Aggregatibacter actinomycetemcomitans Leukotoxin (LtxA; Leukothera®): Mechanisms of Action and Therapeutic Applications
Abstract
1. Introduction
2. Interaction of LtxA with White Blood Cells
2.1. Receptor Independent Interactions with Target Cell Membranes
2.2. Interaction of LtxA with β2 Intergrins
3. Mechanisms of Action
3.1. Monocytes and Macrophages
3.2. Polymorphonuclear Leukocytes
3.3. Lymphocytes
4. Potential Therapeutic Applications
4.1. Leukemia and Lymphoma
4.2. Psoriasis
4.3. Allergic Asthma
4.4. HIV
4.5. The Role of β2 Integrins in Other Autoimmune and Inflammatory Disorders
5. Summary and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fine, D.H.; Kaplan, J.B.; Kachlany, S.C.; Schreiner, H.C. How we got attached to Actinobacillus actinomycetemcomitans: A model for infectious diseases. Periodontology 2000 2006, 42, 114–157. [Google Scholar] [CrossRef] [PubMed]
- Slots, J.; Ting, M. Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis in human periodontal disease: Occurrence and treatment. Periodontology 2000 1999, 20, 82–121. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Badley, A.D.; Cockerill, F.R.; Steckelberg, J.M.; Wilson, W.R. Infective endocarditis caused by HACEK microorganisms. Annu. Rev. Med. 1997, 48, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Shenker, B.J.; McKay, T.; Datar, S.; Miller, M.; Chowhan, R.; Demuth, D. Actinobacillus actinomycetemcomitans immunosuppressive protein is a member of the family of cytolethal distending toxins capable of causing a G2 arrest in human T cells. J. Immunol. (Baltim. Md. 1950) 1999, 162, 4773–4780. [Google Scholar]
- Shenker, B.J.; Besack, D.; McKay, T.; Pankoski, L.; Zekavat, A.; Demuth, D.R. Induction of cell cycle arrest in lymphocytes by Actinobacillus actinomycetemcomitans cytolethal distending toxin requires three subunits for maximum activity. J. Immunol. 2005, 174, 2228–2234. [Google Scholar] [CrossRef] [PubMed]
- Kolodrubetz, D.; Dailey, T.; Ebersole, J.; Kraig, E. Cloning and expression of the leukotoxin gene from Actinobacillus actinomycetemcomitans. Infect. Immun. (Baltim. Md. 1950) 1989, 57, 1465–1469. [Google Scholar]
- Lally, E.T.; Kieba, I.R.; Demuth, D.R.; Rosenbloom, J.; Golub, E.E.; Taichman, N.S.; Gibson, C.W. Identification and expression of the Actinobacillus actinomycetemcomitans leukotoxin gene. Biochem. Biophys. Res. Commun. 1989, 159, 256–262. [Google Scholar] [CrossRef]
- Kachlany, S.C.; Fine, D.H.; Figurski, D.H. Secretion of RTX leukotoxin by Actinobacillus actinomycetemcomitans. Infect. Immun. 2000, 68, 6094–6100. [Google Scholar] [CrossRef]
- Brogan, J.M.; Lally, E.T.; Poulsen, K.; Kilian, M.; Demuth, D.R. Regulation of Actinobacillus actinomycetemcomitans leukotoxin expression: Analysis of the promoter regions of leukotoxic and minimally leukotoxic strains. Infect. Immun. 1994, 62, 501–508. [Google Scholar]
- Sampathkumar, V.; Velusamy, S.K.; Godboley, D.; Fine, D.H. Increased leukotoxin production: Characterization of 100 base pairs within the 530 base pair leukotoxin promoter region of Aggregatibacter actinomycetemcomitans. Sci. Rep. 2017, 7, 1887. [Google Scholar] [CrossRef]
- Balashova, N.V.; Shah, C.; Patel, J.K.; Megalla, S.; Kachlany, S.C. Aggregatibacter actinomycetemcomitans LtxC is required for leukotoxin activity and initial interaction between toxin and host cells. Gene 2009, 443, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Fong, K.P.; Tang, H.Y.; Brown, A.C.; Kieba, I.R.; Speicher, D.W.; Boesze-Battaglia, K.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin is post-translationally modified by addition of either saturated or hydroxylated fatty acyl chains. Mol. Oral Microbiol. 2011, 26, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Lally, E.T.; Golub, E.E.; Kieba, I.R. Identification and immunological characterization of the domain of Actinobacillus actinomycetemcomitans leukotoxin that determines its specificity for human target cells. J. Biol. Chem. 1994, 269, 31289–31295. [Google Scholar] [PubMed]
- Inoue, T.; Tanimoto, I.; Tada, T.; Ohashi, T.; Fukui, K.; Ohta, H. Fermentable-sugar-level-dependent regulation of leukotoxin synthesis in a variably toxic strain of Actinobacillus actinomycetemcomitans. Microbiology (Read. Engl.) 2001, 147, 2749–2756. [Google Scholar] [CrossRef][Green Version]
- Hritz, M.; Fisher, E.; Demuth, D.R. Differential regulation of the leukotoxin operon in highly leukotoxic and minimally leukotoxic strains of Actinobacillus actinomycetemcomitans. Infect. Immun. 1996, 64, 2724–2729. [Google Scholar]
- Kolodrubetz, D.; Phillips, L.; Jacobs, C.; Burgum, A.; Kraig, E. Anaerobic regulation of Actinobacillus actinomycetemcomitans leukotoxin transcription is ArcA/FnrA-independent and requires a novel promoter element. Res. Microbiol. 2003, 154, 645–653. [Google Scholar] [CrossRef]
- Ohta, H.; Miyagi, A.; Kato, K.; Fukui, K. The relationships between leukotoxin production, growth rate and the bicarbonate concentration in a toxin-production-variable strain of Actinobacillus actinomycetemcomitans. Microbiology (Read. Engl.) 1996, 142 Pt 4, 963–970. [Google Scholar] [CrossRef][Green Version]
