Assessment of Toxic Effects of Ochratoxin A in Human Embryonic Stem Cells
Abstract
1. Introduction
2. Results
2.1. Ochratoxin A Reduces the Viability and Decreases the Cellular Proliferation of Human Embryonic Stem Cells (hESCs)
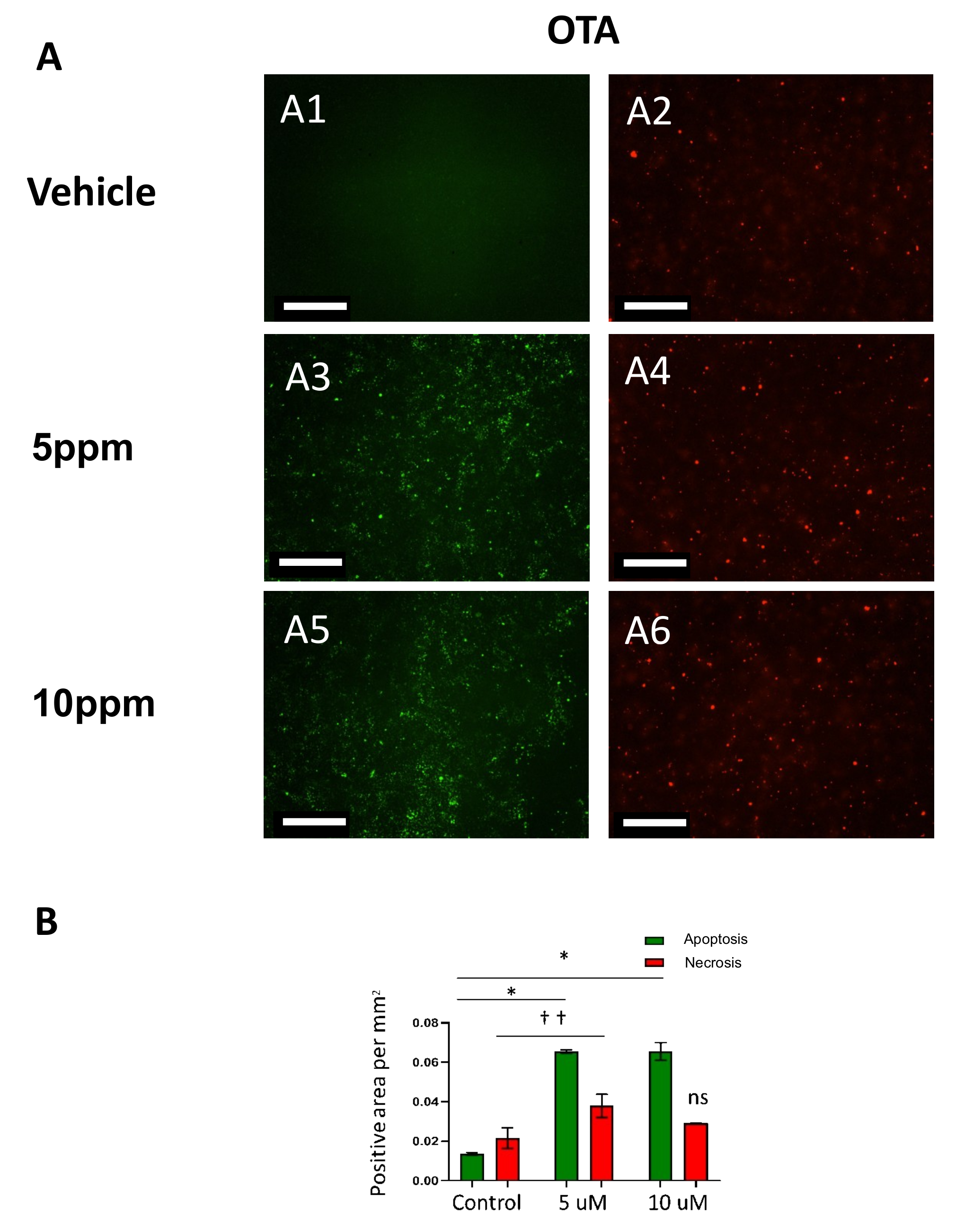
2.2. Ochratoxin A (OTA) Induces Caspase-Mediated Apoptosis in hESCs
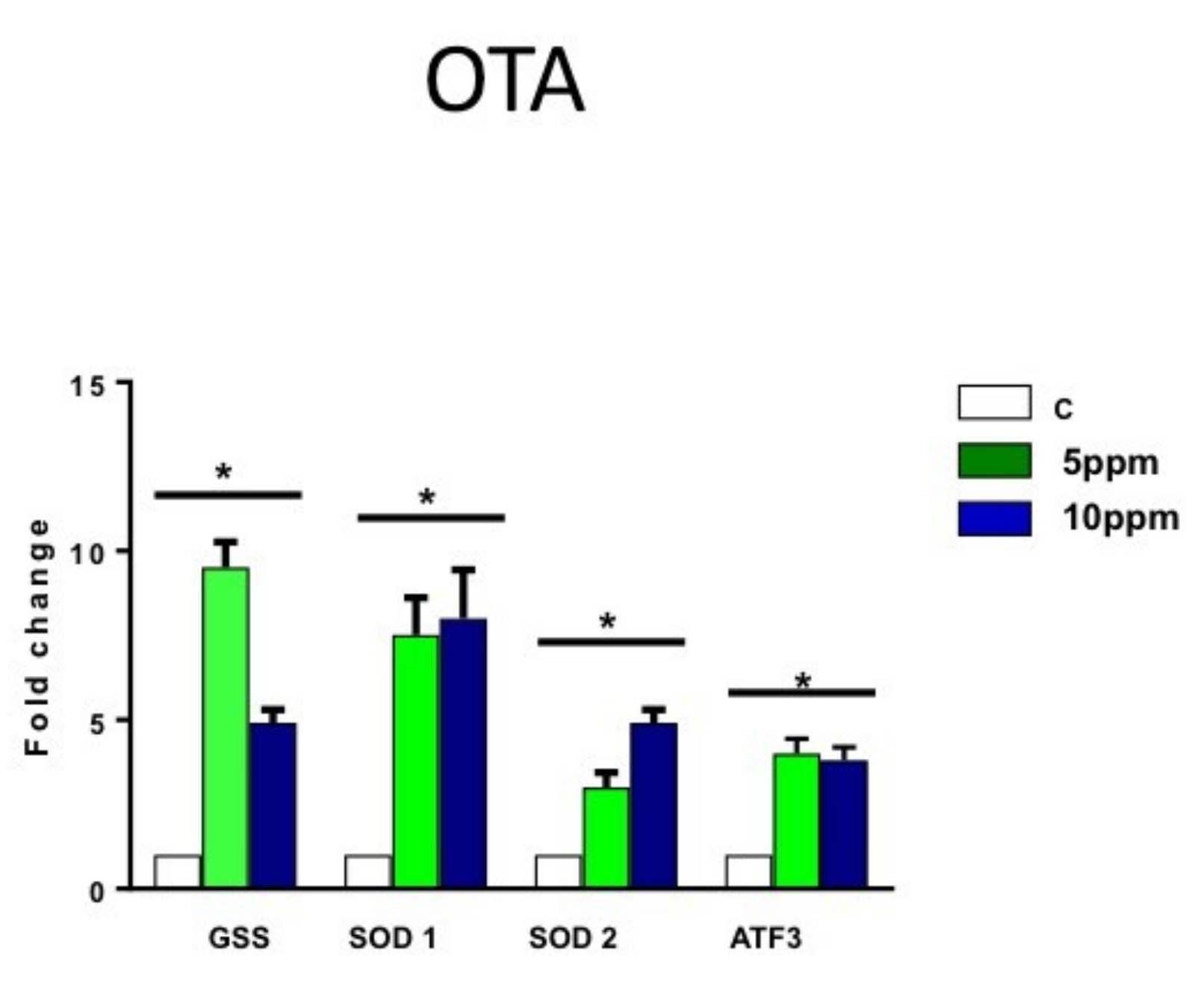
2.3. OTA Increases the Expression of Oxygen Stress Markers in hESCs
3. Discussion
4. Materials and Methods
4.1. Undifferentiated hESC Line Maintenance
4.2. In vitro Culture of hESCs
4.3. Analysis of Cellular Viability
4.4. hESC Compound Exposures
4.5. Toxin Preparation
4.6. RNA Extraction and Reverse Transcriptase Polymerase Chain Reaction (RT-PCR and qRT-PCR)
4.7. Cytotoxicity and Apoptosis Assays
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef] [PubMed]
- Shinde, V.; Klima, S.; Sureshkumar, P.S.; Meganathan, K.; Jagtap, S.; Rempel, E.; Rahnenfuhrer, J.; Hengstler, J.G.; Waldmann, T.; Hescheler, J.; et al. Human Pluripotent Stem Cell Based Developmental Toxicity Assays for Chemical Safety Screening and Systems Biology Data Generation. J. Vis. Exp. 2015, 100, e52333. [Google Scholar] [CrossRef]
- Sokolov, M.V.; Neumann, R.D. Changes in human pluripotent stem cell gene expression after genotoxic stress exposures. World J. Stem Cells 2014, 6, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.O.; Svendsen, A.; Smedsgaard, J. Biochemical characterization of ochratoxin A-producing strains of the genus Penicillium. Appl. Environ. Microbiol. 2001, 67, 3630–3635. [Google Scholar] [CrossRef] [PubMed]
- Medina, A.; Mateo, E.M.; Roig, R.J.; Blanquer, A.; Jimenez, M. Ochratoxin A levels in the plasma of healthy blood donors from Valencia and estimation of exposure degree: comparison with previous national Spanish data. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2010, 27, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Van der Merwe, K.J.; Steyn, P.S.; Fourie, L.; Scott, D.B.; Theron, J.J. Ochratoxin A, a toxic metabolite produced by Aspergillus ochraceus Wilh. Nature 1965, 205, 1112–1113. [Google Scholar] [CrossRef] [PubMed]
- Abarca, M.L.; Bragulat, M.R.; Castella, G.; Cabanes, F.J. Ochratoxin A production by strains of Aspergillus niger var. niger. Appl. Environ. Microbiol. 1994, 60, 2650–2652. [Google Scholar] [PubMed]
- Todescato, F.; Antognoli, A.; Meneghello, A.; Cretaio, E.; Signorini, R.; Bozio, R. Sensitive detection of Ochratoxin A in food and drinks using metal-enhanced fluorescence. Biosens. Bioelectron. 2014, 57, 125–132. [Google Scholar] [CrossRef]
