Aflatoxin in Chili Peppers in Nigeria: Extent of Contamination and Control Using Atoxigenic Aspergillus flavus Genotypes as Biocontrol Agents
Abstract
:1. Introduction
2. Results
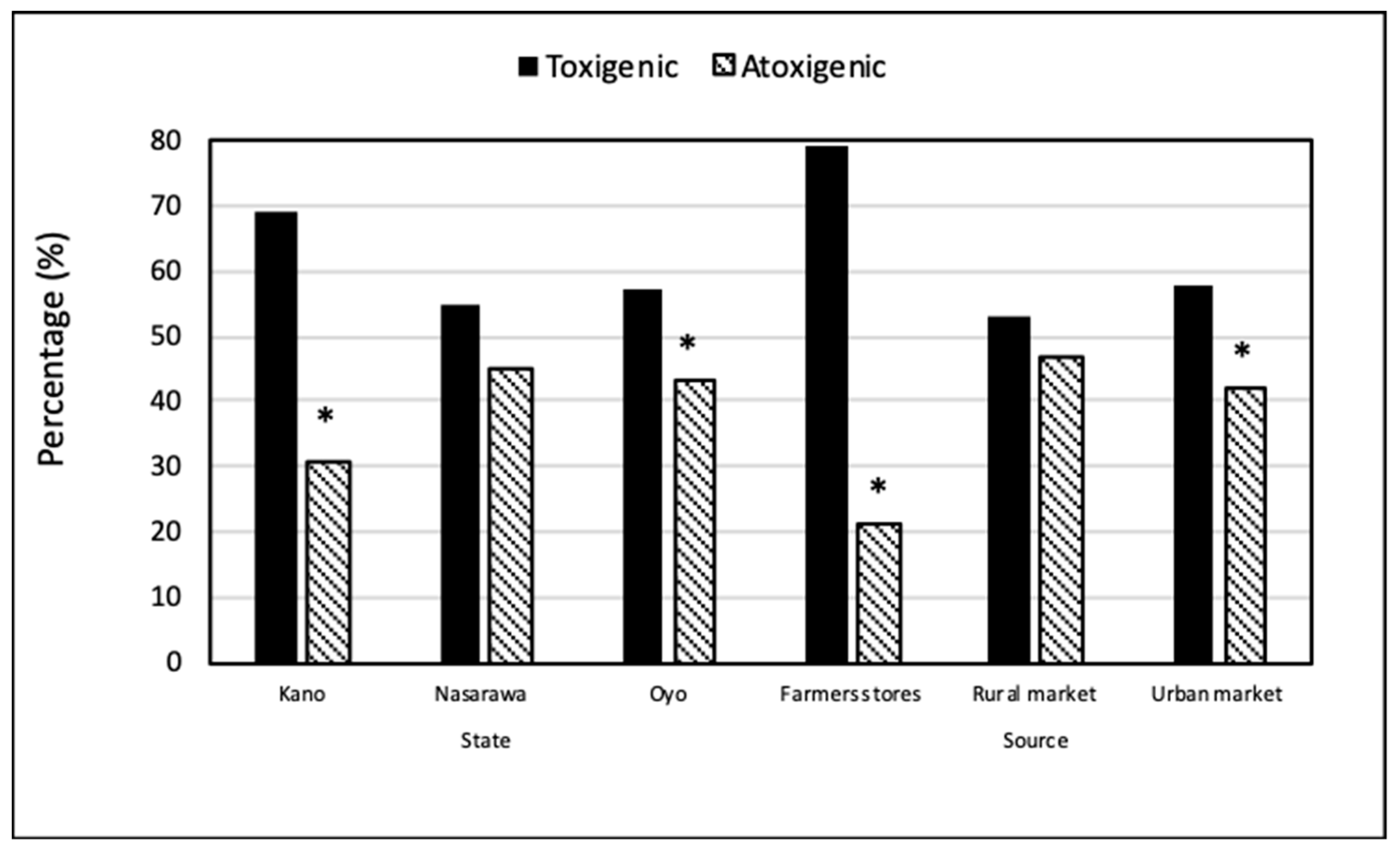
2.1. Aflatoxin Contamination of Chili Pepper from Farmers’ Stores and Markets
2.2. Incidence and Densities of Aspergillus Section Flavi
2.3. Aflatoxin-Producing Ability of the Recovered Fungi
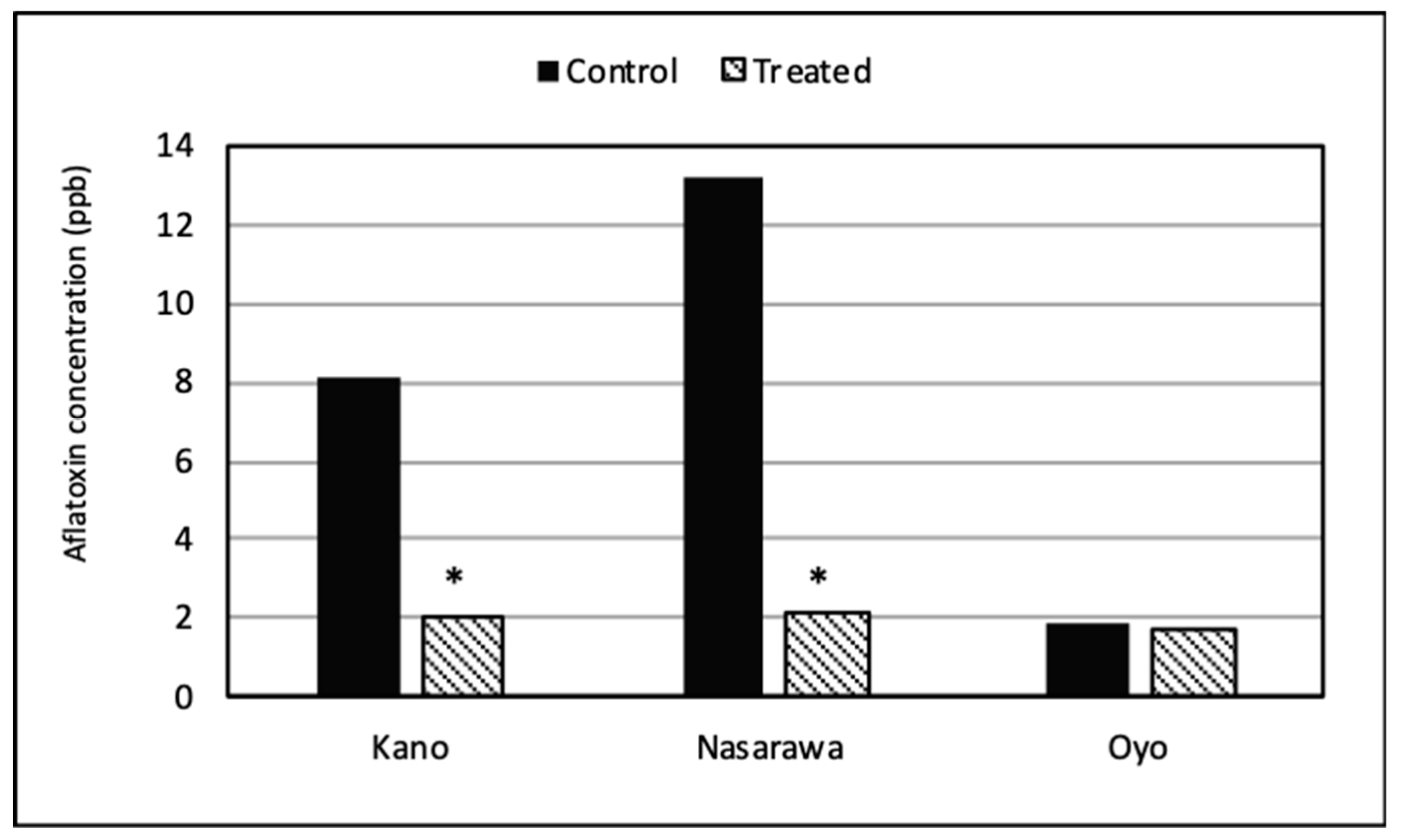
2.4. Aflatoxin Biocontrol in Chili Peppers Using the Product Aflasafe
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Collection of Chili Samples for Baseline Study
5.2. Aspergillus Associated with the Chili Peppers
5.3. Aflatoxin Analysis of Chili Samples
5.4. Aflatoxin-Production Ability of the Recovered Fungi
5.5. Preparation and Quality Control of Aflasafe
5.6. Field Plots and Aflasafe Application
5.7. Soil and Crop Sampling from Field Plots
5.8. Aspergillus Species Associated with Aflasafe-Treated and Untreated Chili Peppers
5.9. Aflatoxin Analyses of Aflasafe-Treated and Control Chili Pepper
5.10. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spivey, D.M. The Peppers, Cracklings, and Knots of Wool Cookbook: The Global Migration of African Cuisine; SUNY Press: Albany, NY, USA, 1999. [Google Scholar]
- Tapsell, L.; Hemphill, I.; Cobiac, L.; Sullivan, D.; Fenech, M.; Patch, C.; Roodenrys, S.; Keogh, J.; Clifton, P.; Williams, P.; et al. Health benefits of herbs and spices: The past, the present, the future. Med. J. Aust. 2006, 185, S7–S9. [Google Scholar]
