FGF23 and Phosphate–Cardiovascular Toxins in CKD
Abstract
1. Introduction
2. FGF23 and its Functions in Phosphate Homeostasis
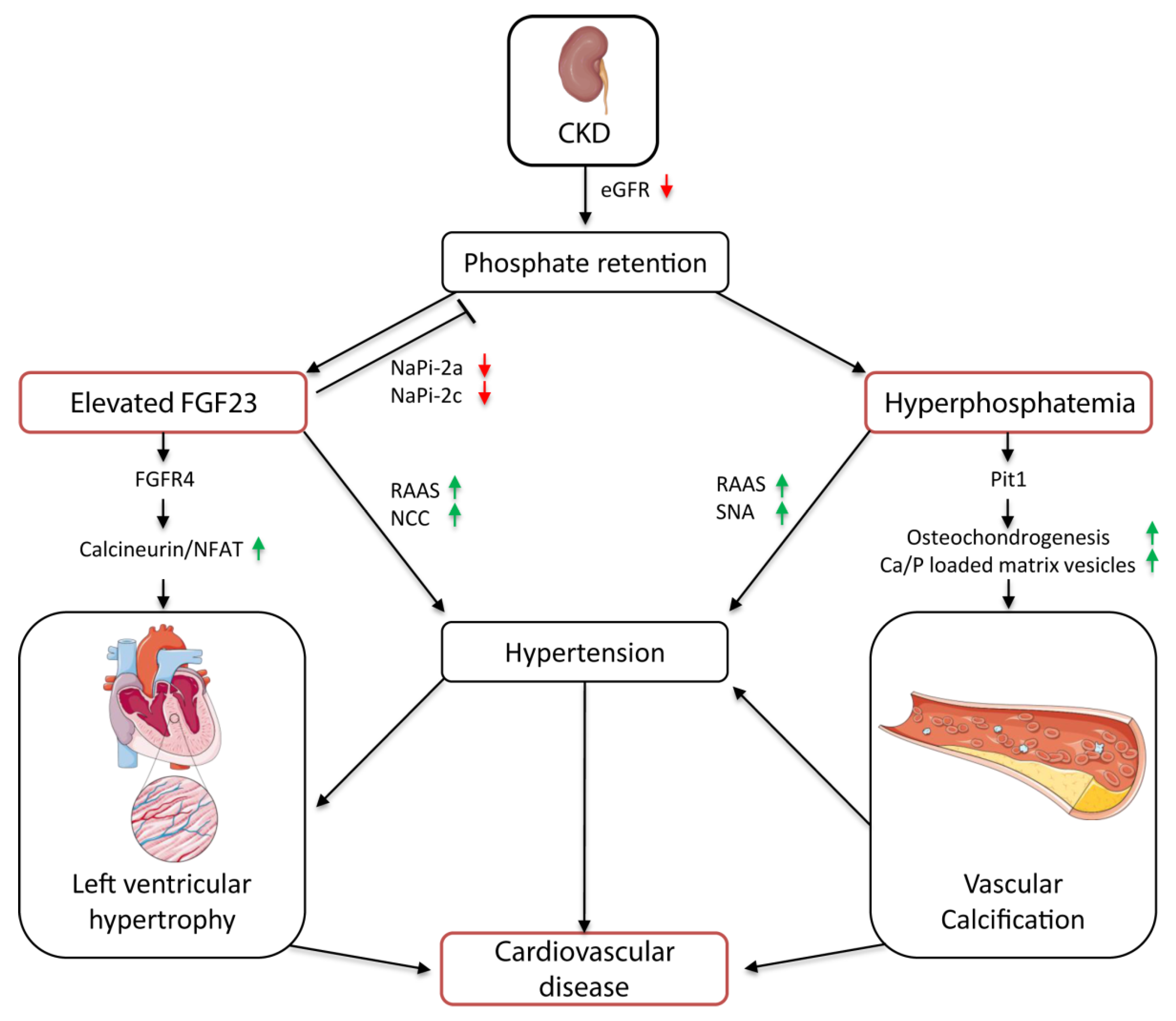
3. The Role of FGF23 and Phosphate Balance in CKD Progression
3.1. Early Stages of CKD
3.2. End-Stage Kidney Disease
4. The Role of FGF23 and Phosphate in CKD-Associated Cardiovascular Diseases
4.1. Hypertension
4.2. Vascular Calcification
4.3. Inflammation-Mediated Vascular Calcification
4.4. Left Ventricular Hypertrophy
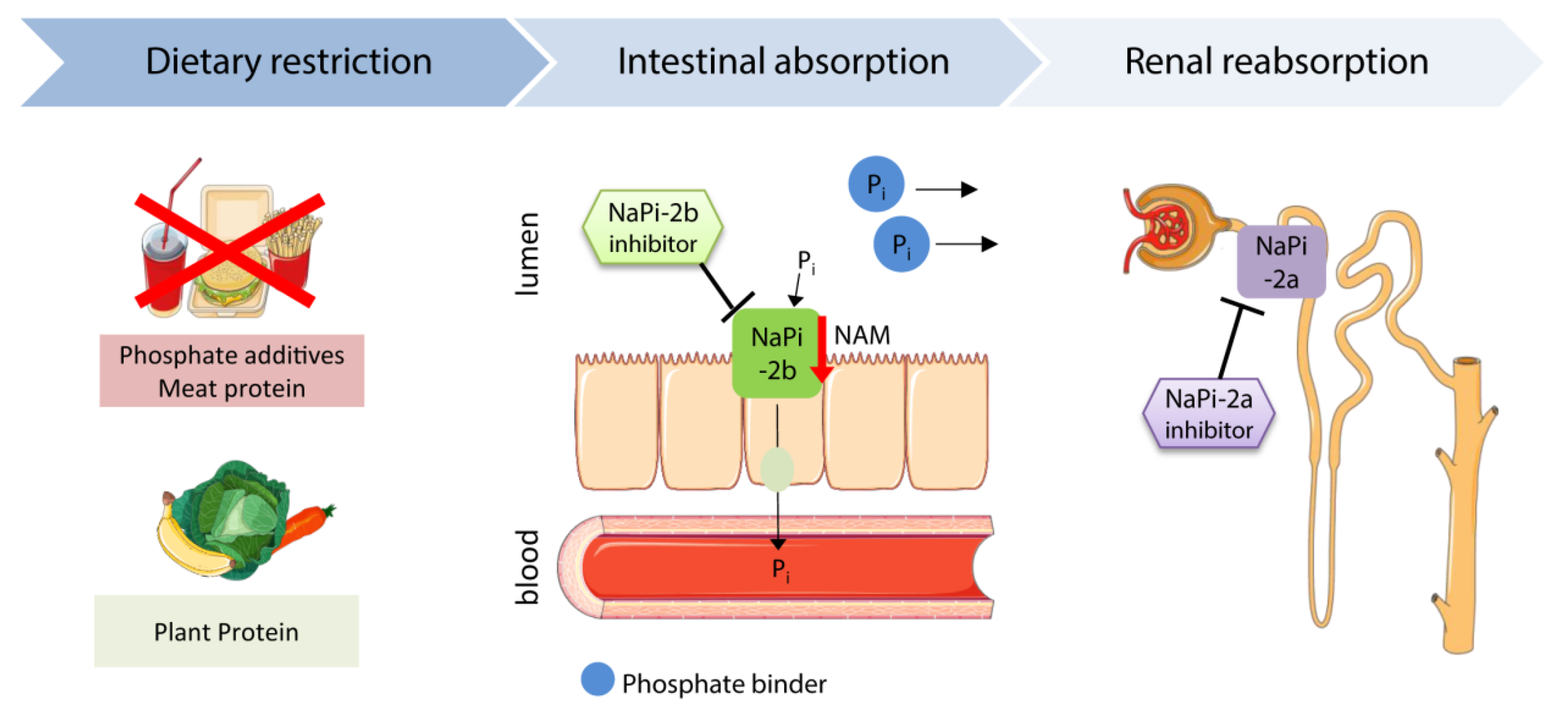
5. Therapeutic Approaches to Inhibit FGF23- and Phosphate-Mediated Cardiovascular Disease
5.1. Restriction of Dietary Phosphate Uptake
5.2. Phosphate Binder
5.3. Nicotinamide
5.4. Magnesium
5.5. Other Approaches
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Thompson, S.; James, M.; Wiebe, N.; Hemmelgarn, B.; Manns, B.; Klarenbach, S.; Tonelli, M. Cause of Death in Patients with Reduced Kidney Function. JASN 2015, 26, 2504–2511. [Google Scholar] [CrossRef] [PubMed]
- Jimbo, R.; Shimosawa, T. Cardiovascular Risk Factors and Chronic Kidney Disease—FGF23: A Key Molecule in the Cardiovascular Disease. Int. J. Hypertens. 2014, 2014, 381082. [Google Scholar] [CrossRef] [PubMed]
- Isakova, T.; Wahl, P.; Vargas, G.S.; Gutiérrez, O.M.; Scialla, J.; Xie, H.; Appleby, D.; Nessel, L.; Bellovich, K.; Chen, J.; et al. Fibroblast Growth Factor 23 is Elevated before Parathyroid Hormone and Phosphate in Chronic Kidney Disease. Kidney Int. 2011, 79, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Hayen, A.; Macaskill, P.; Pellegrini, F.; Craig, J.C.; Elder, G.J.; Strippoli, G.F.M. Serum Levels of Phosphorus, Parathyroid Hormone, and Calcium and Risks of Death and Cardiovascular Disease in Individuals with Chronic Kidney Disease: A Systematic Review and Meta-Analysis. JAMA 2011, 305, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Ritter, C.S.; Slatopolsky, E. Phosphate Toxicity in CKD: The Killer among Us. CJASN 2016, 11, 1088–1100. [Google Scholar] [CrossRef] [PubMed]
- Rodelo-Haad, C.; Santamaria, R.; Muñoz-Castañeda, J.R.; Pendón-Ruiz de Mier, M.V.; Martin-Malo, A.; Rodriguez, M. FGF23, Biomarker Or Target? Toxins 2019, 11, 175. [Google Scholar] [CrossRef]
- Shalhoub, V.; Shatzen, E.M.; Ward, S.C.; Davis, J.; Stevens, J.; Bi, V.; Renshaw, L.; Hawkins, N.; Wang, W.; Chen, C.; et al. FGF23 Neutralization Improves Chronic Kidney Disease–associated Hyperparathyroidism Yet Increases Mortality. J. Clin. Investig. 2012, 122, 2543–2553. [Google Scholar] [CrossRef]
- Clinkenbeard, E.L.; Noonan, M.L.; Thomas, J.C.; Ni, P.; Hum, J.M.; Aref, M.; Swallow, E.A.; Moe, S.M.; Allen, M.R.; White, K.E. Increased FGF23 Protects Against Detrimental Cardio-Renal Consequences during Elevated Blood Phosphate in CKD. Jci Insight 2019, 4, 123817. [Google Scholar] [CrossRef]
- Ben-Dov, I.Z.; Galitzer, H.; Lavi-Moshayoff, V.; Goetz, R.; Kuro-o, M.; Mohammadi, M.; Sirkis, R.; Naveh-Many, T.; Silver, J. The Parathyroid is a Target Organ for FGF23 in Rats. J. Clin. Investig. 2007, 117, 4003–4008. [Google Scholar] [CrossRef]
- Saji, F.; Shigematsu, T.; Sakaguchi, T.; Ohya, M.; Orita, H.; Maeda, Y.; Ooura, M.; Mima, T.; Negi, S. Fibroblast Growth Factor 23 Production in Bone is Directly Regulated by 1α, 25-Dihydroxyvitamin D., but Not PTH. Am. J. Physiol.-Ren. Physiol. 2010, 299, F1212–F1217. [Google Scholar] [CrossRef]
- Perwad, F.; Azam, N.; Zhang, M.Y.; Yamashita, T.; Tenenhouse, H.S.; Portale, A.A. Dietary and Serum Phosphorus Regulate Fibroblast Growth Factor 23 Expression and 1,25-Dihydroxyvitamin D Metabolism in Mice. Endocrinology 2005, 146, 5358–5364. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Yamamoto, L.; Karaplis, A.C.; St–Arnaud, R.; Goltzman, D. Fibroblast Growth Factor 23 Regulation by Systemic and Local Osteoblast-Synthesized 1,25-Dihydroxyvitamin D. J. Am. Soc. Nephrol. 2017, 28, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Chande, S.; Bergwitz, C. Role of Phosphate Sensing in Bone and Mineral Metabolism. Nat. Rev. Endocrinol. 2018, 1, 637–655. [Google Scholar] [CrossRef] [PubMed]
- Tagliabracci, V.S.; Engel, J.L.; Wiley, S.E.; Xiao, J.; Gonzalez, D.J.; Appaiah, H.N.; Koller, A.; Nizet, V.; White, K.E.; Dixon, J.E. Dynamic Regulation of FGF23 by Fam20C Phosphorylation, GalNAc-T3 Glycosylation, and Furin Proteolysis. Proc. Natl. Acad. Sci. USA 2014, 111, 5520–5525. [Google Scholar] [CrossRef] [PubMed]
- Urakawa, I.; Yamazaki, Y.; Shimada, T.; Iijima, K.; Hasegawa, H.; Okawa, K.; Fujita, T.; Fukumoto, S.; Yamashita, T. Klotho Converts Canonical FGF Receptor into a Specific Receptor for FGF23. Nature 2006, 444, 770. [Google Scholar] [CrossRef]
- Shimada, T.; Hasegawa, H.; Yamazaki, Y.; Muto, T.; Hino, R.; Takeuchi, Y.; Fujita, T.; Nakahara, K.; Fukumoto, S.; Yamashita, T. FGF-23 is a Potent Regulator of Vitamin D Metabolism and Phosphate Homeostasis. J. Bone Miner. Res. 2004, 19, 429–435. [Google Scholar] [CrossRef]
- Marks, J.; Srai, S.K.; Biber, J.; Murer, H.; Unwin, R.J.; Debnam, E.S. Intestinal Phosphate Absorption and the Effect of Vitamin D: A Comparison of Rats with Mice. Exp. Physiol. 2006, 91, 531–537. [Google Scholar] [CrossRef]
- Komaba, H.; Fukagawa, M. FGF23–parathyroid Interaction: Implications in Chronic Kidney Disease. Kidney Int. 2010, 77, 292–298. [Google Scholar] [CrossRef]
- Pereira, R.C.; Jűppner, H.; Azucena-Serrano, C.E.; Yadin, O.; Salusky, I.B.; Wesseling-Perry, K. Patterns of FGF-23, DMP1, and MEPE Expression in Patients with Chronic Kidney Disease. Bone 2009, 45, 1161–1168. [Google Scholar] [CrossRef]
- Santamaría, R.; Díaz-Tocados, J.M.; de Mier, M.V.P.R.; Robles, A.; Salmerón-Rodríguez, M.D.; Ruiz, E.; Vergara, N.; Aguilera-Tejero, E.; Raya, A.; Ortega, R.; et al. Increased Phosphaturia Accelerates the Decline in Renal Function: A Search for Mechanisms. Sci. Rep. 2018, 8, 13701. [Google Scholar] [CrossRef]
- Antoniucci, D.M.; Yamashita, T.; Portale, A.A. Dietary Phosphorus Regulates Serum Fibroblast Growth Factor-23 Concentrations in Healthy Men. J. Clin. Endocrinol. Metab. 2006, 91, 3144–3149. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.; Wu, H.; Peng, Y.; Hsu, S.; Chiu, Y.; Chen, H.; Yang, J.; Ko, M.; Pai, M.; Tu, Y. Effects of Lower Versus Higher Phosphate Diets on Fibroblast Growth Factor-23 Levels in Patients with Chronic Kidney Disease: A Systematic Review and Meta-Analysis. Nephrol. Dial. Transplant. 2018, 33, 1977–1983. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, O.; Isakova, T.; Rhee, E.; Shah, A.; Holmes, J.; Collerone, G.; Jüppner, H.; Wolf, M. Fibroblast Growth Factor-23 Mitigates Hyperphosphatemia but Accentuates Calcitriol Deficiency in Chronic Kidney Disease. J. Am. Soc. Nephrol. 2005, 16, 2205–2215. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, H.; Nagano, N.; Urakawa, I.; Yamazaki, Y.; Iijima, K.; Fujita, T.; Yamashita, T.; Fukumoto, S.; Shimada, T. Direct Evidence for a Causative Role of FGF23 in the Abnormal Renal Phosphate Handling and Vitamin D Metabolism in Rats with Early-Stage Chronic Kidney Disease. Kidney Int. 2010, 78, 975–980. [Google Scholar] [CrossRef]
