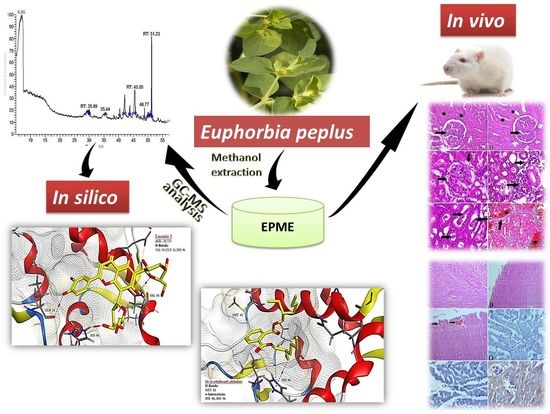
Investigation of the In-Vivo Cytotoxicity and the In Silico-Prediction of MDM2-p53 Inhibitor Potential of Euphorbia peplus Methanolic Extract in Rats
Abstract
1. Introduction
2. Results
2.1. Experimental Observations
2.2. GC-MS Profile of EPME
2.3. Effects of EPME on Cardiac Injury Markers
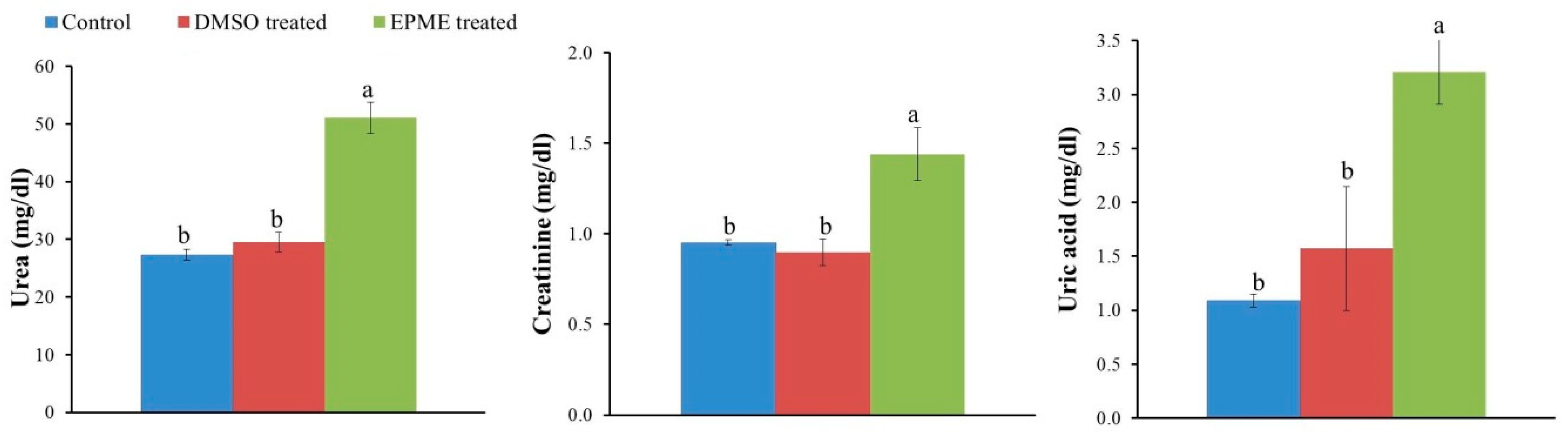
2.4. Effects on EPME on Markers of Kidney Injury
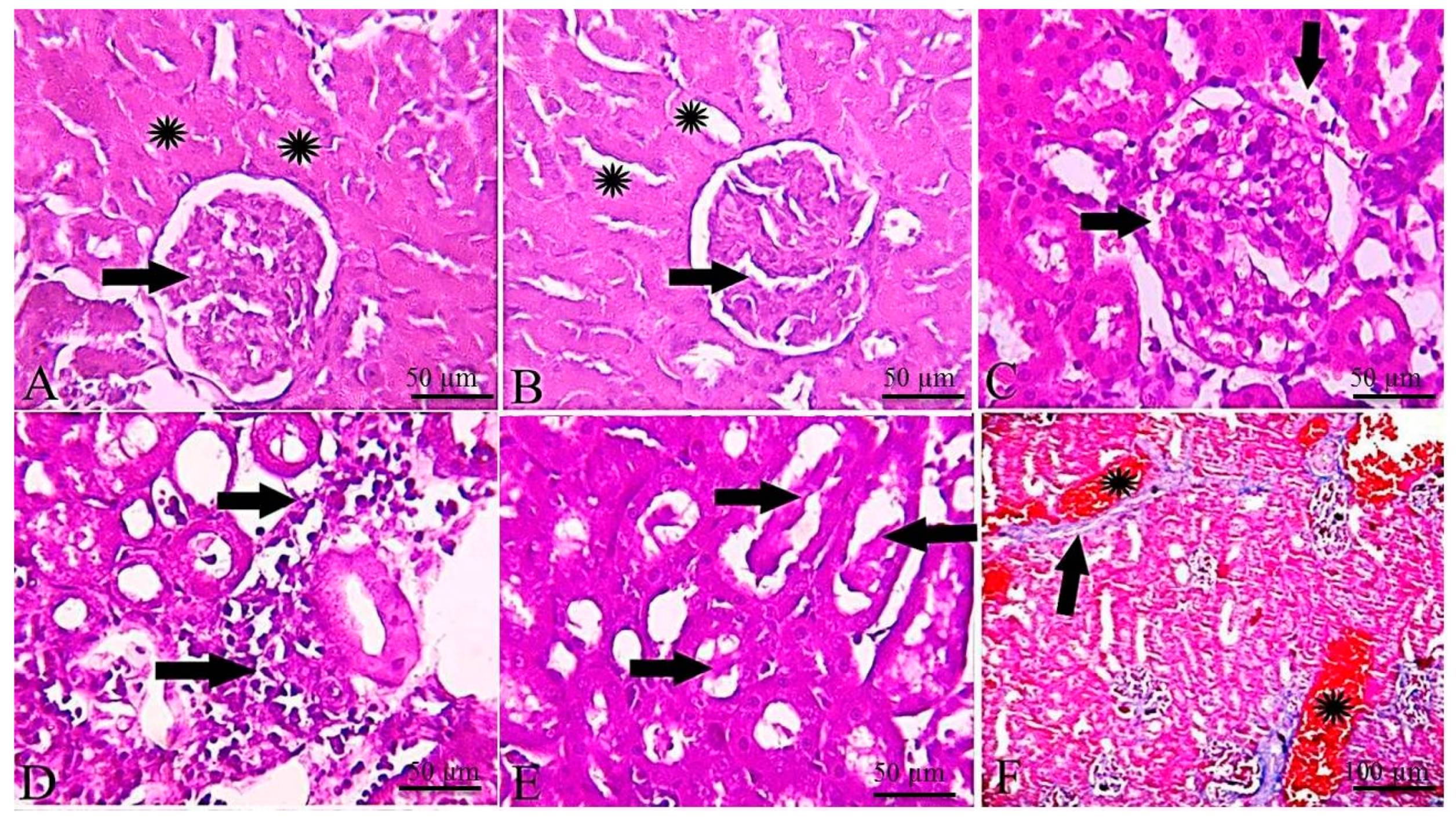
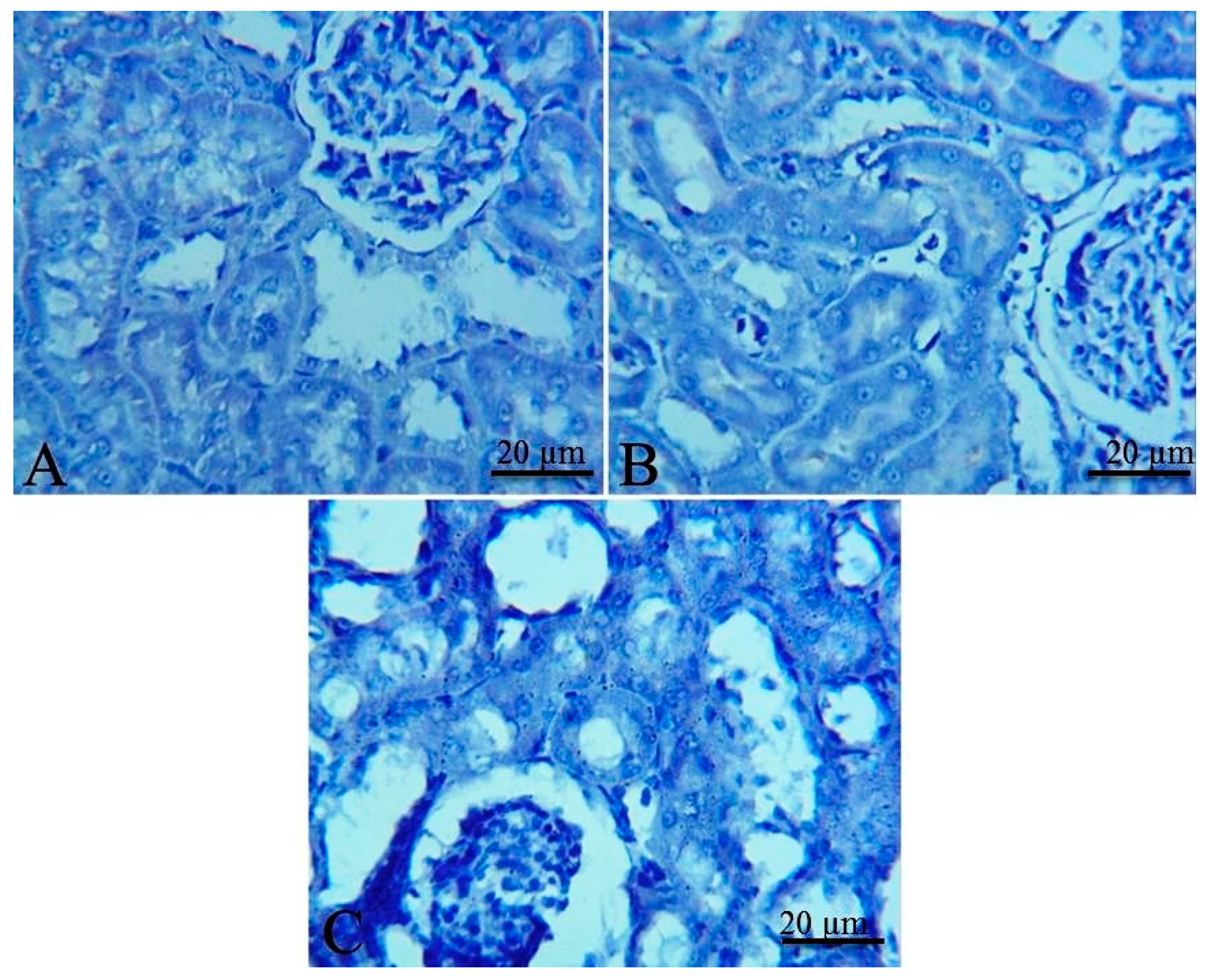
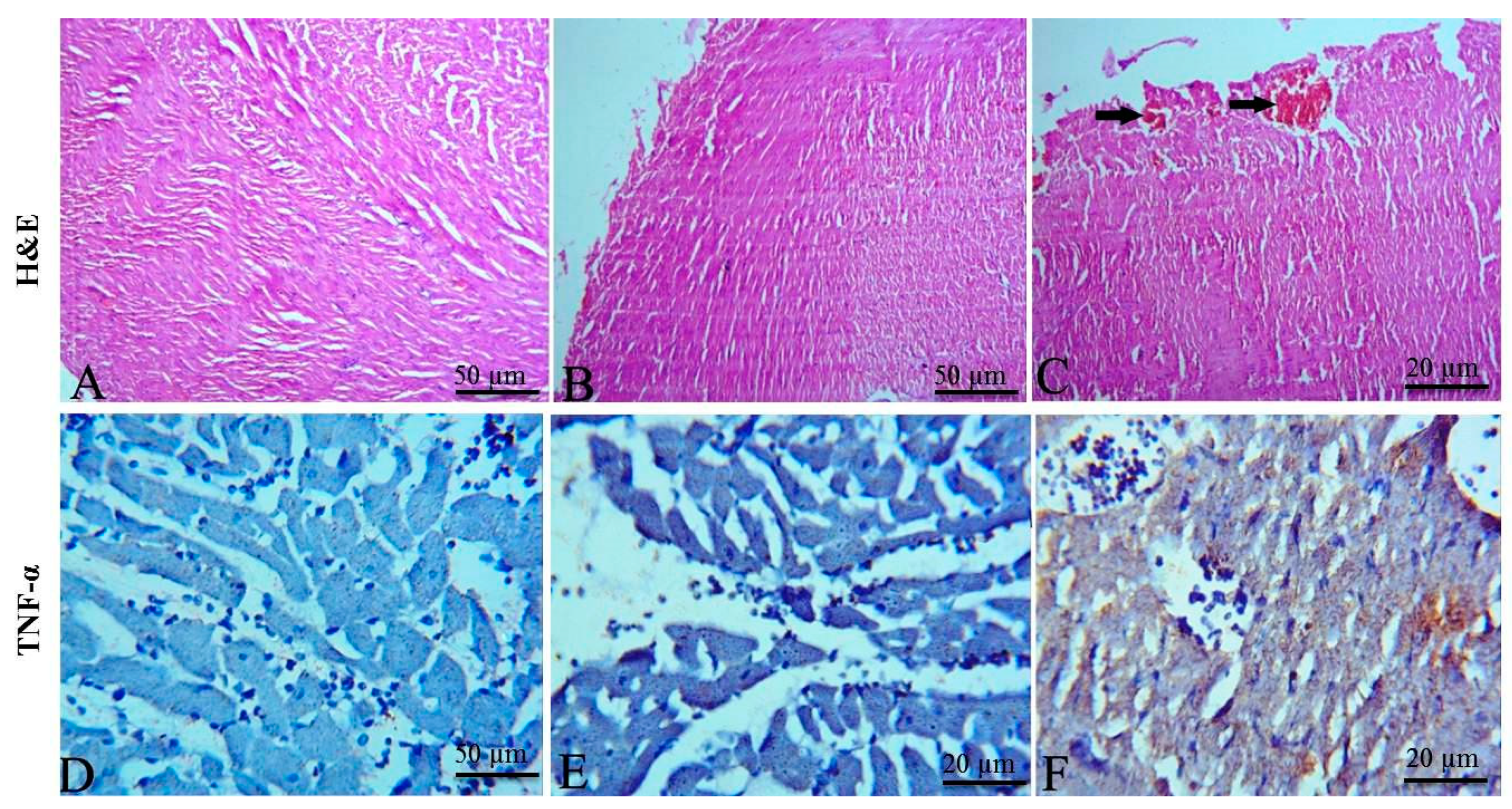
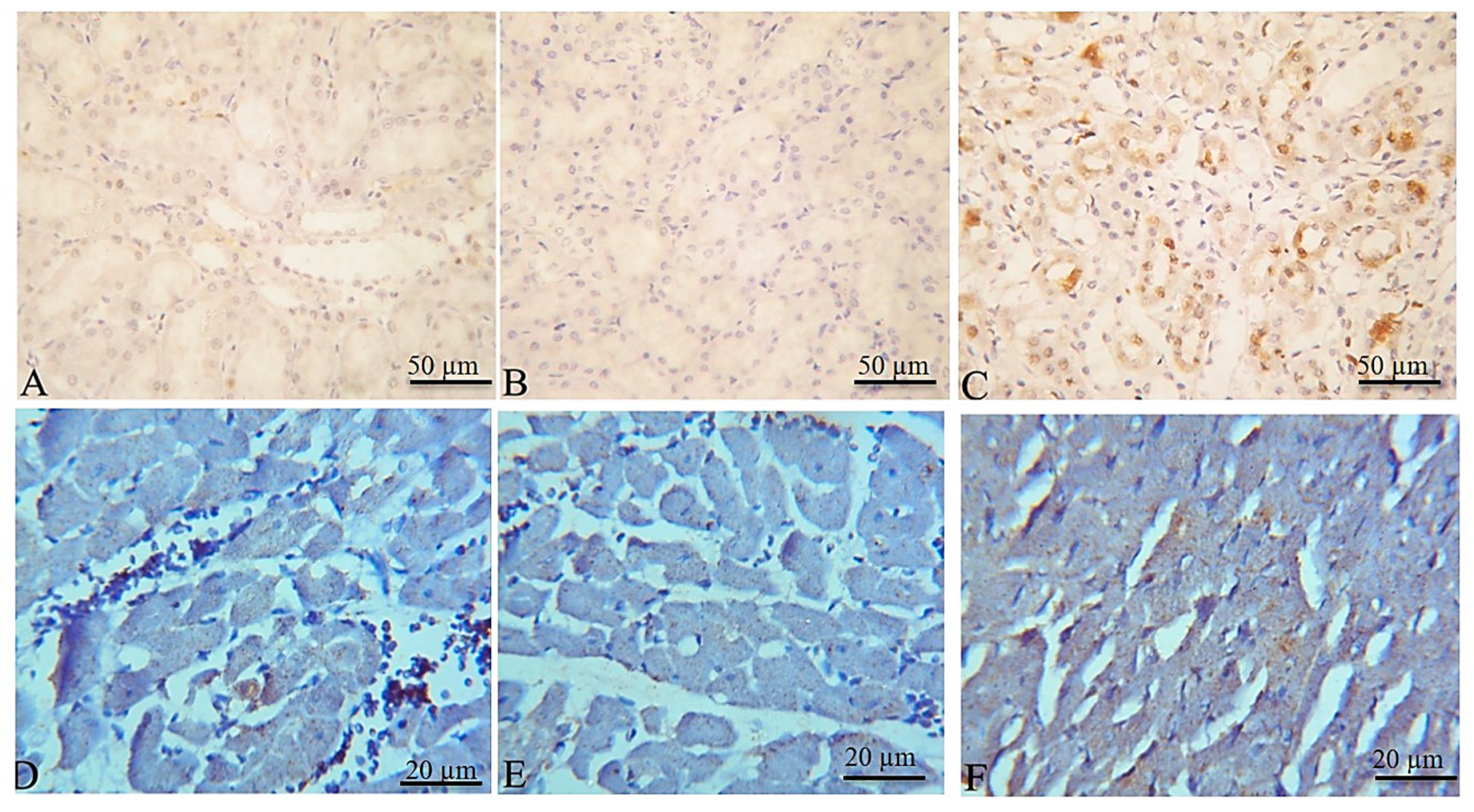
2.5. Histopathological and Immunohistochemical Findings
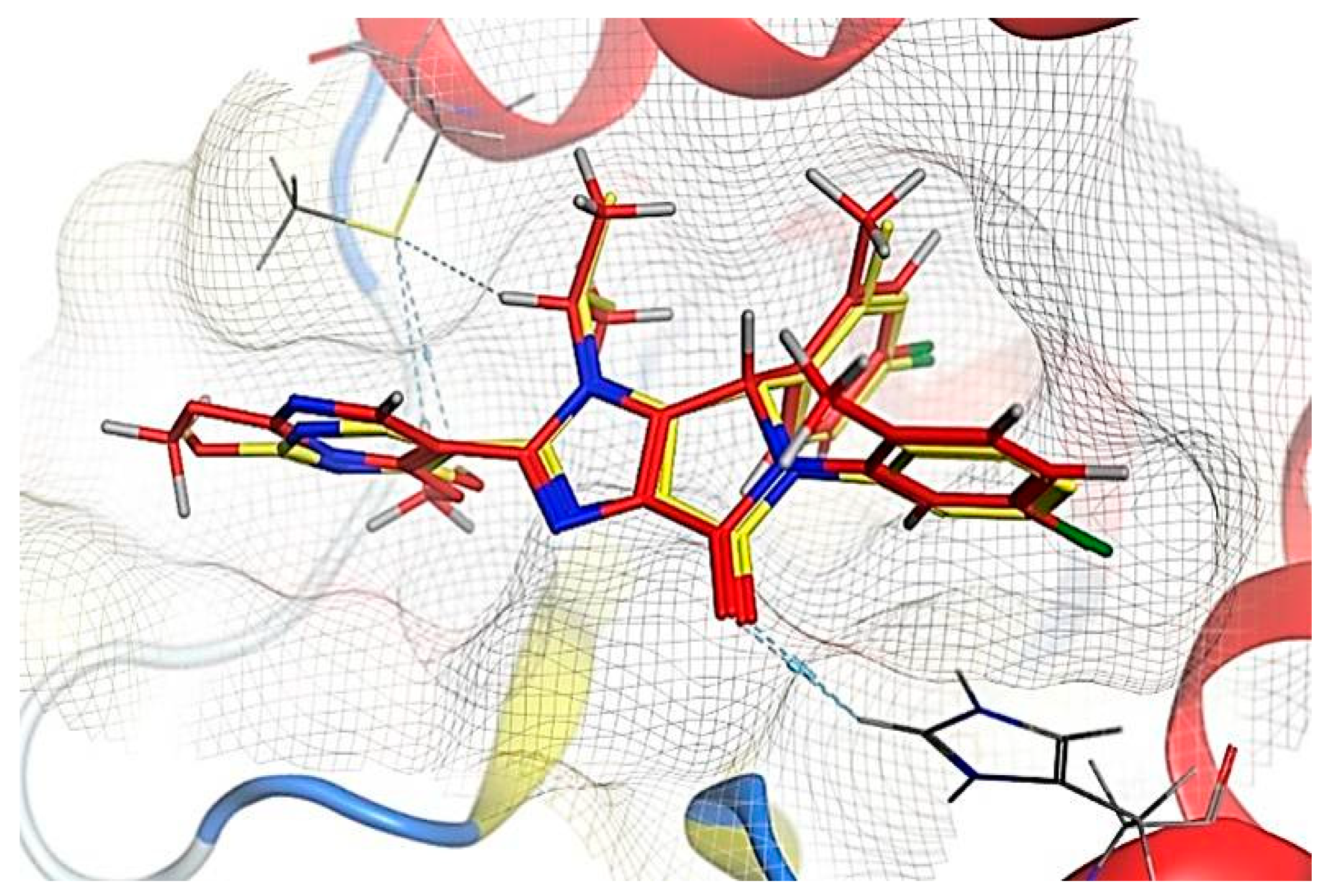
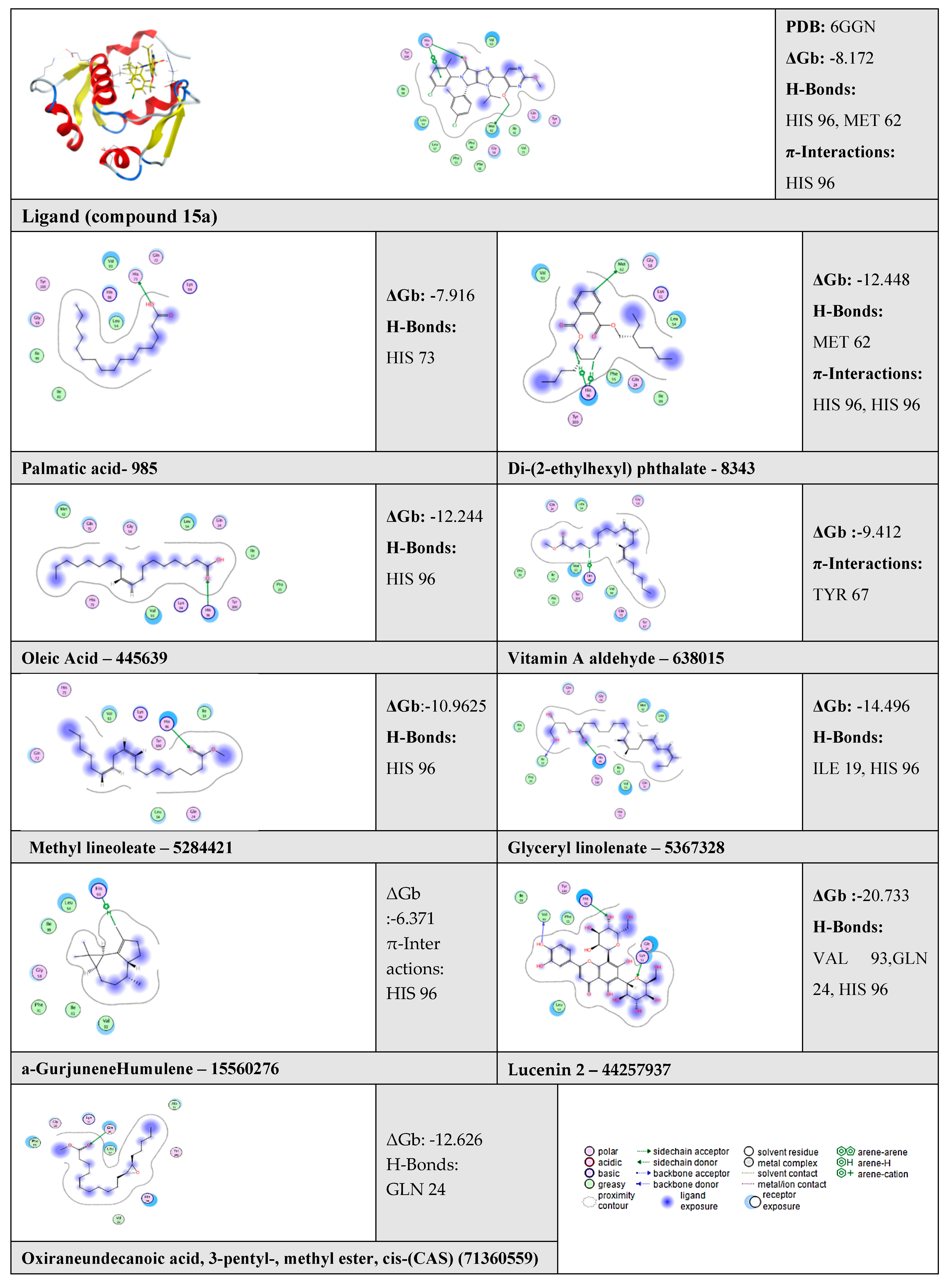
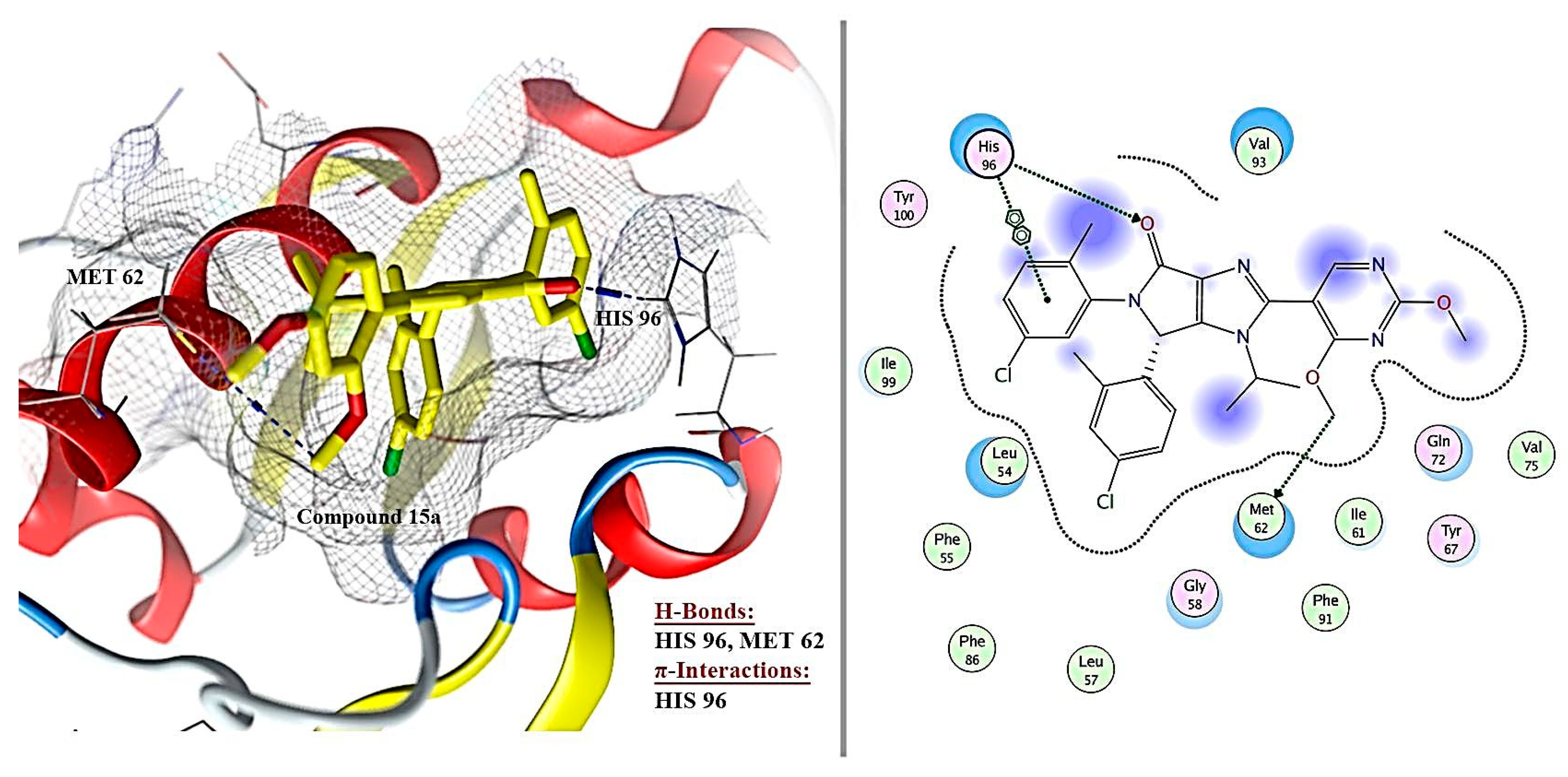
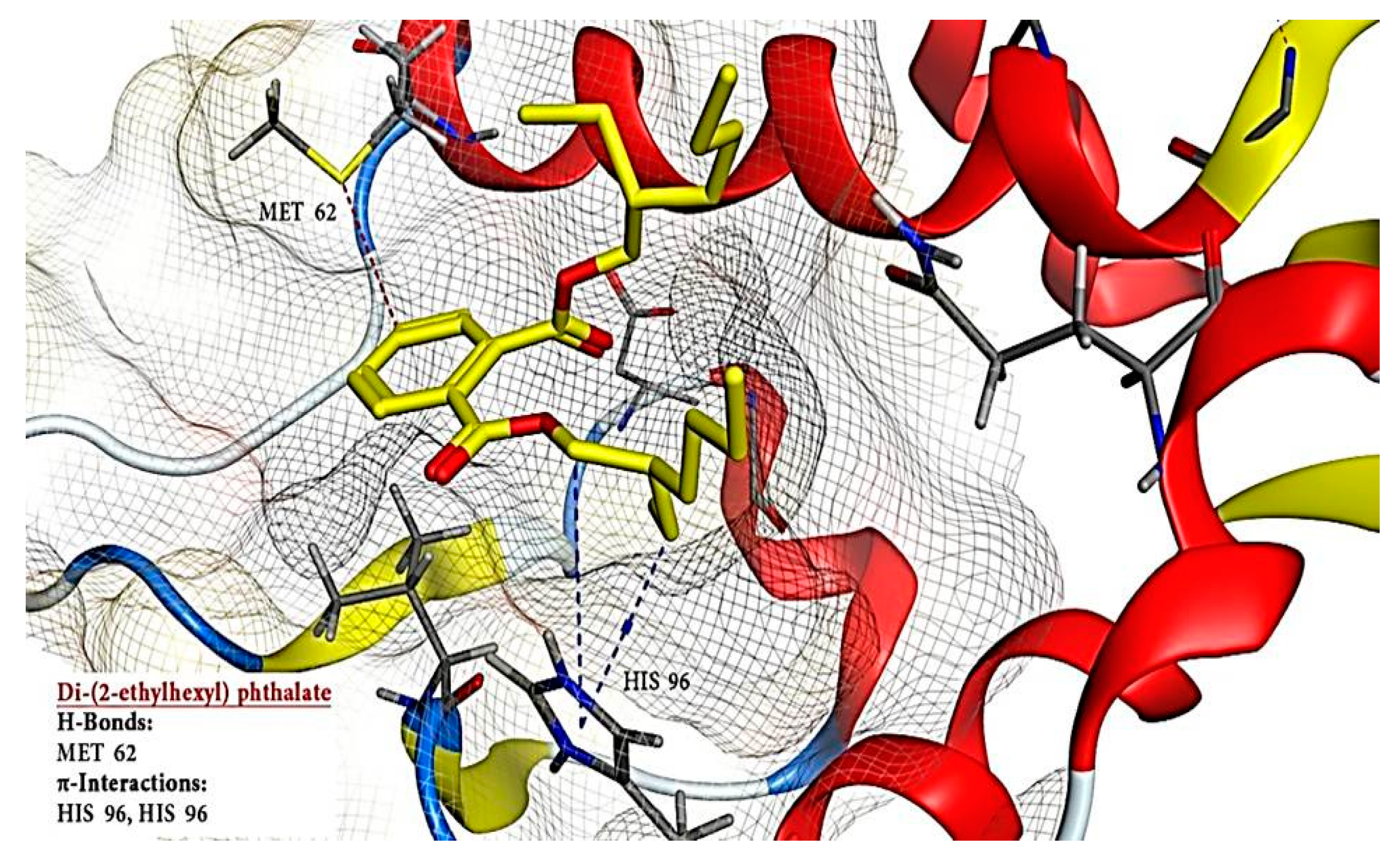
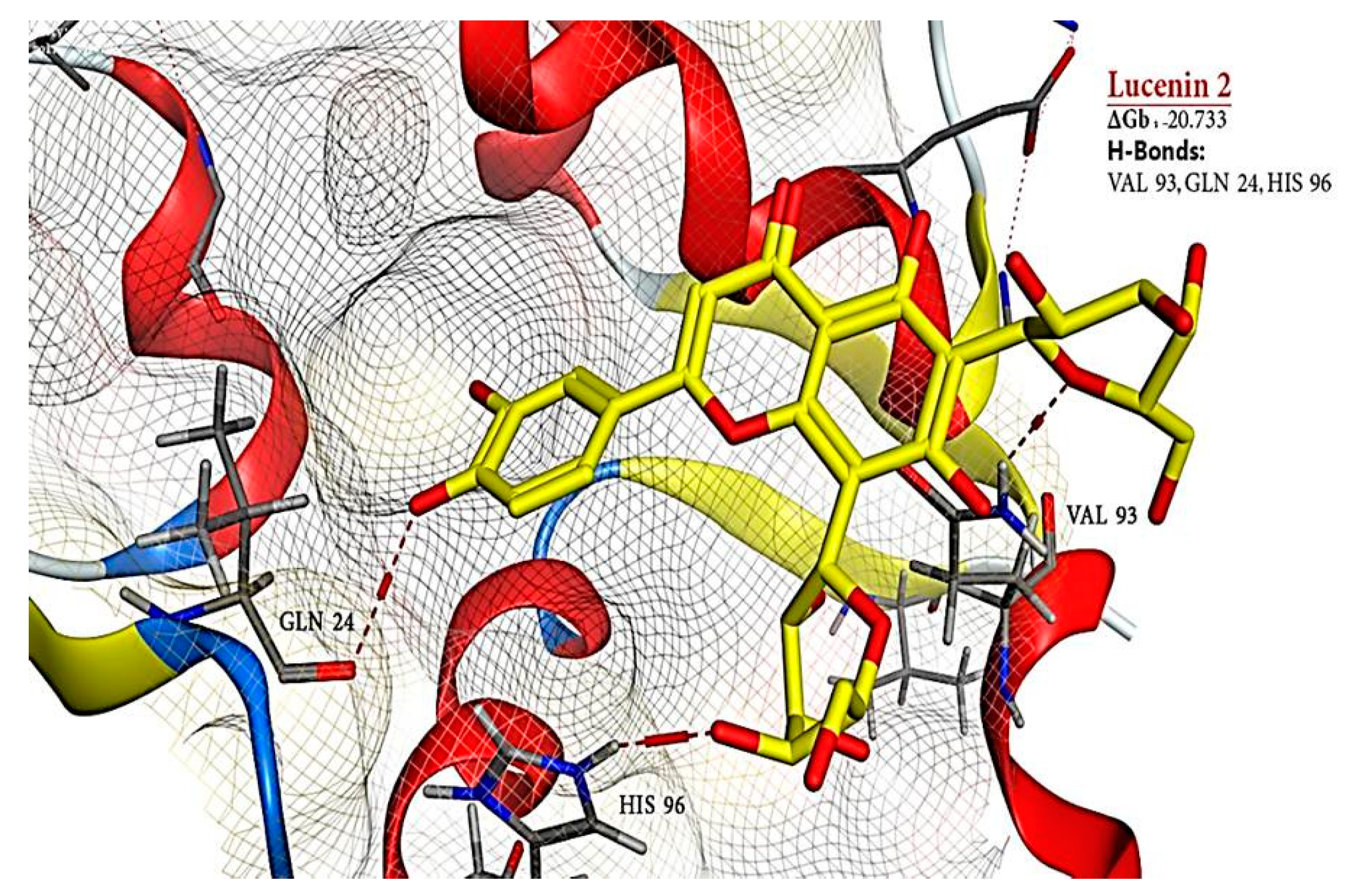
2.6. Molecular Docking Results
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Kits and Chemicals
5.2. Plant Material
5.3. Preparation of Euphorbia Peplus Methanolic Extracts (EPME)
5.4. Gas Chromatography/Mass Spectrometry Analysis (GC–MS) of EPME
5.5. Animal Grouping and Experimental Design
5.6. Sampling
5.7. Serum Biochemical Analysis
5.8. Histopathological and Immunohistochemical Investigation
5.9. Ligand Preparation for Drug-Likeness and Docking
5.10. MDM2 Protein Preparation for Docking
5.11. Docking of Isolated Euphorbia Peplus Compounds into MDM2 Protein
5.12. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Falzon, C.C.; Balabanova, A. Phytotherapy: An Introduction to Herbal Medicine. Prim. Care Clin. Off. Pract. 2017, 44, 217–227. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Traditional Medicine Strategy 2002–2005; World Health Organization: Genev, Switzerland, 2002. [Google Scholar]
- Yuet Ping, K.; Darah, I.; Chen, Y.; Sreeramanan, S.; Sasidharan, S. Acute and subchronic toxicity study of Euphorbia hirta L. methanol extract in rats. BioMed Res. Int. 2013, 2013, 182064. [Google Scholar] [CrossRef] [PubMed]
- Ertekin, V.; Selimoğlu, M.A.; Altinkaynak, S. A combination of unusual presentations of Datura stramonium intoxication in a child: Rhabdomyolysis and fulminant hepatitius. J. Emerg. Med. 2005, 28, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Ukwuani, A.; Abubakar, M.; Hassan, S.; Agaie, B. Toxicological studies of hydromethanolic leaves extract of Grewia crenata. Int. J. Pharm. Sci. Drug Res. 2012, 4, 245–249. [Google Scholar]