- Arora, R.G.; Frolen, H.; Fellner-Feldegg, H. Inhibition of ochratoxin A teratogenesis by zearalenone and diethylstilboestrol. Food Chem. Toxicol. 1983, 21, 779–783. [Google Scholar] [CrossRef]
- Haubeck, H.D.; Lorkowski, G.; Kolsch, E.; Roschenthaler, R. Immunosuppression by ochratoxin A and its prevention by phenylalanine. Appl. Environ. Microbiol. 1981, 41, 1040–1042. [Google Scholar]
- Sava, V.; Reunova, O.; Velasquez, A.; Sanchez-Ramos, J. Can low level exposure to ochratoxin-A cause parkinsonism? J. Neurol. Sci. 2006, 249, 68–75. [Google Scholar] [CrossRef]
- Wilk-Zasadna, I.; Minta, M. Developmental toxicity of Ochratoxin A in rat embryo midbrain micromass cultures. Int. J. Mol. Sci. 2009, 10, 37–49. [Google Scholar] [CrossRef]
- Razafimanjato, H.; Garmy, N.; Guo, X.J.; Varini, K.; Di Scala, C.; Di Pasquale, E.; Taieb, N.; Maresca, M. The food-associated fungal neurotoxin ochratoxin A inhibits the absorption of glutamate by astrocytes through a decrease in cell surface expression of the excitatory amino-acid transporters GLAST and GLT-1. Neurotoxicology 2010, 31, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Pfohl-Leszkowicz, A.; Manderville, R.A. Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef]
- Torovic, L. Aflatoxins and ochratoxin A in flour: A survey of the Serbian retail market. Food Addit. Contam. Part B 2018, 11, 26–32. [Google Scholar] [CrossRef]
- Clarke, R.; Connolly, L.; Frizzell, C.; Elliott, C.T. Challenging conventional risk assessment with respect to human exposure to multiple food contaminants in food: A case study using maize. Toxicol. Lett. 2015, 238, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Jilani, K.; Lupescu, A.; Zbidah, M.; Abed, M.; Shaik, N.; Lang, F. Enhanced apoptotic death of erythrocytes induced by the mycotoxin ochratoxin A. Kidney Blood Press Res. 2012, 36, 107–118. [Google Scholar] [CrossRef]
- Coronel, M.B.; Sanchis, V.; Ramos, A.J.; Marin, S. Review. Ochratoxin A: presence in human plasma and intake estimation. Food Sci. Technol. Int. 2010, 16, 5–18. [Google Scholar] [CrossRef]
- Hope, J.H.; Hope, B.E. A review of the diagnosis and treatment of Ochratoxin A inhalational exposure associated with human illness and kidney disease including focal segmental glomerulosclerosis. J. Environ. Public Health 2012, 2012, 835059. [Google Scholar] [CrossRef]
