1. Introduction
The implementation of monitoring programs for marine biotoxins in the wake of the findings of Yasumoto [
1,
2], at a scale dependent on the geographical scope of each competent administration, started in Andalusia in 1994. These findings demonstrated a relationship between outbreaks of diarrheal intoxication in humans, the ingestion of molluscs and the presence in the environment of
Dinophysis fortii that was identified as the producer of toxins responsible for poisoning. This led to the monitoring programs to carry out both analysis of the mollusc toxin content and cell counts of the phytoplankton species whose toxicity was proven over time.
The European regulations applicable concerning food security [
3,
4] lay down the basic requirements and recommendations for the implementation of these programs. The program implemented in Andalusia has observed the guidelines established for the definition of areas and the frequency of monitoring. This study presents the data obtained from the start of monitoring and their analysis to answer the following question: Are the regulatory requirements for the analysis of toxic phytoplankton effective as an early-warning system for the risk of contamination in molluscs on the Andalusian coast?
The identification of some species of
Dinophysis has led to much discussion and many scientific publications. The main issues have been reviewed by Reguera et al. [
5]. Identification has been carried out based on morphometric characteristics mainly using light microscopy (LM), but the considerable intraspecific variability makes it difficult to clearly discriminate between some of the species. Nevertheless, LM is the method most used by monitoring programs. In any case, in Andalusia, the set of species given in the IOC Taxonomic Reference List of Toxic Microalgae [
6] has been used as the basis for monitoring, and the region has acted according to the principle of maximum food security in situations of any doubt.
A group of new causative agents of shellfish poisoning, the Diarrhetic Shellfish Toxins (DST), was isolated and their structures determined in the 1980s [
1,
2]. In Andalusia, many cases of DST have been detected during the years of monitoring but not all the toxins in this group have been individually characterized.
Accumulation of the toxicity produced by microalgae in filter-feeders has also been the subject of study. However, important intra- and interspecific variability has been measured resulting from causes such as differences in filtration and clearance rates [
7,
8] or from selection mechanisms based on the quality and quantity of prey [
9,
10,
11]. Such variability has become a parameter considered in the monitoring program.
For more than 20 years, species of the genus Dinophysis have been commonly found in almost all the sampling stations in Andalusia. The species clearly associated with toxicity phenomena in molluscs, have been D. acuminata/D. ovum and D. acuta. The accumulation of toxins above permitted levels triggers the activation of closure protocols for fisheries that can sometimes be prolonged for up to 6 months. All the species of the genus identified in the samples that at some time have formed part of the IOC list, are presented in this study along with toxicity levels of molluscs in each case when this has occurred.
