Bee Venom Alleviates Atopic Dermatitis Symptoms through the Upregulation of Decay-Accelerating Factor (DAF/CD55)
Abstract
1. Introduction
2. Results
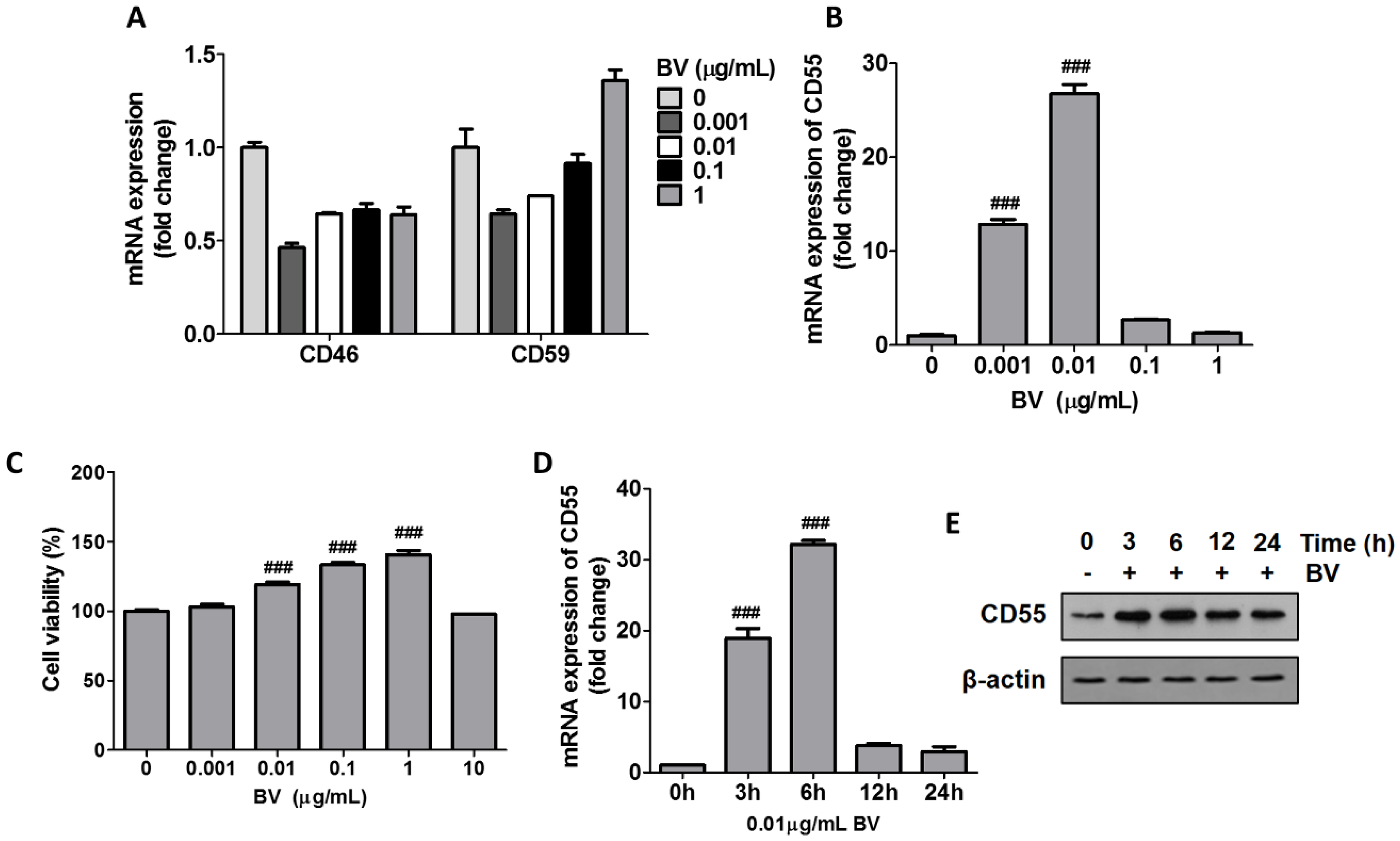
2.1. BV Increased CD55 Production in THP-1 Cells
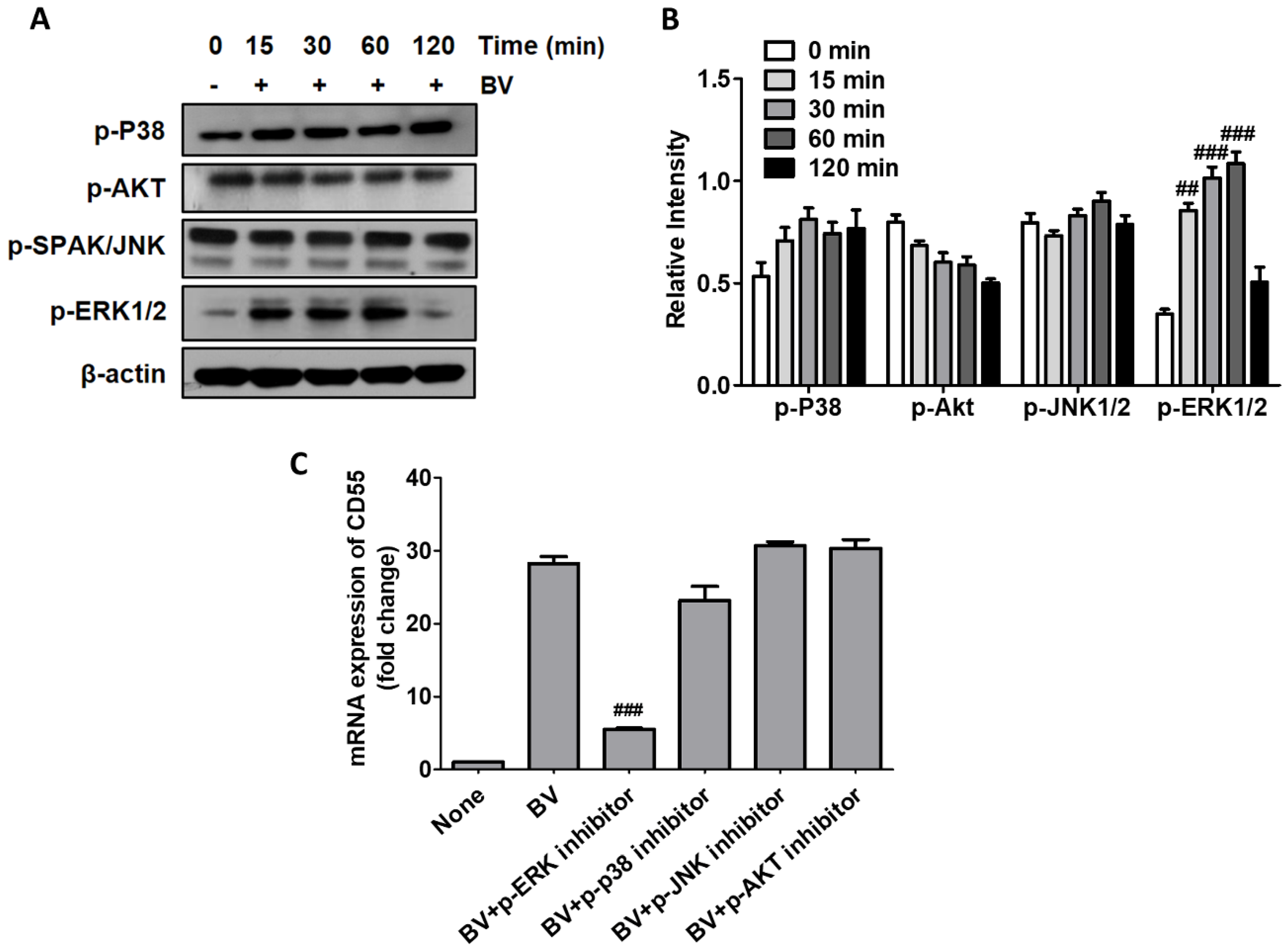
2.2. BV Induced CD55 Through the Activation of ERK
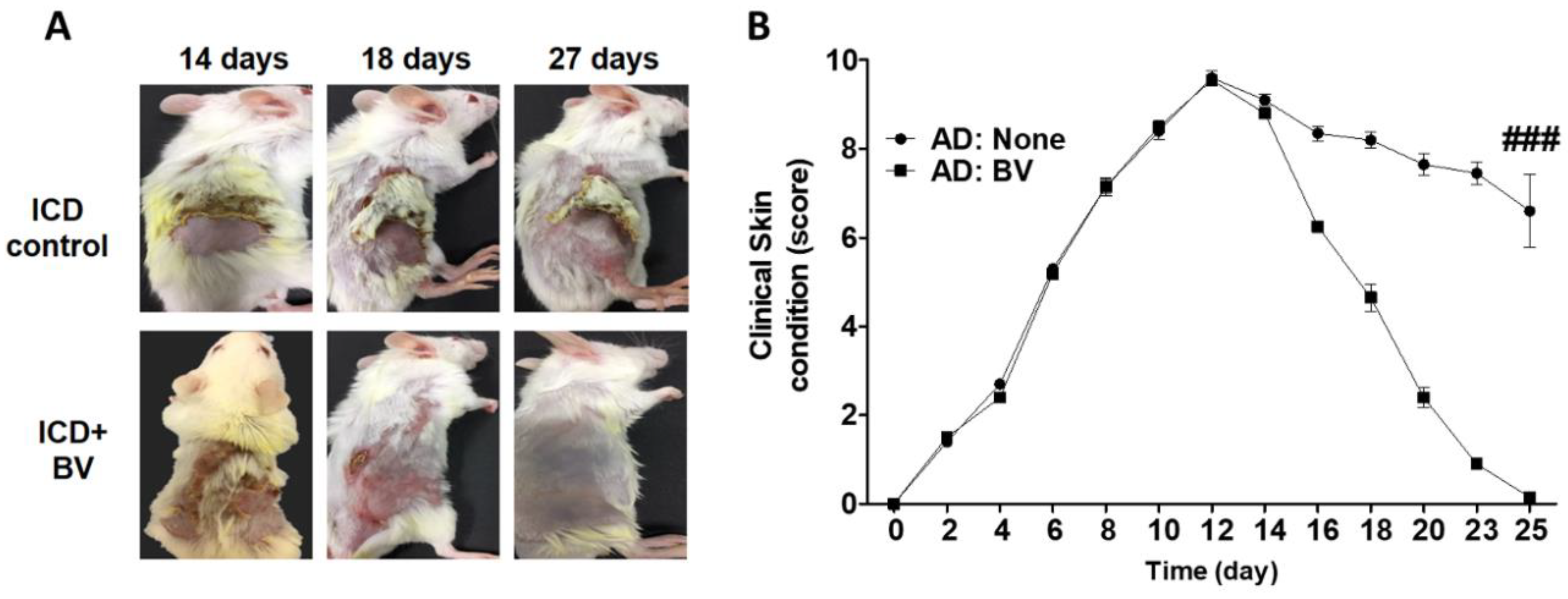
2.3. BV Alleviated AD Symptoms
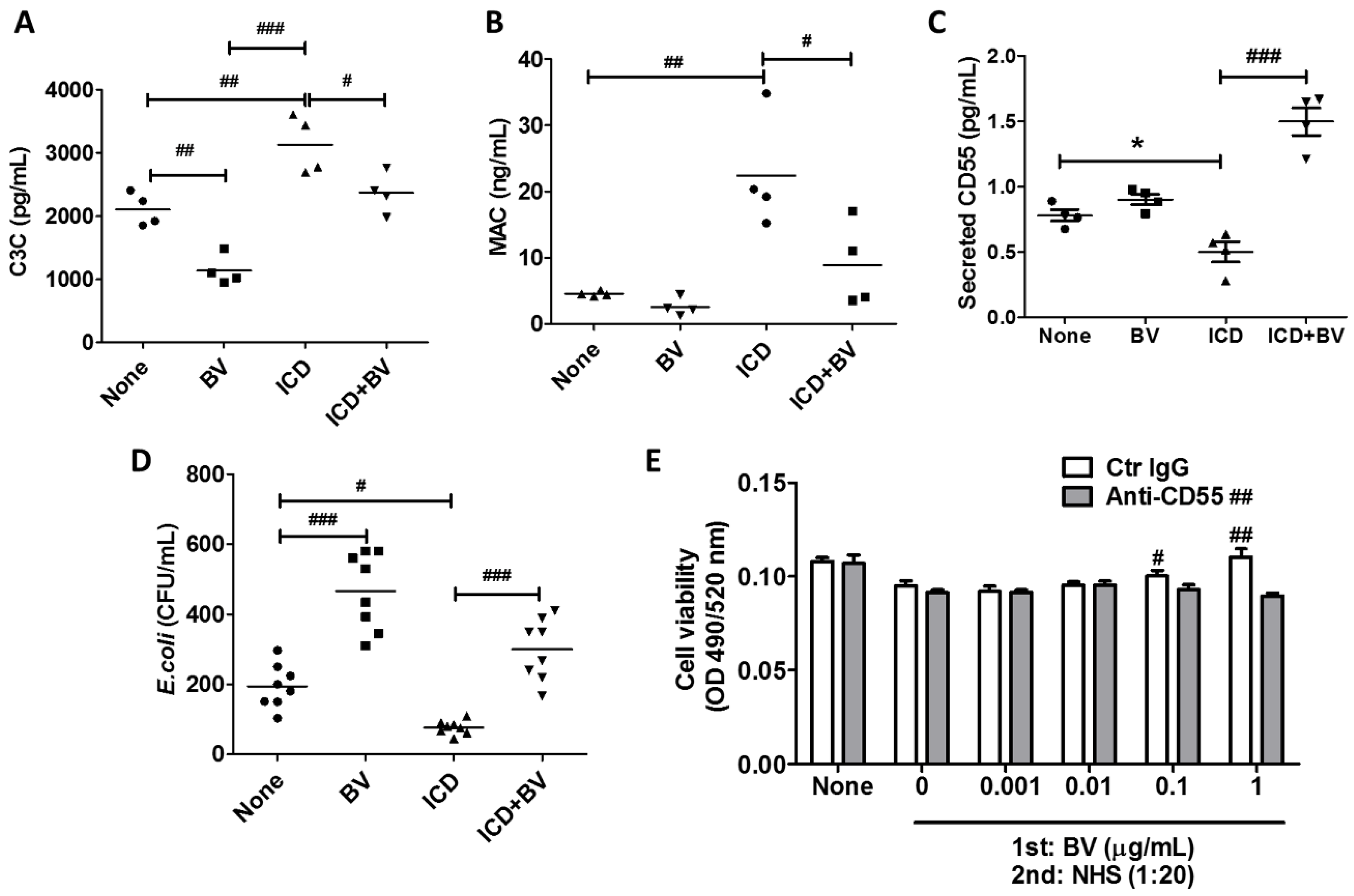
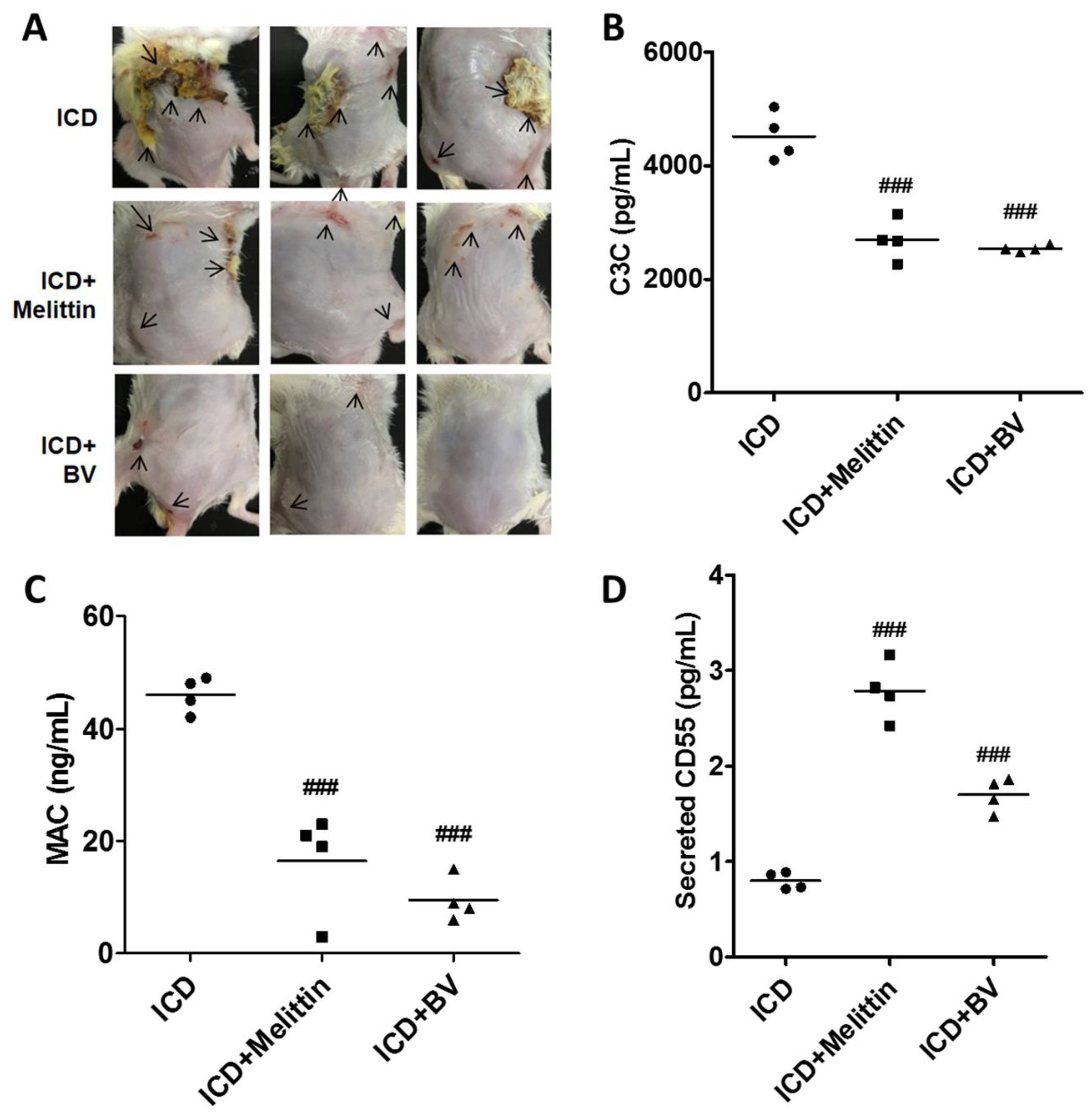
2.4. BV Inactivated Complement System in AD-Like Mice
2.5. Melittin Alleviated AD Symptoms Through the Regulation of Complement
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Drugs
4.3. Real-Time PCR
4.4. Western Blot
4.5. Animals
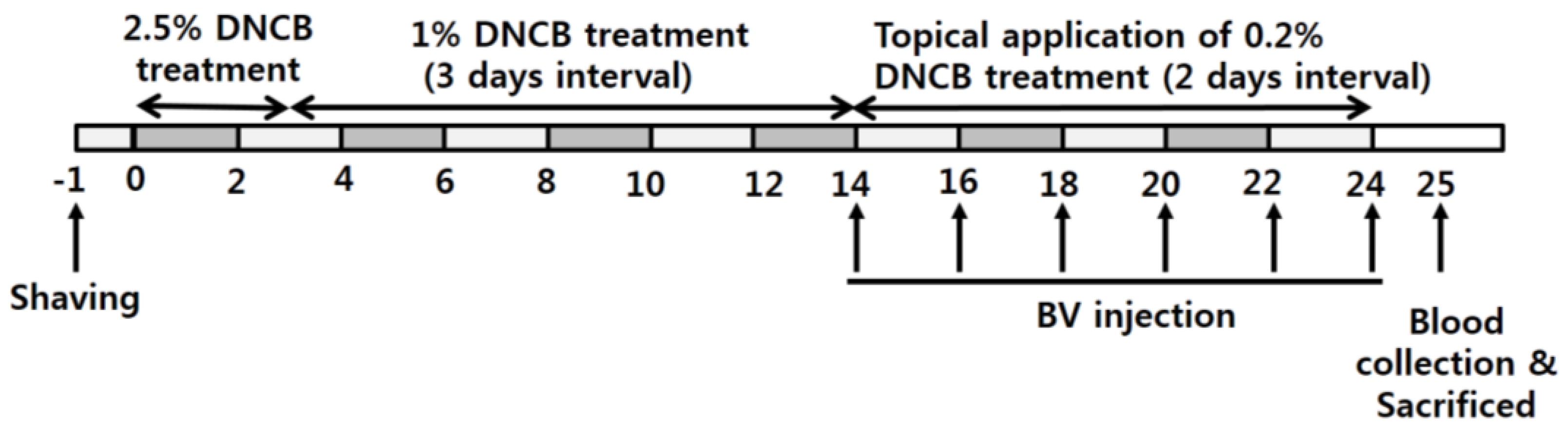
4.6. Development of Irritant Contact Dermatitis (ICD) Mouse
4.7. Treatment of ICD Mice Using BV
4.8. Clinical Skin Score.
4.9. Enzyme-Linked Immunosorbent Assay (ELISA)
4.10. Complement-Dependent Cytotoxicity Assay
4.11. Bactericidal Assay
