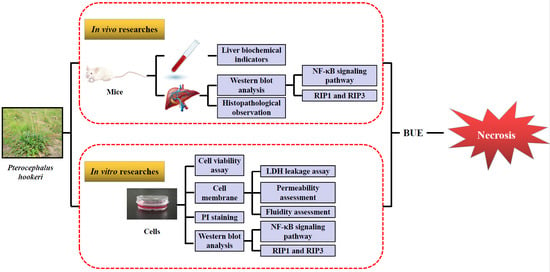
Evaluation of the Liver Toxicity of Pterocephalus hookeri Extract via Triggering Necrosis
Abstract
1. Introduction
2. Results
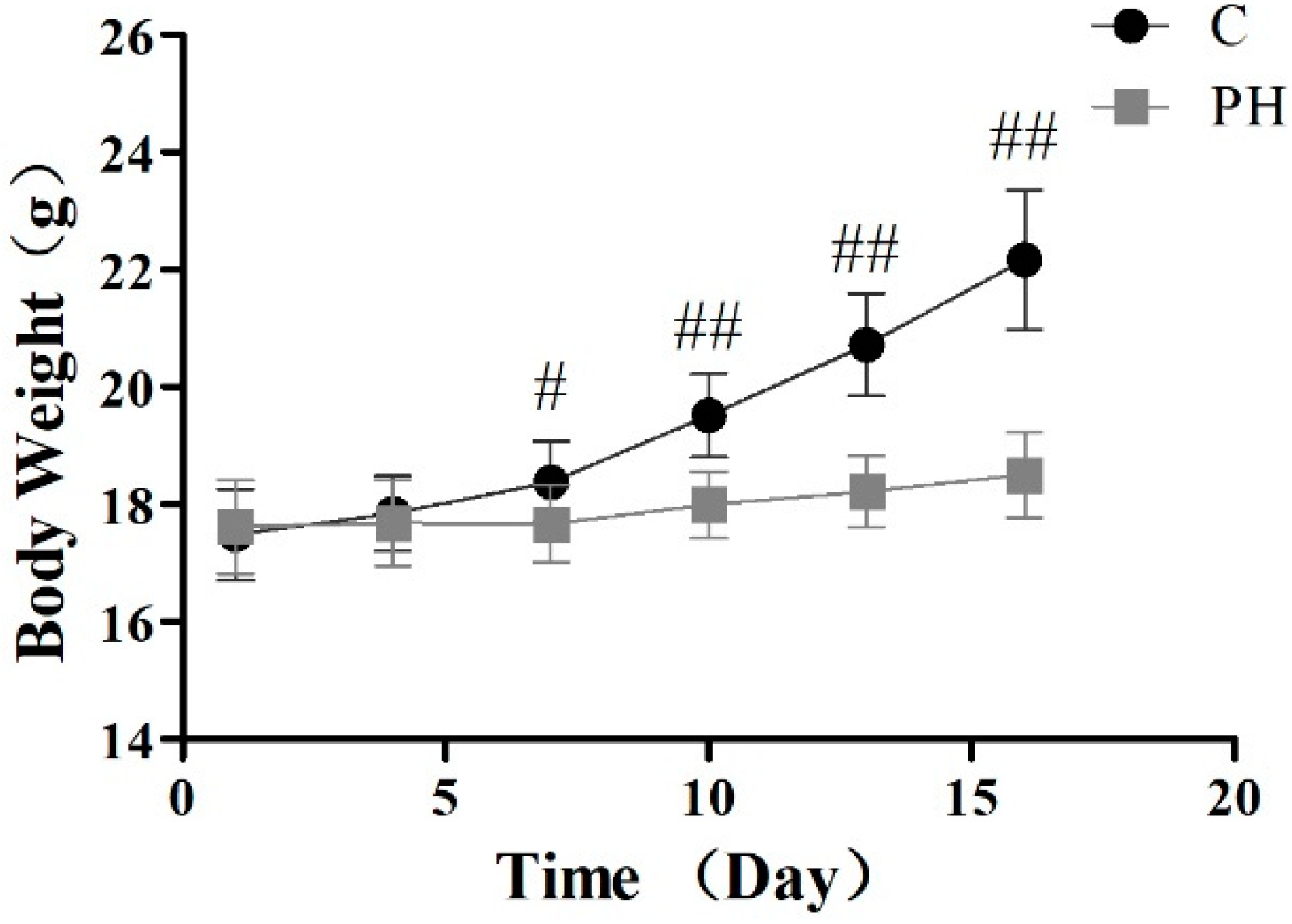
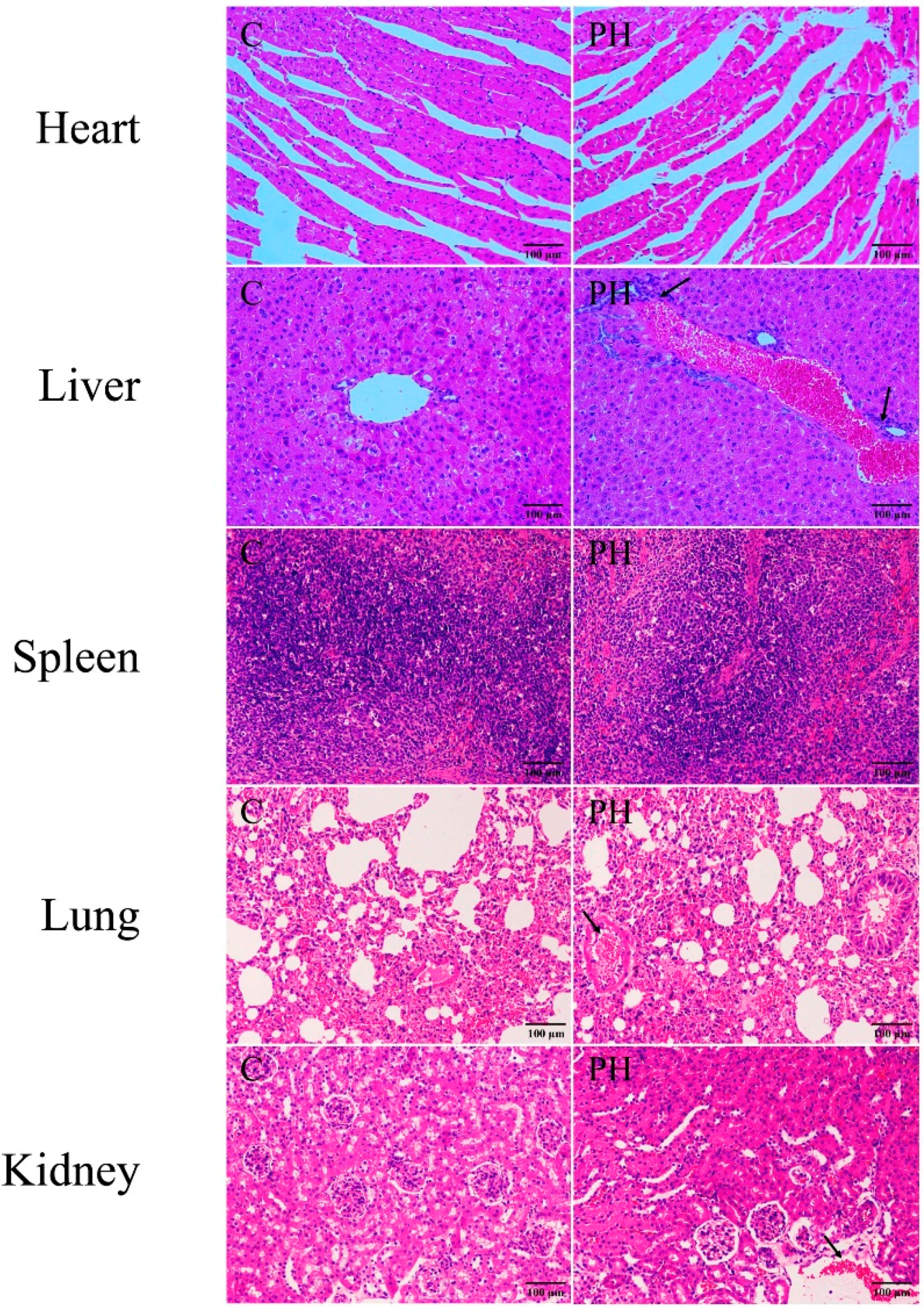
2.1. Toxic Effects of PH in Mice
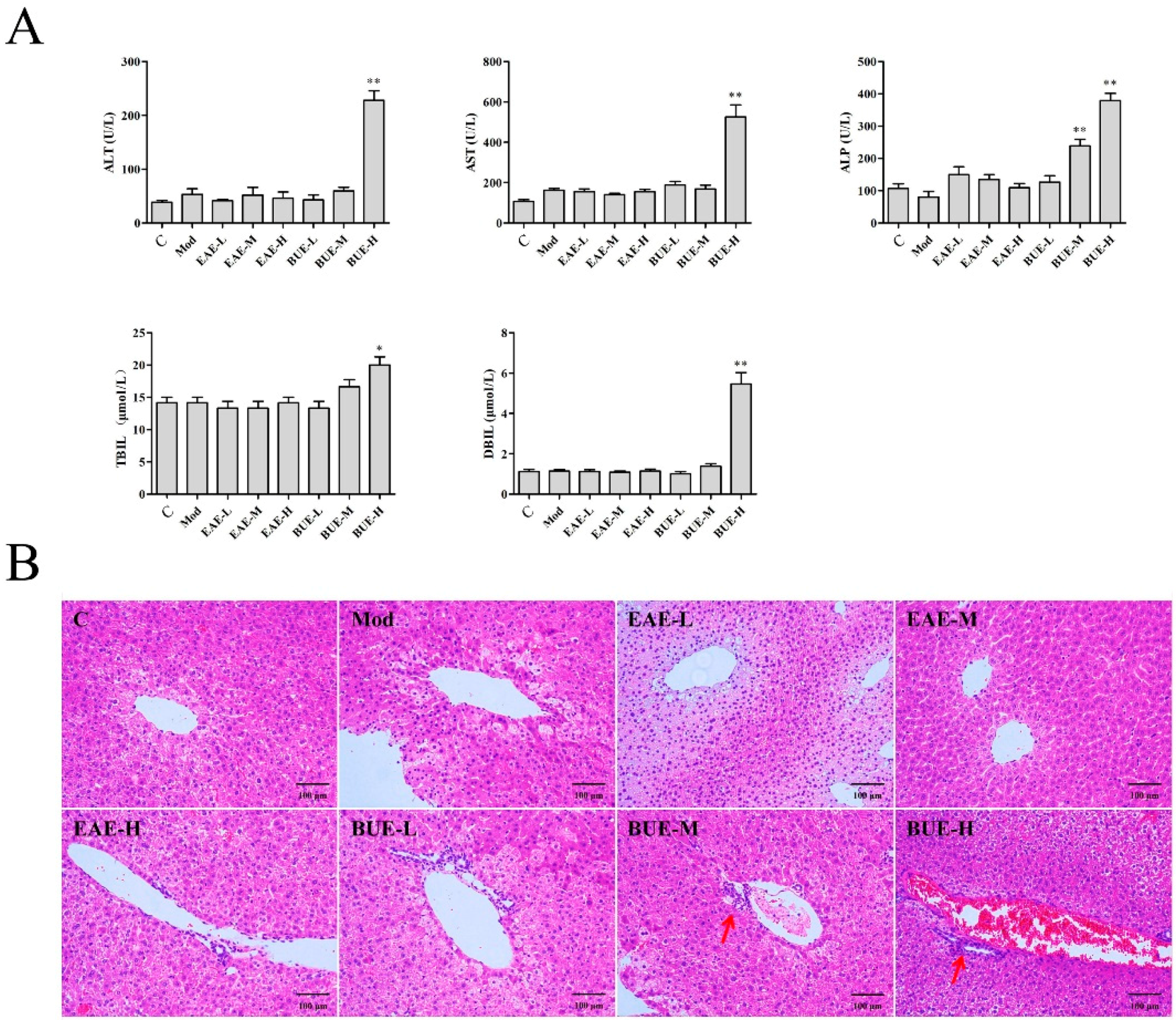
2.2. Liver Toxicity of EAE and BUE on Carrageenan-Induced Paw Edema in Mice
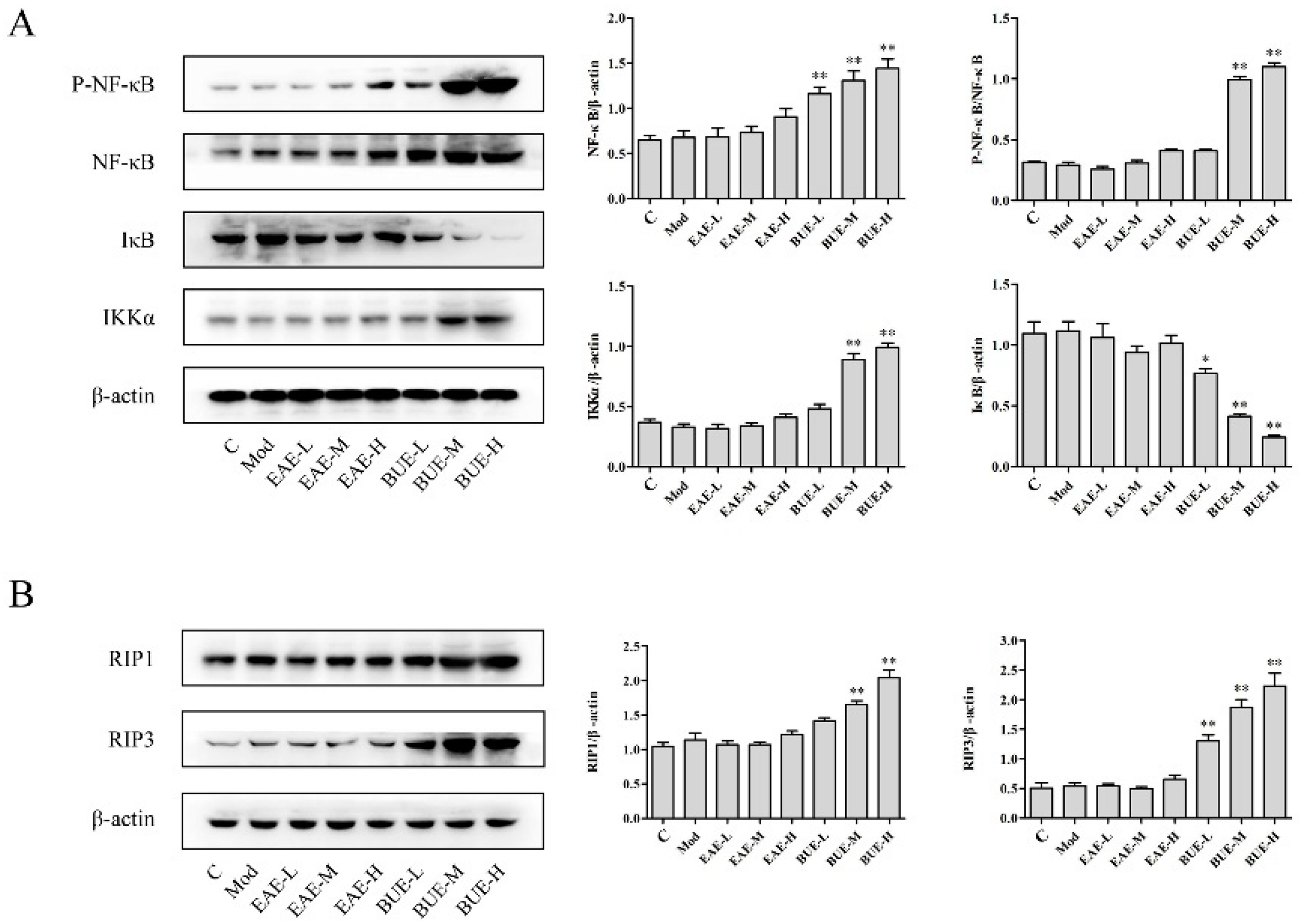
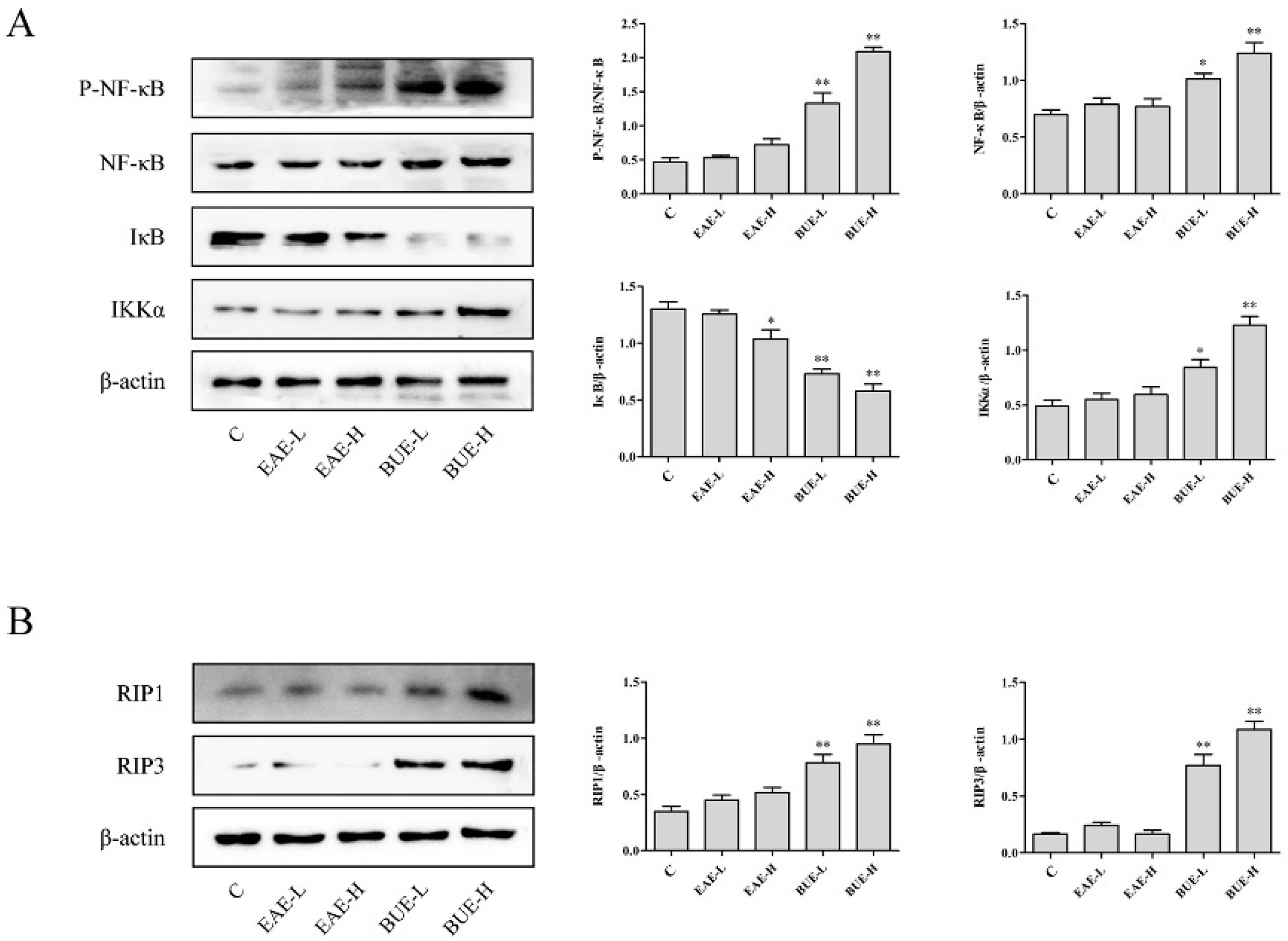
2.3. Effects of EAE and BUE on the Expression of NF-κB Signaling Pathway In Vivo
2.4. Effects of EAE and BUE on the Expression of Necrosis-Related Proteins In Vivo
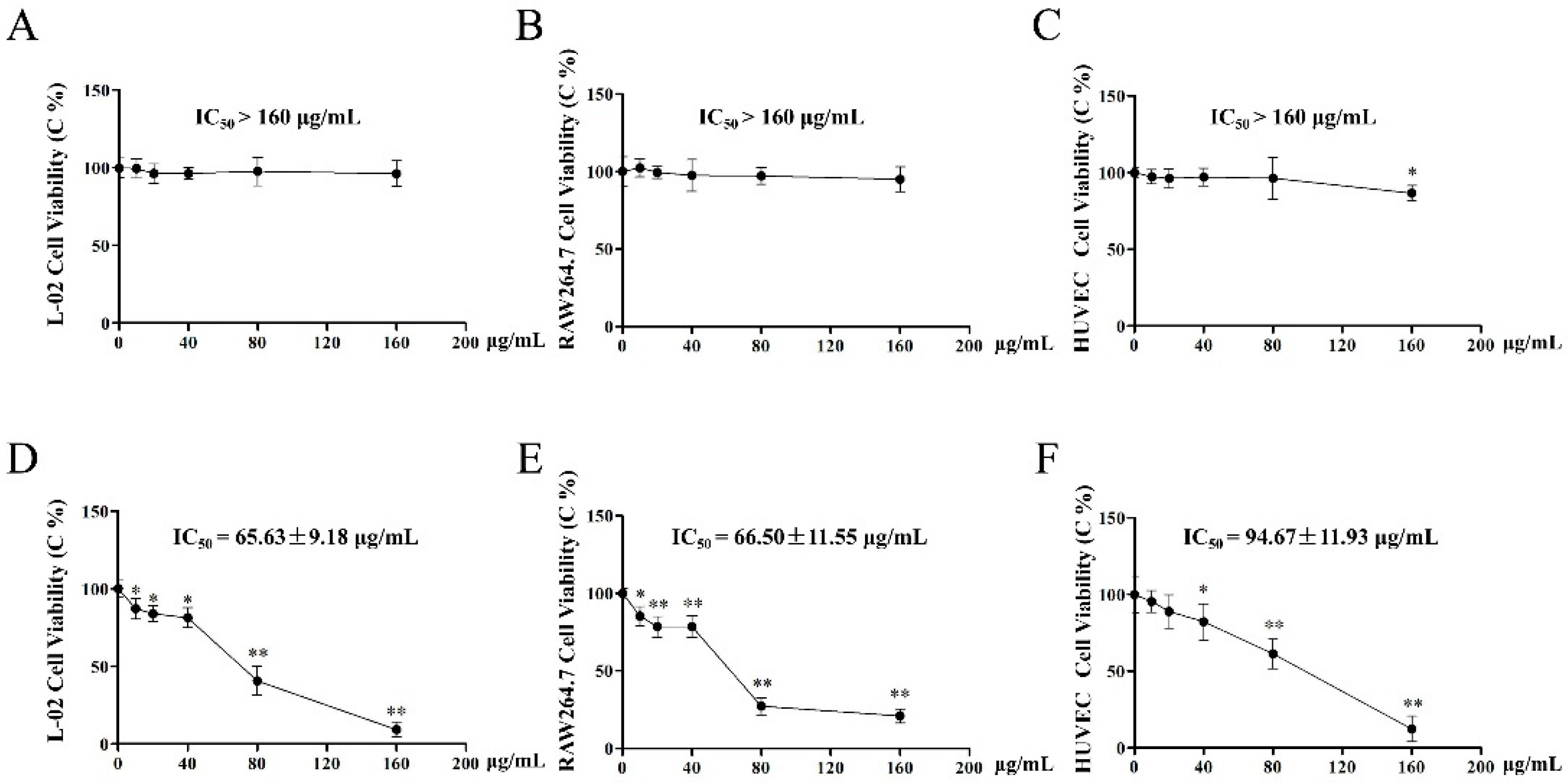
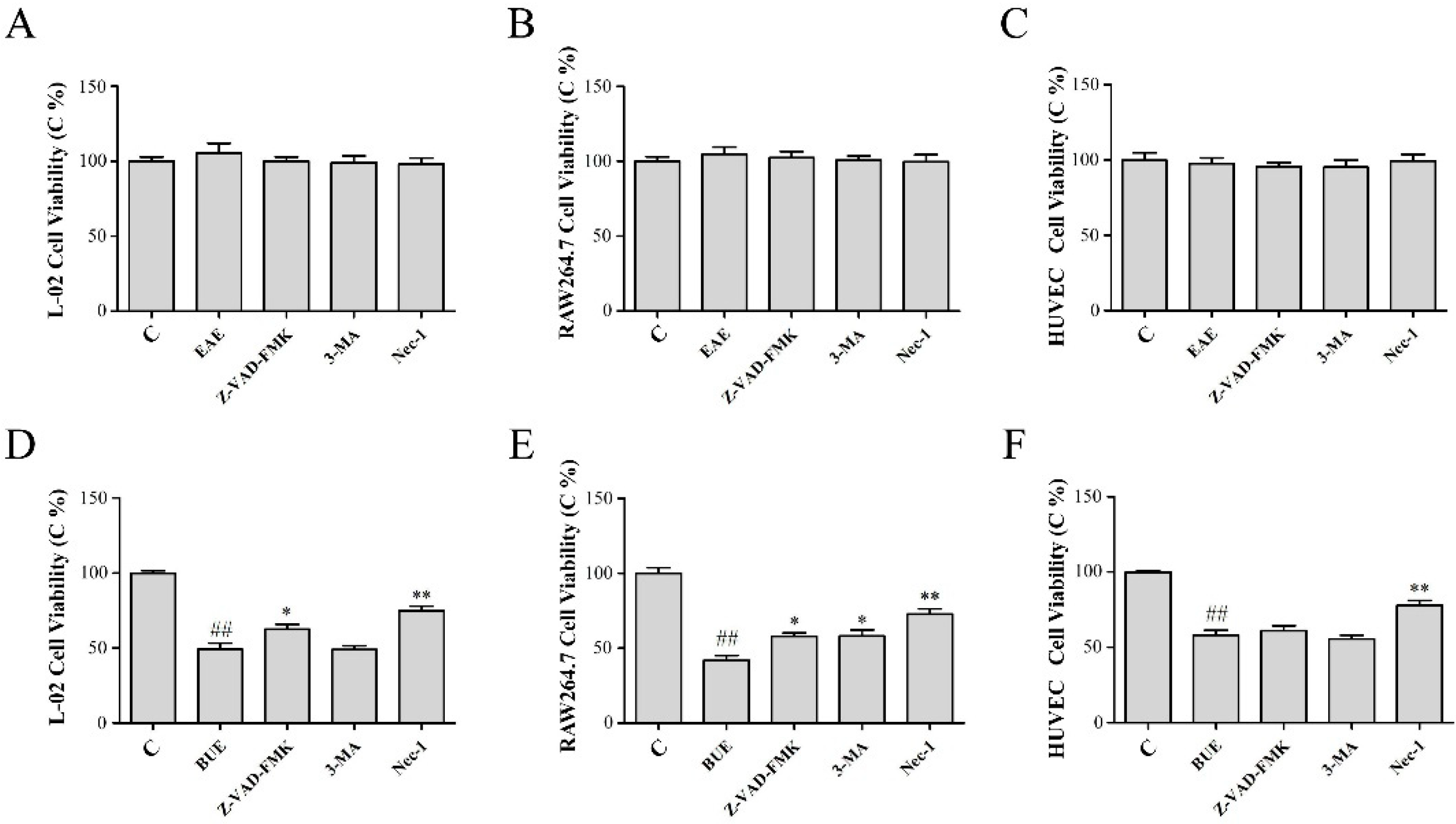
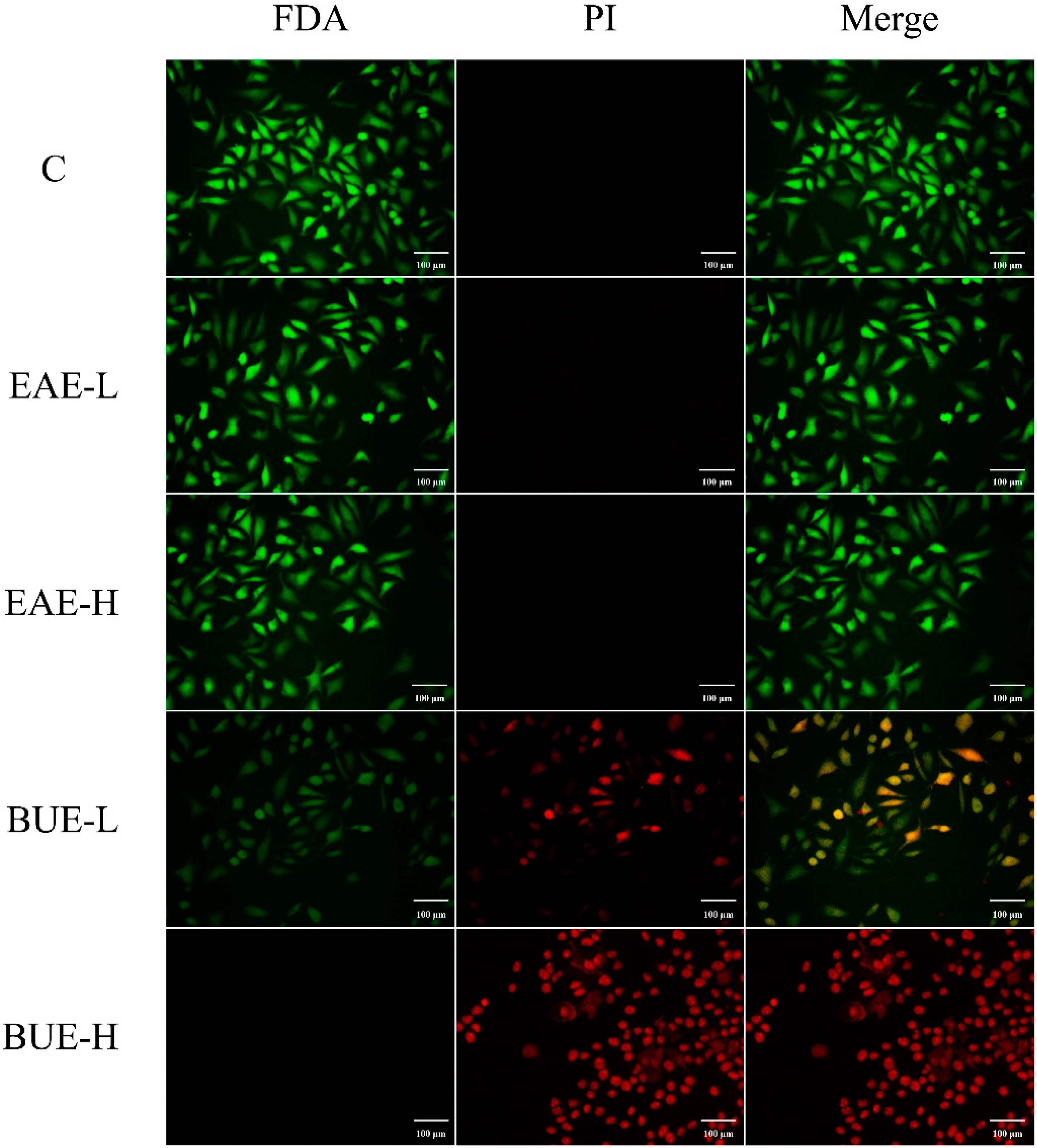
2.5. Effect of EAE and BUE on L-02, RAW264.7, and HUVECs
2.6. Effects of EAE and BUE on NF-κB Signaling Pathway In Vitro
2.7. Effects of EAE and BUE on the Expression of Necrosis-Related Proteins In Vitro
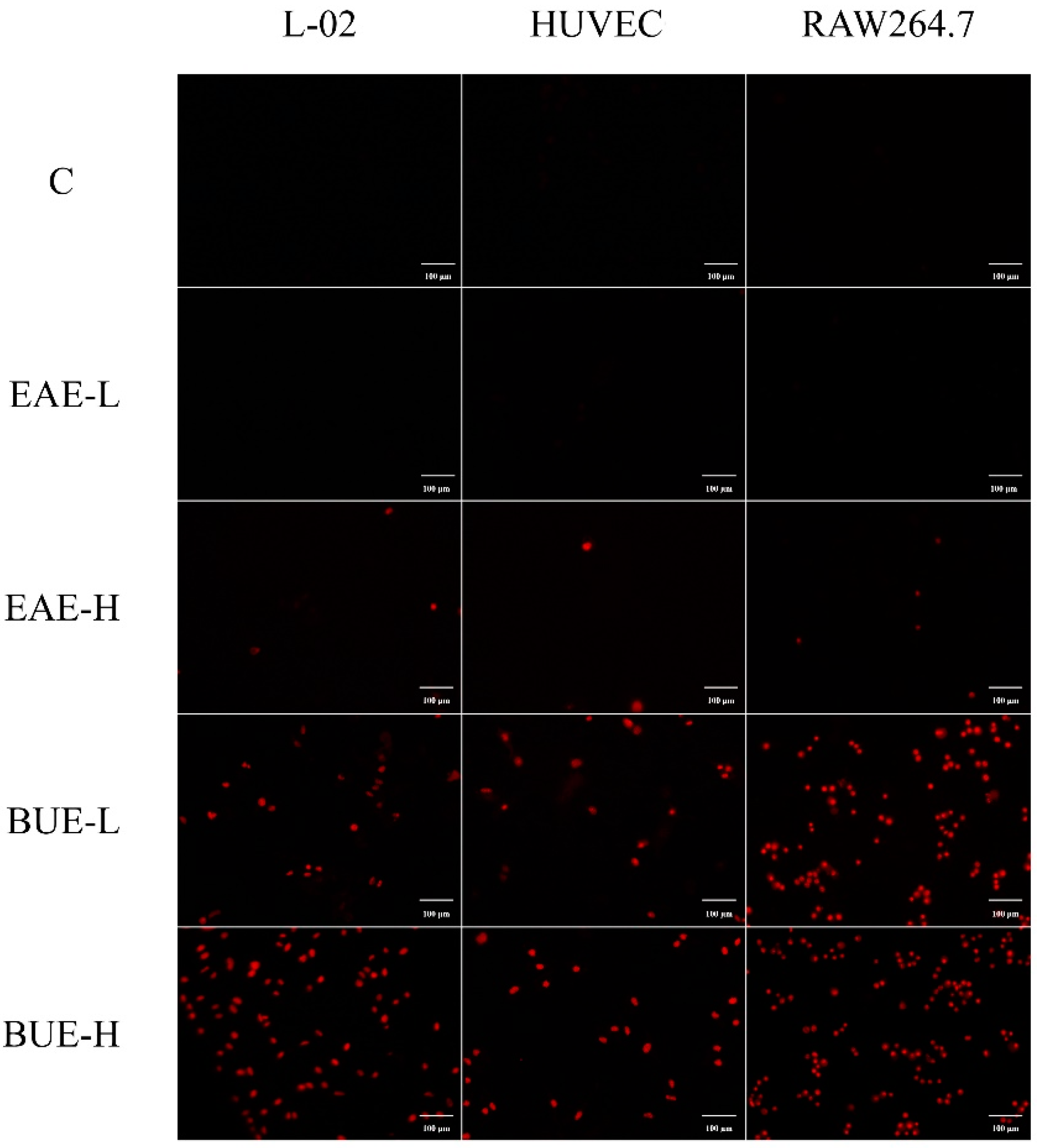
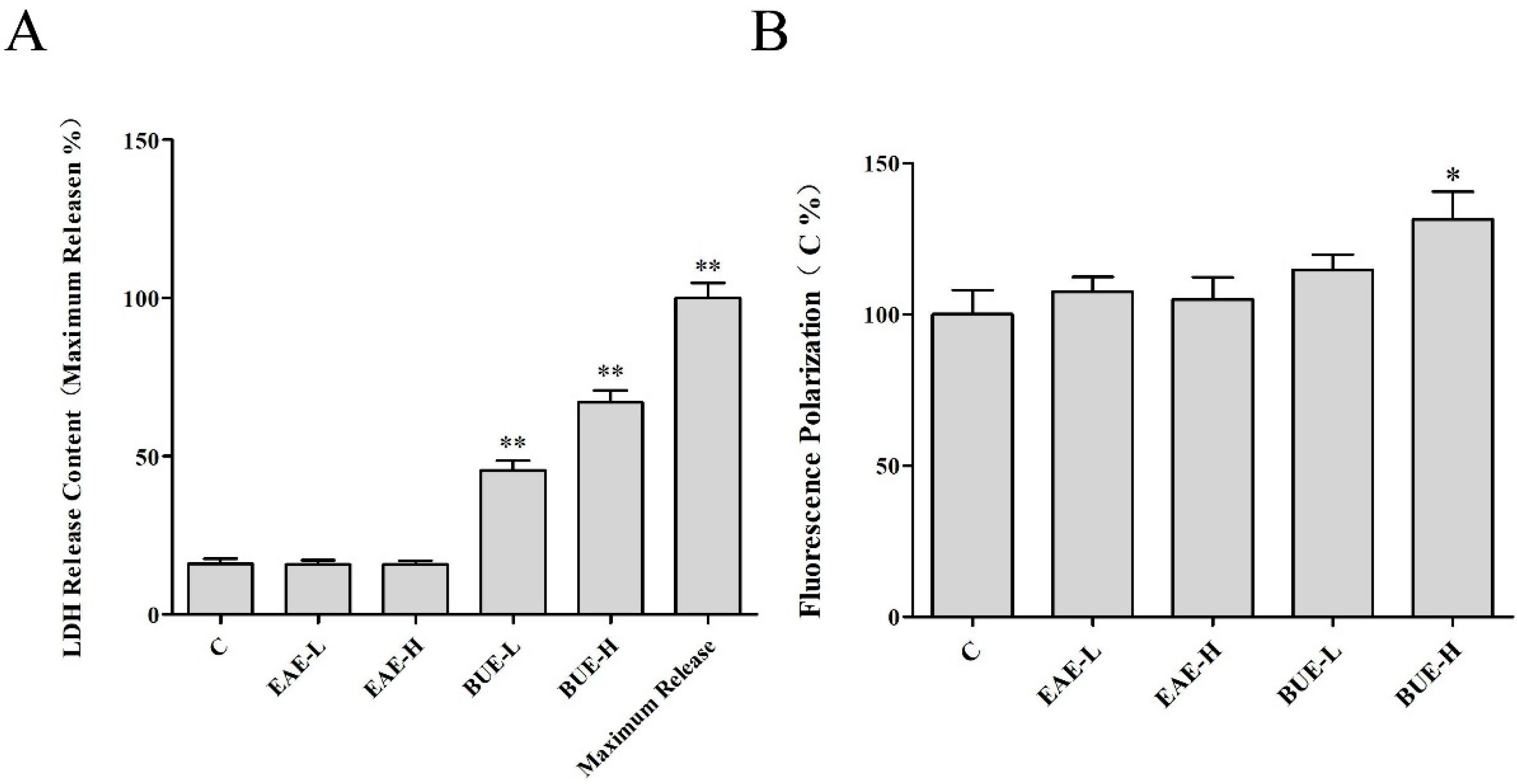
2.8. The Effect of EAE and BUE on L-02 Cell Membrane
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Chemicals and Reagents
5.2. Plant Material and Extraction
