The Diversified O-Superfamily in Californiconus californicus Presents a Conotoxin with Antimycobacterial Activity
Abstract
1. Introduction
2. Results
2.1. Purification of O1_cal29b
2.2. Identification of cDNA Clone Encoding O1_cal29b Precursor
2.3. Transcriptomic Analysis for Members of O-Superfamily
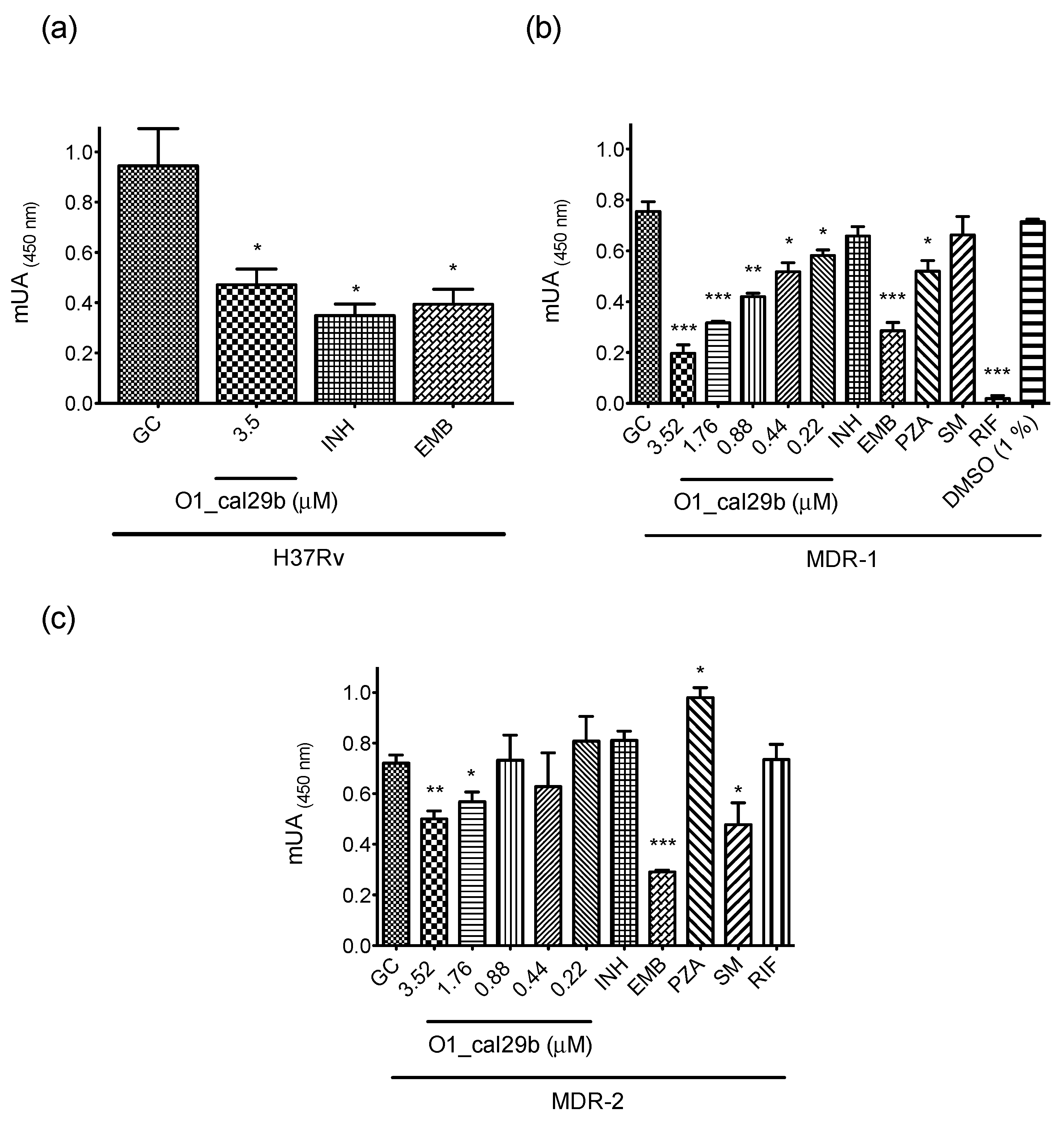
2.4. Antimycobacterial Susceptibility Assay
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Venom Purification
5.2. Peptide Purification
5.3. 3´RACE
5.4. 5´RACE
5.5. Transcriptome of the Venom Gland
5.5.1. Total RNA Extraction
5.5.2. RNA-Seq Library and Venom Duct Transcriptome Assembly
5.5.3. Bioinformatic Analysis of Conotoxin Identification
5.6. Antimycobacterial Susceptibility Assay
5.7. Minimal Inhibitory Concentration (MIC) Assay
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Biggs, J.S.; Watkins, M.; Puillandre, N.; Ownby, J.P.; Lopez, E.V.; Christensen, S.; Juarez, K.M.; Bernaldez, J.; Licea, A.N.; Showers, P.C.; et al. Evolution of Conus peptide toxins: analysis of Conus californicus Reeve, 1844. Mol. Phylogenet. Evol. 2010, 56, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gilly, W.F.; Richmond, T.A.; Duda, T.F.; Elliger, C., Jr.; Lebaric, Z.; Schulz, J.; Bingham, J.P.; Sweedler, J.V. A diverse of novel peptide toxins from an unusual cone snail, Conus californicus. J. Exp. Biol. 2011, 214, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Chao, P.; Jiaan, Y.; Yunhai, Y.; Junqing, Z.; Qiong, S. Cone snails: A big store of conotoxins for novel drug discovery. Toxins 2017, 9, 397. [Google Scholar] [CrossRef] [PubMed]