2. Results
The distribution of areas in Andalusia (see
Section 4.1) represents the complete set of mollusc habitats ranging in size between 12.6 ha and the 35340.5 ha (average area 4842.4 ha). Smaller areas correspond to the perimeter of areas where aquaculture facilities are located.
All the information presented in this study comes from the sampling stations located in the above-mentioned areas. In each sampling, the choice of sampling points has been arbitrary within each area, except in areas where physico-chemical measurements of the water were already being made, at previously fixed points. Mollusc samples were usually selected at those points where there was high fishing activity.
2.1. Species of the Genus Dinophysis
Twenty
Dinophysis species out of more than 100 that make up the genus [
12] were identified on the Andalusian coast. Those
Dinophysis cells considered gametes have been excluded from the reported counts.
Dinophysis cells not identified to species level are reported as
Dinophysis spp.
Table 1 provides a species list along with the frequency of occurrence. The number of samples collected in the study period (20 years) was 34,329.
The difficulty in discriminating between
D. acuminata and
D. ovum has led us to follow the example of Lassus and Bardouil [
13] or Bravo [
14] who introduced the term “
Dinophysis acuminata complex” that encompasses both, reflecting its great morphological variability.
The species with the highest frequency were D. acuminata complex, D. caudata, D. acuta and D. fortii.
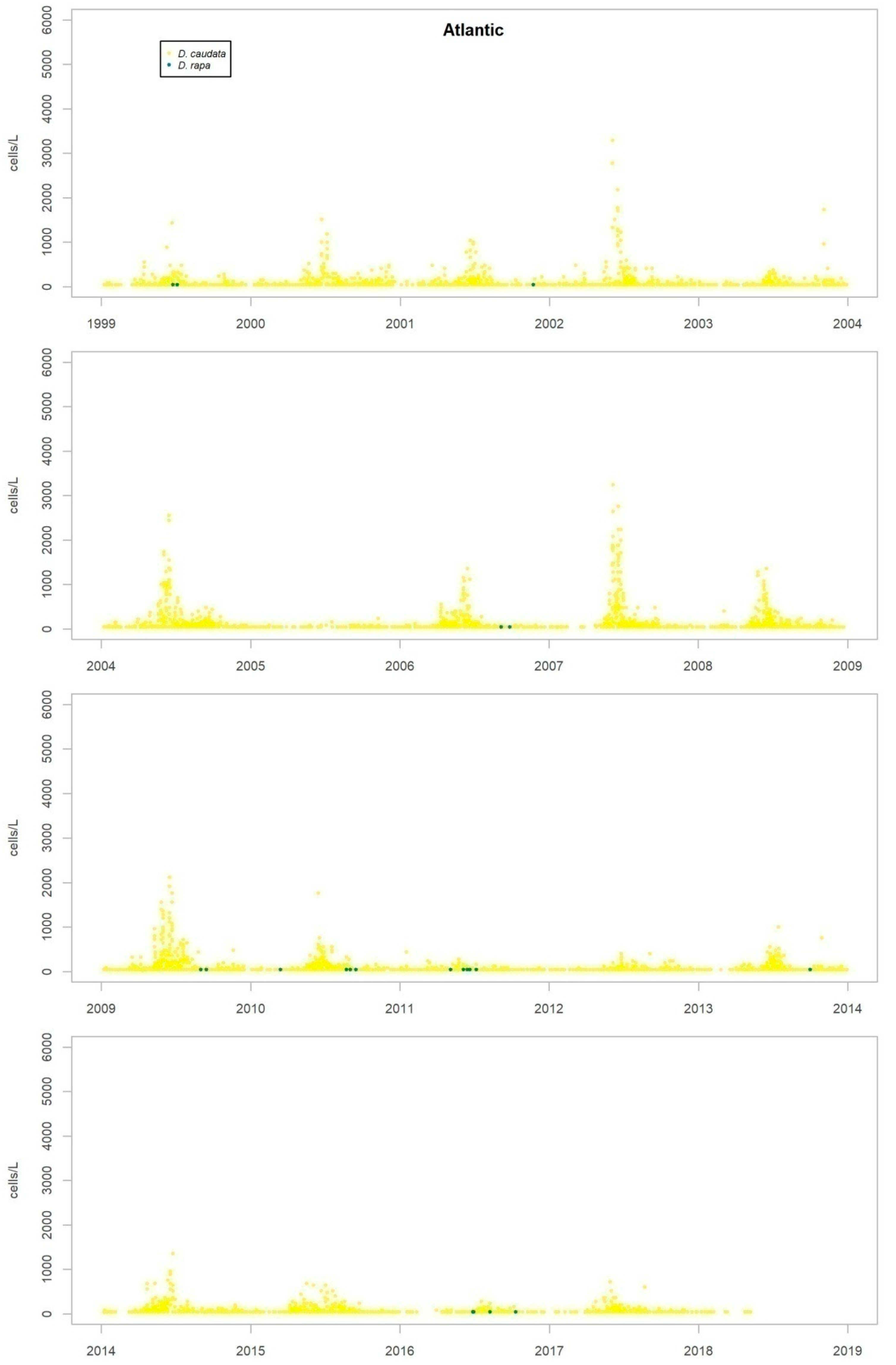
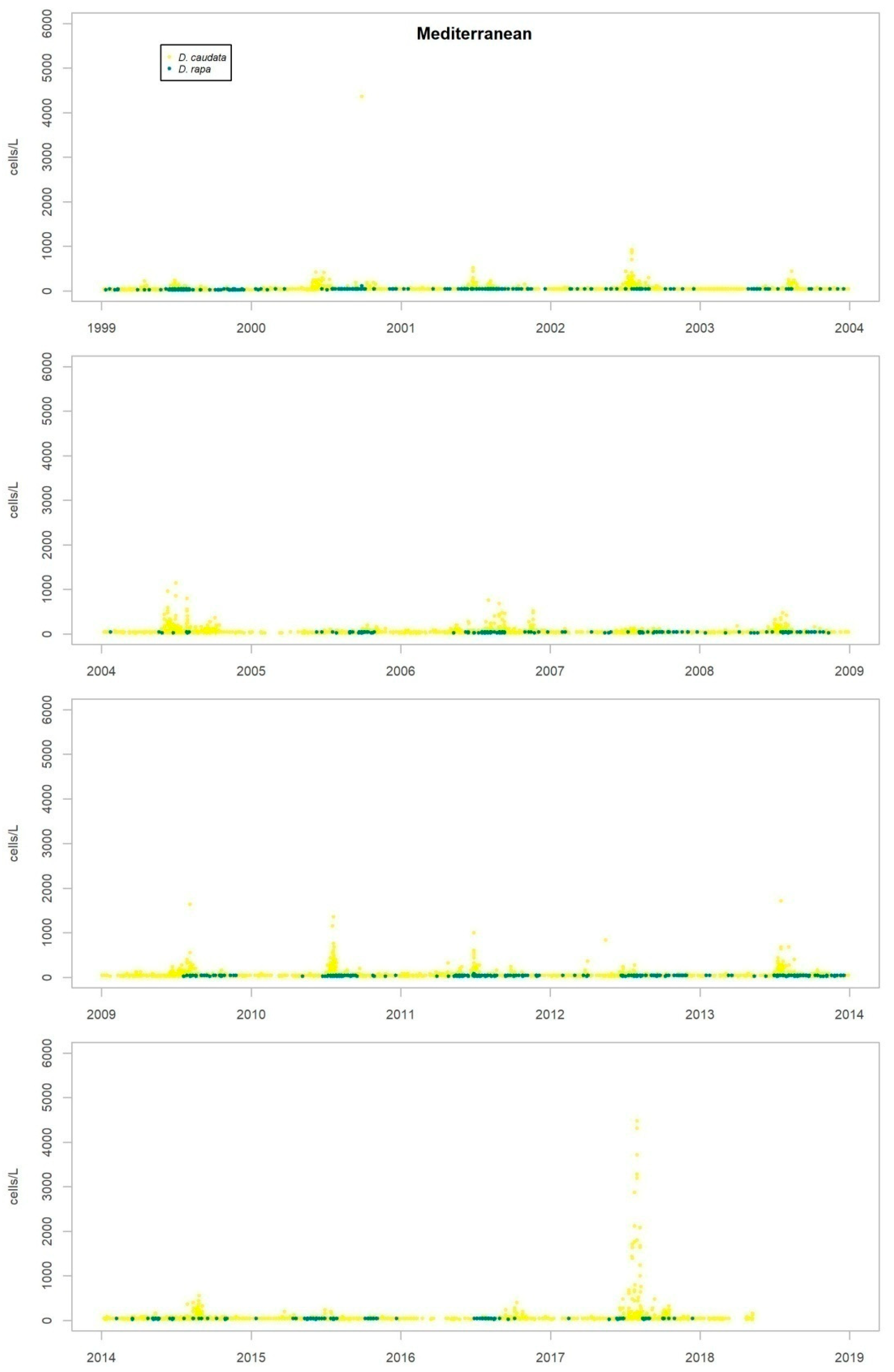
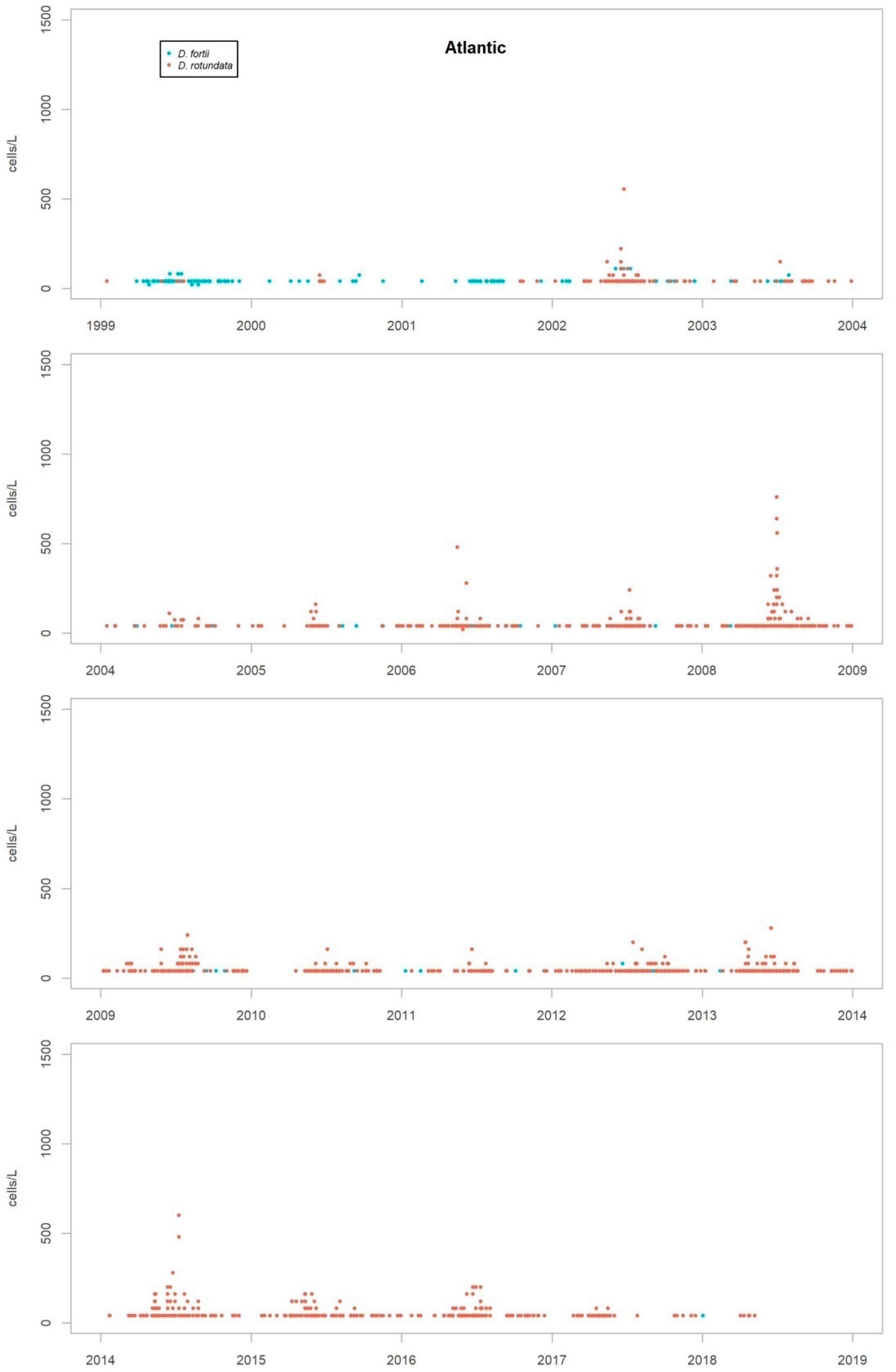
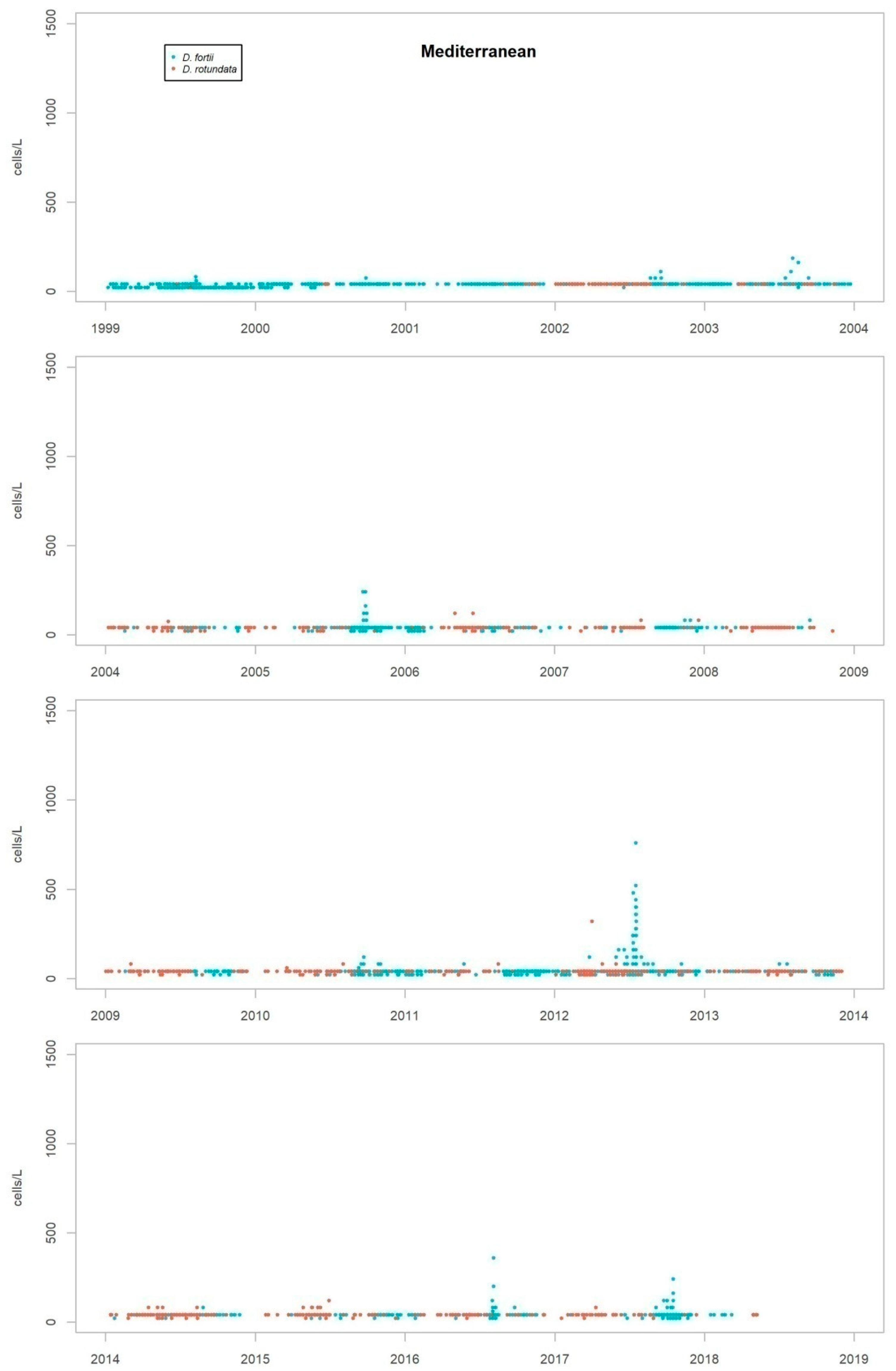
Nearly all the species were detected in both the Atlantic and the Mediterranean areas, but some show marked differences in their repartition between the two areas. While D. acuminata complex, D. acuta and D. rotundata (P. rotundatum) are more frequent on the Atlantic coast, D. fortii and D. rapa (P. rapa) are more frequent in the Mediterranean Sea.
Taking into account that the sampling frequency was uniform all year long, it can be observed that
D. acuminata complex is present almost all throughout the year, although it does not always trigger toxic outbreaks in molluscs (see
Section 2.3).
The couple
D. acuminata complex/
D. caudata is frequent in the samples. In addition, it is accompanied at times by
D. acuta and
D. fortii. Similar associations have already been reported in temperate waters [
5].
The set of species identified as Dinophysis spp. occurs very frequently in both sea areas. Although these species not known to be toxic, unusual high concentrations of some of them activated alert protocols.
2.2. Temporal Episodes of Dinophysis
The volume of data generated during the 20 years of monitoring subject to examination is very high, and data processing is needed to condense the redundancies into manageable form.