4.12. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoyt, J. Literature Review of Bee Venom Therapy: Mechanisms of Action and Selected Therapeutic Uses. Orient. Med. J. 2015, 23, 6–9. [Google Scholar]
- Lariviere, W.R.; Melzack, R. The bee venom test: A new tonic-pain test. Pain 1996, 66, 271–277. [Google Scholar] [CrossRef]
- Lee, G.; Bae, H. Anti-Inflammatory Applications of Melittin, a Major Component of Bee Venom: Detailed Mechanism of Action and Adverse Effects. Molecules 2016, 21, E616. [Google Scholar] [CrossRef]
- Raghuraman, H.; Chattopadhyay, A. Melittin: A membrane-active peptide with diverse functions. Biosci. Rep. 2007, 27, 189–223. [Google Scholar]
- Jung, K.H.; Baek, H.; Kang, M.; Kim, N.; Lee, S.Y.; Bae, H. Bee Venom Phospholipase A2 Ameliorates House Dust Mite Extract Induced Atopic Dermatitis Like Skin Lesions in Mice. Toxins 2017, 9, 68. [Google Scholar] [CrossRef]
- Gittler, J.K.; Shemer, A.; Suárez-Fariñas, M.; Fuentes-Duculan, J.; Gulewicz, K.J.; Wang, C.Q.F.; Mitsui, H.; Cardinale, I.; de Guzman Strong, C.; Krueger, J.G.; et al. Progressive activation of Th2/Th22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis. J. Allergy Clin. Immunol. 2012, 130, 1344–1354. [Google Scholar] [CrossRef]