- Dutertre, S.; Jin, A.; Kaas, Q.; Jones, A.; Alewood, P.F.; Lewis, R.J. Deep Venomics Reveals the Mechanism for Expanded Peptide Diversity in Cone Snail Venom. Mol. Cell. Proteom. 2013, 12, 312–329. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, L.K.E.; Bernaldez, S.J.; Licea, A. Effect of two synthetic disulfide bond variants of a 13-mer toxin from Conus californicus on the transcription of pro-inflammatory cytokines induced by LPS. Toxicon 2013, 70, 82–85. [Google Scholar] [CrossRef] [PubMed]
- De León, N.M.A.; Romero, N.E.; Luna, N.A.; Bernaldez, S.J.; Sánchez, C.L.N.; Licea, N.A.F.; Morales, M.J.; Muñiz, H.S. In vitro effect of the synthetic cal14.1ª conotxin, derived from Conus californicus, on the human parasite Toxoplasma gondii. Mar. Drugs 2016, 8, 66. [Google Scholar] [CrossRef]
- Oroz, P.I.; Navarro, M.; Cervantes, L.K.E.; Alvarez, D.C.; Salvesen, G.; Sanchez, C.L.N.; Licea, N.A.F. Apoptosis activation in human lung cancer cell lines by a novel synthetic peptide derived from Conus californicus venom. Toxins 2016, 8, 38. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, A.M.; Bernaldez, J.; Jimenez, S.; Ueberhide, B.; González, L.J.; Licea, A.N. Antimycobacterial activity: A new pharmacological target for conotoxins found in the first reported conotoxin from Conasprella ximenes. Toxins 2018, 10, 51. [Google Scholar] [CrossRef]
- Global Tuberculosis Report 2018. World Health Organization (WHO). Available online: http://www.who.int/tb/publications/global_report/en/ (accessed on 11 March 2018).
