Staphylococcal Enterotoxin C Is an Important Virulence Factor for Mastitis
Abstract
1. Introduction
2. Results
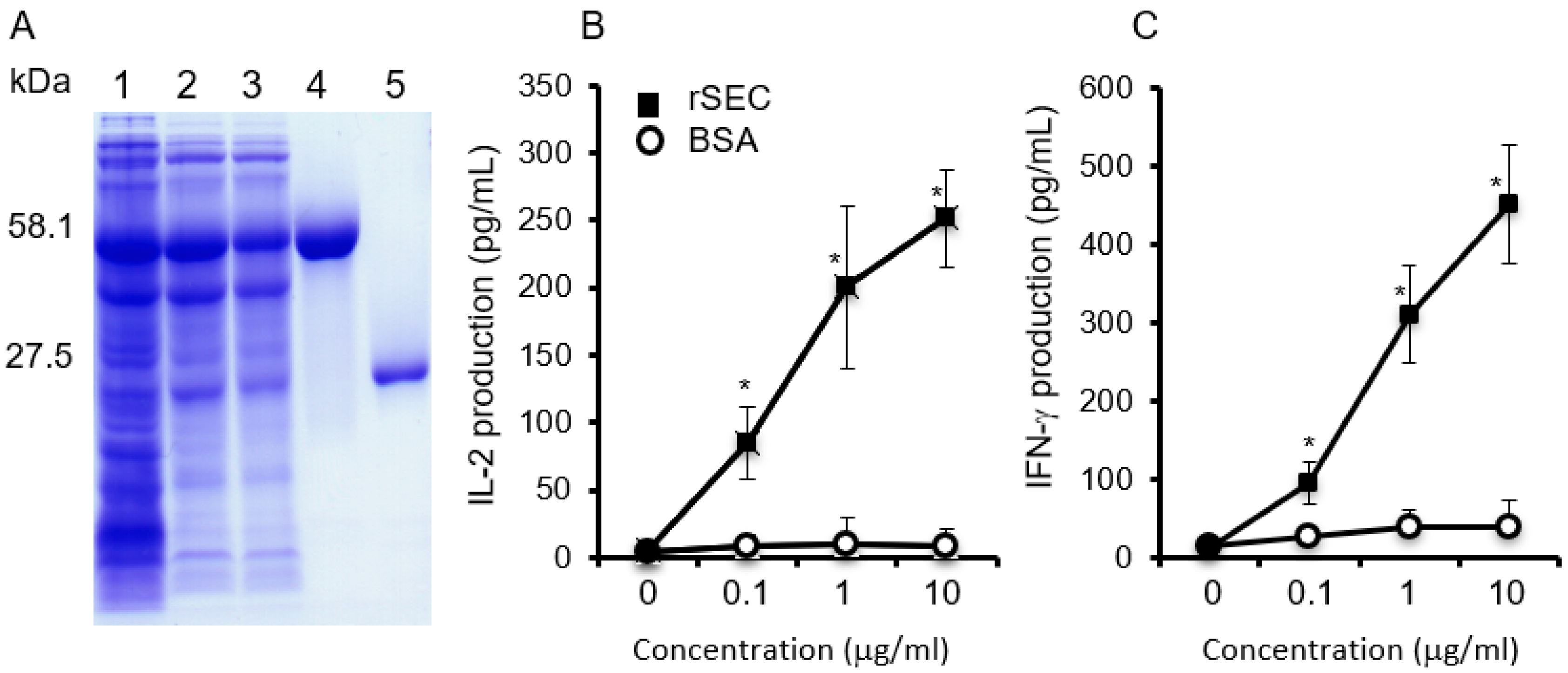
2.1. Biological Activities of Purified Recombinant SEC
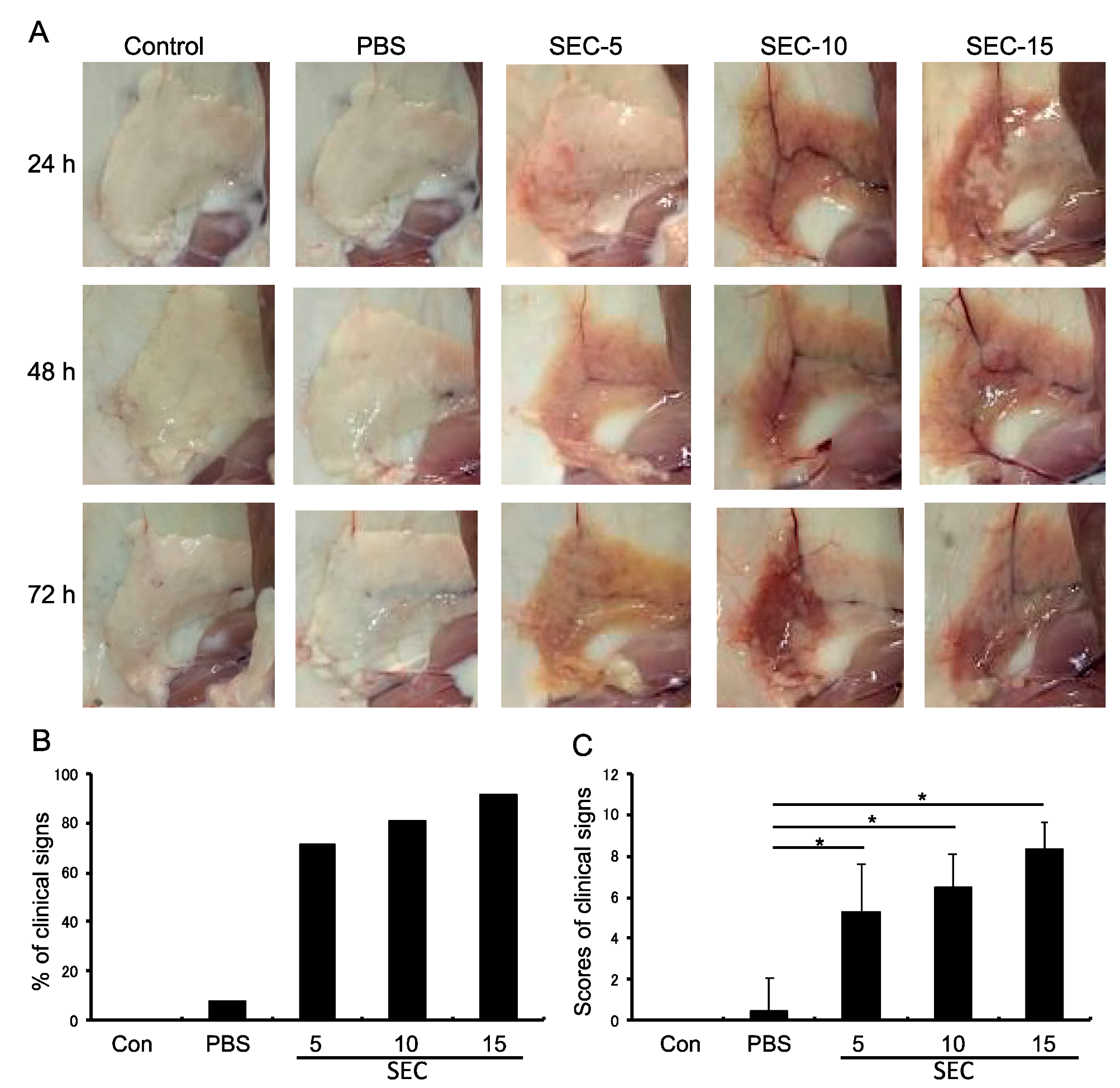
2.2. SEC Exhibited Pathogenic Activity in the Murine Mammary Glands
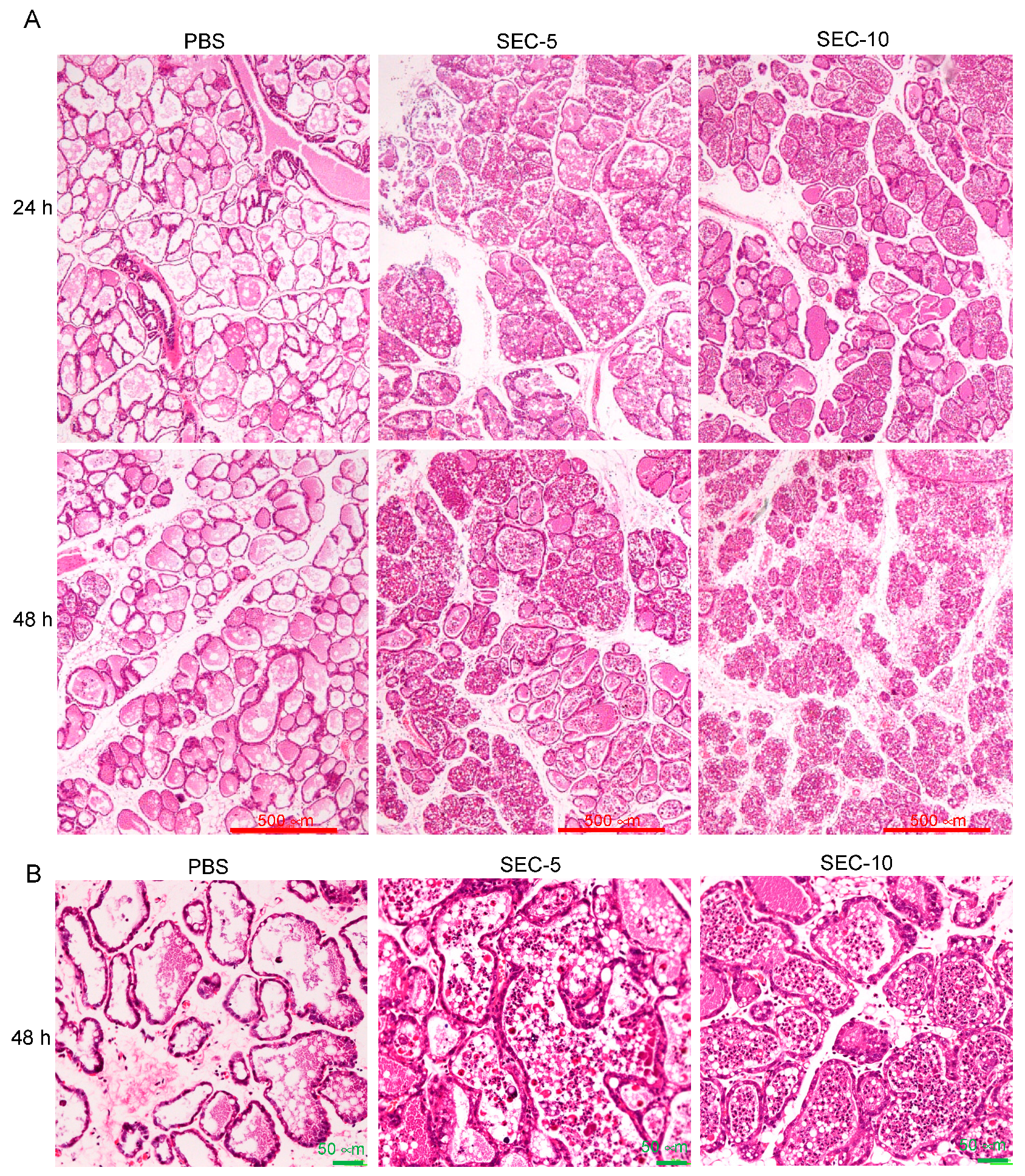
2.3. Histopathological Changes of SEC-Injected Mammary Glands
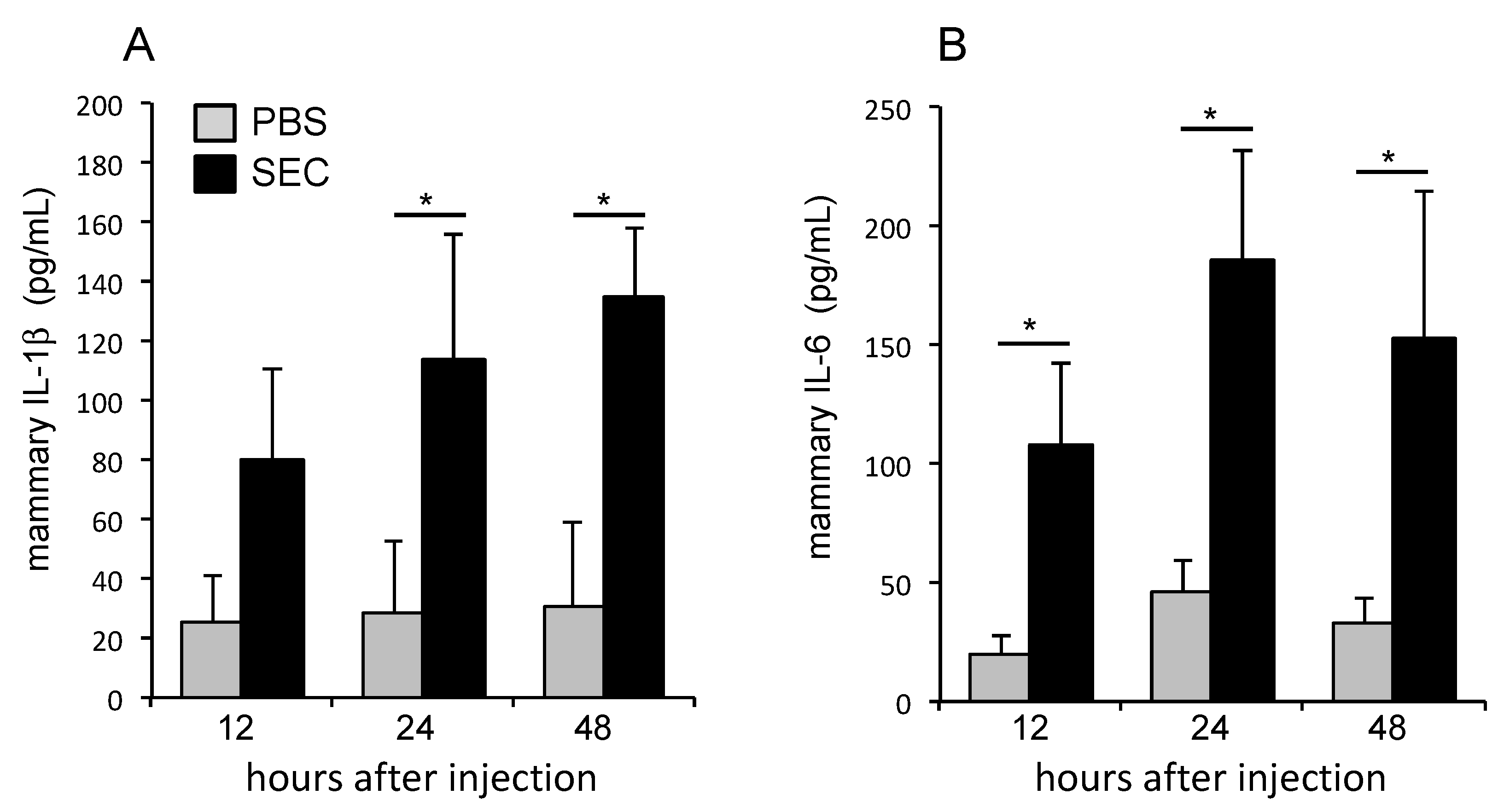
2.4. SEC Induced the Production of Proinflammatory Cytokines
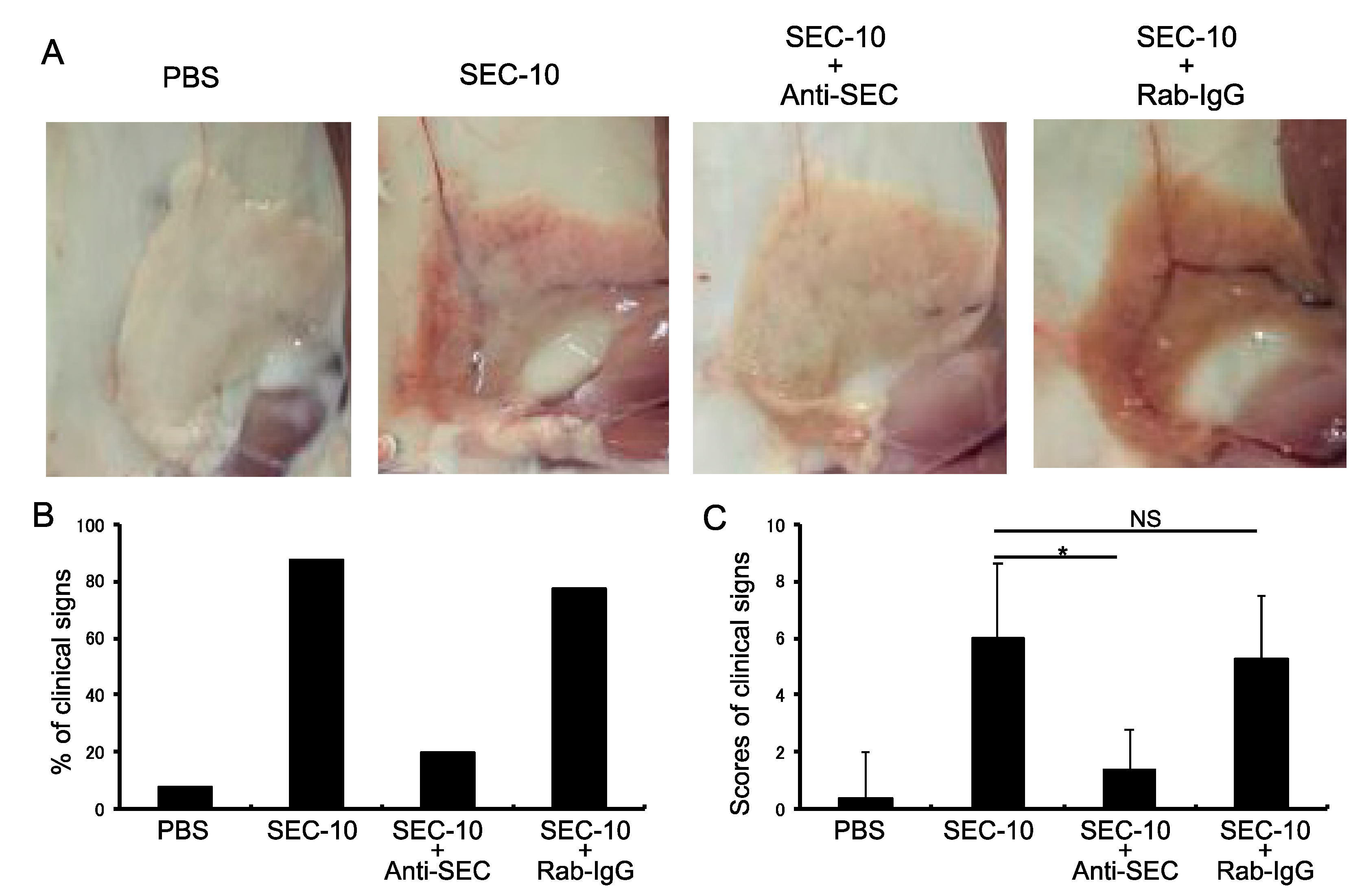
2.5. Anti-SEC Antibody Inhibits the Inflammation of Mammary Gland Induced by SEC
3. Discussion
4. Materials and Methods
4.1. Expression and Purification of SEC
4.2. Assay of Superantigenic Activity of SEC
4.3. Intramammary Inoculation Model
4.4. Clinical Evaluation
4.5. Histopathological Examination
4.6. Determination of Cytokines
4.7. Neutralization Assay of the Anti-SEC Antibody
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sánchez, A.; Sierra, D.; Luengo, C.; Corrales, J.C.; de la Fe, C.; Morales, C.T.; Contreras, A.; Gonzalo, C. Evaluation of the MilkoScan FT 6000 milk analyzer for determining the freezing point of goat’s milk under different analytical conditions. J. Dairy Sci. 2007, 90, 3153–3161. [Google Scholar] [CrossRef] [PubMed]
- Gogoi-Tiwari, J.; Williams, V.; Waryah, C.B.; Costantino, P.; Al-Salami, H.; Mathavan, S.; Wells, K.; Tiwari, H.K.; Hegde, N.; Isloor, S.; et al. Mammary gland pathology subsequent to acute infection with strong versus weak biofilm forming Staphylococcus aureus bovine mastitis isolates: A pilot study using non-invasive mouse mastitis model. PLoS ONE 2017, 27, e0170668. [Google Scholar] [CrossRef] [PubMed]
- White, D.G.; McDermott, P.F. Biocides, drug resistance and microbial evolution. Curr. Opin. Microbiol. 2001, 4, 313–317. [Google Scholar] [CrossRef]
