A Review of the Effect of Trace Metals on Freshwater Cyanobacterial Growth and Toxin Production
Abstract
1. Introduction to Cyanobacteria in Freshwater Systems
2. Nutrient Limitation
3. Sources of Nutrients
4. Colimitation and Optimal Nutrient Ratios
5. Importance of Trace Metals
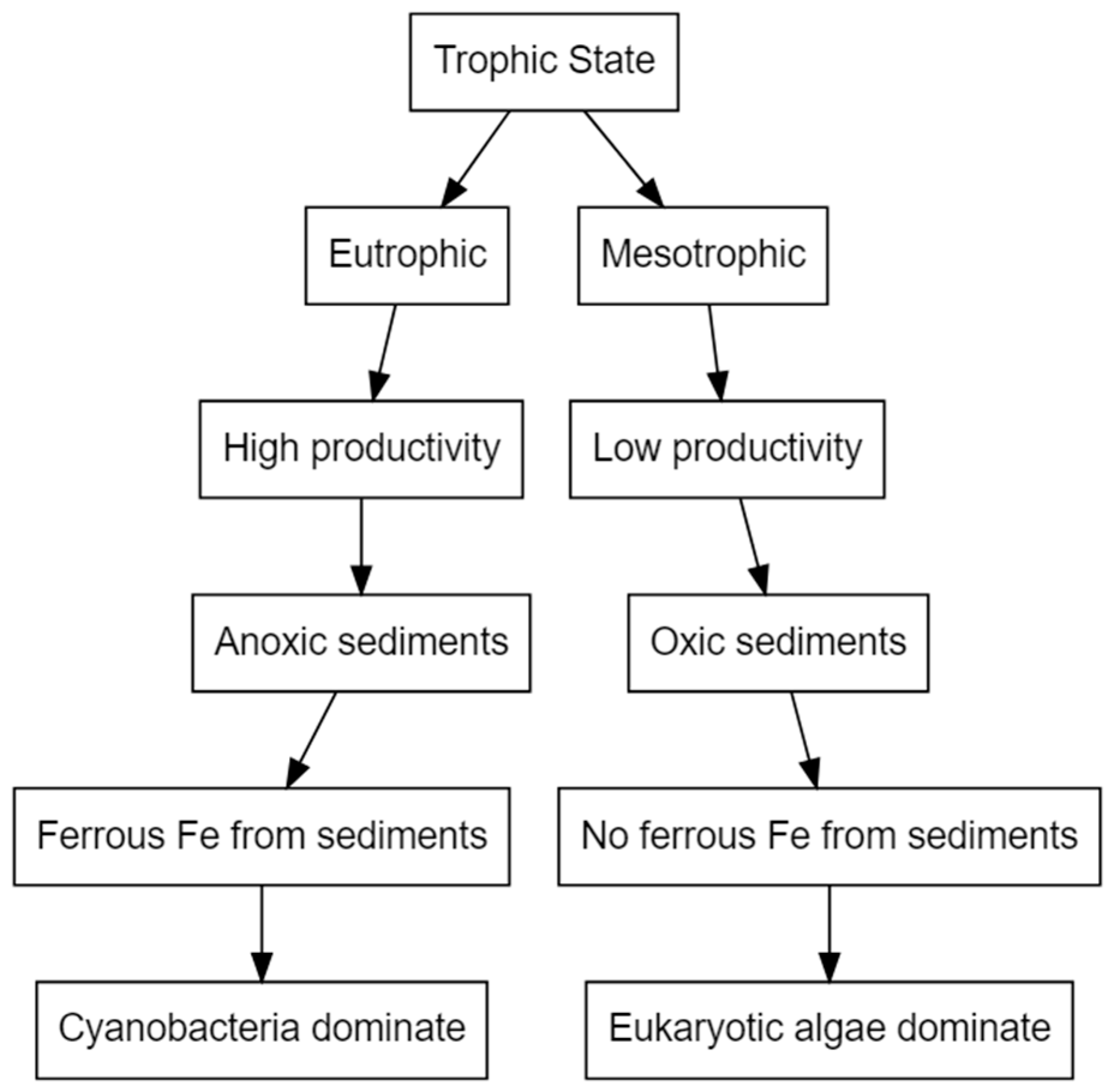
6. Iron
7. Zinc
8. Copper
9. Molybdenum
10. Cobalt
11. Manganese
12. Cyanotoxin Production
13. Trace Metals and Cyanotoxins
14. Knowledge Gaps
Author Contributions
Funding
Conflicts of Interest
References
- Cassardo, C.; Jones, J.A.A. Managing Water in a Changing World. Water 2011, 3, 618–628. [Google Scholar] [CrossRef]
- Jackson, R.B.; Carpenter, S.R.; Dahm, C.N.; McKnight, D.M.; Naiman, R.J.; Postel, S.L.; Running, S.W. Water in a changing world. Ecol. Appl. 2001, 11, 1027–1045. [Google Scholar] [CrossRef]
- Drobac, D.; Tokodi, N.; Simeunović, J.; Baltić, V.; Stanić, D.; Svirčev, Z. Human exposure to cyanotoxins and their effects on health. Arh. Hig. Rada Toksikol. 2013, 64, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Baptista, M.S.; Vasconcelos, M.T. Cyanobacteria metal interactions: Requirements, toxicity, and ecological implications. Crit. Rev. Microbiol. 2006, 32, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Dodds, W.K.; Bouska, W.W.; Eitzmann, J.L.; Pilger, T.J.; Pitts, K.L.; Riley, A.J.; Schloesser, J.T.; Thornbrugh, D.J. Eutrophication of U.S. Freshwaters: Analysis of Potential Economic Damages. Environ. Sci. Technol. 2009, 43, 12–19. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Harmful Cyanobacterial Blooms: Causes, Consequences, and Controls. Microb. Ecol. 2013, 65, 995–1010. [Google Scholar] [CrossRef]
- Baptista, M.S.; Vasconcelos, V.M.; Vasconcelos, M.T.S.D. Trace Metal Concentration in a Temperate Freshwater Reservoir Seasonally Subjected to Blooms of Toxin-Producing Cyanobacteria. Microb. Ecol. 2014, 68, 671–678. [Google Scholar] [CrossRef]
- Quiblier, C.; Susanna, W.; Isidora, E.S.; Mark, H.; Aurélie, V.; Jean-François, H. A review of current knowledge on toxic benthic freshwater cyanobacteria—Ecology, toxin production and risk management. Water Res. 2013, 47, 5464–5479. [Google Scholar]
- Carmichael, W.W. Health Effects of Toxin-Producing Cyanobacteria: “The CyanoHABs”. Hum. Ecol. Risk Assess. An. Int. J. 2001, 7, 1393–1407. [Google Scholar] [CrossRef]
- Ou, H.; Gao, N.; Wei, C.; Deng, Y.; Qiao, J. Immediate and long-term impacts of potassium permanganate on photosynthetic activity, survival and microcystin-LR release risk of Microcystis aeruginosa. J. Hazard. Mater. 2012, 219–220, 267–275. [Google Scholar] [CrossRef]
- Holland, A.; Kinnear, S. Interpreting the Possible Ecological Role(s) of Cyanotoxins: Compounds for Competitive Advantage and/or Physiological Aide? Mar. Drugs 2013, 11, 2239–2258. [Google Scholar] [CrossRef] [PubMed]
- Facey, J.A.; Steele, J.R.; Violi, J.P.; Mitrovic, S.M.; Cranfield, C. An examination of microcystin-LR accumulation and toxicity using tethered bilayer lipid membranes (tBLMs). Toxicon 2019, 158, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Sciuto, K.; Moro, I. Cyanobacteria: The bright and dark sides of a charming group. Biodivers. Conserv. 2015, 24, 711–738. [Google Scholar] [CrossRef]
- Svirčev, Z.B.; Tokodi, N.; Drobac, D.; Codd, G.A. Cyanobacteria in aquatic ecosystems in Serbia: Effects on water quality, human health and biodiversity. Syst. Biodivers. 2014, 12, 261–270. [Google Scholar] [CrossRef]
- Pilotto, L.S.; Douglas, R.M.; Burch, M.D.; Cameron, S. Health effects of exposure to cyanobacteria (blue-green algae) during recreational water–related activities. Aust. N. Z. J. Public Health 1997, 21, 562–566. [Google Scholar] [CrossRef]
- Landsberg, J. The Effects of Harmful Algal Blooms on Aquatic Organisms. Rev. Fish. Sci. 2002, 10, 113–390. [Google Scholar] [CrossRef]
- Paerl, H.W.; Fulton, R.S.; Moisander, P.H.; Dyble, J. Harmful freshwater algal blooms, with an emphasis on cyanobacteria. Sci. World J. 2011, 1, 76–113. [Google Scholar]
- Carpenter, S.R.; Caraco, N.F.; Correll, D.L.; Howarth, R.W.; Sharpley, A.N.; Smith, V.H. Nonpoint Pollution of Surface Waters with Phosphorus and Nitrogen. Ecol. Appl. 1998, 8, 559–568. [Google Scholar] [CrossRef]