- Petkova-Bocharova, T.; Castegnaro, M.; Michelon, J.; Maru, V. Ochratoxin A and other mycotoxins in cereals from an area of Balkan endemic nephropathy and urinary tract tumours in Bulgaria. IARC Sci. Publ. 1991, 115, 83–87. [Google Scholar]
- Belmadani, A.; Tramu, G.; Betbeder, A.M.; Creppy, E.E. Subchronic effects of ochratoxin A on young adult rat brain and partial prevention by aspartame, a sweetener. Hum. Exp. Toxicol. 1998, 17, 380–386. [Google Scholar] [CrossRef]
- Paradells, S.; Rocamonde, B.; Llinares, C.; Herranz-Perez, V.; Jimenez, M.; Garcia-Verdugo, J.M.; Zipancic, I.; Soria, J.M.; Garcia-Esparza, M.A. Neurotoxic effects of ochratoxin A on the subventricular zone of adult mouse brain. J. Appl. Toxicol. 2015, 35, 737–751. [Google Scholar] [CrossRef]
- Talbot, P. In vitro assessment of reproductive toxicity of tobacco smoke and its constituents. Birth Defects Res. C Embryo Today 2008, 84, 61–72. [Google Scholar] [CrossRef]
- Hayes, A.W.; Hood, R.D.; Lee, H.L. Teratogenic effects of ochratoxin A in mice. Teratology 1974, 9, 93–97. [Google Scholar] [CrossRef]
- Wangikar, P.B.; Dwivedi, P.; Sharma, A.K.; Sinha, N. Effect in rats of simultaneous prenatal exposure to ochratoxin A and aflatoxin B1. II. Histopathological features of teratological anomalies induced in fetuses. Birth Defects Res. B Dev. Reprod. Toxicol. 2004, 71, 352–358. [Google Scholar] [CrossRef]
- Hardy, K. Cell death in the mammalian blastocyst. Mol. Hum. Reprod. 1997, 3, 919–925. [Google Scholar] [CrossRef]
- Chan, W.H. Impact of genistein on maturation of mouse oocytes, fertilization, and fetal development. Reprod. Toxicol. 2009, 28, 52–58. [Google Scholar] [CrossRef]
- Chan, W.H. Effects of citrinin on maturation of mouse oocytes, fertilization, and fetal development in vitro and in vivo. Toxicol. Lett. 2008, 180, 28–32. [Google Scholar] [CrossRef]
- Klaric, M.S.; Zeljezic, D.; Rumora, L.; Peraica, M.; Pepeljnjak, S.; Domijan, A.M. A potential role of calcium in apoptosis and aberrant chromatin forms in porcine kidney PK15 cells induced by individual and combined ochratoxin A and citrinin. Arch. Toxicol. 2012, 86, 97–107. [Google Scholar] [CrossRef]
- Li, J.; Yin, S.; Dong, Y.; Fan, L.; Hu, H. p53 activation inhibits ochratoxin A-induced apoptosis in monkey and human kidney epithelial cells via suppression of JNK activation. Biochem. Biophys. Res. Commun. 2011, 411, 458–463. [Google Scholar] [CrossRef]