- Petersen, T.N.; Brunak, S.; von Heijine, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef]
- Lu, J.; Zhang, K.; Wang, S.; Sun, T.; Yu, S.; Dai, Q.; Liu, Z. Cloning, expression and functional characterization of a D-superfamily conotoxin Lt28.1 with previously undescribed cysteine pattern. Peptides 2017, 94, 64–70. [Google Scholar] [CrossRef]
- Bald, D.; Villegas, C.; Lu, P.; Koul, A. Targeting Energy Metabolism in Mycobacterium tuberculosis, a new paradigm in antimycobacterial drug discovery. mBio 2017, 8, e00272-17. [Google Scholar] [CrossRef] [PubMed]
- Abedinzade, M.; Gaeini, M.; Sardari, S. Natural antimicrobial peptides against Mycobacterium tuberculosis. J. Antimicrob. Chemother. 2015, 70, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Niederweis, M. Nutrient acquisition by mycobacteria. Microbiology 2008, 154, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Machado, D.; Pires, D.; Perdigao, J.; Couto, I.; Portugal, I.; Martins, M.; Amaral, L.; Anes, E.; Viveiros, M. Ion channel blockers as antimicrobial agents, efflux inhibitors, and enhancers of macrophage killing activity against drug resistant Mycobacterium tuberculosis. PLoS ONE 2016, 26, e0149326. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.J.; Dutertre, S.; Vetter, I.; MacDonald, J.C. Conus venom peptide pharmacology. Pharmacol. Rev. 2012, 64, 259–298. [Google Scholar] [CrossRef] [PubMed]
- Perumal, R.S.; Stiles, B.G.; Franco, O.L.; Sethi, G.; Lim, L.H.K. Animal venom as antimicrobial agents. Biochem. Pharmacol. 2017, 134, 127–138. [Google Scholar] [CrossRef]
- Lebbe, E.K.M.; Ghequire, M.G.K.; Peigneur, S.; Mille, B.G.; Devi, P.; Ravichandran, S.; Waelkens, E.; D’Souza, L.; De Mot, R.; Tytgat, J. Novel Conopeptides of Largely Unexplored Indo Pacific Conus sp. Mar. Drugs 2016, 14, 199. [Google Scholar] [CrossRef]