- Heisler, J.; Glibert, P.M.; Burkholder, J.M.; Anderson, D.M.; Cochlan, W.; Dennison, W.C.; Dortch, Q.; Gobler, C.J.; Heil, C.A.; Humphries, E.; et al. Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae 2008, 8, 3–13. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Oliver, R.L.; Rees, C.; Bowling, L.C.; Buckney, R.T. Critical flow velocities for the growth and dominance of Anabaena circinalis in some turbid freshwater rivers. Freshw. Biol. 2003, 48, 164–174. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Hardwick, L.; Dorani, F. Use of flow management to mitigate cyanobacterial blooms in the Lower Darling River, Australia. J. Plankton Res. 2011, 33, 229–241. [Google Scholar] [CrossRef]
- Paerl, H.W.; Hall, N.S.; Calandrino, E.S. Controlling harmful cyanobacterial blooms in a world experiencing anthropogenic and climatic-induced change. Sci. Total Environ. 2011, 409, 1739–1745. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Rigosi, A.; Carey, C.C.; Ibelings, B.W.; Brookes, J.D. The interaction between climate warming and eutrophication to promote cyanobacteria is dependent on trophic state and varies among taxa. Limnol. Oceanogr. 2014, 59, 99–114. [Google Scholar] [CrossRef]
- Dignum, M.; Matthijs, H.C.P.; Pel, R.; Laanbroek, H.J.; Mur, L.R. Nutrient Limitation of Freshwater Cyanobacteria. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 65–86. [Google Scholar]
- Xu, H.; Paerl, H.W.; Qin, B.; Zhu, G.; Gao, G. Nitrogen and phosphorus inputs control phytoplankton growth in eutrophic Lake Taihu, China. Limnol. Oceanogr. 2010, 55, 420–432. [Google Scholar] [CrossRef]
- Paerl, H.W.; Xu, H.; McCarthy, M.J.; Zhu, G.; Qin, B.; Li, Y.; Gardner, W.S. Controlling harmful cyanobacterial blooms in a hyper-eutrophic lake (Lake Taihu, China): The need for a dual nutrient (N & P) management strategy. Water Res. 2011, 45, 1973–1983. [Google Scholar] [PubMed]
- Mueller, S.; Mitrovic, S.M. Phytoplankton co-limitation by nitrogen and phosphorus in a shallow reservoir: Progressing from the phosphorus limitation paradigm. Hydrobiologia 2014, 744, 255–269. [Google Scholar] [CrossRef]
- Paerl, H.W.; Fulton, R.S. Ecology of Harmful Cyanobacteria. In Ecology of Harmful Algae; Granéli, E., Turner, J.T., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 95–109. [Google Scholar]
- Schindler, D.W.; Carpenter, S.R.; Chapra, S.C.; Hecky, R.E.; Orihel, D.M. Reducing phosphorus to curb lake eutrophication is a success. Environ. Sci. Technol. 2016, 50, 8923–8929. [Google Scholar] [CrossRef] [PubMed]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Controlling eutrophication: Nitrogen and phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef]
- Bowling, L. Occurrence and possible causes of a severe cyanobacterial bloom in Lake Cargelligo, New South Wales. Mar. Freshw. Res. 1994, 45, 737–745. [Google Scholar] [CrossRef]
- Twiss, M.R.; Auclair, J.; Charlton, M.N. An investigation into iron-stimulated phytoplankton productivity in epipelagic Lake Erie during thermal stratification using trace metal clean techniques. Can. J. Fish. Aquat. Sci. 2000, 57, 86–95. [Google Scholar] [CrossRef]
- North, R.L.; Guildford, S.J.; Smith, R.E.H.; Havens, S.M.; Twiss, M.R. Evidence for phosphorus, nitrogen, and iron colimitation of phytoplankton communities in Lake Erie. Limnol. Oceanogr. 2007, 52, 315–328. [Google Scholar] [CrossRef]
- Downs, T.M.; Schallenberg, M.; Burns, C.W. Responses of lake phytoplankton to micronutrient enrichment: A study in two New Zealand lakes and an analysis of published data. Aquat. Sci. 2008, 70, 347–360. [Google Scholar] [CrossRef]
- Molot, L.A.; Watson, S.B.; Creed, I.F.; Trick, C.G.; Mccabe, S.K.; Verschoor, M.J.; Sorichetti, R.J.; Powe, C.; Venkiteswaran, J.J.; Schiff, S.L. A novel model for cyanobacteria bloom formation: The critical role of anoxia and ferrous iron. Freshw. Biol. 2014, 59, 1323–1340. [Google Scholar] [CrossRef]
- Zhang, X.; Li, B.; Xu, H.; Wells, M.; Tefsen, B.; Qin, B. Effect of micronutrients on algae in different regions of Taihu, a large, spatially diverse, hypereutrophic lake. Water Res. 2019, 151, 500–514. [Google Scholar] [CrossRef]
- Roussiez, V.; Probst, A.; Probst, J.L. Significance of floods in metal dynamics and export in a small agricultural catchment. J. Hydrol. 2013, 499, 71–81. [Google Scholar] [CrossRef]
- Hitchcock, J.N.; Mitrovic, S.M. Highs and lows: The effect of differently sized freshwater inflows on estuarine carbon, nitrogen, phosphorus, bacteria and chlorophyll a dynamics. Estuar. Coast. Shelf Sci. 2015, 156, 71–82. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, B.; Xu, G.; Guan, Y. Characterizing fluvial heavy metal pollutions under different rainfall conditions: Implication for aquatic environment protection. Sci. Total Environ. 2018, 635, 1495–1506. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Søndergaard, M.; Lauridsen, T.; Landkildehus, F. Trophic structure, species richness and diversity in Danish lakes: Changes along a phosphorus gradient. Freshw. Biol. 2000, 45, 201–218. [Google Scholar] [CrossRef]
