First Report of Microcystis Strains Producing MC-FR and -WR Toxins in Japan
Abstract
1. Introduction
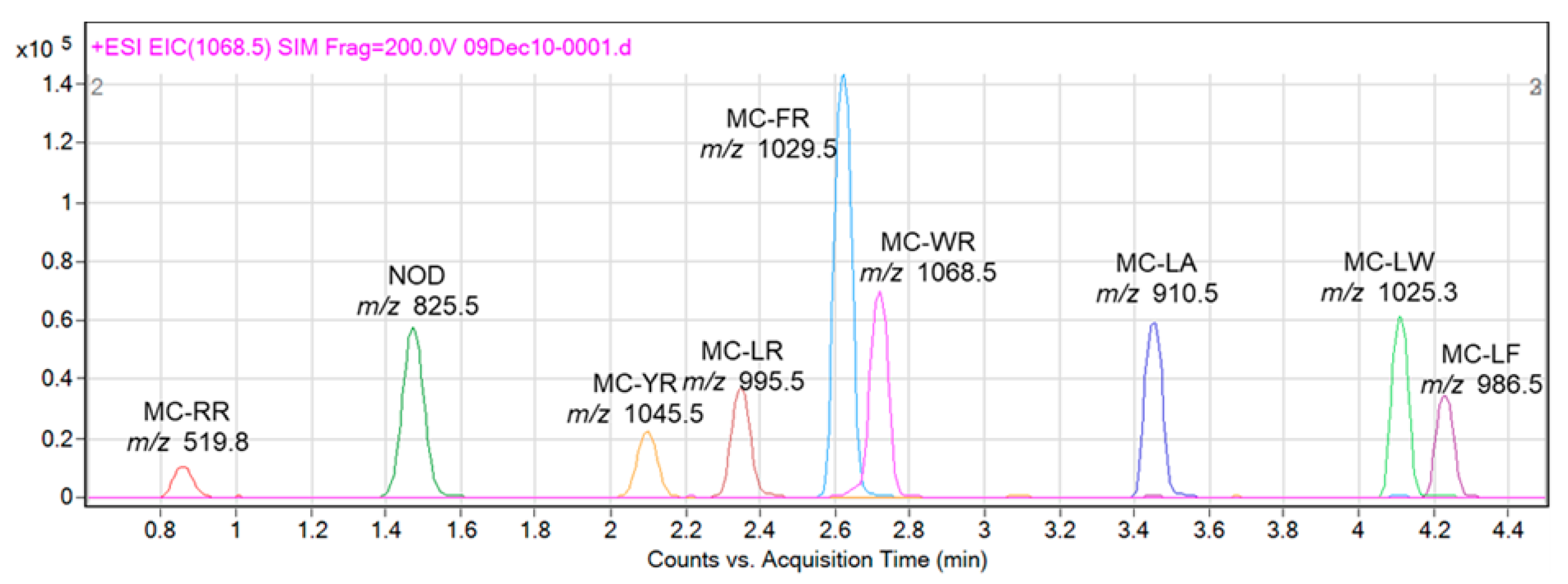
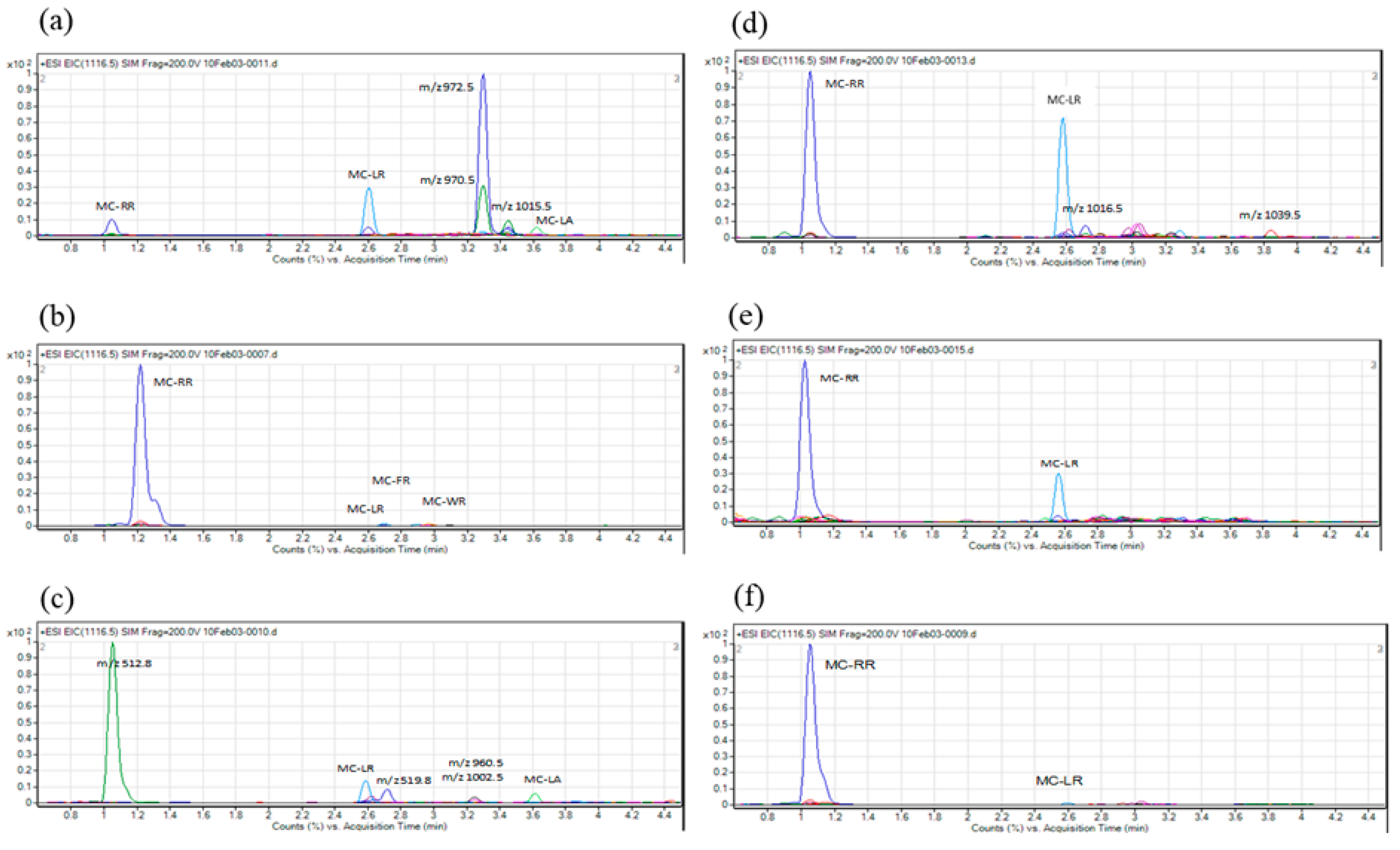
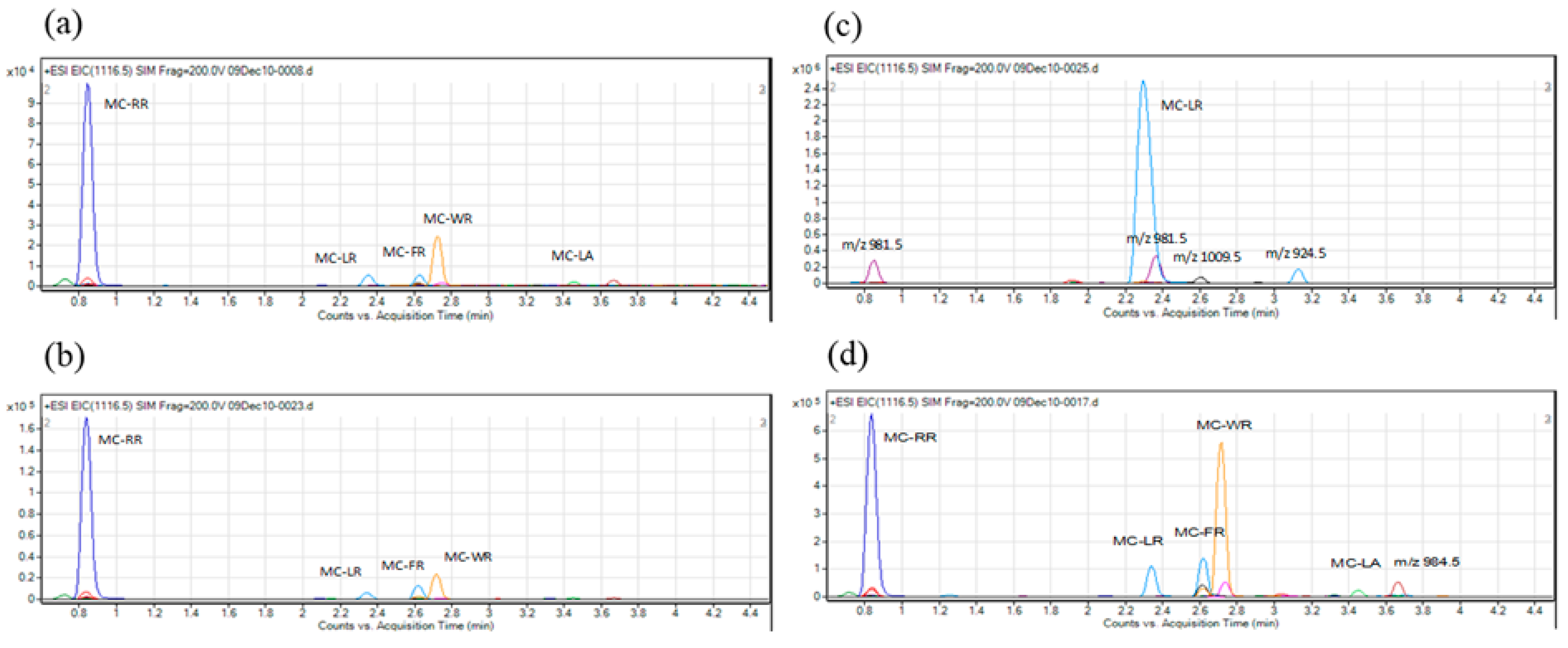
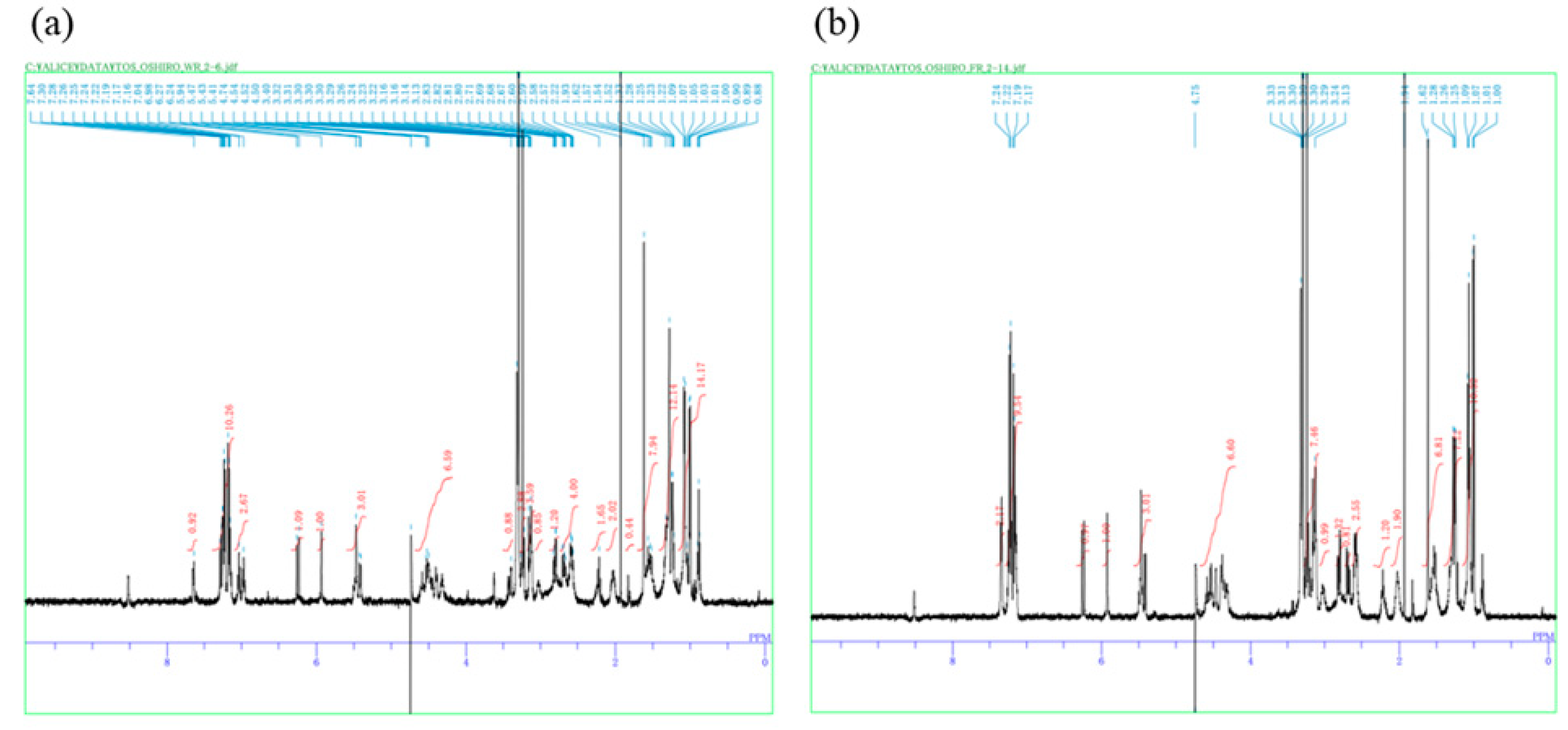
2. Results
3. Discussion
4. Materials and Methods
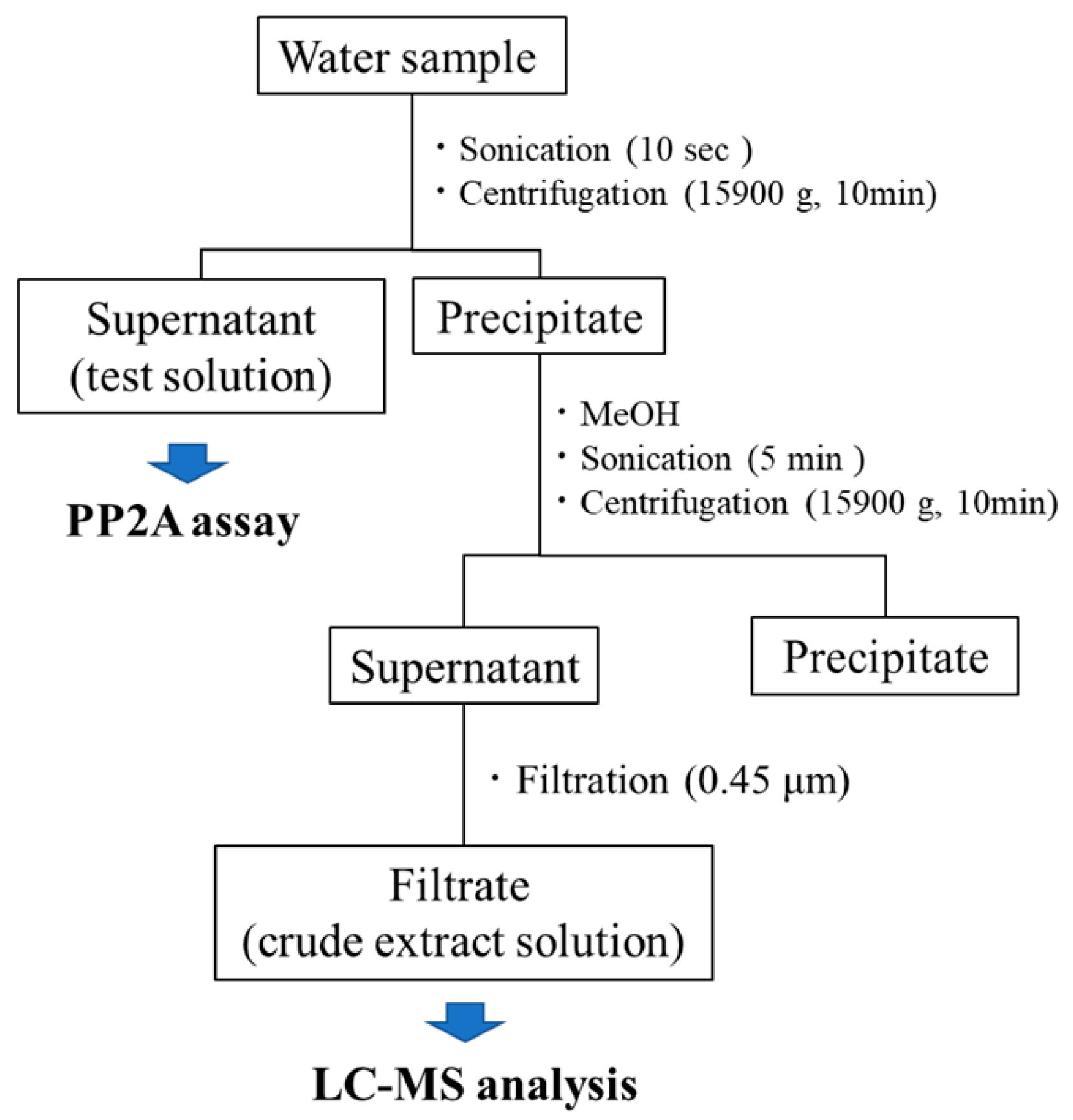
4.1. Water Sample Collection
4.2. Sample Preparation