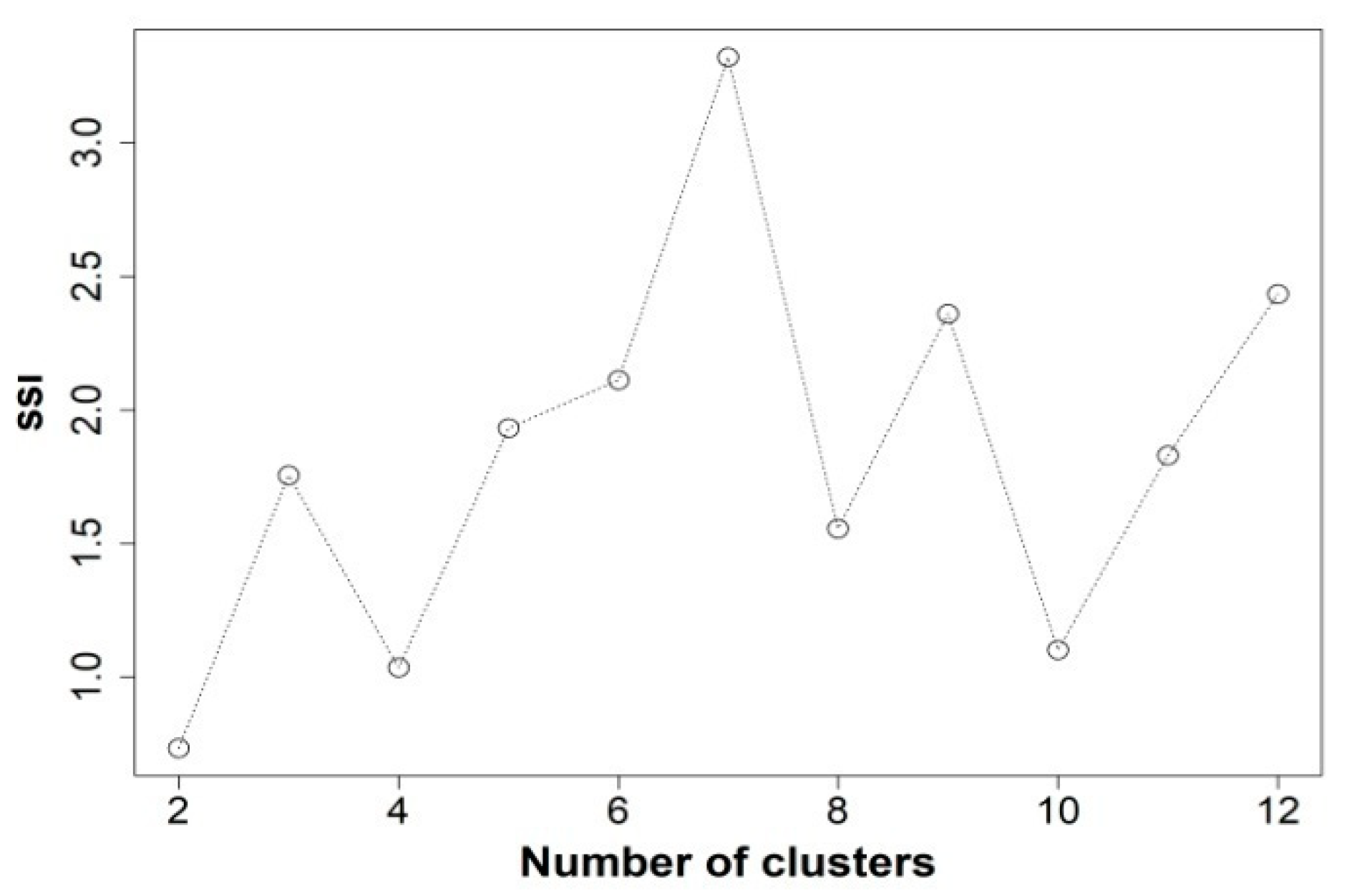
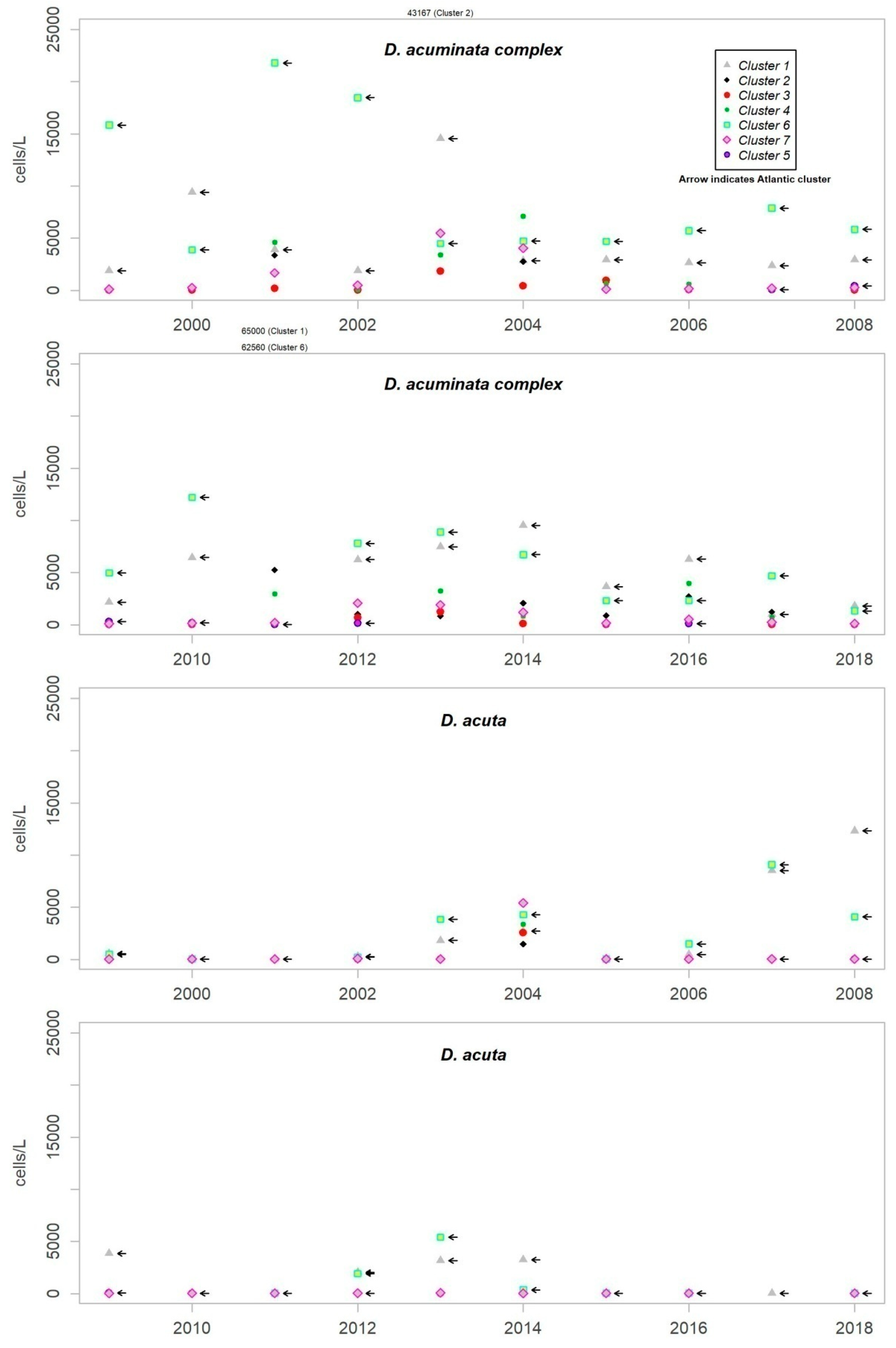
Figure 1 shows the choice of the appropriate number of clusters, to summarize the data, through the highest Simple Structure Index (SSI) [
15]. It shows how the grouping of seven clusters best explains the variance in the data taking up 12 SSI clusters at maximum.
As discussed in the previous section, the most frequent species in the coast of Andalusia are D. acuminata complex, D. caudata, D. acuta and D. fortii. Of these, D. acuminata complex and D. acuta are the only ones which have been partnered by co-occurrence with episodes of DST. Thus, the description of episodes presented in this work will be focused on both species.
Although