5.3. Animals
5.4. Investigation of Body Weight and Pathological Section of Visceral Tissues in Mice
5.5. Investigation of Liver Toxicity on EAE and BUE In Vivo
5.6. Cell Culture and Treatment
5.7. Cell Viability Assay
5.8. PI Staining Assay
5.9. LDH Leakage Assay
5.10. L-02 Cell Membrane Fluidity Assessment
5.11. L-02 Cell Membrane Permeability Assessment
5.12. Western Blot Analysis
5.13. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xia, Q.; Zhang, X.; Xu, K.; Zhang, W.; Fan, J.; Zhao, C.; Feng, Y.; Ni, Y.; Ma, Z.; Lin, R. Review on toxicity of toxic traditional Chinese medicine recorded in Chinese pharmacopoeia (2015 version). Glob. Tradit. Chin. Med. 2017, 377–384. [Google Scholar] [CrossRef]
- Ng, A.W.T.; Poon, S.L.; Huang, M.N.; Lim, J.Q.; Boot, A.; Yu, W.; Suzuki, Y.; Thangaraju, S.; Ng, C.C.Y.; Tan, P.; et al. Aristolochic acids and their derivatives are widely implicated in liver cancers in Taiwan and throughout Asia. Sci. Transl. Med. 2017, 9, eaan6446. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ma, Z.; Niu, M.; Zhu, Y.; Liang, Q.; Zhao, Y.; Song, J.; Bai, Z.; Zhang, Y.; Zhang, P.; et al. Evidence chain-based causality identification in herb-induced liver injury: Exemplification of