Structures of Reaction Products and Degradation Pathways of Aflatoxin B1 by Ultrasound Treatment
Abstract
:1. Introduction
2. Results and Discussion
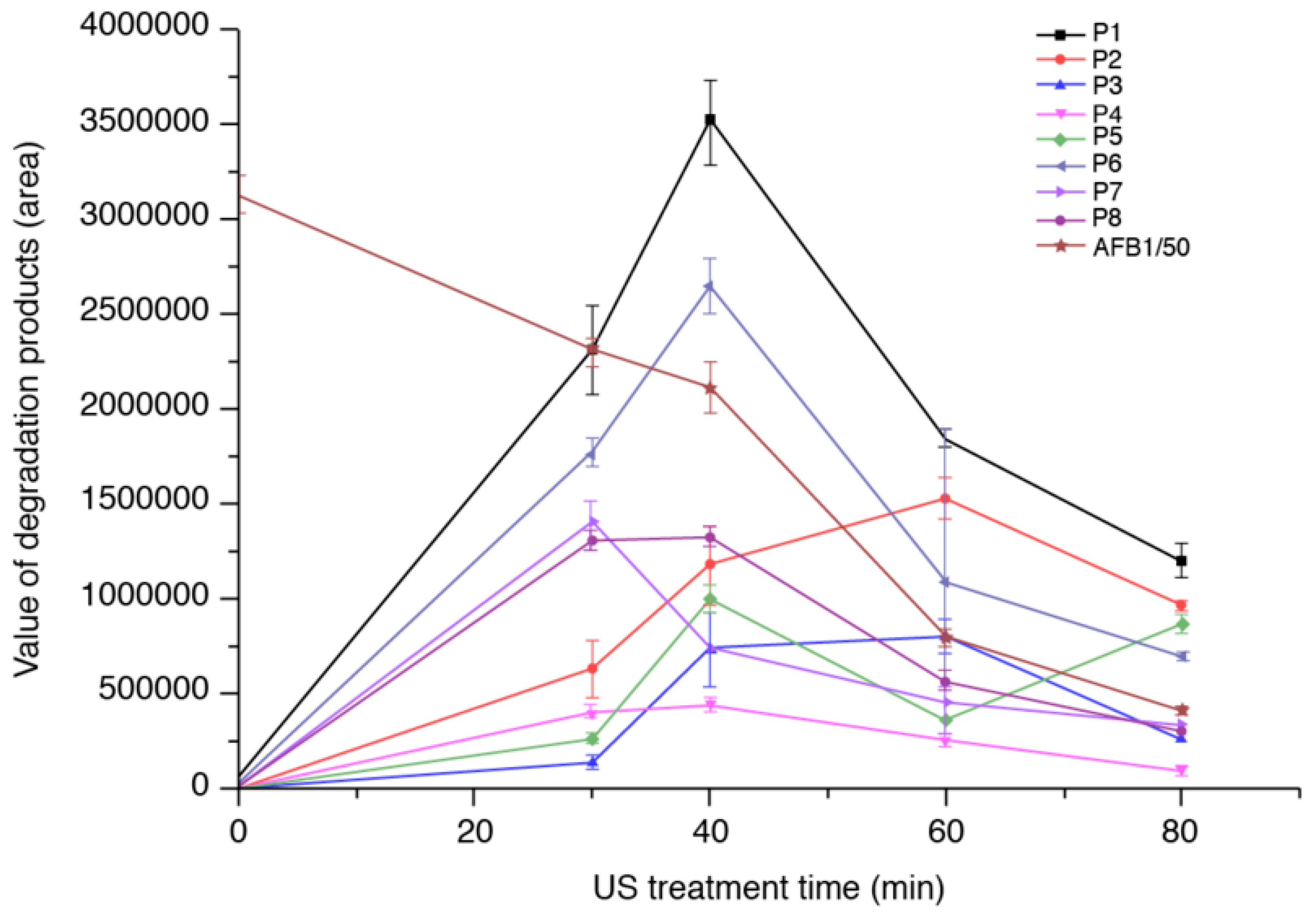
2.1. Formation of AFB1 Reaction Products as A Function of Ultrasound Treatment Time
2.2. Molecular Formulae of the AFB1 Reaction Products
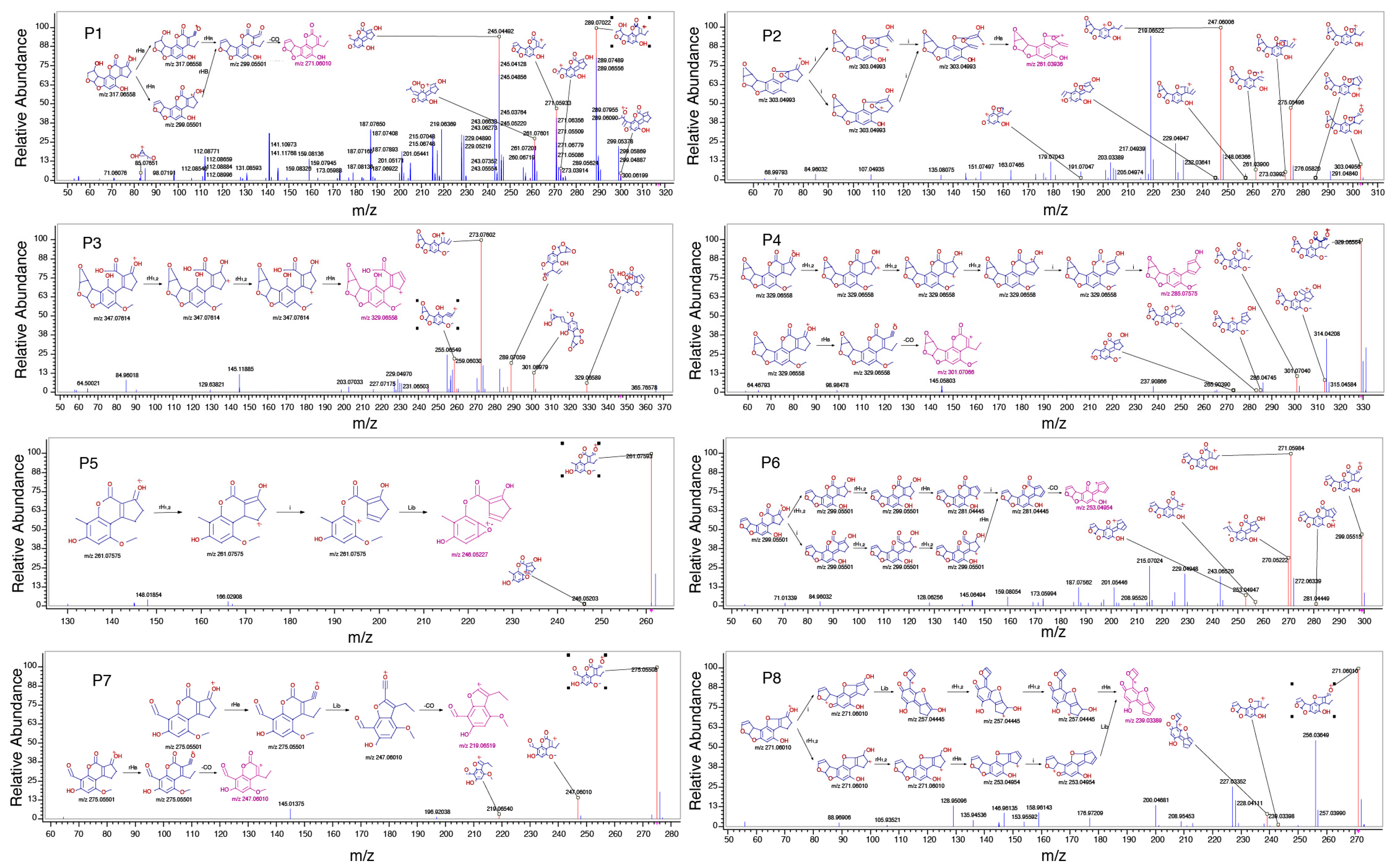
2.3. Proposed Structures of the AFB1 Reaction Products
2.4. Degradation Mechanism and Reaction Pathway of AFB1 upon US Treatment
2.5. Toxicity of the Reaction Products
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. AFB1 Treatment with Power Ultrasound
3.3. UHPLC–MS Analysis
3.4. UHPLC-MS/MS Analysis
3.5. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Battilani, P. Food mycology-a multifaceted approach to fungi and food. World Mycotoxin J. 2008, 1, 223–224. [Google Scholar] [CrossRef]
- Magan, N.; Olsen, M. Rapid detection of mycotoxigenic fungi in plants. In Mocotoxins in Food; Magan, N., Olsen, M., Eds.; Woodhead Publishing: Cambridge, UK, 2004; pp. 111–136. [Google Scholar]
- Pitt, J.I. Food mycology: A discipline comes of age. Food Aust. 1996, 48, 258–261. [Google Scholar]
- Kujawa, M. Some naturally occurring substances: Food items and constituents, heterocyclic aromatic amines and mycotoxins. Mol. Nutr. Food Res. 1994, 38, 351. [Google Scholar] [CrossRef]