D. acuminata complex is present almost all year round, the more substantial episodes are usually produced as a succession of between one and three pulses from the beginning of spring until the end of autumn. The period of growth of
D. acuta is, however, more reduced, from summer to early autumn. Although the pattern mentioned for
D. acuminata complex has been repeated in all the years of monitoring,
D. acuta does not present such a recurrence, and they generally disappear for one or more years. The patterns found show similarity between different years of monitoring, while the abundance reached determines the degree of difference.
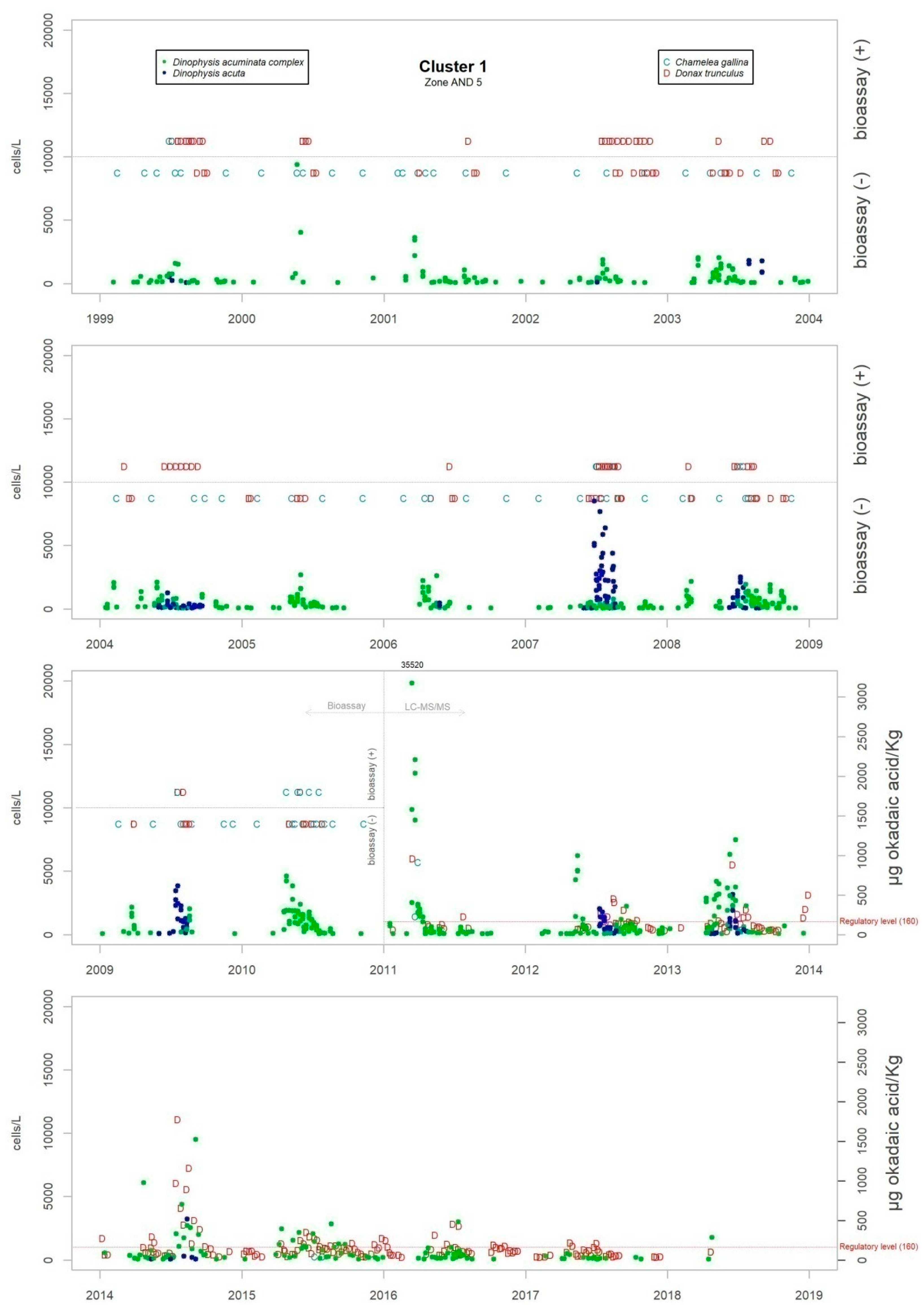
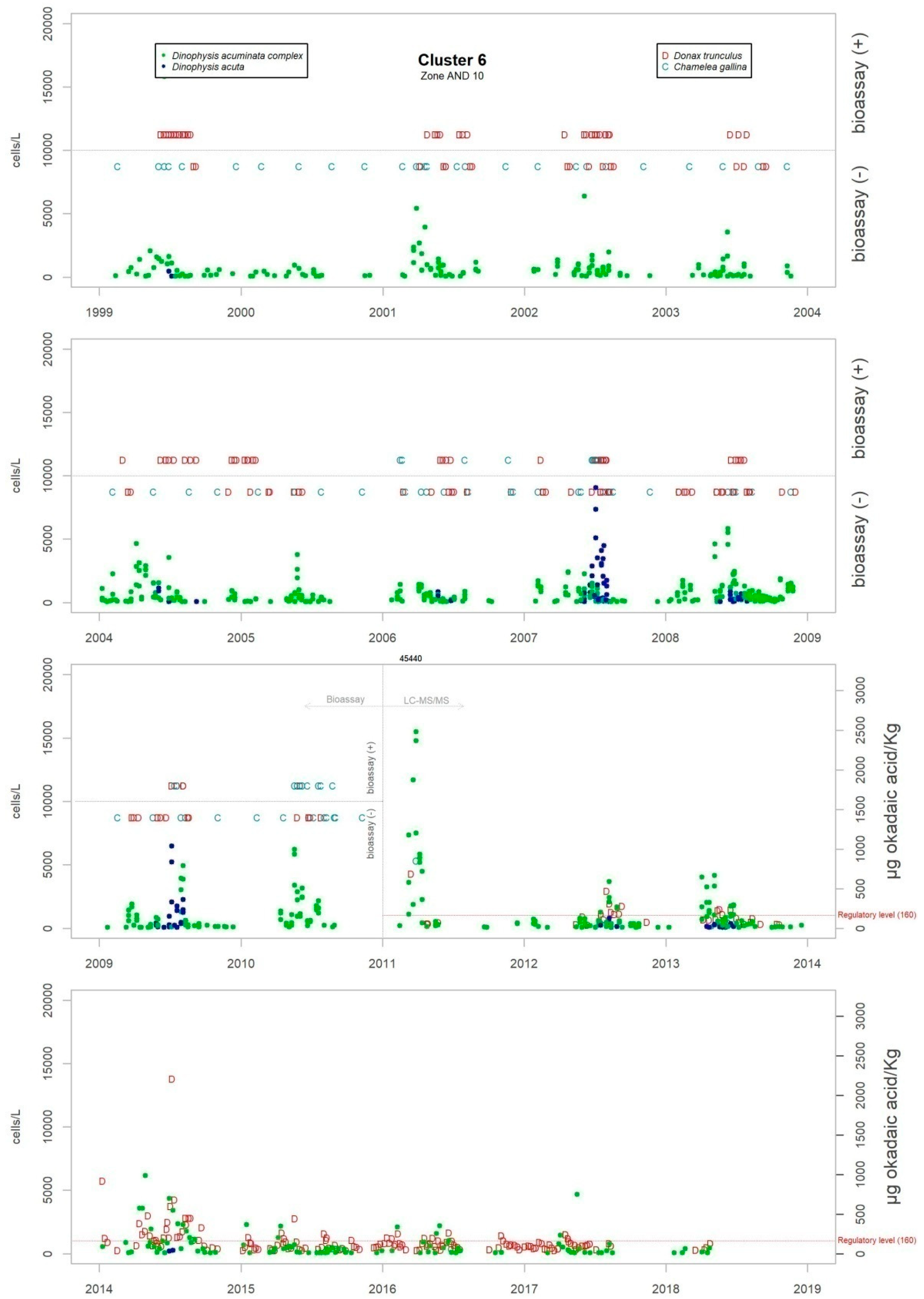
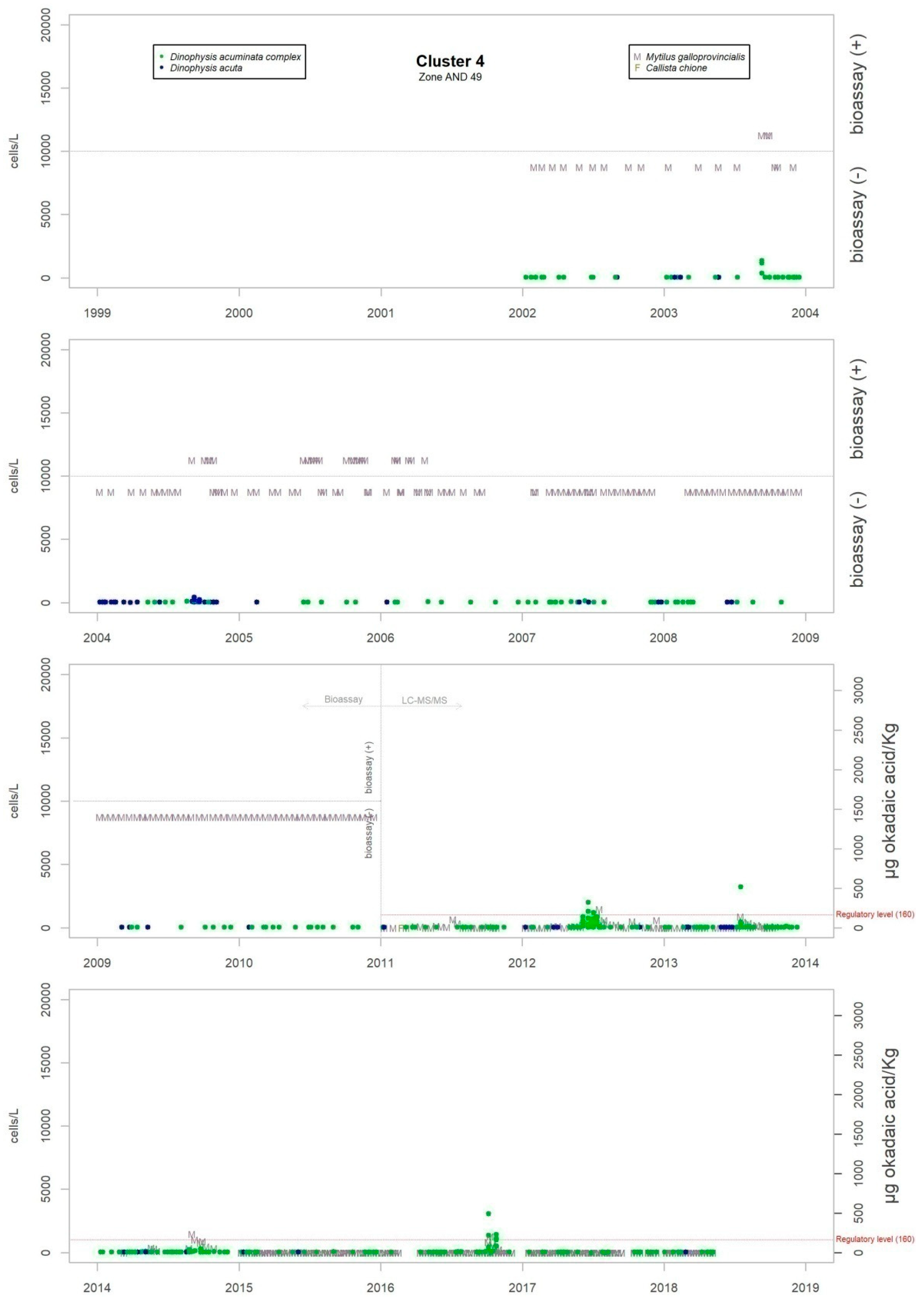
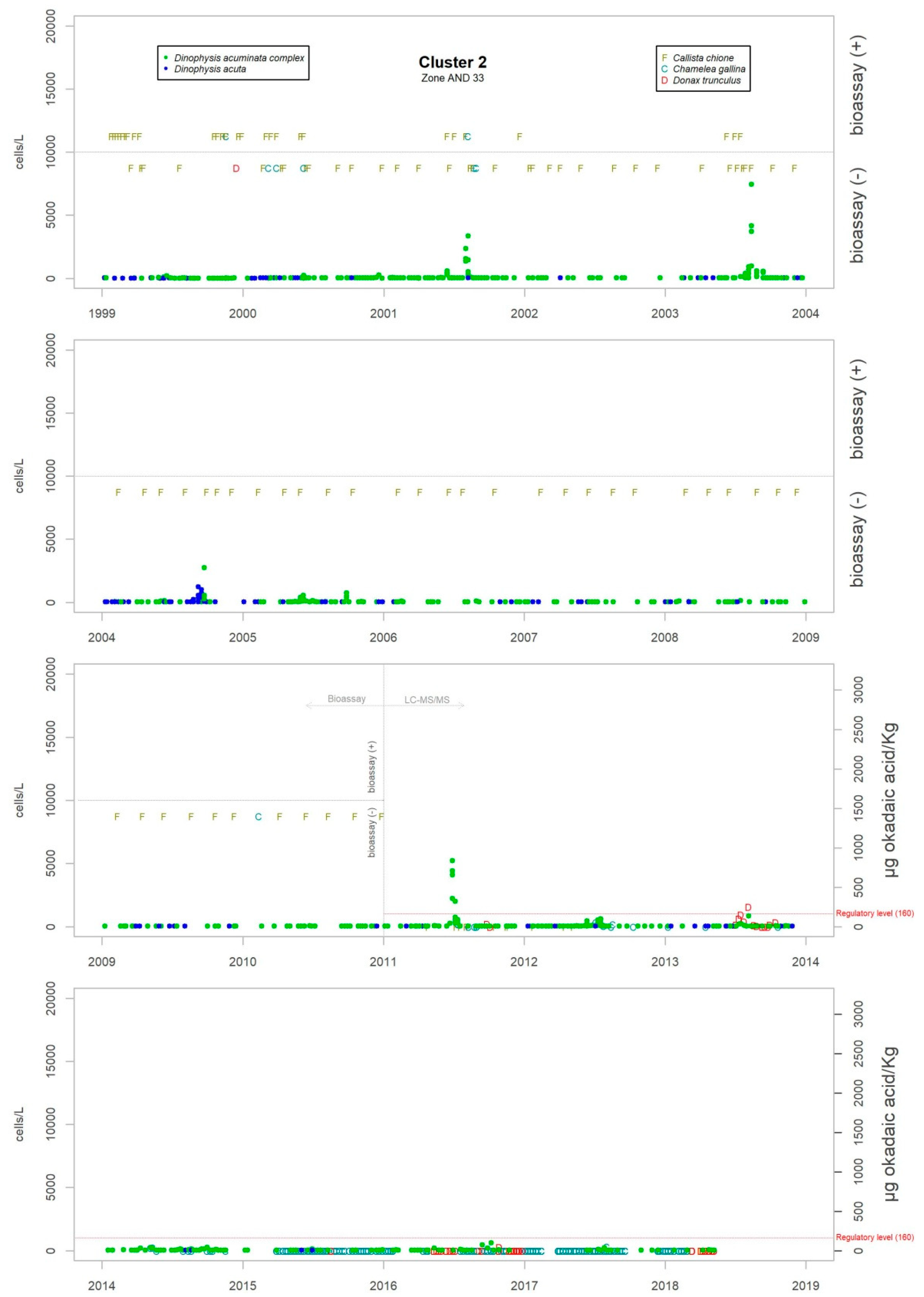
Figure 3 shows the maximum levels of abundance where the difference can be seen.