a well-known liver-restorative herb Polygonum multiflorum. Front. Med. 2015, 9, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Bunchorntavakul, C.; Reddy, K.R. Review article: Herbal and dietary supplement hepatotoxicity. Aliment. Pharm. 2013, 37, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Hu, J.; Lin, J.; Fang, W.; Du, G. Anti-inflammatory and analgesic effects of ethanol and aqueous extracts of Pterocephalus hookeri (C.B. Clarke) Höeck. J. Ethnopharmacol. 2009, 123, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Lin, N.; Liu, B.; Zhang, J.; Long, Y.; Dong, G.; Jin, H.; Ma, B. Acute toxicity, 28-day repeated-dose toxicity and toxicokinetic study of timosaponin BII in rats. Regul. Toxicol. Pharm. 2017, 90, 244–257. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Jin, Y.; Yan, H.; Gao, Z.; Xu, B.; Yang, B.; He, Q.; Shi, Q.; Luo, P. High-mobility group box 1 protein-mediated necroptosis contributes to dasatinib-induced cardiotoxicity. Toxicol. Lett. 2018, 296, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Li, Y.; Yang, D.; Zhang, N.; Xu, X.; Wu, Y.; Chen, J.; Han, J. Quantitative phosphoproteomic analysis of RIP3-dependent protein phosphorylation in the course of TNF-induced