- Williams, J.H.; Phillips, T.D.; Jolly, P.E.; Stiles, J.K.; Jolly, C.M.; Aggarwal, D. Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences, and interventions. Am. J. Clin. Nutr. 2004, 80, 1106–1122. [Google Scholar] [CrossRef] [PubMed]
- Fandohan, P.; Gnonlonfin, B.; Hell, K.; Marasas, W.F.; Wingfield, M.J. Natural occurrence of Fusarium and subsequent fumonisin contamination in preharvest and stored maize in Benin, West Africa. Int. J. Food Microbiol. 2005, 99, 173–183. [Google Scholar] [CrossRef]
- Karlovsky, P. Biological detoxification of fungal toxins and its use in plant breeding, feed and food production. Nat. Toxins 1999, 7, 1–23. [Google Scholar] [CrossRef]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef]
- Liu, R.; Wang, R.; Jian, L.; Chang, M.; Jin, Q.; Du, Z.; Wang, S.; Li, Q.; Wang, X. Degradation of AFB 1 in aqueous medium by electron beam irradiation: Kinetics, pathway and toxicology. Food Control 2016, 66, 151–157. [Google Scholar] [CrossRef]
- Méndez-Albores, A.; Moreno-Martínez, E.; Martínez-Bustos, F.; Gaytán-Martínez, M. Effect of lactic and citric acid on the stability of B-aflatoxins in extrusion-cooked sorghum. Lett. Appl. Microbiol. 2008, 47, 1–7. [Google Scholar] [CrossRef]
- Inan, F.; Pala, M.; Doymaz, I. Use of ozone in detoxification of aflatoxin B in red pepper. J. Stored Prod. Res. 2007, 43, 425–429. [Google Scholar] [CrossRef]