- Artursson, K.; Söderlund, R.; Liu, L.; Monecke, S.; Schelin, J. Genotyping of Staphylococcus aureus in bovine mastitis and correlation to phenotypic characteristics. Vet. Microbiol. 2016, 25, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Schabauer, A.; Pinior, B.; Gruber, C.M.; Firth, C.L.; Käsbohrer, A.; Wagner, M.; Rychli, K.; Obritzhauser, W. The relationship between clinical signs and microbiological species, spa type, and antimicrobial resistance in bovine mastitis cases in Austria. Vet. Microbiol. 2018, 227, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Lacasse, P. Mammary tissue damage during bovine mastitis: Causes and control. J. Anim. Sci. 2008, 86 (Suppl. 13), 57–65. [Google Scholar] [CrossRef] [PubMed]
- Tenhagen, B.A.; Edinger, D.; Baumgärtner, B.; Kalbe, P.; Klünder, G.; Heuwieser, W. Efficacy of a herd-specific vaccine against Staphylococcus aureus to prevent post-partum mastitis in dairy heifers. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2001, 48, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Shkreta, L.; Talbot, B.G.; Diarra, M.S.; Lacasse, P. Immune responses to a DNA/protein vaccination strategy against Staphylococcus aureus induced mastitis in dairy cows. Vaccine 2004, 15, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.L. Vaccination against experimental staphylococcal mastitis in dairy heifers. Res. Vet. Sci. 1992, 53, 346–353. [Google Scholar] [CrossRef]
- Chang, B.S.; Moon, J.S.; Kang, H.M.; Kim, Y.I.; Lee, H.K.; Kim, J.D.; Lee, B.S.; Koo, H.C.; Park, Y.H. Protective effects of recombinant staphylococcal enterotoxin type C mutant vaccine against experimental bovine infection by a strain of Staphylococcus aureus isolated from subclinical mastitis in dairy cattle. Vaccine 2008, 16, 2081–2091. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.J.; Tuffs, S.W.; Wee, B.A.; Seo, K.S.; Park, N.; Connelley, T.; Guinane, C.M.; Morrison, W.I.; Fitzgerald, J.R. Bovine Staphylococcus aureus superantigens stimulate the entire T cell repertoire of cattle. Infect. Immun. 2018, 25, e00505–e00518. [Google Scholar] [CrossRef] [PubMed]
- Krakauer, T. Induction of CC chemokines in human peripheral blood mononuclear cells by staphylococcal exotoxins and its prevention by pentoxifylline. J. Leukoc. Biol. 1999, 66, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Fraser, J.D.; Proft, T. The bacterial superantigen and superantigen-like proteins. Immunol. Rev. 2008, 225, 226–243. [Google Scholar] [CrossRef] [PubMed]
- Spaulding, A.R.; Salgado-Pabón, W.; Kohler, P.L.; Horswill, A.R.; Donald, Y.M.; Leung, D.Y.M.; Schlievert, P.M. Staphylococcal and streptococcal superantigen exotoxins. Clin. Microbiol. Rev. 2013, 26, 422–447. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, T.; Saito, S.; Inoko, H.; Yan, X.J.; Imanishi, K.; Araake, M.; Igarashi, H. Relative activities of distinct isotypes of murine and human major histocompatibility complex class II molecules in binding toxic shock syndrome toxin 1 and determination of CD antigens expressed on T cells generated upon stimulation by the toxin. Infect. Immun. 1990, 58, 3877–3882. [Google Scholar] [PubMed]
- Yarwood, J.M.; McCormick, J.K.; Schlievert, P.M. Identification of a novel two-component regulatory system that acts in global regulation of virulence factors of Staphylococcus aureus. J. Bacteriol. 2001, 183, 1113–1123. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, J.R.; Monday, S.R.; Foster, T.J.; Bohach, G.A.; Hartigan, P.J.; Meaney, W.J.; Smyth, C.J. Characterization of a putative pathogenicity island from bovine Staphylococcus aureus encoding multiple superantigens. J. Bacteriol. 