necroptosis. Proteomics 2014, 14, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Ren, X.; Wang, X.; Wang, H.; Liu, G.; Yuan, X.; Zheng, S.; Yu, L.; Pan, S.; Song, G.; et al. A novel combination of oridonin and valproic acid in enhancement of apoptosis induction of HL-60 leukemia cells. Int J. Oncol. 2016, 48, 734–746. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Mei, Y.; Chen, X.; Jiang, L.; Jiang, Y.; Song, X.; Xiao, H.; Zhang, J.; Wang, J. Autophagy plays a pro-survival role against methamphetamine-induced apoptosis in H9C2 cells. Toxicol. Lett. 2018, 294, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhu, C.J.; Zhu, F.; Dai, B.B.; Song, S.J.; Wang, Z.Q.; Feng, Y.B.; Ge, J.F.; Zhou, R.P.; Chen, F.H. Necrostatin-1 ameliorates adjuvant arthritis rat articular chondrocyte injury via inhibiting ASIC1a-mediated necroptosis. Biochem. Biophys. Res. Commun. 2018, 504, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Xiong, K.; Liao, H.; Long, L.; Ding, Y.; Huang, J.; Yan, J. Necroptosis contributes to methamphetamine-induced cytotoxicity in rat cortical neurons. Toxicol. Vitr. 2016, 35, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.D.; Padmavathi, P.; Bulle, S.; Hebbani, A.V.; Marthadu, S.B.; Venugopalacharyulu, N.C.; Maturu, P.; Varadacharyulu, N.C. Association