4.3. PP2A Inhibition Assay
4.4. MC Standards
4.5. LC-MS Analysis
4.6. Isolation and Culture of Cyanobacterial Strains
4.7. Preparation of MC-FR and -WR
4.8. DNA Extraction, ftsZ Amplification and Next-Generation Sequencing
4.9. Next-Generation Sequencing Data Analysis and Phylogenetic Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carmichael, W.W.; Boyer, G.L. Health impacts from cyanobacteria harmful algae blooms: Implications for the north american great lakes. Harmful Algae 2016, 54, 194–212. [Google Scholar] [CrossRef] [PubMed]
- Bartram, J.; Chorus, I. Toxic Cyanobacteria in Water—A Guide to their Public Health Consequences, Monitoring and Management; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Meriluoto, J.; Spoof, L.; Codd, G. A Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; John Wiley & Sons: San Francisco, CA, USA, 2017; Volume 8. [Google Scholar]
- Jochimsen, E.M.; Carmichael, W.W.; An, J.S.; Cardo, D.M.; Cookson, S.T.; Holmes, C.E.; Antunes, M.B.; de Melo Filho, D.A.; Lyra, T.M.; Barreto, V.S.; et al. Liver failure and death after exposure to microcystins at a hemodialysis center in brazil. N. Engl. J. Med. 1998, 338, 873–878. [Google Scholar] [CrossRef] [PubMed]
- Pouria, S.; de Andrade, A.; Barbosa, J.; Cavalcanti, R.L.; Barreto, V.T.; Ward, C.J.; Preiser, W.; Poon, G.K.; Neild, G.H.; Codd, G.A. Fatal microcystin intoxication in haemodialysis unit in caruaru, brazil. Lancet 1998, 352, 21–26. [Google Scholar] [CrossRef]
- Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum; World Health Organization: Geneva, Switzerland, 2017.