- Isaza, M.P.; Duncan, M.S.; Kaplan, J.B.; Kachlany, S.C. Screen for leukotoxin mutants in Aggregatibacter actinomycetemcomitans: Genes of the phosphotransferase system are required for leukotoxin biosynthesis. Infect. Immun. 2008, 76, 3561–3568. [Google Scholar] [CrossRef]
- Guthmiller, J.M.; Kolodrubetz, D.; Kraig, E. Mutational analysis of the putative leukotoxin transport genes in Actinobacillus actinomycetemcomitans. Microb. Pathog. 1995, 18, 307–321. [Google Scholar] [CrossRef]
- Guthmiller, J.M.; Kolodrubetz, D.; Cagle, M.P.; Kraig, E. Sequence of the lktB gene from Actinobacillus actinomycetemcomitans. Nucleic Acids Res. 1990, 18, 5291. [Google Scholar] [CrossRef]
- Guthmiller, J.M.; Kraig, E.; Cagle, M.P.; Kolodrubetz, D. Sequence of the lktD gene from Actinobacillus actinomycetemcomitans. Nucleic Acids Res. 1990, 18, 5292. [Google Scholar] [CrossRef] [PubMed]
- Crosby, J.A.; Kachlany, S.C. TdeA, a TolC-like protein required for toxin and drug export in Aggregatibacter (Actinobacillus) actinomycetemcomitans. Gene 2007, 388, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Kachlany, S.C. Aggregatibacter actinomycetemcomitans leukotoxin: From threat to therapy. J. Dent. Res. 2010, 89, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Kraig, E.; Dailey, T.; Kolodrubetz, D. Nucleotide sequence of the leukotoxin gene from Actinobacillus actinomycetemcomitans: Homology to the alpha-hemolysin/leukotoxin gene family. Infect. Immun. 1990, 58, 920–929. [Google Scholar] [PubMed]
- Baumann, U.; Wu, S.; Flaherty, K.M.; McKay, D.B. Three-dimensional structure of the alkaline protease of Pseudomonas aeruginosa: A two-domain protein with a calcium binding parallel beta roll motif. EMBO J. 1993, 12, 3357–3364. [Google Scholar] [CrossRef] [PubMed]
- Boehm, D.F.; Welch, R.A.; Snyder, I.S. Domains of Escherichia coli hemolysin (HlyA) involved in binding of calcium and erythrocyte membranes. Infect. Immun. 1990, 58, 1959–1964. [Google Scholar]
- Ostolaza, H.; Soloaga, A.; Goni, F.M. The binding of divalent cations to Escherichia coli alpha-haemolysin. Eur. J. Biochem. FEBS 1995, 228, 39–44. [Google Scholar] [CrossRef]
- Stanley, P.; Koronakis, V.; Hughes, C. Acylation of Escherichia coli hemolysin: A unique protein lipidation mechanism underlying toxin function. Microbiol. Mol. Biol. Rev. MMBR 1998, 62, 309–333. [Google Scholar]
- Stanley, P.; Packman, L.C.; Koronakis, V.; Hughes, C. Fatty acylation of two internal lysine residues required for the toxic activity of Escherichia coli hemolysin. Science (New York N.Y.) 1994, 266, 1992–1996. [Google Scholar] [CrossRef]
- Sanchez-Magraner, L.; Viguera, A.R.; Garcia-Pacios, M.; Garcillan, M.P.; Arrondo, J.L.; de la Cruz, F.; Goni, F.M.; Ostolaza, H. The calcium-binding C-terminal domain of Escherichia coli alpha-hemolysin is a major determinant in the surface-active properties of the protein. J. Biol. Chem. 2007, 282, 11827–11835. [Google Scholar] [CrossRef]
- Brown, A.C.; Balashova, N.V.; Epand, R.M.; Epand, R.F.; Bragin, A.; Kachlany, S.C.; Walters, M.J.; Du, Y.; Boesze-Battaglia, K.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin utilizes a cholesterol recognition/amino acid consensus site for membrane association. J. Biol. Chem. 2013, 288, 23607–23621. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C.; Boesze-Battaglia, K.; Balashova, N.V.; Mas Gomez, N.; Speicher, K.; Tang, H.Y.; Duszyk, M.E.; Lally, E.T. Membrane localization of the Repeats-in-Toxin (RTX) Leukotoxin (LtxA) produced by Aggregatibacter actinomycetemcomitans. PLoS ONE 2018, 13, e0205871. [Google Scholar] [CrossRef]
- DiFranco, K.M.; Kaswala, R.H.; Patel, C.; Kasinathan, C.; Kachlany, S.C. Leukotoxin kills rodent WBC by targeting leukocyte function associated antigen 1. Comp. Med. 2013, 63, 331–337. [Google Scholar] [PubMed]
- Simpson, D.L.; Berthold, P.; Taichman, N.S. Killing of human myelomonocytic leukemia and lymphocytic cell lines by Actinobacillus actinomycetemcomitans leukotoxin. Infect. Immun. 1988, 56, 1162–1166. [Google Scholar] [PubMed]
- Kachlany, S.C.; Schwartz, A.B.; Balashova, N.V.; Hioe, C.E.; Tuen, M.; Le, A.; Kaur, M.; Mei, Y.; Rao, J. Anti-leukemia activity of a bacterial toxin with natural specificity for LFA-1 on white blood cells. Leuk. Res. 2010, 34, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Taichman, N.S.; Iwase, M.; Lally, E.T.; Shattil, S.J.; Cunningham, M.E.; Korchak, H.M. Early changes in cytosolic calcium and membrane potential induced by Actinobacillus actinomycetemcomitans leukotoxin in susceptible and resistant target cells. J. Immunol. (Baltim. Md. 1950) 1991, 147, 3587–3594. [Google Scholar]
- Iwase, M.; Lally, E.T.; Berthold, P.; Korchak, H.M.; Taichman, N.S. Effects of cations and osmotic protectants on cytolytic activity of Actinobacillus actinomycetemcomitans leukotoxin. Infect. Immun. 1990, 58, 1782–1788. [Google Scholar] [PubMed]
- Fong, K.P.; Pacheco, C.M.; Otis, L.L.; Baranwal, S.; Kieba, I.R.; Harrison, G.; Hersh, E.V.; Boesze-Battaglia, K.; Lally, E.T. Actinobacillus actinomycetemcomitans leukotoxin requires lipid microdomains for target cell cytotoxicity. Cell. Microbiol. 2006, 8, 1753–1767. [Google Scholar] [CrossRef] [PubMed]
- Lally, E.T.; Hill, R.B.; Kieba, I.R.; Korostoff, J. The interaction between RTX toxins and target cells. Trends Microbiol. 1999, 7, 356–361. [Google Scholar] [CrossRef]