- Komaba, H.; Fukagawa, M. The Role of FGF23 in CKD—With or without Klotho. Nat. Rev. Nephrol. 2012, 8, 484. [Google Scholar] [CrossRef]
- Hu, M.C.; Kuro-o, M.; Moe, O.W. Klotho and chronic kidney disease. Contrib. Nephrol. 2013, 180, 47–63. [Google Scholar] [CrossRef]
- Kuro-o, M. No Title. Klotho in chronic kidney disease—What’s new? Nephrol. Dial. Transplant. 2009, 24, 1705–1708. [Google Scholar] [CrossRef]
- Koh, N.; Fujimori, T.; Nishiguchi, S.; Tamori, A.; Shiomi, S.; Nakatani, T.; Sugimura, K.; Kishimoto, T.; Kinoshita, S.; Kuroki, T. Severely Reduced Production of Klotho in Human Chronic Renal Failure Kidney. Biochem. Biophys. Res. Commun. 2001, 280, 1015–1020. [Google Scholar] [CrossRef]
- Jean, G.; Terrat, J.; Vanel, T.; Hurot, J.; Lorriaux, C.; Mayor, B.; Chazot, C. High Levels of Serum Fibroblast Growth Factor (FGF)-23 are Associated with Increased Mortality in Long Haemodialysis Patients. Nephrol. Dial. Transplant. 2009, 24, 2792–2796. [Google Scholar] [CrossRef]
- Gutiérrez, O.M.; Mannstadt, M.; Isakova, T.; Rauh-Hain, J.A.; Tamez, H.; Shah, A.; Smith, K.; Lee, H.; Thadhani, R.; Jüppner, H. Fibroblast Growth Factor 23 and Mortality among Patients Undergoing Hemodialysis. N. Engl. J. Med. 2008, 359, 584–592. [Google Scholar] [CrossRef]
- Isakova, T.; Xie, H.; Yang, W.; Xie, D.; Anderson, A.H.; Scialla, J.; Wahl, P.; Gutiérrez, O.M.; Steigerwalt, S.; He, J. Fibroblast Growth Factor 23 and Risks of Mortality and End-Stage Renal Disease in Patients with Chronic Kidney Disease. JAMA 2011, 305, 2432–2439. [Google Scholar] [CrossRef] [PubMed]
- Lavi-Moshayoff, V.; Wasserman, G.; Meir, T.; Silver, J.; Naveh-Many, T. PTH Increases FGF23 Gene Expression and Mediates the High-FGF23 Levels of Experimental Kidney Failure: A Bone Parathyroid Feedback Loop. Am. J. Physiol.-Ren. Physiol. 2010, 299, F882–F889. [Google Scholar] [CrossRef] [PubMed]
- López, I.; Rodríguez-Ortiz, M.E.; Almadén, Y.; Guerrero, F.; De Oca, A.M.; Pineda, C.; Shalhoub, V.; Rodríguez, M.; Aguilera-Tejero, E. Direct and Indirect Effects of Parathyroid Hormone on Circulating Levels of Fibroblast Growth Factor 23 in Vivo. Kidney Int. 2011, 80, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Nishi, H.; Nii-Kono, T.; Nakanishi, S.; Yamazaki, Y.; Yamashita, T.; Fukumoto, S.; Ikeda, K.; Fujimori, A.; Fukagawa, M. Intravenous Calcitriol Therapy Increases Serum Concentrations of Fibroblast Growth Factor-23 in Dialysis Patients with Secondary Hyperparathyroidism. Nephron Clin. Pract. 2005, 101, c94–c99. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Rujirachun, P.; Holick, M.F.; Ungprasert, P. Oral Vitamin D 3 Supplementation Increases Serum Fibroblast Growth Factor 23 Concentration in Vitamin D-Deficient Patients: A Systematic Review and Meta-Analysis. Osteoporos. Int. 2019, 30, 2183–2193. [Google Scholar] [CrossRef] [PubMed]
- Komaba, H.; Goto, S.; Fujii, H.; Hamada, Y.; Kobayashi, A.; Shibuya, K.; Tominaga, Y.; Otsuki, N.; Nibu, K.; Nakagawa, K. Depressed Expression of Klotho and FGF Receptor 1 in Hyperplastic Parathyroid Glands from Uremic Patients. Kidney Int. 2010, 77, 232–238. [Google Scholar] [CrossRef]
- Canalejo, R.; Canalejo, A.; Martinez-Moreno, J.M.; Rodriguez-Ortiz, M.E.; Estepa, J.C.; Mendoza, F.J.; Munoz-Castaneda, J.R.; Shalhoub, V.; Almaden, Y.; Rodriguez, M. FGF23 Fails to Inhibit Uremic Parathyroid Glands. J. Am. Soc. Nephrol. 2010, 21, 1125–1135. [Google Scholar] [CrossRef]
- Galitzer, H.; Ben-Dov, I.Z.; Silver, J.; Naveh-Many, T. Parathyroid Cell Resistance to Fibroblast Growth Factor 23 in Secondary Hyperparathyroidism of Chronic Kidney Disease. Kidney Int. 2010, 77, 211–218. [Google Scholar] [CrossRef]
- Kendrick, J.; Cheung, A.K.; Kaufman, J.S.; Greene, T.; Roberts, W.L.; Smits, G.; Chonchol, M.; HOST Investigators. FGF-23 Associates with Death, Cardiovascular Events, and Initiation of Chronic Dialysis. J. Am. Soc. Nephrol. 2011, 22, 1913–1922. [Google Scholar] [CrossRef]
- Kestenbaum, B.; Sampson, J.N.; Rudser, K.D.; Patterson, D.J.; Seliger, S.L.; Young, B.; Sherrard, D.J.; Andress, D.L. Serum Phosphate Levels and Mortality Risk among People with Chronic Kidney Disease. J. Am. Soc. Nephrol. 2005, 16, 520–528. [Google Scholar] [CrossRef]
- Menon, V.; Greene, T.; Pereira, A.A.; Wang, X.; Beck, G.J.; Kusek, J.W.; Collins, A.J.; Levey, A.S.; Sarnak, M.J. Relationship of Phosphorus and Calcium-Phosphorus Product with Mortality in CKD. Am. J. Kidney Dis. 2005, 46, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Voormolen, N.; Noordzij, M.; Grootendorst, D.C.; Beetz, I.; Sijpkens, Y.W.; Van Manen, J.G.; Boeschoten, E.W.; Huisman, R.M.; Krediet, R.T.; Dekker, F.W. High Plasma Phosphate as a Risk Factor for Decline in Renal Function and Mortality in Pre-Dialysis Patients. Nephrol. Dial. Transplant. 2007, 22, 2909–2916. [Google Scholar] [CrossRef] [PubMed]
- Eddington, H.; Hoefield, R.; Sinha, S.; Chrysochou, C.; Lane, B.; Foley, R.N.; Hegarty, J.; New, J.; O’Donoghue, D.J.; Middleton, R.J. Serum Phosphate and Mortality in Patients with Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2010, 5, 2251–2257. [Google Scholar] [CrossRef]
- Rao, M.V.; Qiu, Y.; Wang, C.; Bakris, G. Hypertension and CKD: Kidney Early Evaluation Program (KEEP) and National Health and Nutrition Examination Survey (NHANES), 1999–2004. Am. J. Kidney Dis. 2008, 51, S30–S37. [Google Scholar] [CrossRef] [PubMed]
- Fyfe-Johnson, A.; Alonso, A.; Selvin, E.; Bower, J.; Pankow, J.; Agarwal, S.; Lutsey, P. Serum Fibroblast Growth Factor-23 and Incident Hypertension: The Atherosclerosis Risk in Communities Study. J. Hypertens. 2016, 34, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Akhabue, E.; Montag, S.; Reis, J.; Pool, L.; Mehta, R.; Yancy, C.; Zhao, L.; Wolf, M.; Gutierrez, O.; Carnethon, M.; et al. FGF23 (Fibroblast Growth Factor-23) and Incident Hypertension in Young and Middle-Aged Adults: The CARDIA Study. Hypertension 2018, 72, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Shi, L.; Liu, Y.; Zhang, H.; Liu, Y.; Huang, X.; Hou, D.; Zhang, M. Plasma Fibroblast Growth Factor 23 is Elevated in Pediatric Primary Hypertension. Front. Pediatrics 2019, 7, 135. [Google Scholar] [CrossRef]