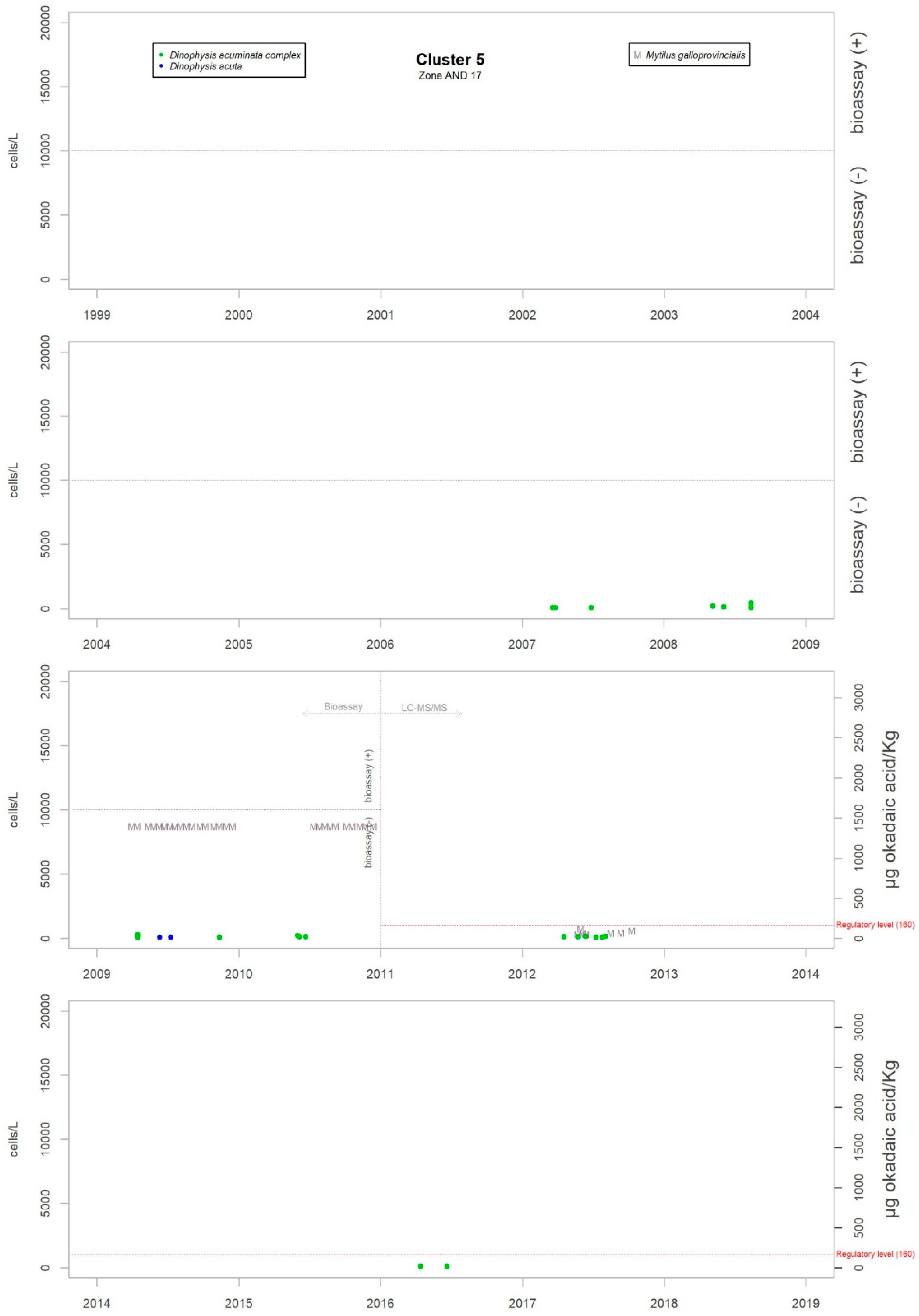
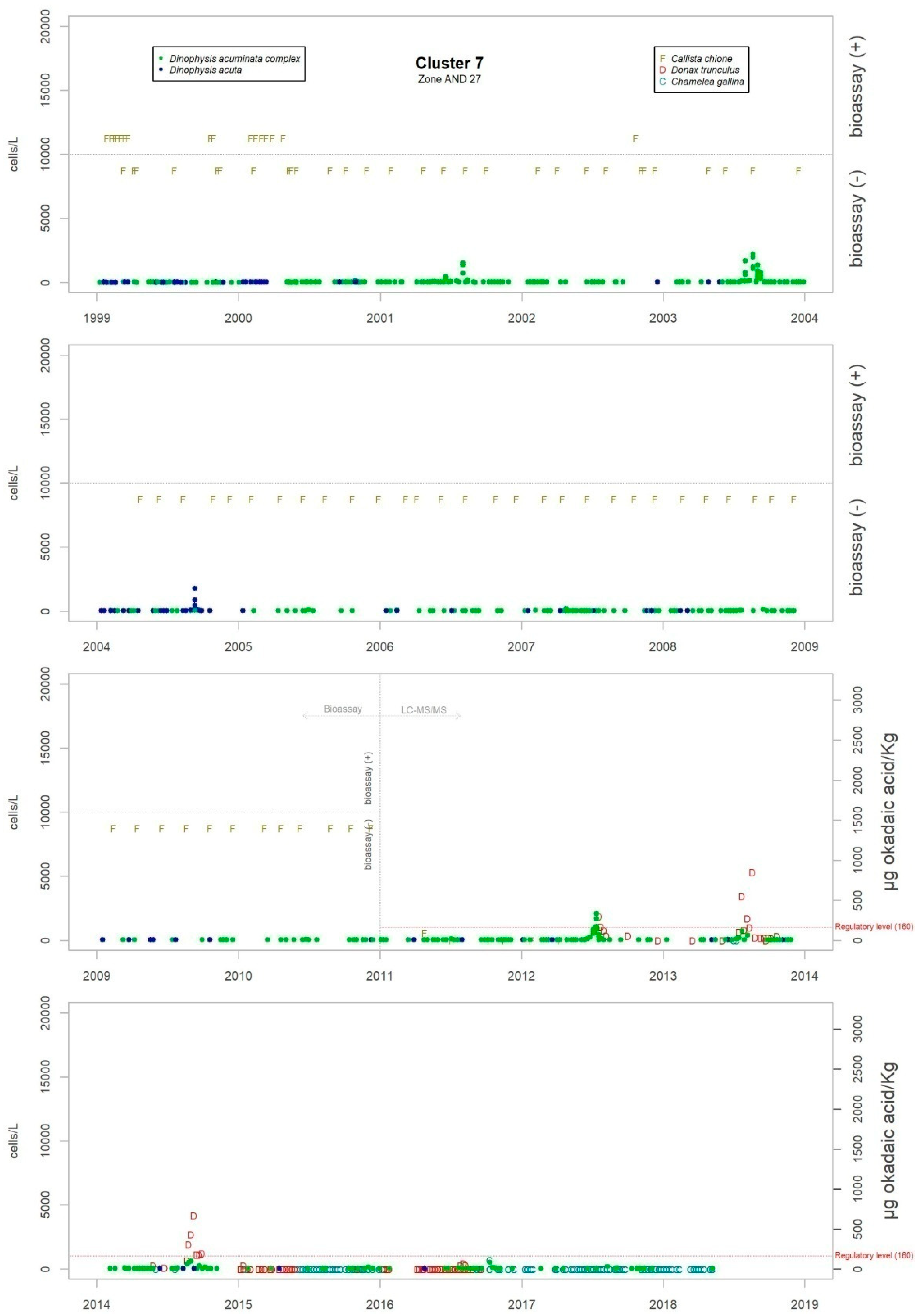
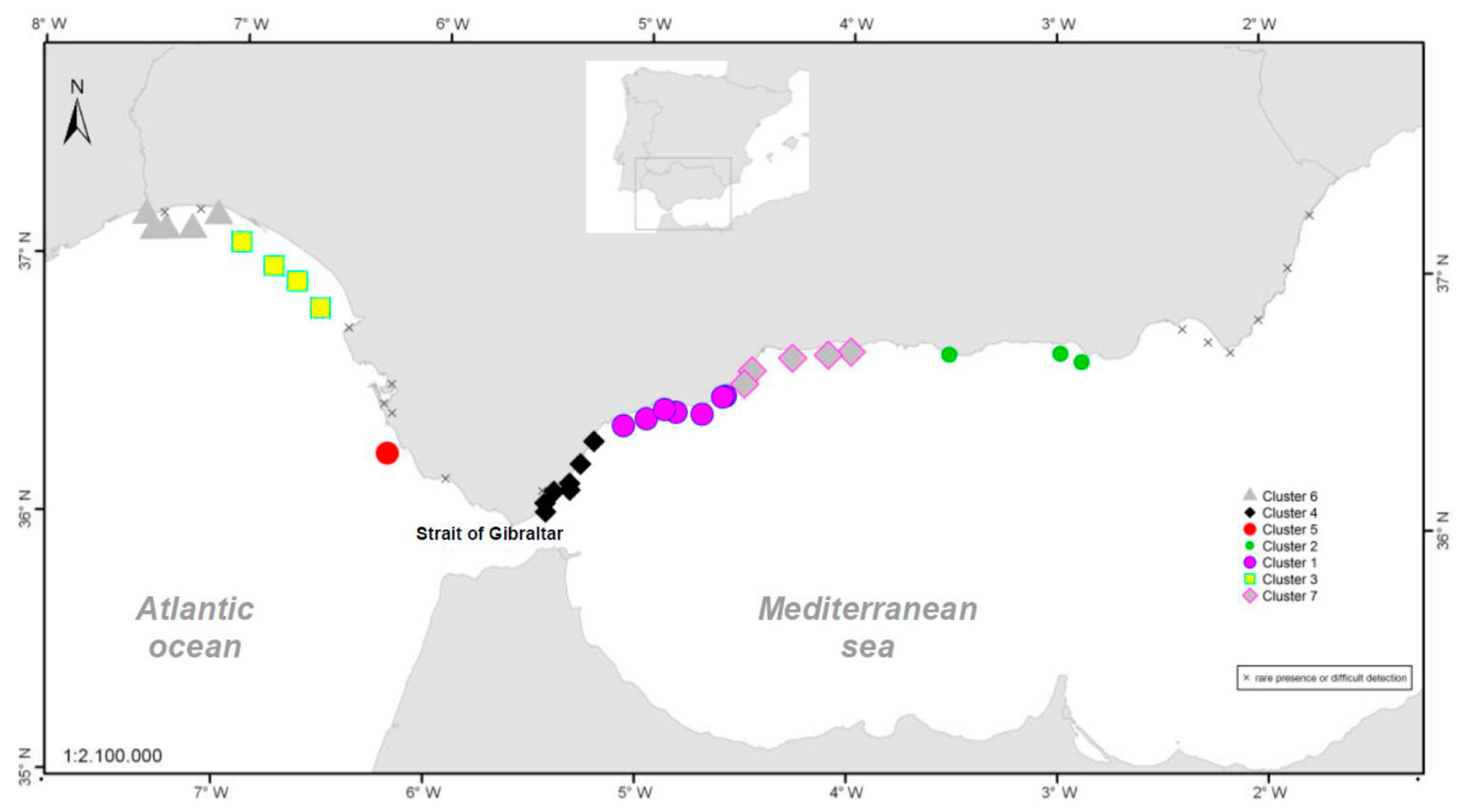
Although the presence of D. acuminata complex and D. acuta in the environment occurs during similar periods all along the coast of Andalusia, in general, there are notable differences in the intensity of the abundance between clusters 1 and 6 (Atlantic area) and the rest. Therefore, although there are frequent blooms of relatively high abundance in the Atlantic area, in the Mediterranean it is rare that either species exceeds 400 cells/l. However, cluster number 5 belongs to the Atlantic area and does not show the behaviour of the other neighbouring Atlantic clusters.