- Hemu, X.; Tam, J.P. Macrocyclic antimicrobial peptides engineered from ω-conotoxin. Curr. Pharm. Des. 2017, 23, 2131–2138. [Google Scholar] [CrossRef]
- Palanisamy, S.K.; Dhanabalan, S.K.; Sundaresan, U. Recent developments and chemical diversity of Cone snails with special reference to Indian Cone snails. In Blue Biotechnology: Production and Use of Marine Molecules, 1st ed.; La Barre, S., Bates, S.S., Eds.; Wiley-CVH Verlag GmmbH & Co. KGaA: Hoboken, NJ, USA, 2018; Volume 1, pp. 443–483. [Google Scholar]
- Robinson, S.D.; Norton, R.S. Conotoxin gene superfamilies. Marine Drugs 2014, 12, 6058–6101. [Google Scholar] [CrossRef]
- Hu, H.; Bandyopadhyay, P.K.; Olivera, B.M.; Yandell, M. Elucidation of the molecular envenomation strategy of the cone snail Conus geographus through transcriptome sequencing of its venom duct. BMC Genom. 2012, 13, 284. [Google Scholar] [CrossRef]
- Walker, C.S.; Steel, D.; Jacobsen, R.B.; Lirazan, M.B.; Cruz, L.J.; Hooper, D.; Shetty, R.; DelaCruz, R.C.; Nielsen, J.S.; Zhou, L.M.; et al. The T-superfamily of Conotoxins. J. Biol. Chem. 1999, 274, 30664–30671. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Haas, B.J.; Papanicolaou, A.; Yassour, M.; Grabherr, M.; Blood, P.D.; Bowden, J.; et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 2013, 8, 1494–1512. [Google Scholar] [CrossRef] [PubMed]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, H. Predicting secretory proteins with SignalP. Methods Mol. Biol. 2017, 1611, 59–73. [Google Scholar] [CrossRef]