- Tilburt, J.C.; Kaptchuk, T.J. Herbal medicine research and global health: An ethical analysis. Bull. World Health Organ. 2008, 86, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.W.; Su, X.H.; Kiyota, H. Chemical and pharmacological research of the plants in genus Euphorbia. Chem. Rev. 2008, 108, 4295–4327. [Google Scholar] [CrossRef] [PubMed]
- Jyothi, T.; Prabhu, K.; Jayachandran, E.; Lakshminarasu, S.; Setty, R. Hepatoprotective and antioxidant activity of Euphorbia antiquorum. Pharmacogn. Mag. 2008, 4, 127. [Google Scholar]
- Teng, Y.N.; Wang, Y.; Hsu, P.L.; Xin, G.; Zhang, Y.; Morris-Natschke, S.L.; Goto, M.; Lee, K.H. Mechanism of action of cytotoxic compounds from the seeds of Euphorbia lathyris. Phytomedicine 2018, 41, 62–66. [Google Scholar] [CrossRef]
- Cruz, L.S.; de Oliveira, T.L.; Kanunfre, C.C.; Paludo, K.S.; Minozzo, B.R.; Prestes, A.P.; Wang, M.; Fernandes, D.; dos Santos, F.A.; Manda, V.K.; et al. Pharmacokinetics and cytotoxic study of euphol from Euphorbia umbellata (Bruyns) Pax latex. Phytomedicine 2018, 47, 105–112. [Google Scholar] [CrossRef]
- Adedapo, A.; Abatan, M.; Idowu, S.; Olorunsogo, O. Effects of chromatographic fractions of Euphorbia hirta on the rat serum biochemistry. Afr. J. Biomed. Res. 2005, 8, 185–189. [Google Scholar] [CrossRef]
- Al-Sultan, S.; Yehia, A. Acute toxicity of Euphorbia heliscopia in rats. Pak. J. Nutr. 2006, 5, 135–140. [Google Scholar]
- Song, Z.Q.; Mu, S.Z.; Di, Y.T.; Hao, X.J. A new jatrophane diterpenoid from Euphorbia peplus. Chin. J. Nat. Med. 2010, 8, 81–83. [Google Scholar] [CrossRef]
- Nambudiri, V. From home remedy to cancer treatment: A history of ingenol mebutate and Euphorbia peplus in dermatology. J. Am. Acad. Dermatol. 2013, 68, AB33. [Google Scholar]
- Ramsay, J.; Suhrbier, A.; Aylward, J.; Ogbourne, S.; Cozzi, S.J.; Poulsen, M.; Baumann, K.; Welburn, P.; Redlich, G.; Parsons, P. The sap from Euphorbia peplus is effective against human nonmelanoma skin cancers. Br. J. Dermatol. 2011, 164, 633–636. [Google Scholar] [CrossRef] [PubMed]
- Al-Okbi, S.Y.; El-Sayed, E.M. Effect of Ruta Graveolens L. and Euphorbia peplus L. Anti-Inflammatory Extracts on Nutritional Status of Rats and the Safety of Their Use. Indian J. Exp. Biol. 2002, 40, 45–48. [Google Scholar]
- Ali, A.A.; Sayed, H.M.; Ibrahim, S.R.; Zaher, A.M. Chemical constituents, antimicrobial, analgesic, antipyretic, and anti-inflammatory activities of Euphorbia peplus L. Phytopharmacology 2013, 4, 69–80. [Google Scholar]