- Sakudo, A.; Toyokawa, Y.; Misawa, T.; Imanishi, Y. Degradation and detoxification of aflatoxin B1 using nitrogen gas plasma generated by a static induction thyristor as a pulsed power supply. Food Control 2017, 73, 619–626. [Google Scholar] [CrossRef]
- Escobedo-González, R.; Méndez-Albores, A.; Villarreal-Barajas, T.; Aceves-Hernández, J.M.; Miranda-Ruvalcaba, R.; Nicolás-Vásquez, I. A theoretical study of 8-chloro hydroxy-aflatoxin B1, the conversion product of aflatoxin B1 by neutral electrolyzed water. Toxins 2016, 8, 225. [Google Scholar] [CrossRef] [PubMed]
- Somayajula, A.; Asaithambi, P.; Susree, M.; Matheswaran, M. Sonoelectrochemical oxidation for decolorization of Reactive Red 195. Ultrason. Sonochem. 2012, 19, 803–811. [Google Scholar] [CrossRef] [PubMed]
- De Lima Leite, R.H.; Cognet, P.; Wilhelm, A.-M.; Delmas, H. Anodic oxidation of 2,4-dihydroxybenzoic acid for wastewater treatment: Study of ultrasound activation. Chem. Eng. Sci. 2002, 57, 767–778. [Google Scholar] [CrossRef]
- Suslick, K.S.; Nyborg, W.L. Ultrasound: Its chemical, physical and biological effects. J. Acoust. Soc. Am. 1990, 87, 919–920. [Google Scholar] [CrossRef]