- Mendes, M.; Resende, L.; Teixeira, A.; Correia, J.; Silva, G. Blood Pressure and Phosphate Level in Diabetic and Non-Diabetic Kidney Disease: Results of the Cross-Sectional “Low Clearance Consultation” Study. Porto Biomed. J. 2017, 2, 301–305. [Google Scholar] [CrossRef]
- Mohammad, J.; Scanni, R.; Bestmann, L.; Hulter, H.N.; Krapf, R. A Controlled Increase in Dietary Phosphate Elevates BP in Healthy Human Subjects. J. Am. Soc. Nephrol. Jasn 2018, 29, 2089–2098. [Google Scholar] [CrossRef]
- Li, Y.C.; Kong, J.; Wei, M.; Chen, Z.F.; Liu, S.Q.; Cao, L.P. 1,25-Dihydroxyvitamin D(3) is a Negative Endocrine Regulator of the Renin-Angiotensin System. J. Clin. Investig. 2002, 110, 229. [Google Scholar] [CrossRef]
- de Borst, M.H.; Vervloet, M.G.; ter Wee, P.M.; Navis, G. Cross Talk between the Renin-Angiotensin-Aldosterone System and Vitamin D-FGF-23-Klotho in Chronic Kidney Disease. J. Am. Soc. Nephrol. 2011, 22, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Crackower, M.A.; Sarao, R.; Oudit, G.Y.; Yagil, C.; Kozieradzki, I.; Scanga, S.E.; Oliveira-dos-Santos, A.J.; da Costa, J.; Zhang, L.; Pei, Y. Angiotensin-Converting Enzyme 2 is an Essential Regulator of Heart Function. Nature 2002, 417, 822. [Google Scholar] [CrossRef] [PubMed]
- Dai, B.; David, V.; Martin, A.; Huang, J.; Li, H.; Jiao, Y.; Gu, W.; Quarles, L.D. A Comparative Transcriptome Analysis Identifying FGF23 Regulated Genes in the Kidney of a Mouse CKD Model. PLoS ONE 2012, 7, e44161. [Google Scholar] [CrossRef] [PubMed]
- Andrukhova, O.; Slavic, S.; Smorodchenko, A.; Zeitz, U.; Shalhoub, V.; Lanske, B.; Pohl, E.E.; Erben, R.G. FGF23 Regulates Renal Sodium Handling and Blood Pressure. Embo Mol. Med. 2014, 6, 744–759. [Google Scholar] [CrossRef]
- Patel, R.K.; Jeemon, P.; Stevens, K.K.; Mccallum, L.; Hastie, C.E.; Schneider, A.; Jardine, A.G.; Mark, P.B.; Padmanabhan, S. Association between Serum Phosphate and Calcium, Long-Term Blood Pressure, and Mortality in Treated Hypertensive Adults. J. Hypertens. 2015, 33, 2046–2053. [Google Scholar] [CrossRef]
- Bozic, M.; Panizo, S.; Sevilla, M.; Riera, M.; Soler, M.; Pascual, J.; Lopez, I.; Freixenet, M.; Fernandez, E.; Valdivielso, J. High Phosphate Diet Increases Arterial Blood Pressure Via a Parathyroid Hormone Mediated Increase of Renin. J. Hypertens. 2014, 32, 1822–1832. [Google Scholar] [CrossRef]
- Suzuki, Y.; Mitsushima, S.; Kato, A.; Yamaguchi, T.; Ichihara, S. High-Phosphorus/Zinc-Free Diet Aggravates Hypertension and Cardiac Dysfunction in a Rat Model of the Metabolic Syndrome. Cardiovasc. Pathol. 2014, 23, 43–49. [Google Scholar] [CrossRef]
- Mizuno, M.; Mitchell, J.H.; Crawford, S.; Huang, C.; Maalouf, N.; Hu, M.; Moe, O.W.; Smith, S.A.; Vongpatanasin, W. High Dietary Phosphate Intake Induces Hypertension and Augments Exercise Pressor Reflex Function in Rats. American journal of physiology. Regul. Integr. Comp. Physiol. 2016, 311, R39–R48. [Google Scholar] [CrossRef]
- Delaney, E.P.; Greaney, J.L.; Edwards, D.G.; Rose, W.C.; Fadel, P.J.; Farquhar, W.B. Exaggerated Sympathetic and Pressor Responses to Handgrip Exercise in Older Hypertensive Humans: Role of the Muscle Metaboreflex. Am. J. Physiol.-Heart Circ. Physiol. 2010, 299, H1318–H1327. [Google Scholar] [CrossRef]
- Vongpatanasin, W.; Wang, Z.; Arbique, D.; Arbique, G.; Adams-Huet, B.; Mitchell, J.H.; Victor, R.G.; Thomas, G.D. Functional Sympatholysis is Impaired in Hypertensive Humans. J. Physiol. (Lond.) 2011, 589, 1209–1220. [Google Scholar] [CrossRef]
- Goodman, W.G.; Goldin, J.; Kuizon, B.D.; Yoon, C.; Gales, B.; Sider, D.; Wang, Y.; Chung, J.; Emerick, A.; Greaser, L. Coronary-Artery Calcification in Young Adults with End-Stage Renal Disease Who are Undergoing Dialysis. N. Engl. J. Med. 2000, 342, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Blacher, J.; Guerin, A.P.; Pannier, B.; Marchais, S.J.; London, G.M. Arterial Calcifications, Arterial Stiffness, and Cardiovascular Risk in End-Stage Renal Disease. Hypertension 2001, 38, 938–942. [Google Scholar] [CrossRef] [PubMed]
- Russo, D.; Corrao, S.; Battaglia, Y.; Andreucci, M.; Caiazza, A.; Carlomagno, A.; Lamberti, M.; Pezone, N.; Pota, A.; Russo, L. Progression of Coronary Artery Calcification and Cardiac Events in Patients with Chronic Renal Disease Not Receiving Dialysis. Kidney Int. 2011, 80, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Nasrallah, M.M.; El-Shehaby, A.R.; Salem, M.M.; Osman, N.A.; El Sheikh, E.; Sharaf El Din, U.A. Fibroblast Growth Factor-23 (FGF-23) is Independently Correlated to Aortic Calcification in Haemodialysis Patients. Nephrol. Dial. Transplant. 2010, 25, 2679–2685. [Google Scholar] [CrossRef]
- Desjardins, L.; Liabeuf, S.; Renard, C.; Lenglet, A.; Lemke, H.D.; Choukroun, G.; Drueke, T.B.; Massy, Z.A.; European Uremic Toxin (EUTox) Work Group. FGF23 is Independently Associated with Vascular Calcification but Not Bone Mineral Density in Patients at various CKD Stages. Osteoporos Int. 2012, 23, 2017–2025. [Google Scholar]
- Scialla, J.J.; Lau, W.L.; Reilly, M.P.; Isakova, T.; Yang, H.; Crouthamel, M.H.; Chavkin, N.W.; Rahman, M.; Wahl, P.; Amaral, A.P. Fibroblast Growth Factor 23 is Not Associated with and does Not Induce Arterial Calcification. Kidney Int. 2013, 83, 1159–1168. [Google Scholar] [CrossRef]
- Lindberg, K.; Olauson, H.; Amin, R.; Ponnusamy, A.; Goetz, R.; Taylor, R.F.; Mohammadi, M.; Canfield, A.; Kublickiene, K.; Larsson, T.E. Arterial Klotho Expression and FGF23 Effects on Vascular Calcification and Function. PLoS ONE 2013, 8, e60658. [Google Scholar] [CrossRef]
- Lim, K.; Lu, T.; Molostvov, G.; Lee, C.; Lam, F.T.; Zehnder, D.; Hsiao, L. Vascular Klotho Deficiency Potentiates the Development of Human Artery Calcification and Mediates Resistance to Fibroblast Growth Factor 23. Circulation 2012, 125, 2243–2255. [Google Scholar] [CrossRef]
- Zhu, D.; Mackenzie, N.C.; Millan, J.L.; Farquharson, C.; MacRae, V.E. A Protective Role for FGF-23 in Local Defence against Disrupted Arterial Wall Integrity? Mol. Cell. Endocrinol. 2013, 372, 1–11. [Google Scholar] [CrossRef]
- Jimbo, R.; Kawakami-Mori, F.; Mu, S.; Hirohama, D.; Majtan, B.; Shimizu, Y.; Yatomi, Y.; Fukumoto, S.; Fujita, T.; Shimosawa, T. Fibroblast Growth Factor 23 Accelerates Phosphate-Induced Vascular Calcification in the Absence of Klotho Deficiency. Kidney Int. 2014, 85, 1103–1111. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, C.; Duan, Z.; Xu, C.; Chen, Y. Klotho/FGF23 Axis Mediates High Phosphate-induced Vascular Calcification in Vascular Smooth Muscle Cells Via Wnt7b/Β-catenin Pathway. Kaohsiung J. Med. Sci. 2019, 35, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Shigematsu, T.; Kono, T.; Satoh, K.; Yokoyama, K.; Yoshida, T.; Hosoya, T.; Shirai, K. Phosphate Overload Accelerates Vascular Calcium Deposition in End-stage Renal Disease Patients. Nephrol. Dial. Transplant. 2003, 18, iii86–iii89. [Google Scholar] [CrossRef] [PubMed]