- Amin, E.; Moawad, A.; Hassan, H. Biologically-guided isolation of leishmanicidal secondary metabolites from Euphorbia peplus L. Saudi Pharm. J. 2017, 25, 236–240. [Google Scholar] [CrossRef]
- Sharif, H.; Mukhtar, M.; Mustapha, Y.; Baba, G.; Lawal, A. Acute and subchronic toxicity profile of Euphorbia pulcherrima methanol extract on Wistar albino rats. Adv. Pharm. 2015, 2015, 1–9. [Google Scholar]
- Saleem, U.; Ahmad, B.; Ahmad, M.; Erum, A.; Hussain, K.; Irfan Bukhari, N. Is folklore use of Euphorbia helioscopia devoid of toxic effects? Drug Chem. Toxicol. 2016, 39, 233–237. [Google Scholar] [CrossRef]
- Moustafa, G.; Mohammed, A.; Abo Hadeed, A.; Kandil, F.; El Kelsh, E. Estimation of toxic hazards caused by methanolic extract of Euphorbia peplus in male albino rats. Adv. Anim. Vet. Sci 2016, 4, 5–11. [Google Scholar] [CrossRef]
- Akinmoladun, A.C.; Olaleye, M.T.; Farombi, E.O. 13—Cardiotoxicity and Cardioprotective Effects of African Medicinal Plants. In Toxicological Survey of African Medicinal Plants; Kuete, V., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 395–421. [Google Scholar] [CrossRef]
- Mohamed, W.A.M.; Abd-Elhakim, Y.M.; Farouk, S.M. Protective effects of ethanolic extract of rosemary against lead-induced hepato-renal damage in rabbits. Exp. Toxicol. Pathol. 2016, 68, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Boubaker, K.; Ounissi, M.; Brahmi, N.; Goucha, R.; Hedri, H.; Abdellah, T.; El Younsi, F.; Maiz, H.; Kheder, A. Acute renal failure by ingestion of Euphorbia paralias. Saudi J. Kidney Dis. Transplant. 2013, 24, 571–575. [Google Scholar] [CrossRef]
- Liu, Z.; Zeng, Y.; Hou, P. Metabolomic evaluation of Euphorbia pekinensis induced nephrotoxicity in rats. Pharm. Biol. 2018, 56, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Kwan, Y.P.; Saito, T.; Ibrahim, D.; Al-Hassan, F.M.S.; Ein Oon, C.; Chen, Y.; Jothy, S.L.; Kanwar, J.R.; Sasidharan, S. Evaluation of the cytotoxicity, cell-cycle arrest, and apoptotic induction by Euphorbia hirta in MCF-7 breast cancer cells. Pharm. Biol. 2016, 54, 1223–1236. [Google Scholar]
- Fu, Z.; Han, X.; Du, J.; Han, X.; Liu, W.; Shao, S.; Liu, X. Euphorbia lunulata extract acts on multidrug resistant gastric cancer cells to inhibit cell proliferation, migration and invasion, arrest cell cycle progression, and induce apoptosis. J. Ethnopharmacol. 2018, 212, 8–17. [Google Scholar] [CrossRef]
- Jian, B.; Zhang, H.; Han, C.; Liu, J. Anti-Cancer Activities of Diterpenoids Derived from Euphorbia fischeriana Steud. Molecules 2018, 23, 387. [Google Scholar] [CrossRef]