- Buck, N.J.; Gobler, C.J.; Sañudo-Wilhelmy, S.A. Dissolved trace element concentrations in the East River - Long Island Sound system: Relative importance of autochthonous versus allochthonous sources. Environ. Sci. Technol. 2005, 39, 3528–3537. [Google Scholar] [CrossRef]
- Schindler, D.W. Recent advances in the understanding and management of eutrophication. Limnol. Oceanogr. 2006, 51, 356–363. [Google Scholar] [CrossRef]
- Withers, P.J.A.; Sharpley, A.N. Characterization and apportionment of nutrient and sediment sources in catchments. J. Hydrol. 2008, 350, 127–130. [Google Scholar] [CrossRef]
- Carey, R.O.; Migliaccio, K.W. Contribution of wastewater treatment plant effluents to nutrient dynamics in aquatic systems. Environ. Manag. 2009, 44, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Oehmen, A.; Lemos, P.C.; Carvalho, G.; Yuan, Z.; Keller, J.; Blackall, L.L.; Reis, M.A.M. Advances in enhanced biological phosphorus removal: From micro to macro scale. Water Res. 2007, 41, 2271–2300. [Google Scholar] [CrossRef] [PubMed]
- Tam, N.F.Y.; Wong, Y.S. Retention and distribution of heavy metals in mangrove soils receiving wastewater. Environ. Pollut. 1996, 94, 283–291. [Google Scholar] [CrossRef]
- Luoma, S.N. Metal uptake by phytoplancton during a bloom in South San Francisco Bay: Implications for metal cycling in estuaries. Limnol. Oceanogr. 1998, 43, 1007–1016. [Google Scholar] [CrossRef]
- Baldwin, D.S.; Williams, J. Differential release of nitrogen and phosphorus from anoxic sediments. Chem. Ecol. 2007, 23, 243–249. [Google Scholar] [CrossRef]
- Özkundakci, D.; Hamilton, D.P.; Gibbs, M.M. Hypolimnetic phosphorus and nitrogen dynamics in a small, eutrophic lake with a seasonally anoxic hypolimnion. Hydrobiologia 2011, 661, 5–20. [Google Scholar] [CrossRef]
- Müller, S.; Mitrovic, S.M.; Baldwin, D.S. Oxygen and dissolved organic carbon control release of N, P and Fe from the sediments of a shallow, polymictic lake. J. Soils Sediments 2016, 16, 1109–1120. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Bowling, L.C.; Buckney, R.T. Vertical disentrainment of Anabaena circinalis in the turbid, freshwater Darling River, Australia: Quantifying potential benefits from buoyancy. J. Plankton Res. 2001, 23, 47–55. [Google Scholar] [CrossRef]
- Bormans, M.; Ford, P.W.; Fabbro, L. Spatial and temporal variability in cyanobacterial populations controlled by physical processes. J. Plankton Res. 2005, 27, 61–70. [Google Scholar] [CrossRef]
- Saito, M.A.; Goepfert, T.J.; Ritt, J.T. Some thoughts on the concept of colimitation: Three definitions and the importance of bioavailability. Limnol. Oceanogr. 2008, 53, 276–290. [Google Scholar] [CrossRef]
- Harpole, W.S.; Ngai, J.T.; Cleland, E.E.; Seabloom, E.W.; Borer, E.T.; Bracken, M.E.S.; Elser, J.J.; Gruner, D.S.; Hillebrand, H.; Shurin, J.B.; et al. Nutrient co-limitation of primary producer communities. Ecol. Lett. 2011, 14, 852–862. [Google Scholar] [CrossRef] [PubMed]
- Schindler, D.W.; Hecky, R.E.; Findlay, D.L.; Stainton, M.P.; Parker, B.R.; Paterson, M.J.; Beaty, K.G.; Lyng, M.; Kasian, S.E.M. Eutrophication of lakes cannot be controlled by reducing nitrogen input: Results of a 37-year whole-ecosystem experiment. Proc. Natl. Acad. Sci. USA 2008, 105, 11254–11258. [Google Scholar] [CrossRef] [PubMed]
- Buitenhuis, E.T.; Geider, R.J. A model of phytoplankton acclimation to iron-light colimitation. Limnol. Oceanogr. 2010, 55, 714–724. [Google Scholar] [CrossRef]
- Tilman, D.; Kilham, S.S.; Kilham, P. Phytoplankton Community Ecology: The Role of Limiting Nutrients. Annu. Rev. Ecol. Syst. 1982, 13, 349–372. [Google Scholar] [CrossRef]
- Redfield, A.C. The biological control of the chemical factors in the environment. Am. Sci. 1958, 46, 205–221. [Google Scholar]
- Saito, M.A.; Moffett, J.W.; DiTullio, G.R. Cobalt and nickel in the Peru upwelling region: A major flux of labile cobalt utilized as a micronutrient. Global Biogeochem. Cycles 2004, 18, 1–14. [Google Scholar] [CrossRef]
- Sunda, W.G.; Huntsman, S.A. Feedback interactions between zinc and phytoplankton in seawater. Limnol. Oceanogr. 1992, 37, 25–40. [Google Scholar] [CrossRef]
