The ADP-Ribosylating Toxins of Salmonella
Abstract
1. Introduction
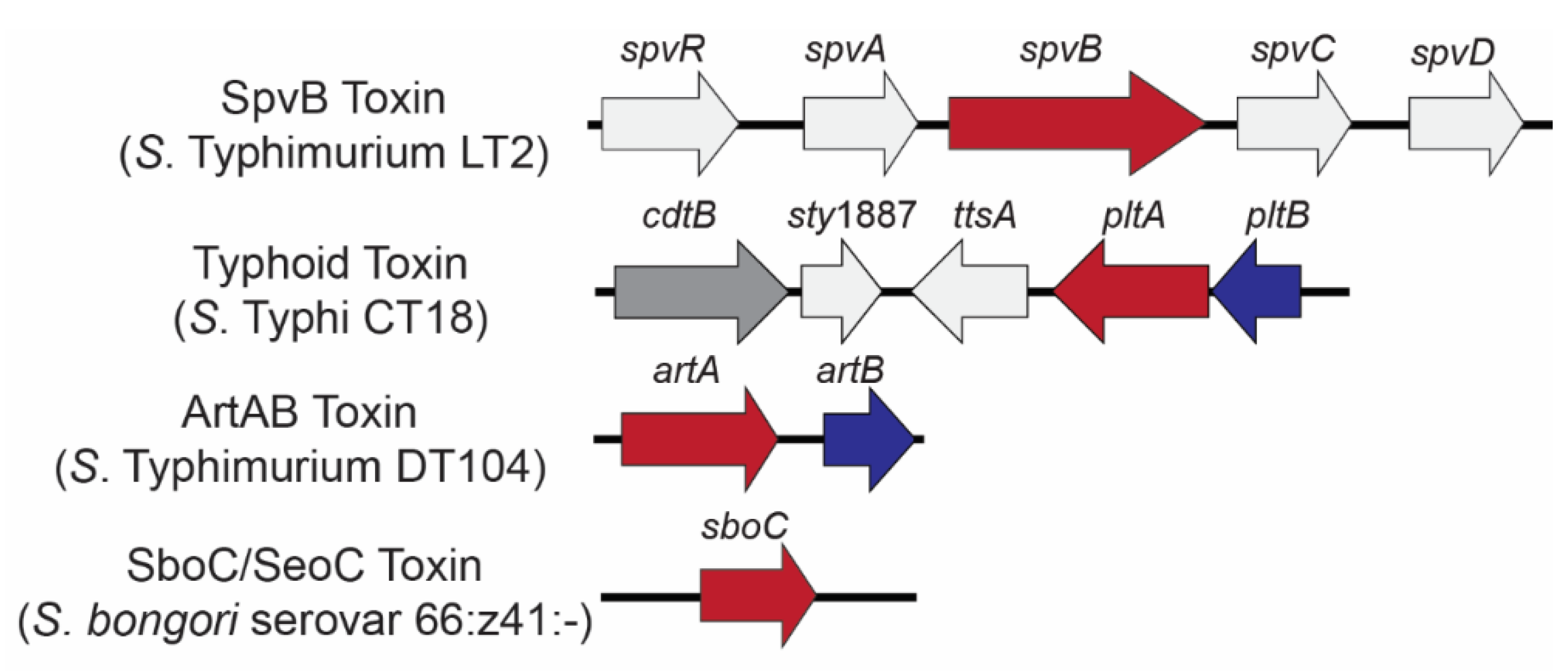
2. SpvB Toxin
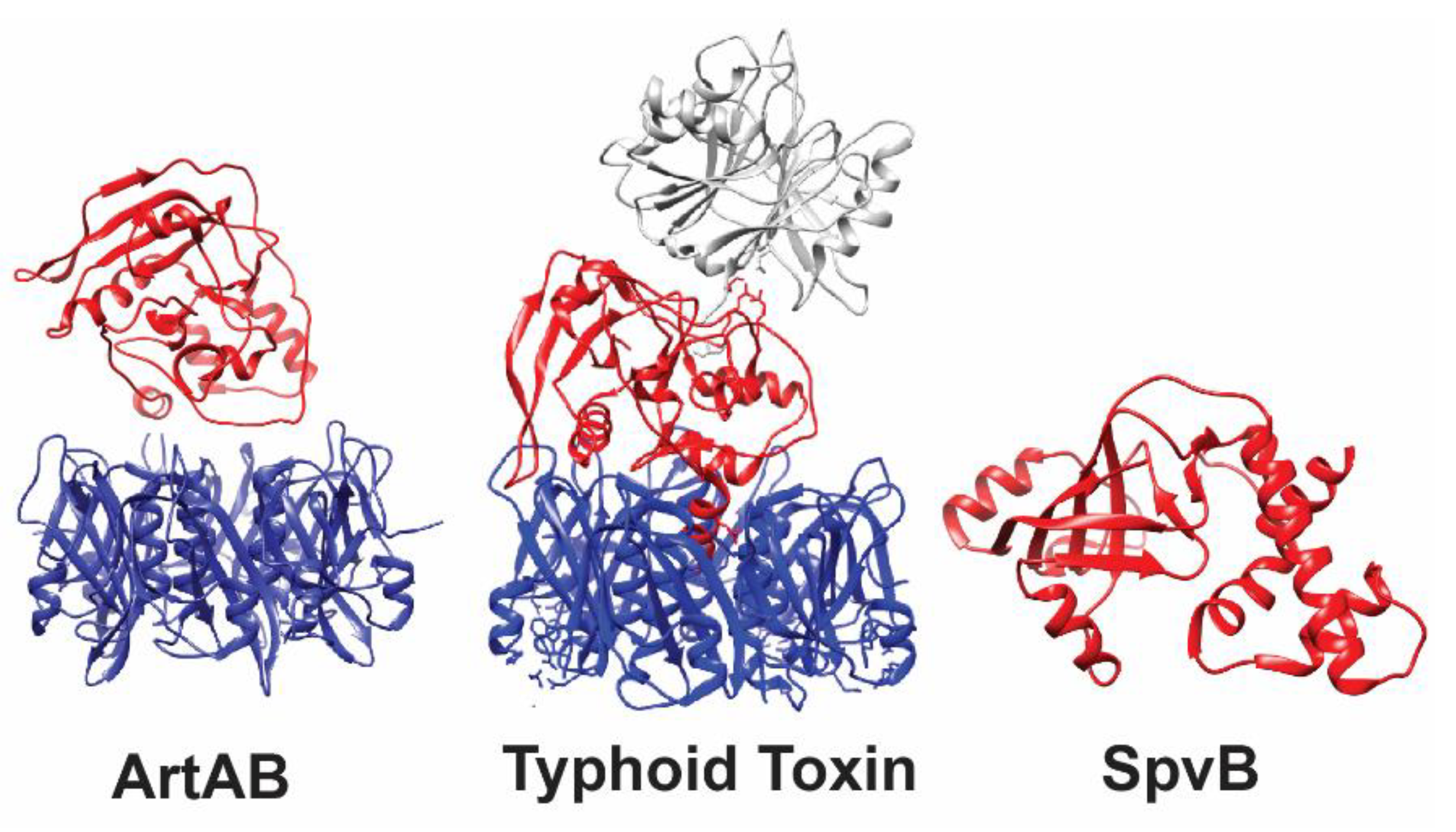
2.1. SpvB Toxin Structure and Activity
2.2. Implications of SpvB-Mediated Salmonella Virulence
3. The Typhoid Toxin
3.1. Typhoid Toxin Structure and ADP-Ribosyltransferase Activity
3.2. Implications of PltA-Mediated Salmonella Virulence
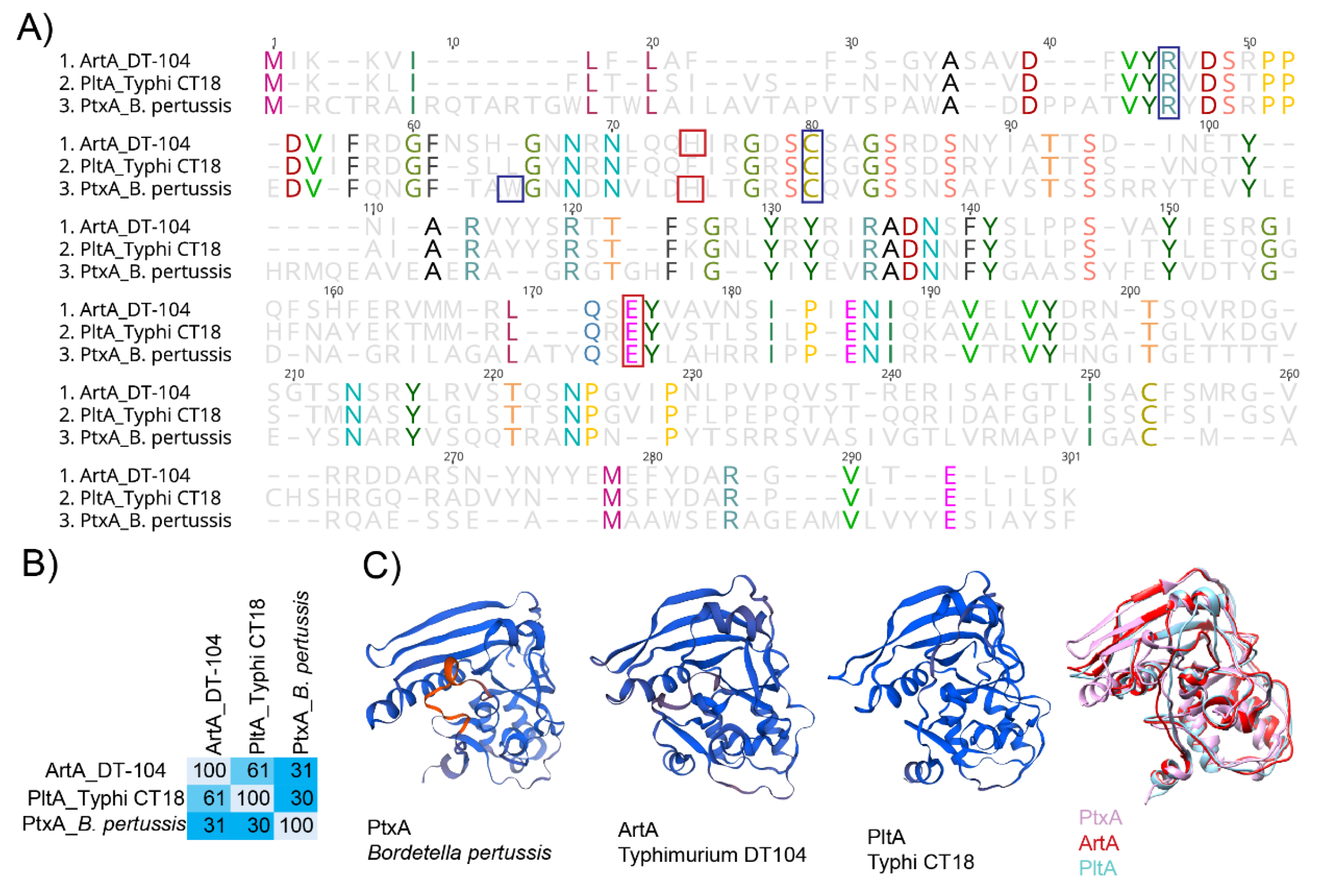
4. ArtAB Toxin
4.1. ArtAB Toxin’s ADP-Ribosyltransferase Activity and Structure
4.2. Implications of ArtAB-Mediated Salmonella Virulence
5. SboC/SeoC Toxin
5.1. SboC/SeoC Toxin’s ADP-Ribosyltransferase Activity and Structure
5.2. Implications of SboC/SeoC-Mediated Salmonella Virulence
6. Taking What We Know About Salmonella bARTTs and Moving the Field Forward
7. Conclusions
Funding
Conflicts of Interest
References
- Brenner, F.; Villar, R.; Angulo, F.; Tauxe, R.; Swaminathan, B. Salmonella nomenclature. J. Clin. Microbiol. 2000, 38, 2465–2467. [Google Scholar] [PubMed]
- Issenhuth-Jeanjean, S.; Roggentin, P.; Mikoleit, M.; Guibourdenche, M.; de Pinna, E.; Nair, S.; Fields, P.I.; Weill, F.-X. Supplement 2008–2010 (no. 48) to the White–Kauffmann–Le Minor scheme. Res. Microbiol. 2014, 165, 526–530. [Google Scholar] [CrossRef] [PubMed]
- Giammanco, G.M.; Pignato, S.; Mammina, C.; Grimont, F.; Grimont, P.A.; Nastasi, A.; Giammanco, G. Persistent endemicity of Salmonella bongori 48: z35:-in southern Italy: Molecular characterization of human, animal, and environmental isolates. J. Clin. Microbiol. 2002, 40, 3502–3505. [Google Scholar] [CrossRef] [PubMed]
- Gal-Mor, O. Persistent infection and long-term carriage of typhoidal and nontyphoidal salmonellae. Clin. Microbiol. Rev. 2018, 32, e00088-18. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.F.; Ingram, L.A.; Cieslak, P.R.; Vugia, D.J.; Tobin-D’Angelo, M.; Hurd, S.; Medus, C.; Cronquist, A.; Angulo, F.J. Salmonellosis outcomes differ substantially by serotype. J. Infect. Dis. 2008, 198, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Havelaar, A.H.; Kirk, M.D.; Torgerson, P.R.; Gibb, H.J.; Hald, T.; Lake, R.J.; Praet, N.; Bellinger, D.C.; De Silva, N.R.; Gargouri, N. World Health Organization global estimates and regional comparisons of the burden of foodborne disease in 2010. PLoS Med. 2015, 12, e1001923. [Google Scholar] [CrossRef] [PubMed]
- Kirk, M.D.; Pires, S.M.; Black, R.E.; Caipo, M.; Crump, J.A.; Devleesschauwer, B.; Döpfer, D.; Fazil, A.; Fischer-Walker, C.L.; Hald, T.; et al. World Health Organization Estimates of the global and regional disease burden of 22 foodborne bacterial, protozoal, and viral diseases, 2010: A data synthesis. PLoS Med. 2015, 12, e1001921. [Google Scholar] [CrossRef]
- Boore, A.L.; Hoekstra, R.M.; Iwamoto, M.; Fields, P.I.; Bishop, R.D.; Swerdlow, D.L. Salmonella enterica infections in the United States and assessment of coefficients of variation: A novel approach to identify epidemiologic characteristics of individual serotypes, 1996–2011. PLoS ONE 2015, 10, e0145416. [Google Scholar] [CrossRef]
- Hendriksen, R.S.; Vieira, A.R.; Karlsmose, S.; Lo Fo Wong, D.M.; Jensen, A.B.; Wegener, H.C.; Aarestrup, F.M. Global monitoring of Salmonella serovar distribution from the World Health Organization Global Foodborne Infections Network Country Data Bank: Results of quality assured laboratories from 2001 to 2007. Foodborne Path. Dis. 2011, 8, 887–900. [Google Scholar] [CrossRef]
- Foley, S.L.; Johnson, T.J.; Ricke, S.C.; Nayak, R.; Danzeisen, J. Salmonella pathogenicity and host adaptation in chicken-associated serovars. Microbiol. Mol. Biol. Rev. 2013, 77, 582–607. [Google Scholar] [CrossRef]