2001, 183, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Ebling, T.L.; Fox, L.K.; Bayles, K.W.; Bohach, G.A.; Byrne, K.M.; Davis, W.C.; Ferens, W.A.; Hillers, J.K. Bovine mammary immune response to an experimental intramammary infection with a Staphylococcus aureus strain containing a gene for staphylococcal enterotoxin C1. J. Dairy Sci. 2001, 84, 2044–2050. [Google Scholar] [CrossRef]
- Saiman, L.; O’Keefe, M.; Graham, P.L.; Wu, F.; Saïd-Salim, B.; Kreiswirth, B.; LaSala, A.; Schlievert, P.M.; Della-Latta, P. Hospital transmission of community-acquired methicillin-resistant Staphylococcus aureus among postpartum women. Clin. Infect. Dis. 2003, 15, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Zecconi, A.; Cesaris, L.; Liandris, E.; Daprà, V.; Piccinini, R. Role of several Staphylococcus aureus virulence factors on the inflammatory response in bovine mammary gland. Microb. Pathog. 2006, 40, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Günaydın, B.; Aslantaş, Ö.; Demir, C. Detection of superantigenic toxin genes in Staphylococcus aureus strains from subclinical bovine mastitis. Trop. Anim. Health Prod. 2011, 43, 1633–1637. [Google Scholar] [CrossRef] [PubMed]
- Aydin, A.; Sudagidan, M.; Muratoglu, K. Prevalence of staphylococcal enterotoxins, toxin genes and genetic-relatedness of foodborne Staphylococcus aureus strains isolated in the Marmara Region of Turkey. Int. J. Food Microbiol. 2011, 148, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Morandi, S.; Brasca, M.; Lodi, R.; Cremonesi, P.; Castiglioni, B. Detection of classical enterotoxins and identification of enterotoxin genes in Staphylococcus aureus from milk and dairy products. Vet. Microbiol. 2007, 124, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.C.; Zhang, B.J.; Lin, Y.C.; Wang, Q.K.; Qian, A.D.; Nakane, A.; Hu, D.L.; Tong, G.Z. Protective effect of glutathione S-transferase-fused mutant staphylococcal enterotoxin C against Staphylococcus aureus-induced bovine mastitis. Vet. Immunol. Immunopathol. 