- Adeney, K.L.; Siscovick, D.S.; Ix, J.H.; Seliger, S.L.; Shlipak, M.G.; Jenny, N.S.; Kestenbaum, B.R. Association of Serum Phosphate with Vascular and Valvular Calcification in Moderate CKD. J. Am. Soc. Nephrol. 2009, 20, 381–387. [Google Scholar] [CrossRef] [PubMed]
- El-Abbadi, M.M.; Pai, A.S.; Leaf, E.M.; Yang, H.; Bartley, B.A.; Quan, K.K.; Ingalls, C.M.; Liao, H.W.; Giachelli, C.M. Phosphate Feeding Induces Arterial Medial Calcification in Uremic Mice: Role of Serum Phosphorus, Fibroblast Growth Factor-23, and Osteopontin. Kidney Int. 2009, 75, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Lau, W.L.; Linnes, M.; Chu, E.Y.; Foster, B.L.; Bartley, B.A.; Somerman, M.J.; Giachelli, C.M. High Phosphate Feeding Promotes Mineral and Bone Abnormalities in Mice with Chronic Kidney Disease. Nephrol. Dial. Transplant. 2012, 28, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Shroff, R.C.; McNair, R.; Skepper, J.N.; Figg, N.; Schurgers, L.J.; Deanfield, J.; Rees, L.; Shanahan, C.M. Chronic Mineral Dysregulation Promotes Vascular Smooth Muscle Cell Adaptation and Extracellular Matrix Calcification. J. Am. Soc. Nephrol. 2010, 21, 103–112. [Google Scholar] [CrossRef]
- Collins, J.F.; Bai, L.; Ghishan, F.K. The SLC20 Family of Proteins: Dual Functions as Sodium-Phosphate Cotransporters and Viral Receptors. Pflügers Arch. 2004, 447, 647–652. [Google Scholar] [CrossRef]
- Li, X.; Yang, H.; Giachelli, C.M. Role of the Sodium-Dependent Phosphate Cotransporter, Pit-1, in Vascular Smooth Muscle Cell Calcification. Circ. Res. 2006, 98, 905–912. [Google Scholar] [CrossRef]
- Chavkin, N.W.; Chia, J.J.; Crouthamel, M.H.; Giachelli, C.M. Phosphate Uptake-Independent Signaling Functions of the Type III Sodium-Dependent Phosphate Transporter, PiT-1, in Vascular Smooth Muscle Cells. Exp. Cell Res. 2015, 333, 39–48. [Google Scholar] [CrossRef]
- Yamada, S.; Leaf, E.M.; Chia, J.J.; Cox, T.C.; Speer, M.Y.; Giachelli, C.M. PiT-2, a Type III Sodium-Dependent Phosphate Transporter, Protects Against Vascular Calcification in Mice with Chronic Kidney Disease Fed a High-Phosphate Diet. Kidney Int. 2018, 94, 716–727. [Google Scholar] [CrossRef]
- Reynolds, J.L.; Joannides, A.J.; Skepper, J.N.; McNair, R.; Schurgers, L.J.; Proudfoot, D.; Jahnen-Dechent, W.; Weissberg, P.L.; Shanahan, C.M. Human Vascular Smooth Muscle Cells Undergo Vesicle-Mediated Calcification in Response to Changes in Extracellular Calcium and Phosphate Concentrations: A Potential Mechanism for Accelerated Vascular Calcification in ESRD. J. Am. Soc. Nephrol. 2004, 15, 2857–2867. [Google Scholar] [CrossRef] [PubMed]
- Speer, M.Y.; Li, X.; Hiremath, P.G.; Giachelli, C.M. Runx2/Cbfa1, but Not Loss of Myocardin, is Required for Smooth Muscle Cell Lineage Reprogramming Toward Osteochondrogenesis. J. Cell. Biochem. 2010, 110, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.X.; O’Neill, K.D.; Duan, D.; Moe, S.M. Phosphorus and Uremic Serum Up-Regulate Osteopontin Expression in Vascular Smooth Muscle Cells. Kidney Int. 2002, 62, 1724–1731. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, H.; Giachelli, C.M. BMP-2 Promotes Phosphate Uptake, Phenotypic Modulation, and Calcification of Human Vascular Smooth Muscle Cells. Atherosclerosis 2008, 199, 271–277. [Google Scholar] [CrossRef]
- Steitz, S.A.; Speer, M.Y.; Curinga, G.; Yang, H.; Haynes, P.; Aebersold, R.; Schinke, T.; Karsenty, G.; Giachelli, C.M. Smooth Muscle Cell Phenotypic Transition Associated with Calcification: Upregulation of Cbfa1 and Downregulation of Smooth Muscle Lineage Markers. Circ. Res. 2001, 89, 1147–1154. [Google Scholar] [CrossRef]
- Shanahan, C.M.; Crouthamel, M.H.; Kapustin, A.; Giachelli, C.M. Arterial Calcification in Chronic Kidney Disease: Key Roles for Calcium and Phosphate. Circ. Res. 2011, 109, 697–711. [Google Scholar] [CrossRef]
- Boström, K.I.; Rajamannan, N.M.; Towler, D.A. The Regulation of Valvular and Vascular Sclerosis by Osteogenic Morphogens. Circ. Res. 2011, 109, 564–577. [Google Scholar] [CrossRef]
- Cheng, L.; Zhang, L.; Yang, J.; Hao, L. Activation of Peroxisome Proliferator-Activated Receptor Γ Inhibits Vascular Calcification by Upregulating Klotho. Exp. Ther. Med. 2017, 13, 467–474. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y.; Liu, L.; Zhang, J.; Zhang, Y.; Zhang, B.; Zhang, Z.; Bi, X.; Nie, L.; Xiong, J.; et al. High Phosphate-Induced Downregulation of PPARγ Contributes to CKD-Associated Vascular Calcification. J. Mol. Cell. Cardiol. 2018, 114, 264–275. [Google Scholar] [CrossRef]
- Mihai, S.; Codrici, E.; Popescu, I.D.; Enciu, A.; Albulescu, L.; Necula, L.G.; Mambet, C.; Anton, G.; Tanase, C. Inflammation-Related Mechanisms in Chronic Kidney Disease Prediction, Progression, and Outcome. J. Immunol. Res. 2018, 2018, 2180373. [Google Scholar] [CrossRef]
- Hénaut, L.; Massy, Z.A. New Insights into the Key Role of Interleukin 6 in Vascular Calcification of Chronic Kidney Disease. Nephrol. Dial. Transplant. 2018, 33, 543–548. [Google Scholar] [CrossRef]
- Mendoza, J.M.; Isakova, T.; Ricardo, A.C.; Xie, H.; Navaneethan, S.D.; Anderson, A.H.; Bazzano, L.A.; Xie, D.; Kretzler, M.; Nessel, L. Fibroblast Growth Factor 23 and Inflammation in CKD. Clin. J. Am. Soc. Nephrol. 2012, 7, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Hanks, L.J.; Casazza, K.; Judd, S.E.; Jenny, N.S.; Gutiérrez, O.M. Associations of Fibroblast Growth Factor-23 with Markers of Inflammation, Insulin Resistance and Obesity in Adults. PLoS ONE 2015, 10, e0122885. [Google Scholar] [CrossRef]
- Wallquist, C.; Mansouri, L.; Norrbäck, M.; Hylander, B.; Jacobson, S.H.; Larsson, T.E.; Lundahl, J. Associations of Fibroblast Growth Factor 23 with Markers of Inflammation and Leukocyte Transmigration in Chronic Kidney Disease. Nephron 2018, 138, 287–295. [Google Scholar] [CrossRef]
- David, V.; Martin, A.; Isakova, T.; Spaulding, C.; Qi, L.; Ramirez, V.; Zumbrennen-Bullough, K.B.; Sun, C.C.; Lin, H.Y.; Babitt, J.L. Inflammation and Functional Iron Deficiency Regulate Fibroblast Growth Factor 23 Production. Kidney Int. 2016, 89, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Durlacher-Betzer, K.; Hassan, A.; Levi, R.; Axelrod, J.; Silver, J.; Naveh-Many, T. Interleukin-6 Contributes to the Increase in Fibroblast Growth Factor 23 Expression in Acute and Chronic Kidney Disease. Kidney Int. 2018, 94, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Egli-Spichtig, D.; Silva, P.H.I.; Glaudemans, B.; Gehring, N.; Bettoni, C.; Zhang, M.; Arroyo, E.P.; Schönenberger, D.; Rajski, M.; Hoogewijis, D. Tumor Necrosis Factor Stimulates Fibroblast Growth Factor 23 Levels in Chronic Kidney Disease and Non-Renal Inflammation. Kidney Int. 2019, 96, 890–905. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Li, L.; Yang, J.; King, G.; Xiao, Z.; Quarles, L.D. Counter-regulatory Paracrine Actions of FGF-23 and 1,25 (OH) 2D in Macrophages. Febs Lett. 2016, 590, 53–67. [Google Scholar] [CrossRef]