- McCarty, M.; DiNicolantonio, J.; O’Keefe, J. Capsaicin may have important potential for promoting vascular and metabolic health. Open Heart 2015, 2, e000262. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Cotty, P.J. Aflatoxin contamination of dried red chilies: Contrasts between the United States and Nigeria, two markets differing in regulation enforcement. Food Control 2017, 80, 374–379. [Google Scholar] [CrossRef]
- Cho, S.H.; Lee, C.H.; Jang, M.R.; Son, Y.W.; Lee, S.M.; Choi, I.S.; Kim, S.H.; Kim, D.B. Aflatoxins contamination in spices and processed spice products commercialized in Korea. Food Chem. 2008, 107, 1283–1288. [Google Scholar] [CrossRef]
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- Williams, J.H.; Phillips, T.D.; Jolly, P.E.; Stiles, J.K.; Jolly, C.M.; Aggarwal, D. Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences, and interventions. Am. J. Clin. Nutr. 2004, 80, 1106–1122. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.Y.; Turner, P.C.; Hall, A.J.; Wild, C.P. Aflatoxin exposure and impaired child growth in West Africa: An unexplored international public health burden. In Mycotoxins: Detection Methods, Management, Public Health and Agricultural Trade; CABI: Wallingford, UK, 2008; pp. 53–66. [Google Scholar]