- Kapp, A.; Wokalek, H.; schöpf, E. Involvement of complement in psoriasis and atopic dermatitis-measurement of C3a and C5a, C3, C4 and C1 inactivator. Arch. Dermatol. Res. 1985, 277, 359–361. [Google Scholar] [CrossRef]
- Kapp, A.; Schöpf, E. Involvement of complement in atopic dermatitis. Acta. Derm. Venereol. Suppl. (Stockh) 1985, 114, 152–154. [Google Scholar]
- Zhuang, Y.; Lyga, J. Inflammaging in skin and other tissues—the roles of complement system and macrophage. Inflamm. Allergy Drug Targets 2014, 13, 153–161. [Google Scholar] [CrossRef]
- Dang, L.; He, L.; Wang, Y.; Xiong, J.; Bai, B.; Li, Y. Role of the complement anaphylatoxin C5a-receptor pathway in atopic dermatitis in mice. Mol. Med. Rep. 2015, 11, 4183–4189. [Google Scholar] [CrossRef]
- Noris, M.; Remuzzi, G. Overview of Complement Activation and Regulation. Semin. Nephrol. 2013, 33, 479–492. [Google Scholar] [CrossRef] [PubMed]
- Son, D.J.; Lee, J.W.; Lee, Y.H.; Song, H.S.; Lee, C.K.; Hong, J.T. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Parmacol. Ther. 2007, 115, 246–270. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Kim, W.H.; An, H.J.; Kim, J.Y.; Gwon, M.G.; Han, S.M.; Leem, J.; Park, K.K. Therapeutic effects of bee venom on experimental atopic dermatitis. Mol. Med. Rep. 2018, 18, 3711–3718. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y.; Guttman-Yassky, E. Deciphering the complexities of atopic dermatitis: Shifting paradigms in treatment approaches. J. Allergy Clin. Immunol. 2014, 134, 769–779. [Google Scholar] [CrossRef]
- Schlapbach, C.; Simon, D. Update on skin allergy. Allergy 2014, 69, 1571–1581. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.J.; Kim, M.; Randy, A.; Nam, E.J.; Nho, C.W. Effects of Hovenia dulcis Thunb. extract and methyl vanillate on atopic dermatitis-like skin lesions and TNF-α/IFN-γ-induced chemokines production in HaCaT cells. J. Pharm. Pharmacol. 2016, 68, 1465–1479. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The development of allergic inflammation. Nature 2008, 454, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Owen, C.E. Immunoglobulin E: Role in asthma and allergic disease: Lessons from the clinic. Pharmacol. Ther. 2007, 113, 121–133. [Google Scholar] [CrossRef]
- Killick, J.; Morisse, G.; Sieger, D.; Astier, A.L. Complement as a regulator of adaptive immunity. Semin. Immunopathol. 2018, 40, 37–48. [Google Scholar] [CrossRef]
- Tsokos, G.C.; Fleming, S.D. Autoimmunity, complement activation, tissue injury and reciprocal effects. Curr. Dir. Autoimmun. 2004, 7, 149–164. [Google Scholar]
- Markiewski, M.M.; Lambris, J.D. The Role of Complement in Inflammatory Diseases From Behind the Scenes into the Spotlight. Am. J. Pathol. 2007, 171, 715–727. [Google Scholar] [CrossRef]
- Sales, T.A.; Marcussi, S.; da Cunha, E.F.F.; Kuca, K.; Ramalho, T.C. Can Inhibitors of Snake Venom Phospholipases A₂Lead to New Insights into Anti-Inflammatory Therapy in Humans? A Theoretical Study. Toxins 2017, 9, 341. [Google Scholar] [CrossRef]
- Harris, J.B.; Scott-Davey, T. Secreted Phospholipases A2 of Snake Venoms: Effects on the Peripheral Neuromuscular System with Comments on the Role of Phospholipases A2 in Disorders of the CNS and Their Uses in Industry. Toxins 2013, 5, 2533–2571. [Google Scholar] [CrossRef]
- Doo, K.H.; Lee, J.H.; Cho, S.Y.; Jung, W.S.; Moon, S.K.; Park, J.M.; Ko, C.N.; Kim, H.; Park, H.J.; Park, S.U. A Prospective Open-Label Study of Combined Treatment for Idiopathic Parkinson’s Disease Using Acupuncture and Bee Venom Acupuncture as an Adjunctive Treatment. J. Altern. Complement Med. 2015, 21, 598–603. [Google Scholar] [CrossRef]
- Lee, J.H.; Li, D.X.; Yoon, H.; Go, D.; Quan, F.S.; Min, B.I.; Kim, S.K. Serotonergic mechanism of the relieving effect of bee venom acupuncture on oxaliplatin-induced neuropathic cold allodynia in rats. BMC Complement Altern. Med. 2014, 14, 471. [Google Scholar] [CrossRef]
- Lim, B.S.; Moon, H.J.; Li, D.X.; Gil, M.; Min, J.K.; Lee, G.; Bae, H.; Kim, S.K.; Min, B.I. Effect of bee venom acupuncture on oxaliplatin-induced cold allodynia in rats. Evid. Based Complement Alternat. Med. 2013, 2013, 369324. [Google Scholar] [CrossRef]