- Singh, S.; Grabner, A.; Yanucil, C.; Schramm, K.; Czaya, B.; Krick, S.; Czaja, M.J.; Bartz, R.; Abraham, R.; Di Marco, G.S. Fibroblast Growth Factor 23 Directly Targets Hepatocytes to Promote Inflammation in Chronic Kidney Disease. Kidney Int. 2016, 90, 985–996. [Google Scholar] [CrossRef]
- Aghagolzadeh, P.; Bachtler, M.; Bijarnia, R.; Jackson, C.; Smith, E.R.; Odermatt, A.; Radpour, R.; Pasch, A. Calcification of Vascular Smooth Muscle Cells is Induced by Secondary Calciprotein Particles and Enhanced by Tumor Necrosis Factor-A. Atherosclerosis 2016, 251, 404–414. [Google Scholar] [CrossRef]
- Pazár, B.; Ea, H.; Narayan, S.; Kolly, L.; Bagnoud, N.; Chobaz, V.; Roger, T.; Lioté, F.; So, A.; Busso, N. Basic Calcium Phosphate Crystals Induce Monocyte/Macrophage IL-1β Secretion through the NLRP3 Inflammasome in Vitro. J. Immunol. 2011, 186, 2495–2502. [Google Scholar] [CrossRef] [PubMed]
- Tintut, Y.; Patel, J.; Parhami, F.; Demer, L.L. Tumor Necrosis Factor-A Promotes in Vitro Calcification of Vascular Cells Via the cAMP Pathway. Circulation 2000, 102, 2636–2642. [Google Scholar] [CrossRef] [PubMed]
- Awan, Z.; Denis, M.; Roubtsova, A.; Essalmani, R.; Marcinkiewicz, J.; Awan, A.; Gram, H.; Seidah, N.G.; Genest, J. Reducing Vascular Calcification by Anti-IL-1β Monoclonal Antibody in a Mouse Model of Familial Hypercholesterolemia. Angiology 2016, 67, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Ceneri, N.; Zhao, L.; Young, B.D.; Healy, A.; Coskun, S.; Vasavada, H.; Yarovinsky, T.O.; Ike, K.; Pardi, R.; Qin, L. Rac2 Modulates Atherosclerotic Calcification by Regulating Macrophage Interleukin-1β Production. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Panguluri, S.K.; Gupta, S.K.; Dahiya, S.; Lundy, R.F.; Kumar, A. Tumor Necrosis Factor-A Regulates Distinct Molecular Pathways and Gene Networks in Cultured Skeletal Muscle Cells. PLoS ONE 2010, 5, e13262. [Google Scholar] [CrossRef]
- Azpiazu, D.; Gonzalo, S.; Villa-Bellosta, R. Tissue Non-Specific Alkaline Phosphatase and Vascular Calcification: A Potential Therapeutic Target. Curr. Cardiol. Rev. 2019, 15, 91–95. [Google Scholar] [CrossRef]
- Zickler, D.; Luecht, C.; Willy, K.; Chen, L.; Witowski, J.; Girndt, M.; Fiedler, R.; Storr, M.; Kamhieh-Milz, J.; Schoon, J. Tumour Necrosis Factor-Alpha in Uraemic Serum Promotes Osteoblastic Transition and Calcification of Vascular Smooth Muscle Cells Via Extracellular Signal-Regulated Kinases and Activator Protein 1/C-FOS-Mediated Induction of Interleukin 6 Expression. Nephrol. Dial. Transplant. 2017, 33, 574–585. [Google Scholar] [CrossRef]
- Agharazii, M.; St-Louis, R.; Gautier-Bastien, A.; Ung, R.; Mokas, S.; Larivière, R.; Richard, D.E. Inflammatory Cytokines and Reactive Oxygen Species as Mediators of Chronic Kidney Disease-Related Vascular Calcification. Am. J. Hypertens. 2014, 28, 746–755. [Google Scholar] [CrossRef]
- Reynolds, J.L.; Skepper, J.N.; McNair, R.; Kasama, T.; Gupta, K.; Weissberg, P.L.; Jahnen-Dechent, W.; Shanahan, C.M. Multifunctional Roles for Serum Protein Fetuin-a in Inhibition of Human Vascular Smooth Muscle Cell Calcification. J. Am. Soc. Nephrol. 2005, 16, 2920–2930. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Thompson, C.R.; Ethier, J.; Carlisle, E.J.; Tobe, S.; Mendelssohn, D.; Burgess, E.; Jindal, K.; Barrett, B.; Singer, J. Left Ventricular Mass Index Increase in Early Renal Disease: Impact of Decline in Hemoglobin. Am. J. Kidney Dis. 1999, 34, 125–134. [Google Scholar] [CrossRef]
- Foley, R.N.; Parfrey, P.S.; Harnett, J.D.; Kent, G.M.; Martin, C.J.; Murray, D.C.; Barre, P.E. Clinical and Echocardiographic Disease in Patients Starting End-Stage Renal Disease Therapy. Kidney Int. 1995, 47, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, E.; Bellino, D.; Cassottana, P.; Rolla, D.; Cannella, G. Left Ventricular Hypertrophy in Nondiabetic Predialysis CKD. Am. J. Kidney Dis. 2005, 46, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.J.; Wu, M. Fibroblast Growth Factor 23: A Possible Cause of Left Ventricular Hypertrophy in Hemodialysis Patients. Am. J. Med. Sci. 2009, 337, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.A.; Larsson, A.; Melhus, H.; Lind, L.; Larsson, T.E. Serum Intact FGF23 Associate with Left Ventricular Mass, Hypertrophy and Geometry in an Elderly Population. Atherosclerosis 2009, 207, 546–551. [Google Scholar] [CrossRef]
- Kirkpantur, A.; Balci, M.; Gurbuz, O.A.; Afsar, B.; Canbakan, B.; Akdemir, R.; Ayli, M.D. Serum Fibroblast Growth Factor-23 (FGF-23) Levels are Independently Associated with Left Ventricular Mass and Myocardial Performance Index in Maintenance Haemodialysis Patients. Nephrol. Dial. Transplant. 2011, 26, 1346–1354. [Google Scholar] [CrossRef]
- Negishi, K.; Kobayashi, M.; Ochiai, I.; Yamazaki, Y.; Hasegawa, H.; Yamashita, T.; Shimizu, T.; Kasama, S.; Kurabayashi, M. Association between Fibroblast Growth Factor 23 and Left Ventricular Hypertrophy in Maintenance Hemodialysis Patients. Circ. J. 2010, 74, 2734–2740. [Google Scholar] [CrossRef]
- Seeherunvong, W.; Abitbol, C.; Chandar, J.; Rusconi, P.; Zilleruelo, G.; Freundlich, M. Fibroblast Growth Factor 23 and Left Ventricular Hypertrophy in Children on Dialysis. Pediatr. Nephrol. 2012, 27, 2129–2136. [Google Scholar] [CrossRef]
- Mitsnefes, M.M.; Betoko, A.; Schneider, M.F.; Salusky, I.B.; Wolf, M.S.; Jüppner, H.; Warady, B.A.; Furth, S.L.; Portale, A.A. FGF23 and Left Ventricular Hypertrophy in Children with CKD. Clin. J. Am. Soc. Nephrol. Cjasn 2018, 13, 45–52. [Google Scholar] [CrossRef]
- Leifheit-Nestler, M.; große Siemer, R.; Flasbart, K.; Richter, B.; Kirchhoff, F.; Ziegler, W.H.; Klintschar, M.; Becker, J.U.; Erbersdobler, A.; Aufricht, C. Induction of Cardiac FGF23/FGFR4 Expression is Associated with Left Ventricular Hypertrophy in Patients with Chronic Kidney Disease. Nephrol. Dial. Transplant. 2015, 31, 1088–1099. [Google Scholar] [CrossRef]
- Faul, C.; Amaral, A.P.; Oskouei, B.; Hu, M.; Sloan, A.; Isakova, T.; Gutiérrez, O.M.; Aguillon-Prada, R.; Lincoln, J.; Hare, J.M.; et al. FGF23 Induces Left Ventricular Hypertrophy. J. Clin. Investig. 2011, 121, 4393–4408. [Google Scholar] [CrossRef]