- Kamala, A.; Shirima, C.; Jani, B.; Bakari, M.; Sillo, H.; Rusibamayila, N.; De Saeger, S.; Kimanya, M.; Gong, Y.Y.; Simba, A. Outbreak of an acute aflatoxicosis in Tanzania during 2016. World Mycotoxin J. 2018, 11, 311–320. [Google Scholar] [CrossRef]
- Probst, C.; Callicott, K.A.; Cotty, P.J. Deadly strains of Kenyan Aspergillus are distinct from other aflatoxin producers. Eur. J. Plant Pathol. 2012, 132, 419–429. [Google Scholar] [CrossRef]
- Cotty, P.J.; Probst, C.; Jaime-Garcia, R. Etiology and management of aflatoxin contamination. In Mycotoxins: Detection Methods, Management, Public Health and Agricultural Trade; CABI: Wallingford, UK, 2008; pp. 287–299. ISBN 9781845930820. [Google Scholar]
- Wu, F. Global impacts of aflatoxin in maize: Trade and human health. World Mycotoxin J. 2015, 8, 137–142. [Google Scholar] [CrossRef]
- Logrieco, A.F.; Miller, J.D.; Eskola, M.; Krska, R.; Ayalew, A.; Bandyopadhyay, R.; Battilani, P.; Bhatnagar, D.; Chulze, S.; De Saeger, S.; et al. The Mycotox Charter: Increasing awareness of, and concerted action for, minimizing mycotoxin exposure worldwide. Toxins 2018, 10. [Google Scholar] [CrossRef]
- Udomkun, P.; Wiredu, A.N.; Nagle, M.; Bandyopadhyay, R.; Müller, J.; Vanlauwe, B. Mycotoxins in Sub-Saharan Africa: Present situation, socio-economic impact, awareness, and outlook. Food Control 2017, 72, 110–122. [Google Scholar] [CrossRef]
- The Rapid Alert System for Food and Feed (RASFF)-Food and Feed Safety Alerts. Available online: https://ec.europa.eu/food/safety/rasff_en (accessed on 5 June 2019).