- Pecoraro, H.L.; Thompson, B.; Duhamel, G.E. Histopathology case definition of naturally acquired Salmonella enterica serovar Dublin infection in young Holstein cattle in the northeastern United States. J. Vet. Diagn. Invest. 2017, 29, 860–864. [Google Scholar] [CrossRef] [PubMed]
- Hoelzer, K.; Moreno Switt, A.I.; Wiedmann, M. Animal contact as a source of human non-typhoidal salmonellosis. Vet. Res. 2011, 42, 32. [Google Scholar] [CrossRef] [PubMed]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.-A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States—Major pathogens. Emerg. Infect. Dis. 2011, 17, 7. [Google Scholar] [CrossRef] [PubMed]
- Ebel, E.D.; Williams, M.S.; Cole, D.; Travis, C.C.; Klontz, K.C.; Golden, N.J.; Hoekstra, R.M. Comparing characteristics of sporadic and outbreak-associated foodborne illnesses, United States, 2004–2011. Emerg. Infect. Dis. 2016, 22, 1193. [Google Scholar] [CrossRef] [PubMed]
- Tsolis, R.M.; Xavier, M.N.; Santos, R.L.; Baumler, A.J. How to become a top model: Impact of animal experimentation on human Salmonella disease research. Infect. Immun. 2011, 79, 1806–1814. [Google Scholar] [CrossRef] [PubMed]
- Sabbagh, S.C.; Forest, C.G.; Lepage, C.; Leclerc, J.-M.; Daigle, F. So similar, yet so different: Uncovering distinctive features in the genomes of Salmonella enterica serovars Typhimurium and Typhi. FEMS Microbiol. Lett. 2010, 305, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Chavez, F.; Baumler, A.J. The pyromaniac inside you: Salmonella metabolism in the host gut. Annu. Rev. Microbiol. 2015, 69, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Raffatellu, M.; Chessa, D.; Wilson, R.P.; Dusold, R.; Rubino, S.; Bäumler, A.J. The Vi capsular antigen of Salmonella enterica serotype Typhi reduces Toll-like receptor-dependent interleukin-8 expression in the intestinal mucosa. Infect. Immun. 2005, 73, 3367–3374. [Google Scholar] [CrossRef]
- Winter, S.E.; Winter, M.G.; Atluri, V.; Poon, V.; Romão, E.L.; Tsolis, R.M.; Bäumler, A.J. The flagellar regulator TviA reduces pyroptosis by Salmonella enterica serovar Typhi. Infect. Immun. 2015, 83, 1546–1555. [Google Scholar] [CrossRef]
- Song, J.; Gao, X.; Galán, J.E. Structure and function of the Salmonella Typhi chimaeric A2B5 typhoid toxin. Nature 2013, 499, 350–354. [Google Scholar] [CrossRef]
- Locht, C.; Coutte, L.; Mielcarek, N. The ins and outs of pertussis toxin. FEBS J. 2011, 278, 4668–4682. [Google Scholar] [CrossRef] [PubMed]
- Aktories, K.; Bärmann, M.; Ohishi, I.; Tsuyama, S.; Jakobs, K.; Habermann, E. Botulinum C2 toxin ADP-ribosylates actin. Nature 1986, 322, 390. [Google Scholar] [CrossRef] [PubMed]
- Collier, R.J. Diphtheria toxin: Mode of action and structure. Bacteriol. Rev. 1975, 39, 54–85. [Google Scholar] [PubMed]
- Gill, D.M.; Meren, R. ADP-ribosylation of membrane proteins catalyzed by cholera toxin: Basis of the activation of adenylate cyclase. Proc. Natl. Acad. Sci. USA 1978, 75, 3050–3054. [Google Scholar] [CrossRef] [PubMed]
- Simon, N.C.; Aktories, K.; Barbieri, J.T. Novel bacterial ADP-ribosylating toxins: Structure and function. Nat. Rev. Microbiol. 2014, 12, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Deng, Q.; Barbieri, J.T. Molecular mechanisms of the cytotoxicity of ADP-ribosylating toxins. Annu. Rev. Microbiol. 2008, 62, 271–288. [Google Scholar] [CrossRef]
- Lang, A.E.; Kuhn, S.; Mannherz, H.G. Photorhabdus luminescens toxins TccC3 and TccC5 affect the interaction of actin with actin-binding proteins essential for treadmilling. Curr. Top. Microbiol. Immunol. 2017, 399, 53–67. [Google Scholar]
- Lang, A.E.; Schmidt, G.; Schlosser, A.; Hey, T.D.; Larrinua, I.M.; Sheets, J.J.; Mannherz, H.G.; Aktories, K. Photorhabdus luminescens toxins ADP-ribosylate actin and RhoA to force actin clustering. Science 2010, 327, 1139–1142. [Google Scholar] [CrossRef]
