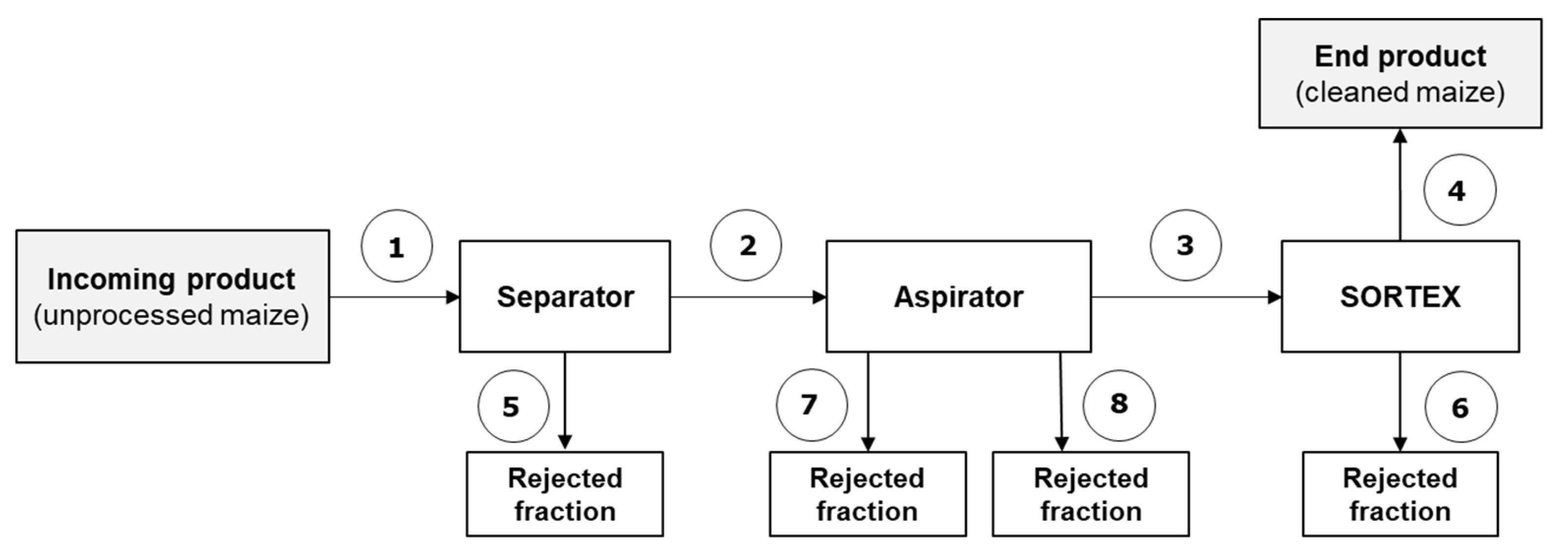
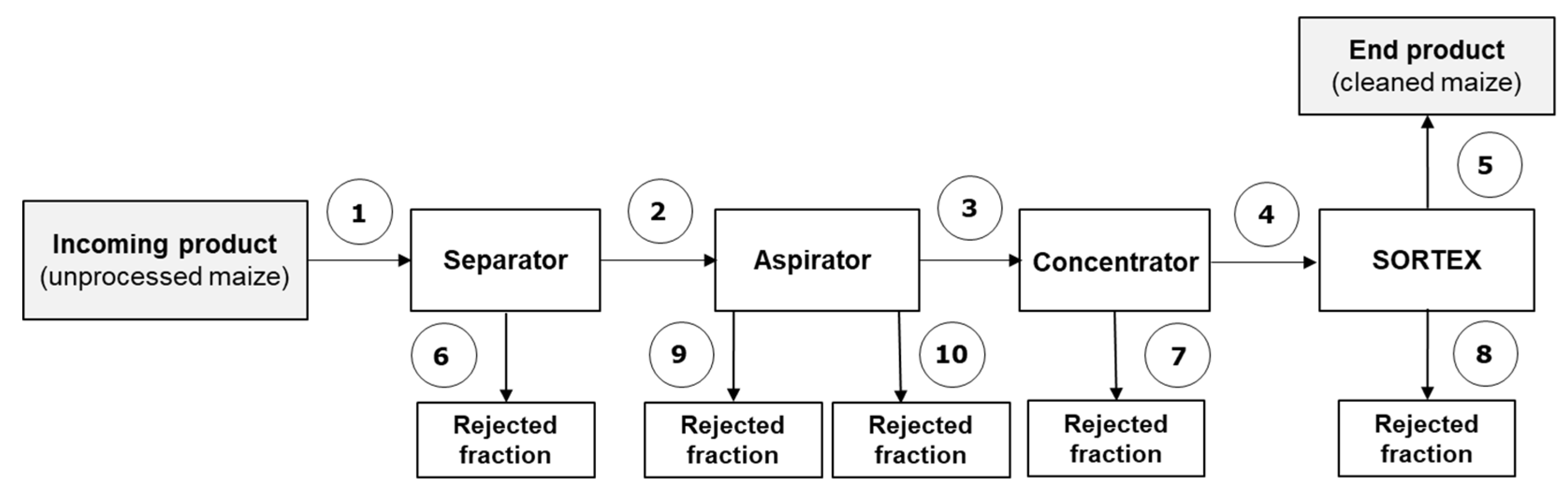
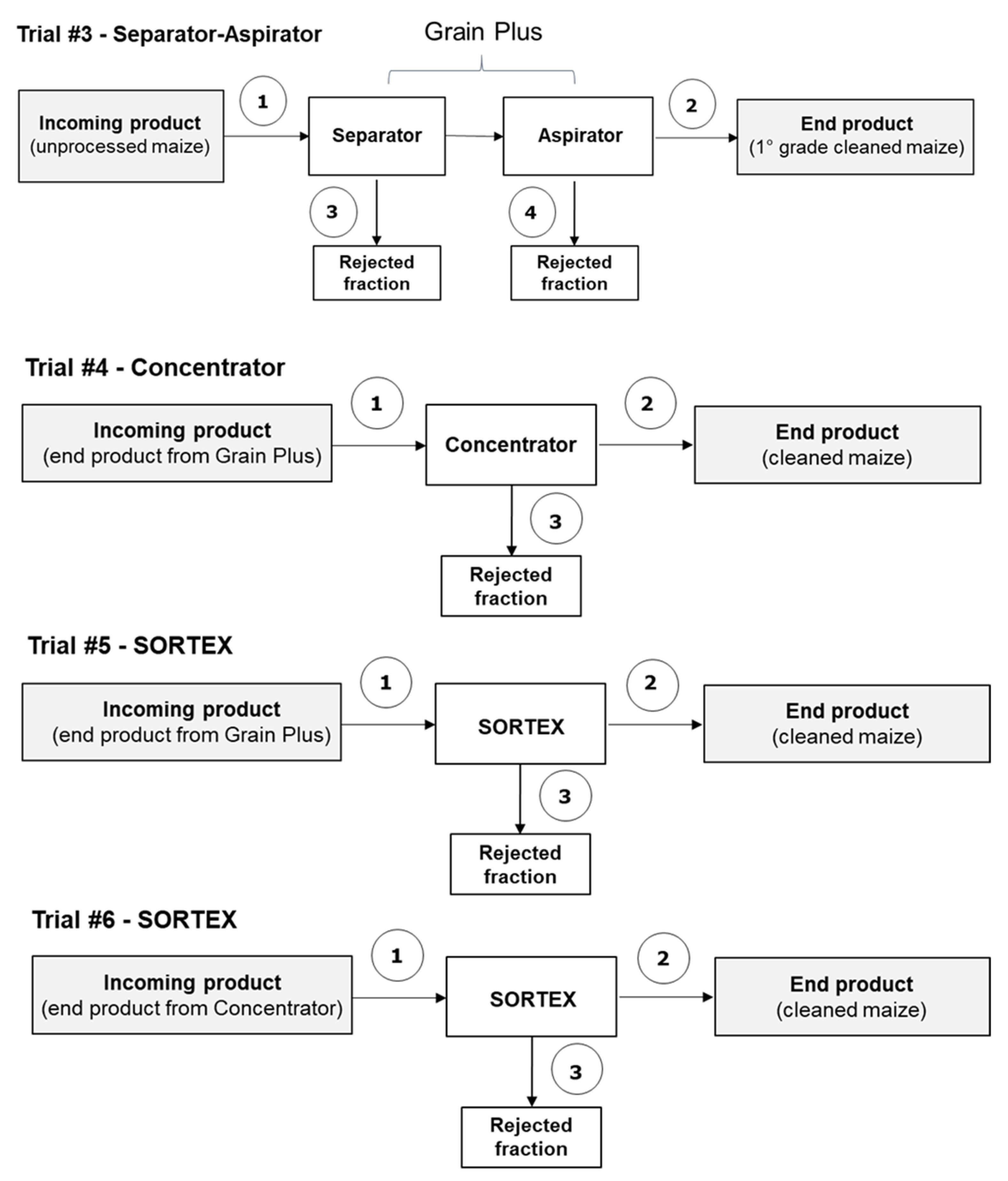
| Maize-Cleaning Fraction | Trial #1 | Trial #2 |
|---|---|---|
| Unprocessed maize | 100 | 100 |
| Rejected fraction from separator Rejected fraction from aspirator | 0.83 0.76 | 0.83 0.32 |
| Rejected fraction from concentrator | 3.49 | |
| Rejected fraction from optical sorter | 4.59 | 2.00 |
| Cleaned maize | 93.82 | 93.36 |
| Batch | Sampled Fraction 1 | AFB1 (µg/kg) | AFB1 Reduction (%) | AFs 2 (µg/kg) | AFs Reduction (%) |
|---|---|---|---|---|---|
| A | 1 | 24.2 | 65.3 | 25.4 | 65.3 |
| 2 | 17.3 | 17.9 | |||
| 3 | 9.5 | 9.8 | |||
| 4 | 8.4 | 8.8 | |||
| 5 | 109.1 | 114.1 | |||
| 6 | 76.0 | 79.5 | |||
| 7 | 165.0 | 170.6 | |||
| 8 | 319.6 | 333.0 | |||
| B | 1 | 62.0 | 78.1 | 64.3 | 77.7 |
| 2 | 28.6 | 29.8 | |||
| 3 | 15.8 | 16.7 | |||
| 4 | 13.5 | 14.3 | |||
| 5 | 110.7 | 115.5 | |||
| 6 | 468.4 | 489.4 | |||
| 7 | 195.6 | 203.2 | |||
| 8 | 309.8 | 322.5 |
| Batch | Sampled Fraction 1 | AFB1 (µg/kg) | AFB1 Reduction (%) | AFs 2 (µg/kg) | AFs Reduction (%) |
|---|---|---|---|---|---|
| C | 1 | 28.8 | 84.4 | 30.2 | 84.4 |
| 2 | 11.4 | 11.8 | |||
| 3 | 12.7 | 13.2 | |||
| 4 | 5.4 | 5.6 | |||
| 5 | 4.5 | 4.7 | |||
| 6 | 120.8 | 125.8 | |||
| 7 | 264.1 | 274.6 | |||
| 8 | 327.2 | 340.3 | |||
| 9 | 182.0 | 189.1 | |||
| 10 | 328.8 | 341.2 | |||
| D | 1 | 23.5 | 75.8 | 24.5 | 75.6 |