- Brown, A.C.; Boesze-Battaglia, K.; Du, Y.; Stefano, F.P.; Kieba, I.R.; Epand, R.F.; Kakalis, L.; Yeagle, P.L.; Epand, R.M.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin cytotoxicity occurs through bilayer destabilization. Cell. Microbiol. 2012, 14, 869–881. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, A.; Jarchau, T.; Benz, R.; Goebel, W. The repeat domain of Escherichia coli haemolysin (HlyA) is responsible for its Ca2+-dependent binding to erythrocytes. Mol. Gen. Genet. MGG 1988, 214, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.; Aranda, F.J.; Villalain, J.; San Martin, C.; Micol, V.; Gomez-Fernandez, J.C. 1,2-Dioleoylglycerol promotes calcium-induced fusion in phospholipid vesicles. Chem. Phys. Lipids 1992, 62, 215–224. [Google Scholar] [CrossRef]
- Ortiz, A.; Killian, J.A.; Verkleij, A.J.; Wilschut, J. Membrane fusion and the lamellar-to-inverted-hexagonal phase transition in cardiolipin vesicle systems induced by divalent cations. Biophys. J. 1999, 77, 2003–2014. [Google Scholar] [CrossRef]
- Walters, M.J.; Brown, A.C.; Edrington, T.C.; Baranwal, S.; Du, Y.; Lally, E.T.; Boesze-Battaglia, K. Membrane association and destabilization by Aggregatibacter actinomycetemcomitans leukotoxin requires changes in secondary structures. Mol. Oral Microbiol. 2013, 28, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Lear, J.D.; Karakelian, D.; Furblur, U.; Lally, E.T.; Tanaka, J.C. Conformational studies of Actinobacillus actinomycetemcomitans leukotoxin: Partial denaturation enhances toxicity. Biochim. Biophys. Acta 2000, 1476, 350–362. [Google Scholar] [CrossRef]
- Brown, A.C.; Koufos, E.; Balashova, N.V.; Boesze-Battaglia, K.; Lally, E.T. Inhibition of LtxA toxicity by blocking cholesterol binding with peptides. Mol. Oral Microbiol. 2016, 31, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Koufos, E.; Chang, E.H.; Rasti, E.S.; Krueger, E.; Brown, A.C. Use of a Cholesterol Recognition Amino Acid Consensus Peptide To Inhibit Binding of a Bacterial Toxin to Cholesterol. Biochemistry 2016, 55, 4787–4797. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.P.; McDowall, A.; Hogg, N. LFA-1-mediated adhesion is regulated by cytoskeletal restraint and by a Ca2+-dependent protease, calpain. J. Cell Biol. 1998, 140, 699–707. [Google Scholar] [CrossRef]
- Kucik, D.F.; Dustin, M.L.; Miller, J.M.; Brown, E.J. Adhesion-activating phorbol ester increases the mobility of leukocyte integrin LFA-1 in cultured lymphocytes. J. Clin. Investig. 1996, 97, 2139–2144. [Google Scholar] [CrossRef]
- Lally, E.T.; Kieba, I.R.; Sato, A.; Green, C.L.; Rosenbloom, J.; Korostoff, J.; Wang, J.F.; Shenker, B.J.; Ortlepp, S.; Robinson, M.K.; et al. RTX Toxins Recognize a 2 Integrin on the Surface of Human Target Cells. J. Biol. Chem. 1997, 272, 30463–30469. [Google Scholar] [CrossRef]
- Reinholdt, J.; Poulsen, K.; Brinkmann, C.R.; Hoffmann, S.V.; Stapulionis, R.; Enghild, J.J.; Jensen, U.B.; Boesen, T.; Vorup-Jensen, T. Monodisperse and LPS-free Aggregatibacter actinomycetemcomitans leukotoxin: Interactions with human beta2 integrins and erythrocytes. Biochim. Biophys. Acta 2013, 1834, 546–558. [Google Scholar] [CrossRef]
- Miller, L.J.; Schwarting, R.; Springer, T.A. Regulated expression of the Mac-1, LFA-1, p150,95 glycoprotein family during leukocyte differentiation. J. Immunol. (Baltim. Md. 1950) 1986, 137, 2891–2900. [Google Scholar]
- Miller, L.J.; Bainton, D.F.; Borregaard, N.; Springer, T.A. Stimulated mobilization of monocyte Mac-1 and p150,95 adhesion proteins from an intracellular vesicular compartment to the cell surface. J. Clin. Investig. 1987, 80, 535–544. [Google Scholar] [CrossRef]
- Kinashi, T. Intracellular signalling controlling integrin activation in lymphocytes. Nat. Rev. Immunol. 2005, 5, 546–559. [Google Scholar] [CrossRef]
- Hogg, N.; Smith, A.; McDowall, A.; Giles, K.; Stanley, P.; Laschinger, M.; Henderson, R. How T cells use LFA-1 to attach and migrate. Immunol. Lett. 2004, 92, 51–54. [Google Scholar] [CrossRef]
- Hogg, N.; Harvey, J.; Cabanas, C.; Landis, R.C. Control of leukocyte integrin activation. Am. Rev. Respir. Dis. 1993, 148, S55–S59. [Google Scholar] [CrossRef]
- de la Fuente, M.T.; Casanova, B.; Moyano, J.V.; Garcia-Gila, M.; Sanz, L.; Garcia-Marco, J.; Silva, A.; Garcia-Pardo, A. Engagement of alpha4beta1 integrin by fibronectin induces in vitro resistance of B chronic lymphocytic leukemia cells to fludarabine. J. Leukoc. Biol. 2002, 71, 495–502. [Google Scholar]
- Damiano, J.S.; Hazlehurst, L.A.; Dalton, W.S. Cell adhesion-mediated drug resistance (CAM-DR) protects the K562 chronic myelogenous leukemia cell line from apoptosis induced by BCR/ABL inhibition, cytotoxic drugs, and gamma-irradiation. Leukemia 2001, 15, 1232–1239. [Google Scholar] [CrossRef]
- Dileepan, T.; Kachlany, S.C.; Balashova, N.V.; Patel, J.; Maheswaran, S.K. Human CD18 is the functional receptor for Aggregatibacter actinomycetemcomitans leukotoxin. Infect. Immun. 2007, 75, 4851–4856. [Google Scholar] [CrossRef]
- Kieba, I.R.; Fong, K.P.; Tang, H.Y.; Hoffman, K.E.; Speicher, D.W.; Klickstein, L.B.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin requires beta-sheets 1 and 2 of the human CD11a beta-propeller for cytotoxicity. Cell. Microbiol. 2007, 9, 2689–2699. [Google Scholar] [CrossRef]