- Guiney, D.G.; Fierer, J. The role of the spv genes in Salmonella pathogenesis. Front. Microbiol. 2011, 2, 129–129. [Google Scholar] [CrossRef]
- Spanò, S.; Ugalde, J.E.; Galán, J.E. Delivery of a Salmonella Typhi exotoxin from a host intracellular compartment. Cell. Host Microbe 2008, 3, 30–38. [Google Scholar] [CrossRef]
- Saitoh, M.; Tanaka, K.; Nishimori, K.; Makino, S.-i.; Kanno, T.; Ishihara, R.; Hatama, S.; Kitano, R.; Kishima, M.; Sameshima, T. The artAB genes encode a putative ADP-ribosyltransferase toxin homologue associated with Salmonella enterica serovar Typhimurium DT104. Microbiology. 2005, 151, 3089–3096. [Google Scholar] [CrossRef]
- Pollard, D.J.; Young, J.C.; Covarelli, V.; Herrera-León, S.; Connor, T.R.; Fookes, M.; Walker, D.; Echeita, A.; Thomson, N.R.; Berger, C.N.; et al. The type III secretion system effector SeoC of Salmonella enterica subsp. salamae and S. enterica subsp. arizonae ADP-ribosylates Src and inhibits opsonophagocytosis. Infect. Immun. 2016, 84, 3618–3628. [Google Scholar]
- Tamamura, Y.; Tanaka, K.; Uchida, I. Characterization of pertussis-like toxin from Salmonella spp. that catalyzes ADP-ribosylation of G proteins. Sci. Rep. 2017, 7, 2653–2653. [Google Scholar] [CrossRef] [PubMed]
- Uchida, I.; Ishihara, R.; Tanaka, K.; Hata, E.; Makino, S.; Kanno, T.; Hatama, S.; Kishima, M.; Akiba, M.; Watanabe, A.; et al. Salmonella enterica serotype Typhimurium DT104 ArtA-dependent modification of pertussis toxin-sensitive G proteins in the presence of [32P]NAD. Microbiology 2009, 155, 3710–3718. [Google Scholar] [CrossRef]
- Young, J.C.; Clements, A.; Lang, A.E.; Garnett, J.A.; Munera, D.; Arbeloa, A.; Pearson, J.; Hartland, E.L.; Matthews, S.J.; Mousnier, A.; et al. The Escherichia coli effector EspJ blocks Src kinase activity via amidation and ADP ribosylation. Nat. Commun. 2014, 5, 5887. [Google Scholar] [CrossRef]
- Hochmann, H.; Pust, S.; von Figura, G.; Aktories, K.; Barth, H. Salmonella enterica SpvB ADP-ribosylates actin at position arginine-177-characterization of the catalytic domain within the SpvB protein and a comparison to binary clostridial actin-ADP-ribosylating toxins. Biochemistry 2006, 45, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Kappeli, R.; Kaiser, P.; Stecher, B.; Hardt, W.D. Roles of spvB and spvC in S. Typhimurium colitis via the alternative pathway. Int. J. Med. Microbiol. 2011, 301, 117–124. [Google Scholar] [CrossRef]
- Gulig, P.A. Virulence plasmids of Salmonella typhimurium and other salmonellae. Microb. Pathog. 1990, 8, 3–11. [Google Scholar] [CrossRef]
- Jones, G.W.; Rabert, D.K.; Svinarich, D.M.; Whitfield, H.J. Association of adhesive, invasive, and virulent phenotypes of Salmonella typhimurium with autonomous 60-megadalton plasmids. Infect. Immun. 1982, 38, 476–486. [Google Scholar] [PubMed]
- Williamson, C.M.; Pullinger, G.D.; Lax, A.J. Identification of an essential virulence region on Salmonella plasmids. Microb. Pathog. 1988, 5, 469–473. [Google Scholar] [CrossRef]
- Lesnick, M.L.; Reiner, N.E.; Fierer, J.; Guiney, D.G. The Salmonella spvB virulence gene encodes an enzyme that ADP-ribosylates actin and destabilizes the cytoskeleton of eukaryotic cells. Mol. Microbiol. 2001, 39, 1464–1470. [Google Scholar] [CrossRef] [PubMed]
- Libby, S.J.; Lesnick, M.; Hasegawa, P.; Kurth, M.; Belcher, C.; Fierer, J.; Guiney, D.G. Characterization of the spv locus in Salmonella enterica serovar Arizona. Infect. Immun. 2002, 70, 3290–3294. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rotger, R.; Casadesus, J. The virulence plasmids of Salmonella. Int. Microbiol. 1999, 2, 177–184. [Google Scholar]
- Haneda, T.; Ishii, Y.; Shimizu, H.; Ohshima, K.; Iida, N.; Danbara, H.; Okada, N. Salmonella type III effector SpvC, a phosphothreonine lyase, contributes to reduction in inflammatory response during intestinal phase of infection. Cell. Microbiol. 2012, 14, 485–499. [Google Scholar] [CrossRef] [PubMed]