2010, 135, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, W.; Ali, T.; Alkasir, R.; Yin, J.; Liu, G.; Han, B. Staphylococcal enterotoxin H induced apoptosis of bovine mammary epithelial cells in vitro. Toxins (Basel) 2014, 6, 3552–3567. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Lin, X.; Jiang, T.; Peng, Z.; Xu, J.; Yi, L.; Li, F.; Fanning, S.; Baloch, Z. Prevalence and characterization of Staphylococcus aureus cultured from raw milk taken from dairy cows with mastitis in Beijing, China. Front. Microbiol. 2018, 9, 1123. [Google Scholar] [CrossRef] [PubMed]
- Piccinini, R.; Borromeo, V.; Zecconi, A. Relationship between S. aureus gene pattern and dairy herd mastitis prevalence. Vet. Microbiol. 2010, 145, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Orwin, P.M.; Fitzgerald, J.R.; Leung, D.Y.; Gutierrez, J.A.; Bohach, G.A.; Schlievert, P.M. Characterization of Staphylococcus aureus enterotoxin L. Infect. Immun. 2003, 71, 2916–2919. [Google Scholar] [CrossRef] [PubMed]
- Kenny, K.; Reiser, R.F.; Bastida-Corcuera, F.D.; Norcross, N.L. Production of enterotoxins and toxic shock syndrome toxin by bovine mammary isolates of Staphylococcus aureus. J. Clin. Microbiol. 1993, 31, 706–707. [Google Scholar] [PubMed]
- Franck, K.T.; Gumpert, H.; Olesen, B.; Larsen, A.R.; Petersen, A.; Bangsborg, J.; Albertsen, P.; Westh, H.; Bartels, M.D. Staphylococcal aureus enterotoxin C and enterotoxin-like L associated with post-partum mastitis. Front. Microbiol. 2017, 8, 173. [Google Scholar] [CrossRef] [PubMed]
- Gogoi-Tiwari, J.; Williams, V.; Waryah, C.B.; Mathavan, S.; Tiwari, H.K.; Costantino, P.; Mukkur, T. Intramammary immunization of pregnant mice with staphylococcal protein A reduces the post-challenge mammary gland bacterial load but not pathology. PLoS ONE 2016, 11, e0148383. [Google Scholar] [CrossRef] [PubMed]