- Teta, R.; Romano, V.; Sala, G.D.; Picchio, S.; Sterlich, C.D.; Mangoni, A.; Tullio, G.D.; Costantino, V.; Lega, M. Cyanobacteria as indicators of water quality in campania coasts, italy: A monitoring strategy combining remote/proximal sensing andin situdata. Environ. Res. Lett. 2017, 12, 024001. [Google Scholar] [CrossRef]
- Esposito, G.; Teta, R.; Marrone, R.; De Sterlich, C.; Casazza, M.; Anastasio, A.; Lega, M.; Costantino, V. A fast detection strategy for cyanobacterial blooms and associated cyanotoxins (fdscc) reveals the occurrence of lyngbyatoxin a in campania (south italy). Chemosphere 2019, 225, 342–351. [Google Scholar] [CrossRef] [PubMed]
- MacKintosh, C.; Klumpp, S. Tautomycin from the bacterium streptomyces verticillatus. Another potent and specific inhibitor of protein phosphatases 1 and 2a. FEBS Lett. 1990, 277, 137–140. [Google Scholar] [CrossRef]
- Honkanen, R.E.; Zwiller, J.; Moore, R.E.; Daily, S.L.; Khatra, B.S.; Dukelow, M.; Boynton, A.L. Characterization of microcystin-lr, a potent inhibitor of type 1 and type 2a protein phosphatases. J. Boil. Chem. 1990, 265, 19401–19404. [Google Scholar]
- Ikehara, T.; Shinjo, F.; Ikehara, S.; Imamura, S.; Yasumoto, T. Baculovirus expression, purification, and characterization of human protein phosphatase 2a catalytic subunits alpha and beta. Protein Expr. Purif. 2006, 45, 150–156. [Google Scholar] [CrossRef]
- Ikehara, T.; Nakashima, S.; Nakashima, J.; Kinoshita, T.; Yasumoto, T. Efficient production of recombinant pp2a at a low temperature using a baculovirus expression system. Biotechnol. Rep. 2016, 11, 86–89. [Google Scholar] [CrossRef]
- Ikehara, T.; Imamura, S.; Sano, T.; Nakashima, J.; Kuniyoshi, K.; Oshiro, N.; Yoshimoto, M.; Yasumoto, T. The effect of structural variation in 21 microcystins on their inhibition of pp2a and the effect of replacing cys269 with glycine. Toxicon 2009, 54, 539–544. [Google Scholar] [CrossRef]
- Ikehara, T.; Imamura, S.; Yoshino, A.; Yasumoto, T. Pp2a inhibition assay using recombinant enzyme for rapid detection of okadaic acid and its analogs in shellfish. Toxins 2010, 2, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Ikehara, T.; Kinoshita, T.; Kurokawa, A.; Nakashima, S.; Maekawa, K.; Ohshiro, N.; Yasumoto, T. Evaluation of protein phosphatase 2a (pp2a) inhibition assay for rapid detection of dsp toxins in scallop. NIPPON SUISAN GAKKAISHI 2017, 83, 367–372. [Google Scholar] [CrossRef][Green Version]
- Ikehara, T.; Imamura, S.; Oshiro, N.; Ikehara, S.; Shinjo, F.; Yasumoto, T. A protein phosphatase 2a (pp2a) inhibition assay using a recombinant enzyme for rapid detection of microcystins. Toxicon 2008, 51, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, Y.; Kasai, F.; Watanabe, M.M. Multilocus sequence typing (mlst) reveals high genetic diversity and clonal population structure of the toxic cyanobacterium microcystis aeruginosa. Microbiology 2007, 153, 3695–3703. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, Y.; Watanabe, M.M. Local expansion of a panmictic lineage of water bloom-forming cyanobacterium microcystis aeruginosa. PLoS ONE 2011, 6, e17085. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tanabe, Y.; Hodoki, Y.; Sano, T.; Tada, K.; Watanabe, M.M. Adaptation of the freshwater bloom-forming cyanobacterium microcystis aeruginosa to brackish water is driven by recent horizontal transfer of sucrose genes. Front. Microbiol. 2018, 9, 1150. [Google Scholar] [CrossRef] [PubMed]