- Lane, D.P. Cancer. p53, guardian of the genome. Nature 1992, 358, 15–16. [Google Scholar] [CrossRef]
- Klein, C.; Vassilev, L. Targeting the p53–MDM2 interaction to treat cancer. Br. J. Cancer 2004, 91, 1415. [Google Scholar] [CrossRef]
- Muller, P.A.; Vousden, K.H. p53 mutations in cancer. Nat. Cell Biol. 2013, 15, 2. [Google Scholar] [CrossRef]
- Bieging, K.T.; Mello, S.S.; Attardi, L.D. Unravelling mechanisms of p53-mediated tumour suppression. Nat. Rev. Cancer 2014, 14, 359. [Google Scholar] [CrossRef]
- Vousden, K.H.; Lane, D.P. p53 in health and disease. Nat. Rev. Mol. Cell Biol. 2007, 8, 275. [Google Scholar] [CrossRef] [PubMed]
- Wade, M.; Wang, Y.V.; Wahl, G.M. The p53 orchestra: Mdm2 and Mdmx set the tone. Trends Cell Biol. 2010, 20, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Lahav, G.; Rosenfeld, N.; Sigal, A.; Geva-Zatorsky, N.; Levine, A.J.; Elowitz, M.B.; Alon, U. Dynamics of the p53-Mdm2 feedback loop in individual cells. Nat. Genet. 2004, 36, 147. [Google Scholar] [CrossRef] [PubMed]
- Wang, X. p53 regulation: Teamwork between RING domains of Mdm2 and MdmX. Cell Cycle 2011, 10, 4225–4229. [Google Scholar] [CrossRef] [PubMed]
- Shangary, S.; Wang, S. Small-molecule inhibitors of the MDM2-p53 protein-protein interaction to reactivate p53 function: A novel approach for cancer therapy. Annu. Rev. Pharmacol. Toxicol. 2009, 49, 223–241. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Swope, D.M.; Dashtipour, K. Comprehensive review of rasagiline, a second-generation monoamine oxidase inhibitor, for the treatment of Parkinson’s disease. Clin. Ther. 2007, 29, 1825–1849. [Google Scholar] [CrossRef]
- Vilar, S.; Cozza, G.; Moro, S. Medicinal chemistry and the molecular operating environment (MOE): Application of QSAR and molecular docking to drug discovery. Curr. Top. Med. Chem. 2008, 8, 1555–1572. [Google Scholar] [CrossRef]
- Guedes, R.; Serra, P.; Salvador, J.; Guedes, R. Computational approaches for the discovery of human proteasome inhibitors: An overview. Molecules 2016, 21, 927. [Google Scholar] [CrossRef]
- Atatreh, N.; Ghattas, M.A.; Bardaweel, S.K.; Al Rawashdeh, S.; Al Sorkhy, M. Identification of new inhibitors of Mdm2–p53 interaction via pharmacophore and structure-based virtual screening. Drug Des. Dev. Ther. 2018, 12, 3741. [Google Scholar] [CrossRef]
- Amr, A.E.G.E.; Elsayed, E.A.; Al-Omar, M.A.; Badr Eldin, H.O.; Nossier, E.S.; Abdallah, M.M. Design, synthesis, anticancer evaluation and molecular modeling of novel estrogen derivatives. Molecules 2019, 24, 416. [Google Scholar] [CrossRef]