- Robinson, S.D.; Safavi-Hemami, H.; McIntosh, L.D.; Purcell, A.W.; Norton, R.S.; Papenfuss, A.T. Diversity of Conotoxin Gene Superfamilies in the Venomous Snail, Conus victoriae. PLoS ONE 2014, 9, e87648. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Yao, G.; Gao, B.-M.; Fan, C.-X.; Bian, C.; Wang, J.; Cao, Y.; Wen, B.; Zhu, Y.; Ruan, Z.; et al. High-throughput identification of novel conotoxins from the Chinese tubular cone snail (Conus betulinus) by multi-transcriptome sequencing. Gigascience 2016, 5, 17. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Sean, E.R. Accelerated profile HMM searches. PLoS Comp. Biol. 2011, 7, e1002195. [Google Scholar] [CrossRef]
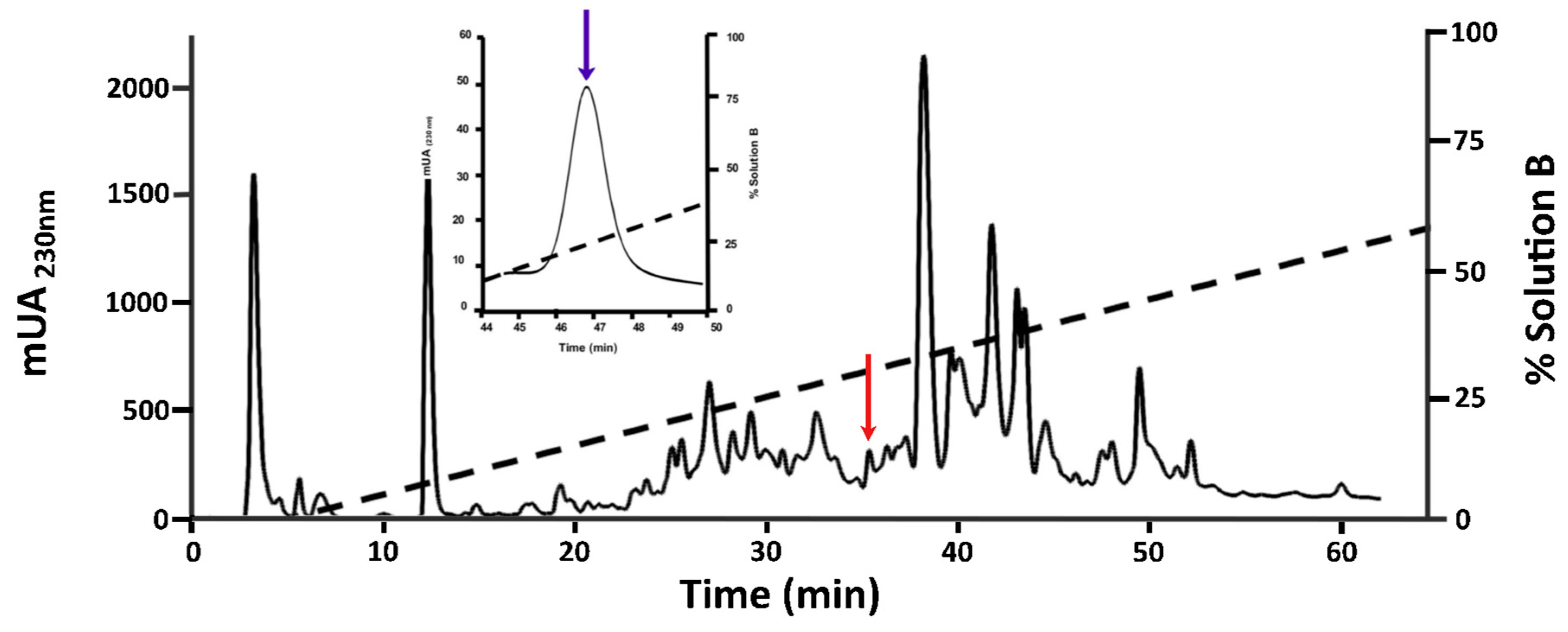
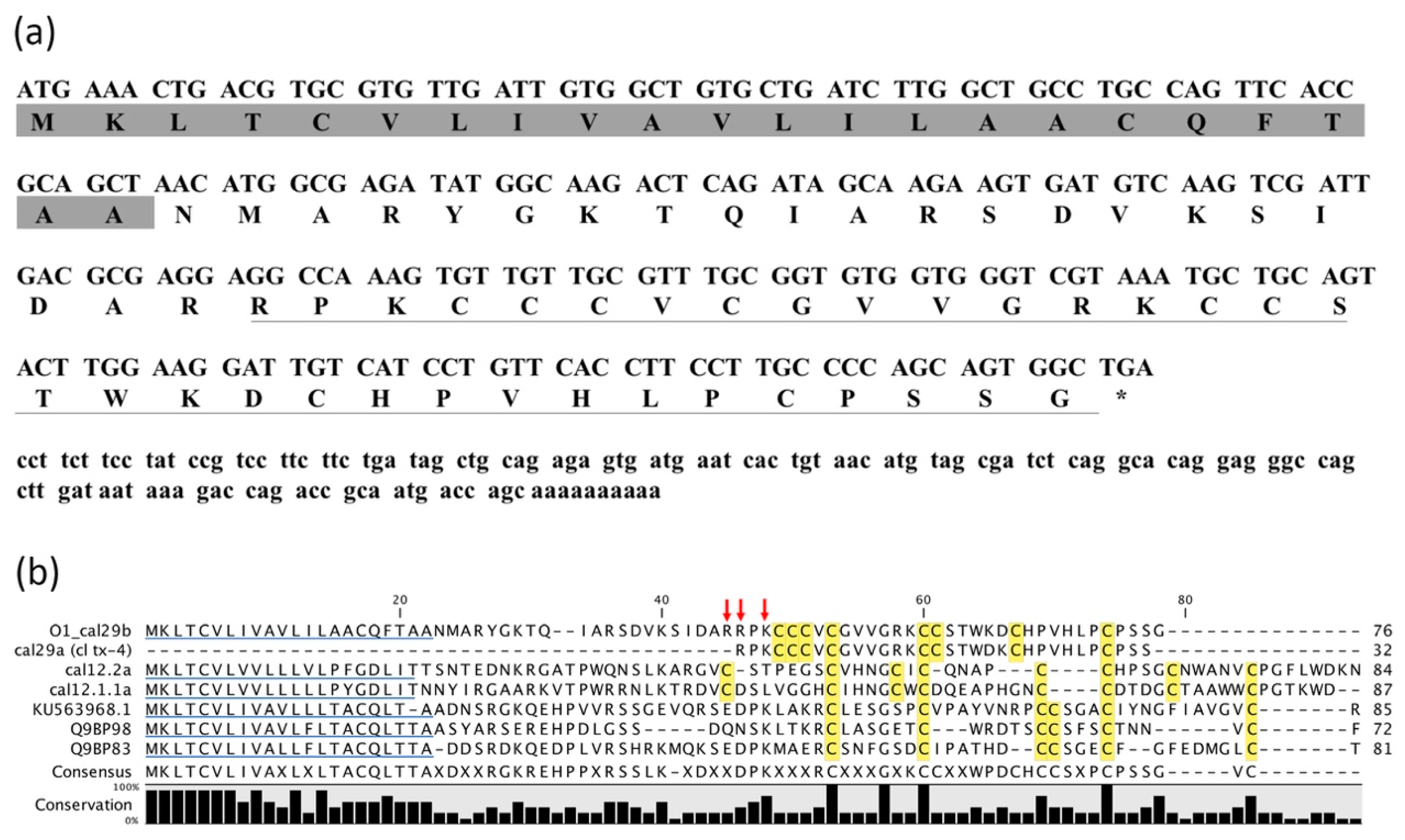
| (a) | ||||
| Name | Cysteine Framework | Signal peptide | Toxin | GenBank # |
| O1_cal1.2 | I | MKLTCVFIIAVLILTACHFIVAD | AGCCPTIMYKTGACRTNRC | ADD97803.1 |
| O1_cl6b/cl6.2 | VI/VII | MKLTCVLIIAVLILTACQFIAAD | NCIPKNHFCGLLHHSRNCCTPTCLIVCF | ADB93121.1 |
| O1_cal6.1b | VI/VII | MKLTTVLVVALLVLAACQFTVTD | CLAGSARCEFHKPSTCCSGHCIFWWCA | ADB93119.1 |
| O1_cl6.6a | VI/VII | MKLTCVLIAAVLLLAVCQLDSADAT | TRGCKSKGSFCWNGIECCGGNCFFACIY | ADB93125.1 |
| O1_cl6.10 | VI/VII | MKLTCVLIAAVLLLAVCQLDSADAT | TRGCKTKGTWCWASRECCLKDCLFVCVY | ADB93112.1 |
| O1_cl6.5 | VI/VII | MKLTCVLIVAVLVLTACQFTAAI | CIPDHHGCGLLHHSRYCCNGTCFFVCIP | ADB93124.1 |
| O1_cal6.1a | VI/VII | MKLTTVLVVALLVLAACQFTVTD | CLAGSARCEFHKPSSCCSGHCIFWWCA | ADB93120.1 |
| O1_cl6.3 | VI/VII | MKLTTVLIVAVLVLAACQFTVTD | GLSRPSKGCIGGGDPCEFHRGYTCCSEHCIIWVCA | ADB93122.1 |
| O1_cal6.1e | VI/VII | MKLTTVLIVAVLVLAACQFTVTD | CIGGGDPCEFHRGYTCCSEHCIIWVCA | ADB04242.1 |
| O1_cal6.1a | VI/VII | MKLTTVLVVALLVLAACQFTVTD | CLAGSARCEFHKPSSCCSGHCIFWWCA | ADB93120.1 |
| O1_cl6.6b | VI/VII | MKLTCVLIAAVLLLAVCQLDSADAT | TRGCKSKGSFCWNGIECCGGNCFFACVY | ADB93126.1 |
| O1_cal12.1.3a | XII | MKLTCVLVVLLLLLPYGDLI | DVCDSLVDGRCIHNGCFCEESKPNGNCCDTGGCVWWWCPGTKWD | ABR92964.1 |
| O1_cal12.2a | XII | MKLTCVLVVLLLLLPYGDLI | GVCSTPEGSCVHNGCICQNAPCCHPSGCNWANVCPGFLWDKN | ABR92966.1 |