- Sauvant, C.; Holzinger, H.; Gekle, M. Proximal tubular toxicity of ochratoxin A is amplified by simultaneous inhibition of the extracellular signal-regulated kinases 1/2. J. Pharmacol. Exp. Ther. 2005, 313, 234–241. [Google Scholar] [CrossRef]
- Flora, S.J.; Mehta, A. Monoisoamyl dimercaptosuccinic acid abrogates arsenic-induced developmental toxicity in human embryonic stem cell-derived embryoid bodies: Comparison with in vivo studies. Biochem. Pharmacol. 2009, 78, 1340–1349. [Google Scholar] [CrossRef]
- He, X.; Imanishi, S.; Sone, H.; Nagano, R.; Qin, X.Y.; Yoshinaga, J.; Akanuma, H.; Yamane, J.; Fujibuchi, W.; Ohsako, S. Effects of methylmercury exposure on neuronal differentiation of mouse and human embryonic stem cells. Toxicol. Lett. 2012, 212, 1–10. [Google Scholar] [CrossRef]
- Lin, S.; Tran, V.; Talbot, P. Comparison of toxicity of smoke from traditional and harm-reduction cigarettes using mouse embryonic stem cells as a novel model for preimplantation development. Hum. Reprod. 2009, 24, 386–397. [Google Scholar] [CrossRef]
- Lin, S.; Fonteno, S.; Weng, J.H.; Talbot, P. Comparison of the toxicity of smoke from conventional and harm reduction cigarettes using human embryonic stem cells. Toxicol. Sci. 2010, 118, 202–212. [Google Scholar] [CrossRef]
- Meganathan, K.; Jagtap, S.; Wagh, V.; Winkler, J.; Gaspar, J.A.; Hildebrand, D.; Trusch, M.; Lehmann, K.; Hescheler, J.; Schluter, H.; et al. Identification of thalidomide-specific transcriptomics and proteomics signatures during differentiation of human embryonic stem cells. PLoS ONE 2012, 7, e44228. [Google Scholar] [CrossRef]
- Schuldiner, M.; Yanuka, O.; Itskovitz-Eldor, J.; Melton, D.A.; Benvenisty, N. Effects of eight growth factors on the differentiation of cells derived from human embryonic stem cells. Proc. Natl. Acad. Sci. USA 2000, 97, 11307–11312. [Google Scholar] [CrossRef]
- Muller, G.; Rosner, H.; Rohrmann, B.; Erler, W.; Geschwend, G.; Grafe, U.; Burkert, B.; Moller, U.; Diller, R.; Sachse, K.; et al. Effects of the mycotoxin ochratoxin A and some of its metabolites on the human cell line THP-1. Toxicology 2003, 184, 69–82. [Google Scholar] [CrossRef]
- Grandjean, P.; Bellinger, D.; Bergman, A.; Cordier, S.; Davey-Smith, G.; Eskenazi, B.; Gee, D.; Gray, K.; Hanson, M.; van den Hazel, P.; et al. The faroes statement: human health effects of developmental exposure to chemicals in our environment. Basic Clin. Pharmacol. Toxicol. 2008, 102, 73–75. [Google Scholar] [CrossRef]