- Nguyen, D.; Nguyen, D.; La, L.H.; Lee, H.B.; Kim, D.; Shin, J.; Kim, E. Isolation of dioctyl phthalate with high depigmenting effect from Chinese herb Nigella glandulifera Freyn. J. Biotechnol. 2007, 2, S43. [Google Scholar] [CrossRef]
- Habib, M.R.; Karim, M.R. Antimicrobial and cytotoxic activity of di-(2-ethylhexyl) phthalate and anhydrosophoradiol-3-acetate isolated from Calotropis gigantea (Linn.) flower. Mycobiology 2009, 37, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Ramalakshmi, S.; Muthuchelian, K. Analysis of bioactive constituents from the leaves of Mallotus tetracoccus (roxb.) Kurz, by gas chromatography-mass spectrometry. Int. J. Pharm. Sci. Res. 2011, 2, 1449. [Google Scholar]
- Zellagui, A.; Gherraf, N.; Ladjel, S.; Hameurlaine, S. Chemical composition and antibacterial activity of the essential oils from Launaea resedifolia L. Org. Med. Chem. Lett. 2012, 2, 2. [Google Scholar] [CrossRef] [PubMed]
- Jia, P.P.; Ma, Y.B.; Lu, C.J.; Mirza, Z.; Zhang, W.; Jia, Y.F.; Li, W.G.; Pei, D.S. The effects of disturbance on Hypothalamus-Pituitary-Thyroid (HPT) axis in zebrafish larvae after exposure to DEHP. PLoS ONE 2016, 11, e0155762. [Google Scholar] [CrossRef]
- David, R.M. Proposed mode of action for in utero effects of some phthalate esters on the developing male reproductive tract. Toxicol. Pathol. 2006, 34, 209–219. [Google Scholar] [CrossRef]
- Lin, H.; Yuan, K.; Li, L.; Liu, S.; Li, S.; Hu, G.; Lian, Q.Q.; Ge, R.S. In utero exposure to diethylhexyl phthalate affects rat brain development: A behavioral and genomic approach. Int. J. Environ. Res. Public Health 2015, 12, 13696–13710. [Google Scholar] [CrossRef]
- Priya, V.M.; Mayilvanan, C.; Akilavalli, N.; Rajesh, P.; Balasubramanian, K. Lactational exposure of phthalate impairs insulin signaling in the cardiac muscle of F1 female albino rats. Cardiovasc. Toxicol. 2014, 14, 10–20. [Google Scholar] [CrossRef]
- Rusyn, I.; Peters, J.M.; Cunningham, M.L. Modes of action and species-specific effects of di-(2-ethylhexyl) phthalate in the liver. Crit. Rev. Toxicol. 2006, 36, 459–479. [Google Scholar] [CrossRef]
- David, R.M.; Moore, M.R.; Finney, D.C.; Guest, D. Chronic toxicity of di (2-ethylhexyl) phthalate in mice. Toxicol. Sci. 2000, 58, 377–385. [Google Scholar] [CrossRef]
- National Industrial Chemicals Notification and Assessment Scheme (NICNAS); Diisooctyl Phthalate. Existing Chemical Hazard Assessment Report; Australian Government: Canberra, Australian, 2008.