between alcohol-induced oxidative stress and membrane properties in synaptosomes: A protective role of vitamin E. Neurotoxicol. Teratol. 2017, 63, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhou, X.; Wang, Z.; Zhu, Y.; Li, P. Elevation of plasma membrane permeability upon laser irradiation of extracellular microbubbles. Laser Med. Sci. 2010, 25, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Nazzal, D.; Cantéro, A.; Therville, N.; Ségui, B.; Nègre-Salvayre, A.; Thomsen, M.; Benoist, H. Chlamydia pneumoniae alters mildly oxidized low-density lipoprotein-induced cell death in human endothelial cells, leading to necrosis rather than apoptosis. J. Infect. Dis. 2006, 193, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Welz, P.; Wullaert, A.; Vlantis, K.; Kondylis, V.; Fernández-Majada, V.; Ermolaeva, M.; Kirsch, P.; Sterner-Kock, A.; van Loo, G.; Pasparakis, M. FADD prevents RIP3-mediated epithelial cell necrosis and chronic intestinal inflammation. Nature 2011, 477, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Holt, M.P.; Ju, C. Mechanisms of drug-induced liver injury. AAPS J. 2006, 8, E48–E54. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, S.; Mousavi Kani, S.N.; Rezazadeh, L.; Pouramir, M.; Ghasemi-Kasman, M.; Moghadamnia, A.A. Low dose administration of Bisphenol A induces liver toxicity in adult rats. Biochem. Biophys. Res. Commun. 2017, 494, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, X.; Wu, Q.; Jin, Y.; Ning, C.; Wang, R.; Mao, J.; Chen, M. The hepatoprotective effects of Sedum sarmentosum extract and its isolated major constituent through Nrf2 activation and NF-κB inhibition. Phytomedicine 2019, 53, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Wallach, D.; Kang, T.B.; Dillon, C.P.; Green, D.R. Programmed necrosis in inflammation: Toward identification of the effector molecules. Science 2016, 352, aaf2154. [Google Scholar] [CrossRef] [PubMed]
- Luedde, T.; Schwabe, R.F. NF-κB in the liver-linking injury, fibrosis and hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Micheau, O.; Seed, B.; Zaru, R.; Thome, M.; Valitutti, S.; Tschopp, J.; Bodmer, J.; Holler, N.; Schneider, P.; Attinger, A. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat. Immunol. 2000, 1, 489–495. [Google Scholar]