- Grabner, A.; Amaral, A.P.; Schramm, K.; Singh, S.; Sloan, A.; Yanucil, C.; Li, J.; Shehadeh, L.A.; Hare, J.M.; David, V. Activation of Cardiac Fibroblast Growth Factor Receptor 4 Causes Left Ventricular Hypertrophy. Cell Metab. 2015, 22, 1020–1032. [Google Scholar] [CrossRef]
- Di Marco, G.S.; Reuter, S.; Kentrup, D.; Grabner, A.; Amaral, A.P.; Fobker, M.; Stypmann, J.; Pavenstädt, H.; Wolf, M.; Faul, C. Treatment of Established Left Ventricular Hypertrophy with Fibroblast Growth Factor Receptor Blockade in an Animal Model of CKD. Nephrol. Dial. Transplant. 2014, 29, 2028–2035. [Google Scholar] [CrossRef]
- Molkentin, J.D. Calcineurin–NFAT Signaling Regulates the Cardiac Hypertrophic Response in Coordination with the MAPKs. Cardiovasc. Res. 2004, 63, 467–475. [Google Scholar] [CrossRef]
- Takashi, Y.; Kinoshita, Y.; Hori, M.; Ito, N.; Taguchi, M.; Fukumoto, S. Patients with FGF23-Related Hypophosphatemic Rickets/Osteomalacia do Not Present with Left Ventricular Hypertrophy. Endocr. Res. 2017, 42, 132–137. [Google Scholar] [CrossRef]
- Leifheit-Nestler, M.; Richter, B.; Basaran, M.; Nespor, J.; Vogt, I.; Alesutan, I.; Voelkl, J.; Lang, F.; Heineke, J.; Krick, S. Impact of Altered Mineral Metabolism on Pathological Cardiac Remodeling in Elevated Fibroblast Growth Factor 23. Front. Endocrinol. 2018, 9, 333. [Google Scholar] [CrossRef]
- Pastor-Arroyo, E.; Gehring, N.; Krudewig, C.; Costantino, S.; Bettoni, C.; Knöpfel, T.; Sabrautzki, S.; Lorenz-Depiereux, B.; Pastor, J.; Strom, T.M. The Elevation of Circulating Fibroblast Growth Factor 23 without Kidney Disease does Not Increase Cardiovascular Disease Risk. Kidney Int. 2018, 94, 49–59. [Google Scholar] [CrossRef]
- Faul, C. FGF23 Effects on the Heart—Levels, Time, Source, And context Matter. Kidney Int. 2018, 94, 7–11. [Google Scholar] [CrossRef]
- Foley, R.N.; Collins, A.J.; Herzog, C.A.; Ishani, A.; Kalra, P.A. Serum Phosphate and Left Ventricular Hypertrophy in Young Adults: The Coronary Artery Risk Development in Young Adults Study. Kidney Blood Press. Res. 2009, 32, 37–44. [Google Scholar] [CrossRef]
- Saab, G.; Whooley, M.A.; Schiller, N.B.; Ix, J.H. Association of Serum Phosphorus with Left Ventricular Mass in Men and Women with Stable Cardiovascular Disease: Data from the Heart and Soul Study. Am. J. Kidney Dis. 2010, 56, 496–505. [Google Scholar] [CrossRef]
- Chue, C.D.; Edwards, N.C.; Moody, W.E.; Steeds, R.P.; Townend, J.N.; Ferro, C.J. Serum Phosphate is Associated with Left Ventricular Mass in Patients with Chronic Kidney Disease: A Cardiac Magnetic Resonance Study. Heart 2012, 98, 219–224. [Google Scholar] [CrossRef]
- Yamamoto, K.T.; Robinson-Cohen, C.; De Oliveira, M.C.; Kostina, A.; Nettleton, J.A.; Ix, J.H.; Nguyen, H.; Eng, J.; Lima, J.A.; Siscovick, D.S. Dietary Phosphorus is Associated with Greater Left Ventricular Mass. Kidney Int. 2013, 83, 707–714. [Google Scholar] [CrossRef]
- Zou, J.; Yu, Y.; Wu, P.; Lin, F.; Yao, Y.; Xie, Y.; Jiang, G. Serum Phosphorus is Related to Left Ventricular Remodeling Independent of Renal Function in Hospitalized Patients with Chronic Kidney Disease. Int. J. Cardiol. 2016, 221, 134–140. [Google Scholar] [CrossRef]
- Amann, K.; Törnig, J.; Kugel, B.; Gross, M.; Tyralla, K.; El-Shakmak, A.; Szabo, A.; Ritz, E. Hyperphosphatemia Aggravates Cardiac Fibrosis and Microvascular Disease in Experimental Uremia. Kidney Int. 2003, 63, 1296–1301. [Google Scholar] [CrossRef]
- Neves, K.R.; Graciolli, F.G.; Dos Reis, L.M.; Pasqualucci, C.A.; Moyses, R.M.; Jorgetti, V. Adverse Effects of Hyperphosphatemia on Myocardial Hypertrophy, Renal Function, and Bone in Rats with Renal Failure. Kidney Int. 2004, 66, 2237–2244. [Google Scholar] [CrossRef]
- Peri-Okonny, P.; Baskin, K.K.; Iwamoto, G.; Mitchell, J.H.; Smith, S.A.; Kim, H.K.; Szweda, L.I.; Bassel-Duby, R.; Fujikawa, T.; Castorena, C.M. High-Phosphate Diet Induces Exercise Intolerance and Impairs Fatty Acid Metabolism in Mice. Circulation 2019, 139, 1422–1434. [Google Scholar] [CrossRef]
- Hu, M.C.; Shi, M.; Cho, H.J.; Adams-Huet, B.; Paek, J.; Hill, K.; Shelton, J.; Amaral, A.P.; Faul, C.; Taniguchi, M. Klotho and Phosphate are Modulators of Pathologic Uremic Cardiac Remodeling. J. Am. Soc. Nephrol. 2015, 26, 1290–1302. [Google Scholar] [CrossRef]
- Hu, M.C.; Scanni, R.; Ye, J.; Zhang, J.; Shi, M.; Maique, J.; Flores, B.; Moe, O.W.; Krapf, R. Dietary Vitamin D Interacts with High Phosphate–induced Cardiac Remodeling in Rats with Normal Renal Function. Nephrol. Dial. Transplant. 2019, gfz156. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, C.; Chang, C.; Chou, C.; Lin, S.; Wang, I.; Hsieh, D.J.; Jong, G.; Huang, C.; Wang, C. Hyperphosphate-Induced Myocardial Hypertrophy through the GATA-4/NFAT-3 Signaling Pathway is Attenuated by ERK Inhibitor Treatment. Cardiorenal Med. 2015, 5, 79–88. [Google Scholar] [CrossRef]
- Ferrari, S.L.; Bonjour, J.; Rizzoli, R. Fibroblast Growth Factor-23 Relationship to Dietary Phosphate and Renal Phosphate Handling in Healthy Young Men. J. Clin. Endocrinol. Metab. 2005, 90, 1519–1524. [Google Scholar] [CrossRef]
- Burnett, S.M.; Gunawardene, S.C.; Bringhurst, F.R.; Jüppner, H.; Lee, H.; Finkelstein, J.S. Regulation of C-terminal and Intact FGF-23 by Dietary Phosphate in Men and Women. J. Bone Miner. Res. 2006, 21, 1187–1196. [Google Scholar] [CrossRef]
- Tsai, W.; Wu, H.; Peng, Y.; Hsu, S.; Chiu, Y.; Yang, J.; Chen, H.; Pai, M.; Lin, W.; Hung, K. Short-Term Effects of very-Low-Phosphate and Low-Phosphate Diets on Fibroblast Growth Factor 23 in Hemodialysis Patients: A Randomized Crossover Trial. Clin. J. Am. Soc. Nephrol. 2019, 14, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Gutekunst, L.; Mehrotra, R.; Kovesdy, C.P.; Bross, R.; Shinaberger, C.S.; Noori, N.; Hirschberg, R.; Benner, D.; Nissenson, A.R. Understanding Sources of Dietary Phosphorus in the Treatment of Patients with Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2010, 5, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Moe, S.M.; Chen, N.X.; Seifert, M.F.; Sinders, R.M.; Duan, D.; Chen, X.; Liang, Y.; Radcliff, J.S.; White, K.E.; Gattone II, V.H. A Rat Model of Chronic Kidney Disease-Mineral Bone Disorder. Kidney Int. 2009, 75, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Moe, S.M.; Zidehsarai, M.P.; Chambers, M.A.; Jackman, L.A.; Radcliffe, J.S.; Trevino, L.L.; Donahue, S.E.; Asplin, J.R. Vegetarian Compared with Meat Dietary Protein Source and Phosphorus Homeostasis in Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2011, 6, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Scialla, J.J.; Appel, L.J.; Wolf, M.; Yang, W.; Zhang, X.; Sozio, S.M.; Miller, E.R., III; Bazzano, L.A.; Cuevas, M.; Glenn, M.J. Plant Protein Intake is Associated with Fibroblast Growth Factor 23 and Serum Bicarbonate Levels in Patients with Chronic Kidney Disease: The Chronic Renal Insufficiency Cohort Study. J. Ren. Nutr. 2012, 22, 37–388. [Google Scholar] [CrossRef]