- Yao, J.J.; Gao, N.Y.; Deng, Y.; Ma, Y.; Li, H.-J.; Xu, B.; Li, L. Sonolytic degradation of parathion and the formation of byproducts. Ultrason. Sonochem. 2010, 17, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; He, Y.; Jeon, J.; O’Shea, K.E. Irradiation of ultrasound to 5 methylbenzotriazole in aqueous phase: Degradation kinetics and mechanisms. Ultrason. Sonochem. 2016, 31, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Musmarra, D.; Prisciandaro, M.; Capocelli, M.; Karatza, D.; Lovino, P.; Canzano, S.; Lancia, A. Degradation of ibuprofen by hydrodynamic cavitation: Reaction pathways and effect of operational parameters. Ultrason. Sonochem. 2016, 29, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, C.; Frontistis, Z.; Antonopoulou, M.; Venieri, D.; Konstantinou, I.; Mantzavinos, D. Sonochemical degradation of ethyl paraben in environmental samples: Statistically important parameters determining kinetics, by-products and pathways. Ultrason. Sonochem. 2016, 31, 62–70. [Google Scholar] [CrossRef]
- Liu, Y.; Li, M.; Liu, Y.; Bai, F.; Bian, K. Effects of pulsed ultrasound at 20 kHz on the sonochemical degradation of mycotoxins. World Mycotoxin J. 2019. [Google Scholar] [CrossRef]
- Mason, T.J.; Chemat, F.; Vinatoru, M. The extraction of natural products using ultrasound or microwaves. Curr. Org. Chem. 2011, 15, 237–247. [Google Scholar] [CrossRef]
- Zheng, H.; Wei, S.; Xu, Y.; Fan, M. Reduction of aflatoxin B1 in peanut meal by extrusion cooking. LWT Food Sci. Technol. 2015, 64, 515–519. [Google Scholar] [CrossRef]