- Huertas, M.J.; López-Maury, L.; Giner-Lamia, J.; Sánchez-Riego, A.M.; Florencio, F.J. Metals in cyanobacteria: Analysis of the copper, nickel, cobalt and arsenic homeostasis mechanisms. Life 2014, 4, 865–886. [Google Scholar] [CrossRef]
- Shcolnick, S.; Keren, N. Metal Homeostasis in Cyanobacteria and Chloroplasts. Balancing Benefits and Risks to the Photosynthetic Apparatus. Plant. Physiol. 2006, 141, 805–810. [Google Scholar] [CrossRef]
- De Wever, A.; Muylaert, K.; Langlet, D.; Alleman, L.; Descy, J.P.; André, L.; Cocquyt, C.; Vyverman, W. Differential response of phytoplankton to additions of nitrogen, phosphorus and iron in Lake Tanganyika. Freshw. Biol. 2008, 53, 264–277. [Google Scholar] [CrossRef]
- Sunda, W.G. Trace Metals and Harmful Algal Blooms. In Ecology of Harmful Algae; Granéli, E., Turner, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 203–214. [Google Scholar]
- Sunda, W.G.; Huntsman, S.A. Processes regulating cellular metal accumulation and physiological effects: Phytoplankton as model systems. Sci. Total Environ. 1998, 219, 165–181. [Google Scholar] [CrossRef]
- Cavet, J.S.; Borrelly, G.P.M.; Robinson, N.J. Zn, Cu and Co in cyanobacteria: Selective control of metal availability. FEMS Microbiol. Rev. 2003, 27, 165–181. [Google Scholar] [CrossRef]
- Alexova, R.; Fujii, M.; Birch, D.; Cheng, J.; Waite, T.D.; Ferrari, B.C.; Neilan, B.A. Iron uptake and toxin synthesis in the bloom-forming Microcystis aeruginosa under iron limitation. Environ. Microbiol. 2011, 13, 1064–1077. [Google Scholar] [CrossRef]
- Vrede, T.; Tranvik, L.J. Iron constraints on planktonic primary production in oligotrophic lakes. Ecosystems 2006, 9, 1094–1105. [Google Scholar] [CrossRef]
- Fujii, M.; Dang, T.C.; Bligh, M.W.; Waite, T.D. Cellular characteristics and growth behavior of iron-limited Microcystis aeruginosa in nutrient-depleted and nutrient-replete chemostat systems. Limnol. Oceanogr. 2016, 61, 2151–2164. [Google Scholar] [CrossRef]
- Glass, J.B.; Wolfe-Simon, F.; Elser, J.J.; Anbar, A.D. Molybdenum-nitrogen co-limitation in freshwater and coastal heterocystous cyanobacteria. Limnol. Oceanogr. 2010, 55, 667–676. [Google Scholar] [CrossRef]
- Harland, F.M.J.; Wood, S.A.; Moltchanova, E.; Williamson, W.M.; Gaw, S. Phormidium autumnale growth and anatoxin-a production under iron and copper stress. Toxins 2013, 5, 2504–2521. [Google Scholar] [CrossRef]
- Kulaksiz, S.; Bau, M. Rare earth elements in the Rhine River, Germany: First case of anthropogenic lanthanum as a dissolved microcontaminant in the hydrosphere. Environ. Int. 2011, 37, 973–979. [Google Scholar] [CrossRef]
- Shen, F.; Wang, L.; Zhou, Q.; Huang, X. Effects of lanthanum on Microcystis aeruginosa: Attention to the changes in composition and content of cellular microcystins. Aquat. Toxicol. 2018, 196, 9–16. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Fernández Amandi, M.; McKenzie, L.; Furey, A.; James, K.J. Effects of selenium, iron and cobalt addition to growth and yessotoxin production of the toxic marine dinoflagellate Protoceratium reticulatum in culture. J. Exp. Mar. Bio. Ecol. 2004, 313, 337–351. [Google Scholar] [CrossRef]
- Pandey, L.K.; Han, T.; Gaur, J.P. Response of a phytoplanktonic assemblage to copper and zinc enrichment in microcosm. Ecotoxicology 2015, 24, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Arrigo, R.K. Marine microorganisms and global nutrient cycles. Nature 2005, 437, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Murphy, T.; Guo, J.; Parr, T.; Nalewajko, C. Iron-stimulated growth and microcystin production of Microcystis novacekii UAM 250. Limnologica 2009, 39, 255–259. [Google Scholar] [CrossRef]
- Raven, J.A.; Evans, M.C.W.; Korb, R.E. The role of trace metals in photosynthetic electron transport in O-2-evolving organisms. Photosynth. Res. 1999, 60, 111–149. [Google Scholar] [CrossRef]
- Sterner, R.W.; Smutka, T.M.; Mckay, R.M.L.; Xiaoming, Q.; Brown, E.T.; Sherrell, R.M. Phosphorus and trace metal limitation of algae and bacteria in Lake Superior. Limnol. Oceanogr. 2004, 49, 495–507. [Google Scholar] [CrossRef]
- Molot, L.A.; Li, G.; Findlay, D.L.; Watson, S.B. Iron-mediated suppression of bloom-forming cyanobacteria by oxine in a eutrophic lake. Freshw. Biol. 2010, 55, 1102–1117. [Google Scholar] [CrossRef]
- Sevilla, E.; Martin-Luna, B.; Vela, L.; Bes, M.T.; Fillat, M.F.; Peleato, M.L. Iron availability affects mcyD expression and microcystin-LR synthesis in Microcystis aeruginosa PCC7806. Environ. Microbiol. 2008, 10, 2476–2483. [Google Scholar] [CrossRef]