- Yewondwossen, M. EU Rejects Ethiopian Red Pepper for Unsafe Levels of Toxins. Available online: https://health-ethiopianism.blogspot.com/2017/05/eu-rejects-ethiopian-red-pepper-for.html (accessed on 5 June 2019).
- Bandyopadhyay, R.; Ortega-Beltran, A.; Akande, A.; Mutegi, C.; Atehnkeng, J.; Kaptoge, L.; Senghor, L.A.; Adhikari, B.N.; Cotty, P.J. Biological control of aflatoxins in Africa: Current status and potential challenges in the face of climate change. World Mycotoxin J. 2016, 9, 771–789. [Google Scholar] [CrossRef]
- Njoroge, S.M.C. A critical review of aflatoxin contamination of peanuts in Malawi and Zambia: The past, present, and future. Plant Dis. 2018, 102, 2394–2406. [Google Scholar] [CrossRef] [PubMed]
- Ayalew, A.; Kimanya, M.; Matumba, L.; Bandyopadhyay, R.; Menkir, A.; Cotty, P.J. Controlling aflatoxins in maize in Africa: Strategies, challenges and opportunities for improvement. In Achieving Sustainable Cultivation of Maize. Volume 2: Cultivation Techniques, Pest and Disease Control; Watson, D., Ed.; Burleigh Doods Science Publishing: Cambridge, UK, 2017; pp. 1–24. [Google Scholar]
- Amaike, S.; Keller, N.P. Aspergillus flavus. Annu. Rev. Phytopathol. 2011, 49, 107–133. [Google Scholar] [CrossRef] [PubMed]
- Klich, M.A. Aspergillus flavus: The major producer of aflatoxin. Mol. Plant Pathol. 2007, 8, 713–722. [Google Scholar] [CrossRef]
- Cotty, P.J. Virulence and cultural characteristics of two Aspergillus flavus strains pathogenic on cotton. Phytopathology 1989, 79, 808–814. [Google Scholar] [CrossRef]
- Donner, M.; Atehnkeng, J.; Sikora, R.A.; Bandyopadhyay, R.; Cotty, P.J. Distribution of Aspergillus section Flavi in soils of maize fields in three agroecological zones of Nigeria. Soil Biol. Biochem. 2009, 41, 37–44. [Google Scholar] [CrossRef]
- Diedhiou, P.M.; Bandyopadhyay, R.; Atehnkeng, J.; Ojiambo, P.S. Aspergillus colonization and aflatoxin contamination of maize and sesame kernels in two agro-ecological zones in Senegal. J. Phytopathol. 2011, 159, 268–275. [Google Scholar] [CrossRef]
- Atehnkeng, J.; Ojiambo, P.S.; Donner, M.; Ikotun, B.; Sikora, R.A.; Cotty, P.J.; Bandyopadhyay, R. Distribution and toxigenicity of Aspergillus species isolated from maize kernels from three agro-ecological zones in Nigeria. Int. J. Food Microbiol. 2008, 122, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Agbetiameh, D.; Ortega-Beltran, A.; Awuah, R.T.; Atehnkeng, J.; Cotty, P.J.; Bandyopadhyay, R. Prevalence of aflatoxin contamination in maize and groundnut in Ghana: Population structure, distribution, and toxigenicity of the causal agents. Plant Dis. 2018, 102, 764–772. [Google Scholar] [CrossRef]