- Heald, C.W. Morphometric study of experimentally induced Staphylococcus bovis mastitis in the cow. Am. J. Vet. Res. 1979, 40, 1294–1298. [Google Scholar] [PubMed]
- Nickerson, S.C.; Heald, C.W. Histopathologic response of the bovine mammary gland to experimentally induced Staphylococcus aureus infection. Am. J. Vet. Res. 1981, 42, 1351–1355. [Google Scholar] [PubMed]
- Paape, M.J.; Bannerman, D.D.; Zhao, X.; Lee, J.W. The bovine neutrophil: Structure and function in blood and milk. Vet. Res. 2003, 34, 597–627. [Google Scholar] [PubMed]
- Bannerman, D.D.; Paape, M.J.; Lee, J.W.; Zhao, X.; Hope, J.C.; Rainard, P. Escherichia coli and Staphylococcus aureus elicit differential innate immune responses following intramammary infection. Clin. Diagn. Lab. Immunol. 2004, 11, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Riollet, C.; Rainard, P.; Poutrel, B. Cells and cytokines in inflammatory secretions of bovine mammary gland. Adv. Exp. Med. Biol. 2000, 480, 247–258. [Google Scholar] [PubMed]
- Lee, J.W.; Bannerman, D.D.; Paape, M.J.; Huang, M.K.; Zhao, X. Characterization of cytokine expression in milk somatic cells during intramammary infections with Escherichia coli or Staphylococcus aureus by real-time PCR. Vet. Res. 2006, 37, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Kauf, A.C.; Vinyard, B.T.; Bannerman, D.D. Effect of intramammary infusion of bacterial lipopolysaccharide on experimentally induced Staphylococcus aureus intramammary infection. Res. Vet. Sci. 2007, 82, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, Y.; Mikami, O.; Yoshioka, M.; Motoi, Y.; Ito, T.; Ishikawa, Y.; Fuse, M.; Nakano, K.; Yasukawa, K. Elevated levels of tumor necrosis factor-alpha (TNF-alpha) and interleukin-6 (IL-6) activities in the sera and milk of cows with naturally occurring coliform mastitis. Res. Vet. Sci. 1997, 62, 297–298. [Google Scholar] [CrossRef]