- Morova, J.; Osicka, R.; Masin, J.; Sebo, P. RTX cytotoxins recognize beta2 integrin receptors through N-linked oligosaccharides. Proc. Natl. Acad. Sci. USA 2008, 105, 5355–5360. [Google Scholar] [CrossRef]
- Munksgaard, P.S.; Skals, M.; Reinholdt, J.; Poulsen, K.; Jensen, M.R.; Yang, C.; Leipziger, J.; Vorup-Jensen, T.; Praetorius, H.A. Sialic acid residues are essential for cell lysis mediated by leukotoxin from Aggregatibacter actinomycetemcomitans. Infect. Immun. 2014, 82, 2219–2228. [Google Scholar] [CrossRef]
- Nygren, P.; Balashova, N.; Brown, A.C.; Kieba, I.; Dhingra, A.; Boesze-Battaglia, K.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin causes activation of LFA-1. Cell. Microbiol. 2019, 21, e12967. [Google Scholar] [CrossRef]
- Ristow, L.C.; Tran, V.; Schwartz, K.J.; Pankratz, L.; Mehle, A.; Sauer, J.D.; Welch, R.A. The Extracellular Domain of the beta2 Integrin beta Subunit (CD18) Is Sufficient for Escherichia coli Hemolysin and Aggregatibacter actinomycetemcomitans Leukotoxin Cytotoxic Activity. mBio 2019. [Google Scholar] [CrossRef]
- Hioe, C.E.; Tuen, M.; Vasiliver-Shamis, G.; Alvarez, Y.; Prins, K.C.; Banerjee, S.; Nadas, A.; Cho, M.W.; Dustin, M.L.; Kachlany, S.C. HIV envelope gp120 activates LFA-1 on CD4 T-lymphocytes and increases cell susceptibility to LFA-1-targeting leukotoxin (LtxA). PLoS ONE 2011, 6, e23202. [Google Scholar] [CrossRef]
- DiFranco, K.M.; Gupta, A.; Galusha, L.E.; Perez, J.; Nguyen, T.V.; Fineza, C.D.; Kachlany, S.C. Leukotoxin (Leukothera(R)) targets active leukocyte function antigen-1 (LFA-1) protein and triggers a lysosomal mediated cell death pathway. J. Biol. Chem. 2012, 287, 17618–17627. [Google Scholar] [CrossRef]
- Korostoff, J.; Wang, J.F.; Kieba, I.; Miller, M.; Shenker, B.J.; Lally, E.T. Actinobacillus actinomycetemcomitans leukotoxin induces apoptosis in HL-60 cells. Infect. Immun. 1998, 66, 4474–4483. [Google Scholar]
- Korostoff, J.; Yamaguchi, N.; Miller, M.; Kieba, I.; Lally, E.T. Perturbation of mitochondrial structure and function plays a central role in Actinobacillus actinomycetemcomitans leukotoxin-induced apoptosis. Microb. Pathog. 2000, 29, 267–278. [Google Scholar] [CrossRef]
- Mangan, D.F.; Taichman, N.S.; Lally, E.T.; Wahl, S.M. Lethal effects of Actinobacillus actinomycetemcomitans leukotoxin on human T lymphocytes. Infect. Immun. 1991, 59, 3267–3272. [Google Scholar]
- Kelk, P.; Johansson, A.; Claesson, R.; Hanstrom, L.; Kalfas, S. Caspase 1 involvement in human monocyte lysis induced by Actinobacillus actinomycetemcomitans leukotoxin. Infect. Immun. 2003, 71, 4448–4455. [Google Scholar] [CrossRef]
- Kelk, P.; Abd, H.; Claesson, R.; Sandstrom, G.; Sjostedt, A.; Johansson, A. Cellular and molecular response of human macrophages exposed to Aggregatibacter actinomycetemcomitans leukotoxin. Cell Death Dis. 2011, 2, e126. [Google Scholar] [CrossRef]
- Fagerberg, S.K.; Jakobsen, M.R.; Skals, M.; Praetorius, H.A. Inhibition of P2X Receptors Protects Human Monocytes against Damage by Leukotoxin from Aggregatibacter actinomycetemcomitans and alpha-Hemolysin from Escherichia coli. Infect. Immun. 2016, 84, 3114–3130. [Google Scholar] [CrossRef]
- Kaur, M.; Kachlany, S.C. Aggregatibacter actinomycetemcomitans leukotoxin (LtxA.; Leukothera) induces cofilin dephosphorylation and actin depolymerization during killing of malignant monocytes. Microbiol. (Read. Engl.) 2014, 160, 2443–2452. [Google Scholar] [CrossRef]
- Kelk, P.; Claesson, R.; Chen, C.; Sjostedt, A.; Johansson, A. IL-1beta secretion induced by Aggregatibacter (Actinobacillus) actinomycetemcomitans is mainly caused by the leukotoxin. Int. J. Med. Microbiol. IJMM 2008, 298, 529–541. [Google Scholar] [CrossRef]
- Kelk, P.; Claesson, R.; Hanstrom, L.; Lerner, U.H.; Kalfas, S.; Johansson, A. Abundant secretion of bioactive interleukin-1beta by human macrophages induced by Actinobacillus actinomycetemcomitans leukotoxin. Infect. Immun. 2005, 73, 453–458. [Google Scholar] [CrossRef]
- Belibasakis, G.N.; Johansson, A. Aggregatibacter actinomycetemcomitans targets NLRP3 and NLRP6 inflammasome expression in human mononuclear leukocytes. Cytokine 2012, 59, 124–130. [Google Scholar] [CrossRef]
- Cookson, B.T.; Brennan, M.A. Pro-inflammatory programmed cell death. Trends Microbiol. 2001, 9, 113–114. [Google Scholar] [CrossRef]
- Balashova, N.; Dhingra, A.; Boesze-Battaglia, K.; Lally, E.T. Aggregatibacter actinomycetemcomitans leukotoxin induces cytosol acidification in LFA-1 expressing immune cells. Mol. Oral Microbiol. 2016, 31, 106–114. [Google Scholar] [CrossRef]
- Johansson, A.; Claesson, R.; Hanstrom, L.; Sandstrom, G.; Kalfas, S. Polymorphonuclear leukocyte degranulation induced by leukotoxin from Actinobacillus actinomycetemcomitans. J. Periodontal Res. 2000, 35, 85–92. [Google Scholar] [CrossRef]
- Aktas, E.; Kucuksezer, U.C.; Bilgic, S.; Erten, G.; Deniz, G. Relationship between CD107a expression and cytotoxic activity. Cell. Immunol. 2009, 254, 149–154. [Google Scholar] [CrossRef]
- Johansson, A.; Sandstrom, G.; Claesson, R.; Hanstrom, L.; Kalfas, S. Anaerobic neutrophil-dependent killing of Actinobacillus actinomycetemcomitans in relation to the bacterial leukotoxicity. Eur. J. Oral Sci. 2000, 108, 136–146. [Google Scholar] [CrossRef]