- Kaya, K.; Watanabe, M.M. Microcystin composition of an axenic clonal strain of microcystis viridis and microcystis viridis-containing waterblooms in japanese freshwaters. J. Appl. Phycol. 1990, 2, 173–178. [Google Scholar] [CrossRef]
- Namikoshi, M.; Rinehart, K.L.; Sakai, R.; Stotts, R.R.; Dahlem, A.M.; Beasley, V.R.; Carmichael, W.W.; Evans, W.R. Identification of 12 hepatotoxins from a homer lake bloom of the cyanobacteria microcystis aeruginosa, microcystis viridis, and microcystis wesenbergii: Nine new microcystins. J. Org. Chem. 1992, 57, 866–872. [Google Scholar] [CrossRef]
- Lehman, P.; Boyer, G.; Satchwell, M.; Waller, S. The influence of environmental conditions on seasonal variation of microcystis abundance and microcystins concentration in san francisco estuary. Hydrobiologia 2008, 600, 187–204. [Google Scholar] [CrossRef]
- Martins, J.; Saker, M.L.; Moreira, C.; Welker, M.; Fastner, J.; Vasconcelos, V.M. Peptide diversity in strains of the cyanobacterium microcystis aeruginosa isolated from portuguese water supplies. Appl. Microbiol. Biotechnol. 2009, 82, 951–961. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.-H.; Chou, H.-N. Isolation and identification of seven microcystins from a cultured m.Tn-2 strain of microcystis aeruginosa. Bot. Bull. Academia Sin. 2000, 41, 197–202. [Google Scholar]
- Bajpai, R.; Sharma, N.K.; Lawton, L.A.; Edwards, C.; Rai, A.K. Microcystin producing cyanobacterium Nostoc sp. BHU001 from a pond in India. Toxicon 2009, 53, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Douma, M.; Ouahid Benkaddour, Y.; Del Campo, F.; Loudiki, M.; Kh, M.; Oudra, B. Identification and quantification of cyanobacterial toxins (microcystins) in two moroccan drinking-water reservoirs (mansour eddahbi, almassira). Environ. Monit. Assess. 2010, 160, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Kurmayer, R.; Christiansen, G.; Chorus, I. The abundance of microcystin-producing genotypes correlates positively with colony size in microcystis sp. And determines its microcystin net production in lake wannsee. Appl. Environ. Microbiol. 2003, 69, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.A.; Briggs, L.R.; Sprosen, J.; Ruck, J.G.; Wear, R.G.; Holland, P.T.; Bloxham, M. Changes in concentrations of microcystins in rainbow trout, freshwater mussels, and cyanobacteria in lakes rotoiti and rotoehu. Environ. toxicol. 2006, 21, 205–222. [Google Scholar] [CrossRef] [PubMed]
- Robillot, C.; Hennion, M.-C. Issues arising when interpreting the results of the protein phosphatase 2a inhibition assay for the monitoring of microcystins. Anal. Chim. Acta 2004, 512, 339–346. [Google Scholar] [CrossRef]
- Sano, T.; Takagi, H.; Kaya, K. A dhb-microcystin from the filamentous cyanobacterium planktothrix rubescens. Phytochemistry 2004, 65, 2159–2162. [Google Scholar] [CrossRef]