- VanBriesen, J.M.; Small, M.; Weber, C.; Wilson, J. Modelling Chemical Speciation: Thermodynamics, Kinetics and Uncertainty. Model. Pollut. Complex. Environ. Syst. 2010, 2, 133–149. [Google Scholar]
- Kraemer, S.M.; Duckworth, O.W.; Harrington, J.M.; Schenkeveld, W.D.C. Metallophores and Trace Metal Biogeochemistry. Aquat. Geochem. 2015, 21, 159–195. [Google Scholar] [CrossRef]
- Ahmed, E.; Holmström, S.J.M. Siderophores in environmental research: Roles and applications. Microb. Biotechnol. 2014, 7, 196–208. [Google Scholar] [CrossRef] [PubMed]
- Polyak, Y.; Zaytseva, T.; Medvedeva, N. Response of toxic cyanobacterium Microcystis aeruginosa to environmental pollution. Water. Air. Soil Pollut. 2013, 224, 224. [Google Scholar] [CrossRef]
- Burnat, M.; Diestra, E.; Esteve, I.; Solé, A. In situ determination of the effects of lead and copper on cyanobacterial populations in microcosms. PLoS ONE 2009, 4, e6204. [Google Scholar] [CrossRef] [PubMed]
- Lehman, J.T.; Bazzi, A.; Nosher, T.; Nriagu, J.O. Copper inhibition of phytoplankton in Saginaw Bay, Lake Huron. Can. J. Fish. Aquat. Sci. 2004, 61, 1871–1880. [Google Scholar] [CrossRef]
- Pinto, E.; Sigaud-Kutner, T.C.S.; Leitao, M.A.S.; Okamoto, O.K.; Morse, D.; Colepicolo, P. Heavy metal-induced oxidative stress in algae. J. Phycol. 2003, 1018, 1008–1018. [Google Scholar] [CrossRef]
- Bishop, W.M.; Willis, B.E.; Horton, C.T. Affinity and Efficacy of Copper Following an Algicide Exposure: Application of the Critical Burden Concept for Lyngbya wollei Control in Lay Lake, AL. Environ. Manag. 2015, 55, 983–990. [Google Scholar] [CrossRef]
- Ter Steeg, P.F.; Hanson, P.J.; Paerl, H.W. Growth-limiting quantities and accumulation of molybdenum in Anabaena oscillarioides (Cyanobacteria). Hydrobiologia 1986, 140, 143–147. [Google Scholar] [CrossRef]
- Healey, F.P. Inorganic Nutrient Uptake and Deficiency in Algae. CRC Crit. Rev. Microbiol. 1973, 3, 69–113. [Google Scholar] [CrossRef]
- Glass, J.B.; Axler, R.P.; Chandra, S.; Goldman, C.R. Molybdenum limitation of microbial nitrogen assimilation in aquatic ecosystems and pure cultures. Front. Microbiol. 2012, 3, 1–11. [Google Scholar] [CrossRef]
- Cole, J.J.; Lane, J.M.; Marino, R.; Howarth, R.W. Molybdenum Assimilation by Cyanobacteria and Phytoplankton in Freshwater and Salt Water. Limnol. Oceanogr. 1993, 38, 25–35. [Google Scholar] [CrossRef]
- Zerkle, A.L.; House, C.H.; Cox, R.P.; Canfield, D.E. Metal limitation of cyanobacterial N2 fixation and implications for the Precambrian nitrogen cycle. Geobiology 2006, 4, 285–297. [Google Scholar] [CrossRef]
- Howarth, R.W.; Cole, J.J. Molybdenum availability, nitrogen limitation, and phytoplankton growth in natural waters. Science 1985, 229, 653–655. [Google Scholar] [CrossRef] [PubMed]
- Quigg, A. Micronutrients. In The Physiology of Microalgae; Borowitzka, M.A., Beardall, J., Raven, J.A., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 211–231. ISBN 978-3-31-924945-2. [Google Scholar]
- Rodriguez, I.B.; Ho, T.Y. Influence of Co and B12 on the growth and nitrogen fixation of Trichodesmium. Front. Microbiol. 2015, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Helliwell, K.E.; Lawrence, A.D.; Holzer, A.; Kudahl, U.J.; Sasso, S.; Kräutler, B.; Scanlan, D.J.; Warren, M.J.; Smith, A.G. Cyanobacteria and Eukaryotic Algae Use Different Chemical Variants of Vitamin B12. Curr. Biol. 2016, 26, 999–1008. [Google Scholar] [CrossRef]
- Intwala, A.; Patey, T.D.; Polet, D.M.; Twiss, M.R. Nutritive Substitution of Zinc by Cadmium and Cobalt in Phytoplankton Isolated from the Lower Great Lakes. J. Great Lakes Res. 2008, 34, 1–11. [Google Scholar] [CrossRef]
- Sunda, W.G.; Huntsman, S.A. Cobalt and zinc interreplacement in marine phytoplankton: Biological and geochemical implications. Limnol. Oceanogr. 1995, 40, 1404–1417. [Google Scholar] [CrossRef]
- Saito, M.A.; Moffett, J.W.; Chisholm, S.W.; Waterbury, J.B. Cobalt limitation and uptake in Prochlorococcus. Limnol. Oceanogr. 2002, 47, 1629–1636. [Google Scholar] [CrossRef]
- Ji, Y.; Sherrell, R.M. Differential effects of phosphorus limitation on cellular metals in Chlorella and Microcystis. Limnol. Oceanogr. 2008, 53, 1790–1804. [Google Scholar] [CrossRef]
- Pandey, M.; Tiwari, D.N. Characteristics of alkaline phosphatase in cyanobacterial strains and in an APasedef mutant of Nostoc muscorum. World J. Microbiol. Biotechnol. 2003, 19, 279–284. [Google Scholar] [CrossRef]
- Salomon, E.; Keren, N. Manganese Limitation Induces Changes in the Activity and in the Organization of Photosynthetic Complexes in the Cyanobacterium Synechocystis sp Strain PCC 6803. Plant. Physiol. 2011, 155, 571–579. [Google Scholar] [CrossRef]