- Furugen, R.; Hayashida, H.; Yoshii, Y.; Saito, T. Neutrophil-derived resistin release induced by Aggregatibacter actinomycetemcomitans. FEMS Microbiol. Lett. 2011, 321, 175–182. [Google Scholar] [CrossRef]
- Claesson, R.; Johansson, A.; Belibasakis, G.; Hanstrom, L.; Kalfas, S. Release and activation of matrix metalloproteinase 8 from human neutrophils triggered by the leukotoxin of Actinobacillus actinomycetemcomitans. J. Periodontal Res. 2002, 37, 353–359. [Google Scholar] [CrossRef]
- Taichman, N.S.; Dean, R.T.; Sanderson, C.J. Biochemical and morphological characterization of the killing of human monocytes by a leukotoxin derived from Actinobacillus actinomycetemcomitans. Infect. Immun. 1980, 28, 258–268. [Google Scholar]
- Tsai, C.C.; McArthur, W.P.; Baehni, P.C.; Hammond, B.F.; Taichman, N.S. Extraction and partial characterization of a leukotoxin from a plaque-derived Gram-negative microorganism. Infect. Immun. 1979, 25, 427–439. [Google Scholar]
- Hiyoshi, T.; Domon, H.; Maekawa, T.; Nagai, K.; Tamura, H.; Takahashi, N.; Yonezawa, D.; Miyoshi, T.; Yoshida, A.; Tabeta, K.; et al. Aggregatibacter actinomycetemcomitans induces detachment and death of human gingival epithelial cells and fibroblasts via elastase release following leukotoxin-dependent neutrophil lysis. Microbiol. Immunol. 2019, 63, 100–110. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science (New York N.Y.) 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Urban, C.F.; Ermert, D.; Schmid, M.; Abu-Abed, U.; Goosmann, C.; Nacken, W.; Brinkmann, V.; Jungblut, P.R.; Zychlinsky, A. Neutrophil extracellular traps contain calprotectin, a cytosolic protein complex involved in host defense against Candida albicans. PLoS Pathog. 2009, 5, e1000639. [Google Scholar] [CrossRef]
- Jorch, S.K.; Kubes, P. An emerging role for neutrophil extracellular traps in noninfectious disease. Nat. Med. 2017, 23, 279–289. [Google Scholar] [CrossRef]
- Hirschfeld, J.; Roberts, H.M.; Chapple, I.L.; Parcina, M.; Jepsen, S.; Johansson, A.; Claesson, R. Effects of Aggregatibacter actinomycetemcomitans leukotoxin on neutrophil migration and extracellular trap formation. J. Oral Microbiol. 2016, 8, 33070. [Google Scholar] [CrossRef]
- Konig, M.F.; Abusleme, L.; Reinholdt, J.; Palmer, R.J.; Teles, R.P.; Sampson, K.; Rosen, A.; Nigrovic, P.A.; Sokolove, J.; Giles, J.T.; et al. Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Sci. Transl. Med. 2016, 8, 369ra176. [Google Scholar] [CrossRef]
- McDonald, B.; Urrutia, R.; Yipp, B.G.; Jenne, C.N.; Kubes, P. Intravascular neutrophil extracellular traps capture bacteria from the bloodstream during sepsis. Cell Host Microbe 2012, 12, 324–333. [Google Scholar] [CrossRef]
- Baehni, P.; Tsai, C.C.; McArthur, W.P.; Hammond, B.F.; Taichman, N.S. Interaction of inflammatory cells and oral microorganisms. VIII. Detection of leukotoxic activity of a plaque-derived gram-negative microorganism. Infect. Immun. 1979, 24, 233–243. [Google Scholar]
- Katagiri, K.; Maeda, A.; Shimonaka, M.; Kinashi, T. RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat. Immunol. 2003, 4, 741–748. [Google Scholar] [CrossRef]
- Shimonaka, M.; Katagiri, K.; Nakayama, T.; Fujita, N.; Tsuruo, T.; Yoshie, O.; Kinashi, T. Rap1 translates chemokine signals to integrin activation, cell polarization, and motility across vascular endothelium under flow. J. Cell Biol. 2003, 161, 417–427. [Google Scholar] [CrossRef]
- Yang, B.; Sun, H.; Li, W.; Zhu, C.; Jian, B.; Hou, W.; Wang, H.; Yuan, J.; Yao, B. Expression of Rap1 during germ cell development in the rat and its functional implications in 2-methoxyacetic acid-induced spermatocyte apoptosis. Urology 2013, 81, 696.e1–698.e8. [Google Scholar] [CrossRef]
- Liu, G.; Shi, Z.; Jiao, S.; Zhang, Z.; Wang, W.; Chen, C.; Hao, Q.; Zhang, M.; Feng, M.; Xu, L.; et al. Structure of MST2 SARAH domain provides insights into its interaction with RAPL. J. Struct. Biol. 2014, 185, 366–374. [Google Scholar] [CrossRef]
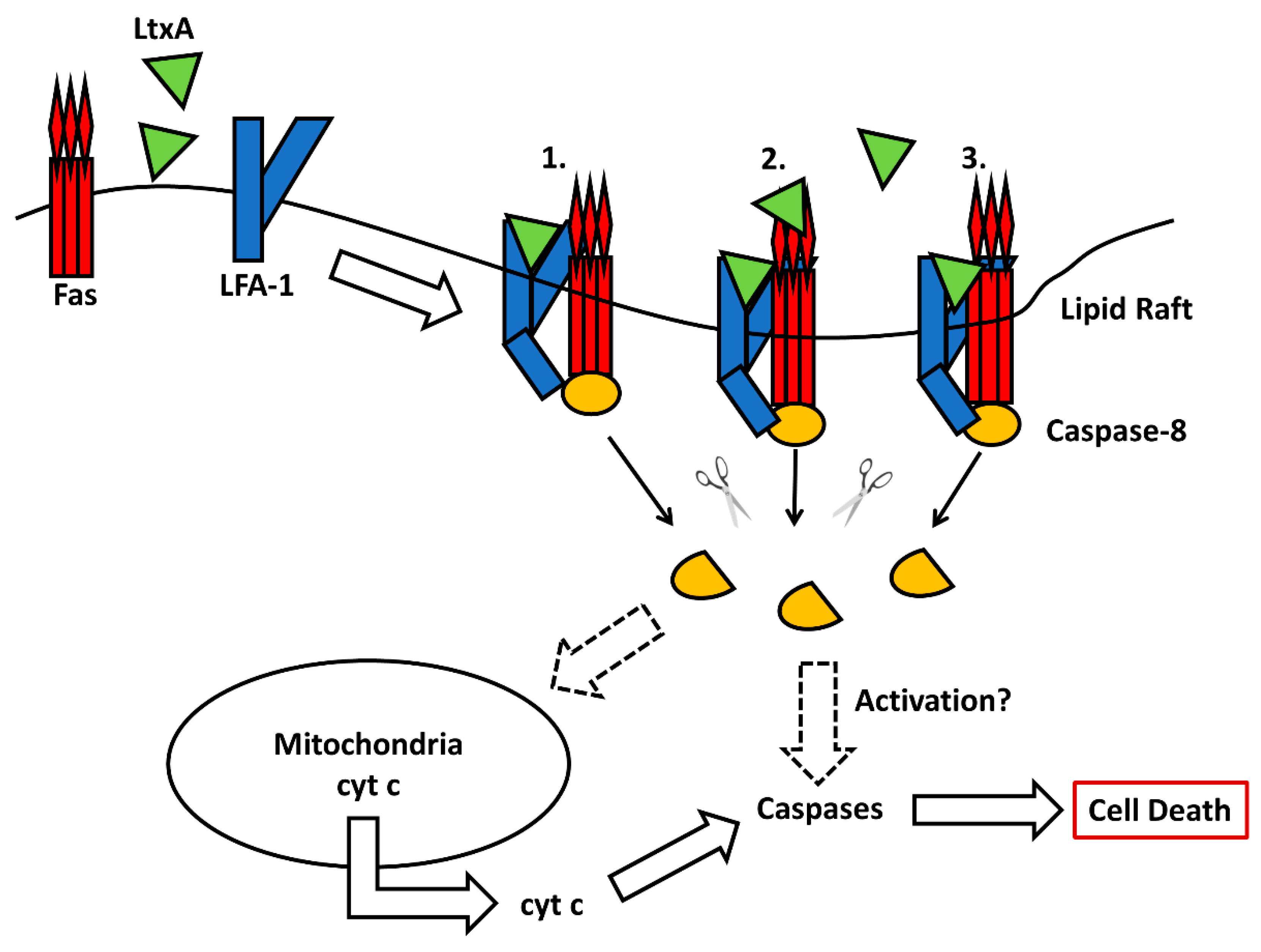
- DiFranco, K.M.; Johnson-Farley, N.; Bertino, J.R.; Elson, D.; Vega, B.A.; Belinka, B.A., Jr.; Kachlany, S.C. LFA-1-targeting Leukotoxin (LtxA.; Leukothera(R)) causes lymphoma tumor regression in a humanized mouse model and requires caspase-8 and Fas to kill malignant lymphocytes. Leuk. Res. 2015, 39, 649–656. [Google Scholar] [CrossRef]
- Vega, B.A.; Schober, L.T.; Kim, T.; Belinka, B.A., Jr.; Kachlany, S.C. Aggregatibacter actinomycetemcomitans Leukotoxin (LtxA) Requires Death Receptor Fas, in Addition to LFA-1, To Trigger Cell Death in T Lymphocytes. Infect. Immun. 2019, 87, e00309-19. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Kieba, I.R.; Korostoff, J.; Howard, P.S.; Shenker, B.J.; Lally, E.T. Maintenance of oxidative phosphorylation protects cells from Actinobacillus actinomycetemcomitans leukotoxin-induced apoptosis. Cell. Microbiol. 2001, 3, 811–823. [Google Scholar] [CrossRef]
- Scaffidi, C.; Fulda, S.; Srinivasan, A.; Friesen, C.; Li, F.; Tomaselli, K.J.; Debatin, K.M.; Krammer, P.H.; Peter, M.E. Two CD95 (APO-1/Fas) signaling pathways. EMBO J. 1998, 17, 1675–1687. [Google Scholar] [CrossRef]