- Saleem, M.; Nazir, M.; Akhtar, N.; Onocha, P.A.; Riaz, N.; Jabbar, A.; Shaiq Ali, M.; Sultana, N. New phthalates from Phyllanthus muellerianus (Euphorbiaceae). J. Asian Nat. Prod. Res. 2009, 11, 974–977. [Google Scholar] [CrossRef] [PubMed]
- Satyan, K.; Prakash, A.; Singh, R.; Srivastava, R. Phthalic acid bis-ester and other phytoconstituents of Phyllanthus urinaria. Planta Med. 1995, 61, 293–294. [Google Scholar] [CrossRef] [PubMed]
- Ertas, A.; Yilmaz, M.A.; Firat, M. Chemical profile by LC–MS/MS, GC/MS and antioxidant activities of the essential oils and crude extracts of two Euphorbia species. Nat. Prod. Res. 2015, 29, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Basile, A.; Giordano, S.; López-Sáez, J.A.; Cobianchi, R.C. Antibacterial activity of pure flavonoids isolated from mosses. Phytochemistry 1999, 52, 1479–1482. [Google Scholar] [CrossRef]
- Fernandes, E.S.; Passos, G.F.; Medeiros, R.; da Cunha, F.M.; Ferreira, J.; Campos, M.M.; Pianowski, L.F.; Calixto, J.B. Anti-inflammatory effects of compounds alpha-humulene and (−)-trans-caryophyllene isolated from the essential oil of Cordia verbenacea. Eur. J. Pharmacol. 2007, 569, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Wright, P.J.; Plummer, D.T. The use of urinary enzyme measurements to detect renal damage caused by nephrotoxic compounds. Biochem. Pharmacol. 1974, 23, 65–73. [Google Scholar] [CrossRef]
- Nebigil, C.G.; Desaubry, L. Updates in Anthracycline-Mediated Cardiotoxicity. Front. Pharmacol. 2018, 9, 1262. [Google Scholar] [CrossRef]
- Kaneko, J.J.; Harvey, J.W.; Bruss, M.L. Clinical Biochemistry of Domestic Animals; Academic Press: Cambridge, MA, USA, 2008. [Google Scholar]
- Adedapo, A.A.; Abatan, M.O.; Olorunsogo, O.O. Toxic effects of some plants in the genus Euphorbia on haematological and biochemical parameters of rats. Vet. Arh. 2004, 74, 53–62. [Google Scholar]
- Hillman, L.S.; Goodwin, S.L.; Sherman, W.R. Identification and measurement of plasticizer in neonatal.
- Posnack, N.G. The adverse cardiac effects of di (2-ethylhexyl) phthalate and bisphenol A. Cardiovasc. Toxicol. 2014, 14, 339–357. [Google Scholar] [CrossRef]
- Cappetta, D.; De Angelis, A.; Sapio, L.; Prezioso, L.; Illiano, M.; Quaini, F.; Rossi, F.; Berrino, L.; Naviglio, S.; Urbanek, K. Oxidative Stress and Cellular Response to Doxorubicin: A Common Factor in the Complex Milieu of Anthracycline Cardiotoxicity. Oxidative Med. Cell. Longev. 2017, 2017, 1521020. [Google Scholar] [CrossRef]
- Ghibu, S.; Delemasure, S.; Richard, C.; Guilland, J.C.; Martin, L.; Gambert, S.; Rochette, L.; Vergely, C. General oxidative stress during doxorubicin-induced cardiotoxicity in rats: Absence of cardioprotection and low antioxidant efficiency of alpha-lipoic acid. Biochimie 2012, 94, 932–939. [Google Scholar] [CrossRef] [PubMed]
- Pecoraro, M.; Del Pizzo, M.; Marzocco, S.; Sorrentino, R.; Ciccarelli, M.; Iaccarino, G.; Pinto, A.; Popolo, A. Inflammatory mediators in a short-time mouse model of doxorubicin-induced cardiotoxicity. Toxicol. Appl. Pharmacol. 2016, 293, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Gourlay, T.; Samartzis, I.; Stefanou, D.; Taylor, K. Inflammatory response of rat and human neutrophils exposed to di-(2-ethyl-hexyl)-phthalate-plasticized polyvinyl chloride. Artif. Organs 2003, 27, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Ward, J.M.; Peters, J.M.; Perella, C.M.; Gonzalez, F.J. Receptor and nonreceptor-mediated organ-specific toxicity of di (2-ethylhexyl) phthalate (DEHP) in peroxisome proliferator-activated receptorα-null mice. Toxicol. Pathol. 1998, 26, 240–246. [Google Scholar] [CrossRef]
- Havasi, A.; Borkan, S.C. Apoptosis and acute kidney injury. Kidney Int. 2011, 80, 29–40. [Google Scholar] [CrossRef]
- Kim, N.H.; Kang, P.M. Apoptosis in cardiovascular diseases: Mechanism and clinical implications. Korean Circ. J. 2010, 40, 299–305. [Google Scholar] [CrossRef]
- Tang, C.; Ma, Z.; Zhu, J.; Liu, Z.; Liu, Y.; Liu, Y.; Cai, J.; Dong, Z. P53 in kidney injury and repair: Mechanism and therapeutic potentials. Pharmacol. Ther. 2019, 195, 5–12. [Google Scholar] [CrossRef]
- Mak, T.W.; Hauck, L.; Grothe, D.; Billia, F. p53 regulates the cardiac transcriptome. Proc. Natl. Acad. Sci. USA 2017, 114, 2331–2336. [Google Scholar] [CrossRef]
- Wu, X.; Bayle, J.H.; Olson, D.; Levine, A.J. The p53-mdm-2 autoregulatory feedback loop. Genes Dev. 1993, 7, 1126–1132. [Google Scholar] [CrossRef]
- Toshiyuki, M.; Reed, J.C. Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell 1995, 80, 293–299. [Google Scholar] [CrossRef]
- El-Deiry, W.S.; Tokino, T.; Velculescu, V.E.; Levy, D.B.; Parsons, R.; Trent, J.M.; Lin, D.; Mercer, W.E.; Kinzler, K.W.; Vogelstein, B. WAF1, a potential mediator of p53 tumor suppression. Cell 1993, 75, 817–825. [Google Scholar] [CrossRef]
- Kastan, M.; Zhan, Q.; El-Deiry, W.S.; Carrier, F.; Jacks, T.; Walsh, W.V.; Plunkett, B.S.; Vogelstein, B.; Fornace, A.J., Jr. . A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxia-telangiectasia. Cell 1992, 71, 587–597. [Google Scholar] [CrossRef]
- Shivakumar, C.V.; Brown, D.R.; Deb, S.; Deb, S.P. Wild-type human p53 transactivates the human proliferating cell nuclear antigen promoter. Mol. Cell. Biol. 1995, 15, 6785–6793. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Bian, J.; Wang, Y.; Jacobs, C. Activation of p53 transcriptional activity by 1,10-phenanthroline, a metal chelator and redox sensitive compound. Oncogene 1997, 14, 385–393. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fu, Z.Y.; Han, X.D.; Wang, A.H.; Liu, X.B. Apoptosis of human gastric carcinoma cells induced by Euphorbia esula latex. World J. Gastroenterol. 2016, 22, 3564–3572. [Google Scholar] [CrossRef] [PubMed]