- Singh, P.; Cotty, P.J. Characterization of Aspergilli from dried red chilies (Capsicum spp.): Insights into the etiology of aflatoxin contamination. Int. J. Food Microbiol. 2019, 289, 145–153. [Google Scholar] [CrossRef]
- Cotty, P.J.; Cardwell, K.F. Divergence of West African and North American communities of Aspergillus section Flavi. Appl. Environ. Microbiol. 1999, 65, 2264–2266. [Google Scholar]
- Frisvad, J.C.; Hubka, V.; Ezekiel, C.N.; Hong, S.-B.; Novakova, A.; Chen, A.J.; Arzanlou, M.; Larsen, T.O.; Sklenar, F.; Mahakarnchanakul, W.; et al. Taxonomy of Aspergillus section Flavi and their production of aflatoxins, ochratoxins and other mycotoxins. Stud. Mycol. 2019, 93, 1–63. [Google Scholar] [CrossRef]
- Probst, C.; Bandyopadhyay, R.; Cotty, P.J. Diversity of aflatoxin-producing fungi and their impact on food safety in sub-Saharan Africa. Int. J. Food Microbiol. 2014, 174, 113–122. [Google Scholar] [CrossRef]
- Cardwell, K.F.; Cotty, P.J. Distribution of Aspergillus section Flavi among field soils from the four agroecological zones of the Republic of Benin, West Africa. Plant Dis. 2002, 79, 1039–1045. [Google Scholar]
- Mehl, H.L.; Jaime, R.; Callicott, K.A.; Probst, C.; Garber, N.P.; Ortega-Beltran, A.; Grubisha, L.C.; Cotty, P.J. Aspergillus flavus diversity on crops and in the environment can be exploited to reduce aflatoxin exposure and improve health. Ann. N. Y. Acad. Sci. 2012, 1273, 7–17. [Google Scholar] [CrossRef]
- Cotty, P.J. Biocompetitive exclusion of toxigenic fungi. In The Mycotoxin Factbook; Barug, D., Bhatnagar, D., van Egdmond, H.P., van der Kamp, J.W., van Osenbruggen, W.A., Visconti, A., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2006; pp. 179–197. [Google Scholar]
- Doster, M.A.; Cotty, P.J.; Michailides, T.J. Evaluation of the atoxigenic Aspergillus flavus strain AF36 in pistachio orchards. Plant Dis. 2014, 98, 948–956. [Google Scholar] [CrossRef]
- Ortega-Beltran, A.; Moral, J.; Puckett, R.D.; Morgan, D.P.; Cotty, P.J.; Michailides, T.J. Fungal communities associated with almond throughout crop development: Implications for aflatoxin biocontrol management in California. PLoS ONE 2018, 13, e0199127. [Google Scholar] [CrossRef]
- Dorner, J.W. Biological control of aflatoxin contamination of crops. J. Toxicol. Toxin Rev. 2004, 23, 425–450. [Google Scholar] [CrossRef]
- Schreurs, F.; Bandyopadhyay, R.; Kooyman, C.; Ortega-Beltran, A.; Akande, A.; Konlambigue, M.; Kaptoge, L.; Van den Bosch, N. Commercial products promoting plant health in African agriculture. In Critical Issues in Plant Health: 50 Years of Research in African Agriculture; Burleigh Dodds Science Publishing: Cambridge, UK, 2019; ISBN 9781786762320. [Google Scholar]
- Food and Agricultural Organization of the United Nations (FAO). FAOSTAT. Available online: http://faostat.fao.org (accessed on 5 February 2019).