- Messmer, U.K.; Briner, V.A.; Pfeilschifter, J. Tumor necrosis factor-alpha and lipopolysaccharide induce apoptotic cell death in bovine glomerular endothelial cells. Kidney Int. 1999, 55, 2322–2337. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.; Redmon, N.; Mazzio, E.; Soliman, K.F. Apigenin inhibits TNFα/IL-1α-induced CCL2 release through IKBK-epsilon signaling in MDA-MB-231 human breast cancer cells. PLoS ONE 2017, 12, e0175558. [Google Scholar] [CrossRef] [PubMed]
- Vieira, S.M.; Lemos, H.P.; Grespan, R.; Napimoga, M.H.; Dal-Secco, D.; Freitas, A.; Cunha, T.M.; Verri, W.A., Jr.; Souza-Junior, D.A.; Jamur, M.C.; et al. A crucial role for TNF-alpha in mediating neutrophil influx induced by endogenously generated or exogenous chemokines, KC/CXCL1 and LIX/CXCL5. Br. J. Pharmacol. 2009, 158, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.L.; Omoe, K.; Sasaki, S.; Yokomizo, Y.; Sashinami, H.; Sakuraba, H.; Shinagawa, K.; Nakane, A. Vaccination with nontoxic TSST-1 protects Staphylococcus aureus infection. J. Infect. Dis. 2003, 188, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.-L.; Omoe, K.; Sashinami, H.; Shinagawa, K.; Nakane, A. Immunization with nontoxic mutant staphylococcal enterotoxin A, D227A, protects against enterotoxin-induced emesis in house musk shrews. J. Infect. Dis. 2009, 199, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Omoe, K.; Hu, D.L.; Takahashi-Omoe, H.; Nakane, A.; Shinagawa, K. Comprehensive analysis of classical and newly described staphylococcal superantigenic toxin genes in Staphylococcus aureus isolates. FEMS Microbiol. Lett. 2005, 246, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.J.; Cui, J.C.; Zhang, X.; Qiang, A.D.; Tong, G.Z.; Hu, D.L.; Nakane, A.; Wang, Q.K. Development and evaluation of Staphylococcus aureus-induced mastitis model. Chin. J. Prevent. Vet. Med. 2009, 31, 365–369. [Google Scholar]