- Petit, P.X.; Lecoeur, H.; Zorn, E.; Dauguet, C.; Mignotte, B.; Gougeon, M.L. Alterations in mitochondrial structure and function are early events of dexamethasone-induced thymocyte apoptosis. J. Cell Biol. 1995, 130, 157–167. [Google Scholar] [CrossRef]
- Petit, P.X.; Goubern, M.; Diolez, P.; Susin, S.A.; Zamzami, N.; Kroemer, G. Disruption of the outer mitochondrial membrane as a result of large amplitude swelling: The impact of irreversible permeability transition. FEBS Lett. 1998, 426, 111–116. [Google Scholar] [CrossRef]
- Jankovic, J. Botulinum toxin in clinical practice. J. Neurol. Neurosurg. Psychiatry 2004, 75, 951–957. [Google Scholar] [CrossRef]
- Duvic, M.; Kuzel, T.M.; Olsen, E.A.; Martin, A.G.; Foss, F.M.; Kim, Y.H.; Heald, P.W.; Bacha, P.; Nichols, J.; Liepa, A. Quality-of-life improvements in cutaneous T-cell lymphoma patients treated with denileukin diftitox (ONTAK). Clin. Lymphoma 2002, 2, 222–228. [Google Scholar] [CrossRef]
- Olsen, E.; Duvic, M.; Frankel, A.; Kim, Y.; Martin, A.; Vonderheid, E.; Jegasothy, B.; Wood, G.; Gordon, M.; Heald, P.; et al. Pivotal phase III trial of two dose levels of denileukin diftitox for the treatment of cutaneous T-cell lymphoma. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2001, 19, 376–388. [Google Scholar] [CrossRef]
- Bechter, O.E.; Eisterer, W.; Dirnhofer, S.; Pall, G.; Kuhr, T.; Stauder, R.; Thaler, J. Expression of LFA-1 identifies different prognostic subgroups in patients with advanced follicle center lymphoma (FCL). Leuk. Res. 1999, 23, 483–488. [Google Scholar] [CrossRef]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef]
- Inghirami, G.; Wieczorek, R.; Zhu, B.Y.; Silber, R.; Dalla-Favera, R.; Knowles, D.M. Differential expression of LFA-1 molecules in non-Hodgkin’s lymphoma and lymphoid leukemia. Blood 1988, 72, 1431–1434. [Google Scholar]
- Horst, E.; Radaszkiewicz, T.; Hooftman-den Otter, A.; Pieters, R.; van Dongen, J.J.; Meijer, C.J.; Pals, S.T. Expression of the leucocyte integrin LFA-1 (CD11a/CD18) and its ligand ICAM-1 (CD54) in lymphoid malignancies is related to lineage derivation and stage of differentiation but not to tumor grade. Leukemia 1991, 5, 848–853. [Google Scholar]
- de Boer, O.J.; Wakelkamp, I.M.; Pals, S.T.; Claessen, N.; Bos, J.D.; Das, P.K. Increased expression of adhesion receptors in both lesional and non-lesional psoriatic skin. Arch. Dermatol. Res. 1994, 286, 304–311. [Google Scholar] [CrossRef]
- McGregor, J.M.; Barker, J.N.; Ross, E.L.; MacDonald, D.M. Epidermal dendritic cells in psoriasis possess a phenotype associated with antigen presentation: In situ expression of beta 2-integrins. J. Am. Acad. Dermatol. 1992, 27, 383–388. [Google Scholar] [CrossRef]
- Lantero, S.; Alessandri, G.; Spallarossa, D.; Scarso, L.; Rossi, G.A. LFA-1 expression by blood eosinophils is increased in atopic asthmatic children and is involved in eosinophil locomotion. Eur. Respir. J. 1998, 12, 1094–1098. [Google Scholar] [CrossRef]
- Gupta, A.; Espinosa, V.; Galusha, L.E.; Rahimian, V.; Miro, K.L.; Rivera-Medina, A.; Kasinathan, C.; Capitle, E.; Aguila, H.A.; Kachlany, S.C. Expression and targeting of lymphocyte function-associated antigen 1 (LFA-1) on white blood cells for treatment of allergic asthma. J. Leukoc. Biol. 2014, 97, 439–446. [Google Scholar] [CrossRef]
- Arthos, J.; Cicala, C.; Martinelli, E.; Macleod, K.; Van Ryk, D.; Wei, D.; Xiao, Z.; Veenstra, T.D.; Conrad, T.P.; Lempicki, R.A.; et al. HIV-1 envelope protein binds to and signals through integrin alpha4beta7, the gut mucosal homing receptor for peripheral T cells. Nat. Immunol. 2008, 9, 301–309. [Google Scholar] [CrossRef]
- Bastiani, L.; Laal, S.; Kim, M.; Zolla-Pazner, S. Host cell-dependent alterations in envelope components of human immunodeficiency virus type 1 virions. J. Virol. 1997, 71, 3444–3450. [Google Scholar]
- Capobianchi, M.R.; Fais, S.; Castilletti, C.; Gentile, M.; Ameglio, F.; Dianzani, F. A simple and reliable method to detect cell membrane proteins on infectious human immunodeficiency virus type 1 particles. J. Infect. Dis. 1994, 169, 886–889. [Google Scholar] [CrossRef]
- Elston, D.M.; McCollough, M.L.; Bergfeld, W.F.; Liranzo, M.O.; Heibel, M. Eosinophils in fibrous tracts and near hair bulbs: A helpful diagnostic feature of alopecia areata. J. Am. Acad. Dermatol. 1997, 37, 101–106. [Google Scholar] [CrossRef]
- Gilhar, A.; David, M.; Ullmann, Y.; Berkutski, T.; Kalish, R.S. T-lymphocyte dependence of psoriatic pathology in human psoriatic skin grafted to SCID mice. J. Investig. Dermatol. 1997, 109, 283–288. [Google Scholar] [CrossRef]
- Ito, T.; Tokura, Y. The role of cytokines and chemokines in the T-cell-mediated autoimmune process in alopecia areata. Exp. Dermatol. 2014, 23, 787–791. [Google Scholar] [CrossRef]
- Santos, Z.; Avci, P.; Hamblin, M.R. Drug discovery for alopecia: Gone today, hair tomorrow. Expert Opin. Drug Discov. 2015, 10, 269–292. [Google Scholar] [CrossRef]
- Wasserman, D.; Guzman-Sanchez, D.A.; Scott, K.; McMichael, A. Alopecia areata. Int. J. Dermatol. 2007, 46, 121–131. [Google Scholar] [CrossRef]
- Pallis, M.; Robson, D.K.; Haskard, D.O.; Powell, R.J. Distribution of cell adhesion molecules in skeletal muscle from patients with systemic lupus erythematosus. Ann. Rheum. Dis. 1993, 52, 667–671. [Google Scholar] [CrossRef]