- Utkilen, H.; Gjolme, N. Iron-stimulated toxin production in Microcystis aeruginosa. Appl. Environ. Microbiol. 1995, 61, 797–800. [Google Scholar] [PubMed]
- Yeung, A.C.Y.; D’Agostino, P.M.; Poljak, A.; McDonald, J.; Bligh, M.W.; Waite, T.D.; Neilan, B.A. Physiological and proteomic responses of continuous cultures of Microcystis aeruginosa PCC 7806 to changes in iron bioavailability and growth rate. Appl. Environ. Microbiol. 2016, 82, 5918–5929. [Google Scholar] [CrossRef] [PubMed]
- Gouvêa, S.P.; Boyer, G.L.; Twiss, M.R. Influence of ultraviolet radiation, copper, and zinc on microcystin content in Microcystis aeruginosa (Cyanobacteria). Harmful Algae 2008, 7, 194–205. [Google Scholar] [CrossRef]
- Lukac, M.; Aegerter, R. Influence of trace metals on growth and toxin production of Microcystis aeruginosa. Toxicon 1993, 31, 293–305. [Google Scholar] [CrossRef]
- Mowe, M.A.D.; Abbas, F.; Porojan, C.; Mitrovic, S.M.; Lim, R.P.; Furey, A.; Yeo, D.C.J. Roles of nitrogen and phosphorus in growth responses and toxin production (using LC-MS/MS) of tropical Microcystis ichthyoblabe and M. flos-aquae. J. Appl. Phycol. 2016, 28, 1543–1552. [Google Scholar] [CrossRef]
- Schatz, D.; Keren, Y.; Vardi, A.; Sukenik, A.; Carmeli, S.; Börner, T.; Dittmann, E.; Kaplan, A. Towards clarification of the biological role of microcystins, a family of cyanobacterial toxins. Environ. Microbiol. 2007, 9, 965–970. [Google Scholar] [CrossRef]
- Pimentel, J.S.M.; Giani, A. Microcystin production and regulation under nutrient stress conditions in toxic Microcystis strains. Appl. Environ. Microbiol. 2014, 80, 5836–5843. [Google Scholar] [CrossRef]
- Briand, E.; Yéprémian, C.; Humbert, J.F.; Quiblier, C. Competition between microcystin- and non-microcystin-producing Planktothrix agardhii (cyanobacteria) strains under different environmental conditions. Environ. Microbiol. 2008, 10, 3337–3348. [Google Scholar] [CrossRef]
- Zilliges, Y.; Kehr, J.C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Börner, T.; Dittmann, E. The cyanobacterial hepatotoxin microcystin binds to proteins and increases the fitness of Microcystis under oxidative stress conditions. PLoS ONE 2011, 6, e17615. [Google Scholar] [CrossRef]
- Rohrlack, T.; Henning, M.; Kohl, J.G. Mechanisms of the inhibitory effect of the cyanobacterium Microcystis aeruginosa on Daphnia galeata’s ingestion rate. J. Plankton Res. 1999, 21, 1489–1500. [Google Scholar] [CrossRef]
- Harke, M.J.; Steffen, M.M.; Gobler, C.J.; Otten, T.G.; Wilhelm, S.W.; Wood, S.A.; Paerl, H.W. A review of the global ecology, genomics, and biogeography of the toxic cyanobacterium, Microcystis spp. Harmful Algae 2016, 54, 4–20. [Google Scholar] [CrossRef] [PubMed]
- Orr, P.T.; Jones, G.J. Relationship between microcystin production and cell division rates in nitrogen-limited Microcystis aeruginosa cultures. Limnol. Oceanogr. 1998, 43, 1604–1614. [Google Scholar] [CrossRef]
- Wiedner, C.; Visser, P.M.; Fastner, J.; Metcalf, J.S.; Codd, G.A.; Mur, L.R. Effects of Light on the Microcystin Content of Microcystis Strain PCC 7806. Applied and Environmental Microbiology 2003, 69, 1475–1481. [Google Scholar] [CrossRef] [PubMed]
- Neilan, B.A.; Pearson, L.A.; Muenchhoff, J.; Moffitt, M.C.; Dittmann, E. Environmental conditions that influence toxin biosynthesis in cyanobacteria. Environ. Microbiol. 2013, 15, 1239–1253. [Google Scholar] [CrossRef]
- Long, B.M.; Jones, G.J.; Orr, P.T. Cellular Microcystin Content in N-Limited Microcystis aeruginosa Can Be Predicted from Growth Rate. Microbiology 2001, 67, 278–283. [Google Scholar] [CrossRef]
- Ross, C.; Santiago-Vázquez, L.; Paul, V. Toxin release in response to oxidative stress and programmed cell death in the cyanobacterium Microcystis aeruginosa. Aquat. Toxicol. 2006, 78, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Humble, A.V.; Gadd, G.M.; Codd, G.A. Binding of copper and zinc to three cyanobacterial microcystins quantified by differential pulse polarography. Water Res. 1997, 31, 1679–1686. [Google Scholar] [CrossRef]
- Saito, K.; Sei, Y.; Miki, S.; Yamaguchi, K. Detection of microcystin-metal complexes by using cryospray ionization-Fourier transform ion cyclotron resonance mass spectrometry. Toxicon 2008, 51, 1496–1498. [Google Scholar] [CrossRef]
- Birch, L.; Bachofen, R. Complexing agents from microorganisms. Experientia 1990, 46, 827–834. [Google Scholar] [CrossRef]