- Makun, H.; Dutton, M.; Njobeh, P.; Gbodi, T.; Ogbadu, G. Aflatoxin contamination in foods and feeds: A special focus on Africa. In Trends in Vital Food and Control Engineering; IntechOpen: London, UK, 2015; pp. 187–234. ISBN 9789537619992. [Google Scholar]
- Atehnkeng, J.; Donner, M.; Ojiambo, P.S.; Ikotun, B.; Augusto, J.; Cotty, P.J.; Bandyopadhyay, R. Environmental distribution and genetic diversity of vegetative compatibility groups determine biocontrol strategies to mitigate aflatoxin contamination of maize by Aspergillus flavus. Microb. Biotechnol. 2016, 9, 75–88. [Google Scholar] [CrossRef]
- Atehnkeng, J.; Ojiambo, P.S.; Cotty, P.J.; Bandyopadhyay, R. Field efficacy of a mixture of atoxigenic Aspergillus flavus Link: FR vegetative compatibility groups in preventing aflatoxin contamination in maize (Zea mays L.). Biol. Control 2014, 72, 62–70. [Google Scholar] [CrossRef]
- Cotty, P.J.; Antilla, L.; Wakelyn, P.J. Competitive exclusion of aflatoxin producers: Farmer-driven research and development. In Biological Control: A Global Perspective; CABI: Wallingford, UK, 2007; pp. 241–253. ISBN 9781845932657. [Google Scholar]
- Ortega-Beltran, A.; Moral, J.; Picot, A.; Puckett, R.D.; Cotty, P.J.; Michailides, T.J. Atoxigenic Aspergillus flavus isolates endemic to almond, fig, and pistachio orchards in California with potential to reduce aflatoxin contamination in these crops. Plant Dis. 2019, 103, 905–912. [Google Scholar] [CrossRef]
- Atehnkeng, J.; Ojiambo, P.S.; Ikotun, T.; Sikora, R.A.; Cotty, P.J.; Bandyopadhyay, R. Evaluation of atoxigenic isolates of Aspergillus flavus as potential biocontrol agents for aflatoxin in maize. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2008, 25, 1264–1271. [Google Scholar] [CrossRef]
- Cotty, P.J. Comparison of four media for the isolation of Aspergillus flavus group fungi. Mycopathologia 1994, 125, 157–162. [Google Scholar] [CrossRef]
- Klich, M.A.; Pitt, J.I. Differentiation of Aspergillus flavus from A. parasiticus and other closely related species. Trans. Br. Mycol. Soc. 1988, 91, 99–108. [Google Scholar] [CrossRef]
- Probst, C.; Bandyopadhyay, R.; Price, L.E.; Cotty, P.J. Identification of atoxigenic Aspergillus flavus isolates to reduce aflatoxin contamination of maize in Kenya. Plant Dis. 2011, 95, 212–218. [Google Scholar] [CrossRef]
- Al-Hadithi, N.; Kössler, P.; Karlovsky, P. Determination of ochratoxin A in wheat and maize by solid bar microextraction with liquid chromatography and fluorescence detection. Toxins 2015, 7, 3000–3011. [Google Scholar] [CrossRef]
- Iqbal, S.; Paterson, R.; Bhatti, I.; Asi, M.; Sheikh, M.; Bhatti, H. Aflatoxin B1 in chilies from the Punjab region, Pakistan. Mycotoxin Res. 2010, 26, 205–209. [Google Scholar] [CrossRef]
- Reiter, E.; Zentek, J.; Razzazi, E. Review on sample preparation strategies and methods used for the analysis of aflatoxins in food and feed. Mol. Nutr. Food Res. 2009, 53, 508–524. [Google Scholar] [CrossRef]