- Takeuchi, T.; Amano, K.; Sekine, H.; Koide, J.; Abe, T. Upregulated expression and function of integrin adhesive receptors in systemic lupus erythematosus patients with vasculitis. J. Clin. Investig. 1993, 92, 3008–3016. [Google Scholar] [CrossRef]
- Takasaki, Y.; Abe, K.; Tokano, Y.; Hashimoto, H. The expression of LFA-1, ICAM-1, CD80 and CD86 molecules in lupus patients: Implication for immunotherapy. Intern. Med. (Tokyo Jpn.) 1999, 38, 175–177. [Google Scholar] [CrossRef][Green Version]
- Kaneko, H.; Tokano, Y.; Hashimoto, H.; Hirose, S. The expression of lymphocyte function associated antigen-1, intercellular adhesion molecule-1 on peripheral blood lymphocytes in patients with systemic lupus erythematosus. Nihon Rinsho Men’eki Gakkai Kaishi Jpn. J. Clin. Immunol. 1996, 19, 60–68. [Google Scholar] [CrossRef][Green Version]
- Elovaara, I.; Ukkonen, M.; Leppakynnas, M.; Lehtimaki, T.; Luomala, M.; Peltola, J.; Dastidar, P. Adhesion molecules in multiple sclerosis: Relation to subtypes of disease and methylprednisolone therapy. Arch. Neurol. 2000, 57, 546–551. [Google Scholar] [CrossRef]
- Lou, J.; Chofflon, M.; Juillard, C.; Donati, Y.; Mili, N.; Siegrist, C.A.; Grau, G.E. Brain microvascular endothelial cells and leukocytes derived from patients with multiple sclerosis exhibit increased adhesion capacity. Neuroreport 1997, 8, 629–633. [Google Scholar] [CrossRef]
- Bernstein, C.N.; Sargent, M.; Rector, E. Alteration in expression of beta 2 integrins on lamina propria lymphocytes in ulcerative colitis and Crohn’s disease. Clin. Immunol. (Orlando Fla.) 2002, 104, 67–72. [Google Scholar] [CrossRef]
- Vainer, B.; Nielsen, O.H.; Horn, T. Comparative studies of the colonic in situ expression of intercellular adhesion molecules (ICAM-1, -2, and -3), beta2 integrins (LFA-1, Mac-1, and p150,95), and PECAM-1 in ulcerative colitis and Crohn’s disease. Am. J. Surg. Pathol. 2000, 24, 1115–1124. [Google Scholar] [CrossRef]
- Palmen, M.J.; Dijkstra, C.D.; van der Ende, M.B.; Pena, A.S.; van Rees, E.P. Anti-CD11b/CD18 antibodies reduce inflammation in acute colitis in rats. Clin. Exp. Immunol. 1995, 101, 351–356. [Google Scholar] [CrossRef]
- Pavlick, K.P.; Ostanin, D.V.; Furr, K.L.; Laroux, F.S.; Brown, C.M.; Gray, L.; Kevil, C.G.; Grisham, M.B. Role of T-cell-associated lymphocyte function-associated antigen-1 in the pathogenesis of experimental colitis. Int. Immunol. 2006, 18, 389–398. [Google Scholar] [CrossRef]
- Takahashi, M.; Mimura, Y.; Hayashi, Y. Role of the ICAM-1/LFA-1 pathway during the development of autoimmune dacryoadenitis in an animal model for Sjogren’s syndrome. Pathobiol. J. Immunopathol. Mol. Cell. Biol. 1996, 64, 269–274. [Google Scholar] [CrossRef]
- Gupta, A.; Le, A.; Belinka, B.A.; Kachlany, S.C. In vitro synergism between LFA-1 targeting leukotoxin (Leukothera) and standard chemotherapeutic agents in leukemia cells. Leuk. Res. 2011, 35, 1498–1505. [Google Scholar] [CrossRef]
- Nestle, F.O.; Kaplan, D.H.; Barker, J. Psoriasis. N. Engl. J. Med. 2009, 361, 496–509. [Google Scholar] [CrossRef]
- Valdimarsson, H.; Thorleifsdottir, R.H.; Sigurdardottir, S.L.; Gudjonsson, J.E.; Johnston, A. Psoriasis—as an autoimmune disease caused by molecular mimicry. Trends Immunol. 2009, 30, 494–501. [Google Scholar] [CrossRef]
- Kunz, M. Current treatment of psoriasis with biologics. Curr. Drug Discov. Technol. 2009, 6, 231–240. [Google Scholar] [CrossRef]
- Poulin, Y.; Langley, R.G.; Teixeira, H.D.; Martel, M.J.; Cheung, S. Biologics in the treatment of psoriasis: Clinical and economic overview. J. Cutan. Med. Surg. 2009, 13 (Suppl. S2), S49–S57. [Google Scholar] [CrossRef]
- Stenderup, K.; Rosada, C.; Dam, T.N.; Salerno, E.; Belinka, B.A.; Kachlany, S.C. Resolution of psoriasis by a leukocyte-targeting bacterial protein in a humanized mouse model. J. Investig. Dermatol. 2011, 131, 2033–2039. [Google Scholar] [CrossRef]
- Wrone-Smith, T.; Nickoloff, B.J. Dermal injection of immunocytes induces psoriasis. J. Clin. Investig. 1996, 98, 1878–1887. [Google Scholar] [CrossRef]
- Chiu, Y.G.; Shao, T.; Feng, C.; Mensah, K.A.; Thullen, M.; Schwarz, E.M.; Ritchlin, C.T. CD16 (FcRgammaIII) as a potential marker of osteoclast precursors in psoriatic arthritis. Arthritis Res. Ther. 2010, 12, R14. [Google Scholar] [CrossRef]
- Kudo, M.; Ishigatsubo, Y.; Aoki, I. Pathology of asthma. Front. Microbiol. 2013, 4, 263. [Google Scholar] [CrossRef]
- Rizzuto, C.D.; Sodroski, J.G. Contribution of virion ICAM-1 to human immunodeficiency virus infectivity and sensitivity to neutralization. J. Virol. 1997, 71, 4847–4851. [Google Scholar]
- Fortin, J.F.; Cantin, R.; Lamontagne, G.; Tremblay, M. Host-derived ICAM-1 glycoproteins incorporated on human immunodeficiency virus type 1 are biologically active and enhance viral infectivity. J. Virol. 1997, 71, 3588–3596. [Google Scholar]
- Jolly, C.; Mitar, I.; Sattentau, Q.J. Adhesion molecule interactions facilitate human immunodeficiency virus type 1-induced virological synapse formation between T cells. J. Virol. 2007, 81, 13916–13921. [Google Scholar] [CrossRef]
- Jolly, C.; Kashefi, K.; Hollinshead, M.; Sattentau, Q.J. HIV-1 cell to cell transfer across an Env-induced, actin-dependent synapse. J. Exp. Med. 2004, 199, 283–293. [Google Scholar] [CrossRef]