| 2 | 10.7 | 11.1 | |||
| 3 | 6.3 | 6.4 | |||
| 4 | 5.4 | 5.5 | |||
| 5 | 5.7 | 6.0 | |||
| 6 | 127.1 | 132.1 | |||
| 7 | 159.9 | 165.7 | |||
| 8 | 244.1 | 265.4 | |||
| 9 | 164.9 | 170.9 | |||
| 10 | 306.9 | 318.7 |
| Trial | Maize-Cleaning Fraction | Batch | |||
|---|---|---|---|---|---|
| A1 | A2 | B1 | B2 | ||
| # 3 | Unprocessed maize | 100 | 100 | 100 | 100 |
| Rejected fraction from separator | 1.7 | 4.3 | 6.8 | 8.8 | |
| Rejected fraction from aspirator | 0.2 | 1.1 | 0.7 | 2.2 | |
| Cleaned maize | 98.1 | 94.6 | 92.5 | 89.0 | |
| # 4 | Cleaned maize from separator + aspirator | 100 | 100 | 100 | 100 |
| Rejected fraction from concentrator | 3.0 | 17.6 | 2.4 | 12.2 | |
| Cleaned maize | 97.0 | 82.4 | 97.6 | 87.8 | |
| # 5 | Cleaned maize from separator + aspirator | 100 | 100 | 100 | 100 |
| Rejected fraction from optical sorter | 5.1 | 3.8 | 7.9 | 5.6 | |
| Cleaned maize | 94.9 | 96.2 | 92.1 | 94.4 | |
| # 6 | Cleaned maize from concentrator | 100 | 100 | 100 | 100 |
| Rejected fraction from optical sorter | 4.3 | 4.1 | 6.9 | 5.4 | |
| Cleaned maize | 95.7 | 95.9 | 93.1 | 94.6 | |
| Batch | Sampled Fraction 1 | AFB1 (µg/kg) | AFB1 Reduction (%) | AFs 2 (µg/kg) | AFs Reduction (%) |
|---|---|---|---|---|---|
| A1 | 1 | n.d. 3 | - | n.d. | - |
| 2 | 0.8 | 1.5 | |||
| 3 | 39.4 | 44.2 | |||
| 4 | 142.4 | 222.0 | |||
| A2 | 1 | 15.3 | 58.9 | 19.1 | 64.4 |
| 2 | 6.3 | 6.8 | |||