- Ince, N.H. Ultrasound-assisted advanced oxidation processes for water decontamination. Ultrason. Sonochem. 2018, 40, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.H.; Doraiswamy, L.K. Sonochemistry: Science and engineering. Ind. Eng. Chem. Res. 1999, 38, 1215–1249. [Google Scholar] [CrossRef]
- Luo, X.; Wang, R.; Wang, L.; Wang, Y.; Chen, Z. Structure elucidation and toxicity analyses of the degradation products of aflatoxin B-1 by aqueous ozone. Food Control 2013, 31, 331–336. [Google Scholar] [CrossRef]
- Zorlugenç, B.; Zorlugenç, F.K.; Öztekin, S.; Evliya, I.B. The influence of gaseous ozone and ozonated water on microbial flora and degradation of aflatoxin B1 in dried figs. Food Chem. Toxicol. 2008, 46, 3593–3597. [Google Scholar] [CrossRef] [PubMed]
- Enjie, D.; Changpo, S.; Hanxue, H.; Shanshan, W.; Minghua, L.; Haizhou, D. Structures of the ozonolysis products and ozonolysis pathway of aflatoxin B1 in acetonitrile solution. J. Agric. Food Chem. 2012, 60, 9364–9370. [Google Scholar] [CrossRef]
- Shi, H.; Cooper, B.R.; Stroshine, R.L.; Ileleji, K.E.; Keener, K. Structures of degradation products and degradation pathways of aflatoxin b1 by high voltage atmospheric cold plasma (HVACP) treatment. J. Agric. Food Chem. 2017, 65, 6222–6230. [Google Scholar] [CrossRef]