- Martínez-Ruiz, E.B.; Martínez-Jerónimo, F. How do toxic metals affect harmful cyanobacteria? An integrative study with a toxigenic strain of Microcystis aeruginosa exposed to nickel stress. Ecotoxicol. Environ. Saf. 2016, 133, 36–46. [Google Scholar] [CrossRef]
- Huang, B.; Xu, S.; Miao, A.; Xiao, L.; Yang, L. Cadmium Toxicity to Microcystis aeruginosa PCC 7806 and Its Microcystin-Lacking Mutant. PLoS ONE 2015, 10, e0116659. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.W.; Maxwell, D.P.; Trick, C.G. Growth, iron requirements, and siderophore production in iron-limited Synechococcus PCC 7002. Limnol. Oceanogr. 1996, 41, 89–97. [Google Scholar] [CrossRef]
- Klein, A.R.; Baldwin, D.S.; Silvester, E. Proton and iron binding by the cyanobacterial toxin microcystin-LR. Environ. Sci. Technol. 2013, 47, 5178–5184. [Google Scholar] [CrossRef] [PubMed]
- Dittmann, E.; Börner, T. Genetic contributions to the risk assessment of microcystin in the environment. Toxicol. Appl. Pharmacol. 2005, 203, 192–200. [Google Scholar] [CrossRef]
- Fujii, M.; Rose, A.L.; Waite, T.D. Iron Uptake by Toxic and Nontoxic Strains of Microcystis aeruginosa. Appl. Environ. Microbiol. 2011, 77, 7068–7071. [Google Scholar] [CrossRef]
- Wang, N.X.; Zhang, X.Y.; Wu, J.; Xiao, L.; Yin, Y.; Miao, A.J.; Ji, R.; Yang, L.Y. Effects of microcystin-LR on the metal bioaccumulation and toxicity in Chlamydomonas reinhardtii. Water Res. 2012, 46, 369–377. [Google Scholar] [CrossRef]
- Amé, M.V.; Wunderlin, D.A. Effects of iron, ammonium and temperature on microcystin content by a natural concentrated Microcystis aeruginosa population. Water. Air. Soil Pollut. 2005, 168, 235–248. [Google Scholar] [CrossRef]
- Attridge, E.M.; Rowell, P. Growth, heterocyst differentiation and nitrogenase activity in the cyanobacteria Anabaena variabilis and Anabaena cylindrica in response to molybdenum and vanadium. New Phytol. 1997, 135, 517–526. [Google Scholar] [CrossRef]
- Auclair, J.C. Implications of increased UV-B induced photoreduction: Iron(II) enrichment stimulates picocyanobacterial growth and the microbial food web in clear-water acidic Canadian Shield lakes. Can. J. Fish. Aquat. Sci. 1995, 52, 1782–1788. [Google Scholar] [CrossRef]
- Cheniae, G.M.; Martin, I.F. Photoreactivation of manganese catalyst in photosynthetic oxygen evolution. Biochem. Biophys. Res. Commun. 1967, 28, 89–95. [Google Scholar] [CrossRef]
- Hyenstrand, P.; Rydin, E.; Gunnerhed, M.; Linder, J.; Blomqvist, P. Response of the cyanobacterium Gloeotrichia echinulata to iron and boron additions—An experiment from Lake Erken. Freshw. Biol. 2001, 46, 735–741. [Google Scholar] [CrossRef]
- Karlsson-Elfgren, I.; Hyenstrand, P.; Riydin, E. Pelagic growth and colony division of Gloeotrichia echinulata in Lake Erken. J. Plankton Res. 2005, 27, 145–151. [Google Scholar] [CrossRef]
- Peschek, G.A. Nitrate and nitrite reductase and hydrogenase in Anacystis nidulans grown in Fe- and Mo-deficient media. FEMS Microbiol. Lett. 1979, 6, 371–374. [Google Scholar] [CrossRef]
- Sorichetti, R.J.; Creed, I.F.; Trick, C.G. Evidence for iron-regulated cyanobacterial predominance in oligotrophic lakes. Freshw. Biol. 2014, 59, 679–691. [Google Scholar] [CrossRef]
- Wurtsbaugh, W.A.; Horne, A.J. Iron in Eutrophic Clear Lake, California: Its Importance for Algal Nitrogen Fixation and Growth. Can. J. Fish. Aquat. Sci. 1983, 40, 1419–1429. [Google Scholar] [CrossRef]
- Corman, J.R.; McIntyre, P.B.; Kuboja, B.; Mbemba, W.; Fink, D.; Wheeler, C.W.; Gans, C.; Michel, E.; Flecker, A.S. Upwelling couples chemical and biological dynamics across the littoral and pelagic zones of Lake Tanganyika, East Africa. Limnol. Oceanogr. 2010, 55, 214–224. [Google Scholar] [CrossRef]
- Omidi, A.; Esterhuizen-Londt, M.; Pflugmacher, S. Still challenging: The ecological function of the cyanobacterial toxin microcystin—What we know so far. Toxin Rev. 2018, 37, 87–105. [Google Scholar] [CrossRef]
- Zurawell, R.W.; Chen, H.; Burke, J.M.; Prepas, E.E. Hepatotoxic Cyanobacteria: A Review of the Biological Importance of Microcystins in Freshwater Environments. J. Toxicol. Environ. Heal. Part. B 2005, 8, 1–37. [Google Scholar] [CrossRef]
- Wiegand, C.; Pflugmacher, S. Ecotoxicological effects of selected cyanobacterial secondary metabolites a short review. Toxicol. Appl. Pharmacol. 2005, 203, 201–218. [Google Scholar] [CrossRef]
- Pearson, L.; Mihali, T.; Moffitt, M.; Kellmann, R.; Neilan, B. On the chemistry, toxicology and genetics of the cyanobacterial toxins, microcystin, nodularin, saxitoxin and cylindrospermopsin. Mar. Drugs 2010, 8, 1650–1680. [Google Scholar] [CrossRef]