| 3 | 21.2 | 32.1 | |||
| 4 | 80.1 | 110.7 | |||
| B1 | 1 | 15.9 | - | 17.6 | - |
| 2 | 19.0 | 29.7 | |||
| 3 | 33.8 | 41.0 | |||
| 4 | 124.8 | 139.5 | |||
| B2 | 1 | 28.3 | 30.7 | 30.4 | 30.6 |
| 2 | 19.6 | 21.1 | |||
| 3 | 22.1 | 31.1 | |||
| 4 | 94.9 | 105.9 |
| Batch | Sampled Fraction 1 | AFB1 (µg/kg) | AFB1 Reduction (%) | AFs 2 (µg/kg) | AFs Reduction (%) |
|---|---|---|---|---|---|
| A1 | 1 2 | 1.7 0.8 | 54.0 | 2.7 1.5 | 43.8 |
| A2 | 1 2 | 7.8 6.3 | 18.7 | 9.0 6.8 | 24.6 |
| B1 | 1 2 | 20.8 19.0 | 8.4 | 31.2 29.7 | 4.9 |
| B2 | 1 2 | 21.5 19.6 | 8.7 | 23.9 21.1 | 11.5 |
| Batch | Sampled Fraction 1 | AFB1 (µg/kg) | AFB1 Reduction (%) | AFs 2 (µg/kg) | AFs Reduction (%) |
|---|---|---|---|---|---|
| A1 | 1 | 2.4 | 45.8 | 2.5 | 48.0 |
| 2 | 1.3 | 1.3 | |||
| 3 | 19.1 | 19.5 | |||
| A2 | 1 | n.d. 3 | - | n.d. | - |
| 2 | n.d. | n.d. | |||
| 3 | n.d. | n.d | |||
| B1 | 1 | 11.5 | 54.8 | 15.4 | 64.9 |
| 2 | 5.2 | 5.4 | |||
| 3 | 73.4 | 77.0 | |||
| B2 | 1 | 2.9 | 48.3 | 3.2 | 53.1 |
| 2 | 1.5 | 1.5 | |||
| 3 | 110.9 | 117.6 |
| Batch | Trial | Sampled Fraction 1 | AFB1 (µg/kg) | AFB1 Reduction (%) | AFs 2 (µg/kg) | AFs Reduction (%) |
|---|---|---|---|---|---|---|
| A1 | 1 | 5.7 | 50.9 | 6.2 | 50.0 | |
| #5 | 2 | 2.8 | 3.1 | |||
| 3 | 58.5 | 62.6 | ||||
| 1 | 1.3 | 7.7 | 1.5 | 13.3 | ||
| #6 | 2 | 1.2 | 1.3 | |||
| 3 | 4.9 | 5.2 | ||||
| A2 | 1 | 0.8 | 25.0 | 0.8 | 25.0 | |