- Akiyamaa, H.; Godaa, Y.; Tanakab, T.; Toyodaa, M. Determination of aflatoxins B1, B2, G1 and G2 in spices using a multifunctional column clean-up. J. Chromatogr. A 2001, 93, 153–157. [Google Scholar] [CrossRef]
- Cotty, P.J. Aflatoxin-producing potential of communities of Aspergillus section Flavi from cotton producing areas in the United States. Mycol. Res. 1997, 11, 698–704. [Google Scholar] [CrossRef]
| Sample Source | States | ||
|---|---|---|---|
| Kano | Nasarawa | Oyo | |
| Farmer store | 10 | 4 | 0 |
| Rural markets | 8 | 14 | 13 |
| Urban markets | 5 | 5 | 11 |
| Total | 23 | 23 | 24 |
| Origin | N a | Aflatoxin Levels (ppb) with Respect to EU Limits | ||||
|---|---|---|---|---|---|---|
| % b | Total Aflatoxin Range | Mean ± SE c | Aflatoxin B1 >5 ppb (%) | Total Aflatoxin >10 ppb (%) | ||
| State | ||||||
| Kano | 23 | 70 | 0–16 | 5.1 ± 1.0 b | 0 (0.0) | 3 (13.0) |
| Nasarawa | 23 | 48 | 0–59 | 6.0 ± 2.8 b | 2 (8.7) | 3 (13.0) |
| Oyo | 24 | 88 | 0–97 | 15.2 ± 4.6 a | 7 (29.2) | 11 (45.8) |
| Source | ||||||
| Farmers’ store | 14 | 71 | 0–18 | 6.4 ± 1.7 a | 0 (0.0) | 3 (21.4) |
| Rural market | 35 | 66 | 0–72 | 8.3 ± 2.6 a | 6 (17.1) | 7 (20.0) |
| Urban market | 21 | 71 | 0–97 | 11.3 ± 4.6 a | 3 (14.3) | 7 (33.3) |
| Origin and Source | N a | Recovered Isolates | Proportion of Species (%) b | CFU/g | ||
|---|---|---|---|---|---|---|
| A. flavus L morphotype | SBG Strains | A. tamarii | ||||
| State | ||||||
| Kano | 23 | 440 | 85.7 ± 1.3 aA | 13.6 ± 1.3 aB | 0.7 ± 0.1 aB | 12,183 b |
| Nasarawa | 23 | 460 | 88.7 ± 1.1 aA | 9.1 ± 1.1 aB | 2.2 ± 0.3 aB | 27,100 ab |
| Oyo | 24 | 480 | 87.9 ± 1.1 aA | 1.9 ± 0.3 aB | 10.2 ± 1.1 aB | 73,012 a |
| Source | ||||||
| Farmers’ store | 14 | 280 | 78.6 ± 2.0 aA | 20.0 ± 2.0 aB | 1.4 ± 0.1 aB | 10,293 b |
| Rural market | 35 | 700 | 86.6 ± 1.0 aA | 6.4 ± 0.7 abB | 7.0 ± 0.7 aB | 31,446 ab |
| Urban market | 21 | 400 | 95.3 ± 0.4 aA | 2.5 ± 0.4 bB | 2.3 ± 0.2 aB | 67,195 a |
| Substrate | Treatment | N a | Proportion of Species (%) b | CFU/g b | ||
|---|---|---|---|---|---|---|
| A. flavus L morphotype | SBG Strains | A. tamarii | ||||
| Kano | ||||||
| Soil before application | Aflasafe | 10 | 96.6 ± 0.0 ns | 1.7 ± 0.1 ns | 1.7 ± 0.1 ns | 99 ns |
| Control | 20 | 99.1 ± 0.5 | 0.6 ± 0.4 | 0.3 ± 0.3 | 143 | |
| Chili pepper at harvest | Aflasafe | 10 | 80.0 ± 9.8 ns | 13.8 ± 8.8 ns | 6.1 ± 3.8 ns | 4,940,181 ns |
| Control | 20 | 83.8 ± 6.0 | 16.3 ± 6.0 | 0.0 | 97,755 | |
| Soil 6 mo. after application | Aflasafe | 10 | 77.9 ± 0.3 ns | 20.5 ± 1.5 ns | 1.6 ± 0.3 ns | 405 ns |
| Control | 20 | 66.3 ± 0.9 | 30.0 ± 1.2 | 3.7 ± 0.5 | 382 | |
| Nasarawa | ||||||
| Soil before application | Aflasafe | 7 | 92.2 ± 0.0 ns | 1.7 ± 0.2 ns | 6.2 ± 0.9 ns | 167 ns |
| Control | 14 | 93.8 ± 0.0 | 3.9 ± 0.4 | 2.3 ± 0.2 | 80 | |