- Rolhion, N.; Furniss, R.C.; Grabe, G.; Ryan, A.; Liu, M.; Matthews, S.A.; Holden, D.W. Inhibition of nuclear transport of NF-kB p65 by the Salmonella type III secretion system effector SpvD. PLoS Pathog. 2016, 12, e1005653. [Google Scholar] [CrossRef] [PubMed]
- Fookes, M.; Schroeder, G.N.; Langridge, G.C.; Blondel, C.J.; Mammina, C.; Connor, T.R.; Seth-Smith, H.; Vernikos, G.S.; Robinson, K.S.; Sanders, M.; et al. Salmonella bongori provides insights into the evolution of the Salmonellae. PLoS Pathog. 2011, 7, e1002191. [Google Scholar] [CrossRef] [PubMed]
- den Bakker, H.C.; Switt, A.I.M.; Govoni, G.; Cummings, C.A.; Ranieri, M.L.; Degoricija, L.; Hoelzer, K.; Rodriguez-Rivera, L.D.; Brown, S.; Bolchacova, E.; et al. Genome sequencing reveals diversification of virulence factor content and possible host adaptation in distinct subpopulations of Salmonella enterica. BMC Genom. 2011, 12, 425. [Google Scholar] [CrossRef]
- Silva, C.; Puente, J.L.; Calva, E. Salmonella virulence plasmid: Pathogenesis and ecology. Pathog. Dis. 2017, 75, ftx070. [Google Scholar] [CrossRef]
- Akiba, M.; Sameshima, T.; Anzai, T.; Wada, R.; Nakazawa, M. Salmonella Abortusequi strains of equine origin harbor a 95kb plasmid responsible for virulence in mice. Vet. Microbiol. 1999, 68, 265–272. [Google Scholar] [CrossRef]
- Uzzau, S.; Gulig, P.A.; Paglietti, B.; Leori, G.; Stocker, B.A.; Rubino, S. Role of the Salmonella abortusovis virulence plasmid in the infection of BALB/c mice. FEMS Microbiol. Lett. 2000, 188, 15–18. [Google Scholar] [CrossRef]
- Bronowski, C.; Fookes, M.C.; Gilderthorp, R.; Ashelford, K.E.; Harris, S.R.; Phiri, A.; Hall, N.; Gordon, M.A.; Wain, J.; Hart, C.A.; et al. Genomic characterisation of invasive non-typhoidal Salmonella enterica subspecies enterica serovar Bovismorbificans isolates from Malawi. PLoS Negl. Trop. Dis. 2013, 7, e2557. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.A.; Wiedmann, M. The cytolethal distending toxin produced by nontyphoidal Salmonella serotypes Javiana, Montevideo, Oranienburg, and Mississippi induces DNA damage in a manner similar to that of serotype Typhi. mBio 2016, 7, e02109-16. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.Q.; Feng, Y.; Wang, Y.; Zou, Q.H.; Chen, F.; Guo, J.T.; Peng, Y.H.; Jin, Y.; Li, Y.G.; Hu, S.N.; et al. Salmonella paratyphi C: Genetic divergence from Salmonella choleraesuis and pathogenic convergence with Salmonella typhi. PLoS ONE 2009, 4, e4510. [Google Scholar] [CrossRef] [PubMed]
- WHO. Collaborating Centre for Reference and Research on Salmonella. Antigenic Formulae of the Salmonella Serovars; WHO: Geneva, Switzerland, 2007; Available online: https://www.pasteur.fr/sites/default/files/veng_0.pdf (accessed on 17 June 2019).
- CDC. National Enteric Disease Surveillance: Salmonella Annual Report, 2016; CDC: Atlanta, GA, USA, 2016. Available online: https://www.cdc.gov/nationalsurveillance/pdfs/2016-Salmonella-report-508.pdf (accessed on 17 June 2019).
- Jennings, E.; Thurston, T.L.M.; Holden, D.W. Salmonella SPI-2 type III secretion system effectors: Molecular mechanisms and physiological consequences. Cell Host Microbe 2017, 22, 217–231. [Google Scholar] [CrossRef] [PubMed]
- LaRock, D.L.; Chaudhary, A.; Miller, S.I. Salmonellae interactions with host processes. Nat. Rev. Microbiol. 2015, 13, 191. [Google Scholar] [CrossRef] [PubMed]
- Steele-Mortimer, O. The Salmonella-containing vacuole: Moving with the times. Curr. Opin. Microbiol. 2008, 11, 38–45. [Google Scholar] [CrossRef]
- Mesa-Pereira, B.; Medina, C.; Camacho, E.M.; Flores, A.; Santero, E. Novel tools to analyze the function of Salmonella effectors show that SvpB ectopic expression induces cell cycle arrest in tumor cells. PLoS ONE 2013, 8, e78458. [Google Scholar] [CrossRef] [PubMed]