It sometimes happens, however, that a particular episode (characterized by a certain intensity) spreads to almost all the Andalusian coast in one direction or the other. Possible advection through the Strait of Gibraltar is suggested by the proximity of the dates of the growth pulses on both sides of the strait, but which also should be investigated using hydrodynamic models. In August 2001, August 2003, October 2004, July 2012, July 2013 and August 2014, episodes of D. acuminata complex were recorded with these features. D. acuta can be often detected along the entire coast, but not always; in August 2004, for example, a single case of unusually high intensity was recorded in all the areas monitored.
Episodes of special intensity of D. acuta occurred from the end of June to end of August 2007 and of D. acuminata complex from March to mid-April 2011, both in clusters 6 and 1. In the first episode, there were more than 2000 cells/L in 24.9% of the samples and more than 5000 cells/L in 4.7% (349 samples). A maximum value of 9080 cells/L occurred in cluster 6. In the second episode, a maximum of 65,000 cells/L (Cluster 1) was recorded, which was the highest concentration of Dinophysis species found during the years of monitoring. In this case, there were more than 2000 cells/L in 69.4% of the samples and more than 5000 cells/L in 47.9% (278 samples).
From 2014 on, a declining trend of abundance has been recorded for both species towards the western end (clusters 1 and 6). The D. acuminata complex had never shown this behavior (frequency of values above 2000 cells/L was drastically reduced) in the monitoring period.
The timing and abundance of other
Dinophysis species is shown in
Appendix B. None of them shows any obvious seasonal pattern or other type of temporary recurrence. The only condition found is spatial, so that while some species are detected in all areas sampled, others preferably appear in the Atlantic or the Mediterranean areas.
2.3. Toxicity in Molluscs
The criteria of the official monitoring programme aims to categorize some species of bivalve molluscs as indicators of each production area (administrative spatial unit). Therefore, in Andalusia, the species which have been monitored in areas of external waters where there have been major incidents with Dinophysis, have mainly been Donax trunculus, Chamelea gallina, Mytilus galloprovincialis, Callista chione, Venus verrucosa and Cerastoderma edule.
Levels of okadaic acid have been detected in all these areas when D. acuminata complex and/or D. acuta were present in the environment. There have also been cases, albeit few, in which samples showed toxicity but in which Dinophysis species or other organisms considered as DSP producers were not found; there were more cases in the period when the mouse bioassay method was used. Other substances that are part of the group of lipophilic toxins have been detected although on a smaller number of occasions. Levels of DTX-2 began to be measured at the beginning of June 2018 in Donax trunculus and Mytilus galloprovincialis. The episode has not finished yet. In the environment, the presence of mainly Prorocentrum cf. texanum has been accompanied by minor amounts of D. acuminata complex and D. acuta. Also, levels of yessotoxin were measured only in Mytilus galloprovincialis in the spring and summer of 2015 when in the water samples the presence mainly of D. acuminata complex was detected and, to a lesser extent, D. acuta, D. fortii, D. caudata and P. rotundatum.
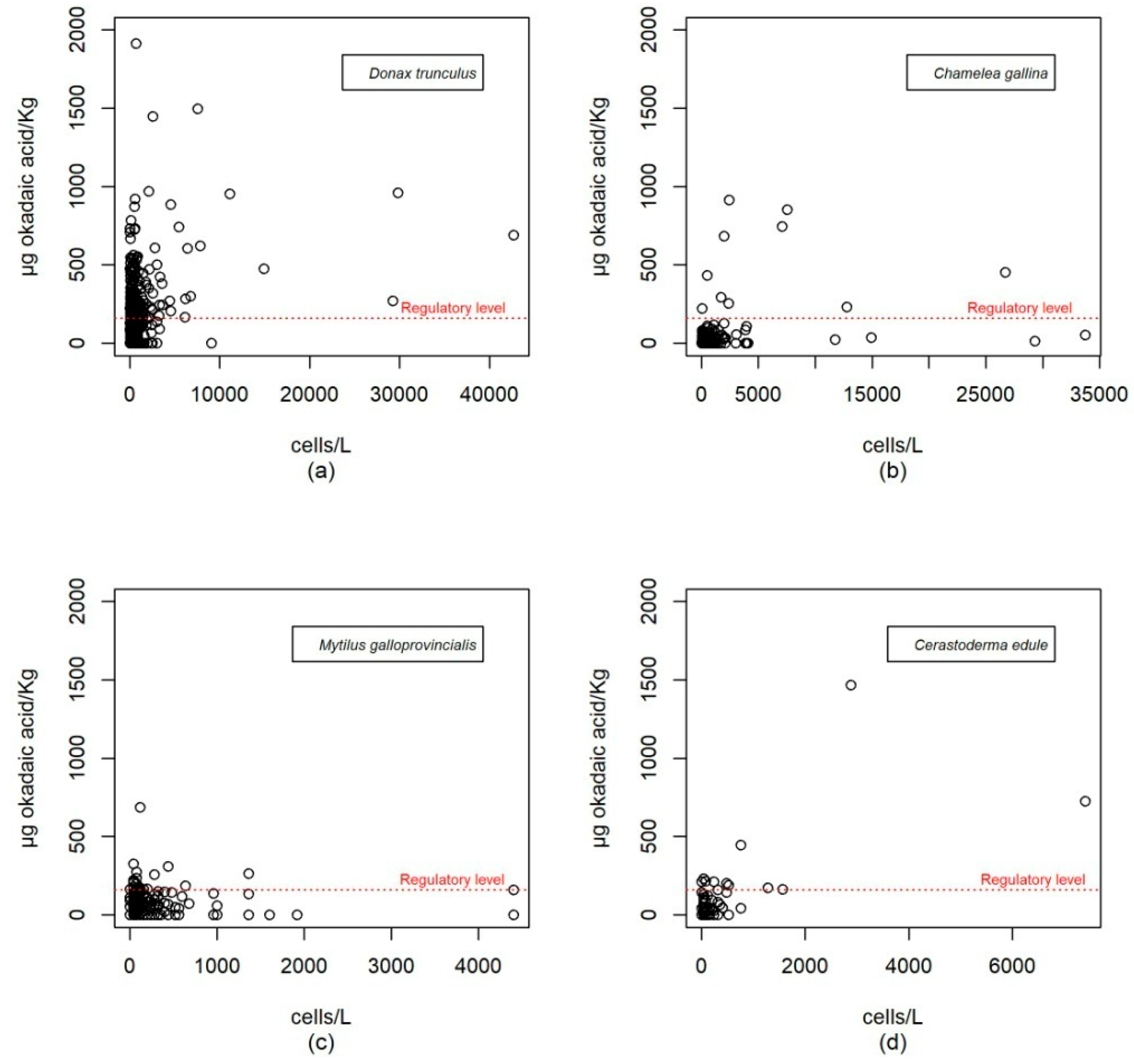
The durations of the episodes usually correspond to the maintenance of the population of that or those phytoplankton species that generate them. There is thus good association between the detection of both parameters in qualitative terms. However, as it can be seen in
Figure 4 which shows the pairs of cell concentration-toxicity values of parallel sampling, the levels of okadaic acid in molluscs do not correlate well with the concentration of the producer microalgae if the expected linear relationships are taken into account. Thus, it is not uncommon to find high values of okadaic acid with low values of phytoplankton and
vice versa.
Although there is not a good correlation between toxicity values in molluscs and the concentration of Dinophysis in parallel sampling, if each episode as a whole is considered, toxicity patterns in molluscs do correspond to the patterns of the producer phytoplankton species as might be expected. Thus, in general, there have been more incidents in the Atlantic area than in the Mediterranean area; episodes have occurred in warm seasons and there has been a trend toward a decrease in measured values over the last three years.