- Sullivan, C.; Sayre, S.S.; Leon, J.B.; Machekano, R.; Love, T.E.; Porter, D.; Marbury, M.; Sehgal, A.R. Effect of Food Additives on Hyperphosphatemia among Patients with End-Stage Renal Disease: A Randomized Controlled Trial. JAMA 2009, 301, 629–635. [Google Scholar] [CrossRef]
- de Fornasari, M.L.L.; dos Santos Sens, Y.A. Replacing Phosphorus-Containing Food Additives with Foods without Additives Reduces Phosphatemia in End-Stage Renal Disease Patients: A Randomized Clinical Trial. J. Ren. Nutr. 2017, 27, 97–105. [Google Scholar] [CrossRef]
- Shinaberger, C.S.; Greenland, S.; Kopple, J.D.; Van Wyck, D.; Mehrotra, R.; Kovesdy, C.P.; Kalantar-Zadeh, K. Is Controlling Phosphorus by Decreasing Dietary Protein Intake Beneficial Or Harmful in Persons with Chronic Kidney Disease? Am. J. Clin. Nutr. 2008, 88, 1511–1518. [Google Scholar] [CrossRef]
- Di Iorio, B.; Di Micco, L.; Torraca, S.; Sirico, M.L.; Russo, L.; Pota, A.; Mirenghi, F.; Russo, D. Acute Effects of very-Low-Protein Diet on FGF23 Levels: A Randomized Study. Clin. J. Am. Soc. Nephrol. 2012, 7, 581–587. [Google Scholar] [CrossRef]
- Oliveira, R.B.; Cancela, A.L.; Graciolli, F.G.; Dos Reis, L.M.; Draibe, S.A.; Cuppari, L.; Carvalho, A.B.; Jorgetti, V.; Canziani, M.E.; Moysés, R.M. Early Control of PTH and FGF23 in Normophosphatemic CKD Patients: A New Target in CKD-MBD Therapy? Clin. J. Am. Soc. Nephrol. 2010, 5, 286–291. [Google Scholar] [CrossRef]
- Block, G.A.; Wheeler, D.C.; Persky, M.S.; Kestenbaum, B.; Ketteler, M.; Spiegel, D.M.; Allison, M.A.; Asplin, J.; Smits, G.; Hoofnagle, A.N. Effects of Phosphate Binders in Moderate CKD. J. Am. Soc. Nephrol. 2012, 23, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Patel, L.; Bernard, L.M.; Elder, G.J. Sevelamer Versus Calcium-Based Binders for Treatment of Hyperphosphatemia in CKD: A Meta-Analysis of Randomized Controlled Trials. Clin. J. Am. Soc. Nephrol. 2016, 11, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Hirakata, H.; Akiba, T.; Fukagawa, M.; Nakayama, M.; Sawada, K.; Kumagai, Y.; Block, G.A. Ferric Citrate Hydrate for the Treatment of Hyperphosphatemia in Nondialysis-Dependent CKD. Clin. J. Am. Soc. Nephrol. 2014, 9, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Parra, E.; Gonzalez-Casaus, M.L.; Galán, A.; Martinez-Calero, A.; Navas, V.; Rodriguez, M.; Ortiz, A. Lanthanum Carbonate Reduces FGF23 in Chronic Kidney Disease Stage 3 Patients. Nephrol. Dial. Transplant. 2011, 26, 2567–2571. [Google Scholar] [CrossRef] [PubMed]
- Isakova, T.; Barchi-Chung, A.; Enfield, G.; Smith, K.; Vargas, G.; Houston, J.; Xie, H.; Wahl, P.; Schiavenato, E.; Dosch, A. Effects of Dietary Phosphate Restriction and Phosphate Binders on FGF23 Levels in CKD. Clin. J. Am. Soc. Nephrol. 2013, 8, 1009–1018. [Google Scholar] [CrossRef] [PubMed]
- Jamal, S.A.; Vandermeer, B.; Raggi, P.; Mendelssohn, D.C.; Chatterley, T.; Dorgan, M.; Lok, C.E.; Fitchett, D.; Tsuyuki, R.T. Effect of Calcium-Based Versus Non-Calcium-Based Phosphate Binders on Mortality in Patients with Chronic Kidney Disease: An Updated Systematic Review and Meta-Analysis. Lancet 2013, 382, 1268–1277. [Google Scholar] [CrossRef]
- Block, G.A.; Pergola, P.E.; Fishbane, S.; Martins, J.G.; LeWinter, R.D.; Uhlig, K.; Neylan, J.F.; Chertow, G.M. Effect of Ferric Citrate on Serum Phosphate and Fibroblast Growth Factor 23 among Patients with Nondialysis-Dependent Chronic Kidney Disease: Path Analyses. Nephrol. Dial. Transplant. 2018, 34, 1115–1124. [Google Scholar] [CrossRef]
- Block, G.A.; Block, M.S.; Smits, G.; Mehta, R.; Isakova, T.; Wolf, M.; Chertow, G.M. A Pilot Randomized Trial of Ferric Citrate Coordination Complex for the Treatment of Advanced CKD. J. Am. Soc. Nephrol. 2019, 30, 1495–1504. [Google Scholar] [CrossRef]
- Francis, C.; Courbon, G.; Gerber, C.; Neuburg, S.; Wang, X.; Dussold, C.; Capella, M.; Qi, L.; Isakova, T.; Mehta, R. Ferric Citrate Reduces Fibroblast Growth Factor 23 Levels and Improves Renal and Cardiac Function in a Mouse Model of Chronic Kidney Disease. Kidney Int. 2019, in press. [Google Scholar] [CrossRef]
- Ix, J.H.; Isakova, T.; Larive, B.; Raphael, K.L.; Raj, D.S.; Cheung, A.K.; Sprague, S.M.; Fried, L.F.; Gassman, J.J.; Middleton, J.P. Effects of Nicotinamide and Lanthanum Carbonate on Serum Phosphate and Fibroblast Growth Factor-23 in CKD: The COMBINE Trial. J. Am. Soc. Nephrol. 2019, 30, 1096–1108. [Google Scholar] [CrossRef]
- Qunibi, W.; Winkelmayer, W.C.; Solomon, R.; Moustafa, M.; Kessler, P.; Ho, C.; Greenberg, J.; Diaz-Buxo, J.A. A Randomized, Double-Blind, Placebo-Controlled Trial of Calcium Acetate on Serum Phosphorus Concentrations in Patients with Advanced Non-Dialysis-Dependent Chronic Kidney Disease. BMC Nephrol. 2011, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- Young, D.O.; Cheng, S.C.; Delmez, J.A.; Coyne, D.W. The Effect of Oral Niacinamide on Plasma Phosphorus Levels in Peritoneal Dialysis Patients. Perit. Dial. Int. 2009, 29, 562–567. [Google Scholar] [PubMed]
- Shahbazian, H.; Shahbazian, H.; Zafar Mohtashami, A.; Zafar Mohtashami, A.; Ghorbani, A.; Ghorbani, A.; Abbaspour, M.R.; Abbaspour, M.R.; Belladi Musavi, S.S.; Musavi, B. Oral Nicotinamide Reduces Serum Phosphorus, Increases HDL, and Induces Thrombocytopenia in Hemodialysis Patients: A Double-Blind Randomized Clinical Trial. Nefrología (Engl. Ed.) 2011, 31, 58–65. [Google Scholar]
- Vasantha, J.; Soundararajan, P.; Vanitharani, N.; Kannan, G.; Thennarasu, P.; Neenu, G.; Reddy, C.U. Safety and Efficacy of Nicotinamide in the Management of Hyperphosphatemia in Patients on Hemodialysis. Indian J. Nephrol. 2011, 21, 245. [Google Scholar] [CrossRef]
- Takahashi, Y.; Tanaka, A.; Nakamura, T.; Fukuwatari, T.; Shibata, K.; Shimada, N.; Ebihara, I.; Koide, H. Nicotinamide Suppresses Hyperphosphatemia in Hemodialysis Patients. Kidney Int. 2004, 65, 1099–1104. [Google Scholar] [CrossRef]
- Cheng, S.C.; Young, D.O.; Huang, Y.; Delmez, J.A.; Coyne, D.W. A Randomized, Double-Blind, Placebo-Controlled Trial of Niacinamide for Reduction of Phosphorus in Hemodialysis Patients. Clin. J. Am. Soc. Nephrol. 2008, 3, 1131–1138. [Google Scholar] [CrossRef]
- Sakaguchi, Y.; Hamano, T.; Obi, Y.; Monden, C.; Oka, T.; Yamaguchi, S.; Matsui, I.; Hashimoto, N.; Matsumoto, A.; Shimada, K. A Randomized Trial of Magnesium Oxide and Oral Carbon Adsorbent for Coronary Artery Calcification in Predialysis CKD. J. Am. Soc. Nephrol. 2019, 30, 1073–1085. [Google Scholar] [CrossRef]