- Koelewijn, P. Epoxidation of olefins by alkylperoxy radicals. Recl. Trav. Chim. Pays Bas 2010, 91, 759–779. [Google Scholar] [CrossRef]
- Hart, E.J.; Henglein, A. Sonolysis of ozone in aqueous solution. J. Phys. Chem. 1986, 90, 3061–3062. [Google Scholar] [CrossRef]
- Authier, O.; Ouhabaz, H.; Bedogni, S. Modeling of sonochemistry in water in the presence of dissolved carbon dioxide. Ultrason. Sonochem. 2018, 45, 17–18. [Google Scholar] [CrossRef] [PubMed]
- Reuter, F.; Lesnik, S.; Ayaz-Bustami, K.; Brenner, G.; Mettin, R. Bubble size measurements in different acoustic cavitation structures: Filaments, clusters, and the acoustically cavitated jet. Ultrason. Sonochem. 2019, 55, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Baertschi, S.W.; Raney, K.D.; Stone, M.P.; Harris, T.M. ChemInform abstract: Preparation of the 8,9-epoxide of the mycotoxin aflatoxin B1: The ultimate carcinogenic species. ChemInform 1989, 20. [Google Scholar] [CrossRef]
- Johnson, W.W.; Guengerich, F.P. Reaction of aflatoxin B1 exo-8,9-epoxide with DNA: Kinetic analysis of covalent binding and DNA-induced hydrolysis. Proc. Natl. Acad. Sci. USA 1997, 94, 6121–6125. [Google Scholar] [CrossRef] [PubMed]