| O1_cal12.1.2b | XII | MKLTCVLVVLLLLLPYGDLI | DVCDSLVDGRCIHNGCYCERDAPNGNCCNTDGCTARWWCPGTKWD | ABR92953.1 |
| O1_cl12.3 | XII | MKLTCVLVVLLLFLPYGDLI | DVCDSLVGGNCIHNGCWCDQEAPHGNCCDTDGCTAAWWCPGTKWD | ADB93095.1 |
| O1_cal12.1p1 | XII | MKLTCVLVVLLLLLPYGDLI | DVCKKSPGKCIHNGCFCEQDKPQGNCCDSGGCTVKWWCPGTKGD | AEC22829.1 |
| O1_cal12a | XII | MKLTCVLVVLLLLLPYGDLI | DVCDSLVGGHCIHNGCWCDQEAPHGNCCDTDGCTAAWWCPGTK | P0DJC1.1* |
| (b) | ||||
| Name | Cysteine Framework | Signal peptide | Toxin | |
| O1_cal6.18 | VI/VII | MKLTYVLIVAMLVLVVCRAD | CFGRGGLCTWFDPSVCCSGICTFVDCW | |
| O1_cal6.19 | VI/VII | MKVTCVLVLTLMALTVCQVATAY | CINVGMCIYDGYCCSNRCWGGMCSPWR | |
| O1_cal6.20 | VI/VII | MKLTCVLIVAVLILTACQVIAAD | GWFGEESSCWWCTGQNKCCEEAQVCQSVNYACPPARR | |
| O1_cal6.21 | VI/VII | MQLTHVLVVGLLVLTSFQPINAV | TNRVDCSAPEDKSEPGYWCGLEPLCCYSGKCFVICFGSKPAGT | |
| O1_cal6.22 | VI/VII | MKLTCVLIVAVLILTACQVIAAD | EADANRLSTRWCACGVNYYCCNEVCTWREDPCP | |
| O1_cal6.23 | VI/VII | MKLTAVLMVAVLVLTACQLITAN | ECSRKGEWCGLESVLCCNGGSWNCWFVCTA | |
| O1_cal6.24 | VI/VII | MKLTCVMIVAVLVLTVCKVVTSD | QLKKLRRECYLEPGDSCFHDDGRGACCEGTCFFGVACVPWS | |
| O1_cal6.25 | VI/VII | MKLTHVLIVAVLVLTVCHLTMAV | CKSGGQACWFLLKKHNCCSGYCIVAVCAG | |
| O1_cal6.26 | VI/VII | MKLTCVMIVAVLVLTVCKVVTSD | QLKKLRRECYLEPGDSCFHDDGRGACCEGTCLFGINCVASW | |
| O1_cal6.27 | VI/VII | MKLTCVLIAAMLLLAVCQLDSADAT | ETGCKKDGSWCWIPSECCIESCLITCWY | |
| O1_cal6.28 | VI/VII | MKLTCVLIVAVLILTACQVIAAD | EATNRATKRGCLMCWGSNVRCCEKANACVSINYECPKARR | |
| O1_cal6.29 | VI/VII | MKLTCVLIVAVLVLTACQFTAAI | SQTQRLSKKCIEDNHACGLLHHSPYCCNGTCFIVCIP | |
| O1_cal6.30 | VI/VII | MKVTCVLTLAVLILTIGQIANAD | STLGQRYCKASGSWCGIHKHRECCSGNCFFWCVYNGK | |
| O1_cal6.31 | VI/VII | MKLTCVLIAAVLLLAVCQLDSADAI | TRDCKTKGYACFASTECCVQDCWLVCLY | |
| O1_cal6.32 | VI/VII | MKLTCVLIVSVLILTACQFTAAV | DCHSTGYLCFWWHECCSNFCIPLQQRCF | |