The time series of contamination episodes of bivalve molluscs, together with the abundance of DSP-producing microalgae is shown in
Appendix A. It shows how smaller bivalve species (larger specimens rarely exceed 3–4 cm in their axis of maximum growth) are more prone to the accumulation of lipophilic toxins. In this way, maximum levels of toxicity (µg okadaic acid/kg) were 2665 in
D. trunculus, 913 in
C. gallina and 1468 in
C. edule, whereas in larger species, there were 687 in
M. galloprovincialis, 91 in
C. chione and 28 in
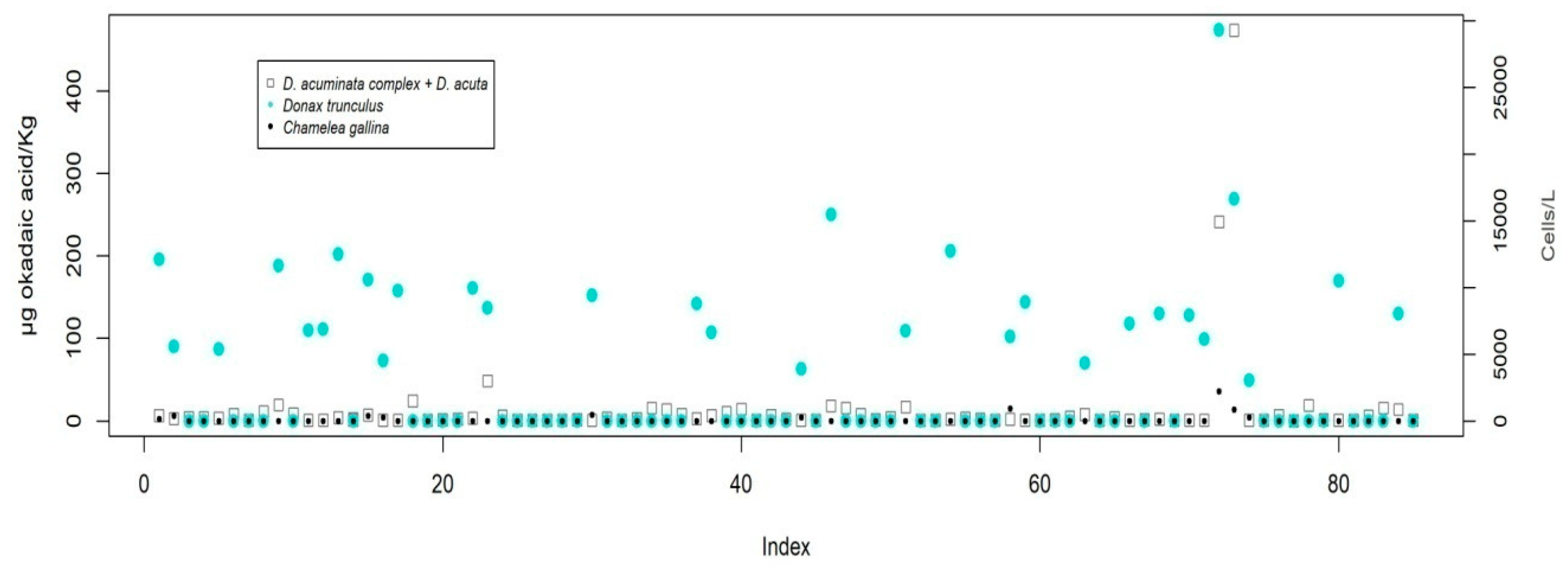
V. verrucosa. This difference has been verified for the pair
D. trunculus and
C. gallina (
Figure 5) in 85 double samples taken, which has contributed to the selection of
Donax trunculus as an indicator species for some harvesting areas.
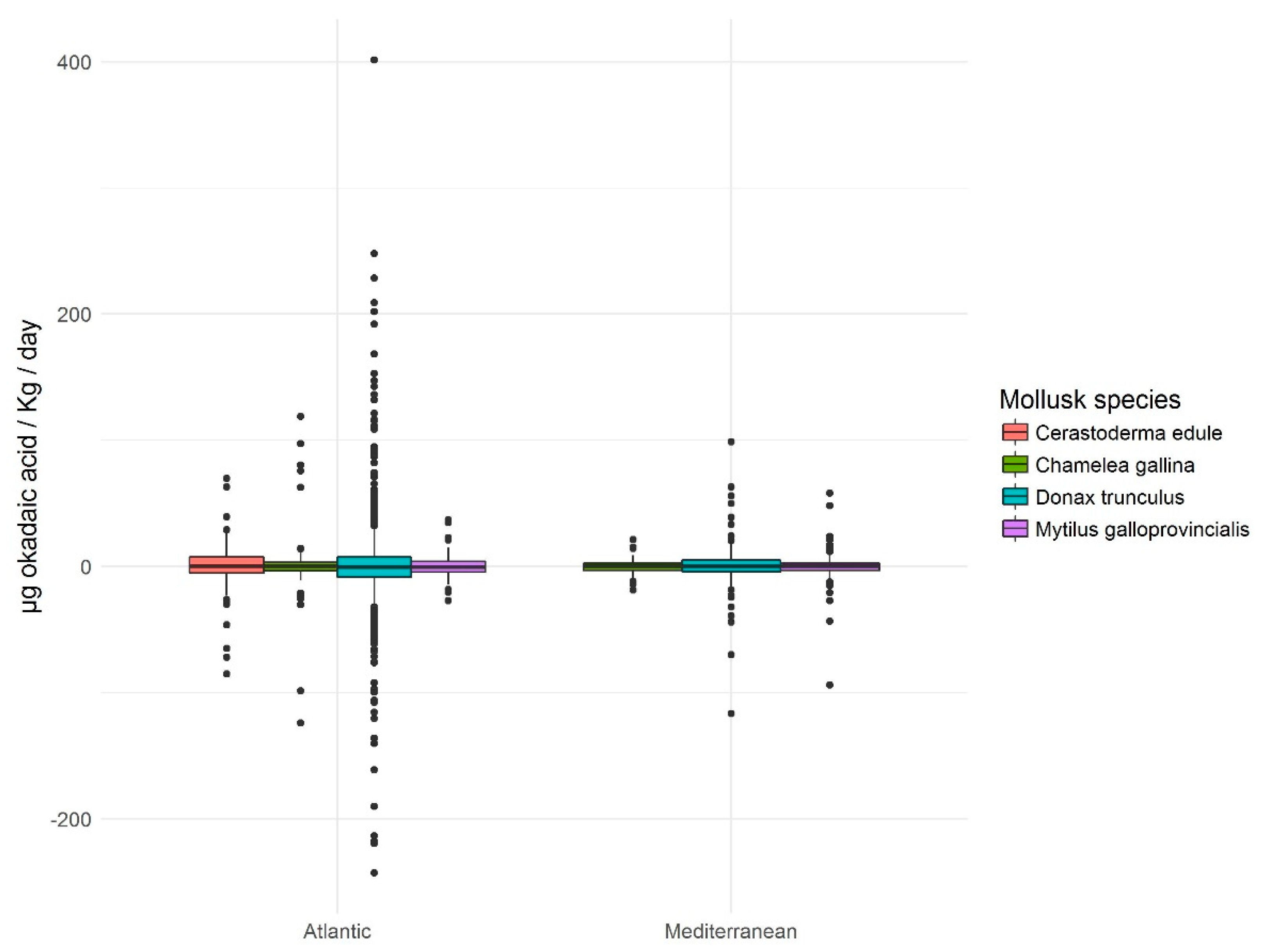
In view of the results of the frequency distribution of accumulation and elimination daily rates (
Figure 6), it is worth noting the symmetry and positive kurtosis. This means that, on the one hand, there are no appreciable differences between the speed of accumulation and elimination and, on the other hand, the values that are statistically considered as normal, do not exceed 25 µg okadaic acid/kg/day which, as has been said, are low values taking into account the extent of the range. This means that the occasions on which, starting from an absence of toxicity, the molluscs analyzed have the capacity to exceed the legal limit (160 µg okadaic acid/kg) for less than a week are statistically not significant (outliers;
Figure 6).
The monitoring program of Andalusia establishes that once levels above the regulatory limit are detected in molluscs, temporary closures of fisheries must be triggered. They have been recorded in digital databases from 2007. The data presented (
Table 2 and
Table 3) correspond to this period.
3. Discussion
Data from monitoring programs are usually complex in their processing. Regulatory changes lead to modifications in sampling plans in a way that they lose their regularity when frequency, spatial distribution or target species change. This study has taken into account such features when calculating parameters or obtaining general conclusions.
The polymorphic life cycle, feeding behaviour and phase of the cell cycle have been given by various authors as the causes of the wide morphological variation of certain species of
Dinophysis [
16,
17]. This variation makes some species intermediate forms similar in shape, a fact that makes it often difficult to discriminate them as it is the case with the pairs:
D. acuminata/
D. sacculus,
D. acuminata/
D. ovum,
D. sacculus/
D. pavilardii,
D. caudata/
D.tripos mentioned by various authors according to the case [
13,
14,
18,
19]. For a monitoring program, this difficulty translates into the possibility of producing false positive or negative results in the risk assessment, taking into account possible differences in the toxic potential between species. An example of this was resolved in the Thermaikos Gulf, Greece [
20] by considering
D. acuminata and
D. ovum as
D. cf
acuminata. A similar treatment has been decided in the monitoring program of Andalusia where
D. acuminata,
D. ovum and
D. sacculus are treated as a whole as
D. acuminata complex.
The species associated with DST outbreaks in Andalusia (
D. acuminata complex and
D. acuta) have also been considered as the main species responsible on the Galician coast in addition to
D. sacculus [
17,
19,
21]. Vale and Sampayo [
22,
23] consider
D. acuminata and
D. acuta as responsible for specific DST episodes along the Portuguese coast. These two species are considered again to be responsible for similar outbreaks in Sweden [
24]. In the North Sea, around the island of Helgoland (Germany),
D. norvegica joins
D. acuminata as co-responsible for episodes of contamination in mussels.
D. acuminata appears as the species of wider distribution concerning its association with toxic events. However, there are cases in which the responsibility of episodes of intoxication is attributed to less common species such as
D. fortii,
D. rotundata,
D. caudata,
D. sacculus and
D. tripos in the east Adriatic [
25]. In our program, although such species have been detected, there were two results: toxicity was not observed in molluscs or the above-mentioned species of
Dinophysis accompanied
D. acuminata and/or
D. acuta to a much lesser abundance.
It is important to mention the spatial coherence of the homogeneous areas obtained in the cluster analysis as there is no cluster covering non-neighbouring stations. It is also noteworthy that clusters mainly occupy coastal areas where the direction of the coast line is constant and that changes in that direction generate a different cluster. This feature seems to define a characteristic length of cluster dependent on such geomorphological characteristics that, in turn, could define hydrodynamic characteristics. This gives consistency to the conclusion derived from the analysis that the patterns and abundances of D. acuminata complex in the different stations of each cluster are similar.
The seasonal patterns of
Dinophysis (growth pulses from the beginning of the spring until the end of autumn) in Andalusia do not differ from those found by the other authors in western Europe. It exceeds at the end points of the period mentioned by Smayda [
26]: “the blooms of dinoflagellates occur in predominantly warm waters which are stratified from late spring to early autumn”. Other authors have limited this same period as the one with the largest development of
Dinophysis [
27,
28,
29,
30,
31]. In general, although the most apparent growth peaks are given within this period, species such as
D. acuminata complex are present throughout the rest of the year even though at very low concentrations. Nincevic-Gladan [
25] revealed that of the 13 species detected in the east Adriatic coast, only
D. tripos appeared in winter. Koukaras [
20] noted that, at genus level, the episodes usually lasted between 3 and 4 months, and sporadically appeared throughout the rest of the year.
Many authors have reported and analysed the possible causes of the discrepancy between the values for toxicity in molluscs and the abundance of microalgae [
24,
32,
33,
34]. The causes mainly proposed are the presence of alternative food, environmental factors, the stratification of the water column that favours the development of thin layers of microalgae, or the variability in the toxin content per cell. In addition, other authors have studied the daily vertical migration of plankton [
13,
35,
36,
37,
38], which translates into irregularity in daily food availability for molluscs. Discrepancy in the pairs of parallel sampling values (if a linear correlation is expected between cell numbers and toxicity) and the explanation of the inaccuracy of dinoflagellate abundance as an indicator of the true exposure suffered by molluscs throughout the previous week has been found in our data. This discrepancy may be a consequence of the factors above mentioned. In this way, the toxicity value in molluscs represents the result of a continuous exposure before the sample is taken, while the value of plankton concentration is representative of the moment of capture as molluscs have not yet responded.