- Kensler, T.W.; Roebuck, B.D.; Wogan, G.N.; Groopman, J.D. Aflatoxin: A 50 year odyssey of mechanistic and translational toxicology. Toxicol. Sci. 2011, 120, S28–S48. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.S.; Dunn, J.J.; Delucca, A.J.; Ciegler, A. Role of lactone ring of aflatoxin B1 in toxicity and mutagenicity. Experientia 1981, 37, 16–17. [Google Scholar] [CrossRef] [PubMed]
- Wogan, G.N.; Edwards, G.S.; Newberne, P.M. Structure-activity relationships in toxicity and carcinogenicity of aflatoxins and analogs. Cancer Res. 1971, 31, 1936–1942. [Google Scholar] [PubMed]
- Feng, W.; Fang, X.; Xiaofeng, X.; Wang, Z.; Fan, B.; Ha, Y. Structure elucidation and toxicity analyses of the radiolytic products of aflatoxin B1 in methanol-water solution. J. Hazard Mater. 2011, 192, 1192–1202. [Google Scholar] [CrossRef]
- Mao, J.; He, B.; Zhang, L.; Li, P.; Zhang, Q.; Ding, X.; Zhang, W. A structure identification and toxicity assessment of the degradation products of aflatoxin B₁ in peanut oil under UV irradiation. Toxins 2016, 8, 332. [Google Scholar] [CrossRef]
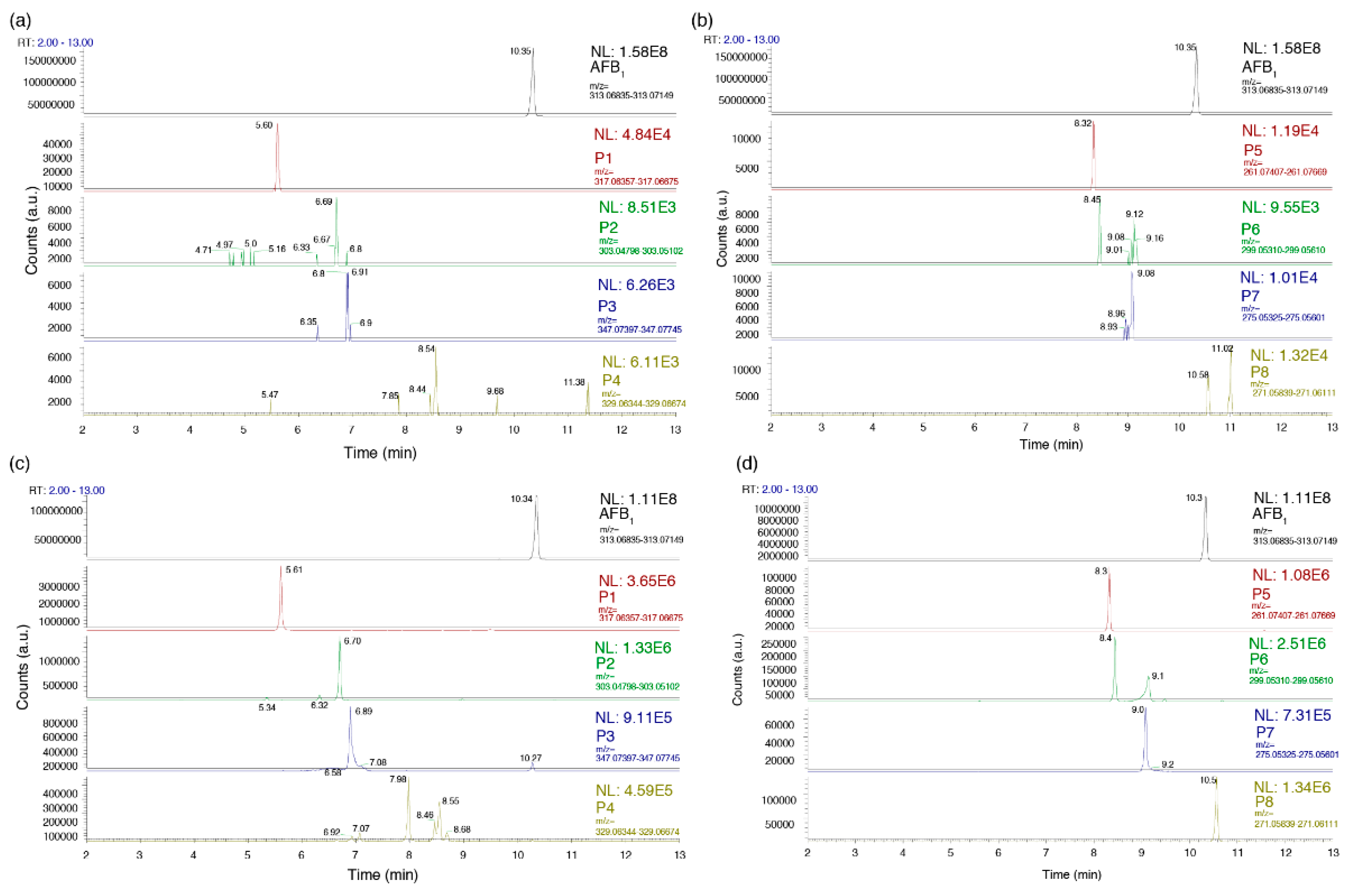
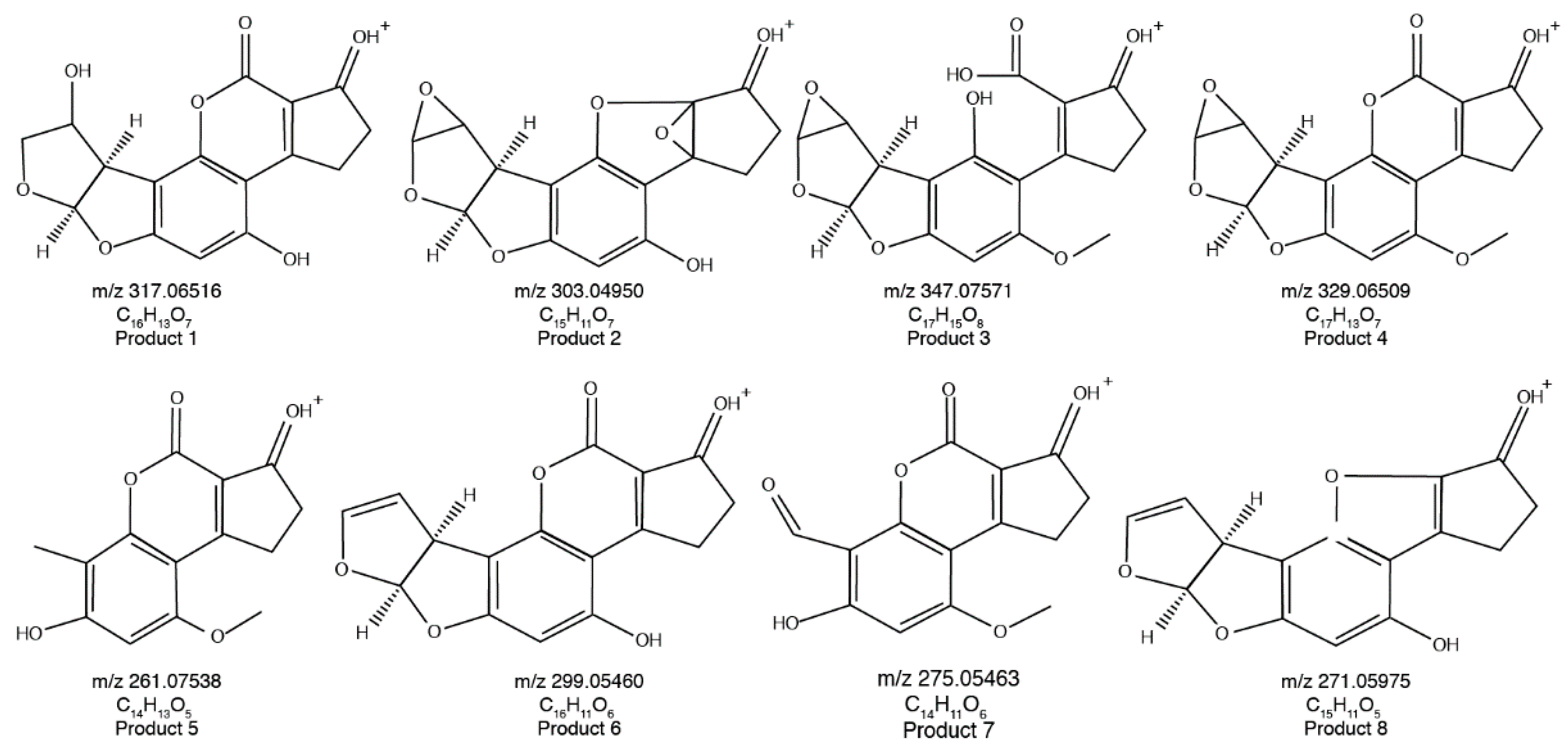
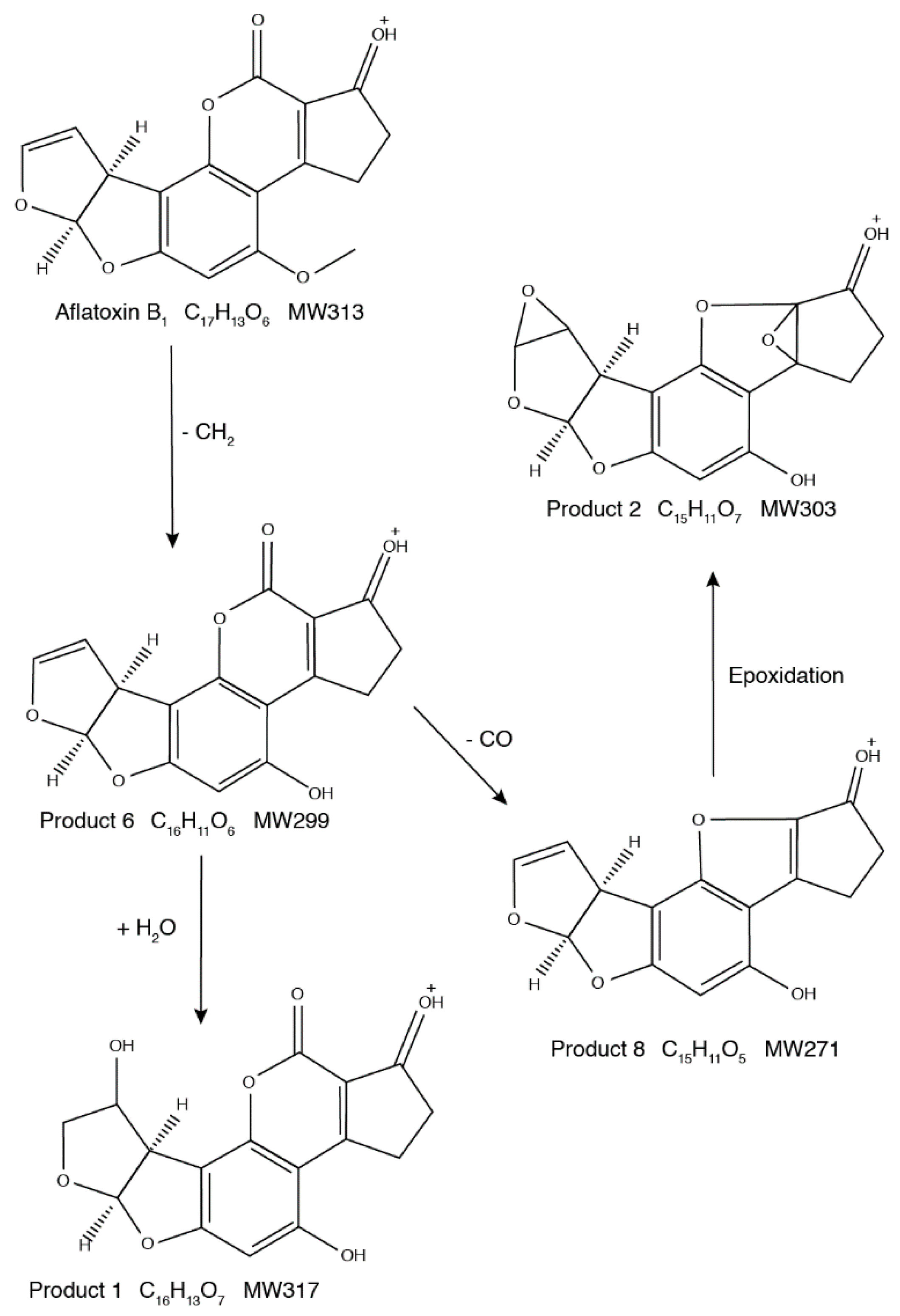
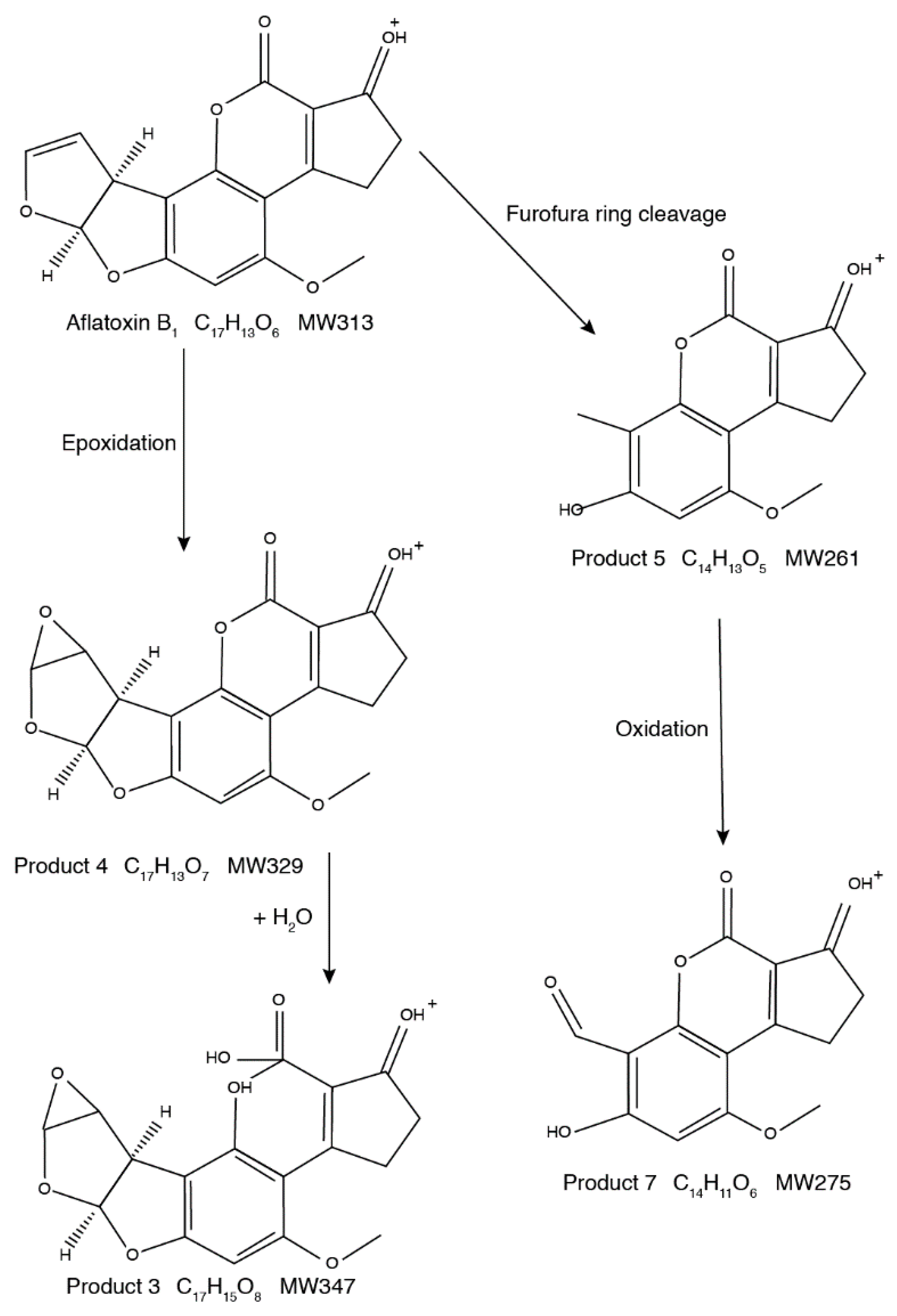
| Proposed Product | Retention Time (min) | Hypothetical Formula | Determined Mass (m/z) 1 | Error (mmu) | IHD 2 | Score (%) |
|---|---|---|---|---|---|---|
| 1 | 5.69 | C16H13O7 | 317.06516 | −0.419 | 10.5 | 89.1 |