| O1_cal6.33 | VI/VII | MKLTCVVIIAVLILTACQFTTAD | DCKPKNNLCLWSSECCSGICFPFAQRCT | |
| O1_cal6.34 | VI/VII | MKLTCVLIVAVLILTACQVIAAD | SSCWFCSTGFNKCCESTGDCMTYPSEYNASCPEA | |
| O1_cal6.35 | VI/VII | MKLTCVLIVAVLILTACQVIAAD | EAEATNRAIKRGWFGEESSCWWCTGFNKCCEAAAVCQSVNSACP | |
| O1_cal6.36 | VI/VII | MKVTCVLTLAVLILTVGQMVTAD | CRSPGSWCFYKHSNCCSGNCFLWCVQNGK | |
| O1_cal6.37 | VI/VII | MKLTCVMIVAVLLLTVCKVVTSD | QLKKLRRECYLEPGDSCFHHDGRGACCEGTCFFGVACVPW | |
| O1_cal6.38 | VI/VII | MKLTFVLIVAVLVLAVCNFTVAD | KANNAEAPEQEKRACTPNGSYCNILSGKLNCCSGWCLALICAG | |
| O1_cal12b | XII | MKLTCMLVVLLLVLPFGDLI | ANTGGLCGMPPGVCYPNGCACGQDTPCCHPSGCNRYNYCGPLLE | |
| O1_cal12c | XII | MKVTCVLVVLLLLLPYGDLLGN | SVCDFGSCVHNGCYCEEHRPCCTPGSCSSWWPRCPGSMMDP | |
| O1_cal12.2e | XII | MKLTCVLVVLLLVLPFGDLI | GVCSTPEGSCVHNGCICQNAPCCHPSGCNWVNVCPGFLWDRS | |
| O1_cal29b | XXIX | MKLTCVLIVAVLILAACQFTAAN | RPKCCCVCGVVGRKCCSTWKDCHPVHLPCPSSG | |
| O2_cal30 | XXX | MEKLIILLLVASLLVTTDSVVKGK | KAARGWLFNEVETCELGGLGDPCSGSGDCCCDQCLCSGSYEHCTQNPDRWFCCRTYGN | |
| O3_cal14d | XIV | MFRLGVFLLTFLLLVSMATSE | YSRGRIMARASECVNECVESGHNTFHCERHCSNT | |
| O3_cal6.1a | VI/VII | MSGSGAMLLGLLILVAMAT | SLDTREICWNHSECDDPSEWCCRMGSGHGSCLPVCRP | |
| O3_cal6.1b | VI/VII | MSGSGAMLLGLLILVAMAT | SLDTREICWNHSECDDPSEWCCRMGSGHGSCQPVCRP | |
| O3_cal6.1c | VI/VII | MSGSGAMLLGLLILVAMAT | SLDTREICWHQSECDDPNEWCCIMGTSYGSCQPVCRP | |
| O3_cal6.2 | VI/VII | MSGSGVLLLTLLLLVPLSAL | AKECSMYYCSGGDFCCPGLKCGDPTGKKICIEPGK | |
| O3_cal6.3 | VI/VII | MSGTTVLLLTCLFLVTMAT | SDCDLYDDSCTGTEICCTPPGDYQGNCMEGEDCPSGGR | |
| O3_cl6d | VI/VII | MSGTGVLLLTLLLLVTMATSD | DACSLLNGDDCGPGELCCTPSGDHQGTCETSCW | |
| O3_cal27 | XXVII | MSGTGVLLLTLLLLVAMAASD | MLSSLIQAHERDSEESCKSYGGGPCPSGEDCCCPPGRSTGTCKRTCNNGSVCA | |
| O3_contryphan-like cal1 | MTRTAVLLLTLLFLVAMAASD | KIKTREVCWTEEECENWE | ||