- Al-Khodairy, F.M.; Khan, M.K.A.; Kunhi, M.; Pulicat, M.S.; Akhtar, S.; Arif, J.M. In Silico prediction of mechanism of Erysolin-induced apoptosis in human breast cancer cell lines. Am. J. Bioinform. Res. 2013, 3, 62–71. [Google Scholar]
- Momand, J.; Zambetti, G.P.; Olson, D.C.; George, D.; Levine, A.J. The mdm-2 oncogene product forms a complex with the p53 protein and inhibits p53-mediated transactivation. Cell 1992, 69, 1237–1245. [Google Scholar] [CrossRef]
- Honda, R.; Yasuda, H. Activity of MDM2, a ubiquitin ligase, toward p53 or itself is dependent on the RING finger domain of the ligase. Oncogene 2000, 19, 1473. [Google Scholar] [CrossRef]
- Fang, S.; Jensen, J.P.; Ludwig, R.L.; Vousden, K.H.; Weissman, A.M. Mdm2 is a RING finger-dependent ubiquitin protein ligase for itself and p53. J. Biol. Chem. 2000, 275, 8945–8951. [Google Scholar] [CrossRef]
- Roth, J.; Dobbelstein, M.; Freedman, D.A.; Shenk, T.; Levine, A.J. Nucleo-cytoplasmic shuttling of the hdm2 oncoprotein regulates the levels of the p53 protein via a pathway used by the human immunodeficiency virus rev protein. EMBO J. 1998, 17, 554–564. [Google Scholar] [CrossRef]
- Vassilev, L.T.; Vu, B.T.; Graves, B.; Carvajal, D.; Podlaski, F.; Filipovic, Z.; Kong, N.; Kammlott, U.; Lukacs, C.; Klein, C.; et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 2004, 303, 844–848. [Google Scholar] [CrossRef] [PubMed]
- Kussie, P.H.; Gorina, S.; Marechal, V.; Elenbaas, B.; Moreau, J.; Levine, A.J.; Pavletich, N.P. Structure of the MDM2 oncoprotein bound to the p53 tumor suppressor transactivation domain. Science 1996, 274, 948–953. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Carlson, H.A. Computational studies and peptidomimetic design for the human p53-MDM2 complex. Proteins 2005, 58, 222–234. [Google Scholar] [CrossRef] [PubMed]
- Dastidar, S.G.; Lane, D.P.; Verma, C.S. Multiple Peptide Conformations Give Rise to Similar Binding Affinities: Molecular Simulations of p53-MDM2. J. Am. Chem. Soc. 2008, 130, 13514–13515. [Google Scholar] [CrossRef] [PubMed]
- Vaupel, A.; Holzer, P.; Ferretti, S.; Guagnano, V.; Kallen, J.; Mah, R.; Masuya, K.; Ruetz, S.; Rynn, C.; Schlapbach, A.; et al. In vitro and in vivo characterization of a novel, highly potent p53-MDM2 inhibitor. Bioorganic Med. Chem. Lett. 2018, 28, 3404–3408. [Google Scholar] [CrossRef] [PubMed]
- Holzer, P.; Masuya, K.; Furet, P.; Kallen, J.; Valat-Stachyra, T.; Ferretti, S.; Berghausen, J.; Bouisset-Leonard, M.; Buschmann, N.; Pissot-Soldermann, C.; et al. Discovery of a Dihydroisoquinolinone Derivative (NVP-CGM097): A Highly Potent and Selective MDM2 Inhibitor Undergoing Phase 1 Clinical Trials in p53wt Tumors. J. Med. Chem. 2015, 58, 6348–6358. [Google Scholar] [CrossRef]
- Furet, P.; Masuya, K.; Kallen, J.; Stachyra-Valat, T.; Ruetz, S.; Guagnano, V.; Holzer, P.; Mah, R.; Stutz, S.; Vaupel, A.; et al. Discovery of a novel class of highly potent inhibitors of the p53-MDM2 interaction by structure-based design starting from a conformational argument. Bioorganic Med. Chem. Lett. 2016, 26, 4837–4841. [Google Scholar] [CrossRef]
- Furet, P.; Guagnano, V.; Holzer, P.; Kallen, J.; Lv, L.I.; Mah, R.; Mao, L.; Masuya, K.; Schlapbach, A.; Stutz, S.; et al. Imidazopyrrolidinone Compounds. U.S. Patent Application 14/327,639, 20 November 2014. [Google Scholar]
- Lemos, A.; Leao, M.; Soares, J.; Palmeira, A.; Pinto, M.; Saraiva, L.; Sousa, M.E. Medicinal Chemistry Strategies to Disrupt the p53-MDM2/MDMX Interaction. Med. Res. Rev. 2016, 36, 789–844. [Google Scholar] [CrossRef]
- Handa, S.S.; Khanuja, S.P.S.; Longo, G.; Rakesh, D.D. Extraction technologies for medicinal and aromatic plants: Earth. In Environmental and Marine Sciences and Technologies; International Centre For Science And High Technology, Trieste: Italy, 2008. [Google Scholar]
- Murray, R. Aspartate aminotransferase. In Clinical Chemistry. Theory, Analysis, and Correlation; Kaplan, L., Pesce, A., Eds.; CV Mosby Company: St Louis, Toronto, Princeton, 1984; pp. 1112–1116. [Google Scholar]
- Wenger, C.; Kaplan, A.; Rubaltelli, F.; Hammerman, C. Alkaline phosphatase. In Clinical Chemistry. Theory, Analysis, and Correlation; Kaplan, L., Pesce, A., Eds.; CV Mosby Company: St Louis, Toronto, Princeton, 1984; pp. 1094–1098. [Google Scholar]
- Pesce, A. Lactate dehydrogenase. In Clinical Chemistry. Theory, Analysis, and Correlation; Kaplan, L., Pesce, A., Eds.; CV Mosby Company: St Louis, Toronto, Princeton, 1984; pp. 1124–1127. [Google Scholar]
- Kaplan, A. Urea. In Clinical Chemistry. Theory, Analysis, and Correlation; Kaplan, L., Pesce, A., Eds.; CV Mosby Company: St Louis, Toronto, Princeton, 1984; pp. 1257–1260. [Google Scholar]
- Fossati, P.; Prencipe, L.; Berti, G. Enzymic creatinine assay: A new colorimetric method based on hydrogen peroxide measurement. Clin. Chem. 1983, 29, 1494–1496. [Google Scholar]
- Barham, D.; Trinder, P. Enzymatic determination of uric acid. Analyst 1972, 97, 142–145. [Google Scholar] [CrossRef]
- Suvarna, S.; Layton, C.; Bancroft, J. The hematoxylins and eosin. In Bancroft’s Theory and Practice of Histological Techniques, 7th ed.; Churchill Livingstone: London, UK, 2013; pp. 172–186. [Google Scholar]