- Hu, D.L.; Cui, J.C.; Omoe, K.; Sashinami, H.; Yokomizo, Y.; Shinagawa, K.; Nakane, A. A mutant of staphylococcal enterotoxin C devoid of bacterial superantigenic activity elicits a Th2 immune response for protection against Staphylococcus aureus infection. Infect. Immun. 2005, 73, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Brouillette, E.; Grondin, G.; Lefebvre, C.; Talbot, B.G.; Malouin, F. Mouse mastitis model of infection for antimicrobial compound efficacy studies against intracellular and extracellular forms of Staphylococcus aureus. Vet. Microbiol. 2004, 101, 253–262. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, R.; Cui, J.; Cui, T.; Guo, H.; Ono, H.K.; Park, C.-H.; Okamura, M.; Nakane, A.; Hu, D.-L. Staphylococcal Enterotoxin C Is an Important Virulence Factor for Mastitis. Toxins 2019, 11, 141. https://doi.org/10.3390/toxins11030141
Fang R, Cui J, Cui T, Guo H, Ono HK, Park C-H, Okamura M, Nakane A, Hu D-L. Staphylococcal Enterotoxin C Is an Important Virulence Factor for Mastitis. Toxins. 2019; 11(3):141. https://doi.org/10.3390/toxins11030141
Chicago/Turabian StyleFang, Rendong, Jingchun Cui, Tengteng Cui, Haiyong Guo, Hisaya K. Ono, Chun-Ho Park, Masashi Okamura, Akio Nakane, and Dong-Liang Hu. 2019. "Staphylococcal Enterotoxin C Is an Important Virulence Factor for Mastitis" Toxins 11, no. 3: 141. https://doi.org/10.3390/toxins11030141
APA StyleFang, R., Cui, J., Cui, T., Guo, H., Ono, H. K., Park, C.-H., Okamura, M., Nakane, A., & Hu, D.-L. (2019). Staphylococcal Enterotoxin C Is an Important Virulence Factor for Mastitis. Toxins, 11(3), 141. https://doi.org/10.3390/toxins11030141