| #5 | 2 | 0.6 | 0.6 | |||
| 3 | 5.1 | 5.3 | ||||
| 1 | 7.0 | 11.4 | 7.9 | 10.1 | ||
| #6 | 2 | 6.2 | 7.1 | |||
| 3 | 24.9 | 26.4 | ||||
| B1 | 1 | 9.0 | 62.2 | 9.7 | 60.8 | |
| #5 | 2 | 3.4 | 3.8 | |||
| 3 | 74.1 | 78.0 | ||||
| 1 | 11.7 | 75.2 | 13.4 | 76.1 | ||
| #6 | 2 | 2.9 | 3.2 | |||
| 3 | 130.7 | 151.0 | ||||
| B2 | 1 | 28.7 | 31.4 | 31.6 | 33.5 | |
| #5 | 2 | 19.7 | 21.0 | |||
| 3 | 180.2 | 209.8 | ||||
| 1 | 7.0 | 42.9 | 7.8 | 46.2 | ||
| #6 | 2 | 4.0 | 4.2 | |||
| 3 | 60.2 | 69.9 |
| Trial | Batch | AFB1 (%) | AFs (%) |
|---|---|---|---|
| #1 Separator–aspirator–optical sorter | A | 58 | 58 |
| B | 60 | 60 | |
| #2 Separator–aspirator–concentrator–optical sorter | C | 76 | 75 |
| D | 75 | 76 | |
| #3 Separator–aspirator | A1 | 102 | 99 |
| A2 | 99 | 100 | |
| B1 | 100 | 100 | |
| B2 | 100 | 100 | |
| #4 Concentrator | A1 | 76 | 74 |
| A2 | -1 | -1 | |
| B1 | 59 | 58 | |
| B2 | 512 | 490 | |
| #5 Optical sorter | A1 | 99 | 99 |
| A2 | 96 | 97 | |
| B1 | 100 | 100 | |
| B2 | 100 | 100 | |
| #6 Optical sorter | A1 | 103 | 98 |
| A2 | 100 | 100 | |
| B1 | 100 | 100 | |
| B2 | 100 | 99 |
| Trial | Sampled Fraction 1 | Fraction Weight (Tons) | Number of Incremental Samples | Aggregate Sample Weight (Kg) |
|---|---|---|---|---|
| #1 | 1, 2, 3, 4 | <10–≤20 | 60 | 6–10 |
| 5 | >0.5–≤1.0 | 10 | 1–2 | |
| 6, 7, 8 | >0.05–≤0.5 | 5 | 1–2 | |
| #2 | 1, 2, 3, 4, 5 | <10–≤20 | 60 | 6–10 |