- Lee, S.M.; Yang, E.J.; Choi, S.M.; Kim, S.H.; Baek, M.G.; Jiang, J.H. Effects of bee venom on glutamate-induced toxicity in neuronal and glial cells. Evid. Based Complement Alternat. Med. 2012, 2012, 368196. [Google Scholar] [CrossRef]
- Mingomataj, E.C.; Bakiri, A.H. Episodic hemorrhage during honeybee venom anaphylaxis: Potential mechanisms. J. Investig. Allergol. Clin. Immunol. 2012, 22, 237–244. [Google Scholar]
- Shaldoum, F.M.; Hassan, M.I.; Hassan, M.S. Natural Honey Bee venom Manipulates Human Immune Response. Egypt J. Hosp. Med. 2018, 72, 4252–4258. [Google Scholar]
- Janeway, C.A., Jr.; Travers, P.; Walport, M.; Shlomchik, M.J. Immunobiology: The Immune System in Health and Disease, 5th ed.; Garland Science: New York, NY, USA, 2001; Available online: https://www.ncbi.nlm.nih.gov/books/NBK27100/ (accessed on 5 January 2019).
- Park, J.H.; Yim, B.K.; Lee, J.H.; Lee, S.; Kim, T.H. Risk associated with bee venom therapy: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0126971. [Google Scholar]