- Lohez, O.D.; Reynaud, C.; Borel, F.; Andreassen, P.R.; Margolis, R.L. Arrest of mammalian fibroblasts in G1 in response to actin inhibition is dependent on retinoblastoma pocket proteins but not on p53. J. Cell Biol. 2003, 161, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Miao, E.A.; Brittnacher, M.; Haraga, A.; Jeng, R.L.; Welch, M.D.; Miller, S.I. Salmonella effectors translocated across the vacuolar membrane interact with the actin cytoskeleton. Mol. Microbiol. 2003, 48, 401–415. [Google Scholar] [CrossRef]
- Meresse, S.; Unsworth, K.E.; Habermann, A.; Griffiths, G.; Fang, F.; Martinez-Lorenzo, M.J.; Waterman, S.R.; Gorvel, J.P.; Holden, D.W. Remodelling of the actin cytoskeleton is essential for replication of intravacuolar Salmonella. Cell. Microbiol. 2001, 3, 567–577. [Google Scholar] [CrossRef]
- Portnoy, D.A.; Auerbuch, V.; Glomski, I.J. The cell biology of Listeria monocytogenes infection: The intersection of bacterial pathogenesis and cell-mediated immunity. J. Cell Biol. 2002, 158, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Reeves, A.Z.; Klein, J.A.; Twedt, D.J.; Knodler, L.A.; Lesser, C.F. The type III secretion system apparatus determines the intracellular niche of bacterial pathogens. Proc. Natl. Acad. Sci. USA 2016, 113, 4794–4799. [Google Scholar] [CrossRef] [PubMed]
- Ray, K.; Marteyn, B.; Sansonetti, P.J.; Tang, C.M. Life on the inside: The intracellular lifestyle of cytosolic bacteria. Nat. Rev. Microbiol. 2009, 7, 333. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, K.; Sørensen, G.; Löfström, C.; Leekitcharoenphon, P.; Nielsen, B.; Wingstrand, A.; Aarestrup, F.M.; Hendriksen, R.S.; Baggesen, D.L. Reappearance of Salmonella serovar Choleraesuis var. Kunzendorf in Danish pig herds. Vet. Microbiol. 2015, 176, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Grandolfo, E.; Parisi, A.; Ricci, A.; Lorusso, E.; de Siena, R.; Trotta, A.; Buonavoglia, D.; Martella, V.; Corrente, M. High mortality in foals associated with Salmonella enterica subsp. enterica Abortusequi infection in Italy. J. Vet. Diagn. Inv. 2018, 30, 483–485. [Google Scholar]
- Pardon, P.; Sanchis, R.; Marly, J.; Lantier, F.; Pepin, M.; Popoff, M. Ovine salmonellosis caused by Salmonella abortus ovis. Ann. Rech. Vet. 1988, 19, 221–235. [Google Scholar] [PubMed]
- Matsui, H.; Bacot, C.M.; Garlington, W.A.; Doyle, T.J.; Roberts, S.; Gulig, P.A. Virulence plasmid-borne spvB and spvC genes can replace the 90-kilobase plasmid in conferring virulence to Salmonella enterica serovar Typhimurium in subcutaneously inoculated mice. J. Bacteriol. 2001, 183, 4652–4658. [Google Scholar] [CrossRef] [PubMed]
- Kidwai, A.S.; Mushamiri, I.; Niemann, G.S.; Brown, R.N.; Adkins, J.N.; Heffron, F. Diverse secreted effectors are required for Salmonella persistence in a mouse infection model. PLoS ONE 2013, 8, e70753. [Google Scholar] [CrossRef] [PubMed]
- Haghjoo, E.; Galán, J.E. Salmonella typhi encodes a functional cytolethal distending toxin that is delivered into host cells by a bacterial-internalization pathway. Proc. Natl. Acad. Sci. USA 2004, 101, 4614–4619. [Google Scholar] [CrossRef]
- Jinadasa, R.N.; Bloom, S.E.; Weiss, R.S.; Duhamel, G.E. Cytolethal distending toxin: A conserved bacterial genotoxin that blocks cell cycle progression, leading to apoptosis of a broad range of mammalian cell lineages. Microbiology 2011, 157, 1851–1875. [Google Scholar] [CrossRef]
- Hodak, H.; Galan, J.E. A Salmonella Typhi homologue of bacteriophage muramidases controls typhoid toxin secretion. EMBO Rep. 2013, 14, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rivera, L.D.; Bowen, B.M.; den Bakker, H.C.; Duhamel, G.E.; Wiedmann, M. Characterization of the cytolethal distending toxin (typhoid toxin) in non-typhoidal Salmonella serovars. Gut Pathoh. 2015, 7, 1. [Google Scholar] [CrossRef] [PubMed]