| Chili pepper at harvest | Aflasafe | 7 | 99.2 ± 0.8 ns | 0.0 ns | 0.8 ± 0.8 ns | 626,086 ns |
| Control | 14 | 99.6 ± 0.4 | 0.0 | 0.4 ± 0.4 | 923,825 | |
| Soil 6 mo. after application | Aflasafe | 7 | 76.1 ± 0.0 * | 21.0 ± 1.7 * | 2.9 ± 0.3 ns | 379 ns |
| Control | 14 | 55.2 ± 1.2 | 40.6 ± 1.8 | 4.2 ± 0.4 | 659 | |
| Oyo | ||||||
| Soil before application | Aflasafe | 9 | 98.8 ± 0.0 ns | 0.0 ns | 3.7 ± 0.2 ns | 377 ns |
| Control | 18 | 94.5 ± 0.0 | 4.5 ± 0.7 | 0.9 ± 0.1 | 159 | |
| Chili pepper at harvest | Aflasafe | 9 | 96.1 ± 2.8 ns | 2.7 ± 2.1 ns | 1.0 ± 0.8 ns | 45,256 ns |
| Control | 18 | 97.4 ± 1.4 | 2.0 ± 1.2 | 0.6 ± 0.4 | 848,469 | |
| Soil 6 mo. after application | Aflasafe | 9 | 84.9 ± 0.0 * | 14.1 ± 1.6 * | 0.4 ± 0.1 ns | 794 ns |
| Control | 18 | 65.6 ± 1.3 | 22.6 ± 2.1 | 0.8 ± 0.3 | 671 | |
| State | Mean Incidence of Aflasafe AAVs (%) a | |||||
|---|---|---|---|---|---|---|
| Soil before Application | Chili Pepper at Harvest | Soil 6 Months after Application | ||||
| Control | Treated | Control | Treated | Control | Treated | |
| Kano | 10.4 ± 2.6 | 7.1 ± 2.5 ns | 6.9 ± 3.7 | 25.0 ± 8.1 * | 7.2 ± 2.4 | 67.0 ± 5.4 * |
| Nasarawa | 8.3 ± 1.8 | 8.8 ± 2.5 ns | 7.6 ± 4.3 | 17.5 ± 6.1 * | 4.5 ± 2.7 | 64.6 ± 2.6 * |
| Oyo | 10.4 ± 2.8 | 3.1 ± 1.7 * | 9.4 ± 4.0 | 8.9 ± 1.3 ns | 4.9 ± 1.9 | 55.4 ± 4.2 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ezekiel, C.N.; Ortega-Beltran, A.; Oyedeji, E.O.; Atehnkeng, J.; Kössler, P.; Tairu, F.; Hoeschle-Zeledon, I.; Karlovsky, P.; Cotty, P.J.; Bandyopadhyay, R. Aflatoxin in Chili Peppers in Nigeria: Extent of Contamination and Control Using Atoxigenic Aspergillus flavus Genotypes as Biocontrol Agents. Toxins 2019, 11, 429. https://doi.org/10.3390/toxins11070429
Ezekiel CN, Ortega-Beltran A, Oyedeji EO, Atehnkeng J, Kössler P, Tairu F, Hoeschle-Zeledon I, Karlovsky P, Cotty PJ, Bandyopadhyay R. Aflatoxin in Chili Peppers in Nigeria: Extent of Contamination and Control Using Atoxigenic Aspergillus flavus Genotypes as Biocontrol Agents. Toxins. 2019; 11(7):429. https://doi.org/10.3390/toxins11070429
Chicago/Turabian StyleEzekiel, Chibundu N., Alejandro Ortega-Beltran, Eniola O. Oyedeji, Joseph Atehnkeng, Philip Kössler, Folasade Tairu, Irmgard Hoeschle-Zeledon, Petr Karlovsky, Peter J. Cotty, and Ranajit Bandyopadhyay. 2019. "Aflatoxin in Chili Peppers in Nigeria: Extent of Contamination and Control Using Atoxigenic Aspergillus flavus Genotypes as Biocontrol Agents" Toxins 11, no. 7: 429. https://doi.org/10.3390/toxins11070429
APA StyleEzekiel, C. N., Ortega-Beltran, A., Oyedeji, E. O., Atehnkeng, J., Kössler, P., Tairu, F., Hoeschle-Zeledon, I., Karlovsky, P., Cotty, P. J., & Bandyopadhyay, R. (2019). Aflatoxin in Chili Peppers in Nigeria: Extent of Contamination and Control Using Atoxigenic Aspergillus flavus Genotypes as Biocontrol Agents. Toxins, 11(7), 429. https://doi.org/10.3390/toxins11070429