- Bressendorff, I.; Hansen, D.; Schou, M.; Pasch, A.; Brandi, L. The Effect of Increasing Dialysate Magnesium on Serum Calcification Propensity in Subjects with End Stage Kidney Disease A Randomized, Controlled Clinical Trial. Clin. J. Am. Soc. Nephrol. 2018, 13, 1373–1380. [Google Scholar] [CrossRef]
- Larsson, T.E.; Kameoka, C.; Nakajo, I.; Taniuchi, Y.; Yoshida, S.; Akizawa, T.; Smulders, R.A. NPT-IIb Inhibition does Not Improve Hyperphosphatemia in CKD. Kidney Int. Rep. 2018, 3, 73–80. [Google Scholar] [CrossRef]
- Katai, K.; Tanaka, H.; Tatsumi, S.; Fukunaga, Y.; Genjida, K.; Morita, K.; Kuboyama, N.; Suzuki, T.; Akiba, T.; Miyamoto, K. Nicotinamide Inhibits Sodium-Dependent Phosphate Cotransport Activity in Rat Small Intestine. Nephrol. Dial. Transplant. 1999, 14, 1195–1201. [Google Scholar] [CrossRef]
- Eto, N.; Miyata, Y.; Ohno, H.; Yamashita, T. Nicotinamide Prevents the Development of Hyperphosphataemia by Suppressing Intestinal Sodium-Dependent Phosphate Transporter in Rats with Adenine-Induced Renal Failure. Nephrol. Dial. Transplant. 2005, 20, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Steffes, M.; Bostom, A.; Ix, J.H. Effect of Niacin on FGF23 Concentration in Chronic Kidney Disease. Am. J. Nephrol. 2014, 39, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Louvet, L.; Büchel, J.; Steppan, S.; Passlick-Deetjen, J.; Massy, Z.A. Magnesium Prevents Phosphate-Induced Calcification in Human Aortic Vascular Smooth Muscle Cells. Nephrol. Dial. Transplant. 2012, 28, 869–878. [Google Scholar] [CrossRef]
- De Oca, A.M.; Guerrero, F.; Martinez-Moreno, J.M.; Madueno, J.A.; Herencia, C.; Peralta, A.; Almaden, Y.; Lopez, I.; Aguilera-Tejero, E.; Gundlach, K. Magnesium Inhibits Wnt/Β-Catenin Activity and Reverses the Osteogenic Transformation of Vascular Smooth Muscle Cells. PLoS ONE 2014, 9, e89525. [Google Scholar]
- ter Braake, A.D.; Tinnemans, P.T.; Shanahan, C.M.; Hoenderop, J.G.; de Baaij, J.H. Magnesium Prevents Vascular Calcification in Vitro by Inhibition of Hydroxyapatite Crystal Formation. Sci. Rep. 2018, 8, 2069. [Google Scholar] [CrossRef]
- Sakaguchi, Y.; Fujii, N.; Shoji, T.; Hayashi, T.; Rakugi, H.; Iseki, K.; Tsubakihara, Y.; Isaka, Y.; Committee of Renal Data Registry of the Japanese Society for Dialysis Therapy. Magnesium Modifies the Cardiovascular Mortality Risk Associated with Hyperphosphatemia in Patients Undergoing Hemodialysis: A Cohort Study. PLoS ONE 2014, 9, e116273. [Google Scholar] [CrossRef]
- Sakaguchi, Y.; Iwatani, H.; Hamano, T.; Tomida, K.; Kawabata, H.; Kusunoki, Y.; Shimomura, A.; Matsui, I.; Hayashi, T.; Tsubakihara, Y.; et al. Magnesium Modifies the Association between Serum Phosphate and the Risk of Progression to End-Stage Kidney Disease in Patients with Non-Diabetic Chronic Kidney Disease. Kidney Int. 2015, 88, 833–842. [Google Scholar] [CrossRef]
- Sakaguchi, Y.; Hamano, T.; Matsui, I.; Oka, T.; Yamaguchi, S.; Kubota, K.; Shimada, K.; Matsumoto, A.; Hashimoto, N.; Isaka, Y. Low Magnesium Diet Aggravates Phosphate-Induced Kidney Injury. Nephrol. Dial. Transplant. 2018, 34, 1310–1319. [Google Scholar] [CrossRef]
- Yao, Z.; Xu, Y.; Ma, W.; Sun, X.; Jia, S.; Zheng, Y.; Liu, X.; Fan, Y.; Wang, C. Magnesium Citrate Protects Against Vascular Calcification in an Adenine-Induced Chronic Renal Failure Rat Model. J. Cardiovasc. Pharm. 2018, 72, 270–276. [Google Scholar] [CrossRef]
- Kaesler, N.; Goettsch, C.; Weis, D.; Schurgers, L.; Hellmann, B.; Floege, J.; Kramann, R. Magnesium but Not Nicotinamide Prevents Vascular Calcification in Experimental Uraemia. Nephrol. Dial. Transplant. 2019, 1–9. [Google Scholar] [CrossRef]
- Schiavi, S.C.; Tang, W.; Bracken, C.; O’Brien, S.P.; Song, W.; Boulanger, J.; Ryan, S.; Phillips, L.; Liu, S.; Arbeeny, C. Npt2b Deletion Attenuates Hyperphosphatemia Associated with CKD. J. Am. Soc. Nephrol. 2012, 23, 1691–1700. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.; Xue, J.; Murali, S.K.; Fenton, R.A.; Rieg, J.A.D.; Rieg, T. Pharmacological Npt2a Inhibition Causes Phosphaturia and Reduces Plasma Phosphate in Mice with Normal and Reduced Kidney Function. J. Am. Soc. Nephrol. 2019, 30, 2128–2139. [Google Scholar] [CrossRef] [PubMed]
- Dussold, C.; Gerber, C.; White, S.; Wang, X.; Qi, L.; Francis, C.; Capella, M.; Courbon, G.; Wang, J.; Li, C. DMP1 Prevents Osteocyte Alterations, FGF23 Elevation and Left Ventricular Hypertrophy in Mice with Chronic Kidney Disease. Bone Res. 2019, 7, 12. [Google Scholar] [CrossRef] [PubMed]

| Class of drug | Drug | Advantages | Disadvantages |
|---|---|---|---|
| Non-calcium phosphate binders | Sevelamer | Lower intact FGF23 and urinary Pi secretion, hypolipidemic [150,151,152] | GI side effects, high pill burden, unchanged serum Pi [150,151,152] |
| Lanthanum carbonate | Good GI tolerance, lower C-term FGF23 [154] | Unchanged serum Pi and intact FGF23 [151,155,160], low solubility: tissue accumulation might cause long-term toxicity | |
| Ferric citrate | Lower serum Pi and intact FGF23, increase hemoglobin and ferritin [153,157,158] | Mild GI side effects [153,157,158] | |
| Calcium-based phosphate binders | Calcium carbonate/acetate | Controlled [150,151] or lower [161] serum Pi | Hypercalcemia, VC, unchanged or increased intact FGF23 [150,151] |
| Nicotinamide | Lower serum Pi and intact FGF23 in dialysis patients [162,163,164] | Unchanged serum Pi and intact FGF23 in non-dialysis patients [160], mild GI side effects, at high doses hepatotoxic/thrombocytopenia [162,163,164,165,166] | |
| Magnesium supplements | Oral magnesium oxide | Slower progression of VC [167] | Unchanged serum Pi, FGF23 not measured, Mild GI side effects [167] |
| Higher dialysate magnesium | Increased conversion time from primary CPP to secondary CPP (T50 test, lower serum Pi [168] | Unchanged serum Pi, FGF23 not measured [168] | |
| Pi-transporter inhibitor | NaPi-2b inhibitor | Not effective in reducing serum Pi [169] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vogt, I.; Haffner, D.; Leifheit-Nestler, M. FGF23 and Phosphate–Cardiovascular Toxins in CKD. Toxins 2019, 11, 647. https://doi.org/10.3390/toxins11110647
Vogt I, Haffner D, Leifheit-Nestler M. FGF23 and Phosphate–Cardiovascular Toxins in CKD. Toxins. 2019; 11(11):647. https://doi.org/10.3390/toxins11110647
Chicago/Turabian StyleVogt, Isabel, Dieter Haffner, and Maren Leifheit-Nestler. 2019. "FGF23 and Phosphate–Cardiovascular Toxins in CKD" Toxins 11, no. 11: 647. https://doi.org/10.3390/toxins11110647
APA StyleVogt, I., Haffner, D., & Leifheit-Nestler, M. (2019). FGF23 and Phosphate–Cardiovascular Toxins in CKD. Toxins, 11(11), 647. https://doi.org/10.3390/toxins11110647