- Fowler, C.; Stack, G.; Jiao, X.; Lara-Tejero, M.; Galán, J.E. Alternate subunit assembly diversifies the function of a bacterial toxin. bioRxiv 2019, 624130. [Google Scholar]
- Gao, X.; Deng, L.; Stack, G.; Yu, H.; Chen, X.; Naito-Matsui, Y.; Varki, A.; Galán, J.E. Evolution of host adaptation in the Salmonella typhoid toxin. Nat. Microbiol. 2017, 2, 1592. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.A.; Lee, S.; Zhao, J.; Thompson, A.J.; McBride, R.; Tsogtbaatar, B.; Paulson, J.C.; Nussinov, R.; Deng, L.; Song, J. In vivo tropism of Salmonella Typhi toxin to cells expressing a multiantennal glycan receptor. Nat. Microbiol. 2018, 3, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.J.; Jin, S.C.; Jiao, X.; Galan, J.E. Unique features in the intracellular transport of typhoid toxin revealed by a genome-wide screen. PLoS Pathog. 2019, 15, e1007704. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Weiss, A.A.; Hewlett, E.L. Virulence factors of Bordetella pertussis. Annu. Rev. Microbiol. 1986, 40, 661–686. [Google Scholar] [CrossRef]
- Miller, R.A.; Betteken, M.I.; Guo, X.; Altier, C.; Duhamel, G.E.; Wiedmann, M. The typhoid toxin produced by the nontyphoidal Salmonella enterica serotype Javiana is required for induction of a DNA damage response in vitro andsystemic spread in vivo. mBio 2018, 9, e00467-18. [Google Scholar] [CrossRef]
- Belluz, L.D.B.; Guidi, R.; Pateras, I.S.; Levi, L.; Mihaljevic, B.; Rouf, S.F.; Wrande, M.; Candela, M.; Turroni, S.; Nastasi, C.; et al. The typhoid toxin promotes host Ssurvival and the establishment of a persistent asymptomatiinfection. PLoS Pathog. 2016, 12, e1005528. [Google Scholar]
- Carbonetti, N.H. Pertussis toxin and adenylate cyclase toxin: Key virulence factors of Bordetella pertussis and cell biology tools. Future Microbiol. 2010, 5, 455–469. [Google Scholar] [CrossRef] [PubMed]
- Otsuji, N.; Sekiguchi, M.; Iijima, T.; Takagi, Y. Induction of phage formation in the lysogenic Escherichia coli K-12 by mitomycin C. Nature 1959, 184, 1079. [Google Scholar] [CrossRef] [PubMed]
- Hiley, L.; Fang, N.X.; Micalizzi, G.R.; Bates, J. Distribution of Gifsy-3 and of variants of ST64B and Gifsy-1 prophages amongst Salmonella enterica serovar Typhimurium isolates: Evidence that combinations of prophages promote clonality. PLoS ONE 2014, 9, e86203. [Google Scholar] [CrossRef] [PubMed]
- Moreno Switt, A.I.; den Bakker, H.C.; Cummings, C.A.; Rodriguez-Rivera, L.D.; Govoni, G.; Raneiri, M.L.; Degoricija, L.; Brown, S.; Hoelzer, K.; Peters, J.E.; et al. Identification and characterization of novel Salmonella mobile elements involved in the dissemination of genes linked to virulence and transmission. PLoS ONE 2012, 7, e41247. [Google Scholar] [CrossRef] [PubMed]
- Avigan, J.; Murtagh, J.J., Jr.; Stevens, L.A.; Angus, C.W.; Moss, J.; Vaughan, M. Pertussis toxin-catalyzed ADP-ribosylation of G(o) alpha with mutations at the carboxyl terminus. Biochemistry 1992, 31, 7736–7740. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Barbieri, J.T. Pertussis toxin-catalyzed ADP-ribosylation of Gi-2 and Gi-3 in CHO cells is modulated by inhibitors of intracellular trafficking. Infect. Immun. 1996, 64, 593–599. [Google Scholar] [PubMed]
- Melvin, J.A.; Scheller, E.V.; Miller, J.F.; Cotter, P.A. Bordetella pertussis pathogenesis: Current and future challenges. Nat. Rev. Microbiol. 2014, 12, 274. [Google Scholar] [CrossRef] [PubMed]
- Hewlett, E.L.; Sauer, K.T.; Myers, G.A.; Cowell, J.L.; Guerrant, R.L. Induction of a novel morphological response in Chinese hamster ovary cells by pertussis toxin. Infect. Immun. 1983, 40, 1198–1203. [Google Scholar]