- Mowe, M.A.D.; Mitrovic, S.M.; Lim, R.P.; Furey, A.; Yeo, D.C.J. Tropical cyanobacterial blooms: A review of prevalence, problem taxa, toxins and influencing environmental factors. J. Limnol. 2015, 74, 205–224. [Google Scholar] [CrossRef]
- Stroom, J.M.; Kardinaal, W.E.A. How to combat cyanobacterial blooms: Strategy toward preventive lake restoration and reactive control measures. Aquat. Ecol. 2016, 50, 541–576. [Google Scholar] [CrossRef]
- Likens, G.E. Inland Waters. In Encyclopedia of Inland Waters; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Downing, T.G.; Phelan, R.R.; Downing, S. A potential physiological role for cyanotoxins in cyanobacteria of arid environments. J. Arid Environ. 2015, 112, 147–151. [Google Scholar] [CrossRef]
- Nogueira, P.; Domingues, R.B.; Barbosa, A.B. Are microcosm volume and sample pre-filtration relevant to evaluate phytoplankton growth? J. Exp. Mar. Biol. Ecol. 2014, 461, 323–330. [Google Scholar] [CrossRef]
| Location | Taxa | Co | Cu | Fe | Mn | Mo | Zn | Mix | Study |
|---|---|---|---|---|---|---|---|---|---|
| Culture | Microcystis aeruginosa | T− | Alexova et al. [68] (2011) | ||||||
| Culture | Microcystis aeruginosa | T+ | Amé and Wunderlin [132] (2005) | ||||||
| Culture | Anabaena spp. | C | Attridge and Rowell [133] (1997) | ||||||
| Canadian Shield lakes | Pico-cyanobacteria | C | Auclair [134] (1995) | ||||||
| Torrão reservoir | Microcystis aeruginosa | N | N | N | N | N | N | Baptista et al. [7] (2014) | |
| Culture | Anacystis sp. | Y | Cheniae and Martin [135] (1967) | ||||||
| Lake Tanganyika, East Africa | Pico-cyanobacteria | Y, C | de Wever et al. [64] (2008) | ||||||
| Lake Waihola, New Zealand | Anabaena flos-aquae | Y | Y | N | Y | Y | Downs et al. [35] (2008) | ||
| Lake Mahinerangi, New Zealand | N | N | N | N | N | ||||
| Culture | Microcystis aeruginosa | Y | Fujii et al. [70] (2016) | ||||||
| Culture | Nostoc sp. | C | Glass et al. [71] (2010) | ||||||
| Culture | Microcystis aeruginosa | T± | T± | Gouvêa et al. [108] (2008) | |||||
| Culture | Phormidium autumnale | Y, T± | Y, T± | Harland et al. [72] (2013) | |||||
| Lake Erken, Sweden | Gloeotrichia echinulate | C | Hyenstrand et al. [136] (2001) | ||||||
| Lake Erken, Sweden | Gloeotrichia echinulate | C | N | Karlsson-Elfgren et al. [137] (2005) | |||||
| Culture | Microcystis novacekii | Y, T+ | Li et al. [78] (2009) | ||||||
| Culture | Microcystis aeruginosa | N, T± | Y, T− | N, T± | Y, T± | Lukac and Aegerter [109] (1993) | |||
| Lake 227, Experimental Lakes Area | Aphanizomenon schindlerii | Y | Molot et al. [81] (2010) | ||||||
| Anabaena flos-aquae, Synechococcus | Y | ||||||||
| Culture | Anacystis nidulans | Y | N | Peschek [138] (1979) | |||||
| Culture | Microcystis aeruginosa | N | Y, T+ | Polyak et al. [86] (2013) | |||||
| Culture | Synechocystis | Y | Salomon and Keren [105] (2011) | ||||||
| Culture | Microcystis aeruginosa | T− | Sevilla et al. [82] (2008) | ||||||
| Laurentian Great Lakes | Total cyanophyta | C | Sorichetti et al. [139] (2014) | ||||||
| Culture | Anabaena oscillarioides | C | ter Steeg et al. [91] (1986) | ||||||
| Culture | Microcystis aeruginosa | T+ | Utkilen and Gjolme [106] (1995) | ||||||
| Clear Lake, California | Aphanizomenon flos-aquae | C | Wurtsbaugh and Horne [140] (1983) | ||||||
| Culture | Microcystis aeruginosa | Y, T− | Yeung et al. [107] (2016) | ||||||
| Lake Taihu, China | Total cyanophyta | N | Y, C | Y, C | N | N | Zhang et al. [37] (2019) | ||
| Microcystis aeruginosa | N | Y, C | C | N | N |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Facey, J.A.; Apte, S.C.; Mitrovic, S.M. A Review of the Effect of Trace Metals on Freshwater Cyanobacterial Growth and Toxin Production. Toxins 2019, 11, 643. https://doi.org/10.3390/toxins11110643
Facey JA, Apte SC, Mitrovic SM. A Review of the Effect of Trace Metals on Freshwater Cyanobacterial Growth and Toxin Production. Toxins. 2019; 11(11):643. https://doi.org/10.3390/toxins11110643
Chicago/Turabian StyleFacey, Jordan A., Simon C. Apte, and Simon M. Mitrovic. 2019. "A Review of the Effect of Trace Metals on Freshwater Cyanobacterial Growth and Toxin Production" Toxins 11, no. 11: 643. https://doi.org/10.3390/toxins11110643
APA StyleFacey, J. A., Apte, S. C., & Mitrovic, S. M. (2019). A Review of the Effect of Trace Metals on Freshwater Cyanobacterial Growth and Toxin Production. Toxins, 11(11), 643. https://doi.org/10.3390/toxins11110643