- Fukui, Y.; Hayasaka, S.; Itoh, M.; Takeuchi, Y. Development of neurons and synapses in ochratoxin A-induced microcephalic mice: A quantitative assessment of somatosensory cortex. Neurotoxicol. Teratol. 1992, 14, 191–196. [Google Scholar] [CrossRef]
- Miki, T.; Fukui, Y.; Uemura, N.; Takeuchi, Y. Regional difference in the neurotoxicity of ochratoxin A on the developing cerebral cortex in mice. Brain Res. Dev. Brain Res. 1994, 82, 259–264. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Bhat, P.V.; Pandareesh, M.; Khanum, F.; Tamatam, A. Cytotoxic Effects of Ochratoxin A in Neuro-2a Cells: Role of Oxidative Stress Evidenced by N-acetylcysteine. Front. Microbiol. 2016, 7, 1142. [Google Scholar] [CrossRef]
- Renzulli, C.; Galvano, F.; Pierdomenico, L.; Speroni, E.; Guerra, M.C. Effects of rosmarinic acid against aflatoxin B1 and ochratoxin-A-induced cell damage in a human hepatoma cell line (Hep G2). J. Appl. Toxicol. 2004, 24, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Hibi, D.; Suzuki, Y.; Ishii, Y.; Jin, M.; Watanabe, M.; Sugita-Konishi, Y.; Yanai, T.; Nohmi, T.; Nishikawa, A.; Umemura, T. Site-specific in vivo mutagenicity in the kidney of gpt delta rats given a carcinogenic dose of ochratoxin A. Toxicol. Sci. 2011, 122, 406–414. [Google Scholar] [CrossRef]
- Palabiyik, S.S.; Erkekoglu, P.; Zeybek, N.D.; Kizilgun, M.; Baydar, D.E.; Sahin, G.; Giray, B.K. Protective effect of lycopene against ochratoxin A induced renal oxidative stress and apoptosis in rats. Exp. Toxicol. Pathol. 2013, 65, 853–861. [Google Scholar] [CrossRef]
- Doi, K.; Uetsuka, K. Mechanisms of mycotoxin-induced neurotoxicity through oxidative stress-associated pathways. Int. J. Mol. Sci. 2011, 12, 5213–5237. [Google Scholar] [CrossRef]
- Van Gurp, M.; Festjens, N.; van Loo, G.; Saelens, X.; Vandenabeele, P. Mitochondrial intermembrane proteins in cell death. Biochem. Biophys. Res. Commun. 2003, 304, 487–497. [Google Scholar] [CrossRef]
- Assaf, H.; Azouri, H.; Pallardy, M. Ochratoxin A induces apoptosis in human lymphocytes through down regulation of Bcl-xL. Toxicol. Sci. 2004, 79, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Darif, Y.; Mountassif, D.; Belkebir, A.; Zaid, Y.; Basu, K.; Mourad, W.; Oudghiri, M. Ochratoxin A mediates MAPK activation, modulates IL-2 and TNF-alpha mRNA expression and induces apoptosis by mitochondria-dependent and mitochondria-independent pathways in human H9 T cells. J. Toxicol. Sci. 2016, 41, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Raghubeer, S.; Nagiah, S.; Chuturgoon, A.A. Acute Ochratoxin A exposure induces inflammation and apoptosis in human embryonic kidney (HEK293) cells. Toxicon 2017, 137, 48–53. [Google Scholar] [CrossRef]