- Hordinsky, M.K. Overview of alopecia areata. J. Investig. Dermatol. Symp. Process. 2013, 16, S13–S15. [Google Scholar] [CrossRef]
- Kachlany, S.C. Mechanisms of LtxA (Leukotoxin), a Potent New Anti-Inflammatory Agent for the Treatment of Alopecia Areata. J. Investig. Dermatol. Symp. Process. 2015, 17, 19–22. [Google Scholar] [CrossRef]
- Kiriakidou, M.; Cotton, D.; Taichman, D.; Williams, S. Systemic lupus erythematosus. Ann. Intern. Med. 2013, 159, ITC4-1. [Google Scholar] [CrossRef]
- Rahman, A.; Isenberg, D.A. Systemic lupus erythematosus. N. Engl. J. Med. 2008, 358, 929–939. [Google Scholar] [CrossRef]
- Lorenz, H.M.; Grunke, M.; Hieronymus, T.; Herrmann, M.; Kuhnel, A.; Manger, B.; Kalden, J.R. In vitro apoptosis and expression of apoptosis-related molecules in lymphocytes from patients with systemic lupus erythematosus and other autoimmune diseases. Arthritis Rheum. 1997, 40, 306–317. [Google Scholar] [CrossRef]
- Nakahara, J.; Maeda, M.; Aiso, S.; Suzuki, N. Current concepts in multiple sclerosis: Autoimmunity versus oligodendrogliopathy. Clin. Rev. Allergy Immunol. 2012, 42, 26–34. [Google Scholar] [CrossRef]
- Xavier, R.J.; Podolsky, D.K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 2007, 448, 427–434. [Google Scholar] [CrossRef]
- Baumgart, D.C.; Carding, S.R. Inflammatory bowel disease: Cause and immunobiology. Lancet (Lond. Engl.) 2007, 369, 1627–1640. [Google Scholar] [CrossRef]
- Baumgart, D.C.; Sandborn, W.J. Inflammatory bowel disease: Clinical aspects and established and evolving therapies. Lancet (Lond. Engl.) 2007, 369, 1641–1657. [Google Scholar] [CrossRef]
- Messmer, E.M. The pathophysiology, diagnosis, and treatment of dry eye disease. Dtsch. Arztebl. Int. 2015, 112, 71–81, quiz 82. [Google Scholar] [CrossRef]
- Stern, M.E.; Schaumburg, C.S.; Dana, R.; Calonge, M.; Niederkorn, J.Y.; Pflugfelder, S.C. Autoimmunity at the ocular surface: Pathogenesis and regulation. Mucosal Immunol. 2010, 3, 425–442. [Google Scholar] [CrossRef]
- Stern, M.E.; Schaumburg, C.S.; Pflugfelder, S.C. Dry eye as a mucosal autoimmune disease. Int. Rev. Immunol. 2013, 32, 19–41. [Google Scholar] [CrossRef]
- Perez, V.L.; Pflugfelder, S.C.; Zhang, S.; Shojaei, A.; Haque, R. Lifitegrast, a novel integrin antagonist for treatment of dry eye disease. Ocul. Surf. 2016, 14, 207–215. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; Stern, M.; Zhang, S.; Shojaei, A. LFA-1/ICAM-1 Interaction as a Therapeutic Target in Dry Eye Disease. J. Ocul. Pharmacol. Ther. Off. J. Assoc. Ocul. Pharmacol. Ther. 2017, 33, 5–12. [Google Scholar] [CrossRef]
- Hogg, N.; Laschinger, M.; Giles, K.; McDowall, A. T-cell integrins: More than just sticking points. J. Cell Sci. 2003, 116, 4695–4705. [Google Scholar] [CrossRef]
- Smith, A.; Stanley, P.; Jones, K.; Svensson, L.; McDowall, A.; Hogg, N. The role of the integrin LFA-1 in T-lymphocyte migration. Immunol. Rev. 2007, 218, 135–146. [Google Scholar] [CrossRef]
- Tauber, J.; Karpecki, P.; Latkany, R.; Luchs, J.; Martel, J.; Sall, K.; Raychaudhuri, A.; Smith, V.; Semba, C.P. Lifitegrast Ophthalmic Solution 5.0% versus Placebo for Treatment of Dry Eye Disease: Results of the Randomized Phase III OPUS-2 Study. Ophthalmology 2015, 122, 2423–2431. [Google Scholar] [CrossRef]
- Murphy, C.J.; Bentley, E.; Miller, P.E.; McIntyre, K.; Leatherberry, G.; Dubielzig, R.; Giuliano, E.; Moore, C.P.; Phillips, T.E.; Smith, P.B.; et al. The pharmacologic assessment of a novel lymphocyte function-associated antigen-1 antagonist (SAR 1118) for the treatment of keratoconjunctivitis sicca in dogs. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3174–3180. [Google Scholar] [CrossRef]


| Indication | Integrins | References |
|---|---|---|
| Leukemia | αLβ2 | [107,109,110] |
| Lymphoma | αLβ2 | [107,109,110] |
| Psoriasis | αLβ2, αMβ2, αXβ2 | [111,112] |
| Asthma | αLβ2 | [113,114] |
| HIV | αLβ2 | [65,115,116,117] |
| Alopecia Areata | αLβ2, | [118,119,120,121,122] |
| Systemic Lupus Erythematosus | αLβ2, αMβ2 | [123,124,125,126] |
| Multiple Sclerosis | β2 | [127,128] |
| Crohn’s Disease | αLβ2, αMβ2 | [129,130,131,132] |
| Ulcerative Colitis | αLβ2, αMβ2 | [129,130,131,132] |
| Dry Eye | αLβ2, | [133] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vega, B.A.; Belinka Jr., B.A.; Kachlany, S.C. Aggregatibacter actinomycetemcomitans Leukotoxin (LtxA; Leukothera®): Mechanisms of Action and Therapeutic Applications. Toxins 2019, 11, 489. https://doi.org/10.3390/toxins11090489
Vega BA, Belinka Jr. BA, Kachlany SC. Aggregatibacter actinomycetemcomitans Leukotoxin (LtxA; Leukothera®): Mechanisms of Action and Therapeutic Applications. Toxins. 2019; 11(9):489. https://doi.org/10.3390/toxins11090489
Chicago/Turabian StyleVega, Brian A., Benjamin A. Belinka Jr., and Scott C. Kachlany. 2019. "Aggregatibacter actinomycetemcomitans Leukotoxin (LtxA; Leukothera®): Mechanisms of Action and Therapeutic Applications" Toxins 11, no. 9: 489. https://doi.org/10.3390/toxins11090489
APA StyleVega, B. A., Belinka Jr., B. A., & Kachlany, S. C. (2019). Aggregatibacter actinomycetemcomitans Leukotoxin (LtxA; Leukothera®): Mechanisms of Action and Therapeutic Applications. Toxins, 11(9), 489. https://doi.org/10.3390/toxins11090489