- Declercq, W.; Vanden Berghe, T.; Vandenabeele, P. RIP Kinases at the Crossroads of Cell Death and Survival. Cell 2009, 138, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Pasparakis, M.; Vandenabeele, P. Necroptosis and its role in inflammation. Nature 2015, 517, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.J.; Hinner, M.J. Getting across the cell membrane: An overview for small molecules, peptides, and proteins. Methods Mol. Biol. 2015, 1266, 29–53. [Google Scholar] [PubMed]
- Levaot, N.; Hershfinkel, M. How cellular Zn2+ signaling drives physiological functions. Cell Calcium 2018, 75, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Yun, J.; Kim, S.I.; Hong, H.; Park, J.; Pyun, J.; Ryu, W. Scalable long-term extraction of photosynthetic electrons by simple sandwiching of nanoelectrode array with densely-packed algal cell film. Biosens. Bioelectron. 2018, 117, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, N.; Pellach, M.; Glick, Y.; Gil, R.; Levy, G.; Avrahami, D.; Barbiro-Michaely, E.; Nahmias, Y.; Gerber, D. Neuregulin 1 discovered as a cleavage target for the HCV NS3/4A protease by a microfluidic membrane protein array. New Biotechnol. 2018, 45, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Bessa, L.J.; Ferreira, M.; Gameiro, P. Evaluation of membrane fluidity of multidrug-resistant isolates of Escherichia coli and Staphylococcus aureus in presence and absence of antibiotics. J. Photochem. Photobiol. B Biol. 2018, 181, 150–156. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, R.; Dong, Z.; Zhang, X.; Mao, J.; Meng, F.; Lan, X.; Liao, Z.; Chen, M. Evaluation of the Liver Toxicity of Pterocephalus hookeri Extract via Triggering Necrosis. Toxins 2019, 11, 142. https://doi.org/10.3390/toxins11030142
Wang R, Dong Z, Zhang X, Mao J, Meng F, Lan X, Liao Z, Chen M. Evaluation of the Liver Toxicity of Pterocephalus hookeri Extract via Triggering Necrosis. Toxins. 2019; 11(3):142. https://doi.org/10.3390/toxins11030142
Chicago/Turabian StyleWang, Rui, Zhaoyue Dong, Xiaolong Zhang, Jingxin Mao, Fancheng Meng, Xiaozhong Lan, Zhihua Liao, and Min Chen. 2019. "Evaluation of the Liver Toxicity of Pterocephalus hookeri Extract via Triggering Necrosis" Toxins 11, no. 3: 142. https://doi.org/10.3390/toxins11030142
APA StyleWang, R., Dong, Z., Zhang, X., Mao, J., Meng, F., Lan, X., Liao, Z., & Chen, M. (2019). Evaluation of the Liver Toxicity of Pterocephalus hookeri Extract via Triggering Necrosis. Toxins, 11(3), 142. https://doi.org/10.3390/toxins11030142