- Joschko, M.A.; Dreosti, I.E.; Tulsi, R.S. The teratogenic effects of nicotine in vitro in rats: a light and electron microscope study. Neurotoxicol. Teratol. 1991, 13, 307–316. [Google Scholar] [CrossRef]
- Zhao, Z.; Reece, E.A. Nicotine-induced embryonic malformations mediated by apoptosis from increasing intracellular calcium and oxidative stress. Birth Defects Res. B Dev. Reprod. Toxicol. 2005, 74, 383–391. [Google Scholar] [CrossRef]
- Seiler, A.E.; Buesen, R.; Visan, A.; Spielmann, H. Use of murine embryonic stem cells in embryotoxicity assays: The embryonic stem cell test. Methods Mol. Biol. 2006, 329, 371–395. [Google Scholar] [CrossRef]
- Yamane, J.; Aburatani, S.; Imanishi, S.; Akanuma, H.; Nagano, R.; Kato, T.; Sone, H.; Ohsako, S.; Fujibuchi, W. Prediction of developmental chemical toxicity based on gene networks of human embryonic stem cells. Nucleic Acids Res. 2016, 44, 5515–5528. [Google Scholar] [CrossRef]
- Aikawa, N.; Kunisato, A.; Nagao, K.; Kusaka, H.; Takaba, K.; Ohgami, K. Detection of thalidomide embryotoxicity by in vitro embryotoxicity testing based on human iPS cells. J. Pharmacol. Sci. 2014, 124, 201–207. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erceg, S.; Mateo, E.M.; Zipancic, I.; Rodríguez Jiménez, F.J.; Pérez Aragó, M.A.; Jiménez, M.; Soria, J.M.; Garcia-Esparza, M.Á. Assessment of Toxic Effects of Ochratoxin A in Human Embryonic Stem Cells. Toxins 2019, 11, 217. https://doi.org/10.3390/toxins11040217
Erceg S, Mateo EM, Zipancic I, Rodríguez Jiménez FJ, Pérez Aragó MA, Jiménez M, Soria JM, Garcia-Esparza MÁ. Assessment of Toxic Effects of Ochratoxin A in Human Embryonic Stem Cells. Toxins. 2019; 11(4):217. https://doi.org/10.3390/toxins11040217
Chicago/Turabian StyleErceg, Slaven, Eva María Mateo, Iván Zipancic, Francisco Javier Rodríguez Jiménez, María Amparo Pérez Aragó, Misericordia Jiménez, José Miguel Soria, and Mª Ángeles Garcia-Esparza. 2019. "Assessment of Toxic Effects of Ochratoxin A in Human Embryonic Stem Cells" Toxins 11, no. 4: 217. https://doi.org/10.3390/toxins11040217
APA StyleErceg, S., Mateo, E. M., Zipancic, I., Rodríguez Jiménez, F. J., Pérez Aragó, M. A., Jiménez, M., Soria, J. M., & Garcia-Esparza, M. Á. (2019). Assessment of Toxic Effects of Ochratoxin A in Human Embryonic Stem Cells. Toxins, 11(4), 217. https://doi.org/10.3390/toxins11040217