- Mohamed, A.A.R.; Mohamed, W.A.; Khater, S.I. Imidacloprid induces various toxicological effects related to the expression of 3β-HSD, NR5A1, and OGG1 genes in mature and immature rats. Environ. Pollut. 2017, 221, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elhakim, Y.M.; Hashem, M.M.; Anwar, A.; El-Metwally, A.E.; Abo-El-Sooud, K.; Moustafa, G.G.; Mouneir, S.M.; Ali, H.A. Effects of the food additives sodium acid pyrophosphate, sodium acetate, and citric acid on hemato-immunological pathological biomarkers in rats: Relation to PPAR-α, PPAR-γ and tnfα signaling pathway. Environ. Toxicol. Pharmacol. 2018, 62, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.M.; Rezk, N.A.; Fawzy, A.; Sabry, M. Protective effects of curcumin and silymarin against paracetamol induced hepatotoxicity in adult male albino rats. Gene 2019, 712, 143966. [Google Scholar] [CrossRef]
- Soliman, M.M.; Nassan, M.A.; Ismail, T.A. Immunohistochemical and molecular study on the protective effect of curcumin against hepatic toxicity induced by paracetamol in Wistar rats. BMC Complement. Altern. Med. 2014, 14, 457. [Google Scholar] [CrossRef] [PubMed]
- Temirak, A.; Shaker, Y.M.; Ragab, F.A.; Ali, M.M.; Ali, H.I.; El Diwani, H.I. Part I. Synthesis, biological evaluation and docking studies of new 2-furylbenzimidazoles as antiangiogenic agents. Eur. J. Med. Chem. 2014, 87, 868–880. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Lu, Y.; Wang, S. Comparative evaluation of 11 scoring functions for molecular docking. J. Med. Chem. 2003, 46, 2287–2303. [Google Scholar] [CrossRef]
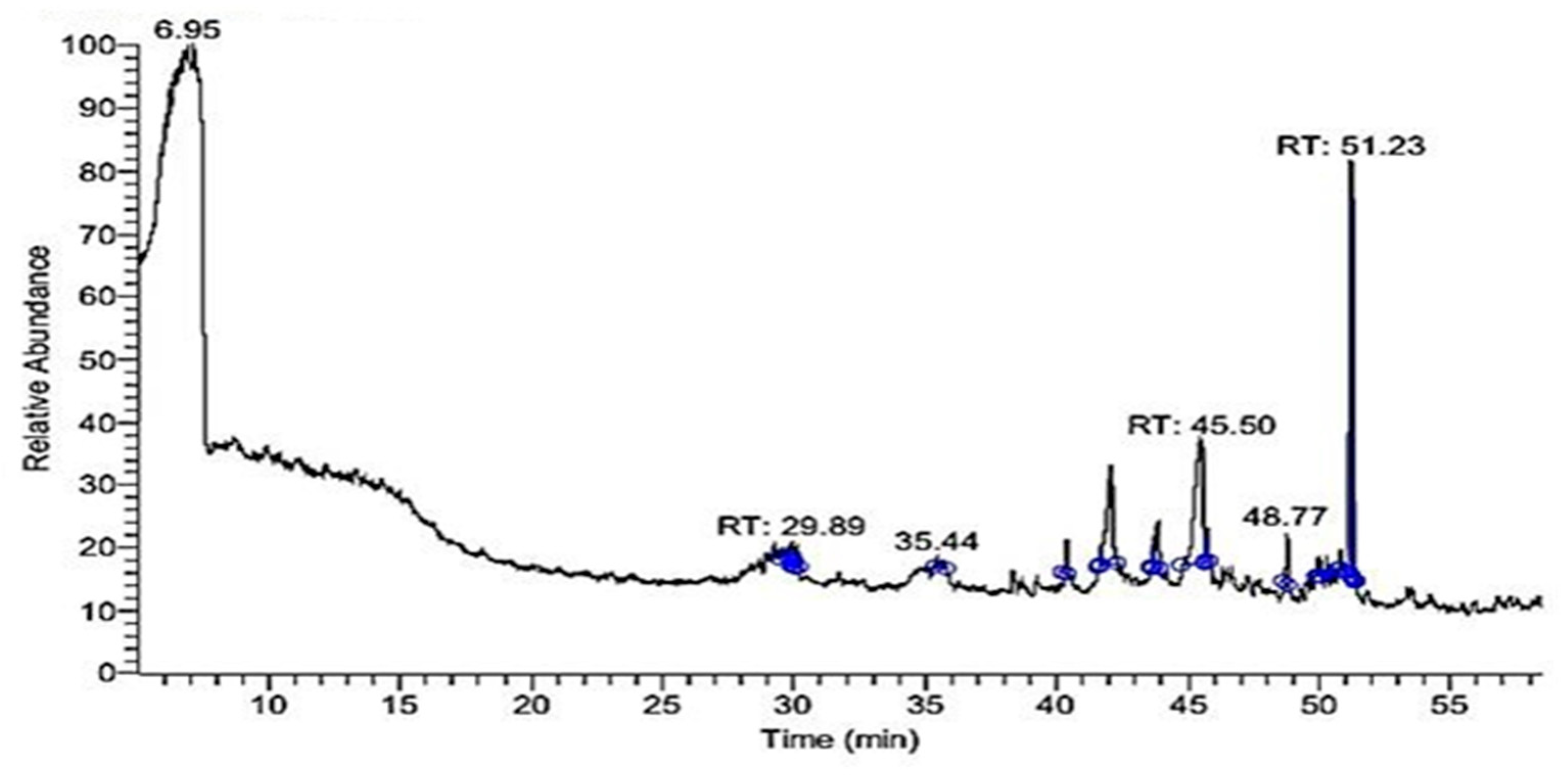
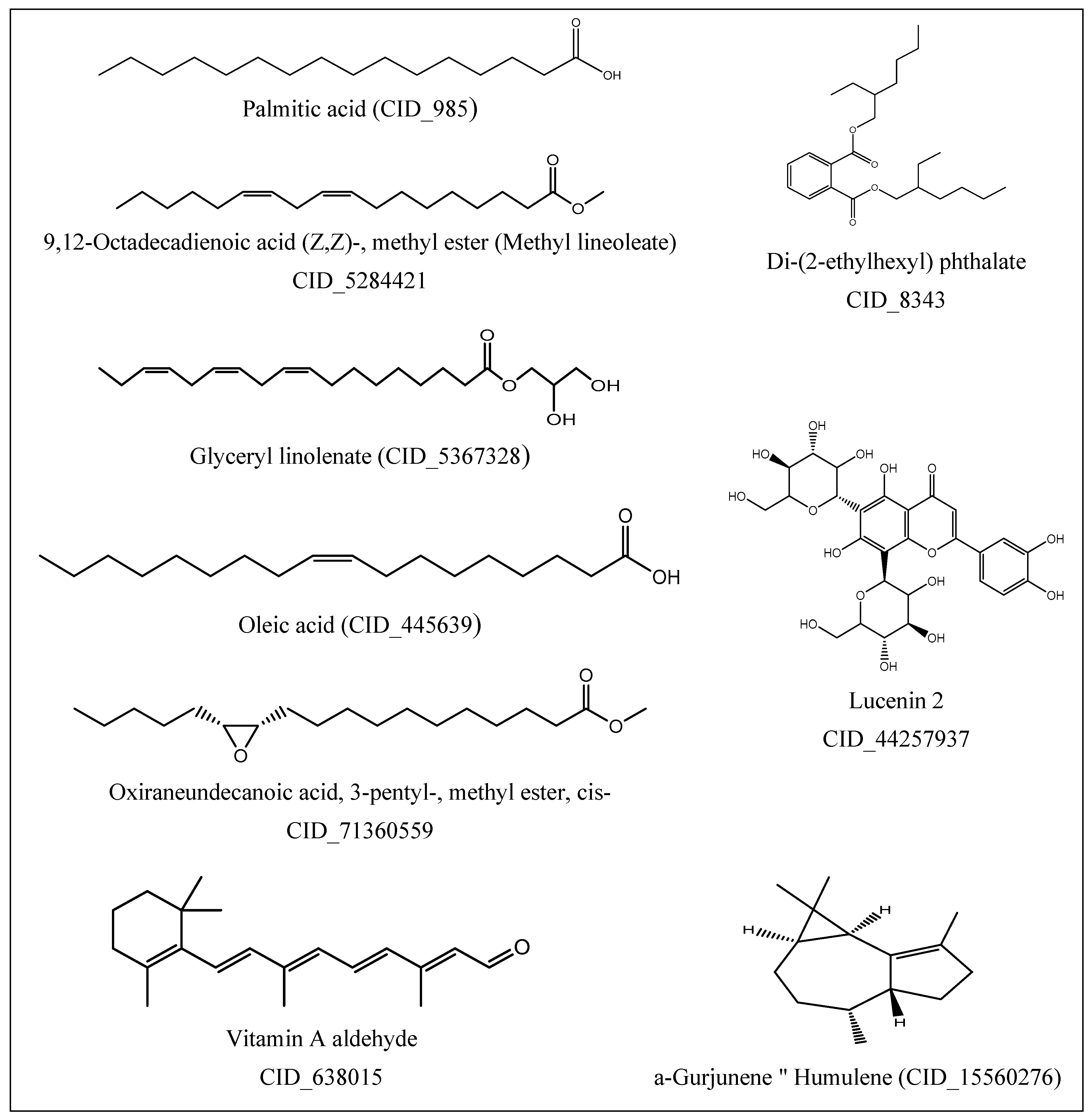
| Compound | MW 1 | Formula | RT (min) | Peak Area% |
|---|---|---|---|---|
| Di-(2-ethylhexyl) phthalate | 390 | C24H38O4 | 51.23 | 37.07 |
| 9-Octadecenoic acid “oleic acid” | 282 | C18H34O2 | 45.50 | 28.82 |
| Hexadecanoic acid “palmitic acid” | 256 | C16H32O2 | 42.07 | 15.74 |
| Lucenin 2 | 610 | C27H30O16 | 48.77 | 4.54 |
| à-Gurjunene “humulene” | 204 | C15H24 | 29.89 | 4.36 |
| 9,12,15-Octadecatrienoic acid, 2,3-dihydroxypropyl ester “glyceryl linolenate” | 352 | C21H36O4 | 43.84 | 3.64 |
| Oxiraneundecanoic acid, 3-pentyl-, methyl ester, cis-(CAS) | 312 | C19H36O3 | 45.72 | 2.23 |
| 9,12-Octadecadienoic acid (Z,Z)-, methyl ester | 294 | C19H34O2 | 43.69 | 2.24 |
| Vitamin A aldehyde | 284 | C20H28O | 35.44 | 1.36 |
| Parameters | Control | DMSO Treated | EPME Treated | P-Value |
|---|---|---|---|---|
| Troponin I (ug/L) | 0.01 b ± 0.001 | 0.02 b ± 0.005 | 0.07 a ± 0.016 | <0.001 |
| CPK (U/L) | 314.0 b ± 2.5 | 350.0 b ± 53.9 | 652.0 a ± 9.7 | <0.001 |
| CK-MB (ng/mL) | 6.0 b ± 0.1 | 8.7 b ± 0.1 | 18.7 a ± 1.7 | <0.001 |
| LDH (U/L) | 1698.3 b ± 11.9 | 2224.0 b ± 279.1 | 3853.7a ± 144.7 | <0.001 |
| AST (U/L) | 92.5 b ± 1.0 | 89.6 b ± 2.1 | 109.9 a ± 3.9 | 0.001 |
| ALP (U/L) | 42.2 b ± 1.2 | 54.9 b ± 1.7 | 81.7 a ± 7.4 | <0.001 |
| N | Compound | ΔGb | RMSD | No. of Binding Interactions | H-Bonds | π-Interactions | |||
|---|---|---|---|---|---|---|---|---|---|
| Name | PubChem CID | Type | Amino Acid | Type | Amino Acid | ||||
| 1 | Ligand | - | −8.172 | 0.49 | 3 | H-acceptor | HIS 96 | pi-pi | HIS 96 |
| H-donor | MET 62 | ||||||||
| 2 | Palmitic acid | 985 | −7.916 | - | 1 | H-donor | HIS 73 | - | - |
| 3 | Di-(2-ethylhexyl) phthalate | 8343 | −12.448 | - | 3 | H-donor | MET 62 | H-pi | HIS 96 |
| H-pi | HIS 96 | ||||||||
| 4 | Oleic Acid | 445639 | −12.244 | - | 1 | H-acceptor | HIS 96 | - | - |
| 5 | Vitamin A aldehyde | 638015 | −9.412 | - | 1 | - | - | H-pi | TYR 67 |
| 6 | 9,12-Octadecadienoic acid (Z,Z)-, methyl ester (Methyl lineoleate) | 5284421 | −10.9625 | - | 1 | H-acceptor | HIS 96 | - | - |
| 7 | Glyceryl linolenate | 5367328 | −14.496 | - | 2 | H-donor | ILE 19 | - | - |
| H-acceptor | HIS 96 | - | - | ||||||
| 8 | α-Gurjunene “Humulene” | 15560276 | −6.371 | - | 1 | - | - | H-pi | HIS 96 |
| 9 | Lucenin 2 | 44257937 | −20.733 | - | 3 | H-donor | VAL 93 | - | - |
| H-acceptor | GLN 24 | - | - | ||||||
| H-acceptor | HIS 96 | - | - | ||||||
| 10 | Oxiraneundecanoic acid, 3-pentyl-, methyl ester, cis-(CAS) | 71360559 | −12.626 | - | 1 | H-acceptor | GLN 24 | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd-Elhakim, Y.M.; Abdo Nassan, M.; Salem, G.A.; Sasi, A.; Aldhahrani, A.; Ben Issa, K.; Abdel-Rahman Mohamed, A. Investigation of the In-Vivo Cytotoxicity and the In Silico-Prediction of MDM2-p53 Inhibitor Potential of Euphorbia peplus Methanolic Extract in Rats. Toxins 2019, 11, 642. https://doi.org/10.3390/toxins11110642
Abd-Elhakim YM, Abdo Nassan M, Salem GA, Sasi A, Aldhahrani A, Ben Issa K, Abdel-Rahman Mohamed A. Investigation of the In-Vivo Cytotoxicity and the In Silico-Prediction of MDM2-p53 Inhibitor Potential of Euphorbia peplus Methanolic Extract in Rats. Toxins. 2019; 11(11):642. https://doi.org/10.3390/toxins11110642
Chicago/Turabian StyleAbd-Elhakim, Yasmina M., Mohamed Abdo Nassan, Gamal A. Salem, Abdelkarim Sasi, Adil Aldhahrani, Khaled Ben Issa, and Amany Abdel-Rahman Mohamed. 2019. "Investigation of the In-Vivo Cytotoxicity and the In Silico-Prediction of MDM2-p53 Inhibitor Potential of Euphorbia peplus Methanolic Extract in Rats" Toxins 11, no. 11: 642. https://doi.org/10.3390/toxins11110642
APA StyleAbd-Elhakim, Y. M., Abdo Nassan, M., Salem, G. A., Sasi, A., Aldhahrani, A., Ben Issa, K., & Abdel-Rahman Mohamed, A. (2019). Investigation of the In-Vivo Cytotoxicity and the In Silico-Prediction of MDM2-p53 Inhibitor Potential of Euphorbia peplus Methanolic Extract in Rats. Toxins, 11(11), 642. https://doi.org/10.3390/toxins11110642