| 7 | >0.5–≤1.0 | 10 | 1–2 | |
| 6, 8 | >0.05–≤0.5 | 5 | 1–2 | |
| 9, 10 | ≤0.05 | 3 | 1–1.5 | |
| #3–6 | 1, 2 | >1.0–≤3.0 | 20 | 2–4 |
| 3 | >0.05–≤0.5 | 5 | 1–2 | |
| 4 | ≤0.05 | 3 | 1–1.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pascale, M.; Logrieco, A.F.; Graeber, M.; Hirschberger, M.; Reichel, M.; Lippolis, V.; De Girolamo, A.; Lattanzio, V.M.T.; Slettengren, K. Aflatoxin Reduction in Maize by Industrial-Scale Cleaning Solutions. Toxins 2020, 12, 331. https://doi.org/10.3390/toxins12050331
Pascale M, Logrieco AF, Graeber M, Hirschberger M, Reichel M, Lippolis V, De Girolamo A, Lattanzio VMT, Slettengren K. Aflatoxin Reduction in Maize by Industrial-Scale Cleaning Solutions. Toxins. 2020; 12(5):331. https://doi.org/10.3390/toxins12050331
Chicago/Turabian StylePascale, Michelangelo, Antonio F. Logrieco, Matthias Graeber, Marina Hirschberger, Mareike Reichel, Vincenzo Lippolis, Annalisa De Girolamo, Veronica M.T. Lattanzio, and Katarina Slettengren. 2020. "Aflatoxin Reduction in Maize by Industrial-Scale Cleaning Solutions" Toxins 12, no. 5: 331. https://doi.org/10.3390/toxins12050331
APA StylePascale, M., Logrieco, A. F., Graeber, M., Hirschberger, M., Reichel, M., Lippolis, V., De Girolamo, A., Lattanzio, V. M. T., & Slettengren, K. (2020). Aflatoxin Reduction in Maize by Industrial-Scale Cleaning Solutions. Toxins, 12(5), 331. https://doi.org/10.3390/toxins12050331