The European regulations [
3,
4] says “production areas must be periodically monitored to check for the presence of toxin-producing plankton in production and relaying waters and biotoxins in live bivalve molluscs. The sampling frequency for toxin analysis in the molluscs is, as a general rule, to be weekly during the periods at which harvesting is allowed. This frequency may be reduced in specific areas, or for specific types of molluscs, if a risk assessment on toxins or phytoplankton occurrence suggests a very low risk of toxic episodes”. On the other hand, it indicates that monitoring must be increased if there is evidence or suspicions of an increased risk. Such indications, in principle, leave no room for a lack of efficiency in the programs established at European level so, the answer to the initial question raised in this study, “Are the regulatory requirements for the analysis of toxic phytoplankton effective as an early-warning system of the risk of contamination of molluscs in the Andalusian coast?” would be “Yes”. The problem lies in the risk assessment. In Andalusia, the check once a week of the concentration of phytoplankton, as discussed above, may have inaccuracies as indicators. An important number of cases in which molluscs had the capacity to exceed the legal limits in less than a week have been detected on our coast. Under these conditions, 11.6% of the cases were of
Donax trunculus, 12.2% of
Cerastoderma edule, 11.5% of
Mytilus galloprovincialis and 11.4% of
Chamelea gallina. Therefore, in order to prevent, to some extent, this risk from 2015 on, the uncertainty of the measure of toxicity needed to activate the intensification of sampling (48 h) was set in the criteria of the monitoring program as a factor to take into account. On the other hand, incidents with the toxicities of bivalve molluscs have been so important that there have been closures of fisheries for even half a year. A fact that shows the magnitude of the problem, bearing in mind that such fisheries represent a significant economic source at a local coastal scale. As has been mentioned, there have been numerous studies aimed at understanding the dynamics of the plankton’s and molluscs’ toxicities. Although there is increasing awareness of them, it remains necessary to expand knowledge which, with large sets of data of different nature, will shed light to the production of other indices or will suggest changes in sampling strategies to reduce the range of health risk.
4. Materials and Methods
All the data that underpin this article correspond to the information generated by the program for phytoplankton and biotoxins official monitoring established in Andalusia in 1994. The data generated were not recorded on a computer system until 1999. Therefore, this article refers to the period from then on. The information is available to the public and can be found at the web page
http://www.cap.junta-andalucia.es/agriculturaypesca/moluzonasprodu/ of the Regional Ministry for Agriculture, Fisheries and Rural Development of Andalusia (Regional Government). That Ministry is conferred with the exclusive jurisdiction for fishing in inland waters, shellfish and aquaculture. The laboratory designated by the competent authority to perform the analyses corresponding to the official monitoring program is the
Laboratory for the Quality Control of Fishery Resources which has developed its functions since 1996. The laboratory has been entitled to develop this monitoring since 2007, according to the UNE-EN-ISO 17025 standard.
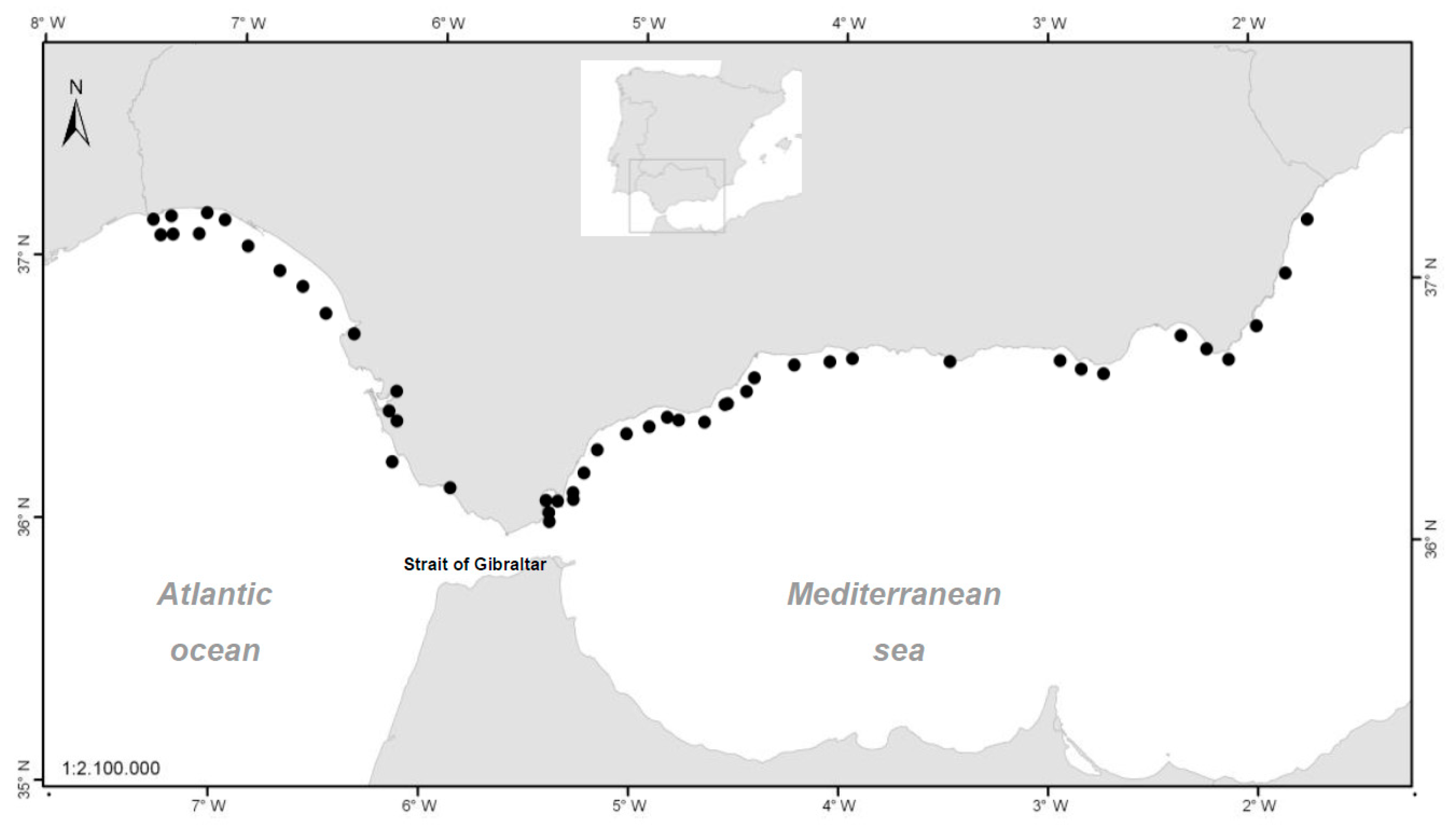
4.1. Sampling Plan
The sampling stations in
Figure 7 are represented by the centroid of the production area to which they belong. However, the point of capture of each sample can be placed in any position within the same area.
Based on the operational capacity of the program, the sampling strategy has been adapted as far as possible to the European Regulations and their successive modifications. Thus, the sampling frequency was weekly at some stations from the beginning while others started on a fortnightly basis for water and quarterly for molluscs. However, this frequency has now been increased to include sampling all parameters in all production areas declared every week. The program has always allowed the possibility of increasing sampling if evidence of an increase of a toxicological risk is detected. The successive changes are detailed in
Appendix C (
Table A1,
Table A2,
Table A3 and
Table A4).
4.2. Sampling Techniques
Water samples were collected using vertical hauls with bongo nets of mesh size 20 μm and a hose system [
40]. Samples of molluscs from natural populations were collected using techniques traditionally used by the Andalusian shellfish sector. Samples were either taken at points of highest productive activity within each production area or provided by producers from their aquaculture facilities.
4.3. Analysis
Testing of lipophilic toxins and toxic phytoplankton are based on official methods harmonized and accredited since 2007 by ENAC (National Accreditation Entity-in Spanish) according to the standard UNE-EN-ISO 17025.
For the identification and enumeration of
Dinophysis spp., the technique of Utermöhl [
41] was used as recommended in European standard UNE-EN 15204
Guidance standard on the enumeration of phytoplankton using inverted microscopy.
Lipophilic toxin levels were determined according to the method described by Yasumoto et al. [
42]. From 2011 on, the data presented correspond to analyses carried out using the European harmonized chemical technique (LC-MS/MS).
4.4. Data Processing
The processing and graphic presentations of the data are carried out using the free software for statistical analysis and graphics R in its 2013 version [
15].
Given the huge volume of records (20 years), and for the submission of the time series, a cluster analysis was carried out on producing areas in order to reduce the datasets to be presented, excluding redundancy without losing relevant information. To do this, the K-means non-hierarchical cluster analysis (function in R: kmeans()) was used after developing the analysis of the optimal number of clusters (function in R: cascadeKM()) over a maximum of 12 clusters. (If more clusters were extracted, the purpose sought with the use of this technique would not be achieved). This is an unsupervised automatic method for pattern recognition, i.e., it is based on the response variable (concentration of
D. acuminata complex) without seeking correlations with explanatory variables (e.g., environmental). For the response variable, the concentration of
Dinophysis acuminata complex was chosen, as it provides more information because it has a high frequency in the samples. A total of 6659 samples were used after excluding very low incidence areas (see
Figure 3) and extracting only quantifiable values. Thus, it is assumed that the homogeneous areas defined by
D. acuminata, probably conditioned by the hydrodynamic characteristics, could be homogeneous also for the other species. In any case, the most relevant species in Andalusia is
D. acuminata complex and the presentation of the information must adapt mainly to it. To obtain the optimal number of areas the “SSI” (Simple Structure Index) criterion has been used [
43]. This is an index that combines three factors: 1) the greatest difference in the response variable between clusters, 2) the sizes of the most contrasting clusters and 3) the deviation of the variable in the cluster centers compared to its overall mean. It can be deduced that each factor separately indicates a more homogeneous selection of groups and thus, a higher SSI score implies a better partition.
The calculation of the daily rate of accumulation or elimination of toxins is carried out through the difference in toxicity measured between the samples from the same area in two successive samplings divided by the number of days, provided that between the two sampling there are not more than 11 days (maximum amplitude in samples obtained in working days of two consecutive weeks). This rate is a calculated value that indicates what would be the accumulation or elimination if every day between the two samples was kept constant. It represents, therefore, a theoretical daily value that must be taken with caution, understanding that there are probably days between the two samples with a higher and lower rate.
The information provided concerning the relative sensitivity among bivalve species identically exposed to microalgae of Dinophysis genus derives from parallel samples, if present, in which such species have been collected at the same time in the same area.