| 2 | 6.82 | C15H11O7 | 303.04950 | −0.429 | 10.5 | 87.6 |
| 3 | 6.99 | C17H15O8 | 347.07571 | −0.434 | 10.5 | 88.9 |
| 4 | 7.17 | C17H13O7 | 329.06509 | −0.487 | 11.5 | 92.5 |
| 5 | 8.42 | C14H13O5 | 261.07538 | −0.370 | 8.5 | 88.3 |
| 6 | 8.54 | C16H11O6 | 299.05460 | −0.415 | 11.5 | 90.9 |
| 7 | 9.21 | C14H11O6 | 275.05463 | −0.385 | 9.5 | 82.5 |
| 8 | 10.68 | C15H11O5 | 271.05975 | −0.350 | 10.5 | 85.2 |
| AFB1 | 10.45 | C17H12O7 | 313.07025 | −0.415 | 11.5 | 99.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Li, M.; Liu, Y.; Bian, K. Structures of Reaction Products and Degradation Pathways of Aflatoxin B1 by Ultrasound Treatment. Toxins 2019, 11, 526. https://doi.org/10.3390/toxins11090526
Liu Y, Li M, Liu Y, Bian K. Structures of Reaction Products and Degradation Pathways of Aflatoxin B1 by Ultrasound Treatment. Toxins. 2019; 11(9):526. https://doi.org/10.3390/toxins11090526
Chicago/Turabian StyleLiu, Yuanfang, Mengmeng Li, Yuanxiao Liu, and Ke Bian. 2019. "Structures of Reaction Products and Degradation Pathways of Aflatoxin B1 by Ultrasound Treatment" Toxins 11, no. 9: 526. https://doi.org/10.3390/toxins11090526
APA StyleLiu, Y., Li, M., Liu, Y., & Bian, K. (2019). Structures of Reaction Products and Degradation Pathways of Aflatoxin B1 by Ultrasound Treatment. Toxins, 11(9), 526. https://doi.org/10.3390/toxins11090526