- Quilliam, M.; Hess, P.; Dell’aversano, C. Recent developments in the analysis of phycotoxins by liquid chromatography-mass spectrometry. In Proceedings of the Xth International IUPAC Symposium on Mycotoxins and Phycotoxins, Guaruja, Brazil, 21–25 May 2000; pp. 383–391. [Google Scholar]
- Otsuka, S.; Suda, S.; Li, R.; Matsumoto, S.; Watanabe, M.M. Morphological variability of colonies of microcystis morphospecies in culture. J. Gen. Appl. Microbiol. 2000, 46, 39–50. [Google Scholar] [CrossRef]
- Sano, T.; Takagi, H.; Nishikawa, M.; Kaya, K. Nies certified reference material for microcystins, hepatotoxic cyclic peptide toxins from cyanobacterial blooms in eutrophic water bodies. Anal. Bioanal. Chem. 2008, 391, 2005–2010. [Google Scholar] [CrossRef]
- Edgar, R.C. Muscle: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. Mega7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Boil. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
| Sampling Site No. | PP2A Inhibition (%), Mean ± SD (Test Solution) | MCs Identified by LC/MS (Crude Extract Solution) | Date of Collection | Sample Origin |
|---|---|---|---|---|
| 1 | 43.27 ± 1.12 | MC-RR, -LR, -LA | 10/18/2008 | Reservoir |
| 2 | 7.03 ± 1.39 | - | 10/21/2008 | Artificial pond |
| 3 | 5.61 ± 1.43 | - | 10/21/2008 | Artificial pond |
| 4 | 3.49 ± 3.31 | - | 10/21/2008 | Reservoir |
| 5 | 5.66 ± 1.40 | - | 11/13/2008 | Reservoir |
| 6 | 9.24 ± 0.67 | - | 11/14/2008 | Reservoir |
| 7 | 82.37 ± 0.86 | MC-RR, -LR, -FR, -WR | 12/24/2008 | Artificial pond |
| 8 | 72.70 ± 0.94 | MC-LR, -LA | 12/24/2008 | Reservoir |
| 9 | 3.40 ± 3.12 | - | 12/24/2008 | Artificial pond |
| 10 | 80.50 ± 0.86 | MC-RR, -LR | 12/24/2008 | Reservoir |
| 11 | 78.71 ± 0.77 | MC-RR, -LR | 1/4/2009 | Reservoir |
| 12 | 71.33 ± 2.32 | MC-RR, -LR | 3/2/2009 | Artificial pond |
| 13 | 9.53 ± 0.29 | - | 10/21/2009 | Reservoir |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ikehara, T.; Kuniyoshi, K.; Yamaguchi, H.; Tanabe, Y.; Sano, T.; Yoshimoto, M.; Oshiro, N.; Nakashima, S.; Yasumoto-Hirose, M. First Report of Microcystis Strains Producing MC-FR and -WR Toxins in Japan. Toxins 2019, 11, 521. https://doi.org/10.3390/toxins11090521
Ikehara T, Kuniyoshi K, Yamaguchi H, Tanabe Y, Sano T, Yoshimoto M, Oshiro N, Nakashima S, Yasumoto-Hirose M. First Report of Microcystis Strains Producing MC-FR and -WR Toxins in Japan. Toxins. 2019; 11(9):521. https://doi.org/10.3390/toxins11090521
Chicago/Turabian StyleIkehara, Tsuyoshi, Kyoko Kuniyoshi, Haruyo Yamaguchi, Yuuhiko Tanabe, Tomoharu Sano, Masahiro Yoshimoto, Naomasa Oshiro, Shihoko Nakashima, and Mina Yasumoto-Hirose. 2019. "First Report of Microcystis Strains Producing MC-FR and -WR Toxins in Japan" Toxins 11, no. 9: 521. https://doi.org/10.3390/toxins11090521
APA StyleIkehara, T., Kuniyoshi, K., Yamaguchi, H., Tanabe, Y., Sano, T., Yoshimoto, M., Oshiro, N., Nakashima, S., & Yasumoto-Hirose, M. (2019). First Report of Microcystis Strains Producing MC-FR and -WR Toxins in Japan. Toxins, 11(9), 521. https://doi.org/10.3390/toxins11090521