- Scanlon, K.M.; Snyder, Y.G.; Skerry, C.; Carbonetti, N.H. Fatal pertussis in the neonatal mouse model is associated with pertussis toxin-mediated pathology beyond the airways. Infect. Immun. 2017, 85, e00355-17. [Google Scholar] [CrossRef]
- Dahan, S.; Wiles, S.; La Ragione, R.M.; Best, A.; Woodward, M.J.; Stevens, M.P.; Shaw, R.K.; Chong, Y.; Knutton, S.; Phillips, A.; et al. EspJ is a prophage-carried type III effector protein of attaching and effacing pathogens that modulates infection dynamics. Infect. Immun. 2005, 73, 679–686. [Google Scholar] [CrossRef]
- Pollard, D.J.; Berger, C.N.; So, E.C.; Yu, L.; Hadavizadeh, K.; Jennings, P.; Tate, E.W.; Choudhary, J.S.; Frankel, G. Broad-spectrum regulation of nonreceptor tyrosine kinases by the bacterial ADP-ribosyltransferase EspJ. mBio 2018, 9, e00170-18. [Google Scholar] [CrossRef] [PubMed]
- Abbott, S.L.; Ni, F.C.Y.; Janda, J.M. Increase in extraintestinal infections caused by Salmonella enterica subspecies II-IV. Emerg. Infect. Dis. 2012, 18, 637–639. [Google Scholar] [CrossRef] [PubMed]
- Lamas, A.; Miranda, J.M.; Regal, P.; Vazquez, B.; Franco, C.M.; Cepeda, A. A comprehensive review of non-enterica subspecies of Salmonella enterica. Microbiolog. Res. 2018, 206, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Fàbrega, A.; Vila, J. Salmonella enterica serovar Typhimurium skills to succeed in the host: Virulence and regulation. Clin. Microbiol. Res. 2013, 26, 308–341. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, C.E.; Kruczkiewicz, P.; Laing, C.R.; Lingohr, E.J.; Gannon, V.P.; Nash, J.H.; Taboada, E.N. The Salmonella in silico typing resource (SISTR): An open web-accessible tool for rapidly typing and subtyping draft Salmonella genome assemblies. PLoS ONE 2016, 11, e0147101. [Google Scholar] [CrossRef]
- Worley, J.; Meng, J.; Allard, M.W.; Brown, E.W.; Timme, R.E. Salmonella enterica phylogeny based on whole-genome sequencing reveals two new clades and novel patterns of horizontally acquired genetic elements. mBio 2018, 9, e02303-18. [Google Scholar] [CrossRef]
- Alikhan, N.-F.; Zhou, Z.; Sergeant, M.J.; Achtman, M. A genomic overview of the population structure of Salmonella. PLOS Genet. 2018, 14, e1007261. [Google Scholar] [CrossRef]
- Yachison, C.A.; Yoshida, C.; Robertson, J.; Nash, J.H.E.; Kruczkiewicz, P.; Taboada, E.N.; Walker, M.; Reimer, A.; Christianson, S.; Nichani, A.; et al. The validation and implications of using whole genome sequencing as a replacement for traditional serotyping for a National Salmonella Reference Laboratory. Front. Microbiol. 2017, 8, 1044. [Google Scholar] [CrossRef]
- Wagner, L.D.; Corvette, L.J.; Ngundi, M.M.; Burns, D.L. Towards replacement of the acellular pertussis vaccine safety test: Comparison of in vitro cytotoxic activity and in vivo activity in mice. Vaccine 2017, 35, 7160–7165. [Google Scholar] [CrossRef]
| bARTT | Host Cell Targets 1 | Known Homologues | Known Contributions to Pathogenesis | References |
|---|---|---|---|---|
| ArtAB | Gαi2 and Gαi3 | PtxA (B. pertussis) PltA (S. enterica) |
| [33,34] |
| PltA (Typhoid toxin) | Unknown | PtxA (B. pertussis) ArtA (Salmonella spp.) |
| [30] |
| SboC/SeoC | Src kinase residue Glu310, Csk kinase residue Glu236 | EspJ (E. coli and C. rodentium) |
| [32,35] |
| SpvB | Actin monomer residue Arg177 | None |
| [36,37] |
| Species/Sub-Species | Serovar/Species | Serogroup 1 | # of Reported Human Cases in 2006–2016 USA 2 | artAB3 | pltAB (Typhoid Toxin) 3 | sboC/seoC3 | spvB3 | References |
|---|---|---|---|---|---|---|---|---|
| S. bongori | Multiple | Multiple | 17 | + | + | +/- | [46] | |
| I (enterica) | Abony | O:4 (B) | 62 | +/- | +/- | [33,47] | ||
| Abortusequi | O:4 (B) | 5 | - | - | + | [33,48,49] | ||
| Abortusovis | O:4 (B) | 0 | + | [50] | ||||