- Lee, W.R.; Pak, S.C.; Park, K.K. The protective effect of bee venom on fibrosis causing inflammatory diseases. Toxins 2015, 7, 4758–4772. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.; Lee, Y.-W.; Kim, H.; Chung, D.K. Bee Venom Alleviates Atopic Dermatitis Symptoms through the Upregulation of Decay-Accelerating Factor (DAF/CD55). Toxins 2019, 11, 239. https://doi.org/10.3390/toxins11050239
Kim Y, Lee Y-W, Kim H, Chung DK. Bee Venom Alleviates Atopic Dermatitis Symptoms through the Upregulation of Decay-Accelerating Factor (DAF/CD55). Toxins. 2019; 11(5):239. https://doi.org/10.3390/toxins11050239
Chicago/Turabian StyleKim, Yenny, Youn-Woo Lee, Hangeun Kim, and Dae Kyun Chung. 2019. "Bee Venom Alleviates Atopic Dermatitis Symptoms through the Upregulation of Decay-Accelerating Factor (DAF/CD55)" Toxins 11, no. 5: 239. https://doi.org/10.3390/toxins11050239
APA StyleKim, Y., Lee, Y.-W., Kim, H., & Chung, D. K. (2019). Bee Venom Alleviates Atopic Dermatitis Symptoms through the Upregulation of Decay-Accelerating Factor (DAF/CD55). Toxins, 11(5), 239. https://doi.org/10.3390/toxins11050239