| O3_contryphan-like cal2 | MTRTAVLLLTLLFLVAMAASD | KIKTRELCWTEEECENWE | ||
| O3_contryphan-like cal3 | MTRTAVLLLTLLFLVAMAASD | KIKTRELCWSERECENGK | ||
| O3_contryphan-like cal4 | MTRTAVLLLTLLFLVAMAASD | KIKTREVCWNEEECENWE | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernáldez-Sarabia, J.; Figueroa-Montiel, A.; Dueñas, S.; Cervantes-Luévano, K.; Beltrán, J.A.; Ortiz, E.; Jiménez, S.; Possani, L.D.; Paniagua-Solís, J.F.; Gonzalez-Canudas, J.; et al. The Diversified O-Superfamily in Californiconus californicus Presents a Conotoxin with Antimycobacterial Activity. Toxins 2019, 11, 128. https://doi.org/10.3390/toxins11020128
Bernáldez-Sarabia J, Figueroa-Montiel A, Dueñas S, Cervantes-Luévano K, Beltrán JA, Ortiz E, Jiménez S, Possani LD, Paniagua-Solís JF, Gonzalez-Canudas J, et al. The Diversified O-Superfamily in Californiconus californicus Presents a Conotoxin with Antimycobacterial Activity. Toxins. 2019; 11(2):128. https://doi.org/10.3390/toxins11020128
Chicago/Turabian StyleBernáldez-Sarabia, Johanna, Andrea Figueroa-Montiel, Salvador Dueñas, Karla Cervantes-Luévano, Jesús A. Beltrán, Ernesto Ortiz, Samanta Jiménez, Lourival D. Possani, Jorge F. Paniagua-Solís, Jorge Gonzalez-Canudas, and et al. 2019. "The Diversified O-Superfamily in Californiconus californicus Presents a Conotoxin with Antimycobacterial Activity" Toxins 11, no. 2: 128. https://doi.org/10.3390/toxins11020128
APA StyleBernáldez-Sarabia, J., Figueroa-Montiel, A., Dueñas, S., Cervantes-Luévano, K., Beltrán, J. A., Ortiz, E., Jiménez, S., Possani, L. D., Paniagua-Solís, J. F., Gonzalez-Canudas, J., & Licea-Navarro, A. (2019). The Diversified O-Superfamily in Californiconus californicus Presents a Conotoxin with Antimycobacterial Activity. Toxins, 11(2), 128. https://doi.org/10.3390/toxins11020128