| Agbeni | O:13 (G) | 522 | - | + | [47] | |||
| Agoueve | O:13 (G) | 37 | + | - | [33] | |||
| Alachua | O:35 (O) | 179 | +/- | - | [33,47] | |||
| Arechavaleta | O:4 (B) | 106 | + | + | [47] | |||
| Barranquilla | O:16 (I) | 89 | + | + | [47] | |||
| Bovismorbificans | O:8 (C2-C3) | 817 | +/- | - | + | [33,47,51] | ||
| Brandenburg | O:4 (B) | 882 | + | + | [33] | |||
| Bredeney | O:4 (B) | 351 | + | + | [33] | |||
| Chester | O:4 (B) | 474 | + | + | [33] | |||
| Choleraesuis | O:7 (C1) | 181 | - | - | + | [33,47,48] | ||
| Corvallis | O:8 (C2-C3) | 261 | +/- | +/- | [33,47] | |||
| Cotham | O:28 (M) | 429 | + | + | [47] | |||
| Cubana | O:13 (G) | 186 | - | +/- | [33,47] | |||
| Dublin | O:9 (D1) | 1,388 | - | - | + | [33,47,48] | ||
| Durban | O:9 (D1) | 156 | + | + | [33] | |||
| Enteritidis | O:9 (D1) | 83,303 | - | - | + | [33,47,48,52] | ||
| Essen | O:4 (B) | 16 | + | + | [33] | |||
| Freetown | O:38 (P) | 8 | - | + | [33] | |||
| Gallinarum | O:9 (D1) | 0 | - | - | + | [47,48] | ||
| Gaminara | O:16 (I) | 981 | + | + | [47] | |||
| Georgia | O:7 (C1) | 19 | + | + | [47] | |||
| Give | O:3,10 (E1) | 1,309 | + | + | [33,47] | |||
| Glostrup | O:8 (C2-C3) | 48 | + | + | [47] | |||
| Indiana | O:4 (B) | 302 | + | + | [47] | |||
| Inganda | O:7 (C1) | 8 | + | + | [33] | |||
| Inverness | O:38 (P) | 620 | + | - | [47] | |||
| Javiana | O:9 (D1) | 25,955 | + | + | [33,47,52] | |||
| Johannesburg | O:40 (R) | 434 | + | + | [33,47] | |||
| Kiambu | O:4 (B) | 656 | - | + | [33,47] | |||
| Kintambo | O:13 (G) | 98 | - | + | [47] | |||
| Kisarawe | O:11 (F) | 27 | + | + | [47] | |||
| Luciana | O:11 (F) | 61 | - | + | [47] | |||
| Miami | O:9 (D1) | 1,203 | - | + | [47] | |||
| Minnesota | O:21 (L) | 301 | + | + | [33,47] | |||
| Mississippi | O:13 (G) | 5,771 | + | +/- | [33,47,52] | |||
| Montevideo | O:7 (C1) | 11,495 | + | + | [33,47,52] | |||
| Muenster | O:3,10 (E1) | 757 | - | +/- | [33,47] | |||
| Oranienburg | O:7 (C1) | 8,012 | + | + | [33,47,52] | |||
| Overschie | O:51 | 16 | + | + | [47] | |||
| Panama | O:9 (D1) | 1,980 | + | + | [33,47] | |||
| Paratyphi A | O:2 (A) | 1,716 | + | + | [47] | |||
| Paratyphi C | O:7 (C1) | 11 | - | - | + | [47,53] | ||
| Pomona | O:28 (M) | 713 | + | + | [47] | |||
| Poona | O:13 (G) | 3,844 | + | + | [47] | |||
| Reading | O:4 (B) | 858 | + | + | [47] | |||
| Rubislaw | O:11 (F) | 1,757 | + | + | [47] | |||
| Ruiru | O:21 (L) | 7 | + | + | [33] | |||
| Sandiego | O:4 (B) | 1,982 | + | + | [47] | |||
| Schwarzengrund | O:4 (B) | 2,934 | + | + | [33,47] | |||
| Sendai | O:9 (D1) | 0 | + | [48] | ||||
| Strasbourg | O:9,46 (D2) | 0 | - | + | [33] | |||
| Telelkebir | O:13 (G) | 339 | + | + | [47] | |||
| Typhi | O:9 (D1) | 4,788 | + | + | [47] | |||
| Typhimurium | O:4 (B) | 63,773 | +/- | - | + | [33,47,48,52] | ||
| Urbana | O:30 (N) | 511 | + | + | [47] | |||
| Wandsworth | O:39 (Q) | 114 | +/- | +/- | [33,47] | |||
| Welikade | O:16 (I) | 5 | + | + | [33] | |||
| Worthington | O:13 (G) | 363 | +/- | - | [33,47] | |||
| II (salamae) | Multiple | Multiple | 271 | +/- | [32] | |||
| IIIa (arizonae) | Multiple | Multiple | 959 | +/- | +/- | [32] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, R.A.; Wiedmann, M. The ADP-Ribosylating Toxins of Salmonella. Toxins 2019, 11, 416. https://doi.org/10.3390/toxins11070416
Cheng RA, Wiedmann M. The ADP-Ribosylating Toxins of Salmonella. Toxins. 2019; 11(7):416. https://doi.org/10.3390/toxins11070416
Chicago/Turabian StyleCheng, Rachel A., and Martin Wiedmann. 2019. "The ADP-Ribosylating Toxins of Salmonella" Toxins 11, no. 7: 416. https://doi.org/10.3390/toxins11070416
APA StyleCheng, R. A., & Wiedmann, M. (2019). The ADP-Ribosylating Toxins of Salmonella. Toxins, 11(7), 416. https://doi.org/10.3390/toxins11070416






